Abstract
trans-10,cis-12 (t10c12) Conjugated linoleic acid (CLA) reduced body lipid deposition in various experimental animals, but the mechanisms involved were still emerging. Carnitine palmitoyltransferase I (CPT I) catalyzes an important regulatory step in lipid metabolism. At present, no studies, to our knowledge, have evaluated the kinetic constants influenced by dietary CLA in fish. In the present study, we tested the hypothesis that changes in body lipid content in fish as a response to dietary t10c12 CLA was related to the change of CPT I kinetic constants [Michaelis constant (K m), maximal velocity and catalytic efficiency for carnitine and palmitoyl-CoA]. Juvenile Synechogobius hasta were fed three experimental diets with fish oil replaced with 0 (control), 1, or 2 % t10c12 CLA for 8 weeks. Weight gain, specific growth rate and protein efficiency rate increased with dietary t10c12 CLA level. Dietary t10c12 CLA addition significantly reduced lipid contents both in liver and muscle. Dietary CLA addition also improved CPT I activities in muscle but did not significantly influence hepatic CPT I activity. CPT I kinetic parameters (K m, V max and catalytic efficiency) were significantly influenced by t10c12 CLA. CPT I catalytic efficiencies with carnitine and palmitoyl-CoA as substrates were higher in muscle and liver of fish fed increasing t10c12 CLA. For the first time, the findings demonstrated effect of dietary CLA addition on CPT I kinetics in fish and supported our starting hypothesis that dietary t10c12 CLA addition induced alterations in CPT I kinetic constants of muscle and liver. Increased CPT I catalytic efficiency might be the main reason for reduced lipid deposition in these tissues by dietary t10c12 CLA supplementation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Conjugated linoleic acid (CLA) is a term used to describe positional and geometric isomers of linoleic acid (18:2n-6) [1]. These compounds occur mainly in beef and dairy products but are widespread in lower levels in many foodstuffs [1]. In animal models and humans, feeding CLA mixtures reduces body fat and increases lean body mass [2], and influences lipid metabolism [3]. The effects of dietary CLA supplements on growth performance and lipid metabolism have been studied in many fish species [4–10]. The CLA preparation used in these studies consist of two major CLA isomers, cis-9,trans-11 (c9t11) and trans-10,cis-12 (t10c12) isomers. However, to our knowledge, no research has reported the effect of a single CLA isomer on growth performance and lipid metabolism in fish. Studies in terrestrial animals indicate the differences between two isomers on a number of biological functions [11–13], and t10c12 CLA rather than c9t11 CLA is the metabolite responsible for the lipid-lowering effect of CLA [14].
Carnitine palmitoyltransferase I (CPT I, EC 2.3.1.21) is a major regulatory enzyme of lipid metabolism, required for the conversion of fatty acid-CoAs into fatty acid-carnitines for entry into the mitochondrial matrix [15]. In vertebrates, CPT I is frequently described as the ‘rate-limiting enzyme’ of β-oxidation flux [16, 17], and therefore is the main regulatory step of fatty acid oxidation [18]. Estimating kinetic constants are one of the most critical parts of the studies related to enzyme-catalyzed reactions. In that instance, elucidation of the kinetic mechanism of CPT I as a result of dietary CLA addition will increase our knowledge about the nutrient physiology of CLA and its critical role in lipid deposition. However, to our knowledge, at present, no information is available on the effect of CLA addition on kinetics of CPT I in animals, including fish.
Synechogobius hasta are widely distributed over the southern coast of Liaoning Province, China. In recent years, commercial farming of this fish has become an increasing interest in northern China because of its euryhalinity, rapid growth, good taste and high market value. The fish is considered to be a potential candidate model for studying lipid metabolism in fish. Recently, we have conducted a series of experiment to investigate the response of the related metabolic enzyme to waterborne and diet-borne materials [19–22]. One of the lingering concerns with S. hasta has been the lipid accumulation in hepatocytes of fish under natural conditions, which in turn increases mortality during farming. The hypotheses of the present study are that t10c12 CLA can reduce lipid deposition of liver and muscle in S. hasta by inducing the change of activity and kinetic constants of CPT I. Growth performance, body composition, CPI I activity and kinetic constant (K m, V max and catalytic efficiency with carnitine and palmitoyl-CoA as substrates, respectively) in muscle and liver are determined in S. hasta after feeding three experimental diets containing different t10c12 CLA levels.
