Abstract
Purpose
The aquaculture sector is the fastest growing food production industry. Life-cycle assessment (LCA) can be a useful tool to assess its environmental impacts and ensure environmentally sustainable development. Years ago, critical reviews of LCA methodology have been conducted in that field to evaluate methodological practice. However, how effective were these reviews in improving LCA application? Are there any remaining issues that LCA practitioners should address in their practice?
Methods
We tackle the above questions by critically reviewing all LCA cases applied to aquaculture and aquafeed production systems from a methodological point of view. A total of 65 studies were retrieved, thus tripling the scope of previous reviews. The studies were analysed following the main phases of the LCA methodology as described in the ISO standards, and the authors’ choices were extracted to identify potential trends in the LCA practice.
Results and discussion
We identified five main methodological issues, which still pose challenges to LCA practitioners: (i) the functional unit not always reflecting the actual function of the system, (ii) the system boundary often being too restricted, (iii) the multi-functionality of processes too often being handled with economic allocation while more recommendable ways exist, (iv) the impact coverage not covering all environmental impacts relevant to aquaculture and (v) the interpretation phase usually lacking critical discussion of the methodological limitations. We analysed these aspects in depth, highlighting trends and tendencies.
Conclusions
For each of the five remaining issues, we provided recommendations to be integrated by practitioners in their future LCA practice. We also developed a brief research agenda to address the future needs of LCA in the aquaculture sector. The first need is that emphasis should be put on the construction of aquaculture life-cycle inventory databases with a special need for developing countries and for post-farming processes. Additionally, method developers should develop and/or refine characterisation models for missing impact pathways to better cover all relevant impacts of seafood farming.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Because of a growing global population, food demand currently faces a significant increase, which is expected to intensify in the future (UN 2017). As a main diet component in many countries and a healthy source of protein, seafood demand is no exception to that trend. Historically, fisheries were the main source of producing seafood, but with a majority of the fish stocks now fished at maximum capacity or at unsustainable levels, seafood production has progressively transitioned to aquaculture, for which production has boomed over the last decades (FAO 2016). However, the aquaculture industry remains associated with a number of impacts on the environment, such as climate change, aquatic eutrophication or loss of biodiversity due to escapes of farmed animals (Naylor et al. 2000; Diana 2009; Ottinger et al. 2016). It is therefore crucial to ensure that the fast development of the aquaculture sector happens in the most sustainable way possible.
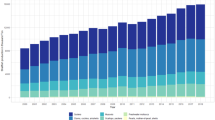
A common tool to assess environmental sustainability of products or systems is life-cycle assessment (LCA; ISO 2006a, b). It has already been widely applied to assess aquaculture systems since the early 2000s. The number of LCA studies published in scientific literature has intensified in the last few years now reaching over 50 publications (Fig. 1; Bohnes et al. 2018). Previous critical reviews have been made, looking at the findings of the LCA studies as well as the methodological choices of LCA practitioners (Henriksson et al. 2012; Parker 2012; Aubin 2013; Cao et al. 2013; Pahri et al. 2015). For instance, Henriksson et al. (2012) analysed methodological practices from 12 LCA studies of aquaculture systems. The authors concluded on a lack of transparency in the data used, and reported a limited coverage in the number of impacts assessed by the studies and too narrowly scoped system boundaries, for which they provided a number of recommendations to future studies. The aforementioned past reviews have provided similar messages to improve LCA practice based on other limited sets of studies (see Fig. 1). However, now that the number of publications has more than quadrupled, how have these messages been taken up by LCA practitioners in the aquaculture sector? For example, has system boundary completeness and environmental impact coverage been improved in recent LCA studies conducted since critical reviews were published?
Here, we conducted a follow-up critical review of all existing LCA studies in the aquaculture sector to address how LCA practice has evolved since previous reviews and recommendations were released and identify potential points that still remain to be addressed by practitioners. In the subsequent sections, we use this review basis to (i) critically evaluate the methodological choices of LCA studies in the aquaculture sector and provide a new set of recommendations wherever needed (Sect. 3) and (ii) outline a research agenda to address the requirements for more consistent LCA practice in the aquaculture sector (Sect. 4).
2 Material and methods
2.1 Identification of the studies
To enter the scope of this review, LCA studies had to comply with the following requirements: (i) assessing at least one production system of aquaculture or aquafeed (i.e. feed for aquatic organisms farmed in aquaculture); (ii) focusing on seafood production for direct human consumption; and (iii) including at least two impact categories (therefore, we excluded, e.g. stand-alone carbon footprinting studies). Only articles in peer-reviewed journals and publicly available peer-reviewed LCA reports published up to June 2017 and written in English were considered. The studies were found using Web of Science online database (http://webofknowledge.com) and Google Scholar research tool (https://scholar.google.dk/), with the keywords “Life-cycle assessment” + “Aquaculture”, “Life-cycle analysis” + “aquaculture”, “LCA” + “aquaculture”, “Life-cycle assessment” + “aquafeed” and “Life-cycle assessment” + “aquaculture” + “feed”. Additional studies were identified by cross-referencing existing reviews in that field (Henriksson et al. 2012; Parker 2012; Aubin 2013; Cao et al. 2013; Clark and Tilman 2017). For further details on the identification and selection of the studies, the readers are referred to Bohnes et al. (2018), who used the same pool of LCA studies to analyse trends and patterns of environmental impacts from different aquaculture systems.
2.2 Review criteria
Studies were analysed following the main phases of the LCA methodology as described by the ISO standards (ISO 2006a, b), i.e. goal definition, scope definition, life-cycle inventory (LCI), life-cycle impact assessment (LCIA) and life-cycle interpretation. Table 1 presents the list of the main methodological choices retrieved. Data quality was categorised as poor, medium or good following the same criteria than Laurent et al. (2014). They were then compiled and analysed to identify potential trends and patterns in practice, and their relevance was critically considered in the context of the ISO14040-4 standards (ISO 2006a, b). Based on this analysis and the recommendations made in previous reviews of LCA methodology (Henriksson et al. 2012; Parker 2012; Aubin 2013), we identified and prioritised five important methodological issues. These mainly relate to the scope definition of the study (one also addresses interpretation of the results), which is an essential phase to ensure consistency and reliability in the LCA results. Using ISO standards, we then established a set of recommendations to LCA practitioners to potentially improve the quality of future LCA studies.
