Abstract
Betula platyphylla and Betula costata are important species in mixed broadleaved-Korean pine (Pinus koraiensis) forests. However, the specific ways in which their growth is affected by warm temperatures and drought remain unclear. To address this issue, 60 and 62 tree-ring cores of B. platyphylla and B. costata were collected in Yichun, China. Using dendrochronological methods, the response and adaptation of these species to climate change were examined. A “hysteresis effect” was found in the rings of both species, linked to May–September moisture conditions of the previous year. Radial growth of B. costata was positively correlated with the standardized precipitation-evapotranspiration index (SPEI), the precipitation from September to October of the previous year, and the relative humidity in October of the previous year. Growth of B. costata is primarily restricted by moisture conditions from September to October. In contrast, B. platyphylla growth is mainly limited by minimum temperatures in May–June of both the previous and current years. After droughts, B. platyphylla had a faster recovery rate compared to B. costata. In the context of rising temperatures since 1980, the correlation between B. platyphylla growth and monthly SPEI became positive and strengthened over time, while the growth of B. costata showed no conspicuous change. Our findings suggest that the growth of B. platyphylla is already affected by warming temperatures, whereas B. costata may become limited if warming continues or intensifies. Climate change could disrupt the succession of these species, possibly accelerating the succession of pioneer species. The results of this research are of great significance for understanding how the growth changes of birch species under warming and drying conditions, and contribute to understanding the structural adaptation of mixed broadleaved-Korean pine (Pinus koraiensis) forests under climate change.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With the intensification of industrialization and rapid population growth, human activities have an unprecedented impact on Earth’s climate, with no parallels in the past two millennia (IPCC 2021). This has translated into extreme changes in climate, such as heightened temperatures and severe drought, with consequences for tree growth, development, and survival (Nitschke et al. 2017; Peng et al. 2023).
In the Northern Hemisphere, warming is more evident at higher latitudes than at middle and lower latitudes. Consequently, certain regions have experienced pronounced warming and drying (Balting et al. 2021). Over the past 100 years, northeastern China has undergone a marked warming trend coupled with reduced precipitation, resulting in increased aridity (Chorography Compilation Committee of Heilongjiang Province 1997). This warmer and more arid climate has a profound influence on the ecosystems of northeastern China (Gao et al. 2020; Ju et al. 2007).
Betula platyphylla Sukaczev. and Betula costata Trantv. belong to the early evolutionary and the late evolutionary species, respectively, of the genus Betula (Jiang 1990). B. platyphylla is dominant in the natural secondary forests formed following the harvesting of mixed broadleaved-Korean pine (Pinus koraiensis Siebold & Zucc.) forests in the Xiaoxing’an Mountains. It is a typical pioneer species of temperate secondary forests, characterized by its adaptability and its resource utilization value. In contrast, B. costata is an important associate species within both primary mixed broadleaved-Korean pine forests and secondary forests in the Xiaoxing’an Mountains. Its wood is hard, and the species has significant economic value (Liu 2004). Both B. platyphylla and B. costata play a vital role in supporting ecosystem structure as well as facilitating forest succession (Hussain et al. 2021). It is therefore important to develop a more comprehensive understanding of the dynamics driving the responses of these species to climate change. Although both belong to the same genus, they are distinct species; understanding their climate change responses is crucial for understanding interspecies variations.
Inconsistencies exist in the factors limiting the growth of B. platyphylla across various regions. In the Changbai Mountain area, characterized by relatively abundant precipitation, temperature has a significantly more pronounced influence on radial growth than precipitation (Liu 2018). Temperature also has a significant influence on growth in the Zhangguangcai Mountains. On the relatively cold Kamchatka Peninsula of Russia, growth is mainly affected by summer (May to August) temperatures (Deck et al. 2017). Some surveys have also found that temperature is a major factor in B. platyphylla growth at high altitudes (Lyu et al. 2020; Li et al. 2022). However, in the relatively arid mountain steppe-coniferous forests of northern Mongolia, water availability is the critical determinant of growth (Gradel et al. 2017a, 2017b). Similarly, there are no correlations between B. platyphylla tree-ring width and temperatures in any month at the highest elevations of Mount Norikura in Japan (Takahashi et al. 2003).
While some research has explored the connection between the growth of B. platyphylla and climate (Watanabe et al. 2016; Gradel et al. 2017b; Khan et al. 2019; Li et al. 2022), few studies have addressed climate change responses of B. costata. Luo et al. (2012) analyzed the changes in diameter at breast height (DBH), tree height, and volume of B. costata in Jilin province but did not consider the influence of climate factors. Yang et al. (2018) studied the correlation between DBH, height and crown width of the species in mixed coniferous broad-leaved forests in the Xiaoxing’an Mountains but did not examine relationships between growth and climate. Studies on wood anatomy of B. costata trunks and branches found that xylem anatomical characteristics varied with location and age (Zhao et al. 2019).