Materials and Methods
Ingredients and Experimental Diets
Three isonitrogenous (40 % CP) experimental diets were formulated to contain increasing t10c12 CLA levels of 0, 1 and 2 %, respectively. The ingredients and proximate analysis of the diets are shown in Table 1. 0.01 % butylated hydroxytoluene (BHT) was used as an antioxidant. Highly purified t10c12 CLA isomer came from Natural Lipids Ltd. (SA, Industriveien, Hovdebygda, Norway).
The experimental diets were produced according to the methods described in our recent study [22]. Briefly, all dry ingredients were ground to pass through a 120-μm sieve, weighed, and mixed to a unanimous homogeneity. t10c12 CLA was added to fish oil and mixed until homogenous. The oil mixture was then added to the homogenous dry ingredients and mixed thoroughly. Then the pre-weighed distilled water (10 %, v/w) was added to form a dough. The diets were extruded through a 1.0-mm sieve using a laboratory model Muyang Extruder Machine (Jiangsu, China). The resulting strands were dried with forced air circulation at room temperature, crumbled to approximately 1 mm length and kept in the freezer at −20 °C until used.
The care and treatment of the experimental animals conformed to the guidelines of Huazhong Agricultural University for the ethical treatment of laboratory animals.
Experimental Procedures
The experiment was conducted at Panjin Guanghe Fishery Co., Ltd., Panjin, China. Three hundred juvenile S. hasta were collected from a local fish pond (Panjin, China) and kept in five 300-L circular fiberglass tanks for 14-day acclimatization. During the acclimatization period, the fish were fed minced trash fish mixed with the control diet (Table 1). The amount of trash fish was more than that of the control diet at the start of acclimatization but the amount of the diet was progressively increased until the fish readily accepted the diet. At the beginning of the experiment, 20 fish [3.54 ± 0.08 g, mean ± standard error of means (SEM)] of equal size and in good condition were randomly distributed in each fiberglass tank (300 L) with continuous aeration to maintain the dissolved oxygen level near saturation. Each experimental diet was randomly allotted to three replicate tanks. In total, nine tanks were used in the present experiment. The fish were fed to apparent satiation twice daily (0900 and 1600 hours) during the week. Water in each tank was replenished 100 % twice daily, before feeding. Care was taken to ensure that no uneaten feed remained in the tanks during feeding. The amount of feed consumed by the fish in each tank was recorded daily. Fecal matter was also quickly removed during the experiment. Fish were bulk weighed every 2 weeks. Tanks were thoroughly cleaned when the fish were removed for weighing. Mortality was checked daily. The experiment continued for 8 weeks.
The experiment was conducted at ambient temperature and subjected to the natural photoperiod (approximately 14-h light/10-h dark). Water quality parameters were monitored twice a week in the morning. Water temperature ranged from 23.1 to 28.7 °C; dissolved oxygen ≥5.6 mg/L; pH 7.9–8.3; total ammonia–nitrogen 0.045–0.058 mg/L.
Sampling and Sample Analysis
At the conclusion of the 8-week period, 24 h after the last feeding, all fish were counted and weighed to determine survival, weight gain (WG), specific growth rate (SGR), feed conversion rate (FCR) and protein efficiency rate (PER). After obtaining the final total weight of fish in each aquarium, six fish from each tank were dissected to obtain liver and muscle samples, and quickly frozen at −80 °C for proximate analysis (moisture, crude protein, lipid and ash). The remaining fish from each aquarium were also dissected to obtain liver and muscle samples, and quickly determined for CPT I enzyme activity and kinetic constants.
For proximate analysis of the experimental diet, muscle and liver, crude protein (N× 6.25) content was determined by the Kjeldahl method after an acid digestion using an auto Kjeldahl System (2300-Auto-analyzer, Sweden), lipids by ether–extraction, moisture by oven drying at 105 °C for 24 h and ash using a muffle furnace at 550 °C for 24 h [23].
