Abstract
In this review paper, the ill effects of pharmaceuticals (PhAs) on the environment and their adsorption on graphene oxide (GO) and graphene oxide-based (GO-based) nanomaterials have been summarised and discussed. The adsorption of prominent PhAs discussed herein includes beta-blockers (atenolol and propranolol), antibiotics (tetracycline, ciprofloxacin and sulfamethoxazole), pharmaceutically active compounds (carbamazepine) and analgesics such as diclofenac. The adsorption of PhAs strictly depends upon the experimental conditions such as pH, adsorbent and adsorbate concentrations, temperature, ionic strength, etc. To understand the adsorption mechanism and feasibility of the adsorption process, the adsorption isotherms, thermodynamics and kinetic studies were also considered. Except for some cases, GO and its derivatives show excellent adsorption capacities for PhAs, which is crucial for their applications in the environmental pollution cleanup.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Water is so crucial to human beings as well as animals and plants that their lives are impossible without it. But recently, it is becoming a hazard to human health as well as to the life of flora and fauna (Biziuk et al. 1996). In 2010, 80 % of the world’s total population was exposed to high threats of water pollution (Vörösmarty et al. 2010). One of the sources responsible for water pollution is the pharmaceuticals (PhAs) (Rivera-Utrilla et al. 2013). About 30–90 % of the PhAs remain undegradable in humans and animal’s bodies and are excreted as active compounds in the environment (Aksu 2005; Rivera-Utrilla et al. 2013). In this way, their continuous entry can pollute the environment, especially the drinking water, which can directly affect the human health (Sirés and Brillas 2012; Yuan et al. 2009).
The PhAs are added to the freshwater mainly through hospital wastes, pharmaceutical industries and therapeutic drugs. Similarly, human beings consume tons of the PhAs and especially antibiotics on the average rate of 100 to 200 × 106 kg per year (Wise 2002) to treat bacterial infections in humans, animals and plants (Martinez 2009). Because of their large-scale consumption, they are gradually discharged into domestic sewage water (Kümmerer et al. 1997), which falls either directly into freshwater or accumulated in the soil when the sewage sludge is used as fertilizer (Kinney et al. 2006). Similarly, due to their partial removal by wastewater treatment plants (WWTPs) and low natural degradation (Martucci et al. 2012), the PhAs are frequently found in natural water. Figure 1 shows a conceptual diagram of the sources, pathways and effects of PhAs on the environment and human beings. Up to now, over 200 PhAs have been detected in freshwaters alone (Sirés and Brillas 2012, Yuan et al. 2009). The representative PhAs that exist both in underground and surface water include painkillers, anti-inflammatory drugs, antibiotics, lipid regulators, antiulcer drugs, antihistamines, antidepressants, antiepileptics, lipid-lowering drugs, psychiatric drugs and β-blockers (Jelic et al. 2011; Lin et al. 2008). However, the largest group detected is the antibiotics followed by painkillers and antidepressants (Hughes et al. 2012). Some of them such as antibiotics, anti-inflammatories, antidepressants and analgesics were worse than others and can affect plants and animal’s life including human beings. The beta-blockers, also called beta-adrenergic antagonists, are generally used for the treatment of heart rhythm disorders, angina pectoris, hypertension, tachycardia or acute myocardial infarction (del Rosario Brunetto et al. 2015) and mainly found in various environmental water bodies, due to their poor removal by WWTPs (Fatta-Kassinos et al. 2011; Godoy et al. 2015).
The other commonly used antibiotics include carbamazepine (CBZ), doxorubicin hydrochloride (DOX), sulfonamides (SAs) such as sulfamethoxazole (SMX), sulfapyridine (SPY), sulfathiazole (STZ), etc. SAs are widely used as broad-spectrum antibiotics against many infectious diseases in humans as well as animals, to promote livestock growth. However, due to incomplete metabolism, a large fraction may often added to the environment in the form of urine or faeces (Díaz-Cruz et al. 2003; Sarmah et al. 2006), and in a very similar way to beta-blockers, they are also frequently detected in wastewaters, due to the partial removal by traditional sewage treatment plants (García-Galán et al. 2012; Göbel et al. 2007). The existence of the PhAs in drinking water is a potential threat for life, since little is known about the ill effects of these long-term existing compounds on human health (Kümmerer 2001; Stackelberg et al. 2004). Their detection and effective removal or lessening their effects on the environment is thus becoming an international issue for the environmental engineers (Kemper 2008, Khetan and Collins 2007; Kümmerer 2004; Stackelberg et al. 2004).
Environmental effects of PhAs
The presence of PhAs in the environment has been monitored and acknowledged as contaminants by a number of countries (Wang et al. 2016b). In 2014, Germany commissioned a global review of the PhAs in the environment and found that, of the 713 PhAs tested, 631 were found above their detection limits. These PhAs were found not only in surface waters such as lakes and rivers but also in groundwater, manure, soil and even in drinking waters (Weber et al. 2015). However, the PhA level in the environment is much lower than their therapeutic dose, and it is therefore difficult to study their effects on the ecosystem. Similarly, it is far from the public eyes to notice the affected organisms such as sea lice and algae because the effect is so small to go unnoticed if the researchers do not pay full attention to them. Unfortunately, we do not give attention until there is a population crash in big animals. One such accident happened in India in 1996 and 2007 when millions of vultures were killed by exposure to an anti-inflammatory drug, diclofenac (Swan et al. 2006). High doses of diclofenac were given to cattle to treat fever and pain. The cattle which were not recovered were left for vultures to feast on them. As the vultures of the genus Gyps are sensitive to diclofenac, therefore, 10–40 million vultures died of kidney failure, driving the bird in near extinction (Weber et al. 2015). Similar toxic effects have also been seen in Europe and Africa. It was observed that antidepressants can change the spawning behaviour of clams, cause disorder in snail’s movements and change the behaviour of crayfish in an aggressive way and also affect learning behaviours in cuttlefish (Lyons 2015). Similarly, ibuprofen (IBU), an anti-inflammatory drug, not only affects reproduction in fish but also causes delay in hatching of the eggs.
The most widely studied pharmaceutical that affects the environment is ethinylestradiol, which is an active ingredient of contraceptive tablets. It has been found in the laboratory that ethinylestradiol affects the sexual development in female fish. Similarly, intersex fish have been found in the downstream of many sewage plants around the world, which led them to near extinction (Jobling et al. 2005; Kidd et al. 2007). It is a common observation that a number of fish have been greatly reduced in the last 10–15 years which may be due to ethinylestradiol and other potential toxic pollutants. Some sedative drugs such as oxazepam can alter the behaviour and feeding of fish such as European perch, at as low concentration as 1.8 μg/L, and result in distortion of food web structure (Brodin et al. 2013). Similarly, low concentrations of antidepressants such as fluoxetine affect the behaviour of the American native fathead minnow fish (Weinberger and Klaper 2014) and cuttlefish (Di Poi et al. 2014) and also induce physiological changes in water fleas (Campos et al. 2012). In the same way, CBZ (an anti-epilepsy drug and mood stabiliser) alters brain physiology in Atlantic salmon even at low concentrations (7.85 μg/mL) (Hampel et al. 2014). The antibiotic ciprofloxacin (CIP) is widely used in infectious disease therapy (Hartmann et al. 1998; Martins et al. 2008). Unfortunately, it undergoes a slow biodegradation process (Hirsch et al. 1999) and causes many side effects such as stomatitis, leukopenia, and vomiting in human beings (Cox et al. 2002; Kümmerer et al. 2000a). In addition, the long-term existence of CIP in aqueous media develops resistance in bacteria (Diwan et al. 2010) and also produces undesirable changes in water quality (Chang et al. 2010).
The PhAs not only affect animals, birds and fish but also have drastic effects on plants, especially on the aquatic plants like blue-green algae, green algae and cyanobacteria. The PhAs affect the metabolic pathways of these plants and damage their chloroplast due to their homology with bacteria. Plants are generally more sensitive towards the PhAs such as fluoroquinolones which affect the replication of chloroplast (Brain et al. 2008). In a similar way, tetracyclines such as macrolides, p-aminoglycosides, lincosamides and pleuromutilins affect the transcription and translation and sulfonamides affect the metabolic pathways, such as folate biosynthesis, whereas triclosan affects the fatty acid biosynthesis. The other classes of the PhAs, such as statin-type blood lipid regulators, affect sterol biosynthesis (Brain et al. 2008).
PhAs are designed to destroy pathogenic microbes. However, apart from killing or weakening of the pathogenic microbes, they also have a direct impact on the growth, development and diversity of microbial population in aquatic and terrestrial ecosystem. In addition, they help pathogenic bacteria to develop antibiotic resistance in them (Drillia et al. 2005). The resistance of microbes against PhAs has brought many problems to health department such as the decrease in efficiency and effectiveness of antibiotics. In other words, it has decreased the number of effective antibiotics available for the treatment of infectious diseases in human beings. Sulfamethoxazole, for example, was a good antibacterial agent but now it is used in combination with trimethoprim to achieve the same purpose which it was doing alone (Drillia et al. 2005). If not eliminated at sewage treatment plants, the PhAs would eventually be a threat to human health. Endocrine disruptors, for instance, cause hormone-mimicking effect even at the level of nanograms per litre (Halling-Sørensen et al. 1998; Larsson et al. 1999). Thereby, it is crucial to eliminate the PhAs efficiently before their discharge into the environmental water.
On the basis of the abovementioned PhA-related potential threats, we focused on the efficient removal of PhAs from the environmental waters. Being simple and fast, the treatment processes using graphene oxide (GO) and GO-based nanomaterials as adsorbents are specially emphasised and encouraged for the efficient removal of PhAs from aqueous media.
Removal of PhAs
The removal of PhAs was sensed for the first time in 1999 when the feminization was observed in fish living downstream of the WWTPs (Larsson et al. 1999). The commonly used techniques to treat wastewaters for various contaminants include photocatalytic degradation (Chamjangali et al. 2015), membranes (Arami et al. 2006), biodegradation (Islam et al. 2015; Schiavon et al. 2015; Silva et al. 2014), ozonation (Carbajo et al. 2016; Lin and Liaw 2015), Fenton process (Khataee et al. 2016; Yuan et al. 2016), electrochemical techniques (Ni et al. 2015; Särkkä et al. 2015), reverse osmosis (Grobert 2007), chemical precipitation (Bruno et al. 1995; Tan et al. 2014; Tang and Reeder 2009), adsorption on chitosan beads (Adriano et al. 2005), adsorption on activated carbons (Kim et al. 2007), etc. Though the advanced oxidation processes (AOPs) (Sillanpää et al. 2011) are more efficient to degrade a number of contaminants, various oxidative intermediates are produced during the process that are much toxic than their parent compounds (Chamjangali et al. 2015). Similarly, AOPs are not only expensive but also difficult to operate (Moussavi et al. 2013). Many WWTPs use activated sludge processes in which bacteria are used to break down the organic molecules to achieve their goals in an economic way. However, the activated sludge processes reduce the biological oxygen demand (BOD) level, ammonia, phosphate as well as disinfection and show low efficiency to remove PhAs (Ternes et al. 2004). Moreover, most of the antibiotics have a stable naphthol ring in their structures and are recalcitrant to biodegradation (Zhao et al. 2010). On the other hand, adsorption is a cheap, easy and quick method in which the main step is usually the optimization of the adsorption capacity of an adsorbent by changing various physicochemical parameters such as pH, temperature, contact time, ionic strength, etc. (Liu et al. 2011; Liu et al. 2013a; Sun et al. 2013b). Moreover, the removal percentage of PhAs depends on the type of adsorbent and the physiochemical structures of PhAs (Buser et al. 1998a; Buser et al. 1998b, 1999, Kümmerer et al. 2000b; Michael et al. 2013; Mohle et al. 1999; Richardson and Bowron 1985; Stamatelatou et al. 2003; Stumpf et al. 1999; Ternes 1998; Zwiener and Frimmel 2003); therefore, structural similarity and the surface charge density which play a key role in the adsorption must be carefully watched while selecting an adsorbent for a certain type of pollutants.
