Abstract
Objective
To describe the clinical and ultrasonography (US) findings of soft tissue hemangiomas, and to compare with the results of histologic diagnosis after US-guided biopsy.
Method and materials
We retrospectively studied the files of 97 patients (48 female, 49 male; mean age, 34 years; range 4–84 years) with soft tissue hemangiomas diagnosed from 2004 to 2011. Mean follow-up was 9 years (range 7–13 years). Clinical presentation included intermittent mild pain associated with a soft tissue swelling/palpable mass in all patients, chronic pain and increased local heat in 29 patients, local swelling and decreased range of motion of the adjacent joint in 45 patients, and all the above symptoms in 23 patients. B-mode and color Doppler US evaluation included the site, location, size, shape, margins, presence of calcifications, echo structure and echogenicity. All patients had US-guided biopsy for histologic analysis.
Results
US-guided biopsy and histology confirmed the diagnosis of soft tissue hemangioma in 92 of the 97 lesions (94.8%). Histologic examination of the remaining five lesions showed nodular fasciitis (two lesions), endometriosis (one lesion), hemangioendothelioma (two lesions); US of these lesions showed variable size, irregular margins, and deep-seated location. Histologically documented soft tissue hemangiomas were most commonly superficial (74 lesions) and arteriovenous (45 lesions). Shape was most commonly oval (fusiform), margins were most commonly not well defined (irregular, hazing but circumscribed), phleboliths were more common in deep-seated lesions, echo structure was heterogeneous, and echogenicity was most commonly hyperechogen and involuting.
Conclusion
Clinical presentation and typical B-mode and color Doppler US findings are adequate for the diagnosis of soft tissue hemangiomas without the need for biopsy and histologic analysis. If any clinical or US doubt, an US-guided biopsy should be performed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soft tissue hemangiomas are benign soft tissue tumors that closely resemble normal vessels [1]. They account for approximately 7% of all benign soft tissue tumors, may arise at various anatomic locations, and are much more commonly superficial than deep-seated [1,2,3,4]. Diagnosis is usually easy for typical superficial lesions; however, atypical and deep-seated hemangiomas cannot be distinguished from malignant soft-tissue tumors without appropriate imaging studies [5,6,7,8].
The imaging modality of choice for the diagnosis of soft tissue hemangiomas is magnetic resonance (MR) imaging. Previous studies described the MR imaging features of hemangiomas [9,10,11,12], and indicated that hemangiomas may be correctly diagnosed and distinguished from malignant soft tissue tumors by their characteristic MR imaging appearances without the need for biopsy [8, 13,14,15]. We concur with these reports. However, soft tissue hemangiomas most commonly show typical clinical and ultrasonography (US) findings that may allow for an accurate and easy diagnosis [1,2,3,4, 16,17,18,19,20,21,22,23,24]. If the typical clinical and US signs are present, then, the diagnosis of soft tissue hemangiomas may be obtained. If any uncertainty, biopsy is required for histologic diagnosis. In this scenario, US can be used successfully in the same setting to guide biopsy for tissue sampling [21,22,23,24].
The purpose of this study is to describe the clinical and US findings of soft tissue hemangiomas, and to compare with the results of histologic diagnosis. Our hypothesis was that US is appropriate as a single diagnostic method for these tumors without the need for a biopsy.
Materials and methods
We performed an observational study on 97 patients with soft tissue hemangiomas admitted and treated at the authors’ institution from 2004 to 2011. The medical files of these patients were retrospectively reviewed. There were 48 female and 49 male patients with a mean age of 34 years (range 4–84 years). The mean follow-up was 9 years (range 7–13 years). No patient was lost to follow-up; all patients or their relatives gave written informed consent for their data to be included in this study.