For assays of CPT I activity, the samples were homogenized on ice using a hand-driven ground-glass tissue grinder. The mitochondria were extracted according to Suarez and Hochachka [24] with modifications by Morash and McClelland [18]. Mitochondrial isolation buffer (MIB) consisted of 250 mM sucrose, 1 mM EDTA, 20 mM HEPES and 0.5 % bovine serum albumin (BSA) (pH 7.4). Homogenates were centrifuged at 800×g for 10 min at 4 °C. The supernatant was centrifuged at 9,000×g for 10 min at 4 °C to obtain the mitochondrial pellet. The latter were resuspended in a small volume of the appropriate MIB lacking BSA. The resuspended homogenate was collected into a 15-mL centrifuge tube and centrifuged again at 9,000×g for 10 min at 4 °C. The mitochondrial pellet was resuspended in an appropriate volume of MIB lacking BSA. The CPT I activity analyses were conducted at 25 °C as described by Bremer et al. [25]. The assay measured the rate for formation of palmitoylcarnitine from palmitoyl-CoA and carnitine. The CPT I activity was expressed as nmol palmitoylcarnitine produced/(min mg protein). Soluble protein concentration of homogenates was determined by the method of Bradford [26] using BSA as standard. These analyses (the related enzymatic activities and soluble protein concentration) were conducted in duplicate.
For the kinetic studies, the ranges of substrate concentrations for carnitine were from 0.25 to 16 mM, and for palmitoyl-CoA from 0.05 to 0.64 mM. The enzymatic reaction was initiated by adding palmitoyl-CoA (100 μM) and carnitine (400 mM) to generate palmitoylcarnitine and incubated at 25 °C. Analysis of the kinetic data was performed as described by Hofstee [27]. The values of the Michaelis–Menten constants (K m) and maximal reaction rates (V max) were analyzed using a non-linear regression method described by the Michaelis–Menten equation. Lineweaver–Burk graphs [28] were drawn by using 1/v versus 1/[S] values. Catalytic efficiency, defined as an enzyme’s efficiency in transforming its substrate, was calculated by the ratio between maximum enzyme activity and K m (V max/K m). All measurements were performed in duplicate.
Statistical Analysis
The results were presented as means ± SEM of three replicates. Data from each treatment were subjected to one-way analysis of variance (ANOVA). When overall differences were significant (P < 0.05), Duncan’s multiple range test was used to compare the mean values among the treatments. Statistical analysis was performed using the SPSS 10.0 for Windows (SPSS, Michigan Avenue, Chicago, IL, USA).
Results
WG and SGR increased with increasing dietary t10c12 CLA levels from 0 to 2 % (Table 2). Dietary t10c12 CLA levels showed no significant effects on FI, FCR and survival. The highest PER were observed for fish fed 2 % of t10c12 CLA. Dietary t10c12 CLA inclusion did not significantly influence moisture and ash contents in muscle. Increasing muscle protein content was observed in fish with increasing dietary t10c12 CLA level but the differences were not statistically significant. Dietary t10c12 CLA levels significantly reduced muscle lipid content. As a result, the ratio of lipid to protein in muscle declined with increasing dietary t10c12 CLA levels. Dietary t10c12 CLA levels did not significantly influence moisture, protein and lipid contents, and the ratio of lipid to protein of liver (Table 3).
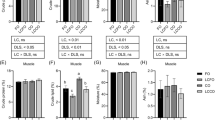
The synthesis of palmitoyl-carnitine was assayed as a function of both substrates: palmitoyl-CoA and l-carnitine. The reaction followed a normal Michaelis–Menten kinetics in the substrate concentration range as shown in Lineweaver–Burk plots (Figs. 1, 2, 3, 4).
