Abstract
The semi-arid mangroves of the Gulf of Kachchh, the largest ecosystems on the west coast of India, are poorly studied in terms of vegetation structure and environmental parameters in spite of their conservation significance. Therefore, it is necessary to document the structural features of these mangroves in view of ongoing coastal industrial development. Mangrove forest structure in 10 locations on the northern and southern coasts of the Gulf of Kachchh were assessed using the line intercept transect method. Descriptions included density of young and mature age classes, tree heights, diameters at breast height (DBH) and aboveground biomass, along with seven significant environmental variables. Mature tree densities ranged from 350 to 1567 ind. ha−1, while average height and girth at breast height ranged from 1.0 to 6.8 m and 3.0 to 137.0 cm, respectively. The majority of trees (55.6%) were in ≤ 1.8 m height class followed by a 1.9 to 2.4 m class (25.1%). DBH was most often in class 2 cm or lower than that. Among the canopy index classes, more trees were recorded in class ≤ 2 cm. The regeneration density was greater than the recruitment class. This study indicates that the poor structural attributes of Avicennia marina Vierth. var. acutissima Stapf and Mold dominated mangroves are largely due to aridity induced by scarce and erratic rainfall and high soil and water salinities. The results should be valuable in conserving and sustainably managing these mangroves in the face of developmental activities.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mangrove structural analysis is an important tool to understanding stand dynamics and is useful in the management of mangrove ecosystems (Adams et al. 2004). Knowledge of mangrove structure helps create an awareness of their status and future stand dynamics (Dahdouh-Guebas et al. 2002). A sound understanding of mangrove dynamics helps managers to determine the appropriate remedial measures to negate the impacts of development. Due to the importance of mangroves, their structure has gained attention worldwide (Fromard et al. 1998; Chen and Twilley 1999; Dahdouh-Guebas and Koedam 2001; Kairo et al. 2002; Satyanarayana et al. 2002; Adams et al. 2004; Coronado-Molina et al. 2004; Chowdhury et al. 2016; Sharma et al. 2016). The objectives of this study are to evaluate the structural attributes of the Gulf of Kachchh mangrove ecosystems and their governing environmental variables in order to promote their conservation. This will help us gain a better understanding of their dynamics.
Mangroves of Gujarat state are located in arid to semi-arid climates over 1140 km2, making them the largest mangrove ecosystems on the west coast of India (FSI 2017). Mangrove forests of the Gulf of Kachchh cover approximately 986 km2 in the state of Gujarat and are dominated by Avicennia marina (Forsk.) Vierth. var. acutissima Stapf and Mold formations with the sporadic occurrence of eight other mangrove species (ICMAM 2004; GEC and BISAG 2008). Despite the designation of the southern coast of Gulf of Kachchh as a Marine National Park in 1982, recent development activities have stressed these mangroves. Industrial development is impacting these mangrove ecosystems by altering edaphic conditions. Systematic studies of the Gulf of Kachchh mangroves are limited (Chavan 1985; Untawale and Wafar1988; Singh 2000; Nayak and Anjali 2001; Thivakaran et al. 2003; Saravanakumar et al. 2009; Patel et al. 2010). Most of the earlier studies have dealt with the coverage and distribution of mangroves by remote sensing (Nayak et al. 1992; Bahuguna and Nayak1996). However, few studies have focused on stand dynamics and structural variations along the coastline. Reliable information about vegetation structure and governing abiotic properties are a necessity for conservation efforts (Selvam 2003; Chowdhury and Maiti 2016a, b, c). Rapid industrialization in this region harboring extensive mangrove formations requires active management and a sound understanding of mangrove structural and demographic characteristics. As noted by Soares and Schaeffer-Novelli (2005), Donato et al. (2011) and Alongi et al. (2016), studying mangrove forest structural variables, including biomass, could address globally significant issues (Sharma and Gobi 2016) such as carbon sequestration and climate change. In addition, their dynamics could also be better understood. Although many descriptive works dealing with the ecology of Indian mangroves have been carried out (Satyanarayana et al. 2002; Thivakaran et al. 2003), structural attributes of the Gulf of Kachchh mangrove formations need to be studied in detail, particularly with ongoing industrial development and the conservation significance of these ecosystems. This study provides a demographic insight of the Gulf of Kachchh mangroves by analyzing their vegetation structure to aid in the conservation efforts.
