Summary
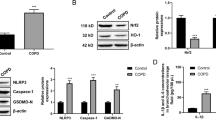
Although several studies confirmed that berberine may attenuate airway inflammation in mice with chronic obstructive pulmonary disease (COPD), its underlying mechanisms were not clear until now. We aimed to establish an experiment mouse model for COPD and to investigate the effects of berberine on airway inflammation and its possible mechanism in COPD model mice induced by cigarette smoke extract (CSE). Twenty SPF C57BL/6 mice were randomly divided into PBS control group, COPD model group, low-dose berberine group and high-dose berberine group, 5 mice in each group. The neutrophils and macrophages were examined by Wright’s staining. The levels of inflammatory cytokines TNF-α and IL-6 in bronchoalveolar lavage fluid (BALF) were determined by enzyme-linked immunosorbent assay. The expression levels of TGF-β1, Smad2 and Smad3 mRNA and proteins in lung tissues were respectively detected by quantitative real-time polymerase chain reaction and Western blotting. It was found that CSE increased the number of inflammation cells in BALF, elevated lung inflammation scores, and enhanced the TGF-β1/Smads signaling activity in mice. High-dose berberine restrained the alterations in the COPD mice induced by CSE. It was concluded that high-dose berberine ameliorated CSE-induced airway inflammation in COPD mice. TGF-β1/Smads signaling pathway might be involved in the mechanism. These findings suggested a therapeutic potential of high-dose berberine on the CSE-induced airway inflammation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease 2017 Report. Am J Respir Crit Care Med, 2017,195(5):557–582
Mun SY, Hwang YI, Kim JH, et al. Awareness of chronic obstructive pulmonary disease in current smokers: a nationwide survey. Korean J Intern Med, 2015,30(2):191–197
Churg A, Cosio M, Wright JL. Mechanisms of cigarette smoke-induced COPD: insights from animal models. Am J Physiol Lung Cell Mol Physiol, 2008,294(4):612–31
Yang ZS, Yan JY, Han NP, et al. Anti-inflammatory effect of Yu-Ping-Feng-San via TGF-β1 signaling suppression in rat model of COPD. Iran J Basic Med Sci, 2016,19(9):993–1002
Ferguson GT, Calverley PM, Anderson JA, et al. Prevalence and Progression of Osteoporosis in Patients With COPD: Results From the Towards a Revolution in COPD Health Study. Chest, 2009,136(6):1456–1465
Eurich DT, Lee C, Marrie TJ, et al. Inhaled Corticosteroids and Risk of Recurrent Pneumonia: A Population-Based, Nested Case-Control Study. Clin Infect Dis, 2013,57(8):1138–1144
O’Byrne PM, Rennard S, Gerstein H, et al. Risk of new onset diabetes mellitus in patients with asthma or COPD taking inhaled corticosteroids. Respir Med, 2012,106(11):1487–1493
Ni WJ, Ding HH, Tang LQ. Berberine as a promising anti-diabetic nephropathy drug: An analysis of its effects and mechanisms. Eur J Pharmacol, 2015,760:103–112
Peng L, Kang S, Yin Z, et al. Antibacterial activity and mechanism of berberine against Streptococcus agalactiae. Int J Clin Exp Pathol, 2015,8(5):5217–5223
Vuddanda PR, Chakraborty S, Singh S. Berberine: a potential phytochemical with multispectrum therapeutic activities. Expert Opin Investig Drugs, 2010,19(10): 1297–1307
Wang Z, Chen Z, Yang S, et al. Berberine Ameliorates Collagen-Induced Arthritis in Rats Associated with Anti-inflammatory and Anti-angiogenic Effects. Inflammation, 2014,37(5):1789–1798
Tillhon M, Guamán Ortiz LM, Lombardi P, et al. Berberine: new perspectives for old remedies. Biochem Pharmacol, 2012,84(10):1260–1267
Lin K, Liu S, Shen Y, et al. Berberine attenuates cigarette smoke-induced acute lung inflammation. Inflammation, 2013,36(5):1079–1086
Xu D, Wan C, Wang T, et al. Berberine attenuates cigarette smoke-induced airway inflammation and mucus hypersecretion in mice. Int J Clin Exp Med, 2015,8(6):8641–8647
Nana-Sinkam SP, Lee JD, Sotto-Santiago S, et al. Prostacyclin prevents pulmonary endothelial cell apoptosis induced by cigarette smoke. Am J Respir Crit Care Med, 2007,175(7):676–685
Zhang Y, Cao J, Chen Y, et al. Intraperitoneal injection of cigarette smoke extract induced emphysema, and injury of cardiac and skeletal muscles in BALB/C mice. Exp Lung Res, 2013,39(1):18–31
