Abstract
Temporarily flooded depressions in arable fields support populations of specialised plant species that are affected by flooding and agricultural management. Depending on the degree of flooding, different proportions of wetland and arable species contribute to the seed bank. This is reflected by high inter-annual variations in plant communities with a high conservation value. Due to ongoing agricultural intensification, the biodiversity of temporarily flooded depressions has declined, and several plant species have become regionally extinct. Because seed banks harbour persistent seeds over long periods, they play a crucial role in the conservation and restoration of temporary wetland vegetation. This study focuses on the effects of different flooding regimes on plant species emerging from seed banks of temporarily flooded depressions in arable fields in northeast Germany. We cultivated soil samples from upper and lower wetland zones under short, intermediate and long-term flooding (5, 15 and 40 cm above soil surface) in a common garden experiment over 2 years. We observed significant changes in species composition depending on the flooding duration. Species richness declined and evenness increased with increasing flooding duration. Upper and lower zones showed similar species richness and evenness, but species compositions differed. Red List species emerged from all treatments although the species differed, indicating that all communities emerging under different flooding regimes have a high conservation value. Seed banks under fluctuating site conditions can constitute a series of alternating plant communities. This could be used to develop management strategies that benefit different communities with high conservation values.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Seed banks have great potential for the conservation and restoration of degraded habitats because they may contain seeds of regionally extinct species (Bernhardt et al. 2004; Poschlod 1993). Particularly, in ecosystems with environmental fluctuations, many species only emerge periodically and they can survive these unfavourable periods in the seed bank (Poschlod et al. 1999). The dynamics of fluctuating systems favour contrasting plant communities depending on the site conditions, and the seed bank will reflect these different communities. Adaptive management may facilitate the establishment of a series of alternating communities, thus, maximising plant diversity over time. Many conservation studies have assessed the soil seed bank and its importance for the conservation and restoration of wetlands in agricultural landscapes (Bissels et al. 2005; Casanova 2012). In the prairie pothole region and in coastal plain depressions in the United States, for example, several studies have focused on different approaches to restore the natural vegetation after long-term agricultural use of these wetlands (De Steven et al. 2006; Galatowitsch and van der Valk 1996). Other studies focused on restoring wet meadows after long periods of intensive (arable) land use, using the soil seed bank (Bekker et al. 2000; Hölzel and Otte 2004; van Dijk et al. 2007). However, studies assessing the soil seed bank of temporarily flooded depressions in arable fields in temperate climates have so far been rare (Bissels et al. 2005).
Because of the intensification of agricultural management, many regions that formerly harboured a great variety of species specifically adapted to traditional farming practices have experienced a considerable decline in arable plant species diversity in the past decades (Burrichter et al. 1993; Meyer et al. 2013; van Zanten et al. 2014). One example is wet field depressions that have been ameliorated by more efficient drainage. Many arable and amphibious plant species are adapted to disturbance and variable site conditions through the development of a long-term persistent seed bank (Brock 2011; Thompson et al. 1998). In temporarily flooded fields, both regular arable weeds and wetland plants are found in the seed bank (Albrecht 1999; Bissels et al. 2005). Thus, the soil seed bank of such sites can harbour complementary plant communities that markedly exceed the species diversity in the apparent vegetation.
In large parts of Europe, deforestation during the medieval age promoted the development of wet field depressions due to rising ground water. This led to the flooding of local depressions, where colluvial clay deposits inhibited water infiltration of Klafs et al. (1973). Such man-made wetlands also developed in abandoned marl or clay pits (Day et al. 1982). As habitat quality has deteriorated due to widespread drainage, the biodiversity of these habitats has markedly declined and many plant species are threatened by the loss of flooding and water level fluctuations (Zacharias and Zamparas 2010). Moreover, climate change is expected to have a negative impact on temporarily flooded depressions as the weather is predicted to become more extreme with mild wet winters and hot dry summers (IPCC 2007). This may shorten the flooding duration, which is a disadvantage for plant species that require flooding for germination (e.g. Kwon et al. 2013). Conversely, precipitation is predicted to become more uneven and more intense (Madsen et al. 2009), which could benefit species assemblages dependent on temporarily flooded depressions (Kappelle et al. 1999). Thus, new concepts are needed to preserve the vegetation of these habitats.
