Abstract
Purpose
Drug-induced sleep endoscopy (DISE) is useful in children with obstructive sleep apnea (OSA) that persists after adenotonsillectomy (AT), but its utility in surgically naïve children is unclear. We report polysomnography outcomes of surgically naïve children who underwent DISE-directed intervention because they were considered high risk for persistent OSA after adenotonsillectomy.
Methods
This study is a case series of 62 surgically naïve children with OSA who were considered high risk for persistence after AT and underwent DISE-directed intervention with pre- and postoperative polysomnography between 2012 and 2016. Analysis was performed with the paired t test.
Results
Children were on average 5.9 (± 5.5, 0.2–18.6) years old at the time of surgery, 68% male, 18% obese, and 60% white. Thirty-eight percent had a syndromic diagnosis: 19% trisomy 21, 11% hypotonic neuromuscular disorder, and 8% craniofacial condition. The remaining 62% were non-syndromic but underwent DISE because they had at least one risk factor for OSA persistence after AT (age > 7 years, black race, 1+ tonsils, obesity, and/or severe OSA). Forty-two percent underwent AT, while 58% underwent treatment other than AT, including 18% who had multilevel surgery. Children improved significantly in 4 out of 5 polysomnography parameters tested, including obstructive apnea-hypopnea index (oAHI; 22.2 to 7.2, p < 0.01) and oxygen nadir (82 to 87, p < 0.01). Thirty-eight (61%) had a postoperative oAHI < 5; 16 (21%) had a postoperative oAHI < 2.
Conclusion
DISE resulted in intervention other than AT in 58% of surgically naïve children at high risk for persistent OSA after AT. DISE-directed intervention resulted in significant mean improvement in postoperative OSA.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pediatric obstructive sleep apnea (OSA) syndrome is characterized by repetitive breathing pauses during sleep, resulting in sleep disruption and negative neurodevelopmental, cardiovascular, and behavioral consequences [1,2,3,4].
Adenotonsillectomy (AT) is the first-line treatment for children with OSA [5]. The Childhood Adenotonsillectomy Trial (CHAT) demonstrated that 79% of normal, healthy children ages 5–9 years will have polysomnographic resolution of their OSA after adenotonsillectomy [6]. However, on average, 40% of all children who undergo the procedure will have persistent sleep apnea, with rates of residual OSA up to 65% in children with comorbidities [7]. Persistent OSA after AT occurs in greater proportions in those with risk factors including age > 7 years [2, 8], severe baseline OSA (oAHI > 10) [2, 8,9,10], obesity [2, 7, 8, 11,12,13,14,15], trisomy 21 [16,17,18], craniofacial syndromes [19,20,21], and hypotonic neuromuscular disorders [10, 20]. Persistence frequently occurs when the tonsils and adenoids are not the only source of obstruction [22, 23]. Drug-induced sleep endoscopy (DISE) is a flexible fiber-optic assessment of the upper airway under a sleep-like state that facilitates targeted surgical intervention at specific sites of obstruction. DISE adds diagnostic benefit to awake endoscopy and clinical exam, and has been shown to influence surgical decision-making in many patients [24,25,26,27].
Previous study of DISE-directed intervention in otherwise healthy surgically naïve patients suggests that tonsillectomy with or without adenoidectomy is the DISE-directed intervention performed in the majority of these patients with a high percentage of success [28,29,30]. We sought to evaluate the utility of DISE in surgically naïve children with risk factors for persistent OSA after AT as there is little published on procedures performed and polysomnography outcomes in this often heterogeneous group of patients. We report the frequency with which the use of DISE resulted in a treatment plan that differed from standard-of-care AT. The primary aim was to evaluate the effectiveness of DISE-directed surgery in this series of surgically naïve children with OSA who underwent DISE prior to AT due to pre-identified risk factors for persistent OSA after AT. We hypothesized that there would be a significant improvement in five preselected polysomnography outcome measures, i.e., obstructive apnea-hypopnea index (oAHI, primary measure), apnea-hypopnea index (AHI), oxygen desaturation index (ODI), oxygen nadir (O2 nadir), and percent of sleep time with end tidal CO2 > 50 mmHg (%TST CO2 > 50).
