Abstract
Background, aim, and scope
The main pathway for human exposure to the highly toxic polychlorinated-p-dioxins and polychlorinated furans [polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/Fs)] is via dietary intake. Other exposure pathways may, however, be important in close proximity to point sources, such as wood preservation sites, where PCDD/F contaminated chlorophenols (CP) were previously used. In this study, a heavily PCDD/F contaminated CP saw mill site in Sweden was investigated. Human exposure through a broad spectrum of exposure pathways was assessed. Such studies are in demand since the question whether contaminated sites represent a current or future risk can only be answered by detailed site-specific risk assessments.
Materials and methods
Sampling of exposure media (soil, air, groundwater, raspberries, carrots, potatoes, grass, milk, eggs, and chicken fodder) was made. Exposure media concentrations and congener distribution patterns were used to investigate the mobilization of PCDD/Fs from soil to the environment and to calculate exposure levels for adults. Blood serum levels from site-exposed and control individuals were also analyzed.
Results
Congener distribution patterns at the site were generally dominated by a specific marker congener (1234678-HpCDF), which is highly abundant in the polluted soil. The dioxin toxic equivalents (TEQ) concentrations were notably elevated as compared to national reference samples for most exposure media, and the marker congener was a major contributor to increased TEQ levels. There were also indications of soil-to-air volatilization of tetra- and penta-CDD/Fs. People who participated in the restoration of a contaminated building showed higher levels of 1234678-HpCDF compared to controls, and calculated exposure levels suggest that several site-specific exposure routes may be of importance for the daily intake of PCDD/F.
Conclusions, recommendations, and perspectives
Despite low mobility of higher chlorinated PCDD/Fs, these contaminants were transferred from the polluted soil to the surroundings and into human tissue. The extent of increased exposure from contaminated sites depends on the PCDD/F source strength of the soil, composition of the pollution, human activities, and dietary patterns of the residents. Impact from the contaminated soil on other exposure media was seen also for areas with low to moderate soil contamination. In the future, not only the levels of PCDD/F soil pollution but also the composition must be considered in risk assessments of contaminated sites.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Background, aim, and scope
The main exposure pathway for polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/Fs) is generally through dietary intake (European Commission 1999), and consumption of fatty foods (e.g., fish, meat, and dairy products) is of particular relevance. Other exposure pathways such as ingestion of soil or inhalation may, however, become important in close proximity to point sources, such as contaminated sites. In this context, it is important to recognize that historic emissions are highly relevant for contemporary exposure and that some of the recent food scandals were caused by dioxin/dioxin-like contamination in connection to contaminated sites (Weber et al. 2008a,b). Land can become contaminated with PCDD/Fs from a variety of primary sources (Weber et al. 2008a). Depending on the original source, the PCDD/F composition in soil varies and can often be characterized by marker congeners that define a “fingerprint” of the source. The former use of chlorophenol (CP) wood preservatives has resulted in PCDD/F contamination of a large number of sites in Sweden (Persson et al. 2007). Although the use of CP was severely restricted in the 1970s, PCDD/Fs are still present at former wood treatment plants at typical levels of 100–1,000 ng kg−1 dw (Tysklind et al. 2006).
The toxic level of a mixture of 2378-substituted PCDD/F congeners and dioxin-like polychlorinated biphenyls (dl-PCBs) is commonly expressed as toxic equivalents (TEQ; van den Berg et al. 1998, 2006). The European Commission (EC) has recommended a tolerable weekly intake (TWI) of 14 pg TEQ kg−1 body weight (bw) week–1 (European Commission 2001a), which often is expressed as a tolerable daily intake (TDI) of 2 pg TEQ kg−1 bw day−1. The average intake of dioxins and dl-PCBs exceeds the TWI for a large proportion of the European population (European Commission 1999). The EC has also introduced maximum residue limit values (MRLs) for food and feed (European Commission 2003, 2006), with the intention to lower the general exposure of the EU population (European Commission 2001b).
PCDD/F contaminated soil has been shown to affect congener composition and increase levels above background levels for food and feed at both local and regional scales (Pirard et al. 2005; Turrio-Baldassarri et al. 2007; Costera et al. 2006; Alcock et al. 2002; Harnly et al. 2000; Lindström et al. 2005; Riss et al. 1990). In several of these cases, MRLs for food and feed set by the EC were exceeded (e.g., Pirard et al. 2005; Turrio-Baldassarri et al. 2007), and unacceptably high daily intakes of PCDD/Fs for local residents are anticipated. Other studies have shown evidence of site-specific human exposure in the form of incrementally higher TEQ levels and altered PCDD/F congener distributions for residents and workers connected to contaminated sites (Dahlgren et al. 2003; Karouna-Renier et al. 2007; Riss et al. 1990; Pirard et al. 2005). However, most case studies related to contaminated sites have so far focused on a limited number of exposure media or exposure routes, while more comprehensive approaches including multiple exposure pathways are rare. Such studies are in demand, since the question whether contaminated sites represent a current or future risk can only be answered by detailed site-specific risk assessments (Weber et al. 2008a,b). This can be done either by site-specific modeling complemented by a few measurement data (Schuhmacher et al. 2004; Wiberg et al. 2007a) or by extensive field measurements.
The aim of the present study was to assess a broad spectrum of exposure routes at a selected CP-contaminated site in Sweden on the basis of extensive field measurement data. A variety of exposure media from the site, including locally produced vegetable and animal food products were analyzed for PCDD/F levels and congener distribution patterns. Exposure levels for ten pathways were calculated and compared to TDI and to exposure from consumption of fatty fish from the Baltic Sea. In parallel, a blood serum study of individuals potentially exposed to PCDD/Fs at the contaminated site and matched control individuals was conducted. The congener composition of the soil at the site suggests that it has been contaminated by a tetrachlorophenol (TeCP) agent, such as Ky-5, and to our best knowledge, no such site has been assessed as an exposure case study before. This paper is one in a series of articles addressing challenges and future relevance of dioxin and POP-contaminated sites (Weber et al. 2008a,b).
2 Materials and methods
2.1 Site description
The former saw mill site studied in this work is situated in northern Sweden (Fig. 1). The saw mill was active during 1862–1970 and used CP preservatives from the mid-1940s until closure. The site is recognized as one of the most PCDD/F polluted sites in Sweden, both in terms of magnitude and spatial extent of the contamination. The saw mill area covered a 1,500-m long and 100–200-m broad stretch on the shore of a deep bay (see Fig. 1). Sawing and impregnation were carried out in area A, while other areas were used for storage (C and G). The former drying house (D) was restored and transformed into an art gallery in the early 2000s but was closed in 2004 due to findings of severe indoor PCDD/F contamination. In area B, there is a permanent resident house with a small garden. Across the small county road, there are pastures for dairy cows (E) and a farm (approximately 700 m east of the road, not shown in Fig. 1). On a small hill south of the pastures, some of the former workmen’s dwellings remain (F), and one of these is today used as a hostel. At the end of the former timber yard, there is a camping area (H). Some campers live in trailers the whole summer, and several of them have returned to the camping area for decades.