A number of adsorbents such as activated carbon (Yu et al. 2005), carbon nanotubes (Yu et al. 2016a), clays (Al-Bayati 2010) and montmorillonite (Wu et al. 2010) were used for the removal of the PhAs. However, as compared with these adsorbents, GO and GO-based nanomaterials have shown high adsorption capacities towards the PhAs. GO is composed of highly functionalised two-dimensional several atoms thick sp2 hybridised carbon atoms that are arranged in hexagonal pattern (Jin et al. 2015; Yu et al. 2015b). The highest theoretical surface area, 2620–2700 m2/g, renders high adsorption capabilities to GO and GO-based nanomaterials (Yang et al. 2012). In our research group, for example, we applied GO as adsorbent for the removal of organic and inorganic pollutants from aqueous solutions and found that GO had the highest adsorption capacity among the natural and manmade nanomaterials (Hu et al. 2014; Sun et al. 2012; Zhao et al. 2011b, 2011c, 2012b). In the same way, GO and GO-based nanomaterials have been widely used as excellent adsorbents for the removal of the PhAs from aqueous solutions (Chen et al. 2015; Ma et al. 2015; Yu et al. 2015a). Moreover, the oxygen-containing functional groups (OCFGs) on GO surfaces can be replaced with other groups or molecules. Surface modification develops selectivity in GO and GO-based nanomaterials and further enhances their adsorption capabilities (Hu et al. 2014; Li et al. 2012; Sun et al. 2012, 2013a, 2015; Wang et al. 2015, 2016c; Yu et al. 2015c; Zhao et al. 2011a, 2011b, 2012a).
Up to now, many reviews have been summarised on the removal of PhAs with broad views. Kyzas et al. (2015a), for example, wrote a review on the removal of the PhAs on different adsorbents and gave a slight touch to all the adsorbents like activated carbons, clays, chitosan, silica, zeolites, graphene, etc. However, it is rather difficult to discuss the adsorption of pollutants on so many adsorbents in a single review. Similarly, Yu et al. (2015b) discussed the adsorption of a number of different classes of pollutants onto graphene nanosheets. In our research group, the application of graphene nanosheets in the elimination of organic pollutants was studied extensively and the results showed that graphene nanosheets or graphene-based nanosheets were very suitable materials in the efficient removal of organic pollutants because of the strong surface complexation and π–π interactions (Zhao et al. 2011a; Yu et al. 2016b; Zou et al. 2016a, 2016b). But in today’s period, every topic is broadened and much more investigated, so what is needed this time is to write reviews on specific topics. One such attempt was made by (Carmalin Sophia et al. 2016); however, synthesis, characterization and properties of graphene and graphene-based materials were mainly focused, whereas the adsorption of PhAs was given less attention. In the present review, we have discussed the adsorption of the PhAs on GO and GO-based nanomaterials in more detail, which are crucial for the application of GOs for the efficient elimination of the PhAs in wastewater treatment.
Synthesis and role of GO and GO-based nanomaterials in the adsorption of PhAs
Adsorption of PhAs on GO
Among the many adsorbents used so far, for the removal of PhAs (Chang et al. 2009a, 2009b; Figueroa et al. 2004; Oleszczuk et al. 2009; Prado et al. 2009), GO, due to its unique physicochemical properties, was the most efficient, which could be synthesised by a number of methods such as chemical vapour deposition (CVD) (Berger et al. 2006), electrical discharge (Subrahmanyam et al. 2009; Wu et al. 2009), unzipping of carbon nanotubes (Hirsch 2009; Jiao et al. 2009), epitaxial growth on silicon carbide (Aristov et al. 2010; Deng et al. 2010), total organic synthesis (Berresheim et al. 1999; Sakamoto et al. 2009; Simpson et al. 2002; Wu et al. 2003) and electrochemical synthesis (Guo et al. 2009; Shao et al. 2010; Zhou et al. 2009). However, these methods could be used to produce GO for special applications and were expensive. On the other hand, for laboratory usage, the GO is readily synthesised by the oxidation of graphite flakes, through Offeman’s (William et al. 1958) or the well-known Hummers method (Marcano et al. 2010); however, it might produce some toxic gases such as NO2 and N2O4 and large amount of heat, which may lead to explosion. Moreover, the health and ecosystem risks, due to the release of GO and GO-based nanomaterials into the environment via discharge from manufacturing sites and deterioration from composite materials, are rapidly an emerging environmental issue and have attracted more concern as its toxicity evidence is accumulating (Hu and Zhou 2013).
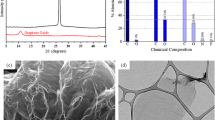
As mentioned earlier, being a pollutant and their ill effects on the environment, the PhAs were crucial to eliminate from the environment and have attracted the attention of many researchers. The GO was extensively investigated for the removal of commonly used PhAs (Table 2), especially those with aromatic centres, such as tetracycline (TCs), CIP, SAs, etc. As shown in Table 1, GO and a number of the PhAs have many common features, in terms of aromatic ring and functional groups, which is why it can be used as efficient adsorbent for them. One such attempt was made by Gao et al. (2012), who prepared GO by modified Hummers method and characterization indicated that GO surface was composed of few atoms that are thick, wrinkled and layer by layer folded structures of uniform thickness, which assigned high surface area to GO. The as-prepared GO was applied for the removal of representative tetracyclines, such as TC, oxytetracycline (OTC) and doxycycline (DOC), from aqueous solutions. In a similar way, Ghadim et al. (2013) further investigated GO for the aqueous elimination of TC, and due structural similarities with GO (Table 1), considerable adsorption results were obtained. The mass formed by sticking of TC with GO was porous in nature and increased the chances for further adsorption. In contrast, water could form hydrogen bonds with OCFGs, present on the surface of GO, and reduce the adsorption sites for TC. Nam et al. (2015) reported that the adsorption ability of GO can be increased further by sonication, which exfoliated the GO sheets, reduced some of the OCFGs and also decreased the hydrodynamic radius as compared with pristine GO. Thus, large amounts of TC (313.0 mg/g), OTC (312.0 mg/g) and DOC (398.0 mg/g) were adsorbed with extremely small amount of GO (0.181 mg/mL), with a time span of 90 min. Moussavi et al. (2016) oxidised graphite two times, first with Hummers method and then with Offeman’s method and achieved a very efficient adsorbent for the removal of acetaminophen (APAP). The adsorption capacity of this adsorbent was very high and removed 704.0 mg/g of APAP with the expense of small amount of DGO (20 mg/L).
As a matter of fact, the PhAs exist in mixture form, both in the surface as well as underground water bodies. A number of researchers, therefore, focused on the coadsorption of the PhAs (Cai and Larese-Casanova 2014; Nam et al. 2015; Wang et al. 2013). Three types of GOs, namely GO-C (xGnp-C-750, XG Sciences, Inc.), GO-M (xGnp-M-25, XG Sciences, Inc.) and GO-A (N006-010-P, Angstron Materials, Inc.), were applied for the coadsorption of CBZ and EDCs (Cai and Larese-Casanova 2014). Due to high surface area and the presence of high number of strongly and moderately electron activating phenolic and ether groups, GO-C (SSA = 720 m2/g) showed high adsorption capacity for CBZ, as compared to GO-M (SSA = 120 m2/g) and GO-A (SSA = 100 m2/g), which is necessary for its application in real-life processes.
The GO was successfully used for the efficient removal of PhAs; however, the current energy usage of GO production was estimated to be 1100 MJ/kg, which was about 50-fold greater than that of activated carbons (Arvidsson et al. 2014). Its costly and time-consuming preparation, exhausting exfoliation and separation and potential secondary pollution were some of the challenging problems associated with the GO. Thus, more efforts are needed to produce low-cost high-surface-area GO at commercial scale.
Adsorption of PhAs on rGO
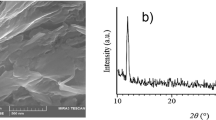
To date, reduction was one of the most important reaction of GO, which can be achieved by a number of ways such as electrochemical (Zhou et al. 2009) or thermal (Kaniyoor et al. 2010; McAllister et al. 2007; Schniepp et al. 2006); however, chemical reduction was more practical (Amarnath et al. 2011; Dreyer et al. 2010; Liu et al. 2012a, 2013b; Pei and Cheng 2012), in which the aqueous colloidal dispersion of GO can be reduced with hydrazine (N2H4), sodium borohydride (NaBH4) and lithium aluminium hydride (LiAlH4). N2H4 was used as an excellent reducing agent (Stankovich et al. 2007); however, the introduction of toxic heteroatomic impurities somewhat limits its practical use. NaBH4, the most widely used reducing agent in research laboratories, was exploited by Gao et al. (2009), for the total reduction of GO. However, unlike N2H4, NaBH4 produced additional alcohols as principal impurities (Shin et al. 2009). LiAlH4, a strong reducing agent, was able to reduce carbonyl, ester, epoxy and carboxylic functionalities to hydroxyl groups (Ambrosi et al. 2012). On the other hand, Fernandez-Merino et al. (2010) found ascorbic acid as more safe and cheap reducing agent for GO, and the reducing capability was comparable to that of hydrazine. Furthermore, unlike GO with high negative charge density due to the presence of OCFGs, which mainly discourage the adsorption of organic molecules with negative charge (Ramesha et al. 2011; Ren et al. 2013), the reduction of GO can significantly reduce the intensity of oxygenated functional groups (Kim et al. 2014; Liu et al. 2012b), can favour the electrostatic interactions as well as hydrogen bonds between negatively charged PhAs such as s-DCF and the rGO surfaces (Jauris et al. 2016) and could remove 59.67 mg/g. In addition, Yu et al. (2015a) found that the adsorption efficiency was directly related to the properties of an adsorbent, such as specific surface area, porosity, pore diameter and functional groups. They found that pristine graphene expressed low adsorption capability for CIP (145.9 mg/g). However, the one-step hydrothermal reduction of graphene was used to increase not only the porosity but also the SSA equal to 2630 m2/g (Ma et al. 2015). Because of the largest surface area and porous nature, it could adsorb 235.6 mg/g of CIP, which was necessary for the efficient elimination of CIP or other similar organic pollutants, from aqueous solutions. Similarly, the presence of π-electron system along with some OCFGs (Fig. 2c), due to the partial reduction of GO (Luo et al. 2011a; Mao et al. 2012), rendered rGOs a hydrophilic–lipophilic nature and were used as super mixed-mode adsorbents for the compounds with a wide polarity range (Khataee et al. 2016; Mohle et al. 1999; Yuan et al. 2016). Liu et al. (2014) prepared rGO1 by reducing GO (obtained from 300 mesh graphite) and rGO2 by reducing GO (obtained from 100 mesh graphite), with ammoniacal solution of hydrazine. The rGOs were applied for the removal of ketoprofen (KEP) and CBZ, and the maximum adsorption calculated for rGO1 (SSA = 331 m2/g) was 60.0 mg/g for KEP and 120.0 mg/g for CBZ, while in case of rGO2 (SSA = 325 m2/g), it was 55.0 mg/g for KEP and 95.0 mg/g for CBZ. One year later, the same group further investigated rGO1 and rGO2 for the aqueous removal of two SAs (SPY and STZ), and the maximum adsorption capacity of rGO1 was 165.0 mg/g for STZ and 138.0 mg/g for SPY, and in case of rGO2, it was 142.0 mg/g for STZ and 117.0 mg/g for SPY (Liu et al. 2015). At the same time, it was found that the naturally dissolved organic matter (DOM), i.e. humic acid (HA), bovine serum albumin (BSA) and sodium dodecyl benzene sulfonate (SDBS), had no considerable effect on the adsorption properties of rGOs, which was beneficial for the potential application of rGOs in environmental water treatment.