Lineweaver–Burk plots of initial velocity against carnitine at varied substrate concentration for liver of S. hasta. Values represent the means and standard error of mean (SEM), n = 3. The values of R 2 for fish fed diet 1 (filled squares), diet 2 (open triangles) and diet 3 (solid line) were 0.99, 0.99, 0.98, respectively
Lineweaver–Burk plots of initial velocity against carnitine at varied substrate concentration for muscle of S. hasta. Values represent the means and standard error of mean (SEM), n = 3. The values of R 2 for fish fed diet 1 (filled squares), diet 2 (open triangles) and diet 3 (solid line) were 0.99, 0.98, 0.99, respectively
Lineweaver–Burk plots of initial velocity against palmitoyl-CoA at varied substrate concentration for liver of S. hasta. Values represent the means and standard error of mean (SEM), n = 3. The values of R 2 for fish fed diet 1 (filled squares), diet 2 (open triangles) and diet 3 (solid line) were 0.98, 0.98, 0.98, respectively
Lineweaver–Burk plots of initial velocity against palmitoyl-CoA at varied substrate concentration for muscle of S. hasta. Values represent the means and standard error of mean (SEM), n = 3. The values of R 2 for fish fed diet 1 (filled squares), diet 2 (open triangles) and diet 3 (solid line) were 0.98, 0.98, 0.97, respectively
No significant differences were observed in hepatic CPT I activities among the treatments (Table 4). When using carnitine as substrate, constant V max, reduced K m and increased catalytic efficiency were observed in livers of fish fed increasing t10c12 CLA isomer. When using palmitoyl-CoA as substrate, constant K m, increasing V max and catalytic efficiency were observed in livers of fish fed increasing t10c12 CLA isomer.
Dietary t10c12 CLA addition significantly improved muscle CPT I activity (Table 5). When using carnitine as substrate, increasing K m, V max and catalytic efficiency were observed in muscle of fish fed increasing t10c12 CLA isomer. When using palmitoyl-CoA as substrate, reduced K m, increasing V max and catalytic efficiency were observed in muscle of fish fed increasing t10c12 CLA isomer.
Discussion
In the present study, dietary t10c12 CLA isomer addition significantly improved growth performance and feed utilization. Similarly, Choi et al. [29] pointed out that common carp fed diets containing 1.0 % CLA exhibited significant improvements in growth rate. Increasing feed efficiency has also been reported by Berge et al. [30]. In contrast, growth performance and feed utilization are not affected by dietary CLA inclusion in other fish species [5–8, 10, 30–32]. In the present study, dietary CLA levels show no significant effect on FI, similar to those reports in mice, rats and pigs [2, 33–36]. In contrast, studies suggest that dietary CLA addition significantly reduces FI in several fish species [4, 9, 32, 37]. From the various results above, we can conclude that the CLA effect on growth performance and feed efficiency is clearly dependent on the species considered and also on the CLA isomers.
Our study indicates that dietary t10c12 CLA inclusion significantly reduces lipid contents of muscle and liver. Similarly, studies on yellow perch and hybrid striped bass have demonstrated reduced hepatic lipids after CLA feeding [4, 5]. The t10c12 CLA reduces hepatic triacylglycerol (TAG) in hamster [12], a similar decrease occurs in total body TAG levels in salmon [8]. Moreover, TAG levels in muscle are also lowered in response to the CLA diets [37]. The view that CLA affects lipid accumulation both by decreasing de-novo FA synthesis and by increasing oxidation is well established in mammals and cell lines. For example, CLA inhibits fatty acid (FA) synthetase activity in rat liver [38], and suppresses TAG accumulation and increases FA oxidation in 3T3-L1 adipocytes [39]. However, it is unclear whether CLA has the ability to alter lipid metabolism in fish in a manner similar to that of mammalian models [30]. Our data indicate that the lipid-lowering effect of t10c12 CLA is, at least partly, due to increased fatty acids entry in mitochondria, as shown in increased catalytic efficiency of CPT I observed in the present study. In contrast, in our recent study, increased liver lipid content is observed in fish fed a diet containing a mixture of c9t11 and t10c12 CLA isomers (Luo et al., in preparation). The differences involved in the effect of CLA obtained in the present study and other reports may be attributable to differences in relative proportion of each isomer in a dietary CLA mixture. Metges et al. [11] demonstrate that c9t11 CLA and t10c12 CLA have distinctly different effects on lipid metabolism in adipocytes. c9t11 CLA increases lipid accumulation in white and brown adipocytes. t10c12 CLA, on the other hand, decreases lipid accumulation in both cell types by decreasing glucose incorporation into FA. Brown and McInstosh [40] find that t10c12 CLA attenuates TAG content and differentiation in primary cultures of human adipocytes, but that the c9t11 CLA isomer increases TAG accumulation.