Map of Scandinavia showing the location of the saw mill site together with an aerial photograph of the industrial area during its active period (1862–1970) (photo: National Land Survey of Sweden, Gävle, Sweden; with permission). A–I refer to the following subareas: A former saw mill and impregnation (the hot spot), B resident house, C former wood storage, D former drying house, E pastures and farm, F culture area with resident houses and hostel, G former timber yard, H cutter shaving tip and present-day camping, I village
Previously measured and interpolated PCDD/F concentrations in surface soil and groundwater from the area are presented in Table 1. Area A, hereafter designated as the hot spot, is the most polluted area, but significantly elevated concentrations in soil are also found around the resident house, at the camping area, the former timber storage, and at the former timber yard. In comparison, soil from a rural Swedish site was found to contain 1 ng WHO-TEQ/kg dw (Matscheko et al. 2002), and a reference groundwater sample taken outside the industrial area contained less than 0.00004 ng WHO-TEQ L−1 (Kramfors community, unpublished data). The national soil guide line values for residential areas is 20 ng WHO-TEQ/kg dw (including the dl-PCBs), while the corresponding value for Germany and Japan is 1,000 ng TEQ/kg dw. Considering this, most areas at the study site can be considered as affected by the point source, generally at a moderate level, and risk for elevated PCDD/F levels in the human food chain at a local scale cannot be excluded.
2.2 Sampling and chemical analysis
Exposure media from different parts of the polluted area were sampled according to Table 2. The sampling of air, groundwater, raspberries, eggs, milk, soil, and carrots were carried out during August–October 2006, while potatoes and grass were sampled in the summer of 2007. The air sampling was conducted during 5 days in October 2006 when the temperature ranged from +2 to +15°C. Details of the sampling, sample pretreatment, clean up and chemical analyses are given in Supporting information (SI).
In addition, blood serum from 16 individuals was sampled in 2006. Among them, eight individuals were potentially exposed at the study site via three exposure scenarios: (1) people that had been working at least 6 months with indoor renovation of the drying house (area D), (2) regularly returning camping residents (at least a total of 60 months of camping; area H) and (3) permanent residents from the village (40–60 years of residence, intake of locally produced food every day; area I). The first exposure scenario was particularly interesting, since it lasted for 6 months and all of the workers performed activities known to enhance particle exposure (i.e., drilling and sweeping) in highly contaminated areas. For each of the eight case individuals, control individuals were selected so that gender and age were matched. However, one of the control individuals had later to be excluded from the study due to medicolegal reasons.
Carrots and potatoes were washed prior to analysis, and soil samples were dried in a fume hood and then homogenized. The collected water was passed through glass fiber filter and polyurethane foam (PUF) plugs. Prior to extraction, all samples were fortified with 13C-labeled internal standards. The analytical procedures for milk, egg, grass, carrots, and potatoes were described by Danielsson et al. (2005) and Wiberg et al. (2006). Extraction of filters, PUF plugs, and soil was carried out by toluene Soxhlet–Dean–Stark extraction, and then the extracts were purified and fractionated using multilayer silica columns and activated carbon. The serum samples were analyzed at a contracted lab (ALS Czech Republic s.r.o., HRMS Laboratory, Pardubice, Czech Republic). In short, serum was mixed with ammonium sulfate, demineralized water, and methanol. The mixture was extracted by using hexane/diethylether mixture, and the extract was then dried by using anhydrous sodium sulfate. The raw extract was purified by liquid–liquid extraction using concentrated sulfuric acid, and then clean-up and fractionation was carried out using multilayer silica and Florisil columns. All instrumental analyses were performed by using gas chromatography–high-resolution mass spectrometry, and analytical conditions for all samples except serum are given in Wiberg et al. (2007b).
2.3 Quantification, quality assurance, and quality control
The details for quantification, quality assurance, and quality control are given in SI. In short, the quantification was made according to the isotope dilution method and by using calibration standards of all of the 17 2378-PCDD/Fs. TEQ levels were calculated using the WHO-TEFs from van den Berg et al. (2006) (WHO-TEF2005). The TEQ values reported in this study include half of the limit of detection (LOD) for congeners that were below LOD (medium-bound value), unless other is given. Average recoveries ranged from 75% to 96%.
3 Results and discussion
3.1 Concentrations of exposure media
The dioxin TEQ concentrations were elevated as compared to national reference samples for all exposure media except raspberries and cow’s milk (Tables 3 and 4). Despite the fact that the raspberries were collected directly at the hot spot at the contaminated area, the TEQ level (0.013 pg g−1 ww) was similar to the value of the reference sample and to the Swedish reference data for strawberries. In contrast, although the concentration in the pasture soil did not indicate extensive soil pollution (1–25 pg g–1 dw; area E in Tables 1 and 5), grass sampled in that area showed higher concentrations (0.26–0.61 pg WHO-TEQ g−1 ww) than Swedish reference data (0.04–0.13 pg WHO-TEQ g−1 ww; hay and silage fodder from different places in Sweden; Swedish Board of Agriculture, unpublished data). Grass sampled at area B, with significantly higher soil concentrations (see Table 4) but with no cows grazing, contained 0.18–0.21 pg WHO-TEQ g−1 ww, which is somewhat lower than the pasture levels (area E) but higher as compared to the reference data. Although clearly elevated, none of the grass samples exceeded the legislated MRL value of 0.75 pg WHO-TEQ g−1 ww. Generally, atmospheric particle deposition and uptake of gaseous PCDD/F are believed to be the most important sources to PCDD/F contamination for aboveground plants (Welsch-Pausch et al. 1995; Welsch-Pausch and McLachlan 1998; Thomas et al. 2002). In this case, however, adsorption of contaminated soil particles, e.g., via rain splash, and deposition of dust particles from the area likely constitute additional major sources. Our results clearly indicate that the grass, which in contrast to the raspberries grows next to the ground, is affected by the soil contamination. There were also signs of PCDD/F soil-to-air volatilization and gaseous uptake of lower chlorinated congeners (2378-TCDD, 2378-TCDF, and 23478-PeCDF). The grass that grew on highly contaminated soil (area B) showed distinctly higher relative abundance of low-chlorinated PCDD/Fs than the grass from the pasture (area E). The “area B/area E ratio” of concentrations in grass were 1.2–1.6 for the low chlorinated congeners (2378-TCDD, 2378-TCDF, and 23478-PeCDF), and 0.3–0.7 for higher chlorinated congeners. Modeling has suggested that 2378-TCDD may readily volatilize from soil (Trapp and Matthies 1997), and it can be assumed that other PCDD/F congeners with similar physical–chemical properties, such as 2378-TCDF and 23478-PeCDF (Åberg et al. 2008), exhibit the same behavior. The observed difference is therefore likely due to gaseous uptake from the highly contaminated soil in the resident garden.