Materials and methods
Study area
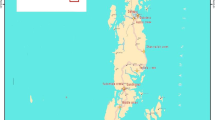
The Gulf of Kachchh (22°15′ to 23°40′N; 68°20′ to 70°40′E) in the northwestern part of India covers some 7350 km2 (Chauhan et al. 2006). The networks of creeks and mudflats and differential macro-tidal levels ranging from 3.5 to 6.7 m, creates several hydrographic peculiarities (Vethamony et al. 2004). The Gulf has more than 40 islands of different sizes, each harbouring important ecosystems such as coral reefs and mangroves with a rich associated fauna and flora (Babu et al. 2005). Only a few seasonal rivers empty freshwater into the Gulf, influencing its biological, physical and chemical characteristics during the monsoon months of June–September. The Gulf of Kachchh is semi-arid with higher mean annual rainfall in the southern coast (565 mm) cf. the northern coast (348 mm). The Gulf area therefore experiences a negative water balance due to the lack of perennial river discharge.
In spite of its rich mangroves and coral formations, in the last two decades the Gulf coastal belt has been subjected to aggressive industrial development such as petrochemical installations, oil refineries and port facilities due to its strategic location and proximity to Middle East countries. Dredging in these ports dislodges enormous quantities of bottom sediments, leading to severe mangrove degradation (ICMAM 2004).
Seven study locations on the southern coast, (Positra, Ashapura, Kalubar, Narara, Chhad, Jodia-Variyali, and Jodia-Dora) and three locations on the northern coast (Kandla, Mundra and Kharo) were selected based on mangrove distribution (Fig. 1). The southern coastline is muddy, interspersed with rocky, sandy intertidal belts. Kalubar is the largest island with many mangrove- lined creek systems and coral formations. Coastal stretches of Jodia-Variali and Jodia-Dora are straight and exposed to open waters of Gulf of Kachchh. The three study locations on the north coast are along minor and major creek systems. Kandla and Mundra sites at the end of the Gulf are characterized by highly muddy shores facing severe threats from heavy port-related activities. Pristine mangrove stands in Kharo in northwestern Kachchh near the international border with Pakistan, have limited anthropogenic activities due to security reasons. The grey mangrove, A. marina, is the dominant species in all the stands studied, with the sporadic occurrence of other species, especially Rhizophora mucronata Lam. and Ceriops tagal (Pers.) C.B. Rob. One species observed as a few individual plants outside the study locations was Aegiceras corniculatum (L.) Blanco at Kandia, thought to be locally extinct.
Mangroves
Gulf of Kachchh mangroves are overwash fringe formations (Ewel et al. 1998) occurring in tide-dominated high salinity regimes and lacking riverine inflow. Vegetation characteristics in the selected locations were studied using the line intercept transect method (Kershaw 1973; Mueller-Dombois and Ellenberg 1974) from November 2006 to March 2007. In the ten different stands along the coastal stretch of 756 km, 60 transects were established with 162 quadrats covering 4351 trees.
The number of transects in each location varied from 3 to 13 in order to accurately represent the particular stand. The length of each transect and distance between quadrats in each transect was based on the width of the mangrove stand as measured by GPS from low to high tide levels. In each plot, the number of trees, tree height and girth at breast height (GBH) were measured using ranging rods and measuring tapes. GBH readings were converted to DBH by dividing by 3.14. The DBH of all trees ≥ 1 m were measured. In case of a branching stem at a specific height, the procedure suggested by English et al. (1997) was followed. Canopy length and width, measured with graduated poles, were multiplied to calculate canopy index (CI). Within each 10 × 10 m plot, 1 × 1 m and 2 × 2 m subplots were laid randomly to determine regeneration and recruitment classes. Seedlings ≤ 50 cm tall were considered regeneration, while the recruitment class was well-established saplings ≥ 50 cm in height. The density of mature, regeneration and recruitment material for each location was expressed as a number per hectare. Tree height, DBH and CI data were segregated into size classes to study the frequency of occurrence of A. marina, Ceriops tagal and Rhizophora mucronata. Bray–Curtis cluster analysis (SPSS 14), which expresses similarity or dissimilarity between different groups in the form of a dendrogram, was used to test the ecological distance between different locations.
For calculating the aboveground biomass (AGB), the general allometric equation (B = 0.251 ρ (D2.46) of Komiyama et al. (2005) was used as it employs DBH as the predictive variable which can be measured more accurately than height. Linear regression plots were constructed for all sites and species to verify the strength of the relationship R2 between the dependent variable AGB and the independent variable DBH. Biomass for the three species and for the whole plot was calculated and expressed as mg ha−1.