Clinical findings at presentation included (1) intermittent mild pain associated with a soft tissue swelling/palpable mass in all patients, (2) chronic pain and increased local heat in 29 patients, (3) local swelling and decreased range of motion of the adjacent joint in 45 patients, and (4) all the above symptoms in 23 patients. All patients had B-mode and power (color) Doppler US examination of their soft tissue lesions. No patient of these series had an MR imaging examination; in all these patients the diagnosis was assumed based on typical clinical and US findings, as previously described [1,2,3,4, 20,21,22,23,24,25,26,27]. US examination was done using a Acuson Antares Premium US device (Siemens Ultrasound, Mountain View, CA) from 2004 to 2009, and a Hi Vision™ Preirus™ device (Hitachi Medical Corporation, Japan) from 2009 to 2011 with a multifrequency (5–12 MHz) linear array transducer. The lesions were studied with a standard US protocol using orthogonal scans along the axis of the lesion, color and power Doppler. The B-mode US protocol evaluated the site and location (superficial, 74 lesions; deep-seated, 23 lesions), size, shape, margins, presence of calcifications, echo structure (homogenous or heterogeneous), and echogenicity (hyper, iso or hypo-echogenicity). The vascularity and flow were evaluated in power Doppler US (PDUS) with scanning the entire lesion and setting the parameters at the lowest Doppler gain to prevent aliasing. Parameters included (1) repetition pulse between 0.650 and 3.650 kHz (average ~ 2 kHz), (2) registration from the area of high vascularity, (3) pulsed Doppler measurement of the area with color flow signal Doppler shifts, and (4) presence of flow. US findings were recorded and evaluated by four senior radiologists (ER, DV, UA, and PP) with absolute homophony for their diagnosis. Based on the clinical and US findings, as reported above, a diagnosis of soft tissue hemangioma was made in all patients.
After US examination, all patients had US-guided biopsy of their lesions using a 14-gauge, 100–150 mm biopsy needle (Biopsy Bell, Mirandola, MO, Italy); biopsies in adults were done under local anesthesia (2% mepivacaine, 6–8 ml), while in children were done under general anesthesia. A direct approach to the tumor was used in all cases after consultation with orthopaedic oncology surgeons (AFM and CE) so that the biopsy tract was put in line with the incision of an anticipated surgical excision of the tumor. Tissue samples were sent for histologic analysis.
Results
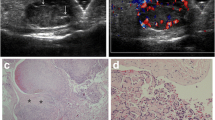
Combined B-mode US and PDUS provided for the diagnosis of a soft tissue hemangioma in 92 of the 97 lesions (94.8%). US-guided biopsy and histology confirmed the diagnosis of soft tissue hemangioma in these lesions (Fig. 1). The sensitivity and specificity of combined B-mode US and PDUS was 100% (95% CI: 96.07–100.00%) and 50% (95% CI: 18.71–81.29%), respectively. Histologic examination of the remaining five lesions showed nodular fasciitis (two lesions, forearm) (Fig. 2), endometriosis (one lesion, abdomen) (Fig. 3), hemangioendothelioma (one lesion each, thigh and leg) (Fig. 4). US of these lesions showed variable size, irregular margins, and deep-seated location.
a B-mode US shows a deep-seated solid lesion with heterogeneous echo structure, mainly hypoechoic with well-defined margins. b PDUS shows vascularity of the lesion with some vacuum anechoic areas. US findings were suggestive of a soft tissue hemangioma but US-guided biopsy and histologic examination showed endometriosis
Histologically documented soft tissue hemangiomas were most commonly superficial (74 lesions) compared to deep-seated (18 lesions). The most common type of hemangiomas was arteriovenous (45 lesions), followed by cavernous intramuscular (37 lesions), and capillary (10 lesions) (Table 1). Shape was most commonly oval (fusiform), margins were most commonly not well defined (irregular, hazing but circumscribed), echo structure was heterogeneous, and echogenicity was most commonly hyperechogen (presence of fat) and involuting (Table 2). No US differences were observed between superficial and deep-seated hemangiomas with respect to site, size, shape, margins, echo structure and echogenicity; phleboliths were more common in deep-seated lesions (11 lesions) compared to superficial lesions (2 lesions). US-guided biopsy-related complications were not observed in any of the patients in this series.
