Abstract
Cardiovascular magnetic resonance (CMR) and computed tomography (CCT) are advanced imaging modalities that recently revolutionized the conventional diagnostic approach to congenital heart diseases (CHD), supporting echocardiography and often replacing cardiac catheterization. Nevertheless, correct execution and interpretation require in-depth knowledge of all technical and clinical aspects of CHD, a careful assessment of risks and benefits before each exam, proper imaging protocols to maximize diagnostic information, minimizing harm. This position paper, written by experts from the Working Group of the Italian Society of Pediatric Cardiology and from the Italian College of Cardiac Radiology of the Italian Society of Medical and Interventional Radiology, is intended as a practical guide for applying CCT and CMR in children and adults with CHD, wishing to support Radiologists, Pediatricians, Cardiologists and Cardiac Surgeons in the multimodality diagnostic approach to these patients. The first part provides a review of the most relevant literature in the field, describes each modality's advantage and drawback, making considerations on the main applications, image quality, and safety issues. The second part focuses on clinical indications and appropriateness criteria for CMR and CCT, considering the level of CHD complexity, the clinical and logistic setting and the operator expertise.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The constant improvements in surgical and interventional techniques have drastically increased the survival rate of congenital heart diseases (CHD) patients over the last decades, with an 85% estimate of children with CHD surviving to adulthood [1].
Imaging techniques are crucial in the multidisciplinary approach to these patients, characterizing anatomical structures and their functional status in order to improve management and guide pre and post-operative evaluation, lifelong surveillance and prognosis. Complementary use of various imaging modalities aims at incrementing accuracy, reproducibility, and cost-effectiveness while minimizing risks [1, 2].
Echocardiography is the first diagnostic tool in the preoperative evaluation and follow-up of CHD [3, 4]. However, it may be limited due to poor acoustic windows for the characterization of extracardiac anatomy and complex cases.
In the last decades, cardiovascular magnetic resonance (CMR) and computed tomography (CCT) have revolutionized the diagnostic approach to CHD [5], being an useful adjunct to echocardiography in multiple instances and they have replaced cardiac catheterization for many diagnostic indications [6–8]. Hence, good knowledge of their potential, limitations, and indications is mandatory [9–11]. Yet, some indications are still a matter of discussion.
The present consensus, proposed by the CMR/CCT working group of the Italian Society of Pediatric Cardiology and by the Italian College of Cardiac Radiology of the Italian Society of Medical and Interventional Radiology (SIRM), is addressed to Radiologists, Pediatricians, Cardiologists and Cardiac surgeons, interested in CHD imaging. It is divided into two parts. The first one provides a review of the most relevant literature in the field, a description of each modality’s advantages and drawbacks, and a comment on future technological perspectives. The aims are:
-
1.
To provide an overview of the technical advantages and disadvantages of CMR and CCT in CHD;
-
2.
To propose clinical recommendations, based on patient's age, CHD complexity, and the required operator expertise.
Further clinical issues are treated in the second part with a specific description of CCT and CMR appropriateness criteria based on this proposed novel approach.
Review of the literature on the indications of CMR and CCT in CHD
From the early ‘80 s several reports and guidelines have been published in this field, progressively attributing a more fundamental role to CMR and CCT in CHD [1, 6, 7].
In 2015, an European Society of Cardiology (ESC) consensus paper was published [9], based on expert opinions concerning CMR use in children with CHD in specific clinical situations, while the same year two expert consensus documents of the Society of CCT were published [10, 11]. Part 1 offered a systematic review of all indications and risks related to CCT in CHD. Part 2 described the optimal technical environment and protocols and the most relevant knowledge for CCT performance in CHD.
In 2018, the European Association of Cardiovascular Imaging released a position paper focused on the multimodality imaging approach to adult CHD [5]. This leading role of CMR and CCT is also established by the American College of Cardiology/American Heart Association (ACC/AHA) guidelines [12, 13]. The latest update re-emphasized how cardiac imaging must be supervised and interpreted by physicians with expertise and/or training in CHD [13]. Additionally, in 2020 the ACC/AHA published appropriateness criteria for using multimodality imaging during the follow-up care of CHD patients [14]. More recently, the new European guidelines for the management of adult CHD by the ESC were published [1], followed by two expert consensus documents by Radiologists and Cardiologists about the appropriateness criteria of CCT/CMR use in different clinical scenarios including CHD, endorsed by the SIRM and by the Italian Society of Cardiology [8, 15].
Strengths and limitations of CMR and CCT in CHD
CMR and CCT imaging have significantly changed the diagnostic approach to CHD, limiting the need for invasive procedures.
Some advantages are common to both techniques. Unlike echocardiography, they are not restricted by body habitus, acoustic windows or geometric assumptions. One of their most notable features is the superb three-dimensional (3D) capability, which allows for excellent visualization of cardiovascular structures with high spatial and temporal resolution (Fig. 1). This is especially useful when planning percutaneous or surgical interventions [5]. Moreover, CMR and CCT have proven very low intra/inter-observer variability in CHD assessment, with particular regard to biventricular function, volumes and mass quantification [16–18] (Fig. 2).
CCT 3D volume rendering images, sagittal view of a 54-year-old woman with bypass aortic coarctation palliation (a). CCT 3D volume rendering images of a double aortic arch with 3D rendering of airways structures (asterisks) showing anatomical relationships between them and the vascular ring (b). CCT Cardiovascular computed tomography, 3D tridimensional
On the other hand, limited availability, high costs, and the need for specific hardware/software equipment and expertise are the main drawbacks to their widespread use in CHD [13, 19, 20]. The risks of CMR and CCT are summarized in Table 1, while a summary of the main advantages and drawbacks of each imaging modality is described in Table 2.