Abiotic parameters
Important abiotic parameters that play a significant role in shaping the mangrove vegetation structure were analysed. These were studied during the dry winter season of November 2006 to March 2007. Creek water salinity (ppt, parts per thousand) was measured during low tides with a pre-calibrated refractometer (Master 3 M, Aatago, Japan). Porewater samples for salinity and pH were collected with a siphon and syringe, and soil samples were collected for soil texture studies. These samples were collected in triplicate at three tidal levels at a depth of 20 cm. Hydrogen ion concentration (pH) was estimated in situ using a pre-calibrated pH pen (Model 331, Hanna, Italy). Samples for texture analysis were oven-dried and mechanically sieved for sand (0.125–0.250 mm); silt (0.0039–0.0625 mm); and clay (0.00098–0.0039 mm). Samples for dissolved oxygen (DO) were collected in biological oxygen demand bottles and a modified Winkler’s titrimetric method (APHA 1998) was used to estimate dissolved oxygen. Total suspended solids (TSS) were estimated by filtering 1-L of water sample through a glass micro fibre filter and the results were expressed as mg L−1. Surface water temperature was measured using a calibrated thermometer (sensitivity ± 0.1 °C). Turbidity of suspended particles was measured in a calibrated nephelometer (Model 331, Hanna, Italy) and the results expressed in nephelometer turbidity units (NTU).
Results
Mature tree density and composition
The grey mangrove, A. marina, is the principle mangrove species in the Gulf of Kachchh, forming pure stands in seven out of the ten locations studied. Rhizophora mucronata and Ceriops tagal were found only at the Chhad and Kalubar sites in the southern coast and at Mundra on the northern coast. For A. marina, the highest density of 3308 ha−1 was recorded at Mundra while the lowest 1462 ha−1 was at Kalubar (Table 1). The densities of C. tagal and R. mucronata were highest at Kalubar with 292.0 and 223.0 ha−1, respectively. At the Chhad site, C. tagal was a mere 7.5% of the total density, while at the Kalubar site, R. mucronata and C. tagal were 11.3% and 14.8%, respectively. At the Mundra site, R. mucronata contributed less than 1.0% to the total density, while C. tagal was 6.0%. A. marina had the highest relative density of 96.4% followed by C. tagal (2.5%) and R. mucronata (1.1%) (Fig. 2).
Mature tree height
The average tree height values ranged from 1.0 to 6.8 m in different locations, with the highest variation at Kalubar and the lowest at Jodia-Dora (Table 1). Mean tree height was 2.2 ± 0.6 m and 2.1 ± 1.1 m for C. tagal and A. marina, respectively, at Kalubar. The mean tree height of C. tagal was larger at Chhad and Kalubar, while it was shorter at Mundra.
Segregation of tree heights into 0.6 m frequency classes showed that more trees (55.6%) were ≤ 1.8 m followed by 25% of the trees in the 1.9–2.4 m class. There were few (1.0%) trees 4.3–4.8 m (Fig. 3).
Diameter at breast height (DBH)
DBH ranged from 0.3 to 43.6 cm, and mean values were 7.0 cm (± 2.8) and 21.1 cm (± 0.2) at Mundra and Kalubar, respectively (Table 1). Stands at Kalubar, Mundra and Chhad had larger DBH ranges while R. mucronata and C. tagal stands showed less variation. Among DBH classes, 25% of the trees 3.1–4.5 cm were predominant followed by 20% of the trees in the 1.6–3.0 cm class (Fig. 4). In all study locations, trees ≥ 16.5 cm were fewest whereas in Positra, Narara, Chhad, Kalubar and Mundra stands it was more in number. For A. marina, all 13 girth classes were represented at Chhad, Mundra and Kharo locations but not in the Kandla and Jodia-Dora ecosystems.
Canopy indices
Canopy index values were higher for A. marina mangroves than for C. tagal and R. mucronata. Among canopy classes, 54.3% of the trees were in the frequency class of ≤ 2. Other higher frequency classes (8–9, 9–10 and 10–11) were least represented among all the sites, whereas lower 2–4 classes were well-represented (Fig. 5). Site locations such as Positra, Ashapura and Narara had 12 out of 13 frequency classes.
A Bray–Curtis coefficient (Bray and Curtis1957) was used to produce a hierarchical cluster for 10 study locations using tree density, DBH and height as variables (Fig. 6). Four groups were identified at a scale of 1–10 (Fig. 6), apparently due to similar structural characters as a function of their geographical proximity and similar environments. The first cluster, Narara, Positra and Chhad is located on the western part of the southern shore, whereas the second cluster, represented by a single location, Kalubar, is an island ecosystem where maximum average tree heights and high floral diversity and density were recorded. The third cluster, Mundra, Kharo and Ashapura, share similar vegetation attributes and the fourth, Jodia-Variyali, Kandla and Jodia-Dora, is located on the inner gulf and is characterized by the lowest floral density.
