Abstract
The Brazilian Amazon is heavily impacted by mining activities, especially by gold and cassiterite mining. Pb is a contaminant released during cassiterite extraction, and it stands out for its high toxicity. The present study aimed to evaluate the effect of flood pulse on Pb dynamics in four fish species with different feeding habits (Cichlapleiozona: carnivorous, Mylossomaduriventre: herbivorous, Prochilodus nigricans: detritivorous, and Serrasalmus rhombeus: piscivorous) over four hydrological periods in Puruzinho Lake (Brazilian Amazon), which receives water from the Madeira River. The risk assessment for daily Pb intake through the consumption of these fish by the local riverside population was also carried out. Fish species were sampled during four Amazonian hydrological periods: rising water, high water, falling water, and low water. Pb and stable isotopes (δ15N and δ13C) measurements were carried out on fish muscle. The flood pulse had the same effect on Pb concentrations of all the fish species regardless of their feeding habits, as we found a nonsignificant interaction between these variables. The Pb in fish muscle increased in the flood season (rising and high water) and decreased in the dry season (falling and low water). The Pb concentration in fish increased significantly with their trophic level (δ15N), evidencing the occurrence of biomagnification of the element along the trophic chain (R2 = 0.24). The main food source of the herbivorous fish (δ13C) was very distinct from that of the others, which had a large overlap in their values. Regarding risk assessment, the daily Pb intake through consumption of the herbivorous species during high water (17.82 ± 19.68 µg∙day−1) exceeded the limit determined by the Food and Drug Administration (FDA) of 12.5 µg∙day−1, representing a health risk to the riverside population of Puruzinho Lake.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Brazilian Amazon is heavily impacted by mining activities, especially by gold and cassiterite mining (Rudke et al. 2020). Although the impacts of gold mining on Hg contamination have been widely studied in the Amazon (e.g., Nriagu et al. 1992; Pfeiffer and Lacerda 1988; Lacerda and Salomons 1998; Bastos et al. 2006; Lacerda and Malm 2008; Guimarães 2020; Pestana et al. 2022), studies on the impacts of cassiterite extraction on the release of contaminants to the environment are still incipient.
The extraction of cassiterite is intense in the Madeira River (Rudke et al. 2020), an important tributary of the Amazon River, especially near the city of Porto Velho (Rondônia State; Longo et al. 2011). Among the contaminants released during cassiterite extraction, Pb stands out for its high toxicity (Stierman et al. 2020; Ishitsuka et al. 2020) and accumulation in soils and sediments (Ribeiro et al. 2017). In addition to the point contamination caused by cassiterite mining, Pb also has important diffuse sources of contamination, such as industrial activities and its former use in gasoline; all of which are associated with the atmospheric phase of its biogeochemical cycle (UNEP 2010). The main routes of human exposure to Pb are inhalation and diet (Mushak 2011). Regarding contaminant intake through diet, fish consumption is of great concern in the Amazon, where the estimated consumption is 140 kg·person−1·year−1 (Oliveira et al. 2010), 15 times higher than the Brazilian average (8.9 kg·person−1·year−1; Sonoda and Shirota 2012). Given this high intake, even a low level of contamination can cause problems.
As a toxic element, Pb concentrations are monitored in food by Brazil’s National Sanitary Surveillance Agency (ANVISA). In commercial fish, the maximum tolerance limit (MTL) is 0.3 µg·g−1 (ANVISA 2021). In addition, there is an established daily Pb intake limit that if exceeded can harm human health. The last provisional reference limit established by the Food and Drug Administration (FDA) for daily Pb intake is 3 µg·day−1 for children and 12.5 µg·day−1 for adults (Dolan et al. 2020; Flannery et al. 2020). Pb intoxication can affect several body functions, especially neurological ones, and is linked to reduced cognitive functions and changes in neurotransmitter secretion (Ishitsuka et al. 2020; Stierman et al. 2020).
Some studies in the Amazon have already reported high levels of Pb exposure in the riverside population (Barbosa-Jr et al. 2009) and cassiterite miners (Hacon et al. 2010; Queiroz et al. 2019). Regarding cassiterite mining, it is clear that the distance from mining areas plays an important role in defining the health risk of populations (Hacon et al. 2010). High cassiterite mining activities are reported in the literature in the Jamari River (Longo et al. 2011), a tributary of the Madeira River. The waste released increases the Pb concentration in the water and can be transported to sedimentation areas like floodplains due to the great transport capacity of these rivers. This has already been observed for other contaminants like Cr, which also has an anthropic source in the Jamari River (tanneries) and whose concentrations increased more than 250 km downstream in the Madeira River until reaching Puruzinho Lake (Sousa et al. 2015; Nascimento et al. 2022).
In the Amazon, the transport capacity of rivers is strongly related to the seasonality of hydrological periods in aquatic ecosystems. Puruzinho Lake is a floodplain lake located in the state of Amazonas (AM) that is heavily influenced by the flood pulse of the Madeira River, to which it is connected by a narrow channel (locally called igarapé; Feitosa et al. 2019). The hydrological periods in the Amazon can be divided according to the rivers’ depths into rising and high water (which comprise the flood period) and falling and low water (which represent the dry period). The rising water period starts in October and ends in February; the high water period is established in February and ends in May; the falling water period begins in May and ends in September; the low water season starts in September and ends in October (Azevedo et al. 2020). During the high water period, the Madeira River overflows and its water invades Puruzinho Lake. At that moment, the contaminants present in the water of the Madeira River are transported to the lake (Nascimento et al. 2007) where they then can be absorbed by the biota.