Discussion
The diagnosis of soft tissue hemangioma is based on clinical history, physical examination, symptoms, and imaging findings [8,9,10,11,12,13,14,15,16,17,18,19,20]. Typical clinical findings include a smooth and soft palpable mass, particularly superficial, painless or painful during activity, slow-growing or constant in time [1,2,3,4]. Typical US findings include small size (usually less than 5 cm), subcutaneous location, heterogeneous echotexture with multiple regular cystic or tubular spaces and echogenicity depending on the prevalent tissue in the lesion (most commonly hyperechogenicity due to presence of fat), hazing margins (not completely well-defined), intralesional regular vascular formations of different caliber and regularly distributed, calcifications (phleboliths), good compressibility, increased flow signal during compression, tubular and cystic structures [4, 20,21,22,23,24]. A high vessel density (> 5/cm2) and a high Doppler shift (> 2 kHz) has been associated with a sensitivity of 84% and a specificity of 98% for the diagnosis of hemangiomas [20]. The above clinical and US findings strongly suggest the diagnosis of soft tissue hemangiomas. Additionally, US has several advantages for the diagnosis of soft tissue hemangiomas; it is a real time, easy, low cost, bedside imaging technique. Most important, it is based on non-ionizing radiation, therefore, it can be repeated as many times as necessary, and can be used to guide biopsy [16,17,18,19,20]. Based on the results of the present series, the diagnostic accuracy of US for soft tissue hemangiomas is high (94.8%); therefore, US diagnosis of soft tissue hemangiomas should be considered accurate without the need for biopsy and histologic analysis. If necessary, US-guided biopsy is safe and successful for tissue sampling for histologic analysis.
We see two limitations in this study. First, this study is retrospective and was based on observational findings. Although prospective studies provide powerful results and conclusions, retrospective studies are often useful to evaluate a diagnostic and treatment approach. We concur that the present study did not evaluate a control group but was based on observational (clinical and imaging) findings of a single center physicians. In this regard, our results could be regarded with caution. However, the observed high diagnostic accuracy and the findings based on the experience of a large tertiary tumor center as the authors’ own center increase the power of our analysis and conclusions. Second, the study lacks a formal statistical analysis. We also acknowledge this limitation. However, we did not compare to a control group of patients with soft tissue tumors to evaluate the diagnostic accuracy of US, but we shorted our analysis only in patients with the clinical diagnosis of soft tissue hemangiomas to evaluate in these patients the diagnostic accuracy of US. We believe that in this setting, a statistical analysis is not applicable in the present study.
Typical US findings of soft tissue hemangiomas were observed in the patients included in this series using combined B-mode US and PDUS [16, 25,26,27]. B-mode US provided for evaluation of the site, size, margins and echostructure (liquid or mixed) of the lesion, and presence of plebolithes and any vascular structures. These features were strongly suggestive for a vascular lesion. PDUS provided for imaging of the vascular spaces of the lesion, therefore, confirming the diagnosis of a hemangioma. Shape of the lesions was most commonly oval/fusiform, mean longitudinal diameter was 5 cm, lesions were most commonly superficial, margins were most commonly not well-defined, and phleboliths were rare, most commonly in deep-seated lesions. Echogenic pattern depends on the presence of fat, muscle and fibrous tissue; presence of fat shows a hyperechogenic pattern, while presence of muscle and fibrous tissues shows a mixed hypo-iso echogenic pattern. Hypoechogenic tubular or cystic/alveolar formations with good compressibility and light attenuation are common because of intralesional vascular formations [4, 16, 18,19,20, 26, 28, 29]. Doppler US shows a flow signal that confirms the vascular nature of these lesions; however, a positive Doppler is not typical of hemangiomas. B-mode US always shows a complex heterogeneous echo structure. Heterogeneity shows a prevalent hyperechogenic pattern with a prominent fatty component, while when a mixed fatty, muscular and fibrous component is present the echo structure is not hyperechogenic but mixed (hyper-hypo-iso) echogenic [4, 16, 18,19,20, 26, 28, 29]. In the present series, most soft tissue hemangiomas (69%) were hyperechogenic with a heterogeneous echo structure secondary to the presence of fat (most common), muscle, fibrous tissue, and tubular or cystic regular or irregular vascular formations. There was always some compressibility and most lesions showed increased color flow and a high flow pattern with an arterial and venous component flow signal, modified during both systole and diastole, in power and color Doppler. Doppler shift of > 2 kHz, low resistance and high vessels density (> 5 vessels/cm2) were a typical pattern in our series, as in the related literature [16, 18,19,20].