While few studies have explored species of the same genus, investigations comparing the impacts of climate change on growth across distinct birch species have been rare. It is important to note that different birch species adapt to different habitats. For example, Betula pendula Roth. thrives in well-drained soils, whereas Betula pubescens Ehrh. is highly tolerant of moist soils and poorly drained heaths (Beck et al. 2016). The core distribution area and maximum climate niche range differ among various birch species. B. platyphylla falls within the category of species adapted to low temperatures, low humidity, and has a broad ecological range, while B. costata is characterized as a low temperature, low humidity species with a narrower one (Du et al. 2019). Comparative studies of birch microanatomy have also been carried out (Nielsen et al. 2017; Yuan et al. 2021). In western Greenland, variations in vessel lumen area are correlated with spring and summer temperatures, while vessel grouping is primarily influenced by winter temperatures (Nielsen et al. 2017). Although B. platyphylla, B. costata, and Betula dahurica Pallas. are diffuse-porous species, B. costata has higher mechanical strength (Gao et al. 1996). B. platyphylla responds to climate change by producing more and smaller vessels. Conversely, B. costata adapts by enhancing water transport efficiency through large vessels. B. dahurica falls between these two extremes, with a moderate number and size of vessels (Bai et al. 2023).
Although the growth response of B. platyphylla to climate change has been widely analyzed, little has been carried out to understand other species within the Betula genus. The impact of varying climatic conditions on the growth of different birch species warrants further exploration. This paper builds upon prior research and addresses the following questions: (1) Do different birch species respond differently to climatic factors? (2) How do the growth patterns of B. platyphylla and B. costata reflect extreme drought conditions?
Materials and methods
Study area
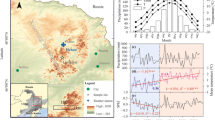
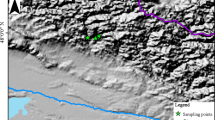
The research area is located in Wuying National Forest Park (47.9° N–48.3° N, 129.1° E–129.5° E) in Yichun, northeastern China. It has a typical temperate, continental monsoon climate with warm, humid summers and cold, dry winters and strong seasonal distinctions. The annual mean temperature is − 0.5 °C, with the mean temperatures of the coldest (January) and hottest (July) months − 23.5 and 20.2 °C, respectively. The annual total precipitation is 780 mm, falling from June to August and accounting for about 83% of the annual amount. Early frosts begin in mid-September, and late frosts last until late May; there are only 100–120 days frost-free. Elevation throughout the study area ranges from 285 to 688 m a.sl. The area is part of the Tangwang River system. Soils are dark brown forest soil.
The zonal vegetation is a temperate coniferous and broad-leaved mixed forest dominated by P. koraiensis with a variety of temperate broad-leaved species, including B. platyphylla, B. costata, Tilia amurensis Rupr., Acer tegmentosum Maxim., and Quercus mongolica Fisch. ex Ledeb. Cold-temperate coniferous species include Picea koraiensis Nakai. and Abies nephrolepis (Trautv. ex Maxim.) Maxim.; common shrub species include Euonymus alatus (Thunb.) Sieb., Corylus mandshurica Maxim. & Rupr., and Philadelphus schrenkii Rupr. Actinidia kolomikta (Maxim. & Rupr.) Maxim. is the dominant liana.
Tree-ring sampling and chronology development
Sampling was carried out in July 2015 and 2016. Samples were taken from healthy individuals without obvious signs of pest infestation or fire disturbance. To have the sampling environment as consistent as possible, samples were taken from closed forest areas. The soil type and soil nutrient contents of the sampling points are basically similar. Following traditional dendrochronological methods, a minimum of 25 healthy trees of each species were selected for sampling (Table 2). Two or three cores per tree at breast height (~ 1.3 m) were taken with a 5.15 mm increment borer (Haglöf, Dalarna, Sweden). A total of 60 B. platyphylla cores and 62 B. costata cores were obtained. The samples were placed in numbered plastic tubes for transport to the laboratory where they were processed. Following the method of Stokes and Smiley (1968), samples were fixed on wooden mounts, air-dried, and polished until the tree-ring boundary was clearly visible under a microscope. The skeleton plot method was used to visually assign each ring to an accurate calendar year. The Velmex tree-ring measurement system (Velmex Inc., Bloomfield, NY, USA) was used to measure tree-ring widths (0.001 mm accuracy). The COFECHA program (Holmes 1983) examined the cross-dating and measurement results. Cores that could not be cross-dated or that correlated poorly with the main sequence were eliminated from the sample set. This process ensured that an accurate calendar year was assigned to each growth ring and eliminated errors in dating and measurement. A smoothing spline 67% of the series length was used to detrend the individual tree-ring series. The process of chronology establishment was completed using the R package dplR (Bunn 2008).
Meteorological data and statistical analyses
Climate data was collected from the Yichun meteorological station (47.73° N, 128.92° E). Monthly mean temperature (T), monthly minimum temperature (Tmin), monthly maximum temperature (Tmax), monthly total precipitation (P), relative humidity (RH), and standardized precipitation evapotranspiration index (1-month scale, SPEI-1) were selected for correlation analysis with the tree-ring indices. Relative humidity is the ratio of air humidity to saturated humidity at the same temperature. SPEI has the characteristics of multiple time scales of 1–48 months. SPEI ≤ − 2.0 is considered extreme drought; when − 2.0 < SPEI ≤ − 1.5, severe drought conditions exist. The SPEI was calculated using the R package SPEI (Vicente-Serrano et al. 2010).
To include the impact of climate on radial growth in the preceding year, we used 17 months of data from May of the preceding year to September of the current year. Correlation analysis was conducted using the standard chronology. Recognizing that climate exerts both persistent and cumulative effects on tree growth, this study simultaneously investigated the relationship between seasonal climatic factors and radial growth for both species. To fully encompass the physiological and ecological significance of climate impacts, the seasons were divided into: winter (December of the previous year to February of the current year), spring (March to May of the current year), summer (June to August of the current year), and autumn (September to November of the current year).