The main route of contaminant absorption by fish is through diet. Depending on the behavior of the contaminant in the food web, biodilution or biomagnification of contaminants can occur (Azevedo et al. 2020; Nascimento et al. 2022). In the latter case, it directly impacts the health of the exposed riverside population and can be investigated through the use of stable isotopes. The stable isotope of carbon (δ13C) is commonly used to determine the source of the organic matter of an aquatic environment or organisms, such as fish, being fractionated mainly by photosynthetic activity and decomposition of organic matter (Fry 2006), while the stable isotope of nitrogen (δ15N) varies on average from 3 to 4‰ per trophic level, so its values can be used for studies on trophic relationships among living beings (Fry 2006).
The aim of this study was to evaluate the effects of the Madeira River flood pulse on the Pb dynamics in four fish species with different feeding habits in Puruzinho Lake, western Amazon. We also evaluated the risk of Pb intake through fish consumption by the riverside population of the region. Our hypothesis was that since the fish are from different trophic guilds, their diets would influence Pb concentrations throughout the four hydrological periods (Azevedo et al. 2020).
Material and methods
Study area
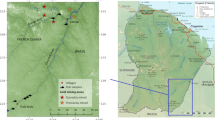
Puruzinho Lake (63°6′0″W; 7°24′0″S) is located near the municipality of Humaitá (Amazon state), on the left bank of the Madeira River, receiving its water during the flood period through an igarapé (Fig. 1). This river-floodplain system is common in the Amazon, and our study site is an ideal model for biogeochemical analysis of trace elements, as it has been used in the past for this purpose (Azevedo et al. 2020, 2021; Nascimento et al. 2022).
Puruzinho lake (left inset), located in the state of Amazonas, Brazil, which receives water from the the Madeira River through a narrow channel (igarapé). Cassiterite mining was reported to occur in the Jamari River (Longo et al. 2011) and is indicated on the map
The Madeira River is classified as a white water ecosystem characterized by brown water, pH close to neutral, and high concentrations of suspended particulate matter (SPM) (Sioli 1968). On the other hand, Puruzinho Lake is considered a black water ecosystem characterized by dark colored water, large amounts of organic matter, and pH tending to acidity (Sioli 1968). This lake is strongly influenced by the hydrological seasonality of the region, and during the high water period, the water column can reach up to 12.5 m in depth, while in the low water period this depth can decrease to only 0.30 m (Almeida et al. 2014; ANA 2019; Supplementary Material 1).
Sampling
The samples used in this study are from a larger project aimed to evaluate the biogeochemistry of trace elements in the Amazon region and have already been used for assessment of Hg and Cr dynamics in fish as well their trophic ecology (Azevedo et al. 2020, 2021; Nascimento et al. 2022). Briefly, fish were sampled with the help of local fishermen at a single point in the lentic part of the lake. Four species with different feeding habits were sampled: Mylossoma duriventre (herbivorous, common name “Pacuprata”), Prochilodus nigricans (detritivorous, “Curimatã”), Cichlapleiozona (carnivorous, “Tucunaréamarelo”), and Serrasalmus rhombeus (piscivorous, “Piranha preta”), totaling 268 specimens. For the capture of fish, gillnets with sizes of 30, 45, and 60 mm were used. After capture, the fish were measured for total length and weight. Also, fish muscle was extracted, freeze-dried (LioTop L108 freeze dryer; São Paulo, Brazil), and reserved for chemical measurements. The sampling was carried out between December 2016 and December 2018 comprising four hydrological periods and four fish species (Table 1, Supplementary Material 1). We defined the feeding habits of the species based on Cella-Ribeiro et al. (2016), who compiled detailed data about the stomach content and biology of several fish species from the Madeira River Basin.
Chemical analyses
Pb measurements
Pb determination was carried out using fish muscle tissue (1.0 g dry weight) as follows (adapted from Bastos et al. 1998): (i) solubilization using 2 mL of 30% hydrogen peroxide (H2O2) and 10 mL of 65% nitric acid (HNO3), (ii) heating in a digester block starting at 60 °C and ending at 120 °C until the complete solubilization of the samples, (iii) reducing the volume to 1 mL at 160 °C, and (iv) filtration with a quantitative filter (Whatman® 40 filter paper) followed by completing the volume up to 35 mL with 0.5 N HNO3. The measurements were performed by inductively coupled plasma-optical emission spectrometry (ICP-OES 720 ES; Varian, Australia) with a detection limit of 0.01 µg·g−1 (wet weight) and a 5-point calibration curve (0.02, 0.05, 0.1, 0.5, and 1 µg∙mL−1).
The analytical quality control of the samples was evaluated through analytical replicates and blanks and by the certified reference material (DORM-3), whose recovery was 107 ± 9% (n = 3). The Pb concentrations by dry weight were converted into wet weight since the regulatory limits are expressed in this unit. For conversion, the following moisture values were used: 77% water for the species Cichlapleiozona (Inhamuns and Franco 2008), 73% for Mylossomaduriventre (Melo et al. 2019), 71% for Prochilodus nigricans (Inhamuns 2000), and 78% for Serrasalmus rhombeus (Hiane et al. 2002).