Our study indicates that no significant differences are observed in hepatic CPT I activities among the treatments. Similarly, studies indicate that dietary CLA does not significantly influence hepatic CPT I activity in several fish species [7, 8]. In the present study, CPT I activity in muscle increases with the dietary CLA level. Similarly, CLA significantly increases CPT I activity in muscle tissue [7, 41]. Several other in-vivo experiments in mice also report that t10c12 CLA causes an increase in CPT activity, with a greater effect being exerted by the t10c12 CLA isomer [42]. On the other hand, in our study, dietary polyunsaturated fatty acid (PUFA) contents decline with t10c12 CLA inclusion at the expense of fish oil. Recently, Morash et al. [43] suggested that fish fed a high PUFA diet significantly increased CPT I mRNA expression in red muscle and liver. They also pointed out that this increased CPT I expression in red muscle and liver was not reflected in CPT I maximum velocity in fish fed the PUFA diet. Accordingly, CPT I activity in muscle and liver obtained in the present study may reflect increasing dietary t10c12 inclusion, or declining PUFA content with t10c12 CLA inclusion, or both in combination.
In the present study, constant V max, reduced K m and increased catalytic efficiency of the CPT I enzyme with carnitine as the substrate were observed in liver of fish fed increasing t10c12 CLA isomer. When using palmitoyl-CoA as the substrate, constant K m, increasing V max and catalytic efficiency were observed in livers of fish fed increasing t10c12 CLA isomer. K m can be a very useful index for evaluation of the substrate status in the tissue [44]. Patterns of enzyme V max are useful in revealing differences in fatty acid oxidation capacity [45]. Enzymatic catalytic efficiency relates total enzyme concentration to the interaction between the enzyme and the substrate. Therefore, reduced hepatic lipid content observed in the control may be due to the increased affinity of the enzyme for substrates (K m) and reduced catalytic efficiency. Reduced muscle lipid content observed in the control may be due to the increased V max and the high catalytic efficiency of CPT I for fish fed the t10c12 CLA-supplemented diet, which indicates that the fish have a high capacity for β-oxidation of long-chain fatty acids. In addition, the differences in kinetic behavior as a response to dietary CLA may be associated with the expression profile of CPT I isoforms. Studies have shown that mammalian tissues express three isoforms of CPT I, a liver, L-CPT I, and a heart/skeletal muscle, M-CPT I, that are 62 % identical in amino acid sequence [46] and a brain isoform, CPT Ic, that is 54 % identical to L- and M-CPT I [47]. The CPT I isoforms are expressed in a variety of tissues other than those for which they are named, and in variable and unpredictable amounts [46]. They also possess very different kinetic properties and sensitivity to inhibitor malonyl-CoA [15]. Accordingly, it is reasonable to speculate that different kinetic constants of CPT I in a response to dietary CLA isomer are related to the levels of CPT I isoforms expressed. However, further experiments will be needed for this aspect.
In summary, dietary t10c12 CLA isomer addition significantly improves growth performance and feed utilization, reduces muscle and liver lipid contents in juvenile S. hasta. The lipid-lowering effect of t10c12 CLA observed in S. hasta juveniles may be due to the change of kinetic constants of CPT I. Thus, t10c12 CLA can hopefully be used as a helpful dietary additive for S. hasta farming since it improves growth performance and reduced hepatic lipid contents.