Adsorption of PhAs on modified GO
Due to the presence of OCFGs and smallest particle size, pristine GO was facing a serious problem of aggregation and challenging separation from the solution, after its application. However, the surface functional groups allow modification (Fig. 2d) and can be replaced or bonded with suitable moieties to assign desirable properties to GO. For instance, GO can be modified with magnetic iron nanoparticles to produce magnetic properties in it, which consequently makes its separation process easy, cost-effective and also avoids the possible secondary pollution associated with GO (Kerkez-Kuyumcu et al. 2016). Our research group achieved a deep black suspension of GO functionalised magnetic nanoparticles (GO-MPs) after the 1-h ultra-sonication of the in situ coprecipitated ammoniacal mixture of Fe2+/Fe3+, with an irregular shape and BET surface area of 142.36 m2/g (Li et al. 2012; Liu et al. 2011; Yang et al. 2012). The as-prepared GO-MPs were applied for the release of four tetracyclines, namely TC, OTC, DOC and chlortetracycline (CTC). Beside its easy separation, it was worth noting that ionic strength and solution pH had no effect on the adsorption behaviour of GO-MPs, which allowed its use for the removal of TCs from aqueous solutions under the environmental conditions. However, the adsorption study indicated that functionalization of GO with Fe3O4 not only decreased its surface area but also the adsorption efficiency of the GO (Lin et al. 2013). The low adsorption capacity of GO-MPs, 39.1, 45.0, 42.6 and 35.5 mg/g for TC, OTC, CTC and DOC, respectively, was ascribed to the low specific surface area and less interactions of Fe3O4 with the pollutants (Jin et al. 2015).
Different attempts were made to modify the GO surface to make it easily separable as well as to increase its adsorption ability. Wu et al. (2013) modified GO with calcium alginate (CA) and obtained a novel adsorbent, the GO/poly(acrylic acid) grafted with chitosan (GO/CA), which could be drawn in the form of fine fibres by using wet spinning technique and were used successfully for the rapid elimination of CIP from aqueous solutions. The adsorption mechanism was sensitive to pH, and maximum adsorption was achieved in acidic medium. The GO/CA fibres were easy to separate mechinically without any centrifugation and produced no secondary pollution, which was crucial for the practical applications of GO/CA in environmental pollution cleanup. However, the turn by turn complex method of preparation and high cost limits the application of GO and its derivatives for the separation of pollutants in bulk. These challenging problems attracted the attentions of many researchers. The cost can be reduced indirectly by creating reusability in the adsorbents. One such adsorbent, GO/poly(acrylic acid) grafted with chitosan (GO/CSA), which could be recycled, was introduced for the first time by Kyzas et al. (2014) for the efficient removal of dorzo with maximum adsorption capacity of 447 mg/g and lost only 10 % of its adsorption ability in 10 cycles, which may recommend its application for the treatment of dorzo contaminated wastewater.
Effect of environmental conditions
Generally, the adsorption behaviour of PhAs on GO and GO-based nanomaterials can be described by batch experiments in various conditions, such as solution pH, ionic strength, contact time, concentration of the adsorbent and adsorbate, temperature, etc. Due to the importance of PhAs in environment, evaluation of the migration, transfer, accumulation and their effects on the ecosystem was important in environmental pollution management.
Among the various experimental parameters, the effect of solution pH was very important, which could be used to optimise the adsorption capacity of an adsorbent. Generally, the pH changes from one to the other environment and could affect the surface properties of the adsorbent and adsorbate species. The pH values of most natural environments usually vary between 5.0 and 9.0, and the adsorption of PhAs, such as CIP, DOX, TCs and SAs, on GO and GO-based nanomaterials can be conducted in this pH range (Gao et al. 2012; Liu et al. 2015; Usca et al. 2015). The adsorption of PhAs was increased by changing the pH from acidic to neutral, whereas a decrease in the adsorption capacity was found, when the pH was moved to more basic side (Liu et al. 2015; Wu et al. 2013), and the pH-dependent behaviour can be explained by the ionization process of the adsorbent and adsorbate. Under more acidic or basic pH conditions, repulsive forces arise between the cationic or anionic species of the adsorbate and the adsorbents, which lead to a decrease in the adsorption capacity. Under acidic conditions, PhAs, such as dorzo, protonate and exist as cation, whereas the adsorbent surface was negatively charged, which is favourable for the efficient adsorption of dorzo via electrostatic interactions, and the percent removal calculated was 52 % on GO, 68 % on CSA and 92 % on GO/CSA surface, as shown in Fig. 3a. It assumes that the adsorption properties of GO and GO-based nanomaterials are strongly dependent to solution pH.
Adsorption evaluation. a Effect of pH on the efficiency of dorzo removal from solution, b dorzo adsorption isotherms (at 25, 45 and 65 °C) for GO, CSA and GO/CSA, respectively, (solid lines represent the fitting to L–F equation) and c reuse cycles of GO, CSA and GO/CSA for dorzo adsorption (Kyzas et al. 2014)
The adsorption mechanism of PhAs and their rate controlling steps such as chemical reaction, mass transport and diffusion-controlled processes can be explained by kinetic models such as pseudo-first-order, pseudo-second-order, Elovich equation and intra-particle diffusion models. For example, the pristine graphene could remove approximately 28 % of CIP in 60 min from aqueous solutions, whereas the highly sonicated GO expressed a higher adsorption capacity of 74 % within 60 min, and the maximum adsorption capacity was calculated to be 379.0 mg/g (C o = 20 mg/L) (Chen et al. 2015). Furthermore, the CIP adsorption data obtained with pristine graphene and sonicated GO could also be fitted by the pseudo-second-order kinetic model, and the activity towards CIP removal by the sonicated GO was about 2.6 times as high as that of the pristine graphene, which expressed that the adsorption process was mainly due to physical interactions, because of the high sorption capacity of sonicated GO, which was necessary for the potential application of GO in environmental pollution cleanup. The removal of TC, OTC and DOC by GO was also fitted better with pseudo-second-order kinetics, and the high adsorption rate showed the adsorption was governed by strong chemisorption and π–π interactions (Ma et al. 2015; Wang et al. 2016a; Wu et al. 2013).
Generally, temperature can alter the adsorption rate and energy of the reaction system. The Freundlich, Langmuir, Temkin and D-R adsorption models are commonly used to represent the sorption of PhAs on GO and GO-based nanomaterials. The thermodynamic studies give the values of important parameters, such as Gibb’s free energy (ΔG 0), entropy (ΔS 0) and enthalpy change (ΔH 0). Understanding the thermodynamic behaviour of PhAs on GO and GO-based nanomaterials is generally conclusive, and the adsorption isotherms can be used to describe the relationship between the concentration of adsorbate in the solid and liquid phase, which is indispensable in the optimization of adsorption capacity of the adsorbents (Kwon and Lee 2015; Rostamian and Behnejad 2016; Chowdhury and Balasubramanian 2014). The adsorption of TCs on GO follows Freundlich as well as Langmuir isotherms (Ghadim et al. 2013; Zhang et al. 2011), while the exothermic and spontaneous nature of the adsorption process showed favourable attractive forces between TCs and the adsorbent. The adsorption isotherms of CIP on graphite follow Freundlich model, which suggested that different sites with several adsorption energy are involved; while in some cases, strong intermolecular forces were involved, and spontaneous adsorption process was confirmed by the negative △G 0 value. Similarly, the negative △H 0 and positive △S 0 values expressed that the adsorption of the PhAs was exothermic and random at solid–liquid interface (Wang et al. 2013). The adsorption isotherms of dorzo by GO and its composites were studied at various temperatures (Fig. 3b). In general, the temperature has a positive effect on the adsorption capacity of GO and GO-based nanomaterials, and it was found that the adsorption process was spontaneous and endothermic, which is useful for the practical application of GO-based adsorbents in wastewater treatment (Kyzas et al. 2014).
The dose amount of the adsorbent is also an important factor, which affects the removal efficiency of pollutants that is why the optimization of dose amount of the adsorbent for the efficient adsorption in the natural environment is convenient for its cost-effective application. According to Kerkez-kuyumcu et al. (2016), a dose of 5.4 mg M-GNPs can be used to remove 84 % of amoxicillin (AA) from aqueous solutions, at initial concentration of 10 mg/L of AA. In another study, the effect of GNPs mass on the adsorption capacity of aspirin (ASA), caffeine (CAF) and APAP was studied (Al-Khateeb et al. 2014), and using 5.0 mg of GNPs, the percent adsorption was 45 % for ASA, 97 % for CAF and 77 % for APAP. However, by increasing adsorbent’s mass from 5.0 to 10.0 mg, the percent adsorption was reached to 62 % for ASA, 98 % for CAF and 85 % for APAP, within 20 min, which indicated the fast transfer of pollutants on the GNP surface. Thus, large amount of the pollutants could readily be adsorbed with small amount of the adsorbent, which was crucial for the PhA-contaminated wastewater treatment.
The effect of coexisting oxyanions and cations on the adsorption of PhAs by GO and GO-based nanomaterials was widely investigated (Ghadim et al. 2013). The overall effect of ionic strength is positive, when it exceeds the driving force of competitive adsorption, otherwise negative. Moreover, an increase in the effect of the ionic strength was observed when the electrolyte was changed from monovalent to divalent (Chen et al. 2015). For example, during the adsorption of CIP, the cations such as Na+ and Ca2+ can compete with CIP and reduce its adsorption by 37 and 41 %, at a concentration of 200 and 10 mM, respectively (Wang et al. 2016a). In general, the effect of cations on the adsorption of different organic pollutants is rather complex depending on the structure and properties of the adsorbent and adsorbate. Equally compared with the effect of solution pH, ionic strength was found to be insignificant to the adsorption process.