Stable isotope (δ 13 C and δ 15 N) analysis
The stable isotope (δ13C and δ15N) analysis was carried out using a Thermo Finnigan Delta V Advantage mass spectrometer (isotopic ratio mass spectrometer, IRMS; Thermo Fisher Scientific, Milan, Italy) coupled to a Flash 2000 organic elemental analyzer (Thermo Fisher Scientific, Milan, Italy). Pee Dee Belemnite (PDB) and atmospheric nitrogen were used as standards for δ13C and δ15N, respectively. For δ13C, the coefficient of variation between replicates was ± 0.2‰, and for δ15N it was ± 0.3‰. The δ13C signature was corrected to remove the influence of lipids (McConnaughey and McRoy 1979; Azevedo et al. 2021).
Risk assessment
The assessment of risk of Pb ingestion through consumption of fish from Puruzinho Lake was calculated using the estimated daily intake (EDI) index (FAO/WHO 2014):
where EDI is the estimated daily intake (µg·day−1), CPb is the Pb concentration determined in the fish samples (µg∙g−1 wet weight), and Ifish is the intake rate (g∙day−1) for each fish species.
The fish intake rate (Ifish) for each species and each hydrological period was obtained from Oliveira et al. (2010), who surveyed the eating habits of residents of Puruzinho Lake (Nascimento et al. 2022). The average daily intake rate of all the fish species consumed by the riverine population is 406 g·person−1·day−1. The mean daily intake rate by the population, considering only the four analyzed species, is 40.4 g·person−1·day−1 (Supplementary Material 2).
Statistical analyses
Statistical analyses were performed in the R program (R Core Team 2022). Two-way ANOVA (aov function, base package, R Core Team 2022) was carried out to evaluate the effect of hydrological periods, eating habits, and the interaction between these two variables on Pb concentrations.
Pb variations over hydrological periods for the four species were evaluated using quadratic regressions (lm function, base package, R Core Team 2022), from which trend lines were extracted. In addition, linear regressions were performed (lm function, base package, R Core Team 2022) between Pb concentrations and stable isotope data in order to assess the association between these variables. In the regression analyses, the Pb-isotope distribution is shown using ellipses (data.ellipse function, car package; Fox and Weisberg 2019), constructed at 1 standard deviation from the centroid (68% confidence interval). Linear regressions were also carried out between Pb concentration and biometric variables (total length and weight; Supplementary Material 3).
When necessary, the data were transformed using a maximum likelihood function (boxcox function, MASS package; Venables and Ripley 2002) in order to meet the assumptions of ANOVA and regressions (linearity, normality, and homoscedasticity of residuals). The ANOVA and regressions were validated using diagnostic plots (Altman and Krzywinski 2016).
Fulton’s condition factor (K) was calculated as a ratio of fish weight (g) by the cube of their total length (cm) multiplied by 100 (Supplementary Material 4; Ricker 1975; Nash et al. 2006) to assess whether the Pb concentrations found in the fish would affect their health status (e.g., K = 1 being a fish in perfect health, K < 1 indicates a “slim” fish, and K > 1 a “fat fish”; Ricker 1975; Nash et al. 2006; Vries et al. 2020), but we found no significant relationship between the variables (Supplementary Material 5).
The assessment of risk of Pb ingestion through fish ingestion was carried out using empirical combinatorial analysis (Monte Carlo; expand.grid function; base package; Khitalishvili 2016) with fish ingestion and Pb concentration data, which generated all possible scenarios among the aforementioned variables, incorporating the variability of both in the final result.
In all applicable cases, an a priori type I error of 5% (α = 0.05) was assumed.
Results
The variation of Pb concentration along the hydrological periods was similar among the analyzed species (ANOVA, interaction term; p = 0.14). The carnivorous, herbivorous, and detritivorous fish showed an increase in Pb concentrations during the flood season (rising and high water) followed by a decrease in concentrations during the dry season (falling and low water; Fig. 2). In contrast, the piscivorous fish showed no relevant Pb variations over the hydrological periods. However, it exceeded the maximum tolerance limit for Pb established by the Brazilian National Sanitary Surveillance Agency (0.3 µg·g−1; ANVISA 2021) in all the hydrological periods, with the highest median of the study being found during the rising water period (0.48 ± 0.18; Fig. 2).
Pb concentrations in Cichla pleiozona, Mylossoma duriventre, Prochilodus nigricans, and Serrasalmus rhombeus in the four hydrological periods of the Brazilian Amazon. The red dashed line indicates the maximum tolerance limit value for Pb in fish (ANVISA 2013). The solid lines indicate the main trends of the medians over the hydrological periods and were extracted from quadratic regressions. The distances between the y-axis values were log-transformed to optimize data visualization
A gradual increase in Pb concentrations was observed with increasing trophic level (δ15N), evidencing the occurrence of Pb biomagnification along the trophic web (Fig. 3A). Although a significant relationship between Pb concentrations and δ13C was not observed, it was possible to observe that the food sources for M. duriventre were 10‰ higher compared to those of the other species, which showed a great overlap in their values (Fig. 3B).
Relationships between isotopic signatures of A nitrogen (δ15N) and B carbon (δ13C) and Pb concentrations in the four fish species analyzed. Ellipses were constructed at 1 standard deviation from their centroids (68% confidence interval). Regression statistics (equation, R2, and p-value) are within each graph. The distances between the y-axis values were log-transfomed to optimize data visualization
The risk assessment of Pb ingestion indicated that consumption of the herbivorous species during the high water period exceeded the provisional safe limit established by the Food and Drug Administration of 12.5 µg∙day−1 for adults (Dolan et al. 2020; Flannery et al. 2020; Fig. 4). Although the Pb concentrations in S. rhombeus exceeded the maximum tolerable limit established by ANVISA (2021) (Fig. 2), the isolated consumption of this species did not lead to exposure to Pb above the safe limit by the riverine population (Fig. 4), mainly because of its low consumption (Supplementary Material 2).