Abbreviations
- BSA:
-
Bovine serum albumin
- CLA:
-
Conjugated linoleic acid
- CPT I:
-
Carnitine palmitoyltransferase I
- FA:
-
Fatty acid
- FBW:
-
Final body weight
- FCR:
-
Feed conversion rate
- FI:
-
Feed intake
- IBW:
-
Initial body weight
- MIB:
-
Mitochondrial isolation buffer
- PER:
-
Protein efficiency rate
- PUFA:
-
Polyunsaturated fatty acid
- SGR:
-
Specific growth rate
- SR:
-
Survival rate
- TAG:
-
Triacylglycerol
- WG:
-
Weight gain
References
Pariza MW, Park Y, Cook M (2001) The biologically active isomers of conjugated linoleic acid. Prog Lipid Res 40:283–298
West DB, Delany JP, Camet PM, Blohm F, Truett AA, Scimeca J (1998) Effects of conjugated linoleic acid on body fat and energy metabolism in the mouse. Am J Physiol 275:R667–R672
deDeckere DA, van Amelsvoort JM, McNeill GP, Jones P (1999) Effects of conjugated linoleic acid (CLA) isomers on lipid levels and peroxisome proliferation in the hamster. Br J Nutr 82:309–317
Twibell RG, Watkins BA, Rogers L, Brown PB (2000) Effects of dietary conjugated linoleic acids on hepatic and muscle lipids in hybrid striped bass. Lipids 35:155–161
Twibell RG, Watkins BA, Brown PB (2001) Dietary conjugated linoleic acids and lipid source alter fatty acid composition of juvenile yellow perch Perca flavescens. J Nutr 131:2322–2328
Figueiredo-Silva AC, Rema P, Bandarra NM, Nunes ML, Valente LMP (2005) Effects of dietary conjugated linoleic acid on growth, nutrient utilization, body composition, and hepatic lipogenesis in rainbow trout juveniles (Oncorhynchus mykiss). Aquaculture 248:163–172
Kennedy SR, Leaver MJ, Campbell PJ, Zheng X, Dick JR, Tocher DR (2006) Influence of dietary oil content and conjugated linoleic acid (CLA) on lipid metabolism enzyme activities and gene expression in tissues of Atlantic salmon (Salmo salar L.). Lipids 41:423–436
Leaver MJ, Tocher DR, Obach A, Jensen L, Henderson RJ, Porter AR, Krey G (2006) Effect of dietary conjugated linoleic acid (CLA) on lipid composition, metabolism and gene expression in Atlantic salmon (Salmo salar) tissues. Comp Biochem Physiol 145A:258–267
Tan XY, Luo Z, Xie P, Li XD, Liu XJ, Xi WQ (2010) Effect of dietary conjugated linoleic acid (CLA) on growth performance, body composition and hepatic intermediary metabolism in juvenile yellow catfish Pelteobagrus fulvidraco. Aquaculture 310:186–191
Luo Z, Tan XY, Liu CX, Li XD, Liu XJ, Xi WQ (2012) Effect of dietary conjugated linoleic acid levels on growth performance, muscle fatty acid profile, hepatic intermediary metabolism and antioxidant responses in genetically improved farmed tilapia strain of Nile tilapia Oreochromis niloticus. Aquacult Res 43:1392–1403
Metges C, Lehmann L, Boeuf S, Petzke KJ, Muller A, Rickert R, Franke W, Steinhart H, Nurnberg G, Klaus S (2003) cis-9,trans-11 and trans-10,cis-12 CLA affect lipid metabolism differently in primary white and brown adipocytes of Djungarian hamsters. Lipids 38:1133–1142
Zabala A, Churruca I, Macarulla MT, Rodriguez VM, Fernandez-Quintela A, Martinez JA, Portillo MP (2004) The trans-10,cis-12 isomer of conjugated linoleic acid reduces hepatic triacylglycerol content without affecting lipogenic enzymes in hamsters. Br J Nutr 92:383–389
Kim JH, Kim J, Park Y (2012) trans-10,cis-12 conjugated linoleic acid enhances endurance capacity by increasing fatty acid oxidation and reducing glycogen utilization in mice. Lipids 47:855–863
Belury MA, Mahon A, Banni S (2003) The conjugated linoleic acid (CLA) isomer, t10c12-CLA, is inversely associated with changes in body weight and serum leptin in subjects with type 2 diabetes mellitus. J Nutr 133:257S–260S