Adsorption–desorption mechanism
The GO and GO-based nanomaterials have been widely investigated for the treatment of PhAs and showed incredible potentials and applications for the aggregation and efficient removal of PhAs. The adsorption process was a multi-step procedure of carrying the adsorbate molecules from liquid to solid phase (Rostamian and Behnejad 2016). The adsorption mechanism of GO and GO-based nanomaterials was not very simple and altered from one to the other type of contaminant. However, mechanistic study of PhAs adsorption by GO and GO-based nanomaterials was very important in explaining the changes occurring on the adsorbent surface, which further assisted the optimization of the adsorption–desorption conditions (Msagati and Nindi 2004; Shao et al. 2005). The possible interaction mechanism of PhAs on GO and GO-based nanomaterials was summarised in Table 2 (under the column adsorption mechanism). It indicated that PhAs showed different adsorption interactions with GO and GO-based nanomaterials ranging from physical forces to chemical interactions. Figure 4 summarises the potentially different and various adsorption interactions of PhAs onto GO and GO-based nanomaterials. It revealed that the main adsorption interactions of PhAs with GO and GO-based nanomaterials were π–π interactions (Ghadim et al. 2013), H-bonding (Jiang et al. 2016), hydrophobic interactions (Moussavi et al. 2016), cation-π bonding and electrostatic forces (Gao et al. 2012) and π–π stacking (Kerkez-Kuyumcu et al. 2016).
Both the internal properties and the surface morphology of the adsorptive materials play an important role in the adsorption of PhAs as those materials have heterogeneous surface areas due to the coexisting heterogeneous groups and expressed different adsorption mechanisms. It was well proposed that the adsorption of PhAs-NH2 (SMX, TCs), on different GO-based adsorbents (GNPs, graphene oxide nanoplatelets (GONPs), rGOs), was dependent on the amount of graphitization, which indicated that π–π EDA interactions between the adsorbate molecules and the graphite surfaces of the GOs were the principal adsorption mechanism (Banerjee et al. 2016, Gao et al. 2012, Rostamian and Behnejad 2016). According to Luo et al. (2011b), SAs were amphoteric and behaved as weak acids in basic medium and vice versa. Due to the protonation–deprotonation of aniline and sulfonylamido groups, SAs exist in cationic form as SA+, in acidic medium, and as anion in the form of SA−, in basic medium (Zhong et al. 2015), which played an important role in the adsorption process. In acidic medium, for example, the sorption of SMX on GO was controlled by the specific driving force, π–π EDA interaction of the electron rich graphite surface and the protonated aniline ring of SMX, referring to as cation-π bonding or π+–π EDA interactions, rather than electrostatic cation exchange. However, in basic medium, the sorption of the anionic SMX species was increased and regulated by the predominant charge-assisted H-bonding (Nam et al. 2015). Moreover, the DFT calculations indicated that the intermolecular distance between DCF and SMX with GO was ~3.5 Å. However, one of the amine aromatic rings of SMX was oriented parallel, while the second ring was oriented away from the GO surface, whereas both the aromatic rings of GO-DCF complex were oriented towards the surface of the GO, maximizing π–π EDA interactions with GO. Similarly, the low adsorption energy of DCF (−18.8 kcal/mol) than SMX (−15.9 kcal/mol) and high surface area exposure to the GO resulted in increased π–π EDA interactions and dispersion forces between the GO and DCF.
Moreover, Geiser et al. (2005) found that antibiotics usually exist as neutral molecules at pH equal to or near to their pK a values and were more hydrophobic than their corresponding ions. Therefore, STZ (pK a1 = 2.4, pK a2 = 7.1) and SPY (pK a1 = 2.3, pK a2 = 8.4) were neutral at pH = 3.0–5.0 and their adsorption on rGOs was governed by strong hydrophobic and π–π interactions (Liu et al. 2015). In a similar way, CIP is a zwitterionic compound and shows two pK a values (pK a1 = 6.1 and pK a2 = 8.7). It exists in cationic form at pH < pK a1 and as anion at pH > pK a2 (Ma et al. 2015). Similarly, the zeta potential value for GO/CA was 3.7. At pH > pHpzc, the surface of GO/CA fibres was negatively charged while at this pH CIP existed in cationic form and, electrostatically, could be adsorbed easily by the negatively charged GO/CA.
It was mentioned that both electron density and aromatic nature of CIP enhanced their adsorption on GO and increased the probability of π–π adsorbate–adsorbent dispersion interactions. Hence, the adsorption process was mainly determined by the adsorbent–adsorbate dispersion interactions (Tang et al. 2013). Yu et al. (2015a) reported that KOH-modified graphene showed similar mechanism, i.e. H-bonding and π–π EDA interactions. The adsorption of CBZ was best explained by the π–π stacking of the planar conjugated π-electron system of CBZ that overlapped with the surface π-electrons of GO in a favourably attractive geometric configuration. In addition, the π–π interactions of GO was enhanced by the strongly and moderately electron activating phenolic and ether groups. These activating groups increased the π-electron density of GO. Due to the π-electron withdrawing ability of the amide group, CBZ was proposed as π-electron acceptor and facilitated the formation of π–π electron donor–acceptor complex (Cai and Larese-Casanova 2014). However, CBZ was highly soluble in ethanol, which could decrease the π–π EDA interactions between CBZ and GO and desorbed 93 % of it. In a similar way, the GONPs, used for the adsorption of IBU, could be regenerated with nitric acid and lost only 3 % of its adsorption ability in 10 cycles (Banerjee et al. 2016). Figure 3c shows that GO did not exhibit a promising cycling behaviour and lost 25 % in 10 cycles (dorzo removal = 52 % in first cycle, 40 % in third cycle, 27 % in fifth cycle, 21 % in seventh cycle and 15 % in 10th cycle). A higher regeneration ability was found for CSA (68, 65, 61, 58 and 55 %, respectively), which lost 13 % of its adsorption ability, whereas the GO/CSA composite showed a balanced capacity losing only 10 % in 10 cycles. These results suggest that GO/CSA is a promising adsorbent for practical application in wastewater treatment technology (Kyzas et al. 2014). Kerkez-kuyumcu et al. (2016) restored M-GNPs with 0.15 M NaOH and recycled for four times, without any considerable loss in adsorption ability. Similarly, the solubility of ATL and PRO was high in methanol, which weakens the H-bonding between them and GO and can be used as excellent desorption solvent to regenerate GO, to avoid secondary pollution (Kyzas et al. 2015b), which is an important requirement of today’s period.
Comparative adsorption study
Due to high specific surface area and favourable structural features, GO has expressed excellent adsorption properties. The adsorption properties of GO were dependent on the experimental conditions and the nature of the adsorbate. The adsorption capacities of different adsorbents can be compared only towards the same adsorbate. Yu et al. (2015a), for example, investigated the adsorption of CIP on pristine graphene and G-KOH. The maximum adsorption capacity of pristine graphene was 145.9 mg/g. However, the alkali activation of pristine graphene (SSA = 138 m2/g) increased the adsorption sites, SSA (512 m2/g), pore volume and subsequently adsorption capacity (194.6 mg/g for CIP). Similarly, the thermal reduction of pristine graphene further increased its pore volume and adsorption capacity from 145.9 to 235.6 mg/g, for CIP (Ma et al. 2015). Furthermore, sonication can be used to increase the adsorption capacity of pristine GO, and the adsorption capacity calculated for CIP was 379.0 mg/g (Chen et al. 2015). On the other hand, the adsorption capacity of modified GO, for CIP, was very low. The adsorption capacity of GO/CA, for example, was 66.0 mg/g (Wu et al. 2013), whereas R-GOM could adsorb only 18.9 mg/g of CIP (Tang et al. 2013).
According to Gao et al. (2012), the highest adsorption capacity of GO, (398.0 mg/g for DOC, 313.0 mg/g for TC and 312.0 mg/g for OTC) compared to commonly used adsorbents such as MWNT10a (190.0 mg/g) (Oleszczuk et al. 2009), rectorite (140.0 mg/g) (Chang et al. 2009a), palygorskite (99.0 mg/g) (Chang et al. 2009b), activated sludge (72.0 mg/g) (Prado et al. 2009) and montmorillonite (54.0 mg/g) (Figueroa et al. 2004), was attributed to its highest surface area, 2620–2700 m2/g, large number of OCFGs and structural fitness to TCs (Sun et al. 2015; Yang et al. 2012; Yu et al. 2015b). The adsorption ability of GO and GO-based nanomaterials, towards CIP, was compared with different adsorbents and graphically summarised in Fig. 5. It indicated that except activated carbon and montmorillonite, the average adsorption capacity of GO and GO-based nanomaterials was very high, which recommends GO and GO-based nanomaterials for their commercial scale application in the environmental pollution management.
Comparison of GO and GO-based nanomaterials with commonly used adsorbents towards the adsorption of CIP. AC (Carabineiro et al. 2011; Carabineiro et al. 2012; Gao et al. 2015; Genç and Dogan 2015; Huang et al. 2014), MMCC (Shi et al. 2013), BFs (Wu et al. 2013), CX (Carabineiro et al. 2011; Carabineiro et al. 2012), CNTs (Carabineiro et al. 2011), CPC (El-Shafey et al. 2012), birnessite (Jiang et al. 2013), Mont (Wang et al. 2011; Wang et al. 2010), rectorite (Wang et al. 2011), MCGO (Wang et al. 2016a), GO (Chen et al. 2015), GO/CA (Wu et al. 2013), GH (Ma et al. 2015), graphene (Yu et al. 2015a), RGO-M (Tang et al. 2013) and G-KOH (Yu et al. 2015a). Note: All the references are given in the order from front to back row in the graph
Conclusion and perspective
This review summarised the adsorption of different classes of PhAs such as antibiotics, beta-blockers, endocrine disrupters and other pharmaceutically active compounds (CBZ) and analgesics such as DCF. The GO and GO-based nanomaterials are successfully used for the adsorption of prominent antibiotics such as CIP, SMX, CBZ, TCs, etc. and some commonly used PhAs like IBU, ASA, APAP, etc. Besides pristine GO, reduced GOs and the modified GOs play an important role in the purification of water. Keeping in view, the use of ease and rapid separation, Fe3O4/GO magnetic nanoparticles are of special importance. Among the GO-based nanomaterials, GO shows the highest adsorption capacity for the PhAs, which can be attributed to its highest theoretical surface area. In most cases, GO-based nanomaterials show high adsorption capacities than activated carbons, carbon nanotubes (CNTs) and others. The adsorption mechanism depends mostly on the structures and functional groups of the adsorbents. Generally, the adsorption process is affected by temperature, contact time, concentration of the adsorbent and adsorbate, ionic strength and pH of the medium. Moreover, the naturally occurring DOM, i.e. HA, BSA and sodium alginate, together with SDBS usually bring no considerable effect on the adsorption properties of GO-based adsorbents as compared to CNTs and graphite. Though in some cases the adsorption of the PhAs is observed over a wide range of pH, most of them prefer slightly acidic conditions. The adsorption of the PhAs onto GO and GO-based nanomaterials occurs mainly through π–π interactions, cation-π bonding, electrostatic forces and H-bonding.
