Abstract
Unnatural amino acids (UAAs) are valuable building blocks in the manufacture of a wide range of pharmaceuticals. UAAs exhibit biological activity as free acids and they can be incorporated into linear or cyclic peptides with biological activity. However, the scope of biotechnological application of UAAs goes beyond this, as they can be used to investigate the structure and dynamics of proteins, to study protein interactions, or to modulate the activity of proteins in living cells. The means to expand nature’s repertoire of amino acids include chemical and biological routes. An UAA can be made through chemical modifications of natural amino acids, or related compounds. These modifications typically rely on utilisation of ligands and palladium catalysts. Employing biocatalysts in the synthesis of UAAs can also afford novel molecules with different physical and chemical properties. A number of transaminases for example have been identified and employed in the production of UAAs. This review will compare the chemical and biological routes for the synthesis of UAAs and provide an overview of their applications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Optically active amines, including amino acids, are estimated to make up approximately 40% of all chiral intermediates involved in the production of active pharmaceutical ingredients (Hönig et al. 2017). Amino acids, in addition to being the essential building blocks, are found in natural products such as antibiotics (Hancock and Chapple 1999), and due to the characteristic that they contain functional groups which could easily be transformed chemically they are attractive molecules for the synthesis of new drugs and materials (Blaskovich 2016; Clerici et al. 2016). In addition to the well-known 20 amino acids often called proteinogenic, ornithine, selenocysteine, hydroxylysine and hydroxyproline are frequently found in nature (Hönig et al. 2017). To date 800 natural amino acids are known in the literature, while thousands of synthetic amino acids have been reported (Xue et al. 2018).
Unnatural amino acids (UAAs) are so called because they are not found in natural polypeptide chains. Many UAAs are formed as secondary metabolites in bacteria, fungi, plants, or marine organisms, or they can be synthesised chemically (Blaskovich 2016). UAAs can be divided into two major groups: UAAs that are structurally similar to canonical amino acids and therefore frequently named as analogues, and surrogate UAAs that differ dramatically from canonical amino acids (Agostini et al. 2017). Whatever group they belong to, their distinct chemical and biological characteristics that stem from their unusual side groups and/or d-stereochemistry make them highly attractive as biologically active molecules or building blocks.
Application of UAAs
UAAs can function as single molecules, part of larger biomolecules, or when inserted in peptides (Table 1). Frequently, single amino acids such as glutamine and its analogues have bioactivity. Glutamine is the major excitatory neurotransmitter of the central nervous system (Albrecht et al. 2010). An UAA named LY404039, a selective glutamine agonist was shown to be efficient in the treatment of schizophrenia (Patil et al. 2007). Furthermore, some UAAs exhibit inhibitory effect on the activity of enzymes (Rando 1974), antimicrobial (Dittmer et al. 1948; Williams et al. 1988) or antiproliferative activity (Kindler et al. 2009).
UAAs are frequently used as scaffolds and building blocks for drug discovery (Bhonsle et al. 2013; Blaskovich 2016), or as reactants for important drug-like compounds such as benzodiazepines (Scott et al. 2009). The unnatural amino acid (2R,3S)-N-benzoyl-3-phenylisoserine is a good example of an important intermediate in the preparation of the anticancer drug called Paclitaxel, used to treat breast and lung cancer (Blaskovich 2016; Njogu et al. 2013; Weaver 2014).
Antimicrobial peptide (AMP) synthesis is one of the application areas for UAAs (Almahboub et al. 2018a). AMPs are biomolecules present in virtually every class of organisms as a host defence mechanism (Yeaman and Yount 2003). Increased interest in AMPs in recent years is due to their activity towards a wide range of bacteria, including some antibiotic-resistant strains, different fungi, viruses and parasites (Ageitos et al. 2017). AMPs are selective and efficient, (Fosgerau and Hoffmann 2015) and unlike conventional antibiotics, such peptides exhibit dual activity: disrupt targeted cells membranes and modulate the immune system while certain peptides have one or the other activity preferentially (Hancock and Sahl 2006). For example, Polymyxin B is a cyclic AMP that has activity against a broad range of Gram-negative bacteria and acts as a surfactant and permeabilises the bacterial cell membrane (Ageitos et al. 2017). Bacterial resistance to AMPs would require severe rearrangements of the membrane structure, which would significantly delay the emergence of resistance (Aoki and Ueda 2013; Chen et al. 2014). Thus, AMPs are promising candidates for the treatment of resistant infections (Shagaghi et al. 2018). Addition of UAAs into the structure of AMPs can increase their activity, hydrophobicity, and decrease their susceptibility to proteolytic activity.
Furthermore, UAAs can be applied in probing protein function (Stevenazzi et al. 2014). By labelling proteins with UAAs it is possible to study protein conformation by Förster resonance energy transfer (FRET), follow intracellular protein localisation or to determine protein structures in vivo by means of 3D X-ray crystallography or 19F NMR spectroscopy (Agostini et al. 2017). In addition, the incorporation of UAAs into enzymes, such as the substitution of methionine by norleucine could be used to protect proteins from methionine oxidation, a strategy valuable in the case of biocatalysts that are inactivated under oxidative conditions or require oxidizing substrates or cofactors (Anderhuber et al. 2016). Using expanded genetic code it is possible to exploit the cell transcription and translation machinery to incorporate UAAs at specific sites of a protein (Xiao and Schultz 2016). This methodology relies on using amber or a nonsense codon and orthogonal aminoacyl-tRNA synthetase/transfer RNA pair to introduce spectroscopic or photo-affinity probes, posttranslational modifications, site-specific modifications and novel properties into proteins (Xiao and Schultz 2016). In addition, the library of genetically encoded UAAs could be expanded by assigning some of the redundant codons to the non-proteinogenic AA (Iwane et al. 2016).
Conjugation of polymers to proteins can improve the stability, solubility, and biocompatibility of these therapeutic agents. Among different strategies for site-specific conjugation of polymers to proteins, the incorporation of UAAs via the expansion of the genetic code or solid-phase peptide synthesis was proven an efficient strategy to improve the pharmacodynamics properties of certain protein or peptide therapeutics (Wang and Wu 2018).
The exploitation of peptides in the development of smart nanomaterials is gaining increasing attention in the last few years. These advances are based on the property of amino acids to drive the self-assembly and the self-organisation at the molecular level (Clerici et al. 2016). Incorporating UAAs in the nanomaterials can expand the scope of the possible applications, ranging from biomaterials, biosensors to drug delivery systems. For example, the incorporation of homoalanine or norvaline in dipeptides created microporous organic materials with novel crystal structures and with the potential to be used as drug carriers as well as absorbents for green-house gasses (Yadav et al. 2015). Furthermore, UAAs such as photoreactive para-azidophenylalanine could be employed to design thermoresponsive gels with tuneable control over the sequestration and delivery of materials (Costa et al. 2018). Elastin-like polypeptides are the usual building blocks of crosslinked gel particles that exist as disordered soluble chains at low temperatures, while at temperatures above their concentration-dependent transition temperature they become globular structures (Meyer and Chilkoti 1999). The major challenge in producing these materials stems from complex processes relying on use of cytotoxic crosslinking agents. The use of UAAs can eliminate the need for external crosslinker, thereby facilitating the fabrication of thermoresponsive gels (Costa et al. 2018).
