Abstract
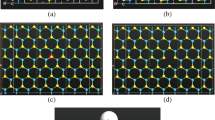
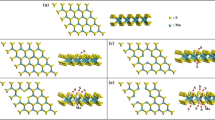
The molybdenum disulfide monolayer (MoS2) is gaining more attention due to its attractive electronic property, and it is extensively used in different electronic applications. The presence of vacancies on the MoS2 monolayer leads to an increase in the conductivity of the material. In this work, we have investigated the adsorption behavior and estimated the gas-sensing properties of the S-vacancy (VS) and the Mo-vacancy (VMo) MoS2 monolayers and the two-probe MoS2 devices with phosphorus pentafluoride (PF5) gas molecule. To explore the sensing and electronic properties of VS and VMo MoS2 towards PF5 gas adsorption, the adsorption distance, adsorption energy, charge transfer, band structure, and density of the states have been analyzed using density functional theory in combination with Non-Equilibrium Green's Function. The results show that the PF5 gas molecule is allowed to adsorb on the S- and Mo-vacancy MoS2 monolayers through van der Waals interaction. The PF5 gas molecule shows adsorption distances of 3.3274 Å and 2.8673 Å, adsorption energies of − 0.1640 eV and − 0.3489 eV, and charge transfers of − 0.025 Q (e) and − 0.053 Q (e) on the VS/VMo MoS2 monolayers. To study the electron transport properties, the device density of the states, the transmission spectra, and the current–voltage characteristics of the VS/VMo MoS2 two-probe devices have been analyzed. The results predicted that the Mo-vacancy MoS2 device shows relatively more adsorption towards the PF5 gas molecule when compared with the VS MoS2 device.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
K.S. Novoselov, D. Jiang, F. Schedin, T.J. Booth, V.V. Khotkevich, S.V. Morozov, and A.K. Geim, Proc. Natl. Acad. Sci. USA 102, 10451 (2005).
C. Lee, R. Sundharam, M. Jaiswal, Y. Lu, and S. Hofmann, J. Phys. D Appl. Phys. 50, 440401 (2017).
Q. Wang, P. Wu, G. Cao, and M. Huang, J. Phys. D Appl. Phys. 46, 505308 (2013).
W. Ju, T. Li, X. Su, H. Li, X. Li, and D. Ma, Phys. Chem. Chem. Phys. 19, 20735 (2017).
A.A. Ramanathan and I.O.P. Conf, Ser. Mater. Sci. Eng. 305, 012001 (2018).
D. Voiry, J. Yang, and M. Chhowalla, Adv. Mater. 28, 6197 (2016).
D.J. Late, Y.K. Huang, B. Liu, J. Acharya, S.N. Shirodkar, J. Luo, A. Yan, D. Charles, U.V. Waghmare, V.P. Dravid, and C.N.R. Rao, ACS Nano 7, 4879 (2013).
B. Cho, M.G. Hahm, M. Choi, J. Yoon, A.R. Kim, Y.J. Lee, S.G. Park, J.D. Kwon, C.S. Kim, M. Song, Y. Jeong, K.S. Nam, S. Lee, T.J. Yoo, C.G. Kang, B.H. Lee, H.C. Ko, P.M. Ajayan, and D.H. Kim, Sci. Rep. 5, 8052 (2015).
J.M. Jasmine, A. Aadhityan, C.P. Kala, and D.J. Thiruvadigal, Appl. Surf. Sci. 489, 841 (2019).
N. Izyumskaya, D.O. Demchenko, V. Avrutin, Ü. Özgür, and H. Morkoç, Turk. J. Phys. 38, 478 (2014).
S.Y. Cho, S.J. Kim, Y. Lee, J.S. Kim, W. Bin Jung, H.W. Yoo, J. Kim, and H.T. Jung, ACS Nano 9, 9314 (2015).
H. Yang, Y. Liu, C. Gao, L. Meng, Y. Liu, X. Tang, and H. Ye, J. Phys. Chem. C 123, 30949 (2019).
B. Zhao, C. Shang, N. Qi, Z.Y. Chen, and Z.Q. Chen, Appl. Surf. Sci. 412, 385 (2017).
Q. Yue, Z. Shao, S. Chang, and J. Li, Nanoscale Res. Lett. 8, 1 (2013).
H.G. Abbas, T.T. Debela, S. Hussain, and I. Hussain, RSC Adv. 8, 38656 (2018).