Apart from their advantages, it is difficult to regenerate GO and GO-based nanomaterials and are usually discarded which may lead to potential secondary pollution. It is therefore crucial to study the physicochemical behaviour of the GO and GO-based nanomaterials in the natural environment, to evaluate their behaviour such as aggregation, deposition, sorption and migration in aqueous solutions. However, some GO-based adsorbents such as G-KOH and Fe3O4/GO magnetic nanoparticles can be separated easily from the solution and do not produce secondary pollution. High cost and reusability are the two main problems associated with GO and GO-based nanomaterials. It is therefore necessary to produce reusability in GO and GO-based nanomaterial to reduce the price. Similarly, the preparation and then purification of GO and GO-based nanomaterials are an exhausting and time-consuming process. So far, large-scale production, high cost and reusability are some of the unresolved problems associated with GO and GO-based nanomaterials, which can hinder the use of GO and GO-based nanomaterials for practical use in the environmental pollution management. However, keeping in view the rapid growth and development in science and technology, such problems are expected to be solved in the near future, which is an important factor for the potential application of GO and GO-based nanomaterials on commercial scale.
Abbreviations
- AA:
-
Amoxicillin
- AC:
-
Activated carbon
- AOPs:
-
Advanced oxidation processes
- APAP:
-
Acetaminophen
- ASA:
-
Aspirin
- ATL:
-
Atenolol
- BFs:
-
Biomass fibres
- BOD:
-
Biological oxygen demand
- BSA:
-
Bovine serum albumin
- CA:
-
Calcium alginate
- CAF:
-
Caffeine
- CBZ:
-
Carbamazepine
- CIP:
-
Ciprofloxacin
- CNTs:
-
Carbon nanotubes
- CPC:
-
Chemically prepared carbon
- CSC:
-
China Scholarship Council
- CTC:
-
Chlortetracycline
- CVD:
-
Chemical vapour deposition
- CX:
-
Carbon xerogel
- DOC:
-
Doxycycline
- DOM:
-
Dissolved organic matter
- Dorzo:
-
Dorzolamide
- DOX:
-
Doxorubicin hydrochloride
- D-R:
-
Dubinin–Radushkevich
- EDCs:
-
Endocrine disrupting compounds
- GO/CSA:
-
GO/poly(acrylic acid) grafted with chitosan
- GO:
-
Graphene oxide
- GO-MPs:
-
Graphene oxide functionalized magnetic nanoparticles
- GONPs:
-
Graphene oxide nanoplatelets
- HA:
-
Humic acid
- IBU:
-
Ibuprofen
- IHM:
-
Improved Hummer’s method
- KEP:
-
Ketoprofen
- MCGO:
-
Magnetic chitosan-grafted graphene oxide nanocomposite
- MHM:
-
Modified Hummer’s method
- MMCC:
-
Magnetic mesoporous carbon composite
- Mont:
-
Montmorillonite
- NOR:
-
Norfloxacin
- OCFGs:
-
Oxygen-containing functional groups
- OTC:
-
Oxytetracycline
- PhAs:
-
Pharmaceuticals
- PRO:
-
Propranolol
- SAs:
-
Sulfonamides
- SDBS:
-
Sodium dodecyl benzene sulfonate
- SMX:
-
Sulfamethoxazole
- SPY:
-
Sulfapyridine
- SSA:
-
Specific surface area
- STZ:
-
Sulfathiazole
- TC:
-
Tetracycline
- WWTPs:
-
Wastewater treatment plants
References
Adriano WS, Veredas V, Santana CC, Gonçalves LRB (2005) Adsorption of amoxicillin on chitosan beads: kinetics, equilibrium and validation of finite bath models. Biochem Eng J 27:132–137
Aksu Z (2005) Application of biosorption for the removal of organic pollutants: a review. Process Biochem 40:997–1026
Al-Bayati RA (2010) Adsorption-desorption isotherm of one of antidiabetic drug from aqueous solutions on some pharmaceutical adsorbents. Eur J Sci Res 137:111–120
Al-Khateeb LA, Almotiry S, Salam MA (2014) Adsorption of pharmaceutical pollutants onto graphene nanoplatelets. Chem Eng J 248:191–199
Amarnath CA, Hong CE, Kim NH, Ku B-C, Kuila T, Lee JH (2011) Efficient synthesis of graphene sheets using pyrrole as a reducing agent. Carbon 49:3497–3502
Ambrosi A, Chua CK, Bonanni A, Pumera M (2012) Lithium aluminum hydride as reducing agent for chemically reduced graphene oxides. Chem Mater 24:2292–2298
Arami M, Limaee NY, Mahmoodi NM (2006) Investigation on the adsorption capability of egg shell membrane towards model textile dyes. Chemosphere 65:1999–2008
Aristov VY, Urbanik G, Kummer K, Vyalikh DV, Molodtsova OV, Preobrajenski AB, Zakharov AA, Hess C, Hänke T, Buchner B (2010) Graphene synthesis on cubic SiC/Si wafers. Perspectives for mass production of graphene-based electronic devices. Nano Lett 10:992–995
Arvidsson R, Kushnir D, Sandén BA, Molander S (2014) Prospective life cycle assessment of graphene production by ultrasonication and chemical reduction. Environ Sci Technol 48:4529–4536
Banerjee P, Das P, Zaman A, Das P (2016) Application of graphene oxide nanoplatelets for adsorption of ibuprofen from aqueous solutions: evaluation of process kinetics and thermodynamics. Process Safety Environ Protect 101:45–53
Berger C, Song Z, Li X, Wu X, Brown N, Naud C, Mayou D, Li T, Hass J, Marchenkov AN (2006) Electronic confinement and coherence in patterned epitaxial graphene. Science 312:1191–1196
Berresheim AJ, Müller M, Müllen K (1999) Polyphenylene nanostructures. Chem Rev 99:1747–1786
Biziuk M, Przyjazny A, Czerwinski J, Wiergowski M (1996) Occurrence and determination of pesticides in natural and treated waters. J Chromatogr A 754:103–123
Brain RA, Hanson ML, Solomon KR, Brooks BW (2008) Aquatic plants exposed to pharmaceuticals: effects and risks. Reviews of environmental contamination and toxicology. Springer, pp. 67–115
Brodin T, Fick J, Jonsson M, Klaminder J (2013) Dilute concentrations of a psychiatric drug alter behavior of fish from natural populations. Science 339:814–815
Bruno J, De Pablo J, Duro L, Figuerola E (1995) Experimental study and modeling of the U (VI)-Fe (OH)3 surface precipitation/coprecipitation equilibria. Geochim Cosmochim Acta 59:4113–4123
Buser H-R, Müller MD, Theobald N (1998a) Occurrence of the pharmaceutical drug clofibric acid and the herbicide mecoprop in various Swiss lakes and in the North Sea. Environ Sci Technol 32:188–192
Buser H-R, Poiger T, Müller MD (1998b) Occurrence and fate of the pharmaceutical drug diclofenac in surface waters: rapid photodegradation in a lake. Environ Sci Technol 32:3449–3456
Buser H-R, Poiger T, Müller MD (1999) Occurrence and environmental behavior of the chiral pharmaceutical drug ibuprofen in surface waters and in wastewater. Environ Sci Technol 33:2529–2535
Cai N, Larese-Casanova P (2014) Sorption of carbamazepine by commercial graphene oxides: a comparative study with granular activated carbon and multiwalled carbon nanotubes. J Colloid Interf Sci 426:152–161
Campos B, Piña B, Barata CC (2012) Mechanisms of action of selective serotonin reuptake inhibitors in Daphnia magna. Environ Sci Technol 46:2943–2950
Carabineiro S, Thavorn-Amornsri T, Pereira M, Figueiredo J (2011) Adsorption of ciprofloxacin on surface-modified carbon materials. Water Res 45:4583–4591
Carabineiro S, Thavorn-Amornsri T, Pereira M, Serp P, Figueiredo J (2012) Comparison between activated carbon, carbon xerogel and carbon nanotubes for the adsorption of the antibiotic ciprofloxacin. Catal Today 186:29–34
Carbajo JB, Petre AL, Rosal R, Berná A, Letón P, García-Calvo E, Perdigón-Melón JA (2016) Ozonation as pre-treatment of activated sludge process of a wastewater containing benzalkonium chloride and NiO nanoparticles. Chem Eng J 283:740–749
Carmalin Sophia A, Lima EC, Allaudeen N, Rajan S (2016) Application of graphene based materials for adsorption of pharmaceutical traces from water and wastewater—a review. Desal Water Treat 57:27573–27586
Chamjangali MA, Bagherian G, Javid A, Boroumand S, Farzaneh N (2015) Synthesis of Ag-ZnO with multiple rods (multipods) morphology and its application in the simultaneous photo-catalytic degradation of methyl orange and methylene blue. Spectrochim Acta Part A: Mol Biomol Spect 150:230–237
Chang P-H, Li Z, Jiang W-T, Jean J-S (2009a) Adsorption and intercalation of tetracycline by swelling clay minerals. Appl Clay Sci 46:27–36
Chang P-H, Li Z, Yu T-L, Munkhbayer S, Kuo T-H, Hung Y-C, Jean J-S, Lin K-H (2009b) Sorptive removal of tetracycline from water by palygorskite. J Hazard Mater 165:148–155
Chang X, Meyer MT, Liu X, Zhao Q, Chen H, Chen J-A, Qiu Z, Yang L, Cao J, Shu W (2010) Determination of antibiotics in sewage from hospitals, nursery and slaughter house, wastewater treatment plant and source water in Chongqing region of Three Gorge Reservoir in China. Environ Pollut 158:1444–1450
Chen H, Gao B, Li H (2015) Removal of sulfamethoxazole and ciprofloxacin from aqueous solutions by graphene oxide. J Hazard Mater 282:201–207
Chowdhury S, Balasubramanian R (2014) Recent advances in the use of graphene-family nanoadsorbents for removal of toxic pollutants from wastewater. Adv Colloid Interf Sci 204:35–56
Cox CE, Marbury TC, Pittman WG, Brown GL, Auerbach SM, Fox BC, Yang JY (2002) A randomized, double-blind, multicenter comparison of gatifloxacin versus ciprofloxacin in the treatment of complicated urinary tract infection and pyelonephritis. Clin Ther 24:223–236
del Rosario Brunetto M, Clavijo S, Delgado Y, Orozco W, Gallignani M, Ayala C, Cerdà V (2015) Development of a MSFIA sample treatment system as front end of GC–MS for atenolol and propranolol determination in human plasma. Talanta 132:15–22
Deng D, Pan X, Zhang H, Fu Q, Tan D, Bao X (2010) Freestanding graphene by thermal splitting of silicon carbide granules. Adv Mater 22:2168–2171
Di Poi C, Bidel F, Dickel L, Bellanger C (2014) Cryptic and biochemical responses of young cuttlefish Sepia officinalis exposed to environmentally relevant concentrations of fluoxetine. Aquat Toxicol 151:36–45
Díaz-Cruz MS, MAJ L d A, Barceló D (2003) Environmental behavior and analysis of veterinary and human drugs in soils, sediments and sludge. TrAC-Trends Anal Chem 22:340–351