Aboveground biomass (AGB)
The grey mangrove ecosystem had the highest aboveground biomass in all locations, followed by C. tagal and R. mucronata. The highest values were in the Kharo site (216.8 mg ha−1) followed by the Narara (105.3 mg ha−1) and Mundra (101.9 mg ha−1) sites. The strongest regression co-efficient (R2) between biomass and DBH was at the Chhad (C. tagal; R2 = 0.92, p < 0.001), Mundra (C. tagal; R2 = 0.93, p < 0.001) and Kandla sites (A. marina; R2 = 0.93, p < 0.001). The relationship between DBH and biomass for the three species followed a second order polynomial trend (non-linear) as evident in Fig. 7a–c. R2 values were highest for C. tagal (R2 = 0.999), followed by R. mucronata (R2 = 0.999), and A. marina (0.994) (Fig. 7a–c).
Younger classes
Regeneration in all locations was higher than recruitment; based on location, the average regeneration density ranged from 21,111 ind. ha−1 at Kandla to 550,733 ind. ha−1 at Jodia-Variyali (Table 2). Regeneration was high in Jodia-Dora (295,911 ind. ha−1) and Ashapura (AS) stands (293,892 ind. ha−1). The ratio between regeneration and recruitment categories was lower in Chhad (1:1.5), Kandla (1:2.3) and Kharo stands (1:5), indicating a high entrance of regeneration into the recruitment category. At the Ashapura, Jodia-Variyali and Positra sites, the regeneration to recruitment ratio was higher, implying poor entrance of this class into the mature category (Table 2). A ratio of 1:2 and 1:2.5 was calculated between recruitment and mature tree categories at Ashapura and Mundra sites, indicating good sapling recruitment.
Abiotic factors
Mangrove creek water and porewater salinity significantly influenced floral diversity, growth, and height. Creek water salinity in the southern coastal locations ranged from 34 to 41 ppt (parts per thousand), with an average of 36.7 ppt which was marginally higher than the average 38.7 ppt at northern locations. The average porewater salinity values of the three quadrats of each transect increased in upper tidal reaches (Table 3). The lowest and highest porewater salinities were at Jodia-Dora and Chhad, recording 51.0 and 40.7 ppt, respectively. Creek water pH ranged from 7.2 to 8.0 at Chhad and Kharo, respectively (Table 3). The mean in all locations was 7.5. However, porewater pH was acidic, ranging from 6.5 to 6.9. Creek water temperatures ranged from 22.0 to 28.4 °C with an average of 25.0 °C, whereas, soil temperatures ranged from 25.0 to 29.0 °C with a mean of 27.6 °C (Table 3).
Turbidity at Kharo was 52 NTU and 162 NTU at Kandla, showing fairly high fluctuations among the locations (Table 3). Suspended solids fluctuated significantly among locations with a minimum level of 52 mg L−1 at Mundra and a maximum 168 mg L−1 at Jodia-Dora. Dissolved oxygen saturation in the waters of mangrove creeks was generally low, with minimum and maximum levels of 2.9 and 4.5 mg L−1 at Ashapura and Kharo, with an overall mean of 3.7 mg L−1. Analysis of sediments for the four fractions (silt, fine and coarse clay, sand) did not reveal any major variations. Only silt and fine clay constituents were dominant in all locations (Table 4).
Discussion
This study shows that structural characters and diversity of the Gulf of Kachchh mangroves are less comparable with other mangrove formations on the Indian subcontinent, such as the Pitchavaram (Kathiresan et al. 1994), the Sundarbans (Saha and Choudhury 1995; Chowdhury et al. 2016) and Kakinada Bay (Satyanarayana et al. 2002), due to their aridity and harsh environmental conditions. Structure of the Gulf of Kachchh mangroves seems to be governed largely by regimes of low, highly variable rainfall and high evaporation as a function of aridity. The importance of various physical and climatic factors in relation to salinity in determining the vegetation structure of mangroves is well-demonstrated (Bunt et al. 1982; Ball 1988; Ball and Pidsley 1988; Duke 1992; Ukpong 1997; Chowdhury et al. 2016).
These mangroves are dominated by A. marina or grey mangrove, the second most dominant mangrove genus worldwide, and are particularly prevalent in harsh climatic conditions (Duke 2001). The arid conditions and erratic rainfall of the Gulf of Kachchh confirms the observation by Blasco (1975) that Gujarat mangroves are floristically poor and semi-arid. In all the stands studied, trees on the waterfront were considerably taller, and a gradual reduction in height could be discerned towards high tide levels due to differing inundation patterns. Although eight true mangrove species have been reported earlier from the Gulf, only A. marina was dominant in the locations studied. This may be due to the well-established, extraordinary ecological plasticity of A. marina to thrive in harsh environmental conditions like those of the Gulf of Kachchh (Downton 1982; Clough 1984; MacNae 1986; Shalom-Gordon and Dubinsky 1993; Ye et al. 2005; Jayatissa et al. 2008). Patel et al. (2010) demonstrated the germination ability and high salt tolerance of A. marina in Gujarat mangroves. Khan and Aziz (2001) showed that A. marina, R. mucronata and C. tagal have higher tolerance to salinity than other species, and thus have wide distribution in Karachi mangroves of Pakistan, which has close geographical proximity to the Gulf of Kachchh. In their study, A. marina was also the tallest of the three species under higher salinity conditions, supporting the results in this study.