Antibody-drug conjugates (ADC) are holding great promise as anticancer therapeutics. Their activity is based on a specific interaction with cells presenting tumour associated antigens, followed by endocytosis of ADC, degradation by lysosome and release of the cytotoxic drug into the cytosol of a tumour cell (Kovtun and Goldmacher 2007; Okeley et al. 2010). Brentuximab vedotin, trade name Adcetris, is an anti-CD30 ADC with proven efficacy in patients with CD30+ malignancies and the first drug approved for Hodgkin’s lymphoma since 1977 (Ansell 2014; Axup et al. 2012). While there are many more ADCs in development, a frequent challenge associated with their design is that a drug is non-selectively conjugated to an antibody via cysteine or lysine residues, which causes formation of heterogenous products upon endocytosis (Hamblett et al. 2004), and therefore poses difficulties in optimising pharmacological and biological effects of ADC. Introducing UAAs was shown to allow more precise control of conjugation and stoichiometry. Site-specific incorporation of p-acetylphenylalanine (pAcPhe) into an antibody using an orthogonal amber suppressor tRNA/aminoacyl-tRNA synthetase pair, and subsequent conjugation of an auristatin derivative to the antibody via pAcPhe keto group was shown to yield an ADC with excellent pharmacokinetics and potent cytotoxicity to mammary fat pad tumours (Axup et al. 2012).
Synthesis of UAAs
Despite their tremendous potential, synthesis of UAAs remains a challenge. While there is a range of chemical synthetic routes available for the preparation of UAAs (Stevenazzi et al. 2014), they are complex and follow multiple reaction steps (Fig. 1), often offering a moderate yield. The particular bottleneck in chemical synthesis of UAAs is the lack of enantio- and stereoselectivity and the need to protect the reactive amine and carboxylic groups. On the other hand, the enzymatic asymmetric synthesis of chiral amino acids is very attractive due to the brief reaction steps, usually in aqueous media without the need for protective groups, and therefore low energy and sustainable means to synthesise these value-added chemicals (Fig. 1). The enzymes involved in the UAAs synthesis in nature may suffer from low activity or stability, or poor substrate range, but these issues may be relatively easy overcome using protein engineering. However, the main challenge associated with biocatalytic production of UAAs at scale is the dependence on cofactors (Fig. 1), which adds to the cost of the process and often requires complex and not so efficient approaches for the cofactor recycling. The examples of enzymatically catalysed synthesis of UAAs and the solutions to cofactor dependency will be addressed below.
Biocatalytic (light green) and chemical (light orange) synthetic routes for the production of unnatural amino acids (UAAs). Chemical synthetic routes for the preparation of UAAs are frequently complex and follow multiple reaction steps (represented by broken arrows), and lack enantio- and stereoselectivity. Biosynthetic routes usually yield enantiomerically pure UAAs, but are frequently challenged by cofactor dependency. AADH amino acid dehydrogenases, TA transaminase, NAD(P)H nicotinamide adenine dinucleotide (phosphate) reduced form, NAD(P)+ nicotinamide adenine dinucleotide (phosphate) oxidised form, PLP pyridoxal 5′-phosphate, PMP pyridoxamine 5′-phosphate
Biocatalytic routes
In biocatalysis enzymes are employed as catalysts that operate under mild reaction conditions with high enantioselectivity, conversion, and space-time yield on an industrial scale (Kelly et al. 2018; Xue et al. 2018). Furthermore, the enzymes are generally harmless and can often be recycled using immobilisation (Cao et al. 2003). Enzyme discovery and advances in protein engineering are major contributors to the expansion of enzyme catalysed synthesis of UAAs. The main biocatalytic routes for the synthesis of UAAs are kinetic resolution and asymmetric synthesis. In kinetic resolution the process is limited by the maximal theoretical yield of 50% while asymmetric synthesis offers maximal theoretical yield of 100% and more importantly synthesis of new to nature molecules (Rudat et al. 2012; Xue et al. 2018). However, as already mentioned, the main challenge associated with biocatalytic production of UAAs is the dependence on cofactors, which adds to the cost of the process and often requires complex and not so efficient approaches for the cofactor recycling. On the other hand, protein engineering, metabolic engineering and synthetic biology offer great possibilities for the advancement of biocatalysts, or design of new biocatalysts.
Amino acid dehydrogenases (AADH) are involved in reductive amination of ketoacids to yield amino acids. They are NAD(P)H dependent (Fig. 1), therefore the cofactor has to be recycled during the process. Stoichiometric supply of NAD(P)H is not economically sustainable since the price ranges from 4500 US $ per kg for NADH to 290,000 US $ per kg for NADPH (Faber 2011). However, efficient cofactor recycling systems were developed to aid large-scale application of AADHs and the current methods for the NAD(P)H regeneration are reviewed in in detail elsewhere (Wang et al. 2017).
Another issue associated with application of AADH is the reversibility of the reaction since these enzymes naturally maintain balance between amino and keto acids. Ammonia could be used as amino donor, which makes the process relatively cheap and thermodynamically favourable (Sharma et al. 2017). Relatively narrow substrate range of AADH could be overcome with the aid of in silico modelling and mutagenesis (Sharma et al. 2017). In addition, the protein engineering was proven successful in designing D-AADH, which are not ubiquitously found in nature for the synthesis of β-UAAs (Vedha-Peters et al. 2006).
However, regardless of the challenges related to AADH application there are numerous examples of synthesis of UAAs using these enzymes as biocatalysts. An efficient strategy to produce unnatural l-amino acids combines an AADH to convert hydrophobic α-keto acids into amino acids and a formate dehydrogenase for the cofactor regeneration (Bommarius et al. 1998; Krix et al. 1997). One of the UAAs produced using this strategy is l-tert-leucine, a precursor in the manufacture of protease inhibitors used in the treatment of hepatitis C, NS3/4 protease inhibitors, HIV protease inhibitor, as well as peptide deformylase inhibitors and matrix metalloprotease inhibitors (Xue et al. 2018). A leucine dehydrogenase was recently engineered using random mutagenesis paired with a high throughput spectrophotometric screening assay to allow the biosynthesis of l-tert-leucine with productivity of 1170 g/l/day (Zhu et al. 2016). Further advances in the production of UAA via AADH biocatalysis could be achieved by using synthetic biology to design a tuneable multienzyme system, such as a whole-cell biocatalyst reported by Jiang and Fang, in which leucine dehydrogenase and cofactor recycling enzyme, formate dehydrogenase are co-ordinately expressed to give good enantioselectivity and high conversion rate combined with high stability and lower cost of the process (Jiang and Fang 2016). The gap between the activities of leucine dehydrogenase and the formate dehydrogenase was bridged by using the ribosome binding sites (RBS) of different strength to finely tune the expression of the leucine dehydrogenase and placing the formate dehydrogenase under a very strong RBS since this enzyme is known to have low activity. This whole-cell catalyst was tested with a 100 mM substrate load, and it performed superior in comparison with purified enzymes, yielding a complete conversion of the substrate trimethylpyruvic acid into optically pure l-tert-leucine in an hour (Jiang and Fang 2016).