Diwan V, Tamhankar AJ, Khandal RK, Sen S, Aggarwal M, Marothi Y, Iyer RV, Sundblad-Tonderski K, Stålsby-Lundborg C (2010) Antibiotics and antibiotic-resistant bacteria in waters associated with a hospital in Ujjain, India. BMC Public Health 10:1–8
Dong S, Sun Y, Wu J, Wu B, Creamer AE, Gao B (2015) Graphene oxide as filter media to remove levofloxacin and lead from aqueous solution. Chemosphere 150:759–764
Dreyer DR, Park S, Bielawski CW, Ruoff RS (2010) The chemistry of graphene oxide. Chem Soc Rev 39:228–240
Drillia P, Dokianakis SN, Fountoulakis MS, Kornaros M, Stamatelatou K, Lyberatos G (2005) On the occasional biodegradation of pharmaceuticals in the activated sludge process: the example of the antibiotic sulfamethoxazole. J Hazard Mater 122:259–265
El-Shafey E-SI, Al-Lawati H, Al-Sumri AS (2012) Ciprofloxacin adsorption from aqueous solution onto chemically prepared carbon from date palm leaflets. J Environ Sci 24:1579–1586
Fatta-Kassinos D, Meric S, Nikolaou A (2011) Pharmaceutical residues in environmental waters and wastewater: current state of knowledge and future research. Anal Bioanal Chem 399:251–275
Fernandez-Merino M, Guardia L, Paredes J, Villar-Rodil S, Solis-Fernandez P, Martinez-Alonso A, Tascon J (2010) Vitamin C is an ideal substitute for hydrazine in the reduction of graphene oxide suspensions. J Phys Chem C 114:6426–6432
Figueroa RA, Leonard A, MacKay AA (2004) Modeling tetracycline antibiotic sorption to clays. Environ Sci Technol 38:476–483
Gao W, Alemany LB, Ci L, Ajayan PM (2009) New insights into the structure and reduction of graphite oxide. Nature Chem 1:403–408
Gao Y, Li Y, Zhang L, Huang H, Hu J, Shah SM, Su X (2012) Adsorption and removal of tetracycline antibiotics from aqueous solution by graphene oxide. J Colloid Interface Sci 368:540–546
Gao Y, Yue Q, Gao B, Sun Y (2015) Optimization preparation of activated carbon from Enteromorpha prolifra using response surface methodology and its adsorption studies of fluoroquinolone antibiotics. Desalination Water Treat 55:624–636
García-Galán MJ, González Blanco S, López Roldán R, Díaz-Cruz S, Barceló D (2012) Ecotoxicity evaluation and removal of sulfonamides and their acetylated metabolites during conventional wastewater treatment. Sci Total Environ 437:403–412
Geiser L, Henchoz Y, Galland A, Carrupt PA, Veuthey JL (2005) Determination of pKa values by capillary zone electrophoresis with a dynamic coating procedure. J Sep Sci 28:2374–2380
Genç N, Dogan EC (2015) Adsorption kinetics of the antibiotic ciprofloxacin on bentonite, activated carbon, zeolite, and pumice. Desalination Water Treat 53:785–793
Ghadim EE, Manouchehri F, Soleimani G, Hosseini H, Kimiagar S, Nafisi S (2013) Adsorption properties of tetracycline onto graphene oxide: equilibrium, kinetic and thermodynamic studies. PLoS One 8:1–9
Göbel A, McArdell CS, Joss A, Siegrist H, Giger W (2007) Fate of sulfonamides, macrolides, and trimethoprim in different wastewater treatment technologies. Sci Total Environ 372:361–371
Godoy AA, Kummrow F, Pamplin PAZ (2015) Occurrence, ecotoxicological effects and risk assessment of antihypertensive pharmaceutical residues in the aquatic environment—a review. Chemosphere 138:281–291
Grobert N (2007) Carbon nanotubes—becoming clean. Mater Today 10:28–35
Guo H-L, Wang X-F, Qian Q-Y, Wang F-B, Xia X-H (2009) A green approach to the synthesis of graphene nanosheets. ACS Nano 3:2653–2659
Halling-Sørensen B, Nielsen SN, Lanzky P, Ingerslev F, Lützhøft HH, Jørgensen S (1998) Occurrence, fate and effects of pharmaceutical substances in the environment—a review. Chemosphere 36:357–393
Hampel M, Bron JE, Taggart JB, Leaver MJ (2014) The antidepressant drug carbamazepine induces differential transcriptome expression in the brain of Atlantic salmon, Salmo salar. Aquat Toxicol 151:114–123
Hartmann A, Alder AC, Koller T, Widmer RM (1998) Identification of fluoroquinolone antibiotics as the main source of umuC genotoxicity in native hospital wastewater. Environ Toxicol Chem 17:377–382
Hirsch A (2009) Unzipping carbon nanotubes: a peeling method for the formation of graphene nanoribbons. Angew Chem Int Ed 48:6594–6596
Hirsch R, Ternes T, Haberer K, Kratz K-L (1999) Occurrence of antibiotics in the aquatic environment. Sci Total Environ 225:109–118
Hu R, Shao D, Wang X (2014) Graphene oxide/polypyrrole composites for highly selective enrichment of U(VI) from aqueous solutions. Polym Chem 5:6207–6215
Hu X, Zhou Q (2013) Health and ecosystem risks of graphene. Chem Rev 113:3815–3835
Huang L, Wang M, Shi C, Huang J, Zhang B (2014) Adsorption of tetracycline and ciprofloxacin on activated carbon prepared from lignin with H3PO4 activation. Desalination Water Treat 52:2678–2687
Hughes SR, Kay P, Brown LE (2012) Global synthesis and critical evaluation of pharmaceutical data sets collected from river systems. Environ Sci Technol 47:661–677
Islam MS, Zhang Y, McPhedran KN, Liu Y, Gamal El-Din M (2015) Granular activated carbon for simultaneous adsorption and biodegradation of toxic oil sands process-affected water organic compounds. J Environ Manag 152:49–57
Jauris I, Matos C, Saucier C, Lima E, Zarbin A, Fagan S, Machado F, Zanella I (2016) Adsorption of sodium diclofenac on graphene: a combined experimental and theoretical study. Phys Chem Chem Phys 18:1526–1536
Jelic A, Gros M, Ginebreda A, Cespedes-Sánchez R, Ventura F, Petrovic M, Barcelo D (2011) Occurrence, partition and removal of pharmaceuticals in sewage water and sludge during wastewater treatment. Water Res 45:1165–1176
Jiang L-H, Liu Y-G, Zeng G-M, Xiao F-Y, Hu X-J, Hu X, Wang H, Li T-T, Zhou L, Tan X-F (2016) Removal of 17β-estradiol by few-layered graphene oxide nanosheets from aqueous solutions: external influence and adsorption mechanism. Chem Eng J 284:93–102
Jiang W-T, Chang P-H, Wang Y-S, Tsai Y, Jean J-S, Li Z, Krukowski K (2013) Removal of ciprofloxacin from water by birnessite. J Hazard Mater 250:362–369
Jiao L, Zhang L, Wang X, Diankov G, Dai H (2009) Narrow graphene nanoribbons from carbon nanotubes. Nature 458:877–880
Jin Z, Wang X, Sun Y, Ai Y, Wang X (2015) Adsorption of 4-n-nonylphenol and bisphenol-A on magnetic reduced graphene oxides: a combined experimental and theoretical studies. Environ Sci Technol 49:9168–9175
Jobling S, Williams R, Johnson A, Taylor A, Gross-Sorokin M, Nolan M, Tyler CR, van Aerle R, Santos E, Brighty G (2005) Predicted exposures to steroid estrogens in UK rivers correlate with widespread sexual disruption in wild fish populations. Environ Health Perspect 114:32–39
Kaniyoor A, Baby TT, Ramaprabhu S (2010) Graphene synthesis via hydrogen induced low temperature exfoliation of graphite oxide. J Mater Chem 20:8467–8469
Kemper N (2008) Veterinary antibiotics in the aquatic and terrestrial environment. Ecolog Indicat 8:1–13
Kerkez-Kuyumcu Ö, Bayazit ŞS, Salam MA (2016) Antibiotic amoxicillin removal from aqueous solution using magnetically modified graphene nanoplatelets. J Ind Eng Chem 36:198–205
Khataee A, Gholami P, Sheydaei M (2016) Heterogeneous Fenton process by natural pyrite for removal of a textile dye from water: effect of parameters and intermediate identification. J Taiwan Inst Chem Eng 58:366–373
Khetan SK, Collins TJ (2007) Human pharmaceuticals in the aquatic environment: a challenge to green chemistry. Chem Rev 107:2319–2364
Kidd KA, Blanchfield PJ, Mills KH, Palace VP, Evans RE, Lazorchak JM, Flick RW (2007) Collapse of a fish population after exposure to a synthetic estrogen. Proc Nat Acad Sci 104:8897–8901
Kim JM, Hong WG, Lee SM, Chang SJ, Jun Y, Kim BH, Kim HJ (2014) Energy storage of thermally reduced graphene oxide. Int J Hydrog Energy 39:3799–3804
Kim SD, Cho J, Kim IS, Vanderford BJ, Snyder SA (2007) Occurrence and removal of pharmaceuticals and endocrine disruptors in South Korean surface, drinking, and waste waters. Water Res 41:1013–1021
Kinney CA, Furlong ET, Werner SL, Cahill JD (2006) Presence and distribution of wastewater-derived pharmaceuticals in soil irrigated with reclaimed water. Environ Toxicol Chem 25:317–326
Kümmerer K, Steger-Hartmann T, Meyer M (1997) Biodegradability of the anti-tumour agent ifosfamide and its occurrence in hospital effluents and communal sewage. Water Res 31:2705–2710
Kümmerer K, Al-Ahmad A, Mersch-Sundermann V (2000a) Biodegradability of some antibiotics, elimination of the genotoxicity and affection of wastewater bacteria in a simple test. Chemosphere 40:701–710
Kümmerer K, Al-Ahmad A, Wiethan J, Hertle W, Henninger A (2000b) Effects and fate of antibiotics in sewage treatment plants. Proceedings of the Third SETAC World Congress, May, pp. 21–25
Kümmerer K (2001) Drugs in the environment: emission of drugs, diagnostic aids and disinfectants into wastewater by hospitals in relation to other sources—a review. Chemosphere 45:957–969
Kümmerer K (2004) Resistance in the environment. J Antimicrob Chemother 54:311–320
Kwon J, Lee B (2015) Bisphenol A adsorption using reduced graphene oxide prepared by physical and chemical reduction methods. Chem Eng Res Design 104:519–529
Kyzas GZ, Bikiaris DN, Seredych M, Bandosz TJ, Deliyanni EA (2014) Removal of dorzolamide from biomedical wastewaters with adsorption onto graphite oxide/poly(acrylic acid) grafted chitosan nanocomposite. Bioresour Technol 152:399–406
Kyzas GZ, Fu J, Lazaridis NK, Bikiaris DN, Matis KA (2015a) New approaches on the removal of pharmaceuticals from wastewaters with adsorbent materials. J Mol Liq 209:87–93
Kyzas GZ, Koltsakidou A, Nanaki SG, Bikiaris DN, Lambropoulou DA (2015b) Removal of beta-blockers from aqueous media by adsorption onto graphene oxide. Sci Total Environ 537:411–420
Larsson D, Adolfsson-Erici M, Parkkonen J, Pettersson M, Berg A, Olsson P-E, Förlin L (1999) Ethinyloestradiol—an undesired fish contraceptive? Aquat Toxicol 45:91–97