Cardiovascular magnetic resonance
CMR is the only imaging modality offering in a single step exam excellent anatomical pictures of cardiovascular structures and functional/hemodynamic information [5, 7]. CMR is justified in each patient with insufficient clinical or echocardiographic data for monitoring, decision-making, or treatment planning [1], and in complex CHD to guide clinical decisions [9]. Lifelong follow-up with serial CMR imaging is also recommended in grown-up congenital heart disease (GUCH) patients [1], often submitted to surgical repairs during childhood (Fig. 3), for its high reproducibility and limited safety issues compared to CCT and/or catheterization, considering the relatively young population.
30-year-old female with repaired Tetralogy of Fallot (ToF). CMR SSFP image, RVOT sagittal plane shows a slightly reduced homograft caliber with post-stenosis pulmonary artery dilation (a). 21-year-old male after aortic coarctation and VSD repair. MRA MPR axial image shows residual pulmonary bifurcation and proximal branch arteries stenosis post pulmonary banding (b). 31-year-old female with repaired ToF. CMR SSFP 4-chamber view demonstrates right ventricular dilatation (c). CMR LGE short-axis view shows RVOT post-surgical scar (arrows) (d). CMR cardiovascular magnetic resonance, Hg Homograft, La left atrium, LPA left pulmonary artery, LGE late gadolinium enhancement, Lv left ventricle, MPA pulmonary artery, MPR Multiplanar Reformation, MRA Magnetic Resonance Angiography, Ra right atrium, RPA right pulmonary artery, Rv right ventricle, RVOT right ventricle outflow tract, SSFP Steady State Free Precession, VSD Ventricular septal defect
To date, CMR is considered the gold standard for volumes and myocardial mass assessment, especially of the right ventricle (RV) (i.e., cine imaging) and highly appropriate for flow and shunt quantification, allowing for hemodynamic assessment of valvular pathology (i.e., phase-contrast sequences). Nevertheless, CMR is not superior to echocardiography in estimating gradients or evaluating atrio-ventricular valvular and sub-valvular pathology and remains inferior in detecting small mobile structures like vegetations or patent foramen ovale [1, 21]. Moreover, CMR can precisely delineate intra and extra-cardiac anatomy by means of several sequences (i.e., black-blood spin echo, Magnetic Resonance Angiography, 3D steady-state free-precession). A further strength of CMR is its ability to perform tissue characterization, achieved with late gadolinium enhancement (LGE) sequences [5]. The acquisition of LGE-CMR, even though not always required in CHD and not to be repeated at each imaging follow-up examination, is a considerable integration to morpho-functional evaluation, allowing for the identification of focal myocardial fibrosis that has been associated to adverse cardiac events (i.e., heart failure, cardiac arrhythmias) in many CHD.
CMR can be performed on either 1.5 or 3 Tesla (T) scanners, with 1.5 T being the clinical standard due to more robust scanning sequences [22].
CMR has broadly a high safety profile, even in children. The lack of radiation exposure and options to avoid contrast administration in many cases are well-established advantages, characterizing its favorable benefit/risk profile compared with catheterization and CCT [23, 24]. Nonetheless, CMR involves exposure to electromagnetic energy (static magnetic fields, gradients and radiofrequency pulses), with intrinsic hazards from ferromagnetic external (i.e., "projectile effect") and/or implanted devices, induced electrical currents, heating, and acoustic noise. Therefore, a safety screening should be performed prior to every exam to rule out contraindications, including non-MR-compatible pacemakers (PMs), implantable cardioverter defibrillators (ICDs), cochlear implants, and other ferromagnetic devices [19, 22]. Most of implanted devices do not represent an absolute contraindication to CMR being classified as CMR-conditional. Nevertheless, CMR-conditional cardiac devices require collaboration with the electrophysiology team for CMR execution and may produce significant artifacts [25, 26]. Remarkably, safe protocols have recently been shown feasible with conventional PMs and ICDs [27, 28].
Despite recent technical advances, CMR is still time-consuming, especially in complex cases [29, 30]. Consequently, young children and uncooperative patients require deep sedation with spontaneous respiration or general anesthesia [1, 5], needing CMR-compatible equipment. CMR has proven to be safe even during general anesthesia in fragile subjects, when adopting a consolidated approach by an experienced multidisciplinary team [31, 32]. Patients affected by complex CHD or Williams syndrome deserve special consideration, because of an increased risk of adverse events during sedation [33, 34].
Intravenous administration of gadolinium-based contrast agents (GBCAs) is commonly performed in CHD, although not mandatory except for tissue characterization, myocardial perfusion or contrast-enhanced angiography. GBCAs can be considered safe in both children and adults [35], as adverse events are very rare and usually mild. A relative contraindication exists for severe kidney disease (glomerular filtration rate < 30 mL/min/1.73m2) due to the risk of nephrogenic systemic fibrosis, a rare but serious condition that can compromise internal organs [22]. Recently, the potential long-term effects of gadolinium deposition in tissues, particularly in the brain, have aroused considerable interest although its real clinical relevance still needs more research [36]. For the abovementioned reasons, since 2018 the European Medicines Agency and subsequently the corresponding Italian authority suspended the distribution of linear non-ionic GBCAs and limited the use of linear ionic agents. Therefore, macrocyclic ionic molecules are those allowed for cardiovascular MR scans.