The actual vegetation that develops in temporarily flooded depressions in arable fields depends on agricultural management, soil and climatic conditions (Pinke et al. 2014). Most importantly, the species diversity of these depressions is affected by the flooding regime (Casanova and Brock 2000), controlling germination via soil temperature and light and oxygen concentrations (e.g. Matsuo et al. 1984). At the initial stage of secondary succession, competition is low, and most species best establish under terrestrial conditions, as long as the soil moisture is sufficient. Only submersed macrophytes and those with floating leaves are unable to survive at terrestrial sites, and most of them cannot establish without inundation (Baskin and Baskin 2014). During flooding, the depth of a waterbody can affect water plant germination through daily changes in light availability and temperature (Pons and Schröder 1986). High water levels and a long flooding duration promote the establishment of water plant species, whereas a short flooding duration is more advantageous for terrestrial species. During the drawdown, amphibious and terrestrial plant species can emerge under damp but not flooded conditions. An intermediate flooding duration with a slow drawdown or slowly rising water levels throughout a vegetation period can stimulate a sequence of water plant, amphibious plants or terrestrial species. Thus, the highest species diversity is expected under intermediate flooding. Moreover, as temporary wetlands harbour many rare and endangered plant species (Casanova and Brock 1990; Lentz and Dunson 1999; Lukacs et al. 2013; Mann and Raju 2002; Pukacz et al. 2009), the number of Red List species will increase concurrently with flooding. Species evenness is often negatively correlated to species richness (Weiher and Keddy 1999). In aquatic ecosystems, the limited availability of light and chemical growth factors can significantly limit competition by reducing the productivity of dominant species (Li et al. 2015; Slivitzky 2002). In contrast, this may also reduce species richness by excluding species without appropriate adaptations. However, as dominance is reduced and most of the remaining species find conditions suitable in which to establish, we expect that species are more likely to be equally abundant and that evenness will increase based on the intensity of flooding. In the lower wetland zone, where flooding occurs more frequently over a longer duration than in the upper wetland zone, seeds can accumulate in the soil because the germination of most species is inhibited under anaerobic conditions (Saatkamp et al. 2014). Moreover, as flooding gradients lead to differences in species composition in the apparent vegetation (Keddy and Reznicek 1986), a similar pattern can be expected in the composition of the soil seed bank. Thus, we expect the soil seed bank to reflect the flooding history of the different wetland zones.
In this study, we investigate how soil seed banks in upper and lower wetland zones of temporarily flooded depressions reflect the past flooding regime, and how flood management could be used to create a series of different plant communities. We analysed the species diversity of plant communities emerging from soil samples in experiments with different flooding durations and water levels. Specifically, we hypothesise that (i) plant communities emerging from the seed bank are different along a flooding gradient, both in the upper and lower zones of temporarily flooded depressions, (ii) seedling densities emerging from the seed bank are highest under intermediate flooding, particularly in samples from the lower zone, (iii) species richness of the seed bank will decrease with flooding in samples from the upper zone, (iv) species evenness is highest at high flooding in both zones and (v) the number of rare species increases with flooding.
Methods
Study sites and seed bank sampling
Soil samples were collected at Parstein, northeast Germany (northeast of Berlin, 53°0′52N, 13°59′E, 55 m above sea level). This region is characterised by a temperate, humid climate with an average annual temperature of 9 °C and 500 mm precipitation (1981–2010; DWD 2014); one-third of the precipitation occurs in the summer. The glacial moraine landscape has fertile luvisols with an almost neutral soil reaction. Glacial processes and former land use has resulted in numerous wet field depressions with great differences in both the frequency and duration of flooding (Klafs et al. 1973). As natural and man-made depressions have many common ecological characteristics, we refer to both of them as ‘temporarily flooded depressions’.