Careful imaging is necessary for the evaluation of soft tissue hemangiomas [4, 30,31,32]. US and MR imaging should be the initial choice and in follow-up studies. Once clinically examined, US assessment of the lesion should differentiate slow- and fast-moving circulation. Doppler US differentiates arteriovenous malformations from other vascular lesions such as venous-malformations and capillary hemangiomas, and evaluates size, volume and vascular pattern of the lesion. Additionally, US provides guidance for preoperative needle biopsy, perioperative guidance for lesion resection by localizing leading margin, and guidance for needle placement during minimally invasive treatments such as percutaneous sclerosis [31]. It has been reported that US depiction of abundant low-flow vascular channels is a predictor for the potential success of percutaneous sclerosis [31, 32]. MR imaging provides for better demonstration of the extent of lesion, volume of the affected area, and surrounding structures [20, 33]. Although MR vascular imaging techniques provide information regarding the feeder vessels, they lack detailed information required for vascular intervention. Computed tomography (CT) angiography provides comprehensive information on long-standing lesions, lesions in adolescent and adult patients, and lesions involving osseous structures [31]. Careful imaging is also necessary for the differential diagnosis of soft tissue hemangiomas from other benign soft tissue lesions and sarcomas. The differential diagnosis should include venous malformations, arteriovenous malformations, lymphatic malformations, rapidly involuting congenital hemangioma, noninvoluting congenital hemangioma, capillary malformation, tufted angioma, Kaposiform hemangioendothelioma, and fibrosarcoma [34,35,36,37,38,39]. Doppler US of malformations shows sponge-like anechoic vessels with arterial and/or venous wave form, or a poorly defined collection of cystic spaces with a high mean restrictive index. Kaposiform hemangioendothelioma usually shows moderate vascular density, while fibrosarcoma is largely avascular [34,35,36,37,38,39]. US is the first line modality for most cases. MR imaging may be considered as first line modality in superficial lesions with atypical clinical presentation, deep lesions difficult to evaluate with clinical examination, large lesions, complex combined vascular malformations, and to evaluate the deep extent in areas difficult to evaluate with US.
Continuous dull pain, large deep-seated lesions, increase in size during time, irregular infiltrating margins, highly heterogeneous echo structure, and irregular mixed tubular and alveolar vascular formations are clinical and US findings that should increase the awareness of the treating physician for another diagnosis than a hemangioma. In these patients, a biopsy, preferably US-guided, should be performed [21,22,23,24]. In the present series, the lesions clinically and US misdiagnosed as soft tissue hemangiomas appeared like solid formations with a heterogeneous echo structure and a mainly hypoechogenic pattern. All were deep-seated, with a size of > 5 cm, and an oval or round shape. The margins were well defined in the benign lesions, and poorly defined in the hemangioendotheliomas. Intralesionally, all lesions showed alveolar (endometriosis) or tubular (fasciitis, hemangioendotelioma and endometriosis) vascular structures of different caliber.
In conclusion, clinical presentation and typical US findings are adequate for the diagnosis of soft tissue hemangiomas without the need for a biopsy and histologic analysis. US shows the size and site, echo structure and echogenicity, compressibility and calcifications with a sensitivity of 94.8%. Therefore, we consider biopsy and histologic analysis not necessary for the diagnosis of soft tissue hemangiomas. If any clinical or US doubt, a biopsy for histologic diagnosis should be performed. In these cases, biopsy should be US-guided.