Cardiovascular computed tomography
CCT has been increasingly used as a non-invasive imaging modality in CHD patients of all ages (15). Current generation multi-slice CCT scanners allow for rapid coverage of large anatomic volumes with excellent spatial and temporal resolution, overall short examination times, and low radiation exposure [10].
Wider availability and lower cost compared to CMR have also favored its success.
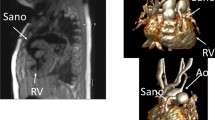
CCT is an alternative to other imaging tools, when they cannot provide good quality images, when CMR is contraindicated or present logistic difficulties such as anesthesia [11, 37]. CCT, due to its higher sub-millimetric isotropic spatial resolution (up to 0.4 × 0.4.0.6 mm for modern multirow detectors scanners), is considered superior to CMR in cardiovascular anatomy delineation (Fig. 4), primarily for small blood vessels analysis such as coronary arteries, collaterals, fistulas, and arteriovenous malformations [20]. Electrocardiographic (ECG)-synchronization is mandatory only for coronary, aortic root, and/or intracardiac imaging. Besides, CCT enables optimal depiction of airways and lung parenchyma and it is the preferred imaging modality when tracheobronchial or pulmonary abnormalities need to be investigated [5, 10, 11].
31-year-old male with a repaired ToF. CCT MPR image, RVOT sagittal plane: infundibular and pulmonary stenosis (a), note the anatomical detail of valvular cusp (arrow). 16-year-old female with a cTGA after Arterial Switch operation. CCT 3D volume rendering images demonstrate high resolution post-surgical anatomy (b), LCA reimplantation kinking and stretching (black arrow) is well depicted (c). 2-year-old child with coronary artery fistula. CCT 3D volume rendering image optimally shows the fistula (white arrows) between LAD artery and Rv chamber (d). Ao Ascending aorta, CCT Cardiovascular computed tomography, cTGA Complete transposition of the great arteries, 3D Tridimensional, D Diagonal artery, LAD Left Anterior descending artery, LCA Left coronary artery, Lv Left ventricle, MPA Main pulmonary artery, MPR Multiplanar Reformation, RPA Right pulmonary artery, Rv Right ventricle, ToF Tetralogy of Fallot
Like CMR, CCT performance is strongly dependent on scanner technology: detector number and size, gantry rotation speed, double source technology, temporal resolution (66–75 ms for dual source scanners and 140–150 ms for single source machines) and dose reduction algorithms significantly influence its accuracy and applicability, especially in newborns/infants and coronary artery imaging. Another major advantage of CCT over CMR is its shorter duration, and no need for specific equipment for sedation, particularly useful in uncooperative patients, namely younger children and critically ill subjects in the acute setting [38]. High pitch (up to 3.4 for dual source scanners) or target mode acquisition with newer generation scanners permits image acquisition in a single or few heartbeats, acquiring data during a small cardiac cycle portion (i.e., prospective scanning) [39, 40], thus drastically diminishing cardiac and respiratory motion artifacts and provide diagnostic images even at higher heart rates encountered in neonates and infants [11]. Consequently, exams performed for most indications can be obtained without sedation and even during free breathing [41, 42] while the use of beta-blockers or nitrates is reduced or eliminated unless detailed coronary artery imaging is sought. In contrast, older generation scanners may be inadequate for some indications, with an increased need for sedation and pre-medication [11].
CCT is less susceptible than CMR to metallic artifacts (Fig. 5). Although stents are not a contraindication to CMR, CCT is superior in diagnosing stent patency and integrity [43] and it is preferable when evaluating metallic devices and calcifications within conduits and vessels [3, 5]. Furthermore, CCT is not limited by implanted cardiac devices [1, 5].
34-year-old male with Mustard repair of c-TGA and loop recorder implantation. CMR SSFP 4-chamber view shows atrio-ventricular concordance and the pulmonary baffle (asterisk). The ventricular apex is canceled by artifacts (a). 18-year-old Fontan patient with pacemaker implantation. CCT axial plane displays the wires and the pacemaker generator (arrows) with minimal artifacts upon thoracic aorta (b). Adult male patient with Mustard repair of c-TGA and baffle leakage. CMR SSFP 4-chamber images show the flow turbulence (arrow) before the treatment (c) and a huge artifact (arrow) caused by the metallic closure device (d). Adult male with ASD after endovascular closure. CCT axial image well depicts the closure device without limitations to cardiac chambers visualization (e). CCT cardiovascular computed tomography, CMR cardiovascular magnetic resonance, cTGA complete transposition of the great arteries, SSFP Steady State Free Precession, La left atrium, Lv left ventricle, Ra right atrium, Rv right ventricle
CCT accurately quantifies right and left ventricular volumes and function [44, 45], although with lower temporal resolution than CMR or echocardiography and at the expense of increased radiation exposure, needing ECG-synchronized data acquisition during the whole cardiac cycle (i.e., retrospective scanning). Therefore, it should be considered with caution for serial measurements, mainly when CMR is contraindicated or degraded by artifacts. Moreover, CCT does not provide hemodynamic information [20].
Obviously, the main drawback of CCT is radiation exposure, which is considered to increase the risk of future cancer development [46]. This is especially concerning in younger patients characterized by longer expected lifespan and greater radiation sensitivity. Recent advancements in scanner technology and reconstruction methods [47] have led to low-dose CCT protocols, which are now consistently inferior to cardiac catheterization with careful selection of scan parameters [48] and can currently be employed for many CHD indications (for many of which it can reach effective dose level equal to or lower than 1 mSv), except for coronary imaging where the exposure reduction is limited by several patients and scanner features.