Mangrove stand density and species composition are influenced by many factors, including salinity regime, seasonality of rainfall, tidal inundation, freshwater inflow, micro-level topography, sediment composition and community structure. Higher entrance of a recruitment class into the mature tree category leads to the latter’s higher density at Mundra (3308 ind. ha) and Ashapura (2742 ind. ha) sites, which had low ratios of recruitment to mature tree densities. Along with factors such as tidal inundation and rainfall, upstream freshwater runoff is vital in determining floristic diversity in mangroves (Venkatesan 1966; Blasco and Aizpuru 1997; Naskar and Mandal 1999). Besides the inherent aridity of the region, the development of numerous water harvesting structures in the upland areas has curtailed downstream runoff to coastal waters, resulting in poor mangrove species diversity.
Close positive relationships among younger age classes and mature age classes, as observed by Dahdouh-Guebas et al. (2004) in Kenyan and Sri Lankan mangroves, could be observed in this study as well, which may be due to the inherent nature of the grey mangrove species to spread propagules widely. In addition, conducive canopy gaps and tidal reach, (by way of propagule dispersal), may also influence the establishment of a regeneration class and their successful entry into recruitment and mature tree categories. Higher densities of younger age classes as recorded at Ashapura and Narara could be due to such conducive factors prevailing in these stands.
Low biomass values in Kalubar, Chhad and Mundra sites could be due to aridity and subsequent hypersaline conditions (Fromard et al. 1998; Komiyama et al. 2008). Even though low latitude tropical countries have higher aboveground biomass in general (Komiyama et al. 2008), values in this study are comparatively lower than those reported for other Asian stands. Arid and semi-arid mangroves worldwide have poor structural attributes as reported for Mexico (Lizarraga et al. 2004), the Caribbean (Cintron et al. 1978) and on the east African coast (Walter and Steiner 1936). With a mean tree height (1.8 m) and mean DBH (4.8 cm), the Gulf of Kachchh mangroves could be classified as structurally inferior as per the classification of Pellegrini et al. (2009).The single species nature of the stands also suggests inferiority since the growth is less than that of the mixed stands (Das et al. 1997).
As a large contiguous formation, the Gulf of Kachchh mangroves are unique from the deltaic (Sundarbans), riverine (Godavari–Krishna) and island (Andamans) formations of the Indian subcontinent in terms of structural attributes such as poor floral diversity and rich faunal association. Given the low annual mean rainfall of 565 mm, the height range of 1.0–6.8 m is normal, although it is not comparable with high rainfall areas of the east coast of India where trees as tall as 10.0 m have been recorded (Blasco 1975). Usually mangrove forests in regions with less than 1500 mm rain have a height range of 1.0–6.0 m (Duke et al. 1998). At Ashapura, Kalubar and Mundra, all height classes are well-represented in contrast to stands at Kandla, Jodia-Dora and Jodia-Variyali which showed anomalous height class distribution lacking many successive classes. Kandla mangroves especially face severe development-related threats, which results in drastic cover reduction and altered physical and chemical properties of the creeks (ICMAM 2004; Shirodkar et al. 2010). Stands at Jodia-Doria and Jodia-Variyali sites appear stunted as higher DBH classes coupled with short height classes were recorded, possibly due to high porewater salinity on these sites (Table 3). Height class distribution of C. tagal was moderately better than R. mucronata as more trees were evenly distributed in different frequency classes (Fig. 2). Their DBH frequency was also less and did not exceed the initial three frequency classes, showing that these two species were not stressed and are rejuvenating in the stands studied. Wider representation of different DBH and height classes at Ashapura, Kalubar and Mundra sites indicate that their stands are regenerating while this is not true for Kandla, Jodia-Dora and Jodia-Variyali stands where DBH and height frequency classes were poorly represented. Use of girth and height frequency classes to construct past dynamics of the mangrove stands was demonstrated in Kenyan and Sri Lankan mangroves (Dahdouh-Guebas et al. 2002). Similarly, in this study, variations in girth and height frequency classes at Ashapura, Kalubar and Mundra sites indicate their young and dynamic state contrary to Kandla, Jodia-Dora and Jodia-Variyali stands where increased porewater salinity might influence the overall mangrove structure.
