Abstract
Purpose
Previous studies in cancer survivors have examined behavioral correlates of physical activity (PA), but no study to date has adopted a broader social-ecological framework in understanding PA. This study examined the associations among demographic, medical, social-cognitive, and environmental correlates of meeting PA guidelines among kidney cancer survivors (KCS).
Methods
All 1985 KCS diagnosed between 1996 and 2010 identified through a Canadian provincial registry were mailed a survey that consisted of medical, demographic, and social-cognitive measures, as well as PA as measured by the Godin Leisure Time Exercise Questionnaire. Environmental constructs were also assessed for both self-report and objective measures using geographic information systems (GIS). A series of binary logistic regression analyses were conducted in this cross-sectional study.
Results
Completed surveys with geographical information were received from 432 KCS with M age = 64.4 ± 11.1 years, 63.2 % male, and 82.2 % having localized kidney cancer. In the final multivariate model, meeting PA guidelines was associated with disease stage (OR = 0.25, p = .005), having drug therapy (OR = 3.98, p = .009), higher levels of instrumental attitudes (OR = 1.66, p = .053), higher levels of intention (OR = 1.72, p = .002), and the perceived presence of many retail shops in the neighborhood (OR = 1.37, p = .032).
Conclusions
Meeting PA guidelines in KCS were associated with various aspects of the social-ecological model.
Implications for Cancer Survivors
Understanding the social-ecological correlates for PA can provide insight into future interventions designed to increase PA in KCS. Prime targets for PA promotion should consider treatment-related factors, promote the benefits of PA, and enhance positive perceptions of the built environment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Physical activity (PA) has beneficial effects on overall quality of life (QoL), cancer-specific concerns, fatigue, and pain across many cancer survivor groups [1] including kidney cancer survivors (KCS) [2]. Despite the established benefits of PA, the majority of cancer survivors are not meeting public health PA guidelines [3, 4], including KCS [2]. Previous research examining the correlates of PA in cancer survivors has been based on individual and interpersonal factors such as psychosocial, medical, and demographic variables [5–12]. While these factors do have merit in explaining PA, larger contextual factors such as environmental factors have been recognized as important determinants of PA and require further study in this population [13, 14].
Social-ecological models of health behavior emphasize the interplay between individuals and their social and physical environments [15, 16]. Specifically, a social-ecological framework proposes multiple levels of influence, including individual (e.g., attitudes, self-efficacy, intentions), interpersonal (e.g., social support from family and friends), and physical environmental (e.g., perceived neighborhood aesthetics, personal safety, built environment) factors for understanding and changing health behaviors [15]. Central to this ecological perspective is the assumption of the interaction and reciprocal causation among the various levels [17]. Knowledge about the comprehensive set of factors influencing PA can inform the development of multilevel interventions to offer the best level of success [13]. Intervention studies have focused largely on these individual characteristics with little emphasis on the perceived and objective built environment [18]. Environmental attributes such as access to services, street connectivity, safety, and land use mix as determinants of PA have been examined in apparently healthy adults [14, 18], but no study has specifically examined the role of the built environment for PA in cancer survivors.
A recent systematic review examined the determinants and exercise maintenance in adult cancer survivors from a socio-ecological perspective [19]. Of the 18 studies included in the review, 12 focused on the determinants of exercise and six studies centered on determinants of exercise maintenance after intervention completion. The majority of the studies examined demographic and clinical factors (e.g., age, stage of disease, date of diagnosis), psychological factors (e.g., motivational variables, QoL), physical factors (e.g., muscular strength, body composition), whereas few studies examined social factors (e.g., family support) and environmental factors (e.g., location of fitness center). There was evidence for a positive relationship between physical determinants such as exercise history and exercise adherence, with insufficient evidence of an association for most demographic and clinical factors with exercise adherence and maintenance. Inconsistent findings were found for exercise adherence in terms of gender; type of treatment; psychological factors including perceived behavioral control (PBC), stage of change, self-efficacy, extraversion; physical factors such as cardiovascular fitness; and environmental factors such as fitness center location. In addition, inconsistent findings were noted for exercise maintenance such as age, education, self-efficacy, fatigue, attitude, QoL, intention, PA intervention adherence, body mass index, baseline PA, and cardiovascular fitness [19].
In apparently healthy adults, a review of 18 reviews identified a small number of correlates including reported health, intention to exercise, male sex, self-efficacy, past exercise behavior, recreation facilities and locations, transportation environments, and aesthetics [13]. Among older adults, previous studies have reported a positive relationship between presence of nearby destinations and older adults’ walking for transportation [20, 21]. However, a recent systematic review found limited associations between the physical environment (e.g., quality of sidewalks, access to parks) and PA in older adults (≥65 years) [22]. Given that KCS are an older population, similar relationships may exist with select correlates of PA that are found in older adults, with medical correlates playing a more central role in KCS.
There has been only one study that used an ecological model of health behavior in colorectal cancer survivors, but this study focused on examining potential barriers to PA into subcategories including physical environment, social environment, personal attributes, and disease-specific barriers [23]. Disease-specific barriers (e.g., fatigue) were perceived as the greatest barrier to PA followed by personal attributes (e.g., already active enough), whereas the physical environment (e.g., lack of suitable facilities) was the least salient barrier.