References
Campanacci M (1999) Bone and soft tissue tumors: clinical features, imaging, pathology and treatment, 2nd edn. Piccin-Springer-Verlag, Wien, pp 215–283
Weiss SW GJ, Enzinger FM (2008) Bening tumors and tumor-like lesions of blood vessels. In: Enzinger and Weiss’ soft tissue tumors, 5th edn. Elsevier, Inc., Mosby, pp 837–890
Mulliken JB, Glowacki J (1982) Hemangiomas and vascular malformations in infants and children: a classification based on endothelial characteristics. Plast Reconstr Surg 69(3):412–422
Donnelly LF, Adams DM, Bisset GS (2000) Vascular malformations and hemangiomas: a practical approach in a multidisciplinary clinic. AJR Am J Roentgenol 174(3):597–608
Resnick D, Niwayama G (1995) Diagnosis of bone and joint disorders, 3rd edn. Saunders, Philadelphia, pp 4491–4498
Pierucci A, Teixeira P, Zimmermann V et al (2013) Tumours and pseudotumours of the soft tissue in adults: perspectives and current role of sonography. Diagn Interv Imaging 94(3):238–254
Greenspan A, Azouz EM, Matthews J 2nd, Décarie JC (1995) Synovial hemangioma: imaging features in eight histologically proven cases, review of the literature, and differential diagnosis. Skeletal Radiol 24(8):583–590
Teo EL, Strouse PJ, Hernandez RJ (2000) MR imaging differentiation of soft-tissue hemangiomas from malignant soft-tissue masses. AJR Am J Roentgenol 174(6):1623–1628
Suh JS, Hwang G, Hahn SB (1994) Soft-tissue hemangiomas: MR manifestations in 23 patients. Skeletal Radiol 23:621–625
Buetow DC, Kransdorf MJ, Moser RP Jr et al (1990) Radiological appearance of intramuscular hemangioma with emphasis on MR imaging. AJR 154:563–567
Kaplan PA, Williams SM (1987) Mucocutaneous and peripheral soft-tissue hemangiomas: MR imaging. Radiology 163:163–166
Jenner G, Soderlund V, Bauer HFC, Brosjo O (1996) MR imaging of skeletal muscle hemangiomas. Acta Radiol 37:140–144
Berquist TH, Ehman RL, King BF et al (1990) Value of MR imaging in differentiating benign from malignant soft-tissue masses: study of 95 lesions. AJR 155:1251–1255
Crim JR, Seeger LL, Yao L et al (1992) Diagnosis of soft-tissue masses with MR imaging: can benign masses be differentiated from malignant ones? Radiology 185:581–586
Moulton JS, Blebea JS, Dunco DM et al (1995) MR imaging of softtissue masses: diagnostic efficacy and value of distinguishing between benign and malignant lesions. AJR 164:1191–1199
Murphey MD, Fairbairn KJ, Parman LM et al (1995) From the archives of the AFIP. Musculoskeletal angiomatous lesions: radiologic–pathologic correlation. Radiographics 15(4):893–917
Chiou HJ, Chou YH, Chiu SY et al (2009) Differentiation of benign and malignant superficial soft-tissue masses using grayscale and color Doppler ultrasonography. J Chin Med Assoc 72(6):307–315
Paltiel HJ, Burrows PE, Kozakewich HP et al (2000) Soft-tissue vascular anomalies: utility of US for diagnosis. Radiology 214(3):747–754
Yang WT, Ahuja A, Metreweli C (1997) Sonographic features of head and neck hemangiomas and vascular malformations: review of 23 patients. J Ultrasound Med 16(1):39–44
Dubois J, Garel L, Grignon A et al (1998) Imaging of hemangiomas and vascular malformations in children. Acad Radiol 5(5):390–400
Rimondi E, Rossi G, Bartalena T et al (2011) Percutaneous CT-guided biopsy of the musculoskeletal system: results of 2027 cases. Eur J Radiol 77(1):34–42
Mavrogenis AF, Rossi G, Calabrò T et al (2012) The role of embolization for hemangiomas. Musculoskelet Surg 96(2):125–135
Mavrogenis AF, Angelini A, Errani C, Rimondi E (2014) How should musculoskeletal biopsies be performed? Orthopedics 37(9):585–588
Mavrogenis AF, Angelini A, Vottis C et al (2015) State-of-the-art approach for bone sarcomas. Eur J Orthop Surg Traumatol 25(1):5–15