The impact of drought events on the growth of B. platyphylla and B. costata was examined using the standardized precipitation evapotranspiration index on 3-month (SPEI-3) and 12-month (SPEI-12) scales (Fig. 1). SPEI-3 captures seasonal (short-term) variations in drought, whereas SPEI-12 reflects annual (long-term) changes. To reveal the specific conditions of drought, SPEI-3 values for February, May, August, and November were selected to represent seasonal drought patterns. The annual drought condition was characterized by the SPEI-12 value in December (Schwalm et al. 2017).
The R package dplR was used to conduct superposed epoch analysis (SEA) on the selected drought years (Table 1) (Bunn 2008). The goal was to clarify distinctions in the mechanisms of growth restraint and release between B. platyphylla and B. costata following seasonal and annual drought events. The SEA approach reduces the interference of background factors and directs attention toward the specific influences of interest. This technique is commonly used to analyze the impact of extreme weather events on the radial growth of trees. In this context, the year of the drought event is used as the critical time point, while alterations in tree-ring width are used as indicators of response changes.
Over the past 62 years (1956–2017), there has been a significant increase (0.36 °C/10 years) in temperatures at Wuying Forest Farm in Yichun, China. At the same time, the region’s climate has exhibited a drying trend (Fig. 2). To explore the impacts of these climate trends on radial growth, a 20-year moving correlation analysis was used. The goal was to establish links between tree growth patterns and climate warming as indicated by the SPEI.
For both species, the correlation and response coefficients were calculated at 20-year intervals starting from 1963 and 1964, respectively. The analysis subsequently progressed backward one year at a time. Pearson correlation analysis between tree-ring chronologies and climate variables was performed using the R package Treeclim (Zang and Biondi 2015). In addition, an evaluation of the correlation between seasonal climate variables and the radial growth of B. platyphylla and B. costata was carried out using the Seascorr function within the Treeclim R package (Zang and Biondi 2015). Visuals were generated using the R package ggplot2 (Villanueva and Chen 2019) and Microsoft Excel 2019.
Results
Tree growth and chronology statistics
The longest effective time spans captured for B. platyphylla and B. costata were 55 and 53 years, respectively (Table 2). B. platyphylla was more sensitive to climate variations, as shown by its higher mean sensitivity and mean correlation compared to B. costata. The robustness of the chronologies is underscored by the high signal-to-noise ratios and expressed population signals observed for both species. These indicators suggest that variations in their ring widths accurately reflect changes in the surrounding environmental conditions. Overall, the dendrochronological analyses of both provide valuable information about the relationship between growth patterns and climatic influences.
Relationships between radial growth and climatic factors
Figure 3 illustrates both shared patterns and distinctive characteristics in the relationship between the radial growth of both species and various climate factors. The radial growth of B. costata was positively correlated with precipitation and SPEI from September to October of the preceding year, as well as with relative humidity in October of the previous year (Fig. 3b, f). However, its growth was negatively correlated with mean temperature and minimum temperature in September of the current year (Fig. 3a, c). Overall, the growth of B. costata was primarily constrained by moisture conditions from September to October. Similarly, the radial growth of B. platyphylla was positively correlated with precipitation and SPEI in both May and September of the previous year, as well as with SPEI in February of the current year (Fig. 3f). For both species, the moisture conditions (monthly precipitation, relative humidity, and SPEI) from May to November of the preceding year had a more significant influence on growth compared to moisture conditions of the current year (Fig. 3).
Correlation coefficients between the tree-ring indices of B. platyphylla (1963–2017) and B. costata (1964–2016) and major monthly climate factors; T: monthly mean temperature; Tmin: monthly minimum temperature; Tmax: monthly maximum temperature; P: monthly total precipitation; RH: monthly relative humidity; SPEI-1: monthly standardized precipitation evapotranspiration index. The gray line is an auxiliary line that can clearly indicate where the correlation coefficients of 0.2 and −0.2 are. *, P < 0.05
From July of the preceding year to August of the current year, the radial growth of B. platyphylla was more consistent in response to temperature variables (mean temperature, minimum temperature, and maximum temperature) as compared to moisture variables (Fig. 3). The contributions of mean temperature and minimum temperature from May to September of the current year to radial growth were greater than that of climatic factors during the preceding year. Both monthly mean temperature and SPEI during the growing season were positively correlated with B. platyphylla growth, with SPEI showing a stronger influence. Moreover, radial growth of B. platyphylla was positively correlated with the minimum temperature in June of the preceding year and May to June of the current year. In summary, B. platyphylla growth is mainly limited by minimum temperatures during the May to June period of both the previous and current years (Fig. 3).
The effect of seasonal temperature on radial growth was less than the effect of monthly temperature (Table 3). Summer mean temperature and precipitation were positively correlated with B. platyphylla growth, while the minimum temperature was significantly positively correlated with B. platyphylla growth.
Effects of extreme drought on radial growth
The radial growth responses of B. platyphylla and B. costata to extreme drought were generally consistent, but B. platyphylla exhibited a greater sensitivity (Fig. 4). This aligns with the conclusion that the growth of B. platyphylla is more sensitive to seasonal climate shifts than B. costata. The occurrence of extreme drought resulted in a reduction in radial growth of both species (Fig. 4a). Following a drought episode, radial growth of both exhibited a gradual recovery. By the fifth-year post-drought, the radial growth of both species had returned to pre-drought levels (Fig. 4a, d, e). Compared to other seasonal droughts, extreme summer droughts had a greater impact on the growth of the two species (Fig. 4d).