To the best of our knowledge, no study to date has examined the correlates of PA behavior in a single cancer type (i.e., KCS) from a population-based and social-ecological approach. In a previous study of KCS by our research group using the full dataset (n = 703), we examined the correlates of PA using the theory of planned behavior (TPB) [24]. While this previous study did find the utility of the TPB in explaining PA, it only focused on individual-level variables, rather than larger contextual factors such as the environment. Furthermore, previous studies have relied on self-report of environmental attributes, and although these measures are related to PA behavior [13, 14, 23], objective measures of environmental attributes such as the use of geographic information systems (GIS) are warranted to provide an understanding of the opportunities and resources for PA situated within the neighborhood. The purpose of this study is to examine the associations between demographic, medical, behavioral, and environmental correlates of PA in KCS using several aspects of the social-ecological model including individual, social-cognitive, and the perceived and built environment variables. We hypothesized that individual correlates (i.e., demographic, medical, social-cognitive variables) may be more proximal predictors of meeting PA guidelines in KCS.
Methods
Geographical context
The province of Alberta spans 661,185 km2 with a population of 3,645,257 [25]. Of the total population of 3,645,257, 83 % (3,030,402) and 17 % (614,885) reside in urban and rural areas, respectively [25]. Approximately 90 % of Alberta is part of North America’s interior plain, with heavily forested areas and peat lands in the north, grasslands in the south, and parkland in the east and central areas. In the western boundary of the province, mountains and foothills comprise the remainder of the province [26]. The province of Alberta has two major cities: Edmonton and Calgary.
Study design and participants
The design and methods of the survey have been reported elsewhere [2]. The study employed a cross-sectional design with a mailed, self-administered survey. Eligibility for the study included (a) 18 years or older, (b) ability to understand English, (c) currently residing in Alberta, and (d) diagnosed with kidney cancer in Alberta between 1996 and 2010. All 1985 KCS from the Alberta Cancer Registry who met our eligibility criteria were mailed the survey between May and September 2010. Ethical approval was obtained from the Alberta Cancer Board Research Ethics Board and the University of Alberta Health Research Ethics Board.
Measures
Demographic information
Demographic variables were assessed using self-report and included age (coded as “0” for <65 years and coded as “1” for ≥65 years), sex (coded 0 for male and coded 1 for female), education level (coded 0 for some/completed high school and coded 1 for some/completed university), marital status (coded 0 for not married and coded 1 for married/common law), employment status (coded 0 for unemployed and coded 1 for employed full-/part-time), ethnicity (coded 0 for other and coded 1 for white), and body mass index (BMI) was coded as 0 for healthy weight and coded 1 for overweight/obese.
Medical information
Medical variables were also assessed using self-report and included time since diagnosis (coded 0 for <60 months and coded 1 as ≥60 months), disease stage (coded 0 for localized and coded 1 for metastatic), surgery treatment (coded 0 for no and coded 1 for yes), drug treatment (coded 0 for no and coded 1 for yes), and current disease status (coded 0 for disease free and coded 1 for existing disease).
Physical activity
PA was assessed using a modified version of the validated leisure score index (LSI) from the Godin Leisure-Time Exercise Questionnaire (GLTEQ) [27]. Participants were asked to recall the average number of times per week and average duration they performed light (minimal effort, no perspiration), moderate (not exhausting, light perspiration), and vigorous (heart beats rapidly, sweating) PA for a minimum of 10 min per session during free time in the past month. The percentage of participants meeting the public health aerobic PA guidelines was calculated based on the PA guidelines recommended for cancer survivors by the American Cancer Society [3] and the American College of Sports Medicine [4]. These guidelines recommend that individuals obtain 75 min of vigorous aerobic PA per week, 150 min of moderate aerobic PA per week, or an equivalent combination. Thus, we calculated “PA minutes” as moderate minutes plus two times the vigorous minutes. These PA minutes were then classified into two categories: (1) not meeting PA guidelines (<150 PA minutes; coded 0) and (2) meeting PA guidelines (≥150 PA minutes; coded 1).
Social-cognitive variables
Social-cognitive factors were assessed using a modified version of TPB. Prior to completing the TPB measures, we defined regular PA for participants as “moderate intensity PA (e.g., brisk walking) performed for at least 150 min per week (2.5 h), OR vigorous intensity PA performed at least 75 min per week (1.25 h).” The TPB items were developed based on guidelines recommended by Ajzen [28, 29], as well as previous studies with cancer survivors [30, 31].
Attitude was measured by four items using a 7-point bipolar adjective scale that examined both instrumental (beneficial/harmful, important/unimportant) and affective (enjoyable/unenjoyable, fun/boring) components of attitude. The verbal descriptors were extremely (points 1 and 7), quite (points 2 and 6), and slightly (points 3 and 5). The stem that preceded the adjectives was “I think that for me to participate in regular PA over the next month would be…”
Subjective norm was measured by three items rated on a 7-point Likert scale. The two items that measured injunctive norm were preceded by the stem “I think that if I participated in regular PA over the next month, most people who are important to me would be…” followed by the scales 1 = extremely discouraging to 7 = extremely encouraging and 1 = extremely unsupportive to 7 = extremely supportive. There was one item measuring descriptive norm, which was “I think that over the next month, most people who are important to me will themselves participate regularly in PA” (1 = strongly disagree to 7 = strongly agree).