CCT requires intravenous administration of iodine-based contrast agents. Non-ionic low or iso-osmolar agents are used because adverse reactions are rarer and usually milder, including allergic-type and non-allergic reactions similar to GBCAs [49]. Severe kidney disease is a contraindication to injection due to the risk of contrast-induced nephropathy [50].
Comparative cost
The cost analysis of imaging modalities in CHD, specifically in children, rarely reaches adequate cost-effectiveness [51]. Overall costs often significantly exceed local reimbursements, particularly for complex examinations such as CMR under general anesthesia [52]. CMR scan costs in Western societies exceed several times those of echocardiography due to purchasing, running, and maintaining the scanner and staff expertise and training requirements. The decision to request a CMR and CCT investigation must be guided by the additional information needed, local facilities, and available resources for scanning [53]. Workflow and technical optimization (scan time, processing, and reporting) may be beneficial. In this sense, the combined use of rapid imaging, artificial intelligence algorithms [54], structured reports, and strategies to reduce the need for general anesthesia ("feed and wrap", MRI-safe audio–video systems for entertainment) could speed up the diagnostic process. On the other hand, CCT is faster and less expensive than MRI, often does not require general anesthesia, and can sometimes be preferred for its feasibility and availability, especially in clinical emergencies.
Levels of recommendation of CMR and CCT in CHD
Based on the literature and expert opinions, this consensus paper proposes a novel approach to recommendations for CMR and CCT in CHD, divided into 3 levels, according to patient's age, disease complexity, and the required imaging experience. This model appears to be more suitable in routine clinical activity due to the extreme variability of the clinical scenarios and issues addressed in CHD [5, 10, 11, 13, 19]. The levels of recommendation are summarized in Table 3.
Level 1 Adolescent/adult patients affected by simple to moderate complexity CHD that require additional cross-sectional imaging investigation with CMR and/or CCT → it refers to any center with cardiovascular imaging experience.
This level includes native or repaired CHD of simple to moderate complexity in older children/adolescents and adults (e.g., aortic coarctation, pulmonary venous return or aortic arch anatomy, shunt quantification). The choice of imaging modality and timing of execution are well defined and widely acknowledged. Since patients are stable, cooperative, and do not necessarily require the latest generation technology, which is usually only available in highly specialized centers, they could be addressed to and/or managed by any center with CMR or CCT equipment and experience [10, 11, 55].
Level 2 Cooperative patients with moderate/complex CHD that are candidate to longitudinal cross-sectional imaging evaluation with CMR and/or CCT → It refers preferably to a specialized center with experience in diagnosis and treatment of CHD.
This category includes well-established CMR and CCT indications for all simple CHDs in children (e.g., single pulmonary vein anomalies), most moderate complexity (e.g., follow-up in Tetralogy of Fallot or aortic coarctation), and some complex CHDs in both children and adults. They generally have largely accepted protocols but imaging modality choice, post-processing and timing are based on the specific information required for patient management (clinical conditions, need for sedation, and previous imaging) or on local availability and expertise. This category also includes limited cases of CHD with uncertain CMR or CCT indications.
It is recommended to coordinate these decisions with a dedicated team (Cardiologists, Cardiac Surgeons, Cardiovascular Radiologists, Anesthesiologists) committed to long-term collaboration within a referral center [10, 11, 55].
Level 3 Uncooperative patients and complex CHD proposed for “optional” and technically difficult cross-sectional imaging investigation with CMR and/or CCT in potentially unsafe scanning condition → it refers to highly specialized and equipped centers with long-standing experience in CHD imaging and treatment.
This group encompasses all situations where a high level of complexity is expected, be it because they involve very complex CHD pre/post repair/palliation (e.g., Fontan procedure), fragile patients (e.g., newborns or critical patients), technical (e.g., coronary anomalies in small children) or anesthesiologic (e.g., Williams syndrome) difficulties requiring advanced technology, highly specific protocols and experienced staff, no definite agreement on the indication, imaging modality or timing, or other selected cases.
These circumstances necessitate a multidisciplinary evaluation performed during joint meetings of experts that only a highly dedicated referral center can provide [10, 11, 55].
Recent advances and future perspective
In the last decades, the technological developments of CMR and CCT have contributed to their incremental use in CHD. Novel emerging techniques like advanced flow evaluation and reduced acquisition and post-processing times [56] could further expand their role in the near future (Fig. 6).
Overview of some of the emergent imaging applications in CHD. T2 map in a short axis basal view of the ventricles in a RV dilatation due to left-to-right shunt (a). High complexity CHD 3D printing model: supero-inferior ventricle with a complex relationship of the ventricular septal defect (b). 4D flow reconstruction of thoracic aorta: flow streamlines panoramic sagittal oblique visualization in a bicuspid aortic valve patient. Color-coding of different flow velocities (c)
Four-dimensional flow technology [57–59] allows for time-resolved 3D blood flow direction and velocity assessment in the whole heart and great vessels, rendering flow analysis in any vessel section available in a single acquisition, which is especially appealing in complex CHD. Moreover, advanced parameters such as flow energetics and wall shear stress might be particularly useful in some conditions such as aortopathies.