Local PCDD/F sources, such as municipal solid waste incinerators, contaminated river systems, and contaminated soils have been shown to affect PCDD/F concentration and composition of cow’s milk and other animal products (Lake et al. 2005; Lindström et al. 2005; Pirard et al. 2003, 2005; Schmid et al. 2003; Riss et al. 1990). If the PCDD/F concentrations in grass are elevated, the milk from cows that graze this grass may also exhibit elevated concentrations, particularly if the contamination is dominated by readily bioavailable congeners. A compilation of feed-to-milk bioconcentration factors (BCFs) show that there is an inverse relation between bioconcentration and chlorination number with higher BCFs for low chlorinated congeners (Huwe and Smith 2005). The concentrations in cow’s milk found in this study (0.26–0.42 pg WHO-TEQ g−1 lw) are not elevated in comparison to national food control data (0.34 pg WHO-TEQ g−1 lw, see Table 3) and are significantly lower than the MRL. Thus, although the pasture where the cows graze showed elevated PCDD/F concentrations of both soil and grass, these circumstances did not affect the cow’s milk quality under current conditions, which likely is related to low bioavailability of the highly abundant congeners and also to the fact that the pasture grass is not the only feed for these cows (see SI).
In contrast, the TEQ concentrations of the eggs (3.91 ± 0.95 pg WHO-TEQ g−1 lw) were significantly higher than the reference data (0.70 ± 0.36 pg WHO-TEQ g−1 lw, see Table 3), although the farm soil contained low levels of PCDD/Fs (area E, farm yard in Table 4). The egg levels also exceeded or were close to the legislated EU limit value of 3 pg WHO-TEQ g−1 lw. It should, however, be noted that the reference values were obtained from large-scale organic breed production, and these are not necessarily comparable to levels in eggs from small-scale free-range breeding. Chicken fodder containing fish residues have earlier been identified as a major contributor to PCDD/Fs in eggs from free-range organic chickens. The fodder used at the study site contained low levels of PCDD/Fs (mean, 0.12 pg WHO-TEQ g−1 ww, adjusted to 12% water content), and most of the congeners were below the LOD. Several investigations have pointed out that chickens and eggs are sensitive receptors to PCDD/F (Schuler et al. 1997; Stephens et al. 1995; Harnly et al. 2000), and this is primarily related to ingestion of soil and insects (Harnly et al. 2000; Schuler et al. 1997) and the bioaccumulative properties of PCDD/Fs. It is particularly the tetra- and penta-substituted congeners that show high soil-to-egg transfer efficiency (Schuler et al. 1997). This means that free range chickens are subjected to relatively high exposure even at background concentrations. It has been shown that PCDD/Fs levels in eggs from free range chickens are generally higher than eggs from chickens that were bred indoors (Schuler et al. 1997). However, the extension of the biotransfer from soil to eggs varies considerably. Pirard et al. (2003) found that the PCDD/F content in eggs from free range chickens held on soil containing 11–13 pg TEQ g−1 dw contained 24–121 pg TEQ g−1 lw, while Air et al. (2003) reported somewhat lower egg concentrations (16–21 pg I-TEQ g−1 lw), although the soil levels were significantly higher (41–148 pg I-TEQ g−1 dw).
The PCDD/F concentrations in root crops from the resident garden were up to 20 times higher than Swedish reference concentrations for carrots (see Table 3). It is well known that carrots hyperaccumulate PCDD/Fs (Schroll and Schneuert 1993; Müller et al. 1994; Engwall and Hjelm 2000). On the other hand, several studies have pointed out that hydrophobic compounds are mainly accumulated in the peel of root crops (Hülster and Marschner 1993; Müller et al. 1994; Zohair et al. 2006). The root crops in the current study were not peeled before analysis, and it is likely that peeled carrots and potatoes contain lower levels of PCDD/Fs than those reported in Table 3.
The groundwater concentrations found in this study were within the range of those reported earlier from the study area (see Tables 1 and 4) but significantly higher (three orders of magnitude) than the reference sample taken outside the study area (<0.04 pg WHO-TEQ L−1, Kramfors community, unpublished data). Considering this, the groundwater at the study site must be considered as seriously polluted, probably as a result of co-transported contaminated colloids (Persson et al. 2008b).
Results from the air sampling at the study site are presented in Table 5. Since a majority of the PCDD/F congeners were present at levels below LOD in the ambient air samples, lower bound TEQ levels were calculated using detected levels only. One of the ambient air samples from area C was severely affected by diesel smoke and showed clearly deviating concentration values and congener pattern as compared to other ambient air samples. This sample was therefore excluded from the evaluation. The ambient air concentrations measured at the highly contaminated industrial area were not significantly different from the air concentration at the farm at the time of the sampling. Air concentrations of environmental pollutants fluctuate greatly over time primarily depending on air mass origin (Tysklind et al. 1993; Oehme et al. 1996; Sellström et al. 2009). Recently reported rural air data from Sweden include measurements from Råö at the Swedish west coast (fall–winter 2004 and summer 2005–2006; Swedish Environmental Research Institute 2006) and from Aspvreten located south of Stockholm (winter 2006/2007; Sellström et al. 2009), and the concentration range spanned from 0.6 to 29 fg TEQ m−3. The air levels in the current study overlapped these data but were in the high end and in some cases even exceeding.
Despite the fact that the drying house was not in use for several decades, indoor air contained highly elevated concentrations of PCDD/Fs (510–8,600 fg WHO-TEQ m−3, see Table 5). The large variation in the indoor data is likely due to various ventilation conditions since the highest value was obtained in a small closet. These values can be compared to PCDD/F levels in indoor air at a daycare center with pentachlorophenol (PCP) treated home interiors where concentrations ranged from 10 to 1,700 fg TEQ m−3 (Sagunski et al. 1989). For the indoor air samples, most of the PCDD/Fs were associated with the particle fraction (58–96% on the filter, see Table 5). In contrast, for the ambient air sampled at the industrial area, a majority of the PCDD/Fs were associated to the gaseous fraction, with only 20–27% on the filters (see Table 5). The farm reference sample and other rural Swedish air showed significantly higher values (see Table 5). The mixed indoor/ambient air sample exhibited an intermediate value of 41% of the PCDD/Fs in the particulate fraction. These findings may reflect variations in air particle abundance, with higher indoor values and possibly also higher at the farm, where human activity is higher than at the industrial area. The finding of high gaseous fractions of the industrial air in contrast to other Swedish rural air may further support a net volatilization in the air–soil gas exchange. Such net soil-to-air net volatilization has been shown for other organochlorines with similar physical–chemical properties as the PCDD/Fs in cases with high source soil source strengths (Bidleman et al. 2006).