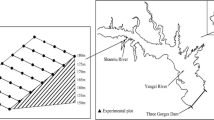
All samples were taken from seven temporarily flooded field depressions within a 60-ha area. They were selected on the presence of Elatine alsinastrum, a rare and strongly endangered amphibious species (Landesumweltamt 2006; Lansdown 2014; Ludwig and Schnittler 1996). Due to the restricted distribution of this species and to facilitate field access and soil collection, samples were taken on one conventionally managed farm. According to information from the farmer, the arable fields are cultivated in a four-course crop rotation (winter barley, winter rape, winter wheat and maize) using a field cultivator and disc harrow (plough level 0.2 m) for soil tillage. Mineral NPK fertilisers and herbicides are applied depending on the nutrient needs of the individual crops and weed abundance. All temporarily flooded parts are cultivated if the water level and soil moisture content allow the use of machinery. In May 2012, the sizes of the waterbodies ranged from 0.3 to 4.2 ha with geographical distances between them of 0.2–1.0 km (mean 0.5 km; see Fig. 1).
Position of the seven temporarily flooded depressions (shaded areas) within two arable fields. Aerial picture from Bing (2012)
As the vegetation at the edges of wetlands follows a clear zonation according to soil moisture and flooding (Keddy and Ellis 1985), we assumed that such a zonation also might exist in the seed bank composition. Therefore, we differentiated between the upper zone that was flooded until late May, and the lower zone that was flooded until late July.
In August 2012, we took soil samples from both of these zones. At depressions with permanently flooded core areas (marked by a shoreline with perennial plants like Salix spp. or Phragmites australis), the permanently flooded parts experienced a different disturbance regime than the arable fields. Therefore, we also presumed that there would be differences in the species composition of the soil seed bank and excluded these parts from sampling. At each depression, 30 soil cores were randomly collected from both the upper and lower zones. Soil corers had a diameter of 3.3 cm, and samples were taken down to 20 cm, corresponding with the depth of ploughing practised on the farm. According to Gruber et al. (2010), regular tillage leads to an even seed distribution within this plough layer. The samples from each zone were combined and mixed thoroughly to remove any site-specific seed bank variation. This resulted in two composite samples, one from the upper and one from the lower zone, representing a contrasting flooding history.
Flooding experiment
To test the effects of different water levels and flooding duration combinations on the emergence of plant communities, the composite samples were transferred to a set of artificial outdoor water basins and split into different treatments. As the depressions in the study area were usually flooded in the winter, we also maintained the samples in flooded basins at that time. We defined the start of the vegetation period as the first week with mean daily temperatures above 5 °C, which was early April in 2013 and early March in 2014. Usually, the upper zone of flooded field depressions dries up within approximately 2 weeks after the start of the vegetation period; in the lower zone, this takes approximately 8 weeks (Altenfelder, pers. obs.). Therefore, we selected flooding durations of 2 and 8 weeks. To examine the full gradient of possible flooding durations, we included a permanently flooded treatment (>16 weeks flooded) and an unflooded control (water level 15 cm below the soil surface). To test for the effects of different water levels, we established water levels of 5, 15 and 40 cm above the soil surface. Moreover, each water level was combined with the three flooding treatments of 2, 8 and >16 weeks. All combinations of flooding duration and water level resulted in 10 treatments that can be interpreted as gradients of flooding intensity.
For each treatment combination we used six replicates, resulting in a total of 118 plastic pots (18 × 18 × 18 cm3). Approximately, three quarters of each pot were filled with sterilised soil and covered with 2 cm of soil from the upper or lower zone. To evaluate seed contamination, we additionally set up controls with autoclaved soil (four replicates per flooding treatment and six replicates for the unflooded control). Because Epilobium spp., Juncus effucus, Lythrum salicaria, Sonchus arvensis and Taraxacum spp. emerged from these pots we considered them as seed contaminants and excluded them from further analyses. Nomenclature follows Wisskirchen and Haeupler (1998) for vascular plants and Blümel and Raabe (2004) for charophytes.
The experiment started in August 2012 and ran for 2 years, finishing in August 2014. All emerging seedlings were identified, counted and removed bi-weekly over the whole study period. Soil was mixed when seedling emergence declined. Plants that were difficult to identify were grown until they could be identified to species. Within the Alisma plantago-aquatica group, three different taxa occurred; however, as most individuals did not flower, it was impossible to determine their abundance. The Red List of the Federal State of Brandenburg (Kabus and Mauersberger 2011; Landesumweltamt 2006) was used to assess the conservation values of plants emerging from the soil samples.