Li J, Zhang S, Chen C, Zhao G, Yang X, Li J, Wang X (2012) Removal of Cu(II) and fulvic acid by graphene oxide nanosheets decorated with Fe3O4 nanoparticles. ACS Appl Mater Interf 4:4991–5000
Lin AY-C, Yu T-H, Lin C-F (2008) Pharmaceutical contamination in residential, industrial, and agricultural waste streams: risk to aqueous environments in Taiwan. Chemosphere 74:131–141
Lin B, Liaw S-L (2015) Simultaneous removal of volatile organic compounds from cooking oil fumes by using gas-phase ozonation over Fe(OH)3 nanoparticles. J Environ Chem Eng 3:1530–1538
Lin Y, Xu S, Li J (2013) Fast and highly efficient tetracyclines removal from environmental waters by graphene oxide functionalized magnetic particles. Chem Eng J 225:679–685
Liu FF, Zhao J, Wang S, Du P, Xing B (2014) Effects of solution chemistry on adsorption of selected pharmaceuticals and personal care products (PPCPs) by graphenes and carbon nanotubes. Environ Sci Technol 48:13197–13206
Liu FF, Zhao J, Wang S, Xing B (2015) Adsorption of sulfonamides on reduced graphene oxides as affected by pH and dissolved organic matter. Environ Pollut 210:85–93
Liu J, Tang J, Gooding JJ (2012a) Strategies for chemical modification of graphene and applications of chemically modified graphene. J Mater Chem 22:12435–12452
Liu M, Chen C, Hu J, Wu X, Wang X (2011) Synthesis of magnetite/graphene oxide composite and application for cobalt(II) removal. J Phys Chem C 115:25234–25240
Liu M, Wen T, Wu X, Chen C, Hu J, Li J, Wang X (2013a) Synthesis of porous Fe3O4 hollow microspheres/graphene oxide composite for Cr(vi) removal. Dalton Trans 42:14710–14717
Liu S, Chen K, Fu Y, Yu S, Bao Z (2012b) Reduced graphene oxide paper by supercritical ethanol treatment and its electrochemical properties. Appl Surf Sci 258:5299–5303
Liu Y, Zhang Y, Ma G, Wang Z, Liu K, Liu H (2013b) Ethylene glycol reduced graphene oxide/polypyrrole composite for supercapacitor. Electrochim Acta 88:519–525
Luo D, Zhang G, Liu J, Sun X (2011a) Evaluation criteria for reduced graphene oxide. J Phys Chem C 115:11327–11335
Luo YB, Shi ZG, Gao Q, Feng YQ (2011b) Magnetic retrieval of graphene: extraction of sulfonamide antibiotics from environmental water samples. J Chromatogr A 1218:1353–1358
Lyons G (2015) Pharmaceuticals in the environment: a growing threat to our tap water and wildlife. A CHEM Trust Report 11:1–36
Ma J, Yang M, Yu F, Zheng J (2015) Water-enhanced removal of ciprofloxacin from water by porous graphene hydrogel. Sci Rep 5:13578
Mao S, Pu H, Chen J (2012) Graphene oxide and its reduction: modeling and experimental progress. RSC Adv 2:2643–2662
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun Z, Slesarev A, Alemany LB, Lu W, Tour JM (2010) Improved synthesis of graphene oxide. ACS Nano 4:4806–4814
Martinez JL (2009) Environmental pollution by antibiotics and by antibiotic resistance determinants. Environ Pollut 157:2893–2902
Martins AF, Vasconcelos TG, Henriques DM, Frank CS, König A, Kümmerer K (2008) Concentration of ciprofloxacin in Brazilian hospital effluent and preliminary risk assessment: a case study. Clean–Soil, Air, Water 36:264–269
Martucci A, Pasti L, Marchetti N, Cavazzini A, Dondi F, Alberti A (2012) Adsorption of pharmaceuticals from aqueous solutions on synthetic zeolites. Micropor Mesopor Mater 148:174–183
McAllister MJ, Li J-L, Adamson DH, Schniepp HC, Abdala AA, Liu J, Herrera-Alonso M, Milius DL, Car R, Prud’homme RK (2007) Single sheet functionalized graphene by oxidation and thermal expansion of graphite. Chem Mater 19:4396–4404
Michael I, Rizzo L, McArdell C, Manaia C, Merlin C, Schwartz T, Dagot C, Fatta-Kassinos D (2013) Urban wastewater treatment plants as hotspots for the release of antibiotics in the environment: a review. Water Res 47:957–995
Mohle E, Kempter C, Kern A, Metzger J (1999) Examination of the degradation of drugs in municipal sewage plants using liquid chromatography-electrospray mass spectrometry. Acta Hydrochim Hydrobiol 27:430–436
Moussavi G, Alahabadi A, Yaghmaeian K, Eskandari M (2013) Preparation, characterization and adsorption potential of the NH4Cl-induced activated carbon for the removal of amoxicillin antibiotic from water. Chem Eng J 217:119–128
Moussavi G, Hossaini Z, Pourakbar M (2016) High-rate adsorption of acetaminophen from the contaminated water onto double-oxidized graphene oxide. Chem Eng J 287:665–673
Msagati TA, Nindi MM (2004) Multiresidue determination of sulfonamides in a variety of biological matrices by supported liquid membrane with high pressure liquid chromatography-electrospray mass spectrometry detection. Talanta 64:87–100
Nam SW, Jung C, Li H, Yu M, Flora JR, Boateng LK, Her N, Zoh KD, Yoon Y (2015) Adsorption characteristics of diclofenac and sulfamethoxazole to graphene oxide in aqueous solution. Chemosphere 136:20–26
Ni X, Zhao Y, Song Q (2015) Electrochemical reduction and in-situ electrochemiluminescence detection of nitroaromatic compounds. Electrochim Acta 164:31–37
Oleszczuk P, Pan B, Xing B (2009) Adsorption and desorption of oxytetracycline and carbamazepine by multiwalled carbon nanotubes. Environ Sci Technol 43:9167–9173
Pei S, Cheng H-M (2012) The reduction of graphene oxide. Carbon 50:3210–3228
Prado N, Ochoa J, Amrane A (2009) Biodegradation and biosorption of tetracycline and tylosin antibiotics in activated sludge system. Process Biochem 44:1302–1306
Ramesha G, Kumara AV, Muralidhara H, Sampath S (2011) Graphene and graphene oxide as effective adsorbents toward anionic and cationic dyes. J Colloid Interface Sci 361:270–277
Ren X, Li J, Tan X, Wang X (2013) Comparative study of graphene oxide, activated carbon and carbon nanotubes as adsorbents for copper decontamination. Dalton Trans 42:5266–5274
Richardson ML, Bowron JM (1985) The fate of pharmaceutical chemicals in the aquatic environment. J Pharm Pharmacol 37:1–12
Rivera-Utrilla J, Sánchez-Polo M, Ferro-García MÁ, Prados-Joya G, Ocampo-Pérez R (2013) Pharmaceuticals as emerging contaminants and their removal from water. A review. Chemosphere 93:1268–1287
Rostamian R, Behnejad H (2016) A comparative adsorption study of sulfamethoxazole onto graphene and graphene oxide nanosheets through equilibrium, kinetic and thermodynamic modeling. Proc Safe Environ Prot 102:20–29
Sakamoto J, van Heijst J, Lukin O, Schlüter AD (2009) Two-dimensional polymers: just a dream of synthetic chemists? Angew Chem Int Ed 48:1030–1069
Särkkä H, Vepsäläinen M, Sillanpää M (2015) Natural organic matter (NOM) removal by electrochemical methods—a review. J Electroanal Chem 755:100–108
Sarmah AK, Meyer MT, Boxall ABA (2006) A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 65:725–759
Schiavon M, Scapinello M, Tosi P, Ragazzi M, Torretta V, Rada EC (2015) Potential of non-thermal plasmas for helping the biodegradation of volatile organic compounds (VOCs) released by waste management plants. J Cleaner Product 104:211–219
Schniepp HC, Li J-L, McAllister MJ, Sai H, Herrera-Alonso M, Adamson DH, Prud’homme RK, Car R, Saville DA, Aksay IA (2006) Functionalized single graphene sheets derived from splitting graphite oxide. J Phys Chem B 110:8535–8539
Shao B, Dong D, Wu Y, Hu J, Meng J, Tu X, Xu S (2005) Simultaneous determination of 17 sulfonamide residues in porcine meat, kidney and liver by solid-phase extraction and liquid chromatography–tandem mass spectrometry. Analy Chim Acta 546:174–181
Shao Y, Wang J, Engelhard M, Wang C, Lin Y (2010) Facile and controllable electrochemical reduction of graphene oxide and its applications. J Mater Chem 20:743–748
Shi S, Fan Y, Huang Y (2013) Facile low temperature hydrothermal synthesis of magnetic mesoporous carbon nanocomposite for adsorption removal of ciprofloxacin antibiotics. Ind Eng Chem Res 52:2604–2612
Shin HJ, Kim KK, Benayad A, Yoon SM, Park HK, Jung IS, Jin MH, Jeong HK, Kim JM, Choi JY (2009) Efficient reduction of graphite oxide by sodium borohydride and its effect on electrical conductance. Adv Funct Mater 19:1987–1992
Sillanpää ME, Kurniawan TA, W-h L (2011) Degradation of chelating agents in aqueous solution using advanced oxidation process (AOP). Chemosphere 83:1443–1460
Silva EJ, Rocha e Silva NMP, Rufino RD, Luna JM, Silva RO, Sarubbo LA (2014) Characterization of a biosurfactant produced by Pseudomonas cepacia CCT6659 in the presence of industrial wastes and its application in the biodegradation of hydrophobic compounds in soil. Colloid Surf B: Biointerfaces 117:36–41
Simpson CD, Brand JD, Berresheim AJ, Przybilla L, Räder HJ, Müllen K (2002) Synthesis of a giant 222 carbon graphite sheet. Chem–Eur J 8:1424–1429
Sirés I, Brillas E (2012) Remediation of water pollution caused by pharmaceutical residues based on electrochemical separation and degradation technologies: a review. Environ Int 40:212–229
Stackelberg PE, Furlong ET, Meyer MT, Zaugg SD, Henderson AK, Reissman DB (2004) Persistence of pharmaceutical compounds and other organic wastewater contaminants in a conventional drinking-water-treatment plant. Sci Total Environ 329:99–113
Stamatelatou K, Frouda C, Fountoulakis M, Drillia P, Kornaros M, Lyberatos G (2003) Pharmaceuticals and health care products in wastewater effluents: the example of carbamazepine. Water Sci Technol Water Supply 3:131–137
Stankovich S, Dikin DA, Piner RD, Kohlhaas KA, Kleinhammes A, Jia Y, Wu Y, Nguyen ST, Ruoff RS (2007) Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 45:1558–1565
Stumpf M, Ternes TA, Wilken R-D, Rodrigues SV, Baumann W (1999) Polar drug residues in sewage and natural waters in the state of Rio de Janeiro, Brazil. Sci Total Environ 225:135–141
Subrahmanyam K, Panchakarla L, Govindaraj A, Rao C (2009) Simple method of preparing graphene flakes by an arc-discharge method. J Phys Chem C 113:4257–4259