PBC was determined by two items on a 7-point Likert scale. The specific items were (a) “If you were really motivated, how much control would you have over doing regular PA over the next month” (1 = very little control to 7 = complete control); (b) “If you were really motivated, how confident would you be that you could do regular PA over the next month?” (1 = not at all confident to 7 = extremely confident).
Planning was measured using the instrument developed and validated by Rise et al. [32]. The four items were (1) “I have made plans concerning ‘when’ I am going to engage in regular PA over the next month;” (2) “I have made plans concerning ‘where’ I am going to engage in regular PA over the next month;” (3) “I have made plans concerning ‘what’ kind of regular PA I am going to engage in over the next month;” and (4) “I have made plans concerning ‘how’ I am going to get to a place to engage in regular PA over the next month.” All items were rated using a 7-point Likert-type scale ranging from 1 (no plans) to 7 (detailed plans).
Perceived environment
Perceived environment characteristics were based on a combination of items from the Neighborhood Environment Walkability Scale (NEWS) [33] and the International Physical Activity Prevalence Study Environmental Survey Module (IPAPSEM) [34] that have been used to predict walking in previous research with healthy adults [35]. The decision on the items was based on measures from previous research that highlight proximity to retail or recreation, aesthetics, crime, traffic, and walking infrastructure quality as key components [36]. Proximity was measured using the following: (1) “Many shops, stores, markets or other places to buy things I need are within easy walking distance of my residence” and (2) “My neighborhood has several free or low cost recreation facilities, such as parks, walking trails, bike paths, and recreation centers.” Quality of walking infrastructure was assessed using the following: “There are well-maintained sidewalks on most of the streets in my neighborhood.” Neighborhood aesthetics was measured using the following: “There are many attractive natural sights in my neighborhood (such as landscaping, views…).” Traffic was assessed using the following: “It feels unsafe to walk along the streets in my neighborhood because there is so much traffic.” Finally, crime was measured with the following: “There is a high crime rate in my neighborhood.” All items were rated on a 4-point scale from strongly disagree (1) to strongly agree (4). Higher scores are indicative of favorable perceptions, whereas lower scores reflect unfavorable perceptions of the perceived environment.
Built environment
GIS-based measures were used to characterize the objective built environment. Survey data were geocoded to the geographic center of the home postal and provide a reasonable proxy for the home address in urban settings [37]. Respondents with missing postal code data (n = 162) or residing in rural postal codes (n = 109) were excluded from this study. Rural postal codes are subject to misclassification in identifying the location of residence, and they tend to cover large geographic areas [37]. Network buffers were compiled based on a distance of 1 km from the participants’ home postal code centroid and used to examine neighborhood characteristics. The network buffer calculates distance based on a walking network, which includes streets, multiuse pathways, and pedestrian walkways. All of the area within the network buffer or home neighborhood can be reached within a 1-km walk. The choice of this distance was informed by previous studies of PA and the built environment [38, 39].
Neighborhood socioeconomic status (SES) was assessed using dissemination area (DA) level data on median household income from census data obtained through Statistics Canada [40]. Dissemination areas are the smallest geographical unit where income data are released in Canada and have a mean size of 0.282 km2 within urban areas of the sample. Population counts were obtained from Statistics Canada [40]. Population density was compiled by dividing the total number of people at census block level by the total area of land in square kilometers within each buffer. Population density is a proxy for neighborhood type and may relate to distances to certain destinations (i.e., shops, recreation centers).
Data on the street network, parks, recreation centers, and shopping centers were derived from DMTI Route Logistics dataset (DMTI 2010). Park density was determined by dividing the total area of public parks within the home neighborhood by the total area of the buffer. Public parks include nature trails, bike paths, playgrounds, athletic fields, and municipal, provincial, and national parks. Access to shopping and recreational centers was determined by calculating the number of centers within the home neighborhood. Recreation centers included private and public fee facilities including physical fitness clubs, sports and recreation clubs, swimming pools, yoga/dance studios, bowling centers, golf courses, and sporting arenas.
Characteristics of the street network were also examined. Street network design varies based on era of development, as older neighborhoods typically have a more connected street network with shorter straighter blocks, a higher density of intersections, and more land dedicated to roadways. While in more suburban areas, the streets are typically curvilinear, the roadways are less connected which can increase distances to destinations. This study examined both the density of local roads and density of intersections. Local road density was calculated by dividing the total distance of local roads divided by the neighborhood area. Intersection density, a measure of street connectivity, was determined by calculating the number of three- or four-way intersections per square kilometer. Areas with a higher intersection density will have a more connected street network and more route options.
Data analyses
All statistical analyses were performed using SPSS Statistics 22 (IBM Corp., Armonk, NY). Chi-squared analysis was used to test for group differences between each demographic and/or medical variable with meeting PA guidelines. All demographic and medical variables were either dichotomized to ensure adequate numbers per cell based either on clinically relevant cutpoints or balanced statistical splits. Logistic regression analyses using a hierarchical approach were conducted given the tenets of an ecological model. In block 1, demographic variables were entered including age, sex, marital status, employment status, education level, and ethnicity. In block 2, medical variables were entered including number of comorbidities, months since diagnosis, disease stage, BMI, surgery treatment, drug therapy, and current disease status. Social-cognitive variables from the TPB were entered in three blocks to account for the TPB structure and causal ordering of the variables. Block 3 included planning, block 4 included intention, and block 5 included affective and instrumental attitudes, descriptive and injunctive norms, and PBC. In block 6, perceived environmental variables were entered including proximity to retail shops and recreation facilities, aesthetics, crime, traffic, and walking infrastructure quality. In block 7, objective measures of the built environment were entered including mean household income, population density, park density, number of shopping centers, number of recreation centers, local road density, and intersection density. The Omnibus chi-squared test (p < .05) and Hosmer and Lemeshow test (p > .05) were used to determine model fit. The Nagelkerke’s R 2 was used as an indication of the amount of variance explained.