Pb-estimated daily intake through the consumption of the studied fish species over the four hydrological periods. The dashed red line indicates the provisional safe limit of 12.5 µg·day−1 established by the Food and Drug Administration for adults (Dolan et al. 2020; Flannery et al. 2020). The distances between the y-axis values were log-transformed to optimize visualization of the data
Discussion
Our results show that the flood pulse directly influences the Pb concentrations observed in the carnivorous, herbivorous, and detritivorous fish. Floodplain lakes such as Puruzinho, receive high concentrations of trace elements during flood pulses (Almeida et al. 2014; Pestana et al. 2019). This is due to the fact that the Madeira River, which invades Puruzinho Lake during flooding, is a white water river that has a high load of suspended particulate matter, one of the main geochemical supports for the transport of trace elements in aquatic ecosystems (Sioli 1968; Pestana et al. 2019). The rising and high water periods, when an increase in Pb concentrations was observed in the carnivorous, herbivorous and detritivorous fish, are precisely when Puruzinho Lake receives the highest contribution of suspended particles carried by the Madeira River. During these periods, the residence time of suspended particles rich in trace elements is longer, making them more bioavailable to the organisms ingested by the fish. With the reduction of water flow in the falling and low water periods, the particles gradually tend to settle to the bottom sediment, which may explain the decrease in the Pb concentrations in this period (Carvalho et al. 2018; Pestana et al. 2019).
These results strongly contrast with data from Nascimento et al. (2022), who analyzed the Cr variation along the hydrological periods in the same species and lake of this study due to Cr contamination via tanneries in the region. The authors identified a temporal variation of Cr concentrations in fish highly modulated by their feeding habits, which we did not observe. This makes it clear that the bioavailability of trace elements to biota in Amazon aquatic ecosystems depends heavily on the biogeochemistry of the chemical element. For example, Pb bonds strongly to the residual fraction of solids, while Cr binds to the oxidizable fraction. This means that the lake’s own seasonal oxygenation change in the water column could release Cr from the sediments back into the water, while the Pb sources would be more strongly associated with the Madeira River flood pulse transporting suspended particulate matter to the lake (Jakubus 2020). Furthermore, Pb bioavailability increases in the presence of organic matter, unlike Cr, which is reduced from Cr VI (more mobile) to Cr III (less mobile). Since the lake is a black water ecosystem naturally enriched in organic matter, this can also explain the relative independence of the Pb variation in the fish in relation to their feeding habits, unlike the case of Cr (Sioli 1968; Huser et al. 2012; Nascimento et al. 2022).
These results also contrast with data from Azevedo et al. (2020), who identified a clear interaction between the hydrological periods and feeding habits modulating Hg concentrations in the same species from the same lake while studying the effects of the flood pulse and artisanal gold mining on Hg contamination in the region. Unlike Pb, it is well established in the literature that the main fish exposure to Hg is through diet, which explains this difference (Arcagni et al. 2018; Azevedo et al. 2020).
Although the ANOVA’s interaction term indicated no statistical differences among slopes, the piscivorous fish appeared not to be influenced by the flood pulse throughout the hydrological periods, unlike the other species. This may be due to the fact that S. rhombeus is a species with a high position in the food web (δ15N), which allows it to feed on prey in a relatively continuous manner throughout the hydrological periods, also explaining the relatively constant concentrations of Pb during these periods.
The fish consumption by the riverside population of Puruzinho Lake, according to Oliveira et al. (2010), is 406 g·person−1·day−1, contrasting with the Brazilian national average of 8.9 g·person−1·day−1 (Sonoda and Shirota 2012). In fact, fish consumption in the Amazon is very high, comparable to the Maldives, whose population has the highest rate of fish consumption in the world (FAO 2016), so even low levels of fish contamination can lead to a high-risk scenario.
In the high water period, the median Pb intake from the consumption of the herbivorous species (17.82 ± 19.68 g·day−1) exceeded the provisional limit established by the FDA (Dolan et al. 2020; Flannery et al. 2020). This is mainly due to the high consumption of this fish species during the high water period (98.8 g∙day−1; Supplementary Material 2) and means that the isolated consumption of this fish can lead to health problems related to Pb contamination among the population of Puruzinho Lake, such as neurological, hematological, and renal problems (Moreira and Moreira 2004; Schifer et al. 2005). Two aggravating factors should be highlighted in this risk analysis. First, this analysis is an oversimplification of the actual eating habits of the community of Puruzinho Lake. To perform this analysis, we evaluated the isolated consumption of each species in each hydrological period, assuming that only one of them is consumed at a time. In practice, fish consumption varies among meals on the same day, making the Pb intake through the consumption of the species consumed in a single day cumulative. This means that the actual (cumulative) risk could be orders of magnitude higher than our estimates, which raises concerns. Furthermore, our data indicate moderate evidence of Pb biomagnification along the lake food chain (R2 = 0.24, p < 0.0001) but higher compared to other studies (R2 = 0.06, p = 0.019; Jiang et al. 2018), which makes consumption of predatory fish the worst case risk scenario. Pb biomagnification is a controversial topic in the literature, as some studies show negative associations with Pb exposure in controlled experiments (Cardwell et al. 2013) and in field observations (Liu et al. 2018; Souza-Araujo et al. 2022). However, our data are in agreement with the findings of other authors in highly industrialized environments (Yi et al. 2017).