Sun Y, Wang Q, Chen C, Tan X, Wang X (2012) Interaction between Eu (III) and graphene oxide nanosheets investigated by batch and extended X-ray absorption fine structure spectroscopy and by modeling techniques. Environ Sci Technol 46:6020–6027
Sun Y, Shao D, Chen C, Yang S, Wang X (2013a) Highly efficient enrichment of radionuclides on graphene oxide-supported polyaniline. Environ Sci Technol 47:9904–9910
Sun Y, Yang S, Zhao G, Wang Q, Wang X (2013b) Adsorption of polycyclic aromatic hydrocarbons on graphene oxides and reduced graphene oxides. Chem Asian J 8:2755–2761
Sun Y, Yang S, Chen Y, Ding C, Cheng W, Wang X (2015) Adsorption and desorption of U (VI) on functionalized graphene oxides: a combined experimental and theoretical study. Environ Sci Technol 49:4255–4262
Swan GE, Cuthbert R, Quevedo M, Green RE, Pain DJ, Bartels P, Cunningham AA, Duncan N, Meharg AA, Oaks JL (2006) Toxicity of diclofenac to Gyps vultures. Biology Lett 2:279–282
Tan X, Ren X, Chen C, Wang X (2014) Analytical approaches to the speciation of lanthanides at solid-water interfaces. TrAC-Trends Anal Chem 61:107–132
Tang Y, Reeder RJ (2009) Uranyl and arsenate cosorption on aluminum oxide surface. Geochim Cosmochim Acta 73:2727–2743
Tang Y, Guo H, Xiao L, Yu S, Gao N, Wang Y (2013) Synthesis of reduced graphene oxide/magnetite composites and investigation of their adsorption performance of fluoroquinolone antibiotics. Colloid Surf A: Physicochem Eng Aspects 424:74–80
Ternes TA (1998) Occurrence of drugs in German sewage treatment plants and rivers. Water Res 32:3245–3260
Ternes TA, Joss A, Siegrist H (2004) Peer reviewed: scrutinizing pharmaceuticals and personal care products in wastewater treatment. Environ Sci Technol 38:392A–399A
Usca GT, Gómez CV, Fiallos DC, Tavolaro P, Martino G, Caputi LS, Tavolaro A (2015) Preparation of graphene oxide as biomaterials for drug adsorption. AIP Conference Proceedings 1646:79–86
Vörösmarty CJ, McIntyre PB, Gessner MO, Dudgeon D, Prusevich A, Green P, Glidden S, Bunn SE, Sullivan CA, Liermann CR (2010) Global threats to human water security and river biodiversity. Nature 467:555–561
Wang C-J, Li Z, Jiang W-T, Jean J-S, Liu C-C (2010) Cation exchange interaction between antibiotic ciprofloxacin and montmorillonite. J Hazard Mater 183:309–314
Wang C-J, Li Z, Jiang W-T (2011) Adsorption of ciprofloxacin on 2:1 dioctahedral clay minerals. Appl Clay Sci 53:723–728
Wang C, Li H, Liao S, Zheng H, Wang Z, Pan B, Xing B (2013) Coadsorption, desorption hysteresis and sorption thermodynamics of sulfamethoxazole and carbamazepine on graphene oxide and graphite. Carbon 65:243–251
Wang F, Yang B, Wang H, Song Q, Tan F, Cao Y (2016a) Removal of ciprofloxacin from aqueous solution by a magnetic chitosan grafted graphene oxide composite. J Mol Liq 222:188–194
Wang X, Chen Z, Wang X (2015) Graphene oxides for simultaneous highly efficient removal of trace level radionuclides from aqueous solutions. Sci China Chem 58:1766–1773
Wang X, Bi W, Zhai P, Wang X, Li H, Mailhot G, Dong W (2016b) Adsorption and photocatalytic degradation of pharmaceuticals by BiOClxIy nanospheres in aqueous solution. Appl Surf Sci 360:240–251
Wang X, Fan Q, Yu S, Chen Z, Ai Y, Sun Y, Hobiny A, Alsaedi A, Wang X (2016c) High sorption of U (VI) on graphene oxides studied by batch experimental and theoretical calculations. Chem Eng J 287:448–455
Weber FA, Bergmann A, Hickmann S, Ebert I, Hein A, Küster A (2015) Pharmaceuticals in the environment—global occurrences and perspectives. Environ Toxicol Chem 35:823–835
Weinberger J, Klaper R (2014) Environmental concentrations of the selective serotonin reuptake inhibitor fluoxetine impact specific behaviors involved in reproduction, feeding and predator avoidance in the fish Pimephales promelas (fathead minnow). Aquat Toxicol 151:77–83
William S, Hummers J, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339
Wise R (2002) Antimicrobial resistance: priorities for action. J Antimicrob Chemother 49:585–586
Wu J, Gherghel L, Watson MD, Li J, Wang Z, Simpson CD, Kolb U, Müllen K (2003) From branched polyphenylenes to graphite ribbons. Macromolecules 36:7082–7089
Wu Q, Li Z, Hong H, Yin K, Tie L (2010) Adsorption and intercalation of ciprofloxacin on montmorillonite. Appl Clay Sci 50:204–211
Wu S, Zhao X, Li Y, Zhao C, Du Q, Sun J, Wang Y, Peng X, Xia Y, Wang Z, Xia L (2013) Adsorption of ciprofloxacin onto biocomposite fibers of graphene oxide/calcium alginate. Chem Eng J 230:389–395
Wu Z-S, Ren W, Gao L, Zhao J, Chen Z, Liu B, Tang D, Yu B, Jiang C, Cheng H-M (2009) Synthesis of graphene sheets with high electrical conductivity and good thermal stability by hydrogen arc discharge exfoliation. ACS Nano 3:411–417
Yang X, Chen C, Li J, Zhao G, Ren X, Wang X (2012) Graphene oxide-iron oxide and reduced graphene oxide-iron oxide hybrid materials for the removal of organic and inorganic pollutants. RSC Adv 2:8821
Yu F, Ma J, Bi D (2015a) Enhanced adsorptive removal of selected pharmaceutical antibiotics from aqueous solution by activated graphene. Environ Sci Pollut Res Int 22:4715–4724
Yu JG, Yu LY, Yang H, Liu Q, Chen XH, Jiang XY, Chen XQ, Jiao FP (2015b) Graphene nanosheets as novel adsorbents in adsorption, preconcentration and removal of gases, organic compounds and metal ions. Sci Total Environ 502:70–79
Yu S, Wang X, Tan X, Wang X (2015c) Sorption of radionuclides from aqueous systems onto graphene oxide-based materials: a review. Inorg Chem Front 2:593–612
Yu F, Sun S, Han S, Zheng J, Ma J (2016a) Adsorption removal of ciprofloxacin by multi-walled carbon nanotubes with different oxygen contents from aqueous solutions. Chem Eng J 285:588–595
Yu SJ, Wang XX, Ai YJ, Tan XL, Hayat T, Wang XK (2016b) Experimental and theoretical study on competitive adsorption of aromatic compounds on reduced graphene oxides. J Mater Chem A 4:5654–5662
Yu Z, Peldszus S, Huck P, Anderson W (2005) Adsorption of selected pharmaceuticals and endocrine disrupting substances by GAC at low concentration levels. Proceedings of AWWA Water Quality and Technology Conference 2005. Pp 1–16
Yuan F, Hu C, Hu X, Qu J, Yang M (2009) Degradation of selected pharmaceuticals in aqueous solution with UV and UV/H2O2. Water Res 43:1766–1774
Yuan Y, Lai B, Tang Y-Y (2016) Combined Fe0/air and Fenton process for the treatment of dinitrodiazophenol (DDNP) industry wastewater. Chem Eng J 283:1514–1521
Zhang Y, Chen B, Zhang L, Huang J, Chen F, Yang Z, Yao J, Zhang Z (2011) Controlled assembly of Fe3O4 magnetic nanoparticles on graphene oxide. Nanoscale 3:1446–1450
Zhao C, Deng H, Li Y, Liu Z (2010) Photodegradation of oxytetracycline in aqueous by 5A and 13X loaded with TiO2 under UV irradiation. J Hazard Mater 176:884–892
Zhao D, Sheng G, Chen C, Wang X (2012a) Enhanced photocatalytic degradation of methylene blue under visible irradiation on graphene@TiO2 dyade structure. Appl Catal B Environ 111-112:303–308
Zhao G, Jiang L, He Y, Li J, Dong H, Wang X, Hu W (2011a) Sulfonated graphene for persistent aromatic pollutant management. Adv Mater 23:3959–3963
Zhao G, Li J, Ren X, Chen C, Wang X (2011b) Few-layered graphene oxide nanosheets as superior sorbents for heavy metal ion pollution management. Environ Sci Technol 45:10454–10462
Zhao G, Ren X, Gao X, Tan X, Li J, Chen C, Huang Y, Wang X (2011c) Removal of Pb(II) ions from aqueous solutions on few-layered graphene oxide nanosheets. Dalton Trans 40:10945–10952
Zhao G, Wen T, Yang X, Yang S, Liao J, Hu J, Shao D, Wang X (2012b) Preconcentration of U(VI) ions on few-layered graphene oxide nanosheets from aqueous solutions. Dalton Trans 41:6182–6188
Zhong Z, Li G, Luo Z, Liu Z, Shao Y, He W, Deng J, Luo X (2015) Carboxylated graphene oxide/polyvinyl chloride as solid-phase extraction sorbent combined with ion chromatography for the determination of sulfonamides in cosmetics. Anal Chim Acta 888:75–84
Zhou M, Wang Y, Zhai Y, Zhai J, Ren W, Wang F, Dong S (2009) Controlled synthesis of large-area and patterned electrochemically reduced graphene oxide films. Chem Eur J 15:6116–6120
Zou YD, Wang XX, Chen ZS, Yao W, Ai YJ, Liu YH, Hayat T, Alsaedi A, Alharbi NS, Wang XK (2016a) Superior coagulation of graphene oxides on nanoscale layered double hydroxides and layered double oxides. Environ Poll 219:107–117
Zou YD, Wang XX, Ai YJ, Liu YH, Li JX, Ji YF, Wang XK (2016b) Coagulation behavior of graphene oxide on nanocrystallined Mg/Al layered double hydroxides: batch experimental and theoretical calculation study. Environ Sci Technol 50:3658–3667
Zwiener C, Frimmel F (2003) Short-term tests with a pilot sewage plant and biofilm reactors for the biological degradation of the pharmaceutical compounds clofibric acid, ibuprofen, and diclofenac. Sci Total Environ 309:201–211
Acknowledgements
The support from the Chinese government under China Scholarship Council (CSC) for foreign postgraduate students, NSFC (21225730, 91326202, 21577032), the Fundamental Research Funds for the Central Universities (JB2015001), the Kunlun Scholarship of Qinghai province, the Jiangsu Provincial Key Laboratory of Radiation Medicine and Protection and the Priority Academic Program Development of Jiangsu Higher Education Institutions is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Guilherme L. Dotto
Rights and permissions
About this article
Cite this article
Khan, A., Wang, J., Li, J. et al. The role of graphene oxide and graphene oxide-based nanomaterials in the removal of pharmaceuticals from aqueous media: a review. Environ Sci Pollut Res 24, 7938–7958 (2017). https://doi.org/10.1007/s11356-017-8388-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-8388-8