Metabolic engineering has proven useful in designing bacterial catalysts for the production of l-homoalanine or 2-aminobutyric acid, which is a precursor of antiepileptic drug levetiracetam and the antituberculosis drug ethambutol. Zhang and co-workers have expanded the metabolism of Escherichia coli to include l-homoalanine in the metabolite family by introducing threonine dehydratase TdcB or IlvA to convert threonine into 2-ketobutyrate (Zhang et al. 2010). Further optimisation of the process was achieved by incorporating an evolved glutamate dehydrogenase (GDH) for efficient reductive amination of 2-ketobutyrate with ammonia. GDH is naturally involved in reductive amination of 2-ketoglutarate, with 10-fold higher activity in the synthesis of glutamine compared to the glutamine degradation (Causey et al. 2003). Using saturation mutagenesis approach and deletion of native transaminases avtA and ilvE a variant of GDH was created with 8.2-fold increased catalytic efficiency with 2-ketobutyrate, and the created synthetic E. coli strain was ultimately able to convert glucose into l-homoalanine. Changing the host strain for a threonine overproducer (Debabov 2003), overexpressing the evolved GDH along with overexpression of IlvA from B. subtilis and deletion of rhtA23 threonine transporter the final synthetic strain had a productivity of 2.7 g/l/day of l-homoalanine from glucose, with 0.18 g/g yield. While this was only 26% of the theoretical maximum, this work is an example of successful metabolic engineering coupled with protein engineering to allow the production of an UAA.
Another example of an AADH used for the production of an α-amino acid with high e.e. is an engineered phenylalanine dehydrogenase (PheDH) from Thermoactinomyces sp. (Hanson et al. 2007) The modified PheDH containing two amino acid changes at the C‐terminus and a 12‐amino acid extension of the C‐terminus was used for the production of 2‐(3‐hydroxy‐1‐adamantyl)‐(2S)‐aminoethanoic acid, a key intermediate required for the synthesis of the anti-diabetic drug saxagliptin. A 100 kg scale process for the production of this UAA was developed to support the development of saxagliptin (Hanson et al. 2007).
Transaminases (TAs) can aminate a wide range of ketones and aldehydes. These ubiquitous enzymes are naturally involved in nitrogen metabolism. While TAs are also cofactor dependent, there is no need for external cofactor recycling which lowers the cost of the process and therefore makes TAs very attractive as biocatalysts (Kaulmann et al. 2007). Pyridoxal 5′-phosphate (PLP) acts as an intermediate amino group acceptor and an electron sink, and is recycled during the course of the reaction (Bugg 2004). Among TAs, a group of ω-TAs known to accept a range of substrates beyond only α-keto acids and α-amino donors are particularly interesting for biocatalysis and production of α- and β-UAAs (Mathew and Yun 2012). However, the use of TAs as biocatalysts also comes with a challenge of reaction equilibrium (Cassimjee et al. 2010). The by-product ketone formed after deamination of the donor competes with the acceptor for the amino group, shifting the reaction equilibrium towards the reactants. This is especially the case when alanine is used as a donor, where the reaction equilibrium favours the recycling of alanine and starting ketone rather than formation of the corresponding amine and pyruvate (Koszelewski et al. 2010b). In some cases high concentrations of the produced amine and by-product ketone can inhibit ω-TA activity (Höhne and Bornscheuer 2009; Shin and Kim 1997).
Numerous studies dealt with the issues of the ω-TA catalysed reactions (Cassimjee et al. 2010; Cho et al. 2003; Shin and Kim 2001; Truppo et al. 2009; Wang et al. 2013). In order to shift the reaction equilibrium towards product formation excess amino donor is often used (Koszelewski et al. 2010b). Moreover, using donors such as isopropylamine (IPA) provide a potential solution to the problem of reaction equilibrium and the inhibitory effect of the by-product ketone (Mathew and Yun 2012). IPA is transformed into acetone upon the amino group transfer, which is easy to remove due to its volatility (Park et al. 2013; Savile et al. 2010). Another solution to shift the equilibrium is using additional enzymes such as lactate dehydrogenase and alanine dehydrogenase that recycle the amino donor and deplete the potentially inhibitory by-product ketone (Höhne and Bornscheuer 2009; Koszelewski et al. 2008). Recently, an in-situ product crystallisation approach was developed to shift the reaction equilibrium towards product formation (Hulsewede et al. 2018).
l-tert-leucine, an UAA which was already discussed in the section dealing with AADHs, could be produced using a cascade reaction in an extractive biphasic system employing a branched-chain amino acid transaminase (BCTA) and an (S)-selective ω-transaminase (ω-TA) and achiral α-keto acid precursors (Park and Shin 2015). While employing a biphasic system in this case led the conversion of 20 mM substrate into the product, which was a nearly 2-fold improvement in l-tert-leucine synthesis compared to using a monophasic system, this approach compares poorly to the synthesis of this UAA using the whole cell biocatalyst reported by Jiang and Fang (Jiang and Fang 2016; Park and Shin 2015).
Norleucine (2-aminohexanoic acid) is a side product of branched chain amino acids metabolism and as methionine analogue is easily incorporated in proteins in place of methionine (Bogosian et al. 1989; Cirino et al. 2003). The bioproduction of norleucine was achieved in a metabolically engineered E. coli strain in which three acetolactate synthases from the metabolism of branched chain amino acids were deleted, and leuABCD operon was over expressed to allow accumulation of 2-ketobutyrate, a precursor in norvaline and norleucine synthesis (Anderhuber et al. 2016). The enzymes encoded by the leuABCD operon convert 2-ketobutyrate via 2-ketopentanoate into 2-ketohexanoate, which is ultimately aminated by a branched chain transaminase IlvE (Anderhuber et al. 2016). This route can provide synthesis of 5 g/l of norleucine. The engineered strain not only produced norleucine but was also demonstrated as an efficient system for the cost-effective labelling of proteins with norleucine at larger scales (Anderhuber et al. 2016).