Our results of Pb intake are concerning, especially regarding the herbivorous species. Since this species consumes mostly plants (as reflected in its δ13C, 10‰ different from the other species on average), it is possible to infer that the Pb in the lake is bioavailable for absorption by primary producers, which transfer the Pb to herbivores. The observed Pb intake values are in the range reported in other industrially polluted areas. Bassey and Chukwu (2019) found Pb intake between 3.9 and 8.3 µg∙day−1 through the consumption of a demersal detritivorous species from two lagoons in southwestern Nigeria. The authors stated that the high Pb concentrations found in the fish were due to wastewater discharges from local textile factories, printing cartridge waste, and other industrial effluents. Differently, Jiang et al. (2018) reported a mean Pb intake of 1.9 µg∙day−1 while studying 12 fish species in the second largest freshwater lake in China. While the authors stated that the area is one of the most industrialized in China, trace element concentrations in fish, including Pb, showed that the species had low levels of contaminants, which reflected the low estimated daily Pb intake.
Conclusion
Pb concentrations in herbivorous, detritivorous, and carnivorous species varied similarly according to hydrological periods, with the highest concentrations being observed in the flood season (rising and high water periods). This variation is probably related to the influence of the Madeira River water, which transports high loads of suspended particles with high adsorption capacity of metals. The piscivorous fish presented relatively constant Pb concentrations throughout the four hydrological periods, suggesting that they have a relatively stable diet throughout the year. Because of that, we can only partially refute our main hypothesis.
The consumption of the herbivorous species during the high water period has the potential to pose risks to the health of the riverside population of Puruzinho Lake. Given the moderate evidence of Pb biomagnification in the food chain, attention should also be paid to the consumption of predatory fish. In addition, it is worth noting that the data presented concern only four species of fish consumed by the community of Puruzinho Lake. Considering other species and their simultaneous ingestion on the same day, the risk scenario may much greater than we estimated.
Data availability
The data that support the findings of this study are available from the corresponding author, I.A. Pestana, upon reasonable request.
References
Altman N, Krzywinski M (2016) Regression diagnostics. Nat Methods 13:385–386. https://doi.org/10.1038/nmeth.3854
Agência Nacional de Águas (ANA), 2019. Rede Hidrometeorológica Nacional, Sistema HIDRO - Telemetria, Estaçao Humaitá, Código 15630000. Available at: http://www.snirh.gov.br/gestorpcd/gerarGrafico.aspx(Accessed 20 March 2022)(in Portuguese)
ANVISA. Agência Nacional de Vigilância Sanitária. 2013. Resolução - RDC Nº 42, de 29 de agosto de 2013. Dispõe sobre o Regulamento Técnico MERCOSUL sobre Limites Máximos de Contaminantes Inorgânicos em Alimentos. Available at: http://www.cidasc.sc.gov.br/defesasanitariaanimal/files/2021/08/RDC-n%C2%BA-42-de-29-de-agosto-de-2013.pdf(Accessed 20 March 2022) (in Portuguese)
ANVISA. Agência de Vigilância Sanitária. 2021. Instrução Normativa - IN N° 88. Estabelece os limites máximos tolerados (LMT) de contaminantes em alimentos.Available at: https://www.in.gov.br/en/web/dou/-/instrucao-normativa-in-n-88-de-26-de-marco-de-2021-311655598(Accessed 20 March 2022) (in Portuguese)
Arcagni M, Juncos R, Rizo A, Pavlin M, Fajon V, Arribére MA, Horvat M, Guevara SR (2018) Species- and habitat-specific bioaccumulation of total mercury and methylmercury in the food web of a deep oligotrophic lake. Sci Total Environ 612:1311–1319. https://doi.org/10.1016/j.scitotenv.2017.08.260
Azevedo LS, Pestana IA, da Costa Nery AF, Bastos WR, Souza CMM (2020) Mercury concentration in six fish guilds from a floodplain lake in western Amazonia: interaction between seasonality and feeding habits. Ecol Indic 111:106056. https://doi.org/10.1016/j.ecolind.2019.106056
Azevedo LS, Pestana IA, Almeida MG, Bastos WR, Souza CMM (2021) Do fish isotopic niches change in an Amazon floodplain lake over the hydrological regime? Ecol Freshw Fish 31(1):72–80. https://doi.org/10.1111/eff.12609
Barbosa-Jr F, Fillion M, Lemire M, Passos CJS, Rodrigues JL, Philibert A, Guimarães JR, Mergler D (2009) Elevated blood lead levels in a riverside population in the Brazilian Amazon. Environ Res 109(5):594–599. https://doi.org/10.1016/j.envres.2009.03.005
Bassey OB, Chukwu LO (2019) Health risk assessment of heavy metals in fish (Chrysichthys nigrodigitatus) from two lagoons in southwestern Nigeria. J Toxicol Risk Assess 5(2):027. https://doi.org/10.23937/2572-4061.1510027