3 Past LCA practices and improvement potentials
We retrieved and reviewed a total of 65 LCA studies on aquaculture and aquafeed systems; 51 of them assessed aquaculture production systems, 10 assessed aquafeed production systems and 4 included the assessment of both types of systems. An exhaustive list of all the LCA studies included in the review is available in Table 2 for the studies assessing aquaculture production and Table 3 for the ones assessing aquafeed production.
3.1 Making the functional unit reflect the actual function of aquaculture systems
More than 70% of the LCA practitioners assessing aquaculture systems have adopted a functional unit (FU) based on a mass of live-weight seafood (see Fig. 2a; Table 2). This particularly high proportion reflects the focus of many LCA studies on the production side, assessing a function based on the needs and benefits of the producer. It contrasts with the few authors (e.g. Avadí and Fréon 2015) that selected a mass of edible or processed product, hence basing their reference on the consumer needs, which convey a consumption approach. With regard to the 14 studies that assessed aquafeed production systems, 11 of them adopted a FU based on mass of aquafeed, while the remaining ones followed a different approach and used a mass of protein (Strazza et al. 2015), a surface of cultivation (Seghetta et al. 2017) or an energy content (Taelman et al. 2013)—see Fig. 2a; Table 3. It should be highlighted that 14% of the studies had not explicitly defined and reported a FU, which thus had to be deduced from the text and tables/figures of the articles. This lack of transparency only slightly decreased since the last review of LCA methodologies, from 16% in the studies prior to 2013 to 12% in the more recent studies.
The FU is particularly important for comparative assessments because of the need to quantify an identical function for both systems to allow a fair comparison. Defining differently the FUs may lead to different ranking of the assessed solutions, as illustrated by Avadí et al. (2015), who tested two different FUs based on either the mass of live-weight product or the mass of edible product. Furthermore, when assessing the life-cycle of a food product, using a FU based on the product total mass does not reflect the actual function of that product, i.e. to provide nutritional benefits to the consumer (Sala et al. 2017; Sonesson et al. 2017). Most past critical reviews in the field already pointed out practitioners’ preference to define a mass-based FU. They highlighted that the lack of consensus on the way to define the FU reduces the possibility of comparison between studies (Aubin 2013; Cao et al. 2013) and stressed the risk that the choice of the FU might change the results of the study (Henriksson et al. 2012; Parker 2012).
To ensure consistency, it is therefore recommended to define the FU of aquaculture LCA studies based on nutritional criteria of the product, such as protein or energy content, as already emphasised by Sala et al. (2017) and Sonesson et al. (2017). A consensus should be reached in the LCA food community to determine which nutritional criteria the defined FU should rely on as a function of the goal of the LCA, so that future studies can align with this same basis and become more comparable. Such recommendation also applies to aquafeed systems. Indeed, the primary function of the aquafeed is to feed the fishes, that function is only captured properly when a nutritional reference is used. For instance, comparing plant-based ingredients with fish-based ingredients based on a mass alone, as done in several past studies (see Table 3), might be highly misleading, because the amount required to fulfil the needs of the fish is highly different for the two ingredients. To prevent such situation, we recommend to compare full diets to ensure comparability of the aquafeeds’ function.
3.2 Including all relevant life-cycle stages of aquaculture production
Several processes constitute the life-cycle stages of an aquaculture production system. As illustrated in Fig. 3, they can be divided as feed production, energy supply, chemical inputs, infrastructures and equipment, seafood production, processing, packaging, distribution, consumption and seafood end-of-life. All these elements need to be included in an LCA to ensure a complete life-cycle. However, 69% of the studies reviewed herein did not consider the last five aforementioned processes and ended their assessments at farm gate, conducting therefore “cradle-to-farm-gate” LCAs. Additionally, the production and use of chemicals and the infrastructures and equipment were often neglected, with only 64% of the studies including the first and 60% considering the latter. The reason stated by the authors for not including these stages are the expected negligible impacts these may have or the lack of primary data and available databases to support a consistent modelling.
Different stages and processes of aquaculture production and types of system boundaries (adapted from Bohnes et al. 2018). The thick arrows represent the stages between which transport can occur (dependent on case study)
Including all elements that may have important environmental impacts is necessary to conduct a comprehensive LCA and avoid burden shifting from one environmental impact to another (Hellweg and Milà i Canals 2014; Ziegler et al. 2016). Some post-farming processes have been demonstrated to be of potentially great importance on the final impact scores and can increase impacts (e.g. transport to distribution; Seves et al. 2016) or decrease them (e.g. reuse or recycling at end-of-life; Iribarren et al. 2010a). Parker (2012) already introduced the benefits of a larger system boundary than cradle-to-farm gate. Additionally, by conducting a detailed contribution analysis (i.e. hotspot analysis) from the documented results, Bohnes et al. (2018) found out that 78 and 84% of the existing studies that adopted a complete life-cycle reported a non-negligible contribution of 5% or more for the production and use of chemicals and for the infrastructures and equipment, respectively. Henriksson et al. (2012) had already highlighted the need of a broadly encompassing system boundary and the importance of including infrastructures. We reiterate this still ignored recommendation to consider a complete life-cycle when performing LCAs of aquaculture systems, using the processes in Fig. 3 as guidance to ensure a comprehensive assessment of the environmental impacts.
3.3 Using system expansion instead of allocation for handling multi-functional processes
It is common in LCA that a single process produces multiple outputs or functions, called therefore a multi-functional process. Usually, only one of the functions needs to be included in the assessment, hence the necessity of methodologies to solve process multi-functionality. From the retrieved studies, 58% of them selected allocation, 13% system expansion, and 3% used both, while 26% of the studies did not explicitly state which method they used—see Fig. 2b. A difference is witnessed between the studies published until 2012 and the more recent ones: the use of system expansion increased from 7 to 16%, and the proportion of studies not stating which method they used dropped from 36 to 19%. The use of allocation did not change considerably. As evidenced in the sensitivity analyses of numerous LCA studies included in the current review (e.g. Winther et al. 2009; Kluts et al. 2012; Wilfart et al. 2013; Aubin et al. 2015; Jonell and Henriksson 2015; McGrath et al. 2015; Nhu et al. 2016; Medeiros et al. 2017), the choice of method to solve process multi-functionality is of great importance for the LCA results.