Superposed epoch analysis of the radial growth of B. platyphylla and B. costata during drought event years; a: year of extreme drought based on the SPEI-12 filter; b: extreme drought events occurring during the preceding winter based on the SPEI-3 filter; c: extreme drought events occurring in spring based on the SPEI-3 filter; d: extreme drought events occurring in summer based on the SPEI-3 filter; e: extreme drought events occurring in autumn based on the SPEI-3 filter. Dotted lines represent 95% confidence intervals
Temporal stability of the relationship between radial growth and climate
The relationship between B. platyphylla radial growth and monthly SPEI changed in 1980 (Fig. 5). Before 1980, there was a negative correlation between growth and SPEI. After 1980, B. platyphylla growth became significantly positively correlated with SPEI from September of the previous year to July of the current year. This correlation increased over time (Fig. 5).
In contrast to B. platyphylla, the relationship between the growth B. costata and the SPEI from May of the previous year to May of the current year did not change significantly in 1980 (Fig. 6). The significant negative correlation between growth and SPEI existed before 1972 and then gradually weakened. After 1986, the relationship became significantly positive (Fig. 6).
As temperatures in northeastern China rise, drought increasingly becomes a limiting factor for the radial growth of both species (Figs. 5 and 6). B. platyphylla is more sensitive to drought than B. costata. Growth is more strongly impacted by extreme droughts before the growing season than B. costata. However, drought during the growing season had a greater impact on B. costata growth (Figs. 5 and 6).
Discussion
Comparison of climate factors limiting radial growth
Moisture is a primary limiting factor for the radial growth of both B. platyphylla and B. costata. B. platyphylla, in particular, is affected by moisture conditions before the growing season (May to November), indicating moisture conditions have a hysteresis effect on ring width growth, leading to the influence being significant in the following year (Yu et al. 2016; Jiao et al. 2022; Li et al. 2022). This agrees with a study on B. platyphylla from the Hanshan region of Inner Mongolia, China (Jia 2019), which found that moisture conditions of the previous year affected the growth of the birch in the current year. The Xiaoxing’an Mountains have a typical temperate, continental monsoon climate with four distinct seasons, well-defined variations in temperature and sunlight throughout the year. Due to these changes, which particularly affect growth at high latitudes, periods of active growth alternate with periods of cold-resistant dormancy on an annual cycle (Weiser 1970). This cycle is the basis of tree survival and is regulated by the interaction of genetic and environmental factors (Sutinen et al. 2009). Moisture conditions during the preceding year affect the amount of nutrients synthesized for growth in the following year, determines their greening state at the start of the new growing season, and regulates xylem development (Marqués et al. 2022).
Low May and June temperatures also have a significant influence on the growth of B. platyphylla. During these months of early growth, trees need more water (Liang et al. 2001). In the Yichun region, winter is cold and long, and the soil remains frozen for a long time (Liu et al. 2019). The increase in minimum temperatures in May causes snow to melt and soil temperatures to rise, increasing soil moisture and promoting root activity and aboveground growth (Toromani et al. 2011). Thus, radial growth benefits from the May increase in minimum temperatures. Temperature affects photosynthesis which in turn impacts transpiration. The increase in minimum temperatures in June enhances photosynthesis and transpiration (Gao et al. 2022). A key characteristic of the climate in Wuying is that temperature and precipitation increase over the same period, concentrated from June to August (Qi and He 2021). Abundant rainfall replenishes the water lost by transpiration due to photosynthesis, allowing radial growth to increase. This result is in line with the results of studies of other broad-leaved tree species in the Zhangguangcai Mountains by Su and Wang (2017).
B. platyphylla and B. costata show differences in their sensitivity to temperatures in September, an important factor affecting B. costata growth in particular. Previous studies have found that rising air temperatures can either promote (Vitasse et al. 2019a, 2019b) or inhibit (Greenwood et al. 2017; Brodribb et al. 2020) radial growth depending on the season and the environmental conditions. September is the end of the growing season for B. costata (Han et al. 2019). At this time, precipitation decreases, and evaporation is made worse by rising temperatures. When an internal water deficit is reached, the stomata of B. costata close, causing a decrease in photosynthesis. This results in a short period of “physiological drought” that restricts growth (Lavrič et al. 2017). The growth of B. costata is more strongly affected by September temperatures because it is a mid-succession species, whereas B. platyphylla is an early-succession species and therefore more accustomed to temperature changes. Genetic-controlled variations in the traits of the two birch species affect their physiological characteristics, resulting in a significantly different radial growth-climate relationships related to the evolutionary adaptation of species to their environments (White et al. 2007). In general, early succession stage habitats are characterized by openness and abundant light. Due to the buffering effect of vegetation, middle or late succession stage habitats are generally closed and stable, with less drastic changes in environmental conditions (Bazzaz 1996). Early succession species are therefore better adapted to fluctuations in light, temperature, and precipitation. In contrast, species in late succession stages are more susceptible to environmental factors such as drought (Li and Ma 2002). It is therefore unsurprising that B. costata growth is more sensitive to moisture conditions from September–October, whereas B. platyphylla growth is limited by May to June minimum temperatures.
Effects of extreme drought on radial growth
The effects of extreme drought on the radial growth of both species varied from season to season. Extreme drought in summer decreased B. costata growth and significantly reduced the ring width of B. platyphylla. Summer is the primary season for growth and development; insufficient water can be a major factor in delaying or even halting wood formation (Pérez-de-Lis et al. 2016). Therefore, the influence of extreme drought in summer on tree growth are of particular importance.