J. Ren, H. Liu, Y. Xue, and L. Wang, Nanoscale Res. Lett. 14, 293 (2019).
D. Burman, R. Ghosh, S. Santra, S. Kumar Ray, and P. Kumar Guha, Nanotechnology 28 (2017).
V. Vincent and J. Forensic, Leg. Investig. Sci. 1, 005 (2015).
F. Larsson, P. Andersson, P. Blomqvist, A. Lorén, and B.E. Mellander, J. Power Sources 271, 414 (2014).
R. Chaurasiya and A. Dixit, Phys. Chem. Chem. Phys. 22, 13903 (2020).
H. Li, M. Huang, and G. Cao, Phys. Chem. Chem. Phys. 18, 15110 (2016).
A. Shokri and N. Salami, Sensors Actuators B 236, 378 (2016).
W. Jin, Y. Guofeng, X. Junjun, L. Jianming, C. Qing, C. Dunjun, L. Hai, Z. Rong, and Z. Youdou, Sci. Rep. 8, 6 (2018).
C.V. Nguyen, N.N. Hieu, and D.T. Nguyen, Nanoscale Res. Lett. 10, 1 (2015).
D. Chen, X. Zhang, J. Tang, H. Cui, and Y. Li, Appl. Phys. A 124, 194 (2018).
C.P. Kala and D.J. Thiruvadigal, J. Comput. Electron. 17, 580 (2018).
K. Janani and D. John Thiruvadigal, Appl. Surf. Sci. 418, 406 (2017).
W. Xu, P. Li, S. Li, B. Huang, C. Zhang, and P. Wang, Physica E 73, 83 (2015).
D. Cao, H.B. Shu, T.Q. Wu, Z.T. Jiang, Z.W. Jiao, M.Q. Cai, and W.Y. Hu, Appl. Surf. Sci. 361, 199 (2016).
J. Noh, H. Kim, and Y. Kim, Phys. Rev. B 89, 205417 (2014).
Y. Cheng, Z. Zhu, W. Mi, Z. Guo, and U. Schwingenschlogl, Phys. Rev. B 87, 100401(R) (2013).
W. Wang, C. Yang, L. Bai, M. Li, and W. Li, Nanomaterials 8, 74 (2018).
M.G. Sensoy, D. Vinichenko, W. Chen, C.M. Friend, and E. Kaxiras, Phys. Rev. B 95, 1 (2017).
S. Sivasathya, D.J. Thiruvadigal, and S.M. Jaya, Chem. Phys. Lett. 609, 76 (2014).
S. Zhao, J. Xue, and W. Kang, Chem. Phys. Lett. 595–596, 35 (2014).
M. Sharma, A. Kumar, P.K. Ahluwalia, A. Kumar, and P.K. Ahluwalia, Physica E 107, 117 (2019).
L. Feng, A. Li, J. Su, Y. Zhang, and Z. Liu, Mater. Chem. Phys. 209, 146 (2018).
C. Liu, H. Dong, Y. Ji, T. Hou, and Y. Li, Sci. Rep. 8, 1 (2018).
S. Ahmad and S. Mukherjee, Graphene 03, 52 (2014).
S.C. Lu and J.P. Leburton, Nanoscale Res. Lett. 9, 1 (2014).
W.H. Khoo and S.M. Sultan, in IEEE Int. Conf. Semicond. Electron. Proceedings, ICSE, pp. 221–224 (2014).
J. Pang, Q. Yang, X. Ma, L. Wang, C. Tan, D. Xiong, H. Ye, and X. Chen, Phys. Chem. Chem. Phys. 19, 30852 (2017).
X. Zhang, H. Cui, and Y. Gui, Sensors 17, 363 (2017).
X.P. Chen, L.M. Wang, X. Sun, R.S. Meng, J. Xiao, H.Y. Ye, and G.Q. Zhang, IEEE Electron Device Lett. 38, 661 (2017).
Funding
Department of Science and Technology, Ministry of Science and Technology (SR/FST/PSI-155/2010).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meribah Jasmine, J., Preferencial Kala, C. & John Thiruvadigal, D. First Principle Study of Adsorption Behavior of PF5 Gas Molecule on S and Mo Vacancy MoS2 Monolayer. J. Electron. Mater. 50, 1668–1677 (2021). https://doi.org/10.1007/s11664-020-08480-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-020-08480-5