Past general reviews already noted the lack of consensus regarding the approach to be used for handling multi-functionality and, without providing explicit recommendations, they highlighted the need for a better argumentation to justify the choice of the approach applied (Henriksson et al. 2012; Parker 2012; Aubin 2013). According to ISO 14044, it is recommended to prioritise sub-division of the system whenever possible (ISO 2006b). However, the cases when this approach is possible are rare, and the second most recommended method is then system expansion, and, if that is not possible, the LCA practitioner should apply allocation and prioritise physical allocation keys over other types such as, e.g. economic allocation (ISO 2006b).
Considering that more than half of the studies applied allocation, it is therefore legitimate to question whether or not system expansion is applicable in aquaculture systems. By analysing the studies that applied system expansion, it appears that this method can be applied in handling the outputs of several co-products related to aquaculture systems. Natural fertilisers can thus fulfil the same function as synthetic fertilisers (see, e.g. Ayer and Tyedmers 2009; Kluts et al. 2012), seafood or agricultural co-products are equivalent to the same products from conventional production ways, usually from monoculture (e.g. Boxman et al. 2016; Medeiros et al. 2017), aquafeed co-products can be functionally equivalent to the marginal corresponding ingredients (see, e.g. Samuel-Fitwi et al. 2013a), and waste products can generally be valorised, e.g. mussels shells used to produce calcium, thus replacing conventional means (Iribarren et al. 2010a). The above examples cover most of the secondary functions arising from aquaculture and aquafeed production systems and demonstrate that using system expansion is possible in that area for most multi-functional processes.
However, some LCA practitioners have argued that some of the multi-functionality cited above are not solvable by using system expansion. We observed that usually this comes from a difference in the definition of the function to isolate. For instance, the production of fish meal always has fish oil as a co-product, and some LCA practitioners would isolate the fish oil production by expanding the system and include the production of other oils, e.g. vegetal ones, whereas other authors would argue that this is not reasonable because of the different nutritional compositions that make fish oil unique, hence the use of allocation. This is a legitimate decision of the LCA practitioner, but it is not always well justified in the articles under review and allocation often seems to be the default solution. Therefore, we recommend to explain in more details the reason why allocation cannot be avoided and to state explicitly the function considered, which has no alternative processes. Once allocation have been selected, Fig. 2b shows that a third of the LCA studies chose an economic allocation key over a physical one, which should be considered as a last resort according to the ISO hierarchy to solve process multi-functionality (see above: ISO 2006a, b). Indeed, economic allocation keys are not stable because of market fluctuations, which leads to constantly changing LCA results (Ayer et al. 2007). In most cases when system expansion cannot be applied, the multi-functionality concerns the production phase and therefore physical allocation such as energy content or mass allocation can be used instead of economic criteria. This was already recommended by Ayer et al. (2007) in their critical review of co-product allocation in fisheries and aquaculture, where they argued that gross-energy allocation is the most scientifically accurate solution for the cases when system expansion is not applicable.
We therefore recommend that LCA practitioners follow more rigorously the hierarchy specified in the ISO standards to handle multi-functionality of processes. In particular, system expansion should be more prioritised over allocation as it is often applicable. Practitioners are thus encouraged to check previous LCA studies that used system expansion (see above examples) and when allocation cannot be avoided, to use physical allocation keys instead of economic ones.
3.4 Covering all environmental impacts of aquaculture
Figure 2c shows that a majority of studies included climate change, aquatic eutrophication, acidification and cumulative energy demand (all four categories covered in more than 50% of studies), but that all other impact categories are rarely included. Only a few studies included toxicity impacts (25% for human toxicity and 28% for ecotoxicity) or land use (38%), and less than half included net primary production use (NPPU) and water dependence, two impact categories specific and of high relevance to food production systems (Aubin et al. 2009; Cashion et al. 2016). Overall, the spectrum of included impact categories was limited, their selection was poorly justified and exclusively based on the argument that previous LCA studies on aquaculture systems had similarly limited impact coverage. Rare were the authors, who justified the selection of their impact assessment on scientific foundations about the potential relevance of different impact categories (see as example of good practice Avadí and Fréon 2015).
In their critical reviews, Henriksson et al. (2012) and Aubin (2013) already highlighted the limited impact coverage of LCA studies on aquaculture. Together with the life-cycle perspective, the impact coverage is a key element in LCA to ensure a holistic dimension and reduce the risk of environmental burden-shifting (Laurent et al. 2012). When some categories for which the system has high environmental impacts are omitted, the results might be biased and the decisions based on the conclusions might lead to suboptimisation, i.e. decreasing some impacts while increasing others as relevant. For instance, toxicity impacts may be of high relevance in aquaculture systems, as showed by Kluts et al. (2012), who found a different ranking in their comparative study for freshwater ecotoxicity than for most of the other impact categories assessed. Other impacts are as relevant. The inclusion of land use impact category thus has been recommended by several authors (Bosma et al. 2011; Kluts et al. 2012; Samuel-Fitwi et al. 2013b; Dekamin et al. 2015; Jonell and Henriksson 2015), although it has until now mainly been assessed at an inventory level (i.e. total area of land occupied or transformed) without impact assessment. Additionally, indicators specific to biomass extraction that also account for the pressure exerted on wild fish stocks have been developed, and a number of approaches have been proposed although no consensus have yet been reached on a specific LCIA method (see, e.g. Lost Potential Yield (LPY) in Emanuelsson et al. 2014 or Biotic Natural Resource Depletion (BNRD) in Langlois et al. 2012). Therefore, we recommend the assessment of a broad variety of relevant impact categories in future LCA studies, including toxicity impacts and land use, as well as NPPU, water dependence and overfishing related impacts, which are not common to LCA applications, albeit relevant to aquaculture systems. LCIA methods for these categories exist and should be used, including, but not limited to, the USEtox model for toxicity impacts (Bijster et al. 2017), land use assessment method developed by Chaudhary et al. (2015) and recommended in Jolliet et al. (2018), the NPPU method described in Papatryphon et al. (2004a) and water dependence introduced and developed by Aubin et al. (2009).