Woods JA, Wheeler JS, Finch CK, et al. Corticosteroids in the treatment of acute exacerbations of chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis, 2014,9:421–430
Silva OR, Montes JF, García-Valero J, et al. Cellular effectors of the inflammatory response in chronic obstructive pulmonary disease (COPD). Rev Med Chil, 2015,143(9):1162–1171
Bafadhel M, McCormick M, Saha S, et al. Profiling of Sputum Inflammatory Mediators in Asthma and Chronic Obstructive Pulmonary Disease. Respiration, 2012,83(1):36–44
Huang AX, Lu LW, Liu WJ, et al. Plasma Inflammatory Cytokine IL-4, IL-8, IL-10, and TNF-α Levels Correlate with Pulmonary Function in Patients with Asthma-Chronic Obstructive Pulmonary Disease (COPD) Overlap Syndrome. Med Sci Monit, 2016,22:2800–2808
Lee TM, Lin MS, Chang NC. Usefulness of C-reactive protein and interleukin-6 as predictors of outcomes in patients with chronic obstructive pulmonary disease receiving pravastatin. Am J Cardiol, 2008,101(4):530–535
Liang R, Zhang W, Song Y. Levels of leptin and IL-6 in lungs and blood are associated with the severity of chronic obstructive pulmonary disease in patients and rat models. Mol Med Rep, 2013,7(5):1470–1476
Ferrari R, Tanni SE, Caram LM, et al. Three-year follow-up of interleukin 6 and C-reactive protein in chronic obstructive pulmonary disease. Respir Res, 2013,14(1):24
Yu SY, Gao XW, Yan ZJ. Relationship between gene polymorphisms of tumor necrosis factor-α-308 and phenotypes of acute exacerbation of chronic obstructive pulmonary diseases. Chin J Tuberc Respir Dis, 2016,39(3):203–207
Chiang CH, Chuang CH, Liu SL. Transforming growth factor-βl and tumor necrosis factor-α are associated with clinical severity and airflow limitation of COPD in an additive manner. Lung, 2014,192(1):95–102
Morris DG, Huang X, Kaminski N, et al. Loss of integrin alpha v beta 6-mediated TGF-beta activation causes Mmp12-dependent emphysema. Nature, 2003,422(6928):169–173
Verhamme FM, Bracke KR, Joos GF, et al. TGF-β Superfamily in Obstructive Lung Diseases: More Suspects than TGF-β Alone. Am J Respir Cell Mol Biol, 2014,52(6):653–662
Su ZG, Wen FQ, Feng YL, et al. Transforming growth factor-βl gene polymorphisms associated with chronic obstructive pulmonary disease in Chinese population. Acta Pharmacol Sin, 2005,26(6):714–720
Morty RE, Königshoff M, Eickelberg O. Transforming growth factor-beta signaling across ages: from distorted lung development to chronic obstructive pulmonary disease. Proc Am Thorac Soc, 2009,6(7):607–613
Podowski M, Calvi C, Metzger S, et al. Angiotensin receptor blockade attenuates cigarette smoke-induced lung injury and rescues lung architecture in mice. J Clin Invest, 2011,122(1):229–240
Chung KF. The role of airway smooth muscle in the pathogenesis of airway wall remodeling in chronic obstructive pulmonary disease. Proc Am Thorac Soc, 2005,2(4):371–372
Yang YC, Zhang N, Van Crombruggen K, et al. Transforming growth factor-beta1 in inflammatory airway disease: a key for understanding inflammation and remodeling. Allergy, 2012,67(10):1193–1202
Halwani R, Al-Muhsen S, Al-Jahdali H, et al. Role of transforming growth factor-β in airway remodeling in asthma. Am J Respir Cell Mol Biol, 2011,44(2):127–133
Chitra P, Saiprasad G, Manikandan R, et al. Berberine inhibits Smad and non-Smad signaling cascades and enhances autophagy against pulmonary fibrosis. J Mol Med, 2015,93(9):1015–1031
Li L, Huang W, Li K, et al. Metformin attenuates gefitinib-induced exacerbation of pulmonary fibrosis by inhibition of TGF-β signaling pathway. Oncotarget, 2015,6(41):43 605–43 619
Li Y, Li JS, Li WW, et al. Long-term effects of three Tiao-Bu Fei-Shen therapies on NF-KB/TGF-β1/smad2 signaling in rats with chronic obstructive pulmonary disease. BMC Complement Altern Med, 2014,14(1):140
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest Statement
There are no conflicts of interest.
This work was supported by the National Natural Science Foundation of China (No. 81400008).
Rights and permissions
About this article
Cite this article
Wang, W., Zha, G., Zou, Jj. et al. Berberine Attenuates Cigarette Smoke Extract-induced Airway Inflammation in Mice: Involvement of TGF-β1/Smads Signaling Pathway. CURR MED SCI 39, 748–753 (2019). https://doi.org/10.1007/s11596-019-2101-8
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-019-2101-8