Bastos WR, Gomes JPO, Oliveira RC, Almeida R, Nascimento EL, Bernardi JVE, Lacerda LD, Silveira EG, Pfeiffer WC (2006) Mercury in the environment and riverside population in the Madeira River Basin, Amazon. Braz Sci Total Environ 368(1):344–351. https://doi.org/10.1016/j.scitotenv.2005.09.048
Bastos WR, Malm O, Pfeifer WC, Cleary D (1998) Establishment and analytical quality control of laboratories for Hg determination in biological and geological samples in the Amazon, Brazil. Rev. Ciên. Cult. 50:255–260. Available at: https://biogeoquimica.unir.br/uploads/42029128/arquivos/Cien_1728929093.pdf(Accessed 20 March 2022)
Cardwell, R.D., DeForest, D.K., Brix, K.V., Adams, W.J. 2013. Do Cd, Cu, Ni, Pb, and Zn biomagnify in aquatic ecosystems?. In: Whitacre, D. (eds) Reviews of environmental contamination and toxicology Volume 226. Reviews of environmental contamination and toxicology, vol 226. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-6898-1_4
Carvalho, D.P. de, Almeida, R. de, Manzatto, Â.G., Freitas, O.B. de, Bastos, W.R., 2018. Dynamics of metals in lacustrine sediments: case study of the Madeira River, Amazon region. Brazilian J. Water Resour. 23. https://doi.org/10.1590/2318-0331.231820170026
Cella-Ribeiro A, Torrente-Vilara G, Lima-Filho JA, Doria CRC (2016) Ecologia e Biologia de peixes do Rio Madeira. EDUFRO, PortoVelho. 350. https://doi.org/10.47209/978-85-7764-086-7
de Almeida R, Bernardi JVE, Oliveira RC, de Carvalho DP, Manzatto AG, de Lacerda LD, Bastos WR (2014) Flood pulse and spatial dynamics of mercury in sediments in Puruzinho lake. Braz Amazon Acta Amaz 44:99–105. https://doi.org/10.1590/s0044-59672014000100010
Dolan LC, Flannery BM, Hoffman-Pennesi D, Gavelek A, Jones OE, Kanwal R, Wolpert B, Gensheimer K, Dennis S, Fitzpatrick S (2020) A review of the evidence to support interim reference level for dietary lead exposure in adults. Regul Toxicol Pharmacol 111:104579. https://doi.org/10.1016/j.yrtph.2020.104579
FAO. Food and Agriculture Organization. 2016. Fishery and aquaculture country profiles–the Republic of Maldives. Avaliable at: https://www.fao.org/figis/pdf/fishery/facp/MDV/en?title=FAO%20Fisheries%20%26(Accessed 20 March 2022)
FAO/WHO, 2014. Codex Alimentarius: guidelines for the simple evaluation of dietary exposure to food 605 additives CAC/GL 3–1989. Avaliable at: https://www.fao.org/input/download/standards/6/cxg_003e.pdf(Accessed 20 March 2022)
Feitosa IB, Huszar VLM, Domingues CD, Appel E, Paranhos R, Almeida RM, Branco CWC, Bastos WR, Sarmento H (2019) Plankton community interactions in an Amazonian floodplain lake, from bacteria to zooplankton. Hydrobiologia 831:55–70. https://doi.org/10.1007/s10750-018-3855-x
Flannery BM, Dolan LC, Hoffman-Pennesi D, Gavelek A, Jones OE, Kanwal R, Wolpert B, Gensheimer K, Dennis S, Fitzpatrick S (2020) US Food and Drug Administration’s interim reference levels for dietary lead exposure in children and women of childbearing age. Regul. Toxicol. Pharmacol. 110:104516. https://doi.org/10.1016/j.yrtph.2019.104516
Fox J, Weisberg S (2019) An {R} companion to applied regression, 3rd edn. Sage, Thousand Oaks CA, p 608. Availablet at: https://socialsciences.mcmaster.ca/jfox/Books/Companion/
Fry B (2006) Stable Isotope Ecology. New York, NY: Springer.308. https://doi.org/10.1007/0-387-33745-8
Guimarães JRD (2020) Mercury in the Amazon: problem or opportunity? A commentary on 30 years of research on the subject. Elementa: Science of the Anthropocene 8(1):032. https://doi.org/10.1525/elementa.032
Hacon S, Vega C, Hacon B, Carvalho DP, Almeida R, Barbosa R, Bastos IB (2010) Lead human health risk assessment case study: cassiterite mining in the western Brazilian Amazon. Braz Toxicol Letters 196:S66. https://doi.org/10.1016/j.toxlet.2010.03.252
Hiane PA, Leal Filho AF, Ramos Filho MM, Ramos MIL (2002) Teores de colesterol e lipídios totais em seis espécies de peixes capturados na região pantaneira do estado do Mato Grosso do Sul. B CEPPA 20(1):5–74. https://doi.org/10.5380/cep.v20i1.1136
Huser BJ, Fölster J, Köhler SJ (2012) Lead, zinc, and chromium concentrations in acidic headwater streams in Sweden explained by chemical, climatic, and land-use variations. Biogeosci 9(11):4323–4335. https://doi.org/10.5194/bg-9-4323-2012
Inhamuns AJ (2000) Composição de ácidos graxos de peixes de água doce da região amazônica brasilieira. PhD thesis. Universidade Estadual de Campinas.Repositório de produção científica e intelectual da Unicamp. Available at: http://repositorio.unicamp.br/jspui/handle/REPOSIP/255038(Accessed 20 March 2022) (in Portuguese)
Inhamuns AJ, Franco MRB (2008) EPA and DHA quantification in two species of freshwater fish from Central Amazonia. Food Chem 107:587–591. https://doi.org/10.1016/j.foodchem.2007.07.032