3.5 Discussing the results with critical thinking and highlighting the limitations of the studies
Out of the 65 reviewed studies, an overall good quality of the data sources used in the studies was observed, with 85% of the studies relying on primary data and adequate literature sources with respect to data specificity and scope (see Sect. 2.2). However, only half of the studies critically discussed the representativeness of the data, which consists of data that are appropriate in terms of their geographical, temporal and technological aspects. To support the interpretation of LCA results, uncertainty and sensitivity analyses are recommended as part of the sensitivity check (ISO 2006a, b; Laurent et al. 2018). However, only 49% of the studies conducted a sensitivity analysis and 28% ran a quantitative uncertainty analysis.
The accuracy and hence the reliability of the LCA results are highly dependent on the quality of the data collected and the sensitivity and uncertainty underlying in the model. Therefore, these matters need to be critically analysed in the interpretation phase of the assessment during the completeness, consistency and sensitivity checks to support the conclusions from the results as well as the recommendations based on them. The review conducted by Henriksson et al. (2012) emphasised a lack of sensitivity analyses in the LCA studies, and the results of the current study also showed a lack of critical analysis, regardless of the time of publication of the studies (problem encountered in recent studies too). This prevents the reader from putting the results in perspective and assessing the robustness of the results.
Therefore, we recommend future LCA practitioners to critically discuss their LCI and include a detailed description of the limitations of study in the interpretation. We also recommend to systematically perform a sensitivity analysis of a large selection of criteria covering the input data and the modelling choices, and to conduct a quantitative uncertainty analysis such as a Monte Carlo simulations (available in most LCA software), wherever possible, to complement a default qualitative analysis. Guidance for performing interpretation of LCA results is available in Laurent et al. (2018).
4 Research needs in LCA for aquaculture
From the critical review of 65 LCA studies, we additionally identified two main research needs that should be addressed to improve LCA applications to the aquaculture sector: constructing comprehensive LCI data sets and developing missing relevant impact pathways. Both are developed in the following subsections.
4.1 Increasing the pool of LCI data sets for aquaculture
Several studies reported a lack of available LCI for modelling processes within the life-cycle of aquaculture systems, hence preventing them from including these elements in their assessments. Data regarding all post-farming stages (e.g. transport, processing, distribution, consumption and end-of-life) are thus extremely scarce, if not inexistent, as highlighted previously by Abdou et al. (2017a). For primary data collection, LCA practitioners are usually in contact with the seafood farmers, who often know little about the processes occurring to their seafood after farm gate. Therefore, the processing, packaging, transport and distribution steps are almost always missing from the assessment because of the lack of information, which might have an important impact on the final results. For instance, Winther et al. (2009) found that transport can be a main contributor to the final scores depending on the distribution zone of the product, and Iribarren et al. (2010b) highlighted the importance that processing and packaging may have on the results. Specific processes of aquaculture are also poorly documented. Infrastructures for instance are problematic because some parts, such as the water filtration systems, are difficult to model by the LCA practitioners due their high complexity in term of number of components and variety of materials.
Additionally, there is a general lack of databases concerning developing countries, leading to only a few LCA studies performed in these regions and to less robust assessments when some have been attempted (Dekamin et al. 2015; Bohnes et al. 2018). This is especially problematic in aquaculture assessments as more than 95% of the world production of seafood from aquaculture takes place in Asia, where only few general LCI are publicly available (Bohnes et al. 2018). In the Ecoinvent database (Weidema et al. 2013), which is the most widely used LCI database in our review (used in 74% of the studies), only few processes are specific to, e.g. Indonesia (35 processes), Vietnam (14 processes) or the Philippines (17 processes), which are the 2nd, 4th and 5th most important aquaculture producers in the world, respectively (FAO 2016).
We therefore encourage all aquaculture stakeholders to share data for enabling the construction of LCI data sets, which would improve the overall quality of future LCA studies and facilitate their applications to relevant systems and locations.
4.2 Missing impact pathways
Several studies have pointed out that the current LCIA methodologies do not cover all the environmental impacts relevant to aquaculture, as highlighted by Ellingsen and Aanondsen (2006), Samuel-Fitwi et al. (2013b), Aubin et al. (2015), Avadi and Fréon (2015), Henriksson et al. (2015, 2017a), Nhu et al. (2016) and Abdou et al. (2017a). Below, two major gaps are highlighted: impacts from escapes and damages related to use of antibiotics and medicine treatment.
The impacts of escapes on the local environments are thus not addressed, albeit being a well-documented issue in that sector (Naylor et al. 2000; Diana 2009). If the escaped species are invasive, they can affect the balance of the local ecosystem because of the introduction of new predators, which can have important consequences as the extinction of local species (Arismendi et al. 2009; Peeler et al. 2011). If the farmed species are already present in the local ecosystems, it can be as problematic because of breeding that changes the genetics of farmed specimens and make them different from the wild ones, thus altering the natural balance of species present in the ecosystem and potentially contributing to biodiversity losses and/or changes in ecosystem functioning (Youngson and Saroglia 2001; Naylor et al. 2005). Some authors already highlighted the need of including that issue in life-cycle impact assessment and proposed ways of accounting for it (Ford et al. 2012). However, no actual impact pathways have been developed yet, and escapes are only suggested to be considered at inventory level (i.e. accounting the number of fish that escaped per year; Ford et al. 2012).