Methods
Data collection
Following approval by the Seattle Children’s Hospital review board, institutional data was queried to identify patients who had undergone DISE-directed surgery along with pre- and postoperative polysomnography from February 2012 to September 2016. Chart review was performed on those identified to collect polysomnography parameters, DISE findings, DISE-directed surgeries performed, and demographic data including age at surgery, sex, race, preoperative tonsil size, previous airway surgery, sleep-related comorbidities, and perioperative body mass index (percentile for age). Data was stored in REDCap, a secure, web-based research database application [31].
All children included in the analysis underwent standard overnight, monitored pre- and postoperative polysomnography in the Seattle Children’s sleep laboratory within 12 months before and within 24 months after DISE-directed surgery. No child underwent any additional surgery or continuous positive airway pressure (CPAP) treatment in the time between DISE-directed surgery and postoperative polysomnography. Polysomnography included electroencephalography, electromyography, electrooculography, electrocardiogram, and measurement of oronasal airflow by a nasal pressure cannula and thermistor, chest/abdominal excursion, capnography/transcutaneous CO2, and pulse oximetry. Studies were scored by board-certified sleep medicine physicians using the pediatric scoring guidelines of the American Academy of Sleep Medicine [32]. Obstructive apnea was defined as ≥ 90% reduction in airflow lasting ≥ 2 breath cycles. Hypopnea was defined as a ≥ 30% reduction in airflow lasting ≥ 2 breath cycles, associated with an arterial desaturation ≥ 3% from baseline and/or a cortical arousal. AHI was defined as the number of apneas (obstructive and central) and hypopneas per hour of sleep. oAHI was defined as the number of obstructive apneas and hypopneas per hour of sleep. Oxyhemoglobin desaturation index was defined as the number of desaturations ≥ 3% from baseline per hour of sleep. Oxygen nadir (O2) was the lowest level of oxygen saturation detected on pulse oximetry, and percent of total sleep time with end tidal carbon dioxide > 50 mmHg (%TST CO2 > 50) was recorded by capnography. Mild OSA was defined as oAHI > 1 to < 5, moderate ≥ 5 to < 10, and severe ≥ 10 events per hour [33].
Children included in this analysis underwent DISE if they had an oAHI > 1, an AHI > 2, and significant risk factors for persistent OSA after AT. Children considered for DISE prior to AT fell into two groups: syndromic and non-syndromic. The syndromic group included children with trisomy 21, craniofacial conditions, and neuromuscular disorders with hypotonia as a prominent feature (referred to as “hypotonia” throughout). The non-syndromic group included otherwise healthy children that underwent DISE due to factors that increased their risk for persistent OSA after AT including the following: age greater than 7 years old, small tonsils (Brodsky 1+), obesity (> 95th percentile for age), and/or severe baseline OSA (oAHI ≥ 10 events/h) [8,9,10, 13, 16, 20]. DISE was performed by three surgeons (SP, KJ, DH) with the patient supine in the operating room under spontaneous breathing conditions. Patients underwent inhaled induction followed by maintenance propofol anesthetic without the use of topical anesthetics or decongestants. A validated scoring system was employed to grade obstruction at five levels, i.e., the adenoid, palate, lateral pharyngeal wall, tongue base, and supraglottis. Obstruction is scored at each level on a scale as follows: 0 (none), 1 (< 50% obstructed), 2 (> 50% obstructed), and 3 (complete) with a total score ranging from 0 to 15 [34].
Where appropriate, surgery was performed at the level of the adenoid, palatine tonsil, lingual tonsil, and/or supraglottis directly following sleep endoscopy. Surgical decision-making was based on DISE findings in combination with individual patient factors and family preferences.
Analysis
Statistical analysis was performed with the STATA/MP 15 software (StataCorp LP, College Station, TX). Descriptive summaries for continuous variables are reported as ranges and means ± standard deviations. Frequencies are reported for categorical variables. Univariate associations between intervention type (AT vs non-AT) and baseline patient characteristics were tested with the χ2 test, Fisher’s exact test, or Wilcoxon rank-sum test, where appropriate. Univariate associations between intervention type (AT vs non-AT) and obstruction scores at each level and overall were compared using the Wilcoxon rank-sum test. Pre- and postoperative polysomnography parameters were statistically compared with the paired t test. The null hypothesis was rejected at p < 0.05.