Wild AT, Raab P, Krauspe R (2000) Hemangioma of skeletal muscle. ArchOrthop Trauma Surg 120(3–4):139–143
Ly JQ, Sanders TG, Mulloy JP et al (2003) Osseous change adjacent to soft tissue hemangiomas of the extremities: correlation with lesion size and proximity to bone. AJR Am J Roentgenol 180(6):1695–1700
Robertson RL, Robson CD, Barnes PD, Burrows PE (1999) Head and neck vascular anomalies of childhood. Neuroimaging Clin N Am 9(1):115–132
Greenspan A, McGahan JP, Vogelsang P, Szabo RM (1992) Imaging strategies in the evaluation of soft-tissue hemangiomas of the extremities: correlation of the findings of plain radiography, angiography, CT, MRI, and ultrasonography in 12 histologically proven cases. Skeletal Radiol 21(1):11–18
Trop I, Dubois J, Guibaud L et al (1999) Soft-tissue venous malformations in pediatric and young adult patients: diagnosis with Doppler US. Radiology 212(3):841–845
Bhat V, Salins PC, Bhat V (2014) Imaging spectrum of hemangioma and vascular malformations of the head and neck in children and adolescents. J Clin Imaging Sci 24(4):31
Donnelly LF, Bissettz GS, Adams DM (1999) Combined sonographic and fluoroscopic guidance: a Modified technique for percutaneous sclerosis of low-flow vascular malformations. AJR Am J Roentgenol 173:655–657
Fishman SJ, Mulliken JB (1993) Hemangiomas and vascular malformations of infancy and childhood. Pediatr Clin North Am 40:1177–1200
Dubois J, Alison M (2010) Vascular anomalies: what a radiologist needs to know. Pediatr Radiol 40:895–905
Kern S, Niemeyer C, Darge K et al (2000) Differentiation of vascular birthmarks by MR imaging. An investigation of hemangiomas, venous and lymphatic malformations. Acta Radiol 41:453–457
Berenguer B, Mulliken JB, Enjolras O et al (2003) Rapidly involuting congenital hemangioma: clinical and histopathologic features. Pediatr Dev Pathol 6:495–510
Grevelink SV, Mulliken JB (2003) Vascular anomalies and tumors of skin and subcutaneous tissues. In: Freedberg IM, Eisen AZ, Wolff K et al (eds) Fitzpatrick’s dermatology in general medicine, vol 1, 6th edn. McGraw-Hill, New York, pp 1002–1026
Enjolras O (2008) Vascular malformations. In: Callen JP, Horn TD, Mancini AJ et al (eds) Dermatology, vol 2, 2nd edn. Elsevier, St. Louis, pp 1581–1595
North PE, Kincannon J (2008) Vascular neoplasms and neoplastic-like proliferations. In: Callen JP, Horn TD, Mancini AJ et al (eds) Dermatology, vol 2, 2nd edn. Elsevier, St. Louis, pp 1771–1794
Requena C, Miranda L, Canete A et al (2008) Congenital fibrosarcoma simulating congenital hemangioma. Pediatr Dermatol 25:141–144
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Author Eugenio Rimondi declares that he has no conflict of interest. Author Andreas F. Mavrogenis declares that he has no conflict of interest. Author Costantino Errani declares that he has no conflict of interest. Author Teresa Calabrò declares that she has no conflict of interest. Author Alberto Bazzocchi declares that he has no conflict of interest. Author Giancarlo Facchini declares that he has no conflict of interest. Author Salvatore Donatiello declares that he has no conflict of interest. Author Paolo Spinnato declares that he has no conflict of interest. Author Daniel Vanel declares that he has no conflict of interest. Author Ugo Albisinni declares that he has no conflict of interest. Author Patrizia Pelotti declares that she has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Rimondi, E., Mavrogenis, A.F., Errani, C. et al. Biopsy is not necessary for the diagnosis of soft tissue hemangiomas. Radiol med 123, 538–544 (2018). https://doi.org/10.1007/s11547-018-0862-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-018-0862-y