This study, as a first attempt to delineate the structural characteristics of Gulf of Kachchh mangroves, indicates that this ecosystem is structurally inferior to other deltaic mangroves of India. The prevailing environmental variables such as salinity, rainfall regimes and the concomitant aridity play a pivotal role in governing structural variation. The on-going industrial development in the northern and southern shores of the Gulf of Kachchh is of concern for the future of this ecosystem. The congregation of twenty medium and major ports which handle ~ 60% of the country’s crude oil requirements, and allied coastal development in close proximity calls for strong management measures to conserve these mangroves. This initial primary information on the Gulf of Kachchh mangroves should assist forest managers for the sustainable management of these ecosystems.
References
Adams JB, Colloty BM, Bate GC (2004) The distribution and state of mangroves along the coast of Transkei, Eastern Cape Province, South Africa. Wetl Ecol Manag 12(5):531–541
Alongi DM, Murdiyarso D, Fourqurean JW, Kauffman JB, Hutahaean A, Crooks S, Pidgeon E (2016) Indonesia’s blue carbon: a globally significant and vulnerable sink for seagrass and mangrove carbon. Wetl Ecol Manag 24(1):3–13
APHA (1998) Standard method for the examination of water and wastewater, 20th edn. American Public Health Association, Baltimore
Babu MT, Vethamony P, Desa E (2005) Modelling tide-driven currents and residual eddies in the Gulf of Kachchh and their seasonal variability: a marine environmental planning perspective. Ecol Model 184:299–312
Bahuguna A, Nayak S (1996) Mangrove community discrimination using IRS-1C data. In: Proceedings of the national symposium on Remote sensing for natural resources with special emphasis to water management. Indian Society of Remote Sensing/NNRMS, Bangalore, pp 311–319
Ball MC (1988) Eco-physiology of mangroves. Trees 2(3):129–142
Ball MC, Pidsley SM (1988) Establishment of tropical mangrove seedlings in relation tosalinity. In: Larson H, Hanley R, Michie M (eds) Proceedings of a workshop on research and management in Darwin Harbour, Darwin, pp 123–134
Blasco F (1975) The mangroves of India. Trav Sec Sci Tech 14:154–156
Blasco F, Aizpuru M (1997) Classification and evolution of the mangroves of India. Trop Ecol 38:357–374
Bray JR, Curtis JT (1957) An introduction of the upland forest communities of southern Wisconsin. Ecol Monogr 27:325–349
Bunt JS, Willams WT, Clay HJ (1982) River water salinity and the distribution of mangrove species along several rivers in North Queensland. Aust J Bot 30:401–412
Chauhan OS, Jayakumar S, Menezes AAA, Rajawat AS, Nayak SR (2006) Anomalous inland influx of the River Indus, Gulf of Kachchh, India. Mar Geol 229:91–100
Chavan SA (1985) Status of mangrove ecosystem in Gulf of Kachchh. Proceedings of symposium on endangered marine animals and marine parks. 3, paper no. 42. Marine Biological Association of India, Cochin, p 15
Chen R, Twilley RR (1999) Patterns of mangrove forest structure and soil nutrient dynamics along the Shark River Estuary, Florida. Estuar Coasts 22(4):955–970
Chowdhury A, Maiti SK (2016a) Identification of metal tolerant plant species in mangrove ecosystem by using community study and multivariate analysis: a case study from Indian Sundarbans. Environ Earth Sci 75(9):744. https://doi.org/10.1007/s12665-016-5391-1
Chowdhury A, Maiti SK (2016b) Assessing the ecological health risk in a conserved mangrove ecosystem due to heavy metal pollution: a case study from Sundarbans Biosphere Reserve, India. Hum Ecol Risk Assess 22(7):1519–1541. https://doi.org/10.1080/10807039.2016.1190636
Chowdhury A, Maiti SK (2016c) Identifying the source and accessing the spatial variations, contamination status, conservation threats of heavy metal pollution in the river waters of Sundarbans Biosphere Reserve, India. J Coast Conserv 20(3):257–269
Chowdhury A, Sanyal P, Maiti SK (2016) Dynamics of mangrove diversity influenced by climate change and consequent accelerated sea level rise at Indian Sundarbans. Int J Glob Warm 9(4):486–506. https://doi.org/10.1504/IJGW.2016.076333
Cintron G, Lugo AE, Pool DJ, Morris G (1978) Mangroves of arid environments in Puerto Rico and adjacent Islands. Biotropica 10:110–121
Clough BF (1984) Growth and salt balance of the mangrove Avicennia marina (FORSK.) VIERH. and Rhizophora stylosa GRIFF. in relation to salinity. Aust J Plant Physiol 11:419–430