Results
Of 117 children who underwent DISE from 2012 to 2016, 86 (74%) were surgically naïve. Of those, 62 (72%) had pre- and postoperative polysomnography data and were included in the analysis. Forty-two percent underwent adenotonsillectomy, while 58% underwent treatment other than adenotonsillectomy.
Baseline demographic characteristics are shown in Table 1. Children were on average 5.9 (± 5.5, 0.2–18.6) years old at the time of DISE, 68% percent male, 18% obese (≥ 95th percentile for age), and 60% percent white. The majority (82%) had Brodsky 2+ or smaller tonsils on preoperative examination; 58% had 1+ tonsils. The distribution of tonsil size was significantly different in the two intervention groups; children who underwent non-AT interventions had a significantly higher proportion of 1+ tonsils than those who underwent AT. Children who underwent AT had a significantly higher proportion of severe baseline OSA (p = 0.01) than those who underwent non-AT intervention. Twenty-four subjects (38%) had an underlying syndromic diagnosis, including trisomy 21 (19%), hypotonic neuromuscular disorder (11%), or a craniofacial condition (8%). The proportion of children with a syndromic diagnosis did not differ significantly in the two intervention groups. A total of 8 subjects (12.9%) had a diagnosis of gastroesophageal reflux disease (GERD). Of those, 6 were on an antacid medication and 2 were not.
Mean sleep endoscopy scores are presented in Table 2. Children who underwent AT had significantly higher mean obstruction scores at the adenoid and lateral pharyngeal wall (tonsil) levels than those who underwent non-AT intervention (all p < 0.01). Children who underwent non-AT intervention had significantly higher obstruction scores at the supraglottis (p < 0.001), but total obstruction scores and scores at the velum and tongue base were not significantly different between the two groups (p > 0.05).
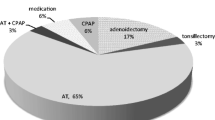
Frequencies of DISE-directed surgery performed are presented in Table 3. While a substantial proportion (42%) underwent AT, more patients (58%) underwent surgery that differed from standard-of-care adenotonsillectomy (non-AT intervention). Overall, 40% had single-level intervention at either the adenoid (7%), tonsil (3%), supraglottis (26%), or lingual tonsil (3%). Of 62 total patients, 11 (18%) had non-AT multilevel surgery. A substantial portion (43%) underwent supraglottoplasty for sleep-state laryngomalacia, either in isolation or in combination with other procedures. Supraglottoplasty was significantly associated with younger age and smaller tonsils (both p < 0.001). The mean age of children who underwent supraglottoplasty was 4.6 ± 5.0 years old (range 0.2–17.5 years) compared with a mean age of 6.9± 5.6 years (range 1.0–18.6 years; p = 0.03). There were no significant differences in any other baseline parameters between children who did and did not undergo supraglottoplasty (all p > 0.05).
DISE-directed surgery was successful in reducing the total number of obstructive breathing events (oAHI) in 79% of subjects, though not all of those children improved in their categorical OSA severity. Figure 1 displays the distribution of pre- and postsurgical OSA severity. Baseline OSA severity was defined by obstructive AHI, with 24% of patients having mild (oAHI < 5), 34% moderate (oAHI ≥ 5 to < 10), and 42% severe OSA (oAHI ≥ 10). None of the children met the criteria for sleep hypoventilation. After DISE-directed surgery, nearly two-thirds (61%) had mild or no OSA, 18% had moderate OSA, and 21% had severe OSA. Overall, 44% of subjects had the same degree of OSA before and after surgery, 48% improved by at least one category (i.e., moderate to mild or severe to either moderate or mild), and 8% worsened by one category. Of the children whose OSA category worsened or did not improve, 47% were mild at baseline, 19% moderate, and 34% severe. Of those who improved, 50% had moderate and 50% severe OSA. This difference in distribution of baseline OSA was significant (p < 0.01). There was a trend for older age in those whose OSA category worsened or failed to improve (7.2 years vs 4.5 years, p = 0.53). There were no other systematic or significant differences between the two groups (all p > 0.05). Furthermore, the five children who worsened did not differ significantly from the remainder with respect to age, sex, race, obesity status, or comorbidity status (all p > 0.05).