McGarry JD, Brown NF (1997) The mitochondrial carnitine palmitoyltransferase system. From concept to molecular analysis. Eur J Biochem 244:1–14
Zammit VA, Fraser F, Orstorphine CG (1997) Regulation of mitochondrial outer-membrane carnitine palmitoyltransferase (CPT I): role of membrane-topology. Adv Enzyme Regul 37:295–317
Kerner J, Hoppel C (2000) Fatty acid import into mitochondria. Biochim Biophys Acta 1486:1–17
Morash AJ, McClelland GB (2011) Regulation of carnitine palmitoyltransferase (CPT) I during fasting in rainbow trout (Oncorhynchus mykiss) promotes increased mitochondrial fatty acid oxidation. Physiol Biochem Zool 84(6):625–633
Liu XJ, Luo Z, Xiong BX, Liu X, Zhao YH, Hu GF, Lv GJ (2010) Effect of waterborne copper exposure on growth, hepatic enzymatic activities and histology in Synechogobius hasta. Ecotoxicol Environ Saf 73:1286–1291
Liu XJ, Luo Z, Li CH, Xiong BX, Zhao YH, Li XD (2011) Antioxidant responses, hepatic intermediary metabolism, histology and ultrastructure in Synechogobius hasta exposed to waterborne cadmium. Ecotoxicol Environ Saf 74:1156–1163
Zheng JL, Luo Z, Chen QL, Liu X, Liu CX, Zhao YH, Gong Y (2011) Effect of waterborne zinc exposure on metal accumulation, enzymatic activities and histology of Synechogobius hasta. Ecotoxicol Environ Saf 74:1864–1873
Luo Z, Tan XY, Li XD, Yin GJ (2012) Effect of dietary arachidonic acid levels on growth performance, hepatic fatty acid profile, intermediary metabolism and antioxidant responses for juvenile Synechogobius hasta. Aquacult Nutr 18:340–348
AOAC (Association of Official Analytical Chemists) (1995) Official methods of analysis, 16th edn. AOAC, Arlington
Suarez RK, Hochachka PW (1981) Preparation and properties of rainbow trout liver mitochondria. J Comp Physiol 143:269–273
Bremer J, Woldegiorgis G, Schalinske K, Shrago E (1985) Carnitine palmitoyltransferase activation by palmitoyl-CoA and inactivation by malonyl-CoA. Biochem Biophys Acta 833:9–16
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Hofstee B (1952) On the evaluation of the constants V m and K m in enzyme reactions. Science 116:329
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56:658–666
Choi BD, Kang SJ, Ha YL, Ackman RG (1999) Accumulation of conjugated linoleic acid (CLA) in tissues of fish fed diets containing various levels of CLA. In: Xiong YL, Ho CT, Shahidi F (eds) Quality attributes of muscle foods. Kluwer/Plenum Publishers, New York, pp 61–71
Berge GM, Ruyter B, Asgard T (2004) Conjugated linoleic acid in diets for juvenile Atlantic salmon (Salmo salar), effects on fish performance, proximate composition, fatty acid and mineral content. Aquaculture 237:365–380
Kennedy SR, Campbell PJ, Porter A, Tocher DR (2005) Influence of dietary conjugated linoleic acid (CLA) on lipid and fatty acid composition in liver and flesh of Atlantic salmon (Salmo salar). Comp Biochem Physiol 141B:168–178
Makol A, Torrecillas S, Fernandez-Vaquero A, Robaina L, Montero D, Caballero MJ, Tort L, Izquierdo M (2009) Effect of conjugated linoleic acid on dietary lipids utilization, liver morphology and selected immune parameters in sea bass juveniles (Dicentrarchus labrax). Comp Biochem Physiol 154B:179–187
Delany JP, Blohm F, Truett AA, Scimeca JA, West DB (1999) Conjugated linoleic acid rapidly reduces body fat content in mice without affecting energy intake. Am J Physiol 276:R1172–R1179
Ostrowska E, Muralitharan M, Cross RF, Bauman DE, Dunshea FR (1999) Dietary conjugated linoleic acids increase lean tissue and decrease fat deposition in growing pigs. J Nutr 129:2037–2042