An ω-TA form Vibrio fluvialis was successfully employed for the synthesis of L-homoalanine in a reaction of reductive amination (Shin and Kim 2009). This study demonstrated the potential to use an amine compound rather than an amino acid as amino donor and by doing so achieving more favourable reaction equilibrium and solving one of the issues of UAAs biosynthesis using TAs as biocatalysts. A severe reaction inhibition that occurred due to the formation of benzaldehyde as a side product was solved by using an extractive biphasic reaction system, finally reaching 96% conversion of 2-oxobutyric acid into L-homoalanine with 99% e.e. (Shin and Kim 2009).
Coupling of two TAs is another strategy that could be used to ensure the favourable reaction equilibrium. In a cascade reaction employing a branched chain amino acid TA from E. coli and an ω-TA from Paracoccus denitrificans l-hydroxyadamantglycine, l-tert-leucine and l-norvaline were produced (Park and Shin 2015). The P. denitrificans ω-TA was used to regenerate 2-oxobutyrate back into the donor, l-homoalanine.
Chromobacterium violaceum ω-TA accepts a wide range of aldehydes and ketones as amino acceptors (Kaulmann et al. 2007), and as such holds a great potential for the synthesis of UAAs. It was recently shown that C. violaceum TA can be used for the synthesis of 2-aminooctanoic or 2-aminocaprylic acid (Almahboub et al. 2018a). This α-UAA can be used as a reagent in the preparation of 1,5-disubstituted-2-aminoimidazoles, which have antibiotic activity towards a variety of bacterial strains (Harris et al. 2011), to design a vaccine delivery system without the need for additional adjuvant (Olive et al. 2003), or to modify antimicrobial peptides for the improved activity (Almahboub et al. 2018a). C. violaceum TA has been engineered via directed mutagenesis and rational design to further expand the range of substrates accepted by this enzyme (Almahboub et al. 2018b; Deszcz et al. 2015), or to change the enantioselectivity (Humble et al. 2012), and therefore increase the repertoire of produced UAAs.
Other enzymes involved in the synthesis of UAAs
While other enzymes could be used in the production of UAAs, such as lipases (Houng et al. 1996), and nitrilases (Martínková and Křen 2010), their application is mainly focused on kinetic resolution, thus providing a maximum theoretical yield of 50% (Koszelewski et al. 2010a). However, with the advancements in the fields of bioinformatics and protein engineering the toolbox of enzymes available for the synthesis of UAAs is growing and includes enzymes such as ammonia lyases and aminomutases (Parmeggiani et al. 2018). Highly enantioselective production of N-substituted aspartic acid derivatives was achieved using an engineered methylaspartate lyase by addition of structurally diverse amines to fumaric acid (Veetil et al. 2013).
Recently an α-ketoglutarate-dependent dioxygenase (Fe/αKG) was characterised and shown to hydroxylate the δ position of various aliphatic amino acids (Zwick and Renata 2018). Selective C–H functionalisation at distal positions is a highly challenging problem in organic synthesis, and this advance opens up possibilities in synthesis of derivatives of amino acids through C–H functionalisation.
Chemical synthetic routes
The development of efficient synthetic methods or new catalytic strategies to access an amino acid–base structural motif in an optically active form has long been an important topic in organic chemistry and catalysis. While numerous challenges are associated with this non-biosynthetic approach i.e. low selectivity and harsh conditions, advances in chemical synthesis yielded some novel and interesting UAAs. An advantage of chemical routes over biocatalysis approach is their broad applicability. The examples of overcoming specific challenges to achieve higher enantioselectivity and design processes that are more environment friendly are given below. However, it is worth noting that the large majority of discussed routes for the synthesis of UAAs are quite complex and require several reaction steps.
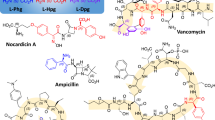
Substituted arylglycines are structural motifs found in many biologically active compounds and natural products, such as β-lactam antibiotics cephalexin, cefadroxi, amoxicillin and peptide antibiotics vancomycin and teicoplanin, P53-MDM2 inhibitor RO-5963, antiplatelet agents clopidogrel and vicagrel, and HCV NS3/4A protease inhibitor (Zhang et al. 2015). While synthetic approaches to racemic arylglycine derivatives have been developed, there is only a few reports of asymmetric synthesis of optically pure arylglycines (Zhang et al. 2015). Contrary to biocatalysts, that most frequently show enantioselectivity, processes employing organic and inorganic catalysts seldom exhibit enantioselectivity when prochiral substrates are used. Zhang and co-workers developed a stereoselective synthesis of optically active arylglycine derivatives using a Pd-catalysed alpha arylation of chiral nickel(II) glycinate complex with various aryl bromides with up to 90% yield and > 99% e.e.
d-Amino acids (d-AAs) can be found in microorganisms, plants, mammals and it has been shown that they have a biological activity either as single molecules or part of a peptide (Genchi 2017). In natural peptides one d-AA is usually positioned close to the N-terminus thus providing protease stability and affects the 3D structure of a peptide, leading to the formation of specific structures (Konno et al. 2007). Owing to the growing interest in d-AAs in biomedicine and pharmaceutical industry, chemical conversion of natural and tailor-made l-α-AAs into d-α-AAs via l-to-d-interconversion was developed (Nian et al. 2016). This approach involves the use of a wide range of free, unprotected l-AAs and cheap, readily available, recyclable proline derived ligands, making it cost-effective and widely applicable.
α,α-Disubstituted amino acids can also modify the structures of peptides or proteins. However, the asymmetric synthesis of these amino acids has not been established due to the challenge of the catalytic enantioselective synthesis (Tsubogo et al. 2010). By expanding chiral calcium chemistry and employing a chiral calcium catalyst system for the asymmetric 1,4-addition of azlactones to acrylates, optically active α-substituted glutamic acid derivatives with stereogenic carbon centres were synthesised (Tsubogo et al. 2010). However, this method requires the use of explosive tetrahydrofuran for the preparation of the catalyst, as well as toluene and low temperature of − 20°C for the synthesis of α-methyl glutamic acid making it non-economical and non-environmental friendly (Tsubogo et al. 2010).
A simple chemical route for the synthesis of racemic norleucine uses nucleophilic substitution reaction between toxic and corrosive 2-bromohexanoic acid and ammonia (Marvel and du Vigneaud 2003). To afford synthesis of optically pure norleucine a non-trivial asymmetric synthesis via alkylation of Ni(II) complexes of Schiff bases derived from (S)-o-[(N-benzylproplyl)amino]benzaldehyde or (S)-o-[(N-benzylproplyl)amino]benzophenone glycine or alanine followed by the separation of diastereoisomeric complexes on SiO2 has been developed (Belokon et al. 1988). However, this chemical route for the synthesis of norleucine is far more complex when compared to the biocatalytic route discussed earlier (Anderhuber et al. 2016).