At baseline, 23% of children in the AT group had mild OSA; 15% had moderate OSA, and 62% had severe OSA. After surgery, the AT group had 38% with mild, 31% with moderate, and 31% with severe OSA. In the group who underwent non-AT intervention, 25% had mild baseline OSA, 47% moderate OSA, and 28% severe OSA. After DISE-directed surgery, the group had 78% with mild, 8% moderate, and 14% severe OSA. The distribution of OSA severity was significantly different between the non-AT and AT groups, with a larger proportion of children in the non-AT than the AT group having mild postoperative OSA (78% vs 38%, χ2 = 10.1; < 0.01).
Sleep test results are displayed in Table 4. The series as a whole improved from severe baseline OSA with a mean preoperative oAHI of 22.2 events per hour to moderate OSA with a mean postoperative oAHI of 7.2 events per hour (p < 0.01). Additionally, there were overall statistically significant improvements in AHI, ODI, and oxygen saturation nadir. Though there was an improvement in percent total sleep time with CO2 > 50 mmHg, this change was not statistically significant.
Discussion
We report improved polysomnography outcomes after DISE-directed surgery in a series of 62 surgically naïve patients with preoperative risk factors for adenotonsillectomy failure. Fifty-eight percent of patients underwent an intervention other than standard-of-care adenotonsillectomy. DISE-directed surgery was successful in reducing the total number of obstructive respiratory events in 79% of subjects. The mean severity of sleep apnea improved from severe to moderate, and postoperative oAHI was mild (< 5) in 61% of patients in this high-risk group. Children who underwent AT did not experience as much improvement overall as those who underwent non-AT intervention.
This study and the series that have come before it are important steps in better understanding DISE as a tool to direct surgical intervention. While some authors argue that DISE should be reserved for cases of OSA that do not improve after AT [27], others suggest that DISE be used routinely in all patients prior to adenotonsillectomy [30]. Studies by Boudewyns et al. and Gazzaz et al. have demonstrated that DISE changes the surgical decision-making in healthy surgically naïve patients in 25–35% of cases [26, 30]. It stands to reason that DISE may be of even greater benefit in patients at high risk for adenotonsillectomy failure than in healthy children.
Polysomnography outcomes after DISE-directed surgery in patients at risk for OSA persistence have been previously reported in two studies; however, even after the use of DISE, the majority underwent intervention at the tonsils, adenoids, or both. Maris et al. reported polysomnography outcomes in 25 surgically naïve children with trisomy 21 [35]. Sixty-four percent underwent adenotonsillectomy, 28% tonsillectomy, and 8% adenoidectomy. Mean oAHI decreased from 11.4 to 5.6 events/h. Truong et al. reported a decrease in AHI from 13.8 to 8.0 events per hour in 39 patients, 28% of whom had a coexisting syndromic diagnosis and the majority (90%) of whom underwent adenotonsillectomy [36]. In addition to the above, He et al. [37] reported a decrease in oAHI from 16.3 to 6.5 events per hour in 21 surgically naïve infants, though 70% underwent tracheostomy for treatment, making it difficult to compare with outcomes of studies reporting primarily on upper airway interventions.
Children in the present series experienced overall similar levels of improvement from severe to moderate OSA. In contrast to the above studies, however, the majority of children in this study did not undergo adenotonsillectomy. An equal proportion of subjects in this study underwent single-level intervention and adenotonsillectomy (each ~ 40%), with a smaller proportion undergoing non-AT multilevel intervention (18%). This may reflect differences in the samples studied, or changes in practice patterns over time or by region.
We performed a substantial number of supraglottoplasties (42%) in isolation or combination with other airway levels to treat sleep-state laryngomalacia. Many infants with awake inspiratory stridor diagnosed with classic congenital laryngomalacia can also have sleep-state laryngeal obstruction and can benefit from polysomnography to evaluate for OSA and use of DISE to confirm the site of obstruction prior to supraglottoplasty [38]. A meta-analysis pooled four studies of 38 infants with laryngomalacia and coincident OSA and found that the AHI improved significantly from 21 to 3.8 events per hour, though 88% still had residual disease [39].