In this study, ring width did not decrease significantly in the four years following summer, autumn, and annual extreme drought events. However, the recovery process was slow and fluctuated slightly until growth release occurred in the 5th year following a drought. A decline in the radial growth of trees experiencing drought conditions leads to the accumulation of non-structural carbohydrates (Gricar et al. 2018), and both gymnosperms and angiosperms have a certain resilience after drought (DeSoto et al. 2020). Our results agree with Ovenden et al. (2021) findings that the growth recovery of Pinus sylvestris L. was significant 4–5 years after a drought. Li et al. (2021) reported that Pinus massoniana Lamb. remained in a recovery phase for two years following an extreme drought, then experienced rapid growth in the third year due to relatively warm and humid conditions in the study area (yearly mean temperature was 20.5 °C and annual rainfall 1600 mm). In this study area, B. platyphylla and B. costata are pioneer species of community succession and the principal species for afforestation of barren hills and bare land (Hussain et al. 2021) due to their drought tolerance and environmental adaptability. This study area has a temperate, continental monsoon climate, with much lower annual precipitation and mean temperatures than the subtropical marine monsoon climate described by Li et al. (2021). We therefore concluded that B. platyphylla and B. costata in the Xiaoxing’an Mountains remain in the growth recovery phase for about four years following an extreme drought, then recover rapidly in the 5th year.
Our analysis shows that B. platyphylla growth recovered more swiftly from the effects of an extreme drought than B. costata. Previous studies have found that many early succession species have higher photosynthetic rates when leaf water potential is reduced (Li and Ma 2002). As a result, early succession species typically recover faster once water stress is relieved (Geis et al. 1971).
Before 1980, the SPEI from September to April showed an insignificant negative correlation with B. platyphylla growth. After 1980, the correlation became significant and positive, and increased over time. B. costata is significantly less drought-limited than B. platyphylla. Although its growth is sensitive to SPEI from July to September, the correlation did not change significantly in or around 1980. Paleoclimate reconstruction studies point to unprecedented warming in the twentieth century. A body of growing data indicates that temperature has increased significantly in northeastern China since 1980 (Zhu et al. 2007). Rising temperatures in Wuying area of Yichun, China from 1956 to 2017 led to enhanced soil water evaporation and faster vegetation transpiration. Precipitation, meanwhile, did not change significantly and therefore could not supplement the evaporated soil water. As a result, soil water content decreased, becoming an important limiting condition for the radial growth of B. platyphylla and B. costata. In general, early succession species often reduce leaf temperature through higher transpiration (Li and Ma 2002). However, intense transpiration also accelerates water loss and exacerbates drought stress in early succession species (Lavrič et al. 2017). This is consistent with our finding that B. platyphylla growth is more significantly restricted by drought than B. costata.
Conclusions
We compared the growth responses of different birch species to climate change in Xiaoxing’an Mountains, China. Radial growth of B. platyphylla and B. costata was affected by moisture conditions of the previous year and temperatures of the current year. Minimum temperatures from May to June and moisture from September to October are major factors limiting the growth of B. platyphylla and B. costata, respectively. With rapid warming after 1980, the correlations between the growth of these two species and climate factors have changed, with water availability becoming an increasingly important limiting factor. At present, the growth of B. platyphylla is more restricted by water conditions than B. costata. B. platyphylla growth is already affected by the warming and drying trend of the last decade; B. costata will also be limited in the future if warming continues. This suggests that climate change may be interfering with the succession processes of B. platyphylla and B. costata, accelerating the succession of pioneer species. Forest managers need to respond to these changes by adjusting the composition and proportion of tree species over time and by promoting forest succession process. Further physiological and ecological experiments are needed to explore the mechanisms by which different birch species respond to changes in climate change. These studies are crucial for developing effective management measures and promoting healthy forest development.