Data analyses
Seed numbers were calculated by extrapolating the numbers of seeds detected in the surface area of the soil cores to 1 m2. Two pots of the upper zone (2 weeks at 40 cm; permanently flooded 40 cm) were lost during the experiment and had to be removed from the analyses.
Response variables were species richness, evenness of seedling communities and number of Red List species as a measure of the conservation value. These variables could not be transformed to achieve a normal distribution. Therefore, we performed Kruskal–Wallis tests using the package agricolae (de Mendiburu 2014) in R (R Core Team 2013) to determine the effects of water level and flooding duration on soil samples. As there was no significant difference in seedling density, species richness, evenness and conservation value between the two wetland zones (Fig. 2), these zones were no longer distinguished in further analyses of diversity. To test for differences between the treatment combinations, group means were compared at the 95 % confidence interval level with Bonferroni corrections. Interaction effects were tested using dummy variables. As a measure for evenness we calculated Pielou’s J (J = H′/ln s), with H′ = −∑ p i × ln p i , where p i is the proportional abundance of species i and s is the number of species. To calculate the number of Red List species, we included those species with a Red List status of ‘near-threatened’, ‘vulnerable’, ‘endangered’ and ‘critically endangered’.
To identify major gradients in species composition, a detrended correspondence analysis (DCA) was used (Hill and Gauch 1980). We preferred this method to non-metric multidimensional scaling (NMDS). even though both operations led to almost identical results (results of NMDS are not shown), because DCA fits species response curves to standard deviations and provides more detailed information on beta diversity along the first ordination axis. The effect of high seedling densities was reduced by square root transformation and subsequent Wisconsin double standardisation. To assess relationships between seedling composition and different water regimes, linear trends of water level, flooding duration and median Ellenberg indicator values for moisture (Ellenberg et al. 2001) were fitted on the ordination results. Both ordination and fitting of linear models were carried out using the R package vegan (Oksanen et al. 2013).
Results
Effects on diversity
During the 2-year sampling period, a total of 19,966 seedlings emerged from all samples. Seedling density was highest under no flooding (74,282 ± 15,015 seedlings m−2, mean ± SD) or short-term flooding (71,427 ± 15,015 seedlings m−2). Density decreased with increasing duration of inundation (Kruskal–Wallis test: H = 105.0, p < 0.0001; Fig. 3). Under intermediate- and long-term flooding, there was an effect of water level with higher seedling densities at water tables of 5 cm compared with higher water levels within the same flooding duration treatment.
A total of 65 species emerged from the soil samples, nine of them are considered to be endangered according to the Red List of the Federal State of Brandenburg (see Online Resource 1). In total, 54 species emerged from soil samples from the upper zone and 60 from the lower zone, with 49 species in common. The most abundant species were Alisma plantago-aquatica agg. and Peplis portula, both of which emerged from 93 % of all pots.
Species richness was highest under no and short-term flooding (22 ± 2 species per pot with no flooding and 22 ± 3 with short-term flooding). Flooding duration significantly reduced species richness (Kruskal–Wallis test: H = 88.1, p < 0.0001); water level had no effect on species richness (Fig. 4). Evenness significantly increased with increasing flooding duration (Kruskal–Wallis test: H = 86.7, p < 0.0001; Fig. 5), while water level had no effect on evenness.
The highest number of Red List species was observed under no or short-term flooding, as well as intermediate flooding with shallow water levels (no flooding: 5.1 ± 0.5; short-term flooding: 5.1 ± 0.6; intermediate with a water level of 5 cm: 5.2 ± 0.6 Red List species per sample). Increasing flooding duration significantly decreased the number of Red List species (Kruskal–Wallis test: H = 67.9, p < 0.0001; Fig. 6). Water level changed the number of Red List species under intermediate- and long-term flooding between 5 and 40 cm water levels. In the unflooded and temporarily flooded treatments almost all emerging Red List species were amphibious plants, including Elatine alsinastrum, Limosella aquatica and Myosurus minimus. Under permanent flooding these species also emerged but more rarely; instead, the threatened charophyte Tolypella prolifera exclusively emerged under intermediate or permanent flooding.