State-dependent laryngomalacia was first described in five patients by Amin and Isaacson [40]. Digoy et al. [41] later reported polysomnography on outcomes of supraglottoplasty in 36 patients > 12 months of age with sleep-state laryngomalacia. They found an overall decrease in AHI from 13.3 to 4.1 events per hour in this series. However, in contrast to our study, 75% of those subjects had undergone previous AT at the time of their supraglottoplasty. A recent meta-analysis of isolated supraglottoplasty for laryngomalacia with obstructive sleep apnea pooled 64 patients with sleep-state laryngomalacia and found that the mean AHI decreased from 14 to 3.3 events per hour [42]. It is still unknown whether relieving obstruction at higher levels of the airway (by performing AT for example) has an impact on sleep-state laryngomalacia. Our decision to perform supraglottoplasty either in isolation or in conjunction with additional levels was driven primarily by the amount of obstruction seen at the supraglottis; 79% of children who underwent supraglottoplasty had maximal obstruction, and an additional 13% had > 50% obstruction at the supraglottic level.
This study has several limitations. This was a retrospective series from a single center. The high degree of comorbidity and the racial breakdown in the series limit the generalizability of results. However, the medical complexity of the series reflects the population seen at a large tertiary pediatric referral center. For many subjects, not all sites of maximal obstruction were addressed simultaneously, due in some cases to parental preference to minimize procedures and in others to the clinical practice of staging upper airway surgery to avoid complications. This series did not capture all stages of sleep surgery for all patients and thus may underrepresent final surgical response after all stages are complete. We did not evaluate for subglottic pathology in conjunction with DISE in this series; however, recent work by Bliss et al. suggests that this is not routinely necessary as subglottic lesions are rare [43]. Undertreatment would bias results toward the null, and despite this bias, the group still experienced significant improvement across respiratory parameters. As this was a case series without a control group, it is unclear how much benefit DISE adds beyond adenotonsillectomy alone in high-risk patients, which warrants further study. Finally, we did not collect data on quality-of-life measures or specific sleep apnea–related symptoms, which are important clinical measures that are not always well-correlated with sleep test parameters [9, 44].
Conclusion
DISE-directed surgery in a surgically naïve population resulted in a surgical plan that differed from adenotonsillectomy in 58% of patients. Intervention was successful in reducing oAHI in 79% of subjects, and significant improvement was seen in 4 out of 5 polysomnography parameters tested. The mean severity of sleep apnea improved overall from severe to moderate, and postoperative oAHI was mild (< 5) in 61% of patients in this high-risk group. Children who underwent non-AT intervention experienced overall greater improvement than children who underwent AT. Our results highlight the severity of baseline disease in this challenging population as well as the potential utility of DISE in directing surgical intervention that may differ from standard-of-care adenotonsillectomy. They also underscore the challenge in completely eliminating obstruction in a highly comorbid population and the need for prospective, controlled studies to identify how much benefit DISE affords over adenotonsillectomy in patients at high risk for persistent OSA after AT.