Ishitsuka K, Yamamoto-Hanada K, Yang L, Mezawa H, Konishi M, Saito-Abe M, Sasaki H, Nishizato M, Sato M, Koeda T, Ohya Y (2020) Association between blood lead exposure and mental health in pregnant women: results from the Japan environment and children’s study. Neurotoxicol 79:191–199. https://doi.org/10.1016/j.neuro.2020.06.003
Jakubus M (2020) Changes in lead and chromium contents in sewage sludge evaluated using both single extractants and sequential method. Environ Pollutants Bioavailab 32(1):87–99. https://doi.org/10.1080/26395940.2020.1778541
Jiang Z, Xu N, Liu B, Zhou L, Wang J, Wang C, Dai B, Xiong W (2018) Metal concentrations and risk assessment in water, sediment and economic fish species with various habitat preferences and trophic guilds from Lake Caizi, Southeast China. Ecotoxicol Environ Saf 157:1–8. https://doi.org/10.1016/j.ecoenv.2018.03.078
Khitalishvili K (2016) Monte Carlo simulation in R: basic example. Avaliable at: https://rpubs.com/Koba/Monte-Carlo-Basic-Example(Accessed 20 March 2022)
Lacerda LD, Salomons W (1998) Mercury from gold and silver mining: a chemical time bomb? Springer, Berlin, Heidelberg 147. https://doi.org/10.1007/978-3-642-58793-1
Lacerda LD, Malm O (2008) Mercury contamination in aquatic ecosystems: an analysis of the critical areas. Dossiê Água: Estudos Avançados 22(63):173–190. https://doi.org/10.1590/S0103-40142008000200011
Liu H, Liu G, Wang S, Zhou C, Yan Z, Da C (2018) Distribution of heavy metals, stable isotope ratios (δ13C and δ15N) and risk assessment of fish from the Yellow River Estuary, China. Chemosphere 208:731–739. https://doi.org/10.1016/j.chemosphere.2018.06.028
Longo RM, Ribeiro AÍ, Melo WJ (2011) Uso da adubação verde na recuperação de solosdegradados por mineração na floresta amazônica. Bragantia 70(1):139–146. https://doi.org/10.1590/S0006-87052011000100020(inPortuguese)
McConnaughey T, McRoy CP (1979) Food-web structure and the fractionation of carbon isotopes in the Bering Sea. Mar Biol 53(3):257–262. https://doi.org/10.1007/BF00952434
Melo, D., Roseno, T.F., Barros, W.M., Faria, R.A.P.G., Paglarini, C.S., Faria, P.B., Mariotto, S., Souza, X.R. 2019. Fatty acid profiles and cholesterol content of five species of pacu-pevas from the pantanal region of Mato Gross, Brazil. Journal of Food Composition and Analysis, 83. https://doi.org/10.1016/j.jfca.2019.103283
Moreira FR, Moreira JC (2004) A cinética do chumbo no organismo humano e sua importância para a saúde. Ciênc Saúde Coletiva 9(1):167–181. https://doi.org/10.1590/S1413-81232004000100017
Mushak P (2011) Lead exposure in human populations: lead intakes. Trace Met Other Contam Environ 10:217–241. https://doi.org/10.1016/B978-0-444-51554-4.00007-9
Nascimento LS, Silva LO, Azevedo SM, Almeida R, Almeida MG, Azevedo LS, Bastos WR, Souza CMM, Pestana IA (2022) Spatial-temporal dynamics of Cr in fish from Puruzinho lake (western Amazon) and dietary risk assessment. Chemosphere 300:134576. https://doi.org/10.1016/j.chemosphere.2022.134576
Nascimento EL, Gomes JPO, Almeida R, Bastos WR, Bernardi, JVE, Miyai RK (2007) Mercúrio no plâncton de um lago natural amazônico, lago Puruzinho (Brasil). J. Braz. Soc. Ecotoxicol, 2(1): 67–72. Available at: https://ecotoxbrasil.org.br/upload/0eafe803266347f3c0fe0bc4a216f5ea-10_v2n1.pdf(Accessed 20 March 2022) (in Portuguese)
Nash RDM, Valencia AH, Geffen AJ (2006) The origin of Fulton’s condition factor—setting the record straight. Fisheries 31(5):236–238.Available at:https://folk.uib.no/nfiag/nfiag/reprints/Nashetal%202006FISH.pdf (Accessed 20 March 2022)
Nriagu Jo, Pfeiffer WC, Malm O, Souza CMM, Mierle G (1992) Mercury pollution in Brazil. Nature 356(2):389–390. https://doi.org/10.1038/356389a0
Oliveira RC, Dórea JG, Bernardi JV, Bastos WR, Almeida R, Manzatto ÂG (2010) Fish consumption by traditional subsistence villagers of the Rio Madeira (Amazon): impact on hair mercury. Ann Hum Biol 37(5):629–642. https://doi.org/10.3109/03014460903525177
Pestana IA, Almeida MG, Bastos WR, Souza CMM (2019) Total Hg and methylmercury dynamics in a river-floodplain system in the western Amazon: influence of seasonality, organic matter and physical and chemical parameters. Sci Total Environ 656:388–399. https://doi.org/10.1016/j.scitotenv.2018.11.388
Pestana IA, Rezende CE, Almeida R, Lacerda LD, Bastos WR (2022) Let’s talk about mercury contamination in the Amazon (again): the case of the floating gold miners’ village on the Madeira River. In press, The Extractive Industries and Society. https://doi.org/10.1016/j.exis.2022.101122
Pfeiffer WC, Lacerda LD (1988) Mercury inputs into the Amazon region, Brazil. Environ Technol Lett 9(4):325–330. https://doi.org/10.1080/09593338809384573
Queiroz TKL, Naka KS, Mendes LCS, Costa BNS, Jesus IM, Câmara VM, Lima MO (2019) Human blood lead levels and the first evidence of environmental exposure to industrial pollutants in the Amazon. Int J Environ Res Public Health 16(17):3047. https://doi.org/10.3390/ijerph16173047