Alkylation of glycine represents the most intuitive synthetic method for the production of UAAs (Stevenazzi et al. 2014). While advances in transition to more economically and environmentally sound chiral phase transfer catalysts have been made, asymmetric alkylation of glycine offers yields of between 60 and 90% with e.e. ranging between 18 and 99.7% (Wang et al. 2015). Furthermore, these synthetic routes usually rely on the use of toxic reagents such as methylsulfonylchloride, needed to achieve efficient reaction (Li et al. 2017; Wang et al. 2015). Li and co-workers developed a protocol with the view of a simplified alkylation workup and reduced use of hazardous reagents (Li et al. 2017). Starting with proline and using EDCI/DMAP as a coupling agent a chiral intermediate is synthesised, which is then alkylated in the presence of sodium tert butoxide. Using this procedure five unnatural amino acids, frequently used for peptide stapling for improved pharmacological activity of peptides, were synthesised with high enantioselectivity > 99% e.e. and yield ranging between 60 and 70% (Li et al. 2017).
Catalytic asymmetric amination which directly generates C-N bond is a simple method to derivatise α-amino acids. However, to achieve high enantioselectivity (97% e.e.) of this process usually very low temperatures of − 40°C are needed (Lan et al. 2009).
Derivatisation of natural amino acids serine, threonine, cysteine and tyrosine containing heavy atoms such as S, O, and N could provide a large number of possible UAAs. Although this strategy represents another intuitive and facile approach for introducing structural and chemical diversity, only a small number of UAAs of this class were successfully synthesised and applied (Stevenazzi et al. 2014).
Concluding remarks
Unnatural amino acids are already used in the market place and there is undoubtedly great potential for expansion of their use. The routes to their production will depend on the price and availability of starting materials but also on the capability of the biological and chemical catalysts. It appears that synthetic routes for the production of UAAs face more challenges than biocatalytic routes, primarily the lack of enantioselectivity. The biosynthesis of enatiomerically pure UAAs from simple and cheap precursors, such as glucose, could remedy the lack of availability and lower the price. To this end there are still significant challenges to broadening catalyst substrate range and achieving high productivity. Thus many possibilities for protein engineering and synthetic catalyst improvement abound. Last decade has seen many advances in the field of biocatalytic synthesis of chiral amines including UAAs, such as metabolic engineering and application of synthetic biology techniques. Synthetic biology in particular offers the potential for the engineering of whole-cell catalysts with finely tuned protein expression that avoid the need of cofactor regeneration.
References
Ageitos JM, Sanchez-Perez A, Calo-Mata P, Villa TG (2017) Antimicrobial peptides (AMPs): Ancient compounds that represent novel weapons in the fight against bacteria. Biochem Pharmacol 133:117–138. https://doi.org/10.1016/j.bcp.2016.09.018
Agostini F, Völler JS, Koksch B, Acevedo-Rocha Carlos G, Kubyshkin V, Budisa N (2017) Biocatalysis with unnatural amino acids: Enzymology meets xenobiology. Angewandte Chemie International Edition 56:9680–9703. https://doi.org/10.1002/anie.201610129
Albrecht J, Sidoryk-Wegrzynowicz M, Zielinska M, Aschner M (2010) Roles of glutamine in neurotransmission. Neuron Glia Biol 6:263–276. https://doi.org/10.1017/S1740925x11000093
Almahboub SA et al (2018a) Biosynthesis of 2-aminooctanoic acid and its use to terminally modify a lactoferricin B peptide derivative for improved antimicrobial activity. Appl Microbiol Biot 102:789–799. https://doi.org/10.1007/s00253-017-8655-0
Almahboub SA, Narancic T, Fayne D, O’Connor KE (2018b) Single point mutations reveal amino acid residues important for Chromobacterium violaceum transaminase activity in the production of unnatural amino acids. Sci Rep-UK. https://doi.org/10.1038/s41598-018-35688-7
Anderhuber N, Fladischer P, Gruber-Khadjawi M, Mairhofer J, Striedner G, Wiltschi B (2016) High-level biosynthesis of norleucine in E. coli for the economic labeling of proteins. J Biotechnol 235:100–111. https://doi.org/10.1016/j.jbiotec.2016.04.033
Ansell SM (2014) Brentuximab vedotin. Blood 124:3197–3200. https://doi.org/10.1182/blood-2014-06-537514
Aoki W, Ueda M (2013) Characterization of antimicrobial peptides toward the development of novel antibiotics. Pharmaceuticals (Basel) 6:1055–1081. https://doi.org/10.3390/ph6081055
Axup JY et al (2012) Synthesis of site-specific antibody-drug conjugates using unnatural amino acids. P Natl Acad Sci USA 109:16101–16106. https://doi.org/10.1073/pnas.1211023109
Belokon YN, Bakhmutov VI, Chernoglazova NI, Kochetkov KA, Vitt SV, Garbalinskaya NS, Belikov VM (1988) General method for the asymmetric-synthesis of alpha-amino acids V via alkylation of the chiral Nickel(II) Schiff base complexes of glycine and alanine. J Chem Soc Perk T 1:305–312. https://doi.org/10.1039/p19880000305
Bhonsle JB, Clark T, Bartolotti L, Hicks RP (2013) A brief overview of antimicrobial peptides containing unnatural amino acids and ligand-based approaches for peptide ligands. Curr Top Med Chem 13:3205–3224
Blaskovich MA (2016) Unusual amino acids in medicinal chemistry. J Med Chem 59:10807–10836. https://doi.org/10.1021/acs.jmedchem.6b00319
Bogosian G, Violand BN, Dorwardking EJ, Workman WE, Jung PE, Kane JF (1989) Biosynthesis and incorporation into protein of norleucine by Escherichia coli. J Biol Chem 264:531–539
Bommarius AS, Schwarm M, Drauz K (1998) Biocatalysis to amino acid-based chiral pharmaceuticals—examples and perspectives. Journal of Molecular Catalysis B: Enzymatic 5:1–11. https://doi.org/10.1016/S1381-1177(98)00009-5