Results
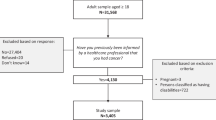
Of the 1985 mailed surveys, 331 were returned to sender due to wrong address, no history of kidney cancer, or deceased. Of the remaining 1654 surveys, 703 were returned, resulting in a 35.4 % completion rate (703/1985) and a 42.5 % response rate (703/1654) excluding the return to sender surveys. For the current study, we had 432 of 703 (61.5 %) KCS that provide valid postal codes for the analyses and living in an urban area. KCS with postal code data were compared to those who did not provide postal code data, and no demographic and medical differences were found.
Previously, responders (n = 703) and nonresponders (n = 1282) were compared and no differences in terms of age, sex, or surgery rate were found [2]. Responders were about 1 year closer to their date of diagnosis compared to nonresponders and had a slightly higher rate of treatment with systemic therapy. Moreover, responders were less likely to have renal cell carcinoma and more likely to have clear cell carcinoma.
Sample characteristics
The demographic and medical characteristics of participants are displayed in Table 1. Descriptive statistics for PA variables are displayed in Table 2. Overall, 28.5 % of KCS were meeting public health PA guidelines. Chi-squared analyses revealed some demographic and medical group differences for meeting PA guidelines as illustrated in Table 1. KCS were more likely to meet PA guidelines if they were female [χ 2(1) = 5.62, p = .018], had some/completed university [χ 2(1) = 9.14, p = .003], and had localized kidney cancer [χ 2(1) = 9.26, p = .002].
Results of the logistic regression for the social-ecological variables are presented separately for each block (see Table 3). When analyzing the models separately, demographic variables such as being female (OR = 1.67, 95 % CI = 1.08–2.61) and having some/completed university (OR = 1.83, 95 % CI = 1.16–2.89) were significantly related to meeting PA guidelines. In terms of medical variables, disease stage was significantly related to meeting PA guidelines (OR = 0.32, 95 % CI = 0.15–0.72). Regarding the social-cognitive variables, higher levels of affective (OR = 1.49, 95 % CI = 1.10–2.00) and instrumental attitudes (OR = 2.34, 95 % CI = 1.47–3.72), higher levels of PBC (OR = 1.62, 95 % CI = 1.29–2.04), higher levels of planning (OR = 1.78, 95 % CI = 1.56–2.05), and higher levels of intention (OR = 2.23, 95 % CI = 1.91–2.78) were significantly related to meeting PA guidelines. In terms of perceived environmental characteristics, land use mix-diversity (i.e., greater number of shops within walking distance from home) (OR = 1.29, 95 % CI = 1.04–1.61) was significantly related to meeting PA guidelines. Objective built environment characteristics were not related to meeting PA guidelines in KCS.
Hierarchical logistic regression consisted of seven models. Model 1 included demographic variables and has been presented above where being female and having some/completed university-level education was associated with meeting PA guidelines. Model 1 was found to have a good fit based on the Omnibus test [χ 2(6) = 16.37, p = .012] and the Hosmer and Lemeshow test [χ 2(8) = 9.83, p = .28]. Model 1 explained 5.3 % (Nagelkerke R 2) of the variance in PA, with the overall accuracy of classification of 71.5 % for meeting and not meeting PA guidelines.
Model 2 included both demographic and medical variables. Model 2 was found to have a good fit based on the Omnibus test [χ 2(13) = 28.43, p = .008], but not a good fit based on the Hosmer and Lemeshow test [χ 2(8) = 16.94, p = .031]. Model 2 explained 9.1 % (Nagelkerke R 2) of the variance in PA, with the overall accuracy of classification of 71.8 %. Of the demographic and medical variables included, having some/completed university (OR = 1.84, 95 % CI = 1.16–2.93) and disease stage (OR = 0.35, 95 % CI = 0.15–0.79) were significant predictors of meeting PA guidelines.
Social-cognitive variables were entered in three steps (models 3–5). Model 5 included demographic and medical variables, as well as the social-cognitive variables. Model 5 was found to have a good fit based on the Omnibus test [χ 2(20) = 158.48, p < .001] and the Hosmer and Lemeshow test [χ 2(8) = 7.66, p = .47]. Model 5 explained 44.0 % (Nagelkerke R 2) of the variance in PA, with the overall accuracy of classification of 80.1 %. Of the variables included, disease stage (OR = 0.27, 95 % CI = 0.11–0.71), having drug therapy (OR = 4.13; 95 % CI = 1.52–11.17), higher levels of instrumental attitudes (OR = 1.62, 95 % CI = 0.99–2.63), and higher levels of intention (OR = 0.71, 95 % CI = 0.59–0.84) were significant predictors of meeting PA guidelines.