Variation in species composition along a flooding gradient
The gradient length of 3.7 along the first DCA ordination axis indicated an almost complete turnover in plant species composition emerged from the soil samples (Fig. 7). This change in species composition was mainly determined by differences between temporary and permanent flooding; few differences were observed among the unflooded and short- to intermediate-term flooded treatments. The gradient was closely correlated with flooding duration and indicator values for moisture (p < 0.001; Table 1). Along the second ordination axis the species composition changed according to the wetland zone (p < 0.001; Table 1). This gradient indicates that species composition among both wetland zones increasingly differed with flooding duration. The experimental water levels were not significantly correlated with the species composition.
Detrended correspondence analysis (DCA) of the species composition in soil samples responding to experimental flooding from the upper (filled symbols) and lower zones (transparent symbols) of temporarily flooded depressions in arable fields. Arrows indicate the correlation of flooding duration and the median Ellenberg indicator values for moisture (EIV moisture) to the ordination results from the linear models. The length of the arrows indicates the strength of the correlation. Different flooding durations are represented by different symbols; species without Red List status are represented by ‘plus’. Full names of Red List species Butomus umbellatus, Elatine alsinastrum, Galium palustre agg., Juncus tenageia, Limosella aquatica, Myosurus minimus, Peplis portula, Potentilla supina and Tolypella prolifera
Discussion
Effects on seedling abundances, diversity and conservation value
Contrary to our hypotheses, the seedling abundance and species richness were highest with no or short-term experimental flooding. It made no difference whether samples came from the upper or lower wetland zone. This is in agreement with other studies, where the highest species richness was observed in unflooded samples (Aponte et al. 2010; Casanova and Brock 2000). The cause for these high numbers of seedlings and species establishing from the soil seed bank is because both terrestrial and amphibious plant species find suitable germination conditions when flooding is short or absent, as long as competition is low. Furthermore, many amphibious plant species emerge in high numbers as soon as environmental conditions favour germination (Bliss and Zedler 1998; von Lampe 1996). Under flooded conditions, where temperatures are constant during day and night, most amphibious plants fail to germinate as they need diurnally fluctuating temperatures (Baskin and Baskin 2014; Poschlod et al. 1999). Furthermore, only few species are known to optimally germinate when they are submerged (Poschlod et al. 1999), and seedling densities are usually higher under unflooded conditions (Collins et al. 2013). Obligate water plants, which depend on continuous flooding, had a minor emergence in all treatments and, therefore, they did not significantly contribute to the species richness in the temporary flooded treatments.
In our study, evenness increased with flooding duration. This observation is consistent with the findings of Weiher and Keddy (1999) in riverine wetlands, where species evenness also increased with decreasing species richness and abundance. These results may be caused by increasing stress associated with the flooding duration. Thus, species unable to tolerate flooding are inhibited, and species richness decreases. The remaining stress-tolerant species are more likely to emerge with similar abundances leading to an increased evenness. As stress is usually associated with decreasing plant biomass and size (Grime 1979), competitive interactions could favour small-sized species contributing to plant communities with low species richness but high evenness, where all species present were equally uncommon (Bock et al. 2007). In contrast, there are also studies that found inconsistent patterns for species richness and evenness (Soininen et al. 2012; Stirling and Wilsey 2001). They concluded that both measures of biodiversity depend on different and independent ecological processes. Thus, Ma (2005) related these patterns to a deviating response to soil conditions, i.e. C:N ratio and phosphorus concentration. However, in our study, such effects can be excluded because we thoroughly mixed the soil samples.
We used the number of Red List species as an indicator of the conservation value of the plant communities emerging from the soil samples. Other studies have shown that the conservation value of temporarily flooded depressions can be very high (Bell et al. 2012; Casanova and Brock 1990; Lukacs et al. 2013). Indeed, we also found endangered species in most of the soil samples. This indicates that the soil seed banks of temporary wetlands are an important reservoir for rare species, which can be activated for conservation purposes or used during the restoration of degraded sites. Faist et al. (2013) reported that rare species in the aboveground vegetation were more abundant in the seed bank in vernal pools in California, showing the potential for the future establishment of rare species from the seed bank.