3.2 Congener distribution patterns
In pollutant fate and source-to-receptor studies, it is often advantageous to study the composition of mixtures of contaminants together with concentration data. While environmental levels generally show large variation, the composition is normally less variable within a limited space. Similarities in congener patterns or homologue profiles indicate a close relationship between environmental matrices. Distribution patterns for 2378-substituted tetra- through octa-CDD/F congener concentrations and for TEQ concentrations of the samples from the polluted site and from reference samples are illustrated in Fig. 2.
All matrices from the study site, except the ambient air, were characterized by a large fraction of hepta-CDFs (HpCDFs) and octachlorodibenzofuran (OCDF) (see Fig. 2a), which together contributed 36–94% of the total PCDD/F abundance in the various media. The sum concentration of HpCDF congeners consisted mainly of 1234678-HpCDF, while 1234789-HpCDF was of less importance. A contamination pattern with a large fraction of 1234678-HpCDF and OCDF is typical for the PCDD/F pattern in TeCP agents, such as Ky-5 (Persson et al. 2007; congener pattern available in Table 1 in SI). Although not always apparent in the TEQ levels reported in Tables 3, 4, and 5, there is no doubt that the soil contamination at the study site has affected all exposure matrices with the possible exception of ambient air. There was a striking difference in the distribution patterns between the samples from the study site as compared to Swedish reference data (see Fig. 2c), which generally showed large fractions of OCDD and a much lower fraction of HpCDFs and OCDF (on average 12–33%). The reference samples were collected in rural areas, and the dominance of OCDD is therefore likely related to diffuse pollution via atmospheric deposition, rather than to other typical OCDD contamination such as PCP contaminated soil.
The congener distribution of indoor and ambient air from the study site was dominated by furans (PCDFs). High fractions of HpCDF and OCDF in the indoor air demonstrated major impact from the contaminated building. In ambient air, there was a higher fraction of tetra- and penta-CDFs, as compared to the reference air. This suggests that the ambient air is affected by volatilization of tetra- and penta-CDFs from the contaminated soil, again supporting the hypothesis of net volatilization of PCDD/F from soil-to-grass. It is further an indication on that, although tetra- and penta-CDFs constitute a minor fraction of the PCDD/F contamination in the soil (and in Ky-5; Table 1 in SI) and the concentration elevation of these congeners therefore is modest, their relatively high solubility in air can result in a change of the atmospheric PCDD/F composition of the air at a local scale. The reason for the difference between the ambient air from the study site and the reference air is, however, uncertain since air mass PCDD/F composition fluctuates greatly over time. Figure 2a and c also demonstrates that the contribution from tetra- through hexa-CDD/Fs was higher in milk and egg samples as compared to abiotic and plant matrices. This illustrates the higher biotransfer of these congeners compared to the higher chlorinated congeners.
Before evaluation of the TEQ patterns (see Fig. 2c, d), it should be noted that half of the LODs are included for congeners that were found to be below LOD. We used state-of the-art analytical methods and thus our LODs are low; nevertheless, this approach means that, in some cases, congeners that were not detected contribute significantly to the calculated TEQ value. In the EU legislation, the MRLs are given as upper-bound values, and the whole LOD is included in the calculation of TEQ values. Thus, the approach we have chosen (medium-bound values) is a compromise between only showing detected levels (lower-bound) and levels used in an EU risk assessment (upper-bound).
In the reference data, the tetra- through hexa-CDDs were generally large contributors to the TEQ levels in biota, together summing up to 49–74% to the total TEQ value. The corresponding fractions for the biotic matrices at the study site were lower (15–60%). Instead, the hexa- to HpCDFs were the major contributors, adding up to 20–81% of the total TEQ with the lowest fractions for milk and egg (20% and 56%, respectively). These observations reflect the impact of the soil contamination and also the limited biotransfer of higher chlorinated congeners. Despite that the distribution patterns in soil and groundwater were quite similar, the furans (PCDFs) contributed to as much as 89% of the TEQ in groundwater compared to approximately 56% in soil. This was mostly attributed to higher fractions of penta- and hexa-CDFs in the groundwater. Persson et al. (2008a) found that PCDFs are more highly associated to colloids than PCDDs and are therefore more mobile than the PCDDs. Our findings further support these observations and similarly suggest a relatively high soil-to-groundwater mobility of lower chlorinated PCDFs.
3.3 Concentrations in serum
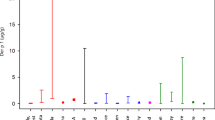
Concentrations of PCDD/Fs in serum found in this study varied largely (Table 6). However, no clear trend of the eight case individuals vs. the seven controls was seen. For several of the case individuals, their respective control individuals showed significantly higher TEQ serum levels. Considering the low number of samples and the limited information on dietary habits, care should be taken in interpreting these data. However, the average congener distribution patterns of each category provided evidence of a site-specific exposure (Fig. 3). The serum concentrations of HpCDF were clearly elevated for workers who participated in the renovation of the drying house. Despite a low TEF for HpCDF (WHO-TEF2005 = 0.01), this marker congener contributed 18% to the total serum TEQ for the worker category, compared to only 1.1% in the control individuals (see Fig. 2b). However, a higher TEQ value as compared to the control individual was only seen for two of the four workers (see Table 6). Neither village residents nor campers showed higher fractions of HpCDF as compared to the controls (see Fig. 3a), indicating that a long residence time at the study site area did not result in elevated exposure.
Since the indoor air was shown to be heavily contaminated by PCDD/Fs (see Table 5), the most likely exposure pathway for the indoor workers was inhalation of gas and particles and possibly also ingestion of air particles. Incremental PCDD/F exposure as a result of indoor work has been shown before. Shih et al. (2006) investigated the exposure to PCDD/F for temporary workers who were hired for maintenance of a municipal waste incinerator. The serum levels were analyzed before and after maintenance, and already after 1 month of work, they found a significant increase in TEQ serum PCDD/F levels (p < 0.05). Elevated PCDD/F serum levels as a result of residence near PCP-contaminated sites have also been reported. The mean serum level for people living close to a former PCP production plant in Pensacola (Florida, USA) was 45 pg WHO-TEQ g−1 lw (range, 8.4–710 WHO-TEQ g−1 lw), and the levels of these residents were shown to be higher than selected reference values (15–21 pg TEQ g−1 lw; Karouna-Renier et al. 2007).