Another uncovered impact pathway is the effect of antibiotics and other medicine used in seafood farms, and their subsequent impacts on human health through, for example antimicrobial resistance. Indeed, the use of antibiotics in food production as growth promoter or medical treatment leads to the development of resistant microorganisms, which will not be treatable by that antibiotic anymore, thereby inducing higher rates of infections by that microorganism in the human population (Cabello et al. 2013). This has recently been highlighted by the World Health Organisation, which recommended addressing this topic urgently (WHO 2018). The use of antibiotics should also be included in the modelling of impact pathways for ecotoxicity because of the potential impacts of these products on natural ecosystems. Antibiotics are designed to affect microorganisms in general and are therefore a threat for bacteria but also fungi and microalgae (Kümmerer 2009). Similarly, the impacts of cleaning products used during the farming stage are not included in some toxicity impact methodologies because these products are usually inorganics and their environmental fate is not always well known. For instance, the USEtox model, which covers 27 inorganics (mainly metals) and 3077 organic substances (Huijbregts et al. 2015a, b), does not include some of the common bleach such as sodium hypochlorite, thus calling for extending the substance coverage in its characterisation factor database.
For the two above methodological gaps, we recommend new method developments in LCIA to complement existing impact pathways and develop characterisation model to integrate these new cause-effect chains.
5 Conclusions
Based on the review of 65 LCA studies in the aquaculture sector, five major issues were identified and analysed. For each of them, recommendations were provided aiming to improve the quality and reproducibility of future LCAs in that sector. In summary, LCA practitioners should (i) choose a functional unit based on nutritional qualities, (ii) prefer system expansion over allocation and seek inspiration and assistance in published studies that applied this rule, (iii) assess a life-cycle as complete as possible in line with the goal of the study, (iv) include an environmental impact coverage as broad as possible and (v) pay special attention to the consistency/completeness check and the sensitivity and uncertainty analysis during the interpretation of the results. Drawing on these, we also identified two key research needs that method developers in LCI and LCIA should undertake, namely expanding LCI database with aquaculture-specific processes and characterising missing impact pathways, respectively. It is also worth noting that as highlighted in Sects. 3.1 to 3.5, a lack of transparency in the methodological choices is latent in many studies, with a non-negligible proportion of them not even stating their choices and assumptions. These not only refer to old studies, i.e. prior to previous critical reviews but also to a number of recent studies. Such poor practice is a great impediment to the credibility and reuse of the LCA results for large-scale analysis or comparative assessments.
We therefore recommend to future practitioners that they undertake these above messages. A few of our recommendations are not new and have already been indicated in previous critical reviews, be it within the field of aquaculture or in other fields. Recent studies have however showed that these key recommendations are not implemented by LCA practitioners. This demonstrates that there is a need for LCA practitioners to better inform themselves on the conduct of LCA in their specific fields of applications, e.g. by reading critical reviews, to integrate consistent guidance and overcome methodological challenges in their cases. Peer reviewers of scientific articles should also be aware of these critical reviews and of the methodological issues indicated therein to prevent studies with insufficient documentation and/or inconsistencies—as some identified in the current review—from being published. Such practice should eventually contribute to bring more consistency and reliability in LCA studies to support decision- and policy-making processes in fields as important and relevant as the aquaculture sector.
References
Abdou K, Aubin J, Romdhane MS, le Loc’h F, Lasram FBR (2017a) Environmental assessment of seabass (Dicentrarchus labrax) and seabream (Sparus aurata) farming from a life cycle perspective: a case study of a Tunisian aquaculture farm. Aquaculture 471:204–2012
Abdou K, Ben Rais Lasram F, Romdhane MS, le Loc’h F, Aubin J (2017b) Rearing performances and environmental assessment of sea cage farming in Tunisia using life cycle assessment (LCA) combined with PCA and HCPC. Int J Life Cycle Assess 23:1049–1062. https://doi.org/10.1007/s11367-017-1339-2
Arismendi I, Soto D, Penaluna B et al (2009) Aquaculture, non-native salmonid invasions and associated declines of native fishes in northern Patagonian lakes. Freshw Biol 54:1135–1147
Aubin J (2013) Life cycle assessment as applied to environmental choices regarding farmed or wild-caught fish. CAB Rev Perspect Agric Vet Sci Nutr Nat Resour 8:1–10
Aubin J, Baruthio A, Mungkung R, Lazard J (2015) Environmental performance of brackish water polyculture system from a life cycle perspective: a Filipino case study. Aquaculture 435:217–227
Aubin J, Fontaine C (2014) Impacts of producing bouchot mussels in Mont-Saint-Michel Bay (France) using LCA with emphasis on potential climate change and eutrophication. In: Proceedings of the 9th international conference on life cycle assessment in the agri-food sector environmental, pp 64–69
Aubin J, Papatryphon E, Van der Werf HMG et al (2006) Characterisation of the environmental impact of a turbot (Scophthalmus maximus) re-circulating production system using life cycle assessment. Aquaculture 261:1259–1268