References
Capdevila OS, Kheirandish-Gozal L, Dayyat E, Gozal D (2008) Pediatric obstructive sleep apnea: complications, management, and long-term outcomes. Proc Am Thorac Soc 5(2):274–282. https://doi.org/10.1513/pats.200708-138MG
Imanguli M, Ulualp SO (2016) Risk factors for residual obstructive sleep apnea after adenotonsillectomy in children. Laryngoscope 126(11):2624–2629. https://doi.org/10.1002/lary.25979
Bixler EO, Vgontzas AN, Lin HM, Liao D, Calhoun S, Fedok F, Vlasic V, Graff G (2008) Blood pressure associated with sleep-disordered breathing in a population sample of children. Hypertension 52(5):841–846
Galland BC, Tripp EG, Gray A, Taylor BJ (2011) Apnea-hypopnea indices and snoring in children diagnosed with ADHD: a matched case-control study. Sleep Breath 15(3):455–462. https://doi.org/10.1007/s11325-010-0357-0
Stillwell PC, Chapman DL, Ward SLD, Howenstine M, Light MJ, McColley SA, Schaeffer DA, Wagener JS (2002) Clinical practice guideline: diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics 109(4):704–712
Marcus CL, Moore RH, Rosen CL, Giordani B, Garetz SL, Taylor HG, Mitchell RB, Amin R, Katz ES, Arens R, Paruthi S, Muzumdar H, Gozal D, Thomas NH, Ware J, Beebe D, Snyder K, Elden L, Sprecher RC, Willging P, Jones D, Bent JP, Hoban T, Chervin RD, Ellenberg SS, Redline S, Childhood Adenotonsillectomy T (2013) A randomized trial of adenotonsillectomy for childhood sleep apnea. N Engl J Med 368(25):2366–2376. https://doi.org/10.1056/NEJMoa1215881
Friedman M, Wilson M, Lin HC, Chang HW (2009) Updated systematic review of tonsillectomy and adenoidectomy for treatment of pediatric obstructive sleep apnea/hypopnea syndrome. Otolaryngol Head Neck Surg 140(6):800–808. https://doi.org/10.1016/j.otohns.2009.01.043
Bhattacharjee R, Kheirandish-Gozal L, Spruyt K, Mitchell RB, Promchiarak J, Simakajornboon N, Kaditis AG, Splaingard D, Splaingard M, Brooks LJ, Marcus CL, Sin S, Arens R, Verhulst SL, Gozal D (2010) Adenotonsillectomy outcomes in treatment of obstructive sleep apnea in children: a multicenter retrospective study. Am J Respir Crit Care Med 182(5):676–683. https://doi.org/10.1164/rccm.200912-1930OC
Mitchell RB (2007) Adenotonsillectomy for obstructive sleep apnea in children: outcome evaluated by pre- and postoperative polysomnography. Laryngoscope 117(10):1844–1854. https://doi.org/10.1097/MLG.0b013e318123ee56
Mitchell RB, Kelly J (2004) Outcome of adenotonsillectomy for severe obstructive sleep apnea in children. Int J Pediatr Otorhinolaryngol 68(11):1375–1379. https://doi.org/10.1016/j.ijporl.2004.04.026
Mitchell RB, Kelly J (2004) Adenotonsillectomy for obstructive sleep apnea in obese children. Otolaryngol Head Neck Surg 131(1):104–108. https://doi.org/10.1016/j.otohns.2004.02.024
Mitchell RB, Kelly J (2007) Outcome of adenotonsillectomy for obstructive sleep apnea in obese and normal-weight children. Otolaryngol Head Neck Surg 137(1):43–48. https://doi.org/10.1016/j.otohns.2007.03.028
Mitchell RB, Boss EF (2009) Pediatric obstructive sleep apnea in obese and normal-weight children: impact of adenotonsillectomy on quality-of-life and behavior. Dev Neuropsychol 34(5):650–661. https://doi.org/10.1080/87565640903133657
Lennon CJ, Wang RY, Wallace A, Chinnadurai S (2017) Risk of failure of adenotonsillectomy for obstructive sleep apnea in obese pediatric patients. Int J Pediatr Otorhinolaryngol 92:7–10. https://doi.org/10.1016/j.ijporl.2016.09.026
Nino G, Gutierrez MJ, Ravindra A, Nino CL, Rodriguez-Martinez CE (2012) Abdominal adiposity correlates with adenotonsillectomy outcome in obese adolescents with severe obstructive sleep apnea. Pulm Med 2012:351037. https://doi.org/10.1155/2012/351037