Tissue mapping parameters (T1,T2, T2* and extracellular volume) offer a quantitative analysis of both focal and overall diffuse myocardial alterations like fibrosis and edema [60–62]. To date their use in CHDs like repaired Tetralogy of Fallot and systemic RV is partially limited by small RV wall thickness, highly exposed to motion artifacts. Future advancements in sequences stability and acquisition speed would probably improve our knowledge of ventricular dysfunction mechanisms and arrhythmogenic risk in these patients [63, 64].
There is growing evidence that myocardial deformation is a more sensitive quantitative assessment of contractile function than ejection fraction. CMR feature tracking [65] measures deformation from simple cine images, overcoming many limitations of myocardial tagging. This opens the possibility to obtain additional diagnostic and prognostic information especially in GUCH, although further research is warranted in this field.
The enhancement of hybrid diagnostic approaches represents a further development in CHD. CMR pulmonary flow regurgitation quantification and invasive pressure measurements [66] are already performed in some centers. CMR guidance of interventional cardiac catheterization has been shown feasible and could be especially useful for electrophysiology procedures, although several issues must still be resolved [67].
A new emerging application in pediatric imaging is fetal CMR [68]. Until recently, this modality was mostly based on static anatomical images. Thanks to advances in fetal cardiac gating techniques, functional imaging is now possible. The combination of flow imaging with oxygen saturation derived from mapping measurements within large fetal vessels allows for calculation of fetal oxygen delivery, consumption, and extraction fraction, providing the only currently available non-invasive insights into fetal hemodynamics [69, 70].
3D printing-prototyping is an ideal manufacturing process for creating patient-specific anatomical models. Its use in CHD is expanding both for surgical and interventional planning and for patients' and families' education [71]. Finally, developments in artificial intelligence and machine/deep learning, including new methods such as radiomics, have a promising role in medical imaging but are still in their early stages [72].
Summary statement
Level 1 | Level 2 | Level 3 | |
|---|---|---|---|
Levels of recommendation of CMR/CCT in CHD | |||
Definition | Cooperative Adolescent/adult patients with simple/moderate CHD that require additional cross-sectional imaging investigation | Cooperative patients with moderate/complex CHD candidate to longitudinal cross-sectional imaging evaluation | Uncooperative patients and complex CHD proposed for “optional” and technically difficult cross-sectional imaging investigation in potentially unsafe scanning condition |
Refer to | Any center with cardiovascular imaging experience | Specialized center with experience in the diagnosis and treatment of CHD | Tertiary center with long-standing experience in the diagnosis and treatment of CHD |
References
Baumgartner H, De Backer J, Babu-Narayan SV et al (2021) 2020 ESC Guidelines for the management of adult congenital heart disease. Eur Heart J 42:563–645. https://doi.org/10.1093/eurheartj/ehaa554
Ntsinjana HN, Hughes ML, Taylor AM (2011) The role of cardiovascular magnetic resonance in pediatric congenital heart disease. J. Cardiovasc Magn Reson 13:66
Orwat S, Diller G-P, Baumgartner H (2014) Imaging of congenital heart disease in adults: choice of modalities. Eur Hear J Cardiovasc Imaging 15:6–17. https://doi.org/10.1093/ehjci/jet124
Tworetzky W, McElhinney DB, Brook MM et al (1999) Echocardiographic diagnosis alone for the complete repair of major congenital heart defects. J Am Coll Cardiol 33:228–233. https://doi.org/10.1016/S0735-1097(98)00518-X
Di Salvo G, Miller O, Babu Narayan S et al (2018) Imaging the adult with congenital heart disease: a multimodality imaging approach—position paper from the EACVI. Eur Heart J Cardiovasc Imaging 19:1077–1098. https://doi.org/10.1093/ehjci/jey102
Higgins CB, Byrd BF, Farmer DW et al (1984) Magnetic resonance imaging in patients with congenital heart disease. Circulation 70:851–860. https://doi.org/10.1161/01.CIR.70.5.851
Sechtem U, Neubauer S, Revel D, De Roos A, Van Rossum A, Von Schulthess G, Sieverding L, Van Der Wall E (1998) The clinical role of magnetic resonance in cardiovascular disease. Task Force of the European Society of Cardiology, in collaboration with the Association of European Paediatric Cardiologists. Eur Heart J 19:19–39
Pontone G, Di Cesare E, Castelletti S et al (2021) Appropriate use criteria for cardiovascular magnetic resonance imaging (CMR): SIC—SIRM position paper part 1 (ischemic and congenital heart diseases, cardio-oncology, cardiac masses and heart transplant). Radiol Medica 126:365–379. https://doi.org/10.1007/s11547-020-01332-6
Valsangiacomo Buechel ER, Grosse-Wortmann L, Fratz S et al (2015) Indications for cardiovascular magnetic resonance in children with congenital and acquired heart disease: an expert consensus paper of the Imaging Working Group of the AEPC and the Cardiovascular Magnetic Resonance Section of the EACVI. Eur Heart J Cardiovasc Imaging 16:281–297. https://doi.org/10.1093/ehjci/jeu129