References
Bai YX, Yuan DY, Wang XC, Liu YL, Wang XC (2023) Comparison of xylem vessels characteristics of the trunks of three species of Betula in northeast China and their relationship with climate. Chin J Plant Ecol 47(8):1144–1158. https://doi.org/10.17521/cjpe.2022.0300. (in Chinese)
Balting DF, AghaKouchak A, Lohmann G, Lonita M (2021) Northern hemisphere drought risk in a warming climate. NPJ Clim Atmos Sci 4:61. https://doi.org/10.1038/s41612-021-00218-2
Bazzaz FA (1996) Plants in changing environments: linking physiological, population, and community ecology. Cambridge University Press, Cambridge
Beck PSA, Caudullo G, Rigo Dd, Tinner W (2016) Betula pendula, Betula pubescens and other birches in Europe: distribution, habitat, usage and threats. European Atlas of Forest Tree Species. Publication Office of the European Union, pp 70−73. https://www.researchgate.net/publication/299405431_Betula_pendula_Betula_pubescens_and_other_birches_in_Europe_distribution_habitat_usage_and_threats
Brodribb TJ, Powers J, Cochard H, Choat B (2020) Hanging by a thread? Forests and drought. Science 368:261–266. https://doi.org/10.1126/science.aat7631
Bunn AG (2008) A dendrochronology program library in R (dplR). Dendrochronologia 26(2):115–124. https://doi.org/10.1016/j.dendro.2008.01.002
Chorography Compilation Committee of Heilongjiang Province (1997) Chorography of Heilongjiang Province. Heilongjiang People Publishing House, Harbin
Deck C, Wiles G, Frederick S, Matsovsky V, Kuderina T, D’Arrigo R, Solomina O, Wiesenberg N (2017) Climate response of larch and birch forests across an elevational transect and hemisphere-wide comparisons, Kamchatka Peninsula, Russian Far East. Forests 8(9):315. https://doi.org/10.3390/f8090315
DeSoto L, Cailleret M, Sterck F, Jansen S, Kramer K, Robert EMR, Aakala T, Amoroso MM, Bigler C, Camarero JJ, Čufar K, Gea-Izquierdo G, Gillner S, Haavik LJ, Hereş A-M, Kane JM, Kharuk VI, Kitzberger T, Klein T, Levanič T, Linares JC, Mäkinen H, Oberhuber W, Papadopoulos A, Rohner B, Sangüesa-Barreda G, Stojanovic DB, Suárez ML, Villalba R, Martínez-Vilalta J (2020) Low growth resilience to drought is related to future mortality risk in trees. Nat Commun 11(1):545. https://doi.org/10.1038/s41467-020-14300-5
Du X, Dong X, Zheng Y, Dong L, Chen BW (2019) Several important Betula Linn. biomes climatic ecological niches and potential distribution areas. J Arid Land Resour Environ 33(8):179–185. https://doi.org/10.13448/j.cnki.jalre
Gao ZY, Wang XM, Guo AL, Xu CM, Wu YT (1996) Study on the three kinds of birch from Big Xian Mountain. J Neimenggu for Coll 18(4):32–36 (in Chinese)
Gao WQ, Lei XD, Fu LY (2020) Impacts of climate change on the potential forest productivity based on a climate-driven biophysical model in northeastern China. J for Res 31:2273–2286. https://doi.org/10.1007/s11676-019-00999-6
Gao S, Liang EY, Liu RS, Babst F, Camarero JJ, Fu YH, Piao SL, Rossi S, Shen MG, Wang T, Peñuelas J (2022) An earlier start of the thermal growing season enhances tree growth in cold humid areas but not in dry areas. Nat Ecol Evol 6:397–404. https://doi.org/10.1038/s41559-022-01668-4
Geis J, Tortorelli RL, Boggess WR (1971) Carbon dioxide assimilation of hardwood seedlings in relation to community dynamics in central illinois. Oecologia 7(3):276–289. https://doi.org/10.1007/BF00345218
Gradel A, Ganbaatar B, Nadaldorj O, Dovdondemberel B, Kusbach A (2017a) Climate-growth relationships and pointer year analysis of a Siberian larch (Larix sibirica Ledeb.) chronology from the Mongolian mountain forest steppe compared to white birch (Betula platyphylla Sukaczev). For Ecosyst 4(1):4–22. https://doi.org/10.1186/s40663-017-0110-2
Gradel A, Haensch C, Ganbaatar B, Dovdondemberel B, Nadaldorj O, Günther B (2017b) Response of white birch (Betula platyphylla Sukaczev) to temperature and precipitation in the mountain forest steppe and taiga of northern Mongolia. Dendrochronologia 41(5):24–33. https://doi.org/10.1016/j.dendro.2016.03.005
Greenwood S, Ruiz-Benito P, Martínez-Vilalta J, Lloret F, Kitzberger T, Allen CD, Fensham R, Laughlin DC, Kattge J, Bönisch G, Kraft NJB, Jump AS (2017) Tree mortality across biomes is promoted by drought intensity, lower wood density and higher specific leaf area. Ecol Lett 20(4):539–553. https://doi.org/10.1111/ele.12748
Gricar J, Zavadlav S, Jyske T, Lavric M, Laakso T, Hafner P, Eler K, Vodnik D (2018) Effect of soil water availability on intra-annual xylem and phloem formation and non-structural carbohydrate pools in stem of Quercus pubescens. Tree Physiol 39(2):222–233. https://doi.org/10.1093/treephys/tpy101
Han JS, Zhao YH, Zhu LJ, Zhang YD, Li ZS, Wang XC (2019) Comparing the responses of radial growth between Quercus mongolica and Phellodendron amurense to climate change in Xiaoxing’ an Mountains, China. Chin J Appl Ecol 30(7):2218–2230. https://doi.org/10.13287/j.1001-9332.201907.012. (in Chinese)
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-ring Bull 43:69–78. http://hdl.handle.net/10150/261223
Hussain A, Shahzad MK, Burkhart HE, Jiang L (2021) Stem taper functions for white birch (Betula platyphylla) and costata birch (Betula costata) in the Xiaoxing’an Mountains, northeast China. Forestry 94(5):714–733. https://doi.org/10.1093/forestry/cpab014