Bugg TDH (2004) Introduction to enzyme and coenzyme chemistry, 3rd edn. Blackwell publishing Ltd, London
Cao LQ, van Langen L, Sheldon RA (2003) Immobilised enzymes: carrier-bound or carrier-free? Curr Opin Biotech 14:387–394. https://doi.org/10.1016/S0958-1669(03)00096-X
Cassimjee KE, Branneby C, Abedi V, Wells A, Berglund P (2010) Transaminations with isopropyl amine: equilibrium displacement with yeast alcohol dehydrogenase coupled to in situ cofactor regeneration. Chem Commun 46:5569–5571. https://doi.org/10.1039/c0cc00050g
Causey TB, Zhou S, Shanmugam KT, Ingram LO (2003) Engineering the metabolism of Escherichia coli W3110 for the conversion of sugar to redox-neutral and oxidized products: Homoacetate production. P Natl Acad Sci USA 100:825–832. https://doi.org/10.1073/pnas.0337684100
Chen C, Hu J, Zeng P, Chen Y, Xu H, Lu JR (2014) High cell selectivity and low-level antibacterial resistance of designed amphiphilic peptide G(IIKK)(3)I-NH(2). ACS Appl Mater Interfaces 6:16529–16536. https://doi.org/10.1021/am504973d
Cho B-K, Seo J-H, Kang T-W, Kim B-G (2003) Asymmetric synthesis of L-homophenylalanine by equilibrium-shift using recombinant aromatic L-amino acid transaminase. Biotechnology and bioengineering 83:226–234. https://doi.org/10.1002/bit.10661
Cirino PC, Tang Y, Takahashi K, Tirrell DA, Arnold FH (2003) Global incorporation of norleucine in place of methionine in cytochrome P450 BM-3 heme domain increases peroxygenase activity. Biotechnology and bioengineering 83:729–734. https://doi.org/10.1002/bit.10718
Clerici F, Erba E, Gelmi ML, Pellegrino S (2016) Non-standard amino acids and peptides: From self-assembly to nanomaterials. Tetrahedron Lett 57:5540–5550. https://doi.org/10.1016/j.tetlet.2016.11.022
Coleman MW (1983) Determination of the enantiomeric purity of oxfenicine by High-Performance Liquid-Chromatography. Chromatographia 17:23–26. https://doi.org/10.1007/Bf02265103
Costa SA et al (2018) Photo-crosslinkable unnatural amino acids enable facile synthesis of thermoresponsive nano- to microgels of intrinsically disordered polypeptides. Adv Mater. https://doi.org/10.1002/adma.201704878
Debabov VG (2003) The threonine story. In: Faurie R, Thommel J (eds) Microbial production of L-amino acids, vol 79. Springer, Berlin, Heidelberg
Deszcz D, Affaticati P, Ladkau N, Gegel A, Ward JM, Hailes HC, Dalby PA (2015) Single active-site mutants are sufficient to enhance serine:pyruvate alpha-transaminase activity in an omega-transaminase. The FEBS journal 282:2512–2526. https://doi.org/10.1111/febs.13293
Dittmer K, Goering HL, Goodman I, Cristol SJ (1948) The Inhibition of microbiological growth by allylglycine, methallylglycine and crotylglycine. J Am Chem Soc 70:2499–2501. https://doi.org/10.1021/ja01187a057
Faber K (2011) Biocatalytic applications. In: Biotransformations in organic chemistry. Springer, Berlin, Heidelberg
Farmer LJ et al (2015) Discovery of VX-509 (Decernotinib): A potent and selective Janus kinase 3 inhibitor for the treatment of autoimmune diseases. J Med Chem 58:7195–7216. https://doi.org/10.1021/acs.jmedchem.5b00301
Fosgerau K, Hoffmann T (2015) Peptide therapeutics: current status and future directions. Drug Discov Today 20:122–128. https://doi.org/10.1016/j.drudis.2014.10.003
Genchi G (2017) An overview on D-amino acids. Amino Acids 49:1521–1533. https://doi.org/10.1007/s00726-017-2459-5
Hamblett KJ et al (2004) Effects of drug loading on the antitumor activity of a monoclonal antibody drug conjugate. Clin Cancer Res 10:7063–7070. https://doi.org/10.1158/1078-0432.Ccr-04-0789
Hancock REW, Chapple DS (1999) Peptide antibiotics. Antimicrob Agents Ch 43:1317–1323
Hancock RE, Sahl HG (2006) Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat Biotechnol 24:1551–1557. https://doi.org/10.1038/nbt1267
Hanson RL et al (2007) Preparation of an amino acid intermediate for the dipeptidyl peptidase IV inhibitor, saxagliptin, using a modified phenylalanine dehydrogenase. Advanced Synthesis & Catalysis 349:1369–1378. https://doi.org/10.1002/adsc.200700013
Harris TL, Worthington RJ, Melander C (2011) A facile synthesis of 1,5-disubstituted-2-aminoimidazoles: antibiotic activity of a first generation library. Bioorganic & medicinal chemistry letters 21:4516–4519. https://doi.org/10.1016/j.bmcl.2011.05.123
Höhne M, Bornscheuer UT (2009) Biocatalytic routes to optically active amines. Chemcatchem 1:42–51. https://doi.org/10.1002/cctc.200900110
Hönig M, Sondermann P, Turner NJ, Carreira EM (2017) Enantioselective chemo- and biocatalysis: Partners in retrosynthesis. Angew Chem Int Edit 56:8942–8973. https://doi.org/10.1002/anie.201612462
Houng JY, Wu ML, Chen ST (1996) Kinetic resolution of amino acid esters catalyzed by lipases. Chirality 8:418–422. https://doi.org/10.1002/(Sici)1520-636x(1996)8:6%3c418:Aid-Chir2%3e3.0.Co;2-8
Hulsewede D, Tanzler M, Suss P, Mildner A, Menyes U, von Langermann J (2018) Development of an in situ-product crystallization (ISPC)-concept to shift the reaction equilibria of selected amine transaminase-catalyzed reactions. Eur J Org Chem:2130-2133 https://doi.org/10.1002/ejoc.201800323
Humble MS, Cassimjee KE, Abedi V, Federsel H-J, Berglund P (2012) Key amino acid residues for reversed or improved enantiospecificity of an omega-transaminase. Chemcatchem 4:1167–1172. https://doi.org/10.1002/cctc.201100487
Iwane Y, Hitomi A, Murakami H, Katoh T, Goto Y, Suga H (2016) Expanding the amino acid repertoire of ribosomal polypeptide synthesis via the artificial division of codon boxes. Nature Chemistry 8:317–325. https://doi.org/10.1038/Nchem.2446
Iwata R, Furumoto S, Pascali C, Bogni A, Ishiwata K (2003) Radiosynthesis ofO-[11C]methyl-L-tyrosine andO-[18F]Fluoromethyl-L-tyrosine as potential PET tracers for imaging amino acid transport. Journal of Labelled Compounds and Radiopharmaceuticals 46:555–566. https://doi.org/10.1002/jlcr.696