A large variety of factors, such as diet (Tsukino et al. 2006; University of Michigan 2006; Chen et al. 2003), age (Tsukino et al. 2006; University of Michigan 2006), and body mass index (Collins et al. 2007), determine the blood serum PCDD/F levels of humans. The serum levels of Swedish men of various ages and feed habits were shown to vary from 3 to 66 pg WHO-TEQ g−1 lw (n = 26; 34–71 years of age; Rylander et al. 2009). The factors considered when choosing control individuals in the current study were age, gender, and to some extent dietary habits. Although limited information can be extracted from this survey because of the small number of individuals studies, the results clearly indicate that activities at or near a CP-contaminated site may affect the serum PCDD/F congener composition, but it does not necessarily result in increased serum TEQ levels.
4 Significance of exposure pathways related to PCDD/F-contaminated sites
Daily exposure doses were calculated for ten different exposure pathways related to the study site by using exposure media concentrations from Tables 3, 4, and 5 and by assuming a body weight of 70 kg. When applicable, average values were used. If only two values were available, the highest value was chosen. The average daily food intake rates for adults were taken from a Swedish food intake survey (Swedish National Food Administration 2002), and it was assumed that all of the ingested food types were of local origin, e.g., the average intake of fruit and berries of 126 g day−1 was here represented by 126 g of raspberries from the study area. The soil ingestion value (50 mg day−1) is recommended by US EPA for adult soil ingestion exposure (US Environmental Protection Agency 1996). The calculated doses were compared to the TDI of PCDD/F and dl-PCBs (European Commission 2001a) and to consumption of fatty fish (herring) from the Baltic Sea (Table 7).
Although interpretation of results from this kind of dose calculation is inevitably complicated by large variability, particularly in soil ingestion rates (US Environmental protection Agency 1996) and soil and groundwater concentrations (see Tables 1 and 5), it can be used to indicate whether exposure routes for an average adult person at a PCDD/F-contaminated site appears to pose a risk. The results show that several of the studied exposure pathways may contribute significantly to TDI. Ingestion of contaminated soil, groundwater, and potatoes contributed each 10% or more to the TDI and so did also inhalation of contaminated indoor air. It should also be noted that the TEQ contributions from the site-specific exposure routes to the total TDI are somewhat higher in reality, since the contribution from dl-PCBs were note measured and therefore not included in the comparison.
Despite that both egg and milk were produced on soil with relatively low levels of PCDD/Fs, these exposure pathways each made up approximately 4% of the TDI. Thus, as soon as the soil concentrations are elevated, these exposure pathways may rapidly become more important. Despite high cultivation soil concentrations (580 pg WHO-TEQ kg−1 dw), the carrot ingestion exposure pathway was of less importance in this scenario. The risk from ingestion of contaminated root crops in this scenario is probably even lower since peeled carrots and potatoes usually are consumed. The data also indicate that ingestion of berries grown on bushes pose very low risk even at high soil concentrations.
Since TDI refers to an external dose, no consideration is taken to the availability of the contaminants after ingestion/inhalation (i.e., the internal dose). It is conceivable that the intestinal tract availability of PCDD/Fs from a particulate matrix (e.g., soil and groundwater particles) is lower than for, e.g., fatty food (Budinsky et al. 2008; Ruby et al. 2002), and the real risk associated to soil and groundwater ingestion is possibly lower than indicated in Table 7. The pulmonary availability of 2378-TCDD was found to be high (95%; Diliberto et al. 1996), and thus, the inhalation exposure pathway may be critical for the total internal dose.
To be able to relate the contaminated site exposure to the ingestion of fish, the amount of Baltic herring (in gram) that would yield the same exposure dose as the actual exposure route was calculated. A portion of fish is approximately 120 g, and the average daily intake of fish and sea shells in Sweden is ~34 g (Swedish National Food Administration 2002). As illustrated in Table 7, only small amounts of Baltic Sea herring can be ingested before the site-specific exposure doses are exceeded, and each of the site-related exposure pathways constitutes <10% of a normal portion of Baltic herring.
The sum of the individual exposure routes in Table 7 makes up to 1.7 pg PCDD/F-WHO-TEQ kg−1 day−1 (corresponding to ~85% of TDI), where intake of food and drinking water constitutes approximately half of the dose, and the rest is mainly from indoor inhalation and unintentional soil ingestion. On top of this value, the PCB-TEQs should be added (not measured). In comparison, the average daily PCDD/F and dl-PCB exposure of an adult Swedish individual has been estimated to 1.3 pg WHO-TEQ kg−1 day−1 in a study based on concentration data from 1999 to 2004 and food intake data from 1997 to 1998 (Swedish National Food Administration 2002). It can thus be concluded that there are circumstances where living and working at or near a CP-contaminated site may pose a health risk due to elevated PCDD/F exposure. However, also considering that the exposure scenario depicted above approaches a worst case scenario and that drinking water in this area origins from elsewhere (provided by the municipality), a broad majority of the individuals at the site will likely not be at elevated risk, as also was indicated by the serum study (although the serum study does not reflect the external dose but the internal dose). This means that a conscious selection of food and a conscious human behavior (peeling of locally produced root crops, avoiding ingestion and indoor inhalation of particles) most likely eliminates risk for incremental PCDD/F exposure at this site. In this context, it is, however, important to stress that children ingest higher amounts of soil compared to adults and that children with pica behavior (deliberate soil ingestion) may ingest as much as several grams of soil per (US Environmental Protection Agency 1996). Considering this and the much lower body weight of children, it is evident that PCDD/F ingestion via soil may result in seriously high exposure for small children at PCDD/F-contaminated sites.
5 Conclusions, Recommendations and Perspectives
High abundance of a marker congener (1234678-HpCDF) in almost all of the exposure media demonstrates that PCDD/F-contaminated soils may constitute point sources affecting local food-chain contamination. Most exposure media also showed higher TEQ concentrations as compared to Swedish reference samples, and 1234678-HpCDF contributed to a large fraction of the TEQ. Since biota-soil accumulation factors vary considerably between PCDD/F congeners, the composition of the source must be considered when assessing the risks. In this case, the soil was dominated by 1234678-HpCDF and OCDF, which have relatively low bioaccumulation factors and are less available compared to lower chlorinated congeners. Despite this, the high source strength of 1234678-HpCDF in the soil (relative to the other congeners) made 1234678-HpCDF one of the most abundant congener in all the exposure media except in ambient air. A similar pollution pattern is found in the sediments of River Kymijoki, Southeastern Finland and in the coastal region of the Gulf of Finland, together constituting a mega-site that has been severely polluted by previous manufacturing of Ky-5 (Salo et al. 2008). In this case, the aquatic food chain was shown to be affected by the contamination as shown by slightly elevated PCDD/F levels in fish muscle and orders of magnitude higher in the liver compared with reference freshwater sites and the Baltic Sea. The concentrations in human fat did, however, not reveal generally high human exposure for the residents of the area, and levels were lower than in sea fishermen (Verta et al. 2009).