Thottam PJ, Trivedi S, Siegel B, Williams K, Mehta D (2015) Comparative outcomes of severe obstructive sleep apnea in pediatric patients with trisomy 21. Int J Pediatr Otorhinolaryngol 79(7):1013–1016. https://doi.org/10.1016/j.ijporl.2015.04.015
Maris M, Verhulst S, Wojciechowski M, Van de Heyning P, Boudewyns A (2017) Outcome of adenotonsillectomy in children with Down syndrome and obstructive sleep apnoea. Arch Dis Child 102(4):331–336. https://doi.org/10.1136/archdischild-2015-310,351
Shete MM, Stocks RM, Sebelik ME, Schoumacher RA (2010) Effects of adeno-tonsillectomy on polysomnography patterns in Down syndrome children with obstructive sleep apnea: a comparative study with children without Down syndrome. Int J Pediatr Otorhinolaryngol 74(3):241–244. https://doi.org/10.1016/j.ijporl.2009.11.006
Amonoo-Kuofi K, Phillips SP, Randhawa PS, Lane R, Wyatt ME, Leighton SE (2009) Adenotonsillectomy for sleep-disordered breathing in children with syndromic craniosynostosis. J Craniofac Surg 20(6):1978–1980. https://doi.org/10.1097/SCS.0b013e3181bd2c9a
Mitchell RB, Kelly J (2005) Outcome of adenotonsillectomy for obstructive sleep apnea in children under 3 years. Otolaryngol Head Neck Surg 132(5):681–684. https://doi.org/10.1016/j.otohns.2004.12.010
Willington AJ, Ramsden JD (2012) Adenotonsillectomy for the management of obstructive sleep apnea in children with congenital craniosynostosis syndromes. J Craniofac Surg 23(4):1020–1022. https://doi.org/10.1097/SCS.0b013e31824e6cf8
Lin AC, Koltai PJ (2009) Persistent pediatric obstructive sleep apnea and lingual tonsillectomy. Otolaryngol Head Neck Surg 141(1):81–85. https://doi.org/10.1016/j.otohns.2009.03.011
Mase CA, Chen ML, Horn DL, Parikh SR (2015) Supraglottoplasty for sleep endoscopy diagnosed sleep dependent laryngomalacia. Int J Pediatr Otorhinolaryngol 79(4):511–515. https://doi.org/10.1016/j.ijporl.2015.01.018
Fishman G, Zemel M, DeRowe A, Sadot E, Sivan Y, Koltai PJ (2013) Fiber-optic sleep endoscopy in children with persistent obstructive sleep apnea: inter-observer correlation and comparison with awake endoscopy. Int J Pediatr Otorhinolaryngol 77(5):752–755. https://doi.org/10.1016/j.ijporl.2013.02.002
Ulualp SO, Szmuk P (2013) Drug-induced sleep endoscopy for upper airway evaluation in children with obstructive sleep apnea. Laryngoscope 123(1):292–297. https://doi.org/10.1002/lary.23832
Gazzaz MJ, Isaac A, Anderson S, Alsufyani N, Alrajhi Y, El-Hakim H (2017) Does drug-induced sleep endoscopy change the surgical decision in surgically naive non-syndromic children with snoring/sleep disordered breathing from the standard adenotonsillectomy? A retrospective cohort study. J Otolaryngol Head Neck Surg 46(1):12. https://doi.org/10.1186/s40463-017-0190-6
Galluzzi F, Pignataro L, Gaini RM, Garavello W (2015) Drug induced sleep endoscopy in the decision-making process of children with obstructive sleep apnea. Sleep Med 16(3):331–335. https://doi.org/10.1016/j.sleep.2014.10.017
Boudewyns A, Saldien V, Van de Heyning P, Verhulst S (2018) Drug-induced sedation endoscopy in surgically naive infants and children with obstructive sleep apnea: impact on treatment decision and outcome. Sleep Breath 22(2):503–510. https://doi.org/10.1007/s11325-017-1581-7
Boudewyns A, Van de Heyning P, Verhulst S (2017) Drug-induced sedation endoscopy in children < 2 years with obstructive sleep apnea syndrome: upper airway findings and treatment outcomes. Eur Arch Otorhinolaryngol 274(5):2319–2325. https://doi.org/10.1007/s00405-017-4481-3
Boudewyns A, Verhulst S, Maris M, Saldien V, Van de Heyning P (2014) Drug-induced sedation endoscopy in pediatric obstructive sleep apnea syndrome. Sleep Med 15(12):1526–1531. https://doi.org/10.1016/j.sleep.2014.06.016