Jiang W, Fang BS (2016) Construction of a tunable multi-enzyme-coordinate expression system for biosynthesis of chiral drug intermediates. Sci Rep-UK 6:30426. https://doi.org/10.1038/srep30462
Kaulmann U, Smithies K, Smith MEB, Hailes HC, Ward JM (2007) Substrate spectrum of omega-transaminase from Chromobacterium violaceum DSM30191 and its potential for biocatalysis. Enzyme Microb Technol 41:628–637. https://doi.org/10.1016/j.enzmictec.2007.05.011
Kelly SA, Pohle S, Wharry S, Mix S, Allen CCR, Moody TS, Gilmore BF (2018) Application of omega-transaminases in the pharmaceutical industry. Chem Rev 118:349–367. https://doi.org/10.1021/acs.chemrev.7b00437
Kindler H, Burris H, Sandler A, Oliff I (2009) A phase II multicenter study of L-alanosine, a potent inhibitor of adenine biosynthesis, in patients with MTAP-deficient cancer. Invest New Drug 27:75–81. https://doi.org/10.1007/s10637-008-9160-1
Konno R, Brückner H, D’Aniello A, Fischer G, Fujii N, Homma H (2007) D-Amino acids: A new frontier in amino acids and protein research − Practical methods and protocols. Nova Science Publishers Inc, New York
Koszelewski D, Lavandera I, Clay D, Rozzell D, Kroutil W (2008) Asymmetric synthesis of optically pure pharmacologically relevant amines employing omega-transaminases. Adv Synt Catal 350:2761–2766. https://doi.org/10.1002/adsc.200800496
Koszelewski D, Goritzer M, Clay D, Seisser B, Kroutil W (2010a) Synthesis of optically active amines employing recombinant omega-transaminases in E. coli cells. Chemcatchem 2:73–77. https://doi.org/10.1002/cctc.200900220
Koszelewski D, Tauber K, Faber K, Kroutil W (2010b) Omega-transaminases for the synthesis of non-racemic alpha-chiral primary amines. Trends in biotechnology 28:324–332. https://doi.org/10.1016/j.tibtech.2010.03.003
Kovtun YV, Goldmacher VS (2007) Cell killing by antibody-drug conjugates. Cancer Lett 255:232–240. https://doi.org/10.1016/j.canlet.2007.04.01
Krix G, Bommarius AS, Drauz K, Kottenhahn M, Schwarm M, Kula MR (1997) Enzymatic reduction of α-keto acids leading to l-amino acids, d- or l-hydroxy acids. J Biotechnol 53:29–39. https://doi.org/10.1016/S0168-1656(96)01657-4
Lacoste AM, Darriet M, Neuzil E, Legoffic F (1988) Inhibition of alanine racemase by vinylglycine and its phosphonic analog - a H-1 nuclear magnetic-resonance spectroscopy study. Biochem Soc T 16:606–608. https://doi.org/10.1042/bst0160606
Lan Q, Wang XS, He RJ, Ding CH, Maruoka K (2009) Highly efficient asymmetric amination of beta-keto esters catalyzed by chiral quaternary ammonium bromides. Tetrahedron Lett 50:3280–3282. https://doi.org/10.1016/j.tetlet.2009.02.041
Li B, Zhang J, Xu YJ, Yang XX, Li L (2017) Improved synthesis of unnatural amino acids for peptide stapling. Tetrahedron Lett 58:2374–2377. https://doi.org/10.1016/j.tetlet.2017.05.007
Lloyd KG, Davidson L, Hornykiewicz O (1975) The neurochemistry of Parkinson’s disease: effect of L-dopa therapy. J Pharmacol Exp Ther 195:453–464
Martínková L, Křen V (2010) Biotransformations with nitrilases. Current opinion in chemical biology 14:130–137. https://doi.org/10.1016/j.cbpa.2009.11.018
Marvel CS, du Vigneaud V (2003) α-amino-n-caproic acid. https://doi.org/10.1002/0471264180.os004.02
Mathew S, Yun H (2012) Omega-transaminases for the production of optically pure amines and unnatural amino acids. ACS Catal 2:993–1001. https://doi.org/10.1021/cs300116n
Meyer DE, Chilkoti A (1999) Purification of recombinant proteins by fusion with thermally-responsive polypeptides. Nat Biotechnol 17:1112–1115
Nian Y et al (2016) Purely chemical approach for preparation of D-alpha-amino acids via (S)-to-(R)-interconversion of unprotected tailor-made alpha-amino acids. J Organ Chem 81:3501–3508. https://doi.org/10.1021/acs.joc.5b02707
Njogu PM, Gut J, Rosenthal PJ, Chibale K (2013) Design, synthesis, and antiplasmodial activity of hybrid compounds based on (2R,3S)-N-benzoyl-3-phenylisoserine. ACS Med Chem Lett 4:637–641. https://doi.org/10.1021/ml400164t
Okeley NM et al (2010) Intracellular activation of SGN-35, a potent anti-CD30 antibody-drug conjugate. Clin Cancer Res 16:888–897. https://doi.org/10.1158/1078-0432.Ccr-09-2069
Olive C, Batzloff M, Horvath A, Clair T, Yarwood P, Toth I, Good MF (2003) Potential of lipid core peptide technology as a novel self-adjuvanting vaccine delivery system for multiple different synthetic peptide immunogens. IAI 71:2373–2383. https://doi.org/10.1128/iai.71.5.2373-2383.2003
Park ES, Shin JS (2015) Biocatalytic cascade reactions for asymmetric synthesis of aliphatic amino acids in a biphasic reaction system. J Mol Catal B-Enzym 121:9–14. https://doi.org/10.1016/j.molcatb.2015.07.011
Park ES, Dong JY, Shin JS (2013) Omega-transaminase catalyzed asymmetric synthesis of unnatural amino acids using isopropylamine as an amino donor. Organ Biomol Chem 11:6929–6933. https://doi.org/10.1039/c3ob40495a
Parmeggiani F, Weise NJ, Ahmed ST, Turner NJ (2018) Synthetic and therapeutic applications of ammonia-lyases and aminomutases. Chem Rev 118:73–118. https://doi.org/10.1021/acs.chemrev.6b00824
Patil ST et al (2007) Activation of mGlu2/3 receptors as a new approach to treat schizophrenia: a randomized Phase 2 clinical trial. Nat Med 13:1102. https://doi.org/10.1038/nm1632
Rando RR (1974) Irreversible inhibition of aspartate aminotransferase by 2-amino-3-butenoic acid. Biochemistry 13:3859–3863. https://doi.org/10.1021/bi00716a006
Rudat J, Brucher BR, Syldatk C (2012) Transaminases for the synthesis of enantiopure beta-amino acids. AMB Express 2:11. https://doi.org/10.1186/2191-0855-2-11
Savile CK et al (2010) Biocatalytic asymmetric synthesis of chiral amines from ketones applied to sitagliptin manufacture. Science 329:305–309. https://doi.org/10.1126/science.1188934
Scott WL et al (2009) Distributed drug discovery, part 2: global rehearsal of alkylating agents for the synthesis of resin-bound unnatural amino acids and virtual D3 catalog construction. J Comb Chem 11:14–33