In the current study area, there were also indications of soil-to-air vaporization of tetra-to-penta CDD/Fs. Thus, contaminated soil with a higher abundance of lower chlorinated congeners (e.g., from a chloralkali source) will probably be of even higher importance as a point source. From a toxicological point of view, contamination of tetra–penta CDD/Fs poses the highest risk, since these congeners have both relatively high bioavailability and high toxic potency (high TEFs).
Site-related PCDD/F exposure was evident in the blood serum levels only for a few case individuals. Workers in the severely contaminated building appear to have been exposed since their fraction of HpCDFs to total PCDD/F levels in serum were notably higher as compared to the control individuals, although total TEQs for workers and controls were in the same range.
Exposure dose calculations showed that several of the studied exposure pathways may contribute significantly to TDI, especially ingestion of contaminated soil, groundwater, potatoes, and egg and inhalation of contaminated indoor air. Although none of the exposure routes resulted in daily exposure levels that exceed the TDI, the calculations showed that production of animal food products at or close PCDD/F-contaminated sites is potentially important for the soil-to-human exposure, and ingestion of eggs and milk may result in high exposure levels already at slightly elevated soil concentrations. For most residents living at or near PCDD/F contaminated sites, however, it is likely that their dietary habits and food preparation routines (peeling root crops, rinsing of vegetables, etc.) are more important for the internal body burden of PCDD/F rather than the magnitude of the measured soil concentrations at the site. It should, however, be noted that for small children and particularly those showing the pica behavior (i.e., deliberate soil ingestion), the intake of soil at a CP-contaminated site will probably be the major exposure pathway, and unacceptably high daily intake may occur for children. It is also worth to highlight that, in addition to CPs, also other types of wood protection agents were normally used at sawmills, e.g., heavy metal and polycyclic aromatic hydrocarbons containing formulations. Therefore, in comprehensive site-specific risk assessments of former saw mill sites, the risk for elevated exposure of other wood preservation contaminants than PCDD/Fs must also be considered.
References
Åberg A, MacLeod M, Wiberg K (2008) Physical-chemical property data for dibenzo-p-dioxin (DD), dibenzofuran (DF), and chlorinated DD/Fs: a critical review and recommended values. J Phys Chem Ref Data 37(4):1997–2008
Air V, Pless-Mulloli T, Schilling B, Paepke O (2003) Environmental non-feed contributors to PCDD/PCDF in free-range allotment poultry eggs: many questions and some answers. Organohalog Compd 63:126–129
Alcock RE, Sweetman AJ, Anderson DR, Fisher R, Jennings RA, Jones KC (2002) Using PCDD/F congener patterns to determine the source of elevated TEQ concentrations in cow’s milk: a case study. Chemosphere 46(3):383–391
Bidleman TF, Leone AD, Wong F, van Vliet L, Szeto S, Ripley BD (2006) Emission of legacy chlorinated pesticides from agricultural and orchard soils in British Columbia, Canada. Environ Toxicol Chem 25(3):1448–1457
Budinsky RA, Rowlands JC, Casteel S, Fent G, Cushing CA, Newsted J, Giesy JP, Ruby MV M, Aylward LL (2008) A pilot study of oral bioavailability of dioxins and furans from contaminated soils: impact of differential hepatic enzyme activity and species differences. Chemosphere 70(10):1774–1786
Chen HL, Lee CC, Liao PC, Guo YL, Chen CH, Su HJ (2003) Associations between dietary intake and serum polychlorinated dibenzo-p-dioxin and dibenzofuran (PCDD/F) levels in Taiwanese. Environ Res 91(3):172–178
Collins JJ, Bodner K, Burns CJ, Budinsky RA, Lamparski LL, Wilken M, Martin GD, Carson ML (2007) Body mass index and serum chlorinated dibenzo-p-dioxin and dibenzofuran levels. Chemosphere 66(6):1079–1085
Costera A, Feidt C, Marchand P, Le Bizec B, Rychen G (2006) PCDD/F and PCB transfer to milk in goats exposed to a long-term intake of contaminated hay. Chemosphere 64(4):650–657
Dahlgren J, Warshaw R, Horsak RD, Parker FM, Takhar H (2003) Exposure assessment of residents living near a wood treatment plant. Environ Res 92(2):99–109
Danielsson C, Wiberg K, Korytar P, Bergek S, Brinkman UAT, Haglund P (2005) Trace analysis of polychlorinated dibenzo-p-dioxins, dibenzofurans and WHO polychlorinated biphenyls in food using comprehensive two-dimensional gas chromatography with electron-capture detection. J Chromatogr A 1086(1–2):61–70
Diliberto JJ, Jackson JA, Birnbaum LS (1996) Comparison of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) disposition following pulmonary, oral, dermal, and parenteral exposures to rats. Toxicol Appl Pharm 138(1):158–168
Engwall M, Hjelm K (2000) Uptake of dioxin-like compounds from sewage sludge into various plant species—assessment of levels using a sensitive bioassay. Chemosphere 40(9–11):1189–1195
European Commission (1999) Compilation of EU dioxin exposure and health data. AEAT/EEQC/0016
European Commission (2001a) Opinion of the Scientific Committee on Food on the risk assessment of dioxins and dioxin-like PCBs in food. CS/CNTM/DIOXIN/20 final
European Commission (2001b) Community strategy for dioxins, furans and polychlorinated biphenyls. No 2001/C 322/02
European Commission (2003) Commission directive amending directive 2002/32/EC of the European Parliament and of the Council on undesirable substances in animal feed. No 2003/57/EC
European Commission (2006) Commission regulation setting maximum levels for certain contaminants in foodstuffs. EC No 1881/2006
Harnly ME, Petreas MX, Flattery J et al (2000) Polychlorinated dibenzo-p-dioxin and polychlorinated dibenzofuran contamination in soil and home-produced chicken eggs near pentachlorophenol sources. Environ Sci Technol 34:1143–1149