Model 6 included demographic, medical, social-cognitive, and perceived environment variables. Model 6 was found to have a good fit based on the Omnibus test [χ 2(26) = 165.59, p < .001] and the Hosmer and Lemeshow test [χ 2(8) = 7.51, p = .48]. Model 6 explained 45.7 % (Nagelkerke R 2) of the variance in PA, with the overall accuracy of classification of 81.3 %. Of the variables included, disease stage (OR = 0.25, 95 % CI = 0.10–0.66), having drug therapy (OR = 4.16, 95 % CI = 1.51–11.46), higher levels of instrumental attitudes (OR = 1.68, 95 % CI = 1.02–2.76), higher levels of intention (OR = 1.70, 95 % CI = 1.23–2.35), and the presence of many retail shops in the neighborhood (OR = 1.33, 95 % CI = 1.01–1.75) were significant predictors of meeting PA guidelines.
The final model included demographic, medical, social-cognitive, perceived environment, and built environment variables. Model 7 was found to have a good fit based on the Omnibus test [χ 2(33) = 169.02, p < .001] and the Hosmer and Lemeshow test [χ 2(8) = 4.57, p = .80]. Model 7 explained 46.4 % (Nagelkerke R 2) of the variance in PA, with the overall accuracy of classification of 80.3 %. Of the variables included, disease stage (OR = 0.25, 95 % CI = 0.10–0.66), having drug therapy (OR = 3.98; 95 % CI = 1.41–11.21), higher levels of instrumental attitudes (OR = 1.66, 95 % CI = 1.00–2.78), higher levels of intention (OR = 1.71; 95 % CI = 1.23–2.39), and the presence of many retail shops in the neighborhood (OR = 1.37, 95 % CI = 1.03–1.81) were significant predictors of meeting PA guidelines. These variables were in the range of small to medium effect sizes [41].
Discussion
This study provides a comprehensive examination of the correlates of PA using a social-ecological approach in KCS. To the best of our knowledge, this is the first study addressing the correlates of PA using a social-ecological model that includes objective measures of the physical environment in KCS and in any cancer survivor group. Notably, the introduction of the social-cognitive variables (i.e., TPB) contributed to the largest variance explained in meeting PA guidelines resulting in a total variance explained of 44.0 %, but the effect sizes were small. The addition of the perceived and objective built environment characteristics contributed an additional 1.7 and 2.4 % of the variance explained in meeting PA guidelines, respectively, but the effect sizes were small. This resulted in a total explained variance of 45.7 % when the perceived environment characteristics were added. The final model including the objective built environment characteristics resulted in a total explained variance of 46.4 %, but none of the objectively measured environmental attributes were significant predictors of meeting PA guidelines for KCS. In the final model, only perceived proximity of retail shops was a significant predictor of meeting PA guidelines.
In the seven-step prediction model, the introduction of the social-cognitive variables contributed to an additional 34.9 % of the variance in meeting PA guidelines. These findings are consistent with previous TPB studies in cancer survivors where 11–42 % of the variance in PA was accounted for [6–10, 24, 42, 43]. In a systematic review examining the determinants of exercise adherence and maintenance in cancer survivors using a socio-ecological approach, inconsistent evidence was found for psychological factors such as stage of change, attitude, intention, PBC, and self-efficacy with exercise adherence and maintenance [19]. In contrast to this review, Husebo et al. [44] found in their meta-analysis exercise stage of change, intention, PBC, and subjective norm to be significant determinants of exercise adherence. These inconsistent findings may be attributed to the relatively small sample sizes and low variability of exercise adherence and maintenance. Although most demographic and medical variables are not modifiable, focusing on social-cognitive factors that are modifiable could contribute to behavior change in future PA interventions.
Demographic variables were not significant predictors of meeting PA guidelines, which was inconsistent with previous findings in cancer survivors. Speed-Andrews et al. [5] found that demographic variables such as age, marital status, employment, education level, and income status were related to meeting PA guidelines. D’Andrea and colleagues [12] reported colorectal cancer survivors with some college education/degree and with less than two chronic conditions were more likely to comply with PA guidelines. The only medical variables to be significant predictors of meeting PA guidelines in KCS were having localized disease and drug therapy. This is consistent with a previous study in colorectal cancer survivors, where those that were disease-free, in better health, had less comorbidities, and not receiving radiation had higher PA levels [5]. Our findings were also somewhat consistent with a previous study in a mixed sample of breast, prostate, and colorectal cancer survivors, where prostate cancer survivors were more likely to meet PA guidelines given the type of treatment they received (i.e., having surgery, but not chemotherapy) [6]. There has been limited research in the area of demographic and medical correlates of PA in KCS, and it is possible that treatment-related variables also play an important role in engaging in PA. For example, metastatic KCS often receive drug therapy in the form of targeted therapy which avoids harm to healthy cells, which, in turn, has fewer side effects than standard chemotherapy, and is well tolerated [45]. Many of the treatment-related adverse reactions can be readily managed in most cases after a dosage reduction, treatment interruption, or other supportive care measures, and thus, KCS may be more motivated to engage in PA [45]. PA interventions should consider treatment-related correlates of PA, especially in terms of type and mode of delivery for PA.
Social-cognitive variables including instrumental attitudes and intention were significant predictors of meeting PA guidelines. These correlates are consistent with previous studies in other cancer survivor groups [5, 7, 8, 10, 46]. In a study of KCS by our research group using the same dataset but with the full sample (N = 703), Trinh and colleagues [24] reported that PBC, intention, and planning were significant predictors of PA, and instrumental attitude, descriptive norm, and PBC were significant predictors of intention.