Aubin J, Papatryphon E, van der Werf HMG, Chatzifotis S (2009) Assessment of the environmental impact of carnivorous finfish production systems using life cycle assessment. J Clean Prod 17:354–361
Avadí A, Fréon P (2015) A set of sustainability performance indicators for seafood: direct human consumption products from Peruvian anchoveta fisheries and freshwater aquaculture. Ecol Indic 48:518–532
Avadí A, Pelletier N, Aubin J, Ralite S, Núñez J, Fréon P (2015) Comparative environmental performance of artisanal and commercial feed use in Peruvian freshwater aquaculture. Aquaculture 435:52–66
Ayer N, Martin S, Dwyer RL, Gace L, Laurin L (2016) Environmental performance of copper-alloy net-pens: life cycle assessment of Atlantic salmon grow-out in copper-alloy and nylon net-pens. Aquaculture 453:93–103
Ayer NW, Tyedmers PH (2009) Assessing alternative aquaculture technologies: life cycle assessment of salmonid culture systems in Canada. J Clean Prod 17:362–373. https://doi.org/10.1016/j.jclepro.2008.08.002
Ayer NW, Tyedmers PH, Pelletier NL, Sonesson U, Scholz A (2007) Co-product allocation in life cycle assessments of seafood production systems: review of problems and strategies. Int J Life Cycle Assess 12:480–487
Baruthio A, Aubin J, Mungkung R et al (2008) Environmental assessment of Filipino fish/prawn polyculture using life cycle assessment. In: Proceedings of the 6th international conference on LCA in the agri-food sector. Zurich, Switzerland, pp 242–247
Besson M, Aubin J, Komen H, Poelman M, Quillet E, Vandeputte M, van Arendonk JAM, de Boer IJM (2016) Environmental impacts of genetic improvement of growth rate and feed conversion ratio in fish farming under rearing density and nitrogen output limitations. J Clean Prod 116:100–109
Bohnes FA, Hauschild MZ, Schlundt J, Laurent A (2018) Life cycle assessments of aquaculture systems: a critical review of reported findings with recommendations for policy and system development. Reviews in Aquaculture. https://doi.org/10.1111/RAQ.12280
Bijster M, Guignard C, Hauschild M et al (2017) USEtox 2.0 Documentation (v1)
Boissy J, Aubin J, Drissi A, van der Werf HMG, Bell GJ, Kaushik SJ (2011) Environmental impacts of plant-based salmonid diets at feed and farm scales. Aquaculture 321:61–70
Bosma R, Anh PT, Potting J (2011) Life cycle assessment of intensive striped catfish farming in the Mekong Delta for screening hotspots as input to environmental policy and research agenda. Int J Life Cycle Assess 16:903–915
Boxman SE, Zhang Q, Bailey D, Trotz MA (2016) Life cycle assessment a commercial-scale freshwater aquaponic system. Environ Eng Sci 34:299–311
Cabello FC, Godfrey HP, Tomova A, Ivanova L, Dölz H, Millanao A, Buschmann AH (2013) Antimicrobial use in aquaculture re-examined: its relevance to antimicrobial resistance and to animal and human health. Environ Microbiol 15:1917–1942
Cao L, Diana JS, Keoleian GA (2013) Role of life cycle assessment in sustainable aquaculture. Rev Aquac 5:61–71
Cao L, Diana JS, Keoleian GA, Lai Q (2011) Life cycle assessment of Chinese shrimp farming systems targeted for export and domestic sales. Environ Sci Technol 45:6531–6538
Cashion T, Hornborg S, Ziegler F, Hognes ES, Tyedmers P (2016) Review and advancement of the marine biotic resource use metric in seafood LCAs: a case study of Norwegian salmon feed. Int J Life Cycle Assess 21:1106–1120
Cashion T, Tyedmers P, Parker RWR (2017) Global reduction fisheries and their products in the context of sustainable limits. Fish Fish. https://doi.org/10.1111/faf.12222,18,1026,1037
Chaudhary A, Verones F, De Baan L, Hellweg S (2015) Quantifying land use impacts on biodiversity: combining species-area models and vulnerability indicators. Environ Sci Technol 49:9987–9995
Chen X, Samson E, Tocqueville A, Aubin J (2015) Environmental assessment of trout farming in France by life cycle assessment: using bootstrapped principal component analysis to better define system classification. J Clean Prod 87:87–95
Clark M, Tilman D (2017) Comparative analysis of environmental impacts of agricultural production systems, agricultural input efficiency, and food choice. Environ Res Lett 12:1–11
Dekamin M, Veisi H, Safari E, Liaghati H, Khoshbakht K, Dekamin MG (2015) Life cycle assessment for rainbow trout (Oncorhynchus mykiss) production systems: a case study for Iran. J Clean Prod 91:43–55
Diana JS (2009) Aquaculture production and biodiversity conservation. Bioscience 59:27–38
Efole Ewoukem T, Aubin J, Mikolasek O, Corson MS, Tomedi Eyango M, Tchoumboue J, van der Werf HMG, Ombredane D (2012) Environmental impacts of farms integrating aquaculture and agriculture in Cameroon. J Clean Prod 28:208–214
Emanuelsson A, Ziegler F, Pihl L, Sköld M, Sonesson U (2014) Accounting for overfishing in life cycle assessment: new impact categories for biotic resource use. Int J Life Cycle Assess 19:1156–1168
Ellingsen H, Aanondsen SA (2006) Environmental impacts of wild caught cod and farmed salmon—a comparison with chicken. Int J Life Cycle Assess 11:60–65
FAO (2016) The state of world fisheries and aquaculture. food and agriculture organisation of the United Nations, Rome, IT
Forchino AA, Lourguioui H, Brigolin D, Pastres R (2017) Aquaponics and sustainability: the comparison of two different aquaponic 2 techniques using the life cycle assessment (LCA). Aquac Eng 77:80–88
Ford JS, Pelletier NL, Ziegler F, Scholz AJ, Tyedmers PH, Sonesson U, Kruse SA, Silverman H (2012) Proposed local ecological impact categories and indicators for life cycle assessment of aquaculture: a Salmon aquaculture case study. J Ind Ecol 16:254–265
Fréon P, Durand H, Avadí A, Huaranca S, Orozco Moreyra R (2017) Life cycle assessment of three Peruvian fishmeal plants: toward a cleaner production. J Clean Prod 145:50–63
García García B, Rosique Jiménez C, Aguado-Giménez F, García García J (2016) Life cycle assessment of gilthead seabream (Sparus aurata) production in offshore fish farms. Sustainability 8:1228
Grönroos J, Seppälä J, Silvenius F, Mäkinen T (2006) Life cycle assessment of Finnish cultivated rainbow trout. Boreal Environ Res 11:401–414
