Abstract
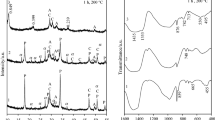
Hydrothermal treatment has been widely applied in the synthesis of well crystalline calcium silicate hydrate (CSH), such as tobermorite and xonotlite. However, both morphology and crystallinity of CSH are greatly affected by the conditions of hydrothermal treatment including siliceous materials, temperature increase rate and isothermal periods. In this study, the influence of hydrothermal conditions on the growth of nano-crystalline CSH was investigated based on XRD analysis. Results showed that siliceous materials with amorphous nature (i e, nano silica powder) are beneficial to synthesize pure amorphous CSH, while the use of more crystallized siliceous materials (i e, diatomite and quartz powder) leads to producing crystalline CSH. Results also indicate that the formation of tobermorite and xonotlite is greatly affected by the temperature rise rate during hydrothermal treatment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Snell DS. Review of Synthesis and Properties of Tobermorite, C-S-H(I), and C-S-H Gel[J]. Journal of the American Ceramic Society, 1975, 58: 292–295
Kurdowski W. Cement and Concrete Chemistry[M]. London: Springer, 2014
Byrappa K, Yoshimura M. Handbook of Hydrothermal Technology[M]. Oxford: William Andrew, 2013
Бабушкин ВИ, Матвеев ГМ, О П М Петросян. Thermodynamic of Silicates[M]. Beijing: China Architecture & Building Press, 1983
Taylor HFW Cement Chemistry[M]. London: Thomas Telford, 1997
Hou D, Ma H, Zhu Y, et al. Calcium Silicate Hydrate from Dry to Saturated State: Structure, Dynamics and Mechanical Properties[J]. Acta Materialia, 2014, 67: 81–94
Taylor HFW. The Calcium Silicate Hydrates[M]. London: Academic Press, 1964
Meller N, Kyritsis K, Hall C. The Mineralogy of the CaO–Al2O3–SiO2–H2O (CASH) Hydroceramic System from 200 to 350 °C[J]. Cement and Concrete Research, 2009, 39: 45–53
Shen W, Zhang T, Hu J, et al. Self Similarity of the Spherical C-S-H Particle in Cement Paste[J]. J. Wuhan Univ. Technol.-Mat. Sci. Edit., 2009, 24 (4): 684–687
Nelson S, Bell SV, Petra M, et al. Morphological Forms of Tobermorite in Hydrothermally Treated Calcium Silicate Hydrate Gels[J]. Journal of American Ceramic Society, 1996, 79: 2175–2178
Karatepe N, Ersoy-MeriÇboyu A, Küçükbayrak S. Activation of Ca(OH)2 Using Different Siliceous Materials[J]. Environmental Technology, 1999, 20: 377–385
Chen JJ, Thomas JJ, Taylor HFW, et al. Solubility and Structure of Calcium Silicate Hydrate[J]. Cement and Concrete Research, 2004, 34: 1499–1519
Wang SP. Calcium Silicate Hydrates Synthesis and Application from Steel Slag[D]. Chongqing: Chongqing Univeristy, 2011
Guan W, Ji F, Fang Z, et al. Low Hydrothermal Temperature Synthesis of Porous Calcium Silicate Hydrate with Enhanced Reactivity SiO2[J], Ceramics International, 2014, 40: 4415–4420
Jing ZZ, Jin FM, Yamasaki N, et al. Hydrothermal Synthesis of a Novel Tobermorite based Porous Material from Municipal Incineration Bottom Ash[J]. Ind. Eng. Chem. Res., 2007, 46: 2657–2660
Wang SP, Peng XQ, Geng JQ, et al. Synthesis of Calcium Silicate Hydrate based on Steel Slag with Various Alkalinities[J]. Journal of Wuhan University of Technology-Materials Science Edition, 2014 (4), 29: 789–794
Guan W, Ji F, Fang D, et al. Porosity Formation and Enhanced Solubility of Calcium Silicate Hydrate in Hydrothermal Synthesis[J]. Ceramics International, 2014, 40: 1667–1674
Li M, Liang H. Formation of Micro-porous Spherical Particles of Calcium Silicate (Xonotlite) in Dynamic Hydrothermal Process[J]. China Particuology, 2004, 2: 124–127
Wang S, Peng X, Tang L, et al. Influence of Inorganic Admixtures on the 11 Å-Tobermorite Formation Prepared from Steel Slags: XRD and FTIR Analysis[J]. Construction and Building Materials, 2014, 60: 42–47
Prevéy P. X-ray Diffraction Characterization of Crystallinity and Phase Composition in Plasma-sprayed Hydroxyapatite Coatings[J]. J. Therm. Spray Tech., 2000, 9: 369–376
Huang J. MDI Jade User's Manual-Guide of X-ray Diffraction Operation[M]. Changsha: Central South University, 2006
Connolly JR. Introduction to DataScan and Jade on the Scintag PADV System[OL]. https://doi.org/www.xray.cz/xray/csca/kol2011/kurs/Dalsi-cteni/Connolly-2005/08-DataScan-Jade-Intro.pdf.2003
Scherrer P. Bestimmung der Grösse und der Inneren Struktur von Kolloidteilchen Mittels Röntgenstrahlen Nachrichten von der Gesellschaft der Wissenschaften, Göttingen[J]. Mathematisch-Physikalische Klasse, 1918, 2: 98–100
Langford JI, Wilson AJC. Scherrer after Sixty Years: A Survey and Some New Results in the Determination of Crystallite Size[J]. Journal of Applied Crystallography, 1978, 11: 102–113
Meier M. Crystallite Size Measurement Using X-ray Diffraction[OL]. https://doi.org/www.kstreetstudio.com/science/experiments/files/XRD-CSize1.pdf
Greenberg SA. Reaction between Silica and Calicum Hydroxide Solutions I. Kinetics in the Temperature Range 30 to 85 ?[J]. The Journal of Physical Chemistry, 1961, 65: 12–16
Björnström J, Martinelli A, Matic A, et al. Accelerating Effects of Colloidal Nano-silica for Beneficial Calcium–Silicate–Hydrate Formation in Cement[J]. Chemical Physics Letters, 2004, 392: 242–248
Shaw S, Clark SM, Henderson CMB. Hydrothermal Formation of the Calcium Silicate Hydrates, Tobermorite (Ca5Si6O16(OH)2·4H2O) and Xonotlite (Ca6Si6O17(OH)2): An in Situ Synchrotron Study[J]. Chemical Geology, 2000, 167: 129–140
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the Fundamental Research Funds for the Central Universities (No.2018CDXYCL0018), the National Natural Science Foundation of China (NSFC) (No.51678093), and the National Youth Fund (No.51402029)
Rights and permissions
About this article
Cite this article
Wang, S., Peng, X., Tang, L. et al. Influence of Hydrothermal Synthesis Conditions on the Formation of Calcium Silicate Hydrates: from Amorphous to Crystalline Phases. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 33, 1150–1158 (2018). https://doi.org/10.1007/s11595-018-1947-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-018-1947-0