Coronado-Molina C, Day JW, Reyes E, Perez BC (2004) Standing crop and aboveground biomass partitioning of a dwarf mangrove forest in Taylor River Slough, Florida. Wetl Ecol Manag 12(3):157–164
Dahdouh-Guebas F, Koedam N (2001) Are the northernmost mangroves of West Africa viable?—A case study in Banc d’Arguin National Park, Mauritania. Hydrobiologia 458(1):241–253
Dahdouh-Guebas F, Kairo JG, Jayatissa LP, Cannicci S, Koedam N (2002) An ordination study to view vegetation structure dynamics in disturbed and undisturbed mangrove forests in Kenya and Sri Lanka. Plant Ecol 161:123–135
Dahdouh-Guebas F, Pottelbergh IV, Kairo JG, Cannicci S, Koedam N (2004) Human-impacted mangroves in Gazi (Kenya): predicting future vegetation based on retrospective remote sensing, social surveys and tree distribution. Mar Ecol Prog Ser 272:77–92
Das P, Basak UC, Das AB (1997) Restoration of mangrove vegetation in Mahadadi delta, Orissa, India. Mangroves Salt Marshes 1:155–161
Donato DKJB, Kauffman JB, Murdiyarso D, Kurnianto S, Stidham M, Kanninen M (2011) Mangroves among the most carbon-rich forests in the tropics. Nat Geosci 4(5):293–297
Downton WJS (1982) Growth and osmotic relations of the mangrove, Avicennia marina as influenced by salinity. Aust J Plant Physiol 9:519–552
Duke NC (1992) Mangrove floristics and biogeography. In: Robertson AI, Alongi DM (eds) Tropical mangrove ecosystems. American Geophysical Union, Washington, pp 63–100
Duke NC (2001) Gap creation and regenerative processes driving diversity and structure of mangrove ecosystems. Wetl Ecol Manag 9:257–269
Duke NC, Ball MC, Joanna C, Ellison JC (1998) Factors influencing mangroves biodiversity and distributional gradients. Glob Ecol Biogeogr Lett 7:27–47
English SC, Wilkinson, Basker V (1997) Survey manual for tropical marine resources, 2nd edn. Australian Institute of Mar. Sci Townsville, Townsville, pp 119–195
Ewel K, Twilley R, Ong JIN (1998) Different kinds of mangrove forests provide different goods and services. Glob Ecol Biogeogr Lett 7(1):83–94
Fromard F, Puig H, Mougin E, Marty G, Betoulle JL, Cadamuro L (1998) Structure, above-ground biomass and dynamics of mangrove ecosystems: new data from French Guiana. Oecologia 115(1):39–53
FSI (2017) India state of forest report. Forest Survey of India, Ministry of Environment, Forests and Climate Change, Govt. of India. Dehradun, India
GEC and BISAG (2008) Mangrove Atlas of Gujarat State. Gujarat Ecology Commission and Bhaskaracharya Institute for Space Applications and Geo-informatics, Gandhinagar, India
ICMAM (2004) Model integrated coastal and marine area management plan for Gulf of Kachchh, Department of Ocean Development (Ministry of Earth Sciences) Chennai, pp 154
Jayatissa LP, Wickramasinghe WAADL, Dahdough-Guebas F, Huxham M (2008) Interspecific variations in responses of mangrove seedlings to two contrasting salinities. Int Rev Hydrobiol 93:700–710
Kairo JG, Dahdouh-Guebas F, Gwada PO, Ochieng C, Koedam N (2002) Regeneration status of mangrove forests in Mida Creek, Kenya: a compromised or secured future? Ambio 31(7):562–568
Kathiresan K, Ramesh MX, Venkatesan V (1994) Forest structure and prawn seeds in Pichavaram mangroves. Environ Ecol 12:465–468
Kershaw KA (1973) Sampling test of comparison and application of quadrat measures. In: Quantitative and dynamic plant ecology. 2nd edn. William Clowes & Sons Limited, London, pp 21–39
Khan MA, Aziz I (2001) Salinity tolerance in some mangrove species from Pakistan. Wetl Ecol Manag 9:219–223
Komiyama A, Poungparn S, Kato S (2005) Common allometric equations for estimating the tree weight of mangroves. J Trop Ecol 21:471–477
Komiyama A, Ong JE, Poungparn S (2008) Allometry, biomass, and productivity of mangrove forests: a review. Aquat Bot 89:128–137
Lizarraga JAA, Verdugo FJF, Rubio AO (2004) Structure and litterfall of an arid mangrove stand on the Gulf of Califormia, Mexico. Aquat Bot 79:137–143
MacNae W (1986) A general account of the fauna and flora of mangrove swamps and forest in the Indo-west-Pacific region. Adv Mar Biol 6:73–270
Mueller-Dombois D, Ellenberg H (1974) Aims and method of vegetation ecology. Wiley, New York, p 547
Naskar KR, Mandal RN (1999) Ecology and biodiversity of Indian mangroves. Daya Publishing House, New Delhi, p 783