Thiel-Cooper RL, Parrish FC Jr, Sparks JC, Wiegand BR, Ewan RC (2001) Conjugated linoleic acid changes swine performance and carcass composition. J Anim Sci 79:1821–1828
Terpstra AH, Beynen AC, Everts H, Kocsis S, Katan MB, Zock PL (2002) The decrease in body fat in mice fed conjugated linoleic acid is due to increases in energy expenditure and energy loss in the excreta. J Nutr 132:940–945
Diez A, Menoyo D, Perez-Benavente S, Calduch-Giner JA, Vega-Rubin de Celis S, Obach A, Favre-Krey L, Boukouvala E, Leaver MJ, Tocher DR, Perez-Sanchez J, Krey G, Bautista JM (2007) Conjugated linoleic acid affects lipid composition, metabolism and gene expression in gilthead sea bream (Sparus aurata). J Nutr 137:1363–1369
Oku H, Wongtangtintharn S, Iwasaki H, Toda T (2003) Conjugated linoleic acid (CLA) inhibits fatty acid synthetase activity in vitro. Biosci Biotechnol Biochem 67:1584–1586
Evans M, Lin X, Odle J, McIntosh M (2002) trans-10,cis-12 conjugated linoleic acid increases fatty acid oxidation in 3T3-L1 preadipocytes. J Nutr 132:450–455
Brown JM, McInstosh MK (2003) Conjugated linoleic acid in humans: regulation of adiposity and insulin sensitivity. J Nutr 133:3041–3046
Bouthegourd JC, Even PC, Gripois D, Toffon B, Blouquit MF, Roseau S, Lutton C, Tome D, Martin JC (2002) A CLA mixture prevents body triglyceride accumulation without affecting energy expenditure in Syrian hamsters. J Nutr 132:2682–2689
Degrace P, Demizieux L, Gresti J, Chardigny JM, Sébédio JL, Coulet P (2004) Hepatic steatosis is not due to impaired fatty acid oxidation capacities in C57BL/6j mice fed the conjugated trans-10,cis-12-isomer of linoleic acid. J Nutr 134:861–867
Morash AJ, Bureau DP, McClelland GB (2009) Effects of dietary fatty acid composition on the regulation of carnitine palmitoyltransferase (CPT) I in rainbow trout (Oncorhynchus mykiss). Comp Biochem Physiol 152B:85–93
Lin X, Odle J (2003) Changes in kinetics of carnitine palmitoyltransferase in liver and skeletal muscle of dogs (Canis familiaris) throughout growth and development. J Nutr 133:1113–1119
Morash AJ, Kajimura M, McClelland GB (2008) Intertissue regulation of carnitine palmitoyltransferase I (CPT I): mitochondrial membrane properties and gene expression in rainbow trout (Oncorhynchus mykiss). Biochim Biophys Acta 1778:1382–1389
Weis BC, Esser V, Foster DW, McGarry JD (1994) Rat heart expresses two forms of mitochondrial carnitine palmitoyltransferase I. J Biol Chem 269:18712–18715
Price N, van der Leij F, Jackson V, Corstorphine C, Thomson R, Sorensen A, Zammit V (2002) A novel brain-expressed protein related to carnitine palmitoyltransferase I. Genomics 80:433–442
Acknowledgments
This work was funded by a program for New Century Excellent Talents in University, Ministry of Education, China (Grant No. NCET-08-0782), by the Special Fund for Central University, Ministry of Education, China (Grant No. 52204-10078), by the National Natural Science Foundation of China (Grant Nos. 30800850, 31072226). We wish to thank the staff of the Panjin Guanghe Fisheries Co., Ltd., for providing the experimental base and for their logistic support during this study.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Tan, XY., Luo, Z., Zeng, Q. et al. trans-10,cis-12 Conjugated Linoleic Acid Improved Growth Performance, Reduced Lipid Deposition and Influenced CPT I Kinetic Constants of Juvenile Synechogobius hasta . Lipids 48, 505–512 (2013). https://doi.org/10.1007/s11745-013-3759-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-013-3759-5