Hellweg S, Mila i Canals L (2014) Emerging approaches, challenges and opportunities in life cycle assessment. Science 344 (6188):1109–1113
Henriksson P, Rico A, Zhang W (2015) Comparison of Asian aquaculture products by use of statistically supported life cycle assessment—supporting information. Sci Technol 49:14176–14183
Henriksson PJG, Dickson M, Allah AN, al-Kenawy D, Phillips M (2017a) Benchmarking the environmental performance of best management practice and genetic improvements in Egyptian aquaculture using life cycle assessment. Aquaculture 468:53–59
Henriksson PJG, Guinée JB, Kleijn R, De Snoo GR (2012) Life cycle assessment of aquaculture systems—a review of methodologies. Int J Life Cycle Assess 17:304–313
Henriksson PJG, Tran N, Mohan CV, Chan CY, Rodriguez UP, Suri S, Mateos LD, Utomo NBP, Hall S, Phillips MJ (2017b) Indonesian aquaculture futures - evaluating environmental and socioeconomic potentials and limitations. J Clean Prod 162:1482–1490
Huijbregts M, Hauschild M, Margni M et al (2015a) USEtox 2.0 user manual: inorganic substances (v2)
Huijbregts M, Meent D van de, Margni M et al (2015b) USEtox 2.0 user manual: organic substances (v2)
Iribarren D, Dagá P, Moreira MT, Feijoo G (2012a) Potential environmental effects of probiotics used in aquaculture. Aquac Int 20:779–789
Iribarren D, Moreira MT, Feijoo G (2010a) Revisiting the life cycle assessment of mussels from a sectorial perspective. J Clean Prod 18:101–111
Iribarren D, Moreira MT, Feijoo G (2010b) Life cycle assessment of fresh and canned mussel processing and consumption in Galicia (NW Spain). Resour Conserv Recycl 55:106–117
Iribarren D, Moreira MT, Feijoo G (2010c) Implementing by-product management into the life cycle assessment of the mussel sector. Resour Conserv Recycl 54:1219–1230
Iribarren D, Moreira MT, Feijoo G (2012b) Life cycle assessment of aquaculture feed and application to the turbot sector. Int J Environ Res 6:837–848
ISO (2006a) ISO 14040:2006—environmental management—life cycle assessment - principles and framework. Geneva, Switzerland
ISO (2006b) ISO 14044:2006—environmental management—life cycle assessment—requirements and guidelines. Geneva, Switzerland
Jerbi MA, Aubin J, Garnaoui K, Achour L, Kacem A (2012) Life cycle assessment (LCA) of two rearing techniques of sea bass (Dicentrarchus labrax). Aquac Eng 46:1–9
Jolliet O, Antón A, Boulay A-M, Cherubini F, Fantke P, Levasseur A, McKone TE, Michelsen O, Milà i Canals L, Motoshita M, Pfister S, Verones F, Vigon B, Frischknecht R (2018) Global guidance on environmental life cycle impact assessment indicators: impacts of climate change, fine particulate matter formation, water consumption and land use. Int J Life Cycle Assess. https://doi.org/10.1007/s11367-018-1443-y
Jonell M, Henriksson PJG (2015) Mangrove-shrimp farms in Vietnam-comparing organic and conventional systems using life cycle assessment. Aquaculture 447:66–75
Kluts IN, Potting J, Bosma RH, Phong LT, Udo HMJ (2012) Environmental comparison of intensive and integrated agriculture-aquaculture systems for striped catfish production in the Mekong Delta, Vietnam, based on two existing case studies using life cycle assessment. Rev Aquac 4:195–208
Kümmerer K (2009) Antibiotics in the aquatic environment—a review—part I. Chemosphere 75:417–434
Langlois J, Fréon P, Delgenes J-P et al (2012) Biotic resources extraction impact assessment in LCA of fisheries. In: 8th international conference on life cycle assessment in the agri-food sector. Saint-Malo (France), pp 517–522
Laurent A, Olsen SI, Hauschild MZ (2012) Limitations of Carbon Footprint as Indicator of Environmental Sustainability. Environmental Science & Technology 46(7):4100–4108
Laurent A, Bakas I, Clavreul J et al (2014) Review of LCA studies of solid waste management systems. Part I: Lessons learned and perspectives. Waste Manag 34:573–588
Laurent A, Weidema B, Bare J et al (2018) Methodological review and detailed guidance for the life cycle interpretation phase. Submitted to international journal of life cycle assessment (06/2018)
Lourguioui H, Brigolin D, Boulahdid M, Pastres R (2017) A perspective for reducing environmental impacts of mussel culture in Algeria. Int J Life Cycle Assess 22:1266–1277
Lozano S, Iribarren D, Moreira MT, Feijoo G (2010) Environmental impact efficiency in mussel cultivation. Resour Conserv Recycl 54:1269–1277
McGrath KP, Pelletier NL, Tyedmers PH (2015) Life cycle assessment of a novel closed-containment salmon aquaculture technology. Environ Sci Technol 49:5628–5636
Medeiros MV, Aubin J, Camargo AFM (2017) Life cycle assessment of fish and prawn production: comparison of monoculture and polyculture freshwater systems in Brazil. J Clean Prod 156:528–537
Mungkung R, Aubin J, Prihadi TH, Slembrouck J, van der Werf HMG, Legendre M (2013) Life cycle assessment for environmentally sustainable aquaculture management: a case study of combined aquaculture systems for carp and tilapia. J Clean Prod 57:249–256
Mungkung R, Udo de Haes H, Clift R (2006) Potentials and limitations of life cycle assessment in setting ecolabelling criteria: a case study of Thai shrimp aquaculture product. Int J Life Cycle Assess 11:55–59
Naylor R, Hindar K, Fleming IA et al (2005) Fugitive salmon: assessing the risks of escaped fish from net-pen aquaculture. Bioscience 55:427–437
Naylor RL, Goldburg RJ, Primavera JH, Kautsky N, Beveridge MCM, Clay J, Folke C, Lubchenco J, Mooney H, Troell M (2000) Effect of aquaculture on world fish supplies. Nature 405:1017–1024
Nhu TT, Schaubroeck T, Henriksson PJG, Bosma R, Sorgeloos P, Dewulf J (2016) Environmental impact of non-certified versus certified (ASC) intensive Pangasius aquaculture in Vietnam, a comparison based on a statistically supported LCA. Environ Pollut 219:156–165
Ottinger M, Clauss K, Kuenzer C (2016) Aquaculture: relevance, distribution, impacts and spatial assessments - a review. Ocean Coast Manag 119:244–266