Han BK, Rigsby CK, Hlavacek A et al (2015) Computed tomography imaging in patients with congenital heart disease part I: rationale and utility. An expert consensus document of the Society of Cardiovascular Computed Tomography (SCCT). Endorsed by the Society of Pediatric Radiology (SPR) and the Nor. J Cardiovasc Comput Tomogr 9:475–492. https://doi.org/10.1016/j.jcct.2015.07.004
Han BK, Rigsby CK, Leipsic J et al (2015) Computed tomography imaging in patients with congenital heart disease, part 2: technical recommendations. An expert consensus document of the Society of Cardiovascular Computed Tomography (SCCT). Endorsed by the Society of Pediatric Radiology (SPR) and th. J Cardiovasc Comput Tomogr 9:493–513. https://doi.org/10.1016/j.jcct.2015.07.007
Warnes CA, Williams RG, Bashore TM et al (2008) ACC/AHA 2008 guidelines for the management of adults with congenital heart disease: executive summary—a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines (writing committee to develop guidelines fo. Circulation 118:2395–2451
Stout KK, Daniels CJ, Aboulhosn JA et al (2019) 2018 AHA/ACC guideline for the management of adults with congenital heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines. Circulation 139:e637–e697. https://doi.org/10.1161/CIR.0000000000000602
Sachdeva R, Valente AM, Armstrong AK et al (2020) ACC/AHA/ASE/HRS/ISACHD/SCAI/SCCT/SCMR/SOPE 2020 appropriate use criteria for multimodality imaging during the follow-up care of patients with congenital heart disease. J Am Coll Cardiol 75:657–703. https://doi.org/10.1016/j.jacc.2019.10.002
Esposito A, Francone M, Andreini D et al (2021) SIRM-SIC appropriateness criteria for the use of Cardiac Computed Tomography. Part 1: Congenital heart diseases, primary prevention, risk assessment before surgery, suspected CAD in symptomatic patients, plaque and epicardial adipose tissue characterization, and functional assessment of stenosis. Radiol Med 126(9):1236–1248. https://doi.org/10.1007/s11547-021-01378-0
Luijnenburg SE, Robbers-Visser D, Moelker A et al (2010) Intra-observer and interobserver variability of biventricular function, volumes and mass in patients with congenital heart disease measured by CMR imaging. Int J Cardiovasc Imaging 26:57–64. https://doi.org/10.1007/s10554-009-9501-y
Sharma A, Einstein AJ, Vallakati A et al (2014) Meta-analysis of global left ventricular function comparing multidetector computed tomography with cardiac magnetic resonance imaging. Am J Cardiol 113:731–738. https://doi.org/10.1016/j.amjcard.2013.11.016
Kilner PJ, Geva T, Kaemmerer H, Trindade PT, Schwitter J, Webb GD (2010) Recommendations for cardiovascular magnetic resonance in adults with congenital heart disease from the respective working groups of the European Society of Cardiology. Eur Heart J 31:794–805. https://doi.org/10.1093/eurheartj/ehp586
Babu-Narayan SV, Giannakoulas G, Valente AM et al (2016) Imaging of congenital heart disease in adults. Eur Heart J 37:1182–1195. https://doi.org/10.1093/eurheartj/ehv519
Powell AJ, Geva T (2000) Blood flow measurement by magnetic resonance imaging in congenital heart disease. Pediatr Cardiol 21:47–58
Fratz S, Chung T, Greil GF et al (2013) Guidelines and protocols for cardiovascular magnetic resonance in children and adults with congenital heart disease: SCMR expert consensus group on congenital heart disease. J Cardiovasc Magn Reson 15:66. https://doi.org/10.1186/1532-429X-15-51
Picano E, Vañó E, Rehani MM et al (2014) The appropriate and justified use of medical radiation in cardiovascular imaging: a position document of the ESC Associations of Cardiovascular Imaging, Percutaneous Cardiovascular Interventions and Electrophysiology. Eur Heart J 35:665–672. https://doi.org/10.1093/eurheartj/eht394
Knuuti J, Bengel F, Bax JJ et al (2014) Risks and benefits of cardiac imaging: an analysis of risks related to imaging for coronary artery disease. Eur Heart J 35:633–638. https://doi.org/10.1093/eurheartj/eht512
Sasaki T, Hansford R, Zviman MM et al (2011) Quantitative assessment of artifacts on cardiac magnetic resonance imaging of patients with pacemakers and implantable cardioverter-defibrillators. Circ Cardiovasc Imaging 4:662–670. https://doi.org/10.1161/CIRCIMAGING.111.965764
Garg R, Powell AJ, Sena L et al (2005) Effects of metallic implants on magnetic resonance imaging evaluation of Fontan palliation. Am J Cardiol 95:688–691. https://doi.org/10.1016/j.amjcard.2004.10.053
Nazarian S, Hansford R, Rahsepar AA et al (2017) Safety of magnetic resonance imaging in patients with cardiac devices. N Engl J Med 377:2555–2564. https://doi.org/10.1056/NEJMoa1604267
Nazarian S, Beinart R, Halperin HR (2013) Magnetic resonance imaging and implantable devices. Circ Arrhythmia Electrophysiol 6:419–428. https://doi.org/10.1161/CIRCEP.113.000116
Salerno M, Sharif B, Arheden H et al (2017) Recent advances in cardiovascular magnetic resonance. Circ Cardiovasc Imaging 10:e003951. https://doi.org/10.1161/CIRCIMAGING.116.003951