Beyond these social-cognitive variables, the only environmental variable to be a significant predictor of meeting PA guidelines was perceived proximity of retail shops. These findings parallel previous studies in the general population of adults where close proximity to retail infrastructure was associated with walking [35, 47]. None of the objective built environmental characteristics were significant predictors of meeting PA guidelines in KCS. Research examining the environmental correlates of PA in cancer survivors has remained scant making comparative efforts difficult. There have been mixed reviews regarding research examining environmental correlates of PA. Previous reviews in apparently healthy adults identified a small number of correlates including environmental attributes such as recreation facilities and locations, transportation environments, and aesthetics as correlates of PA [13]. Among older adults, a recent systematic review found limited associations between the physical environment (e.g., quality of sidewalks, access to parks) and PA in older adults (≥65 years) [22]. In another systematic review of 67 studies examining predictors of PA change among adults, Rhodes and Quinlan [48] found no associations between environmental factors such as access, aesthetics, and safety with PA change.
Only one study in colorectal cancer survivors has examined the physical environment as a potential barrier to PA [23]. For example, limited access to recreation facilities was not related to achieving sufficient levels of PA [23]. In addition, one systematic review examining the determinants of exercise adherence and maintenance among cancer survivors found that the location of the fitness center was not associated with exercise maintenance, and inconsistent findings were noted for location of the fitness center with exercise adherence [19]. Given that KCS are an older cancer survivor group, it may be that older adults tend to live in similar types of areas or in smaller communities such as condominiums or apartment complexes. These types of dwellings may have easy access to amenities located within the building premises, and thus, the characteristics of the built environment may be less relevant for PA [20].
PA in KCS is largely explained by social-cognitive factors, followed by treatment-related factors and the perceived environment. It is likely that the inclusion of the social-cognitive factors influenced the associations for the treatment-related variables, where the odds ratios increased for disease stage and drug therapy. These stronger associations in the treatment-related variables may be contributing to additional variance when taken together with the social-cognitive variables. Nevertheless, there were few factors at any of these levels that explained PA. It is possible that enhancing the social-cognitive correlates of PA such as the benefits of PA may be more important for KCS when forming an intention to engage in PA. This may be due to differences in health and age, where KCS are more likely to be overweight or obese and have other comorbidities compared to other survivor groups. Therefore, KCS may be more likely to intend to engage in PA if they feel that it would be beneficial. In terms of environmental variables, community planners should consider factors such as neighborhood design when building new communities. Overall, few factors may need to be taken into consideration when designing PA interventions in KCS, and it may be likely that interventions designed for apparently healthy older adults may result in behavior change in KCS.
Although the built environment was not related to PA in this sample, we cannot rule out its influence due to methodological concerns. Both objective and perceived measures of the physical environment relate differently to PA, and previous studies suggest that perceptions of the environment are more strongly related to PA compared to objective measures [22]. Ecological models have posited that environmental factors influence behavior directly and indirectly through individual perceptions [15]. More research is needed to understand the complex interaction between the objective and perceived environment for predicting PA.
Our study should be interpreted within the context of important strengths and limitations. To the best of our knowledge, this is the first study examining the relative influence of demographic, medical, behavioral, and environmental correlates of PA in KCS within a social-ecological model. This also appears to be the first study to employ both perceived and objective measures of the physical environment to understand PA in a large, population-based sample of KCS. Limitations of our study include the cross-sectional nature of the study in which causation cannot be implied. Future studies should consider employing longitudinal designs to further understand the social-ecological correlates of PA in cancer survivors. Another limitation of our study was the reliance on self-reported measures of PA which can be subject to recall biases and error. Given the large number of comparisons made in this study without adjustment for multiple testing, there may be a possibility of some chance findings. However, many of these findings are consistent with previous research examining predictors of PA with cancer survivors. Future research should consider objective measures of PA such as accelerometry. Finally, future research should consider using multiple buffer radii (i.e., 0.5 km) given that it may contribute to differences in correlates of meeting PA guidelines in nearby and distant environments for KCS. Additional analyses were conducted using a 2 km buffer radius, but there were no differences in the PA correlates of KCS compared with the 1 km buffer radius. Obtaining residential addresses are also necessary for future research to compare KCS residing in rural and urban locations.
In conclusion, our study found preliminary support for using the social-ecological framework for understanding PA in KCS. However, only a few select medical, behavioral, and perceived environmental factors predicted PA, with social-cognitive factors largely contributing to KCS meeting PA guidelines. Our study expands upon previous studies examining the correlates of PA by considering the physical environment to gain a comprehensive understanding of these factors for achieving PA guidelines. PA interventions should still address multiple facets of the social-ecological model such as at the community, organizational, and policy level given its impact on behavior change. Within these multilevel interventions, particular emphasis on social-cognitive factors such as the benefits of PA may contribute to behavior change. These interventions should also consider treatment-related factors, especially disease stage and drug therapy regimes that may influence the mode and delivery of PA. Few correlates of the physical environment have been identified, suggesting that PA interventions targeting environmental attributes could be ineffective in behavior change. More research is needed to understand the influence of the built environment on PA, in addition to demographic, medical, and behavioral factors. Overall, focusing on social-cognitive aspects of PA may be a more immediate strategy for KCS to achieve PA guidelines for enhancing QoL and health outcomes.