IPCC (2021) Climate Change 2021: The physical science basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge
Jia L (2019) Difference analysis of climate change of radix growth in Betula Platyphylla in Hanshan secondary forest area. College of Forestry. Master's Thesis. Inner Mongolia Agricultural University. (in Chinese)
Jiang JM (1990) The study of the geographical distribution of the Betula in China. For Res 3(1):55–62 (in Chinese)
Jiao L, Wang SJ, Chen K, Liu XP (2022) Dynamic response to climate change in the radial growth of Picea schrenkiana in western Tien Shan, China. J for Res 33:147–157. https://doi.org/10.1007/s11676-021-01336-6
Ju H, Xiong W, Xu YL, Lin ED (2007) Climate change and its impacts in northeast China. Chin Agric Sci Bull 23(4):345–349. https://doi.org/10.11924/j.issn.1000-6850.0704345. (in Chinese)
Khan D, Muneer MA, Nisa Z-U, Shah S, Amir M, Saeed S, Uddin S, Munir MZ, Lushuang G, Huang H (2019) Effect of climatic factors on stem biomass and carbon stock of Larix gmelinii and Betula platyphylla in Daxing’anling Mountain of Inner Mongolia, China. Adv Meteorol 2019:5692574. https://doi.org/10.1155/2019/5692574
Lavrič M, Eler K, Ferlan M, Vodnik D, Gričar J (2017) Chronological sequence of leaf phenology, xylem and phloem formation and sap flow of Quercus pubescens from abandoned Karst grasslands. Front Plant Sci 8:314. https://doi.org/10.3389/fpls.2017.00314
Li QK, Ma KP (2002) Advances in plant succession ecophysiology. Chin J Plant Ecol 26(S1):9–19 (in Chinese)
Li YJ, Fang KY, Bai MW, Cao XG, Dong ZP, Tang WR, Mei ZP (2021) Ecological resilience of ancient Pinus massoniana trees to climate change and insect infestation in southeastern Fujian China. Chin J Appl Ecol 32(10):3539–3547. https://doi.org/10.13287/j.1001-9332.202110.010. (in Chinese)
Li X, Bounthong P, Kang WH, Ji XD, Zhang HJ, Xue ZG, Zhang ZQ (2022) Responses of radial growth to climate change over the past decades in secondary Betula platyphylla forests in the mountains of northwest Hebei China. Chin J Plant Ecol 46:919–931. https://doi.org/10.17521/cjpe.2021.0253. (in Chinese)
Liang EY, Shao XM, Hu YX, Lin JX (2001) Dendroclimatic evaluation of climate-growth relationships of Meyer spruce (Picea meyeri) on a sandy substrate in semi-arid grassland, north China. Trees 15:230–235. https://doi.org/10.1007/s004680100097
Liu YX (2004) Manual on the nature and use of wood in Northeast China. Chemical Industry Press, Beijng
Liu Y (2018) Effects of environmental change on radial growth of dominant tree species in northeast China. Heilongjiang University. (in Chinese)
Liu CS, Liu HN, Zhang HL, Xu YQ, Wei L (2019) Seasonal frozen soil in Heilongjiang: climatic characteristics analysis. Chin Agric Sci Bull 35(16):126–132. https://doi.org/10.11924/j.issn.1000-6850.casb18120063. (in Chinese)
Luo M, Zheng XX, Wang F (2012) Analysis of growth process of Betula costata in Jingouling forest farm. J Centl South Univ for Technol 32(07):45–48. https://doi.org/10.14067/j.cnki.1673-923x.2012.07.025. (in Chinese)
Lyu ZY, Yun RX, Wu T, Ma YJ, Chen ZJ, Jin YT, Li JX (2020) Altitudinal differentiation in the radial growth of Betula platyphylla and its response to climate in cold temperate forest: a case of Oakley Mountain, Northeast China. Chin J Appl Ecol 31(6):1889–1897. https://doi.org/10.13287/j.1001-9332.202006.011. (in Chinese)
Marqués L, Peltier DMP, Camarero JJ, Zavala MA, Madrigal-González J, Sangüesa-Barreda G, Ogle K (2022) Disentangling the legacies of climate and management on tree growth. Ecosystem 25:215–235. https://doi.org/10.1007/s10021-021-00650-8
Nielsen SS, Arx G, Damgaard CF, Abermann J, Buchwal A, Büntgen U, Treier UA, Barfod AS, Normand S (2017) Xylem anatomical trait variability provides insight on the climate-growth relationship of Betula nana in Western Greenland. Arct Antarct Alp Res 49(3):359–371. https://doi.org/10.1657/AAAR0016-041
Nitschke CR, Nichols S, Allen K, Dobbs C, Livesley SJ, Baker PJ, Lynch Y (2017) The influence of climate and drought on urban tree growth in southeast Australia and the implications for future growth under climate change. Landsc Urban Plan 167:275–287. https://doi.org/10.1016/j.landurbplan.2017.06.012
Ovenden TS, Perks MP, Clarke TK, Mencuccini M, Jump AS (2021) Life after recovery: increased resolution of forest resilience assessment sheds new light on post-drought compensatory growth and recovery dynamics. J Ecol 109(9):3157–3170. https://doi.org/10.1111/1365-2745.13576
Peng ZT, Zhang YD, Zhu LJ, Guo MM, Lu QG, Xu K, Shao H, Mo QF, Liu SR (2023) Spatial and temporal patterns of the sensitivity of radial growth response by Picea schrenkiana to regional climate change in the Tianshan Mountains. J for Res 34:1669–1681. https://doi.org/10.1007/s11676-023-01629-y
Pérez-de-Lis G, García-González I, Rozas V, Olano JM (2016) Feedbacks between earlywood anatomy and non-structural carbohydrates affect spring phenology and wood production in ring-porous oaks. Biogeosciences 13(19):5499–5510. https://doi.org/10.5194/bg-13-5499-2016