Banka P, Geva T (2016) Advances in pediatric cardiac MRI. Curr Opin Pediatr 28:575–583. https://doi.org/10.1097/MOP.0000000000000400
Odegard KC, Dinardo JA, Tsai-Goodman B et al (2004) Anaesthesia considerations for cardiac MRI in infants and small children. Paediatr Anaesth 14:471–476. https://doi.org/10.1111/j.1460-9592.2004.01221.x
Stockton E, Hughes M, Broadhead M et al (2012) A prospective audit of safety issues associated with general anesthesia for pediatric cardiac magnetic resonance imaging. Paediatr Anaesth 22:1087–1093. https://doi.org/10.1111/j.1460-9592.2012.03833.x
Gottlieb EA, Andropoulos DB (2013) Anesthesia for the patient with congenital heart disease presenting for noncardiac surgery. Curr Opin Anaesthesiol 26:318–326. https://doi.org/10.1097/ACO.0b013e328360c50b
Matisoff AJ, Olivieri L, Schwartz JM, Deutsch N (2015) Risk assessment and anesthetic management of patients with Williams syndrome: a comprehensive review. Paediatr Anaesth 25:1207–1215. https://doi.org/10.1111/pan.12775
Dillman JR, Ellis JH, Cohan RH et al (2007) Frequency and severity of acute allergic-like reactions to gadolinium-containing IV contrast media in children and adults. Am J Roentgenol 189:1533–1538. https://doi.org/10.2214/AJR.07.2554
Rozenfeld MN, Podberesky DJ (2018) Gadolinium-based contrast agents in children. Pediatr Radiol 48:1188–1196. https://doi.org/10.1007/s00247-018-4165-1
Chan FP (2008) MR and CT imaging of the pediatric patient with structural heart disease. Semin Thorac Cardiovasc Surg 20:393–399. https://doi.org/10.1053/j.semtcvs.2008.11.006
Mangal R, Jain A, Amin P et al (2018) Role of multi-detector computed tomography in congenital heart diseases. Int J Res Med Sci 6:3423. https://doi.org/10.18203/2320-6012.ijrms20184058
Secinaro A, Curione D, Mortensen KH et al (2019) Dual-source computed tomography coronary artery imaging in children. Pediatr Radiol 49:1823–1839. https://doi.org/10.1007/s00247-019-04494-2
Jadhav SP, Golriz F, Atweh LA et al (2015) CT angiography of neonates and infants: comparison of radiation dose and image quality of target mode prospectively ECG-gated 320-MDCT and ungated helical 64-MDCT. AJR Am J Roentgenol 204:W184–W191. https://doi.org/10.2214/AJR.14.12846
Han BK, Overman DM, Grant K et al (2013) Non-sedated, free breathing cardiac CT for evaluation of complex congenital heart disease in neonates. J Cardiovasc Comput Tomogr 7:354–360. https://doi.org/10.1016/j.jcct.2013.11.006
Lell MM, May M, Deak P et al (2011) High-pitch spiral computed tomography: effect on image quality and radiation dose in pediatric chest computed tomography. Invest Radiol 46:116–123. https://doi.org/10.1097/RLI.0b013e3181f33b1d
Nordmeyer J, Gaudin R, Tann OR et al (2010) MRI may be sufficient for noninvasive assessment of great vessel stents: an in vitro comparison of MRI, CT, and conventional angiography. Am J Roentgenol 195:865–871. https://doi.org/10.2214/AJR.09.4166
Rigolli M, Anandabaskaran S, Christiansen JP, Whalley GA (2016) Bias associated with left ventricular quantification by multimodality imaging: a systematic review and meta-analysis. Open Hear 3:e000388. https://doi.org/10.1136/openhrt-2015-000388
Raman SV, Shah M, McCarthy B et al (2006) Multi-detector row cardiac computed tomography accurately quantifies right and left ventricular size and function compared with cardiac magnetic resonance. Am Heart J 151:736–744. https://doi.org/10.1016/j.ahj.2005.04.029
Einstein AJ, Henzlova MJ, Rajagopalan S (2007) Estimating risk of cancer associated with radiation exposure from 64-slice computed tomography coronary angiography. J Am Med Assoc 298:317–323. https://doi.org/10.1001/jama.298.3.317
Dougeni E, Faulkner K, Panayiotakis G (2012) A review of patient dose and optimisation methods in adult and paediatric CT scanning. Eur J Radiol 81:e665–e683. https://doi.org/10.1016/j.ejrad.2011.05.025
Gherardi GG, Iball GR, Darby MJ, Thomson JDR (2011) Cardiac computed tomography and conventional angiography in the diagnosis of congenital cardiac disease in children: recent trends and radiation doses. Cardiol Young 21:616–622. https://doi.org/10.1017/S1047951111000485
Han BK, Lindberg J, Grant K et al (2011) Accuracy and safety of high pitch computed tomography imaging in young children with complex congenital heart disease. Am J Cardiol 107:1541–1546. https://doi.org/10.1016/j.amjcard.2011.01.065
Beckett KR, Moriarity AK, Langer JM (2015) Safe use of contrast media: what the radiologist needs to know. Radiographics 35:1738–1750. https://doi.org/10.1148/rg.2015150033
van der Molen AJ, Reimer P, Dekkers IA et al (2018) Post-contrast acute kidney injury—Part 1: definition, clinical features, incidence, role of contrast medium and risk factors: recommendations for updated ESUR Contrast Medium Safety Committee guidelines. Eur Radiol 28:2845–2855. https://doi.org/10.1007/s00330-017-5246-5
Prakash A, Powell AJ, Geva T (2010) Multimodality Noninvasive Imaging for Assessment of Congenital Heart Disease. Adv Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.109.875021
Tomà P, Magistrelli A, Secinaro A et al (2020) Sustainability of paediatric radiology in Italy. Pediatr Radiol 6:66