Harris P, Taylor R, Thielke R, Payne J, Gonzalez N, Conde J (2009) Research electronic data capture (REDCap)--a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42(2):377–381
Berry RB, Budhiraja R, Gottlieb DJ, Gozal D, Iber C, Kapur VK, Marcus CL, Mehra R, Parthasarathy S, Quan SF, Redline S, Strohl KP, Davidson Ward SL, Tangredi MM, American Academy of Sleep M (2012) Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med 8(5):597–619. https://doi.org/10.5664/jcsm.2172
Sheldon SH, Ferber R, Kryger MH (2014) Principles and practice of pediatric sleep medicine. Second edition. Elsevier Saunders, London
Chan DK, Liming BJ, Horn DL, Parikh SR (2014) A new scoring system for upper airway pediatric sleep endoscopy. JAMA Otolaryngol Head Neck Surg 140(7):595–602. https://doi.org/10.1001/jamaoto.2014.612
Maris M, Verhulst S, Saldien V, Van de Heyning P, Wojciechowski M, Boudewyns A (2016) Drug-induced sedation endoscopy in surgically naive children with Down syndrome and obstructive sleep apnea. Sleep Med 24:63–70. https://doi.org/10.1016/j.sleep.2016.06.018
Truong MT, Woo VG, Koltai PJ (2012) Sleep endoscopy as a diagnostic tool in pediatric obstructive sleep apnea. Int J Pediatr Otorhinolaryngol 76(5):722–727. https://doi.org/10.1016/j.ijporl.2012.02.028
He S, Peddireddy NS, Smith DF, Duggins AL, Heubi C, Shott SR, Ishman SL (2018) Outcomes of drug-induced sleep endoscopy-directed surgery for pediatric obstructive sleep apnea. Otolaryngol Head Neck Surg 158(3):559–565. https://doi.org/10.1177/0194599817740332
Bhushan B, Schroeder JW Jr, Billings KR, Giancola N, Thompson DM (2019) Polysomnography outcomes after supraglottoplasty in children with obstructive sleep apnea. Otolaryngol Head Neck Surg 161(4):694–698. https://doi.org/10.1177/0194599819844512
Farhood Z, Ong AA, Nguyen SA, Gillespie MB, Discolo CM, White DR (2016) Objective outcomes of supraglottoplasty for children with laryngomalacia and obstructive sleep apnea: a meta-analysis. JAMA Otolaryngol Head Neck Surg 142(7):665–671. https://doi.org/10.1001/jamaoto.2016.0830
Amin MR, Isaacson G (1997) State-dependent laryngomalacia. Ann Otol Rhinol Laryngol 106(11):887–890. https://doi.org/10.1177/000348949710601101
Digoy GP, Shukry M, Stoner JA (2012) Sleep apnea in children with laryngomalacia: diagnosis via sedated endoscopy and objective outcomes after supraglottoplasty. Otolaryngol Head Neck Surg 147(3):544–550. https://doi.org/10.1177/0194599812446903
Camacho M, Dunn B, Torre C, Sasaki J, Gonzales R, Liu SY, Chan DK, Certal V, Cable BB (2016) Supraglottoplasty for laryngomalacia with obstructive sleep apnea: a systematic review and meta-analysis. Laryngoscope 126(5):1246–1255. https://doi.org/10.1002/lary.25827
Bliss M, Yanamadala S, Koltai P (2018) Utility of concurrent direct laryngoscopy and bronchoscopy with drug induced sleep endoscopy in pediatric patients with obstructive sleep apnea. Int J Pediatr Otorhinolaryngol 110:34–36. https://doi.org/10.1016/j.ijporl.2018.04.009
Baldassari CM, Mitchell RB, Schubert C, Rudnick EF (2008) Pediatric obstructive sleep apnea and quality of life: a meta-analysis. Otolaryngol Head Neck Surg 138(3):265–273. https://doi.org/10.1016/j.otohns.2007.11.003
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kirkham, E., Ma, CC., Filipek, N. et al. Polysomnography outcomes of sleep endoscopy–directed intervention in surgically naïve children at risk for persistent obstructive sleep apnea. Sleep Breath 24, 1143–1150 (2020). https://doi.org/10.1007/s11325-019-02006-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-019-02006-y