References
Mishra S, Scherer R, Geigle P, Berlanstein D, Topaloglu O, Gotay C, et al. Exercise interventions on health-related quality of life for cancer survivors (review). Cochrane Database Syst Rev. 2012;8:CD007566.
Trinh L, Plotnikoff RC, Rhodes RE, North S, Courneya KS. Associations between physical activity and quality of life in a population-based sample of kidney cancer survivors. Cancer Epidemiol Biomarkers Prev. 2011;20:859–68.
Rock CL, Doyle C, Demark‐Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL, et al. Nutrition and physical activity guidelines for cancer survivors. CACancer J Clin. 2012;62:242–74.
Schmitz KH, Courneya KS, Matthews C, Demark-Wahnefried W, Galvão DA, Pinto BM, et al. American college of sports medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42:1409–26.
Speed-Andrews A, Rhodes R, Blanchard C, Culos-Reed S, Friedenreich C, Belanger L, et al. Medical, demographic and social cognitive correlates of physical activity in a population‐based sample of colorectal cancer survivors. Eur J Cancer Care. 2012;21:187–96.
Forbes CC, Blanchard CM, Mummery WK, Courneya KS. A comparison of physical activity correlates across breast, prostate and colorectal cancer survivors in Nova Scotia, Canada. Support Care Cancer. 2014;22:891–903.
Stevinson C, Tonkin K, Capstick V, Schepansky A, Ladha AB, Valance JK, et al. A population-based study of the determinants of physical activity in ovarian cancer survivors. J Phys Act Health. 2009;6:339–46.
Karvinen KH, Courneya KS, Plotnikoff RC, Spence JC, Venner PM, North S. A prospective study of the determinants of exercise in bladder cancer survivors using the theory of planned behavior. Support Care Cancer. 2009;17:171–9.
Courneya KS, Vallance JK, Jones LW, Reiman T. Correlates of exercise intentions in non-Hodgkin's lymphoma survivors: an application of the Theory of Planned Behavior. JSEP. 2005;27:335–49.
Karvinen KH, Courneya KS, Campbell KL, Pearcey RG, Dundas G, Capstick V, et al. Correlates of exercise motivation and behavior in a population-based sample of endometrial cancer survivors: an application of the theory of planned behavior. Int J Behav Nutr Phys Act. 2007;4:21.
Coups EJ, Park BJ, Feinstein MB, Steingart RM, Egleston BL, Wilson DJ, et al. Correlates of physical activity among lung cancer survivors. Psycho-Oncology. 2009;18:395–404.
D'Andrea AP, Fernandez CA, Tannenbaum SL, Clarke TC, McClure LA, LeBlanc WG, et al. Correlates of leisure time physical activity compliance in colorectal cancer survivors. Prev Med. 2014;62:78–82.
Bauman AE, Reis RS, Sallis JF, Wells JC, Loos RJ, Martin BW. Correlates of physical activity: why are some people physically active and others not? Lancet. 2012;380:258–71.
Hall KS, McAuley E. Individual, social environmental and physical environmental barriers to achieving 10 000 steps per day among older women. Health Educ Res. 2010;25:478–88.
Sallis JF, Owen N, Fisher EB. Ecological models of health behavior. Health Behav Health Educ: Theory, Res Pract. 2008;4:465–85.
Stokols D. Translating social ecological theory into guidelines for community health promotion. Am J Health Promot. 1996;10:282–98.
McLaren L, Hawe P. Ecological perspectives in health research. J Epidemiol Community Health. 2005;59:6–14.
Saelens BE, Handy SL. Built environment correlates of walking: a review. Med Sci Sports Exerc. 2008;40:S550–66.
Kampshoff CS, Jansen F, van Mechelen W, May AM, Brug J, Chinapaw MJ, et al. Determinants of exercise adherence and maintenance among cancer survivors: a systematic review. Int J Behav Nutr Phys Act. 2014;11:80-5868–11-80.
King AC, Sallis JF, Frank LD, Saelens BE, Cain K, Conway TL, et al. Aging in neighborhoods differing in walkability and income: associations with physical activity and obesity in older adults. Soc Sci Med. 2011;73:1525–33.
Frank L, Kerr J, Rosenberg D, King A. Healthy aging and where you live: community design relationships with physical activity and body weight in older Americans. J Phys Act Health. 2010;7:82–90.
Van Cauwenberg J, De Bourdeaudhuij I, De Meester F, Van Dyck D, Salmon J, Clarys P, et al. Relationship between the physical environment and physical activity in older adults: a systematic review. Health Place. 2011;17:458–69.
Lynch BM, Owen N, Hawkes AL, Aitken JF. Perceived barriers to physical activity for colorectal cancer survivors. Support Care Cancer. 2010;18:729–34.
Trinh L, Plotnikoff RC, Rhodes RE, North S, Courneya KS. Correlates of physical activity in population-based sample of kidney cancer survivors: an application of the theory of planned behavior. Int J Behav Nutr Phys Act. 2012;9:96.
Statistics Canada. 2011 Census of population: population, urban and rural, by province and territory. Available from: http://www.statcan.gc.ca/tables-tableaux/sum-som/l01/cst01/demo62j-eng.htm.