Huwe JK, Smith DJ (2005) Laboratory and on-farm studies on the bioaccumulation and elimination of dioxins from a contaminated mineral supplement fed to dairy cows. J Agr Food Chem 53(6):2362–2370
Hülster A, Marschner H (1993) Transfer of PCDD/PCDF from contaminated soils to food and crop plants. Chemosphere 27(1–3):439–446
Karouna-Renier NK, Rao KR, Lanza JJ, Davis DA, Wilson PA (2007) Serum profiles of PCDDs and PCDFs, in individuals near the Escambia Wood Treating Company Superfund Site in Pensacola, FL. Chemosphere 69(8):1312–1319
Kemakta (2007). Kramfors Community: Mariebergs f.d. sågverk, Huvudstudie om markföroreningar m.m. Huvudstudierapport: Miljöteknisk del.2007-10-12, Kemakta AR 2007-08 (in Swedish)
Lake IR, Foxall CD, Lovett AA, Fernandes A, Dowding A, White S, Rose M (2005) Effects of river flooding on PCDD/F and PCB levels in cow’s milk, soil and grass. Environ Sci Technol 39(23):9033–9038
Lindström G, Henriksson S, Hagberg J, Björnfoth H, van Bavel B (2005) Uptake of PCDDs, PCDFs and non-ortho PCBs in sheep from PCP contaminated sawmill soil. Organohalog Compd 67:1387–1389
Matscheko N, Tysklind M, de Wit C, Bergek S, Andersson R, Sellström U (2002) Application of sewage sludge to arable land-soil concentrations of polybrominated diphenyl ethers and polychlorinated dibenzo-p-dioxins, dibenzofurans, and biphenyls, and their accumulation in earthworms. Environ Toxicol Chem 21(12):2515–2525
Müller JF, Hülster A, Päpke O, Ball M, Marschner H (1994) Transfer of PCDD/PCDF from contaminated soils into carrots, lettuce and peas. Chemosphere 29(9–11):2175–2181
Oehme M, Haugen JE, Schlabach M (1996) Seasonal changes and relations between levels of organochlorines in arctic ambient air: first results of an all-year-round monitoring program at Ny-Alesund, Svalbard, Norway. Environ Sci Technol 30(7):2294–2304
Persson Y, Lundstedt S, Öberg L, Tysklind M (2007) Levels of chlorinated compounds (CPs, PCPPs, PCDEs, PCDFs and PCDDs) in soils at contaminated sawmill sites in Sweden. Chemosphere 66(2):234–242
Persson Y, Hernström K, Öberg L, Tysklind M, Enell A (2008a) Use of a column leaching test to study the mobility of chlorinated HOCs from a contaminated soil and the distribution of compounds between soluble and colloid phases. Chemosphere 71(6):1035–1042
Persson Y, Shchukarev A, Öberg L, Tysklind M (2008b) Dioxins, chlorophenols and other chlorinated organic pollutants in colloidal and water fractions of groundwater from a contaminated sawmill site. Environ Sci Poll R 15(6):463–471
Pirard C, Focant J-F, Massart A-C, De Pauw E (2003) Measurable impact of an old MSWI on the level of dioxins in free-range chickens and eggs grown in its vicinity. Organohalog Compd 64:158–161
Pirard C, Eppe G, Massart A-C, Fierens S, De Pauw E, Focant JF (2005) Environmental and human impact of an old timer incinerator in terms of dioxin and PCB level: a case study. Environ Sci Technol 39(13):4721–4728
Riss A, Hagenmaier H, Weberruss U, Schlatter C, Wacker R (1990) Comparison of PCDD/PCDF levels in soil, grass, cow’s milk, human blood and spruce needles in an area of PCDD/PCDF contamination through emissions from a metal reclamation plant. Chemosphere 21(12):1451–1456
Ruby MV, Fehling KA, Paustenbach DJ, Landenberger BD, Holsapple MP (2002) Oral bioaccessibility of dioxins/furans at low concentrations (50–350 ppt toxicity equivalent) in soil. Environ Sci Technol 36(12):4905–4911
Rylander L, Hagmar L, Wallin E, Kitti Sjöström A, Tysklind M (2009) Intra-individual variations and temporal trends in dioxin levels in human blood 1987 to 2000. Chemosphere. doi:10.1016/j.chemosphere.2009.05.036
Sagunski H, Forschner S, Kappos AD (1989) Indoor air pollution by dioxins in day-nurseries. Risk assessment and management. Chemosphere 18(1–6):1139–1142
Salo S, Verta M, Malve O, Korhonen M, Lehtoranta J, Kiviranta H, Isosaari P, Ruokojarvi P, Koistinen J, Vartiainen T (2008) Contamination of River Kymijoki sediments with polychlorinated dibenzo-p-dioxins, dibenzofurans and mercury and their transport to the Gulf of Finland in the Baltic Sea. Chemosphere 73(10):1675–1683
Schmid P, Gujer E, Zennegg M, Studer C (2003) Temporal and local trends of PCDD/F levels in cow’s milk in Switzerland. Chemosphere 53(2):129–136
Schroll R, Schneuert I (1993) Uptake pathways of octachlorodibenzo-p-dioxin from soil by carrot. Chemosphere 26(9):1631–1640
Schuhmacher M, Domingo JL, Garreta J (2004) Pollutants emitted by a cement plant: health risks for the population living in the neighborhood. Environ Res 95(2):198–206
Schuler F, Schmid P, Schlatter C (1997) The transfer of polychlorinated dibenzo-p-dioxins and dibenzofurans from soil into eggs of foraging chicken. Chemosphere 34(4):711–718
Sellström U, Egebäck A-L, McLachlan MS (2009) Identifying source regions for the atmospheric input of PCDD/Fs to the Baltic Sea. Atmos Environ 43(10):1730–1736
Shih TS, Chen HL, Wu YL, Lin YC, Lee CC (2006) Exposure assessment of polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/Fs) in temporary municipal-waste-incinerator maintenance workers before and after annual maintenance. Chemosphere 64(9):1444–1449
Stephens RD, Petreas MX, Hayward DG (1995) Biotransfer and bioaccumulation of dioxins and furans from soil: chicken as a model for foraging animals. Sci Total Environ 175(3):253–273
SWECO VIAK (2005) Kramfors Community: Mariebergs f.d. sågverk, fördjupad förstudie, Sundsvall 2005-11-25, rev 051129 (in Swedish)
Swedish Environmental Research Institute (2006) Dioxins in the Swedish Atmosphere: results from monitoring activities in 2004 and 2005. U 1969
Swedish National Food Administration (2002) Riksmaten 1997–1998. Kostvanor och Näringsintag i Sverige (in Swedish)
Swedish National Food Administration (2005) Intagsberäkningar för dioxin (PCDD/DF), dioxin-lika PCBer och metylkvicksilver via livsmedel. Report no. 25