Pahri SDR, Mohamed AF, Samat A (2015) LCA for open systems: a review of the influence of natural and anthropogenic factors on aquaculture systems. Int J Life Cycle Assess 20:1324–1337
Pahri SDR, Mohamed AF, Samat A (2016) Life cycle assessment of cockles (Anadara granosa) farming: a case study of Malaysia. Environ Asia 9:80–90
Papatryphon E, Petit J, Kaushik SJ, Van Der Werf HMG (2004a) Environmental impact assessment of salmonid feeds using life cycle assessment (LCA). Ambio 33:316–323
Papatryphon E, Petit J, Werf HMG van der, Kaushik SJ (2004b) Life cycle assessment of trout farming in France: a farm level approach. In: Proceedings from the 4th international conference, October 6–8, 2003, Bygholm, Denmark, pp 71–77
Parker R (2012) Review of life cycle assessment research on products derived from fisheries and aquaculture: a report for seafish as part of the collective action to address greenhouse gas emissions in seafood. Sea Fish Industry Authority, Edinburgh, UK
Parker RWR, Tyedmers PH (2012) Life cycle environmental impacts of three products derived from wild-caught Antarctic krill (Euphausia superba). Environ Sci Technol 46:4958–4965
Peeler EJ, Oidtmann BC, Midtlyng PJ, Miossec L, Gozlan RE (2011) Non-native aquatic animals introductions have driven disease emergence in Europe. Biol Invasions 13:1291–1303
Pelletier N, Tyedmers P (2007) Feeding farmed salmon: is organic better? Aquaculture 272:399–416
Pelletier N, Tyedmers P (2010) Life cycle assessment of frozen tilapia fillets from indonesian lake-based and pond-based intensive aquaculture systems. J Ind Ecol 14:467–481
Pelletier N, Tyedmers P, Sonesson U, Scholz A, Ziegler F, Flysjo A, Kruse S, Cancino B, Silverman H (2009) Not all salmon are created equal: life cycle assessment (LCA) of global salmon farming systems. Environ Sci Technol 43:8730–8736
Phong LT, de Boer IJM, Udo HMJ (2011) Life cycle assessment of food production in integrated agriculture-aquaculture systems of the Mekong Delta. Livest Sci 139:80–90
Roque d’Orbcastel E, Blancheton J-P, Aubin J (2009) Towards environmentally sustainable aquaculture: comparison between two trout farming systems using life cycle assessment. Aquac Eng 40:113–119
Sala S, Anton A, McLaren SJ et al (2017) In quest of reducing the environmental impacts of food production and consumption. J Clean Prod 140:387–398
Samuel-Fitwi B, Meyer S, Reckmann K, Schroeder JP, Schulz C (2013a) Aspiring for environmentally conscious aquafeed: comparative LCA of aquafeed manufacturing using different protein sources. J Clean Prod 52:225–233
Samuel-Fitwi B, Nagel F, Meyer S, Schroeder JP, Schulz C (2013b) Comparative life cycle assessment (LCA) of raising rainbow trout (Oncorhynchus mykiss) in different production systems. Aquac Eng 54:85–92
Samuel-Fitwi B, Schroeder JP, Schulz C (2013c) System delimitation in life cycle assessment (LCA) of aquaculture: striving for valid and comprehensive environmental assessment using rainbow trout farming as a case study. Int J Life Cycle Assess 18:577–589
Santos AAO, Aubin J, Corson MS et al (2015) Comparing environmental impacts of native and introduced freshwater prawn farming in Brazil and the influence of better effluent management using LCA. Aquaculture 444:151–159
Seghetta M, Romeo D, D’Este M et al (2017) Seaweed as innovative feedstock for energy and feed—evaluating the impacts through a life cycle assessment. J Clean Prod 150:1–15. https://doi.org/10.1016/j.jclepro.2017.02.022
Seves SM, Temme EHM, Brosens MCC, Zijp MC, Hoekstra J, Hollander A (2016) Sustainability aspects and nutritional composition of fish: evaluation of wild and cultivated fish species consumed in the Netherlands. Clim Chang 135:597–610
Smárason BÖ, Ögmundarson Ó, Árnason J et al (2017) Life cycle assessment of Icelandic Arctic char fed three different feed types. Turkish J Fish Aquat Sci 17:79–90
Sonesson U, Davis J, Flysjö A, Gustavsson J, Witthöft C (2017) Protein quality as functional unit—a methodological framework for inclusion in life cycle assessment of food. J Clean Prod 140:470–478
Strazza C, Magrassi F, Gallo M, Del Borghi A (2015) Life cycle assessment from food to food: a case study of circular economy from cruise ships to aquaculture. Sustain Prod Consum 2:40–51
Taelman SE, De Meester S, Roef L et al (2013) The environmental sustainability of microalgae as feed for aquaculture: a life cycle perspective. Bioresour Technol 150:513–522
UN (2017) World population prospects the 2017 revision key findings and advance tables. New York, USA
Weidema BP, Bauer C, Hischier R et al (2013) Data quality guideline for the Ecoinvent database version 3. The Ecoinvent Centre, St. Gallen, Switzerland
WHO (2018) Antimicrobial resistance. http://www.who.int/antimicrobial-resistance/en/. Accessed 1 Mar 2018
Wilfart A, Prudhomme J, Blancheton JP, Aubin J (2013) LCA and emergy accounting of aquaculture systems: towards ecological intensification. J Environ Manag 121:96–109
Winther U, Ziegler F, Hognes ES et al (2009) Carbon footprint and energy use of Norwegian seafood products. SINTEF Fisheries and Aquaculture
Yacout DMM, Soliman NF, Yacout MM (2016) Comparative life cycle assessment (LCA) of Tilapia in two production systems: semi-intensive and intensive. Int J Life Cycle Assess 21:806–819
Youngson D, Saroglia J (2001) Genetic interactions between marine finfish species European aquaculture and wild conspecifics. J Appl Ichthyol 17:153–162
Ziegler F, Hornborg S, Green BG, Eigaard OR, Farmery AK, Hammar L, Hartmann K, Molander S, Parker RWR, Hognes ES, Vázquez-Rowe I, Smith ADM (2016) Expanding the concept of sustainable seafood using Life Cycle Assessment. Fish and Fisheries 17(4):1073–1093
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Ian Vázquez-Rowe
Rights and permissions
About this article
Cite this article
Bohnes, F.A., Laurent, A. LCA of aquaculture systems: methodological issues and potential improvements. Int J Life Cycle Assess 24, 324–337 (2019). https://doi.org/10.1007/s11367-018-1517-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11367-018-1517-x