Government of Alberta Climate and Geography. Available from: http://www.albertacanada.com/opportunity/choosing/province.aspx.
Godin G, Shephard RJ. A simple method to assess exercise behavior in the community. Can J Appl Sport Sci. 1985;10:141–6.
Ajzen I. Perceived Behavioral Control, Self-Efficacy, Locus of Control, and the Theory of Planned Behavior. J Appl Soc Psychol. 2002;32:665–83.
Ajzen I. The theory of planned behavior. Organ Behav Hum Decis Process. 1991;50:179–211.
Vallance JK, Courneya KS, Plotnikoff RC, Mackey JR. Analyzing theoretical mechanisms of physical activity behavior change in breast cancer survivors: results from the activity promotion (ACTION) trial. Ann Behav Med. 2008;35:150–8.
Courneya KS, Stevinson C, McNeely ML, Sellar CM, Friedenreich CM, Peddle-McIntyre CJ, et al. Effects of supervised exercise on motivational outcomes and longer-term behavior. Med Sci Sports Exerc. 2012;44:542–9.
Rise J, Thompson M, Verplanken B. Measuring implementation intentions in the context of the theory of planned behavior. Scand J Psychol. 2003;44:87–95.
Cerin E, Saelens BE, Sallis JF, Frank LD. Neighborhood Environment Walkability Scale: validity and development of a short form. Med Sci Sports Exerc. 2006;38:1682–91.
Sallis JF, Bowles HR, Bauman A, Ainsworth BE, Bull FC, Craig CL, et al. Neighborhood environments and physical activity among adults in 11 countries. Am J Prev Med. 2009;36:484–90.
Rhodes RE, Courneya KS, Blanchard CM, Plotnikoff RC. Prediction of leisure-time walking: an integration of social cognitive, perceived environmental, and personality factors. Int J Behav Nutr Phys Act. 2007;4:51.
Owen N, Humpel N, Leslie E, Bauman A, Sallis JF. Understanding environmental influences on walking: review and research agenda. Am J Prev Med. 2004;27:67–76.
Bow CJ, Waters NM, Faris PD, Seidel JE, Galbraith PD, Knudtson ML, et al. Accuracy of city postal code coordinates as a proxy for location of residence. Int J Health Geogr. 2004;3:5.
Frank LD, Schmid TL, Sallis JF, Chapman J, Saelens BE. Linking objectively measured physical activity with objectively measured urban form: findings from SMARTRAQ. Am J Prev Med. 2005;28:117–25.
Kemperman A, Timmerman H. Influences of built environment on walking and cycling by latent segments of aging population. Transp Res Rec: J Transp Res Board. 2009;2134:1–9.
Statistics Canada. Median total income, by family type, by province and territory. Available from: http://www.statcan.gc.ca/tables-tableaux/sum-som/l01/cst01/famil108a-eng.htm.
Chen H, Cohen P, Chen S. How big is a big odds ratio? Interpreting the magnitudes of odds ratios in epidemiological studies. Commun Stat Simul Comput. 2010;39:860–4.
Jones LW, Courneya KS, Vallance JK, Ladha AB, Mant MJ, Belch AR, et al. Understanding the determinants of exercise intentions in multiple myeloma cancer survivors: an application of the theory of planned behavior. Cancer Nurs. 2006;29:167–75.
Blanchard CM, Courneya KS, Rodgers WM, Murnaghan DM. Determinants of exercise intention and behavior in survivors of breast and prostate cancer: an application of the theory of planned behavior. Cancer Nurs. 2002;25:88–95.
Husebø AML, Dyrstad SM, Søreide JA, Bru E. Predicting exercise adherence in cancer patients and survivors: a systematic review and meta‐analysis of motivational and behavioural factors. J Clin Nurs. 2013;22:4–21.
Hutson TE, Figlin RA, Kuhn JG, Motzer RJ. Targeted therapies for metastatic renal cell carcinoma: an overview of toxicity and dosing strategies. Oncologist. 2008;13:1084–96.
Bélanger LJ, Plotnikoff RC, Clark AM, Courneya KS. Determinants of physical activity in young adult cancer survivors. Am J Health Behav. 2012;36:483–94.
Rhodes RE, Brown SG, McIntyre CA. Integrating the perceived neighborhood environment and the theory of planned behavior when predicting walking in a Canadian adult sample. Am J Health Promot. 2006;21:110–8.
Rhodes RE, Quinlan A. Predictors of physical activity change among adults using observational designs. Sports Med. 2014:1-19.
Acknowlegments
LT is supported by a research trainee award from the Kidney Cancer Research Network of Canada (KCRNC). GEF is supported by a Canadian Institutes of Health Research-Public Health Agency of Canada (CIHR-PHAC) Chair in Applied Public Health. RCP is supported by a Senior Research Fellowship from the National Health and Medical Research Council of Australia. RER is supported by an Investigator award from the Canadian Cancer Society. KSC is supported by the Canada Research Chairs Program. We thank Carol Russell and Lorraine Cormier from the Alberta Cancer Registry for their assistance in conducting this study.
Conflicts of interest
The authors have no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trinh, L., Larsen, K., Faulkner, G.E. et al. Social-ecological correlates of physical activity in kidney cancer survivors. J Cancer Surviv 10, 164–175 (2016). https://doi.org/10.1007/s11764-015-0462-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-015-0462-y