Qi HL, He P (2021) Climate change characteristics of temperature and precipitation in Yichun area in recent 60 years. Environ Prot Circ Econ 41(12):70–72 (in Chinese)
Schwalm CR, Anderegg WRL, Michalak AM, Fisher JB, Biondi F, Koch G, Litvak M, Ogle K, Shaw JD, Wolf A, Huntzinger DN, Schaefer K, Cook R, Wei Y, Fang Y, Hayes D, Huang M, Jain A, Tian H (2017) Global patterns of drought recovery. Nature 548:202–205. https://doi.org/10.1038/nature23021
Stokes M, Smiley T (1968) Tree-ring dating. The University of Chicago Press, Chicago
Su JJ, Wang XC (2017) Spatio-temporal variations in climate-growth relationships of three hardwood tree species across the north Zhangguangcai Mountains, northeast China. Acta Ecol Sin 37(5):1484–1495. https://doi.org/10.5846/stxb201509251973. (in Chinese)
Sutinen S, Partanen J, Viherä-Aarnio A, Häkkinen R (2009) Anatomy and morphology in developing vegetative buds on detached Norway spruce branches in controlled conditions before bud burst. Tree Physiol 29(11):1457–1465. https://doi.org/10.1093/treephys/tpp078
Takahashi K, Azuma H, Yasue K (2003) Effects of climate on the radial growth of tree species in the upper and lower distribution limits of an altitudinal ecotone on Mount Norikura, central Japan. Ecol Res 18(5):549–558. https://doi.org/10.1046/j.1440-1703.2003.00577.x
Toromani E, Sanxhaku M, Pasho E (2011) Growth responses to climate and drought in silver fir (Abies alba) along an altitudinal gradient in Southern Kosovo. Can J for Res 41(9):1795–1807. https://doi.org/10.1139/x11-096
Vicente-Serrano SM, Beguería S, López-Moreno JI (2010) A multiscalar drought index sensitive to global warming: the standardized precipitation evapotranspiration index. J Clim 23(7):1696–1718. https://doi.org/10.1175/2009jcli2909.1
Villanueva RAM, Chen ZJ (2019) Ggplot2: elegant graphics for data analysis (2nd ed.). Measurement 17(3):160–167. https://doi.org/10.1080/15366367.2019.1565254
Vitasse Y, Bottero A, Cailleret M, Bigler C, Fonti P, Gessler A, Lévesque M, Rohner B, Weber P, Rigling A, Wohlgemuth T (2019a) Contrasting resistance and resilience to extreme drought and late spring frost in five major European tree species. Glob Chang Biol 25(11):3781–3792. https://doi.org/10.1111/gcb.14803
Vitasse Y, Bottero A, Rebetez M, Conedera M, Augustin S, Brang P, Tinner W (2019b) What is the potential of silver fir to thrive under warmer and drier climate? Eur J for Res 138(4):547–560. https://doi.org/10.1007/s10342-019-01192-4
Watanabe Y, Wakabayashi K, Kitaoka S, Satomura T, Eguchi N, Watanabe M, Nakaba S, Takagi K, Sano Y, Funada R, Koike T (2016) Response of tree growth and wood structure of Larix kaempferi, Kalopanax septemlobus and Betula platyphylla saplings to elevated CO2 concentration for 5 years exposure in a FACE system. Trees 30(5):1569–1579. https://doi.org/10.1007/s00468-016-1390-9
Weiser CJ (1970) Cold resistance and injury in woody plants. Science 169(3952):1269–1278. https://doi.org/10.1126/science.169.3952.1269
White TL, Adams WT, Neale DB (2007) Forest genetics. CABI Publishing, Cambridge
Yang YZ, Lin WS, Sun YW (2018) Study on tree growth models of dominant tree species in the coniferous and broad-leaved mixed forest in the Lesser Khingan Mountains. For Res Manag. https://doi.org/10.13466/j.cnki.lyzygl.2018.03.010. (in Chinese)
Yu J, Xu QQ, Liu WH, Luo CW, Yang JL, Li JQ, Liu QJ (2016) Response of radial growth to climate change for Larix olgensis along an altitudinal gradient on the eastern slope of Changbai Mountain, Northeast China. Chin J Plant Ecol 40(1):24–35. https://doi.org/10.17521/cjpe.2015.0216. (in Chinese)
Yuan DY, Zhu LJ, Cherubini P, Li ZS, Zhang YD, Wang XC (2021) Species-specific indication of 13 tree species growth on climate warming in temperate forest community of northeast China. Ecol Indic 133:108389. https://doi.org/10.1016/j.ecolind.2021.108389
Zang C, Biondi F (2015) Treeclim: an R package for the numerical calibration of proxy-climate relationships. Ecography 38(4):431–436. https://doi.org/10.1111/ecog.01335
Zhao XP, Guo PP, Zhang ZL, Peng HX (2019) Anatomical features of branchwood and stemwood of Betula costata Trautv. from natural secondary forests in China. BioResources 14(1):1980–1991. https://doi.org/10.15376/biores
Zhu JH, Hou ZH, Zhang ZJ, Luo YJ, Zhang XQ (2007) Climate change and forest ecosystem: impacts, vulnerability and adaptation. Sci Silvae Sin 43(11):138–145 (in Chinese)
Acknowledgements
We thank Binqing Zhao and Wenqi Song, for their assistance in the laboratory. We also thank the relevant forestry bureau staff and managers for facilitating field sampling.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This study was supported by the Key Project of the China National Key Research and Development Program (2021YFD2200401) and the National Natural Science Foundation of China (42177421 and 41877426).
Corresponding editor: Tao Xu.
The online version is available at http://www.springerlink.com.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, D., An, Y., Li, Z. et al. Differences and similarities in radial growth of Betula species to climate change. J. For. Res. 35, 40 (2024). https://doi.org/10.1007/s11676-023-01690-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11676-023-01690-7