Swedish National Food Administration (2007) Risk assessment of persistent chlorinated and brominated environmental pollutants in food. Report no. 9 (in English)
Thomas GO, Jones JL, Jones KC (2002) Polychlorinated dibenzo-p-dioxin and furan (PCDD/F) uptake by pasture. Environ Sci Technol 36(11):2372–2378
Trapp S, Matthies M (1997) Modeling volatilization of PCDD/F from soil and uptake into vegetation. Environ Sci Technol 31(1):71–74
Tsukino H, Hanaoka T, Sasaki H, Motoyama H, Hiroshima M, Tanaka T, Kabuto M, Turner W, Patterson DG, Needham L, Tsugane S (2006) Fish intake and serum levels of organochlorines among Japanese women. Sci Total Environ 359(1–3):90–100
Turrio-Baldassarri L, Abate V, Alivernini S, Battistelli CL, Carasi S, Casella M, Iacovella N, Iamiceli AL, Indelicato A, Scarcella C, La Rocca C (2007) A study on PCB, PCDD/PCDF industrial contamination in a mixed urban-agricultural area significantly affecting the food chain and the human exposure. Part I: soil and feed. Chemosphere 67(9):1822–1830
Tysklind M, Fängmark I, Marklund S, Lindskog A, Thaning L, Rappe C (1993) Atmospheric transport and transformation of polychlorinated dibenzo-p-dioxins and dibenzofurans. Environ Sci Technol 27(10):2190–2197
Tysklind M, Persson Y, Frankki S, Andersson R, Öberg L, Skyllberg U (2006) Chlorophenol sites in Sweden—a major dioxin reservoir with complex contamination pattern. Organohalog Compd 68:895–898
University of Michigan (2006) Measuring people’s exposure to dioxin contamination along the Tittabawassee River and surrounding areas. Findings from the University of Michigan Dioxin Exposure Study. August 2006
US Environmental Protection Agency (1996) Exposure factors handbook. Volume I of III—General factors. Update to Exposure Factors Handbook EPA/600/8-89/043, May 1989. Office of Research and Development, National Center for Environmental Assessment, US Environmental Protection Agency, Washington, DC, EPA/600/P-95/002Ba. SAB Review Draft
Van den Berg M, Birnbaum L, Bosveld TC, Brunström B, Cook P, Feeley M, Giesy JP, Hanberg A, Hasegawa R, Kennedy SW, Kubiak T, Larsen JC, van Leeuwen FXR, Liem AKD, Nolt C, Peterson RE, Poellinger L, Safe S, Schrenk D, Tillitt D, Tysklind M, Younes M, Waern F, Zacharewski T (1998) Toxic equivalency factors (TEFs) for PCBs, PCDDs, PCDFs for humans and wildlife. Environ Health Persp 106:775–792
Van den Berg M, Birnbaum LS, Denison M, De Vito M, Farland W, Feeley M, Fiedler H, Håkansson H, Hanberg A, Haws L, Rose M, Safe S, Schrenk D, Tohyama C, Tritscher A, Tuomisto J, Tysklind M, Walker N, Peterson RE (2006) The 2005 World Health Organization reevaluation of human and mammalian toxic equivalency factors for dioxins and dioxin-like compounds. Toxicol Sci 93(2):223–241
Verta M, Kiviranta H, Salo S, Malve O, Korhonen M, Verkasalo PK, Ruokojarvi P, Rossi E, Hanski A, Paatalo K, Vartiainen T (2009) A decision framework for possible remediation of contaminated sediments in the River Kymijoki, Finland. Environ Sci Poll R 16(1):95–105
Weber R, Gaus C, Tysklind M (2008a) Dioxin—contemporary and future challenges of historical legacies. Environ Sci Poll R 15(2):96–100
Weber R, Gaus C, Tysklind M, Johnston P, Forter M, Hollert H, Heinisch E, Holoubek I, Lloyd-Smith M, Masunaga S, Moccarelli P, Santillo D, Seike N, Symons R, Torres JPM, Verta M, Varbelow G, Vijgen J, Watson A, Costner P, Woelz J, Wycisk P, Zennegg M (2008b) Dioxin- and POP-contaminated sites-contemporary and future relevance and challenges. Environ Sci Poll R 15(5):363–393
Welsch-Pausch K, McLachlan MS (1998) Fate of airborne polychlorinated dibenzo-p-dioxins and dibenzofurans in an agricultural ecosystem. Environ Poll 102(1):129S–137S
Welsch-Pausch K, McLachlan M, Umlauf G (1995) Determination of the principal pathways of polychlorinated dibenzo-p-dioxins and dibenzofurans to Lolium multiflorum (Welsh ray grass). Environ Sci Technol 29(4):1090–1098
Wiberg K, Andersson PL, Berg H, Olsson P-E, Haglund P (2006) The fate of chiral organochlorine compounds and selected metabolites in intraperitoneally exposed Arctic char (Salvelinus alpinus). Environ Toxicol Chem 25(6):1465–1473
Wiberg K, Åberg A, McKone TE, Tysklind M, Hanberg A, MacLeod M (2007a) Model selection and evaluation for risk assessment of dioxin contaminated sites. AMBIO 36(6):458–466
Wiberg K, Sporring S, Haglund P, Björklund E (2007b) Selective pressurized liquid extraction of polychlorinated dibenzo-p-dioxins, dibenzofurans and dioxin-like polychlorinated biphenyls from food and feed samples. J Chromatogr A 1138(1–2):55–64
Zohair A, Salim A-B, Soyibo AA, Beek AJ (2006) Residues of polycyclic aromatic hydrocarbons (PAHs), polychlorinated biphenyls (PCBs) and organochlorine pesticides in organically-farmed vegetables. Chemosphere 63(4):541–553
Acknowledgements
The authors wish to thank residents of the study site who gave us permission to establish a garden plot on private land, allowed sampling at private land, and assisted in exposure media sampling. Kramfors community and contracted environmental consultant agencies are acknowledged for guidance of the area and data sharing. Katarina Stenman and Viktor Sjöblom (Umeå University) are acknowledged for plant identification and for grass and soil sampling, respectively. We also wish to thank Jaruslav Jurenka (ALS Czech Republic), who provided details about the blood serum analyses. This work was a part of a project financed by the Swedish Environmental Protection Agency; contract numbers E-92-05 and E-5-06.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Peter Luthardt
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Table 1
(PDF 27 kb)
Rights and permissions
About this article
Cite this article
Åberg, A., Tysklind, M., Nilsson, T. et al. Exposure assessment at a PCDD/F contaminated site in Sweden—field measurements of exposure media and blood serum analysis. Environ Sci Pollut Res 17, 26–39 (2010). https://doi.org/10.1007/s11356-009-0223-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-009-0223-4