Nayak S, Anjali B (2001) Application of remote sensing data to monitor mangroves and other coastal vegetation of India. Indian J Mar Sci 30:195–213
Nayak SR, Bahuguna A, Shaikh MG, Chauhan SB, Rao RS, Arya AS (1992) Coastal environment. Scientific Note. Space Application Centre, Ahmedabad
Patel NT, Gupta A, Pandey AN (2010) Salinity tolerance of Avicennia marina (Forssk.) Vierh. from Gujarat coast of India. Aquat Bot 93:9–16
Pellegrini JAC, Soares MLG, Chaves FO, Estrada GCD, Cavalcanti VF (2009) A method for the classification of mangrove forests and sensitivity/vulnerability analysis. J Coast Res 56:443–447
Saha S, Choudhury A (1995) Vegetation analysis of restored and natural mangrove forest in Sagar Island, Sundarbans, east coast of India. Indian J Mar Sci 24:133–136
Saravanakumar A, Rajkumar M, Sun J, Serebiah JS, Thivakaran GA (2009) Forest structure of arid zone mangroves in relation to their physical and chemical environment in the western Gulf of Kachchh, Gujarat, Northwest coast of India. J Coast Conserv 13:217–234
Satyanarayana B, Raman AV, Dehairs F, Kalavati C, Chandramohan P (2002) Mangrove floristic and zonation patterns of Coringa, Kakinada Bay, east coast of India. Wetl Ecol Manag 10(1):25–37
Selvam V (2003) Environmental classification of mangrove wetlands of India. Curr Sci 84:757–765
Shalom-Gordon N, Dubinsky N (1993) Diurnal pattern of salt secretion in leaves of the black mangrove Avicennia marina on the Sinai coast of the Red Sea. Pacific Sci 47:51–58
Sharma SB, Gobi TA (2016) Impact of drought on soil and microbial diversity in different agro-ecosystems of the semiarid zones. In: Hakeem KR et al (eds) Plant, soil and microbes. Springer, Switzerland, p 278
Sharma SB, Gobi TA, Thakkar MG (2016) Invasion of alien species: an impending threat. Indian J Mar Sci 45(11):1409–1411
Shirodkar PV, Pradhan UK, Fernandes D, Haldankar SR, Rao GS (2010) Influence of anthropogenic activities on the existing environmental conditions of Kandla Creek (Gulf of Kutch). Curr Sci 98:815–828
Singh HS (2000) Mangroves in Gujarat: current status and strategy for conservation. GEER Foundation, Gandhinagar, p 128
Soares MLG, Schaeffer-Novelli Y (2005) Above-ground biomass of mangrove species I analysis of models. Estuar Coast Shelf Sci 65:1–18
Thivakaran GA, Saravanakumar A, Serebiah JS, Joshua J, Sunderraj W, Vijayakumar V (2003) Vegetation structure of Kachchh Mangroves, Gujarat, Northwest Coast of India. Indian J Mar Sci 32:37–44
Ukpong IE (1997) Vegetation and its relation to soil nutrient and salinity in the Calabar mangrove swamp, Nigeria. Mangroves and Salt Marshes 1:211–218
Untawale AG, Wafar S (1988) Distribution of mangroves along the Gulf of Kutch. In: Marine archaeology of Indian ocean countries, National Institute of Oceanography, Goa, pp 135–138
Venkatesan KR (1966) The mangroves of Madras state. Indian For 92:27–32
Vethamony P, Reddy GS, Babu MT, Desa E, Sudheesh K (2004) Tidal eddies in a semi-enclosed basin: a model study. Mar Environ Res 59:519–532
Walter H, Steiner M (1936) Die Ökologie der Ost-Afrikanischen Mangroven. Zeitschr f Bot 30:65–193
Ye Y, Tam NFY, Lu CY, Wong SH (2005) Effects of salinity on germination, seedling growth and physiology of three salt secreting mangrove species. Aquat Bot 83:193–205
Acknowledgements
This study was made possible by the support and encouragement of Shri Shyamal Tikadar, IFS, Conservator of Forest (Working Plan), who organized and directed the study. Valuable field assistance by many forest officials of Junagadh and Jamnagar Marine National Park and Sanctuary is gratefully acknowledged. The authors also thank Gujarat Institute of Desert Ecology (GUIDE) at Bhuj for financial support for this research work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project funding: This study was financially supported by the Marine National Park and Sanctuary (MNP & S), Jamnagar, Gujarat State, India through the project, “Mangrove Vegetation Characteristics of Gulf of Kachchh”.
The online version is available at http://www.springerlink.com
Corresponding editor: Yu Lei.
Rights and permissions
About this article
Cite this article
Thivakaran, G.A., Sharma, S.B., Chowdhury, A. et al. Status, structure and environmental variations in semi-arid mangroves of India. J. For. Res. 31, 163–173 (2020). https://doi.org/10.1007/s11676-018-0793-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-018-0793-4