Francis SA, Daly C, Heydari B et al (2013) Cost-effectiveness analysis for imaging techniques with a focus on cardiovascular magnetic resonance. J Cardiovasc Magn Reson 15:52
Petersen SE, Abdulkareem M, Leiner T (2019) Artificial intelligence will transform cardiac imaging—opportunities and challenges. Front Cardiovasc Med 6:133. https://doi.org/10.3389/fcvm.2019.00133
Kilner PJ (2011) The role of cardiovascular magnetic resonance in adults with congenital heart disease. Prog Cardiovasc Dis 54:295–304. https://doi.org/10.1016/j.pcad.2011.07.006
Friedrich MG (2017) The future of cardiovascular magnetic resonance imaging. Eur Heart J 38:1698–1701. https://doi.org/10.1093/eurheartj/ehx243
Zhong L, Schrauben EM, Garcia J et al (2019) Intracardiac 4D flow MRI in congenital heart disease: recommendations on behalf of the ISMRM Flow & Motion Study Group. J Magn Reson Imaging 50:677–681. https://doi.org/10.1002/jmri.26858
Azarine A, Garçon P, Stansal A et al (2019) Four-dimensional flow MRI: principles and cardiovascular applications. Radiographics 39:632–648. https://doi.org/10.1148/rg.2019180091
Vasanawala SS, Hanneman K, Alley MT, Hsiao A (2015) Congenital heart disease assessment with 4D flow MRI. J Magn Reson Imaging 42:870–886. https://doi.org/10.1002/jmri.24856
Ferreira VM, Piechnik SK, Robson MD et al (2014) Myocardial tissue characterization by magnetic resonance imaging: novel applications of T1 and T2 mapping. J Thorac Imaging 29:147–154. https://doi.org/10.1097/RTI.0000000000000077
Salerno M, Kramer CM (2013) Advances in parametric mapping with CMR imaging. JACC Cardiovasc Imaging 6:806–822. https://doi.org/10.1016/j.jcmg.2013.05.005
Messroghli DR, Moon JC, Ferreira VM et al (2017) Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2 and extracellular volume: a consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imagin. J Cardiovasc Magn Reson 19:75. https://doi.org/10.1186/s12968-017-0389-8
Riesenkampff E, Messroghli DR, Redington AN, Grosse-Wortmann L (2015) Myocardial T1 mapping in pediatric and congenital heart disease. Circ Cardiovasc Imaging 8:e002504. https://doi.org/10.1161/CIRCIMAGING.114.002504
Broberg CS, Burchill LJ (2015) Myocardial factor revisited: the importance of myocardial fibrosis in adults with congenital heart disease. Int J Cardiol 189:204–210. https://doi.org/10.1016/j.ijcard.2015.04.064
Dardeer AM, Hudsmith L, Wesolowski R et al (2018) The potential role of feature tracking in adult congenital heart disease: advantages and disadvantages in measuring myocardial deformation by cardiovascular magnetic resonance. J Congenit Cardiol 2:3. https://doi.org/10.1186/s40949-018-0015-0
Muthurangu V, Taylor A, Andriantsimiavona R, Hegde S, Miquel ME, Tulloh R, Baker E, Hill DL, Razavi RS (2004) Novel method of quantifying pulmonary vascular resistance by use of simultaneous invasive pressure monitoring and phase-contrast magnetic resonance flow. Circulation 110:826–834. https://doi.org/10.1161/01.CIR.0000138741.72946.84
Rhode KS, Sermesant M, Brogan D et al (2005) A system for real-time XMR guided cardiovascular intervention. IEEE Trans Med Imaging 24:1428–1440. https://doi.org/10.1109/TMI.2005.856731
Roy CW, Van Amerom JFP, Marini D et al (2019) Fetal cardiac MRI: a review of technical advancements. Top Magn Reson Imaging 28:235–244. https://doi.org/10.1097/RMR.0000000000000218
Portnoy S, Seed M, Sled JG, Macgowan CK (2017) Non-invasive evaluation of blood oxygen saturation and hematocrit from T1 and T2 relaxation times: In-vitro validation in fetal blood. Magn Reson Med 78:2352–2359. https://doi.org/10.1002/mrm.26599
Rodríguez-Soto AE, Langham MC, Abdulmalik O et al (2018) MRI quantification of human fetal O2 delivery rate in the second and third trimesters of pregnancy. Magn Reson Med 80:1148–1157. https://doi.org/10.1002/mrm.27094
Valverde I, Gomez-Ciriza G, Hussain T et al (2017) Three-dimensional printed models for surgical planning of complex congenital heart defects: an international multicentre study. Eur J Cardio-thoracic Surg 52:1139–1148. https://doi.org/10.1093/EJCTS/EZX208
Nicol ED, Norgaard BL, Blanke P et al (2019) The future of cardiovascular computed tomography: advanced analytics and clinical insights. JACC Cardiovasc Imaging 12:1058–1072. https://doi.org/10.1016/j.jcmg.2018.11.037
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design and to the writing of the first draft of the manuscript. All authors commented or contributed to the revision of previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare they have no relevant financial or non-financial interests as to the content of this paper to disclose.
Ethical standards
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Secinaro, A., Ait-Ali, L., Curione, D. et al. Recommendations for cardiovascular magnetic resonance and computed tomography in congenital heart disease: a consensus paper from the CMR/CCT working group of the Italian Society of Pediatric Cardiology (SICP) and the Italian College of Cardiac Radiology endorsed by the Italian Society of Medical and Interventional Radiology (SIRM) Part I. Radiol med 127, 788–802 (2022). https://doi.org/10.1007/s11547-022-01490-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-022-01490-9