Shagaghi N, Palombo EA, Clayton AHA, Bhave M (2018) Antimicrobial peptides: biochemical determinants of activity and biophysical techniques of elucidating their functionality. World J Microb Biot 34:62. https://doi.org/10.1007/s11274-018-2444-5
Sharma M, Mangas-Sanchez J, Turner NJ, Grogan G (2017) NAD(P)H-dependent dehydrogenases for the asymmetric reductive amination of ketones: structure, mechanism, evolution and application. Adv Synth Catal 359:2011–2025. https://doi.org/10.1002/adsc.201700356
Shin JS, Kim BG (1997) Kinetic resolution of alpha-methylbenzylamine with omega-transaminase screened from soil microorganisms: application of a biphasic system to overcame product inhibition. Biotechnol Bioeng 55:348–358. https://doi.org/10.1002/(Sici)1097-0290(19970720)55:2%3c348:Aid-Bit12%3e3.0.Co;2-D
Shin J-S, Kim B-G (2001) Comparison of the omega-transaminases from different microorganisms and application to production of chiral amines. Biosci Biotechnol Biochem 65:1782–1788
Shin JS, Kim BG (2009) Transaminase-catalyzed asymmetric synthesis of L-2-aminobutyric acid from achiral reactants. Biotechnol Lett 31:1595–1599. https://doi.org/10.1007/s10529-009-0057-7
Soth M et al (2012) 3-Amido pyrrolopyrazine JAK kinase inhibitors: development of a JAK3 vs JAK1 selective inhibitor and evaluation in cellular and in vivo models. J Med Chem 56:345–356. https://doi.org/10.1021/jm301646k
Stevenazzi A, Marchini M, Sandrone G, Vergani B, Lattanzio M (2014) Amino acidic scaffolds bearing unnatural side chains: an old idea generates new and versatile tools for the life sciences. Bioorg Med Chem Lett 24:5349–5356. https://doi.org/10.1016/j.bmcl.2014.10.016
Truppo MD, Turner NJ, Rozzell JD (2009) Efficient kinetic resolution of racemic amines using a transaminase in combination with an amino acid oxidase. Chem Commun 16:2127–2129. https://doi.org/10.1039/b902995h
Tsubogo T, Kano Y, Ikemoto K, Yamashita Y, Kobayashi S (2010) Synthesis of optically active, unnatural alpha-substituted glutamic acid derivatives by a chiral calcium-catalyzed 1,4-addition reaction. Tetrahedron Asymmetr 21:1221–1225. https://doi.org/10.1016/j.tetasy.2010.03.004
Vedha-Peters K, Gunawardana M, Rozzell JD, Novick SJ (2006) Creation of a broad-range and highly stereoselective D-amino acid dehydrogenase for the one-step synthesis of D-amino acids. J Am Chem Soc 128:10923–10929. https://doi.org/10.1021/ja0603960
Veetil VP, Raj H, de Villiers M, Tepper PG, Dekker FJ, Quax WJ, Poelarends GJ (2013) Enantioselective synthesis of N-substituted aspartic acids using an engineered variant of methylaspartate ammonia lyase. Chemcatchem 5:1325–1327. https://doi.org/10.1002/cctc.201200906
Velkov T, Roberts KD, Nation RL, Thompson PE, Li J (2013) Pharmacology of polymyxins: new insights into an ‘old’ class of antibiotics. Future Microbiol 8:711–724. https://doi.org/10.2217/Fmb.13.39
Wang YJ, Wu C (2018) Site-specific conjugation of polymers to proteins. Biomacromolecules 19:1804–1825. https://doi.org/10.1021/acs.biomac.8b00248
Wang B, Land H, Berglund P (2013) An efficient single-enzymatic cascade for asymmetric synthesis of chiral amines catalyzed by omega transaminase. Chem Commun 49:161–163. https://doi.org/10.1039/c2cc37232k
Wang Z, Huang D, Xu P, Dong X, Wang X, Dai Z (2015) The asymmetric alkylation reaction of glycine derivatives catalyzed by the novel chiral phase transfer catalysts. Tetrahedron Lett 56:1067–1071. https://doi.org/10.1016/j.tetlet.2015.01.063
Wang X, Saba T, Yiu HHP, Howe RF, Anderson JA, Shi J (2017) Cofactor NAD(P)H regeneration inspired by heterogeneous pathways. Chem 2:621–654. https://doi.org/10.1016/j.chempr.2017.04.009
Weaver BA (2014) How Taxol/paclitaxel kills cancer cells. Mol Biol Cell 25:2677–2681. https://doi.org/10.1091/mbc.E14-04-0916
Williams RM, Sinclair PJ, Zhai D, Chen D (1988) Practical asymmetric syntheses of alpha-amino acids through carbon-carbon bond constructions on electrophilic glycine templates. J Am Chem Soc 110:1547–1557. https://doi.org/10.1021/ja00213a031
Xiao H, Schultz PG (2016) At the interface of chemical and biological synthesis: An expanded genetic code. Csh Perspect Biol 8:023945. https://doi.org/10.1101/cshperspect.a023945
Xue YP, Cao CH, Zheng YG (2018) Enzymatic asymmetric synthesis of chiral amino acids. Chem Soc Rev 47:1516–1561. https://doi.org/10.1039/c7cs00253j
Yadav VN, Comotti A, Sozzani P, Bracco S, Bonge-Hansen T, Hennum M, Gorbitz CH (2015) Microporous molecular materials from dipeptides containing non-proteinogenic residues. Angew Chem Int Edit 54:15684–15688. https://doi.org/10.1002/anie.201507321
Yeaman MR, Yount NY (2003) Mechanisms of antimicrobial peptide action and resistance. Pharmacol Rev 55:27–55. https://doi.org/10.1124/pr.55.1.2
Zhang KC, Li H, Cho KM, Liao JC (2010) Expanding metabolism for total biosynthesis of the nonnatural amino acid L-homoalanine. Proc Natl Acad Sci USA 107:6234–6239. https://doi.org/10.1073/pnas.0912903107
Zhang F, Sun HZ, Song Z, Zhou SX, Wen XA, Xu QL, Sun HB (2015) Stereoselective synthesis of arylglycine derivatives via palladium-catalyzed alpha-arylation of a chiral nickel(II) glycinate. J Organ Chem 80:4459–4464. https://doi.org/10.1021/acs.joc.5b00314
Zhu L, Wu Z, Jin J-M, Tang S-Y (2016) Directed evolution of leucine dehydrogenase for improved efficiency of l-tert-leucine synthesis. Appl Microbiol Biotechnol 100:5805–5813. https://doi.org/10.1007/s00253-016-7371-5
Zwick CR, Renata H (2018) Remote C-H hydroxylation by an alpha-ketoglutarate-dependent dioxygenase enables efficient chemoenzymatic synthesis of manzacidin C and proline analogs. J Am Chem Soc 140:1165–1169. https://doi.org/10.1021/jacs.7b12918
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Narancic, T., Almahboub, S.A. & O’Connor, K.E. Unnatural amino acids: production and biotechnological potential. World J Microbiol Biotechnol 35, 67 (2019). https://doi.org/10.1007/s11274-019-2642-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-019-2642-9