R Core Team, 2022. R: a language and environment for statistical computing. RFoundation for Statistical Computing. Austria, Vienna. Availablet at: http://www.R-project.org/
Ribeiro DRG, Faccin H, Molin TRD, Carvalho LM, Amado LL (2017) Metal and metalloid distribution in different environmental compartments of the middle Xingu River in the Amazon, Brazil. Sci Total Environ 605–606:66–74. https://doi.org/10.1016/j.scitotenv.2017.06.143
Ricker WE (1975) Computation and interpretation of biological statistics of fish populations. Bulletin of the Fisheries Research Board of Canada 191:1–382. Available at: https://waves-vagues.dfo-mpo.gc.ca/library-bibliotheque/1485.pdf (Accessed 20 March 2022)
Rudke AP, Souza VAS, Santos AM, Xavier ACF, Filho OCR, Martins JA (2020) Impact of mining activities on areas of environmental protection in the southwest of the Amazon: a GIS- and remote sensing-based assessment. J Environ Manage 263:110392. https://doi.org/10.1016/j.jenvman.2020.110392
Schifer TS, Bogusz-Jr S, Montano MAE (2005) Aspectos toxicológicos do chumbo. Infarma, (5):67–72.Available at: https://revistas.cff.org.br/infarma/article/view/273 (Accessed 20 March 2022) (in Portuguese)
Sioli H (1968) Hydrochemistry and geology in the Brazilian Amazon region. Amazoniana: Limnologia et Oecologia Regionalis Systematis Fluminis Amazonas, 1(3):267–277.Available at: http://hdl.handle.net/21.11116/0000-0004-5124-8 (Accessed 20 March 2022)
Sonoda DY, & Shirota R (2012) Consumo de pescado no Brasil fica abaixo da média internacional. Visão agrícola, 8(11):145–147. Available at: https://www.esalq.usp.br/visaoagricola/sites/default/files/va11-mercado-e-consumo01.pdf (Accessed 20 March 2022) (in Portuguese)
Sousa, E. A. D., Miranda, M. R., dos Santos, M. H., Costa Júnior, W. A., Lauthartte, L.C., Mussy, M. H., ... & Bastos, W. R. (2015). Assessment of trace metals in Amazonian fish exposed to untreated urban sewage: high chromium concentrations in fish tissues. Revista Ambiente & Água, 10(3), 499-509. https://doi.org/10.4136/ambi-agua.1594
Souza-Araujo J, Hussey N, Hauser-Davis RA, Rosa AH, Lima MO, Giarrizzo T (2022) Human risk assessment of toxic elements (As, Cd, Hg, Pb) in marine fish from the Amazon. Chemosphere 301:134575. https://doi.org/10.1016/j.chemosphere.2022.134575
Stierman B, Hauptman M, Woolf AD (2020) Severe lead poisoning requiring hospitalization: a case report. Curr Probl Pediatr Adolesc Health Care 50:1–5. https://doi.org/10.1016/j.cppeds.2020.100757
UNEP. 2010. United Nations Environment Programme. Final review of scientific information on lead. Available at: https://www.cms.int/sites/default/files/document/UNEP_GC26_INF_11_Add_1_Final_UNEP_Lead_review_and_apppendix_Dec_2010.pdf(Accessed 20 March 2022)
Venables WN, Ripley BD (2002) Modern applied statistics with S. Fourth Edition. Springer, New York. ISBN 0–387–95457–0.https://doi.org/10.1007/978-0-387-21706-2
Vries ANV, Govoni D, Árnason SH, Carlsson P (2020) Microplastic ingestion by fish: body size, condition factor and gut fullness are not related to the amount of plastics consumed. Mar Pollut Bull 151:110827. https://doi.org/10.1016/j.marpolbul.2019.110827
Yi Y, Tang C, Yi T, Yang Z, Zhang S (2017) Health risk assessment of heavy metals in fish and accumulation patterns in food web in the upper Yangtze River, China. Ecotoxicol Environ Saf 145:295–302. https://doi.org/10.1016/j.ecoenv.2017.07.022
Funding
This work was supported by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) through the CNPq/CTUniversal project (Grant no. 458977 2014–4). Inácio A. Pestana received support from Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ; grant numbers E-26/200.586/2022and E-26/210.350/2022).
Author information
Authors and Affiliations
Contributions
Sophia Menezes de Azevedo: writing (original draft and review and editing); Luiza Silva do Nascimento: writing (review and editing); Layra de Oliveira Silva: writing (review and editing); Marcelo Gomes de Almeida: validation and methodology; Lucas Silva Azevedo: writing (review and editing); Wendel Dias Constantino: writing (review and editing); Wanderley Rodrigues Bastos: resources and funding acquisition; Inácio Abreu Pestana: conceptualization, project administration, supervision, and writing (review and editing).
Corresponding author
Ethics declarations
Consent to publish
All the authors agreed with the content of this manuscript and gave explicit consent for submission.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Severine Le Faucheur
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Azevedo, S.M., do Nascimento, L.S., de Oliveira Silva, L. et al. Flood pulse as a driving force of Pb variation in four fish guilds from Puruzinho Lake (western Amazon). Environ Sci Pollut Res 30, 38728–38737 (2023). https://doi.org/10.1007/s11356-022-25015-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-25015-z