As in other studies on the soil seed bank of wetlands (e.g. Aponte et al. 2010), a few species account for the main share of the seed bank, which is a common characteristic for this habitat type (Harper 1987). Some rare plants that are reported for the study area, i.e. Schoenoplectus supinus and Chara baueri, could not be found (Hoffmann 1996; Raabe 2009) although this might be due to their rarity and the limited amount of soil analysed. The differences in the number of Red List species between the different experimental flooding regimes were marginal, but all species groups from terrestrial, amphibious and water plants were included. This result indicates the favourable conditions to promote endangered species from different plant communities along a flooding gradient.
There were no differences in seedling abundance, diversity or conservation value between the upper and lower wetland zones. This shows that even in the areas that are less frequently flooded and farmed when dry enough, the diversity and conservation value are comparable with the more frequently and longer flooded lower wetland zone. The year in which our soil samples were collected was at the end of several years of high precipitation, which resulted in regular flooding of the upper wetland zone and a lack of farming, except for soil tillage. Thus, the seed bank might have recovered from a prior phase of more regular farming. In semi-arid gypsum vegetation, Olano et al. (2012) observed that the soil seed bank can rapidly recover after disturbance, if the seed bank in the surrounding area is still intact. For the conservation management of such habitats, this suggests that populations of rare plant species can recover, as long as flooding recurs within a certain period. Consequently, supporting a fluctuating hydrological regime is crucial for preserving these rare species. In contrast, a changing climate and altering the flooding regime might lead to a further decline of temporarily flooded depressions and the related plant communities. In the Mediterranean region, the predicted decrease of precipitation (IPCC 2007) may lead to shortened hydro-periods that are likely to reduce the spatial extent of amphibious plant communities (Ghosn et al. 2010).
Species composition changes along flooding gradients
The emerging seedling communities were strongly differentiated by flooding duration, indicating that the seed bank of temporarily flooded depressions has the potential to promote different plant communities depending on the actual water regime. The increasing duration of inundation impaired the living conditions for terrestrial species and improved the establishment of water plants, and the gradient from temporary to permanent flooding caused a nearly complete species turnover. Similar to field studies, which observed a clear moisture gradient in the communities of temporarily flooded depressions (Altenfelder et al. 2014; Hoffmann 1996), the species composition of seed bank samples from the upper and lower wetland zones differed. This is consistent with findings of Aponte et al. (2010), who found a gradient in seed bank composition along a moisture gradient. Additionally, regular water level changes, transporting hydrochoerus seeds at the water surface or zoochorous dispersal by birds may contribute to this difference between the two wetland zones.
The experimental water level had no effect on the species composition, probably because flooding at any depth provides constant temperatures under which only a certain set of species are able to germinate (Pons and Schröder 1986). It might also be that plant species react more strongly to light availability, which is not necessarily dependent on the depth of the water column (Wetzel 2001). Light availability can be affected by the turbidity of the water as well as by shading from vegetation cover. During our experiment, all ponds were mostly clear, and light availability was high even at the flooding level of 40 cm. This is consistent with seed bank analyses by Casanova and Brock (2000) who found minor effects of inundation depth on species composition in Australian temporary wetlands. There, depth had an effect in separating groups under waterlogged conditions or no flooding. Among treatments with fluctuating water levels there were no effects of inundation depth as in our experiment. As the influence of flooding depth on seedling establishment might have been of greater importance in turbid water, and as arable field depressions are often muddy, further investigations with water samples of standardised light transmissivities would be reasonable.
Management implications
Our results suggest that the most promising approach to establish and maintain a series of plant communities with high conservation values is to manage water level fluctuations. The most effective way to create diverse communities would be to realise the whole spectrum of flooding situations in a rotational order. According to our results, a short flooding duration leads to the highest species richness but evenness is highest under long-term flooding. Therefore, creating shallow zones around temporarily flooded depressions that can slowly dry up after flooding would benefit many plant species and prevent the dominance of a single competitive species. In contrast, it is important to create adequate conditions for water plants by ensuring that flooding takes place regularly. This can be done by preventing or removing drainage of temporarily flooded depressions. Of course, the flooding regime should particularly focus on the habitat requirements of the species in individual field depressions. To achieve wide species diversity, different field depressions should be included in corresponding programs.
Besides these management treatments that directly affect the depressions, more variable agricultural land use may improve the diversity of the ephemeral plant communities. Devictor et al. (2007) showed that soil tillage is an important management aspect that can enhance the species richness of these plant communities. Also, rare species that occur at temporarily flooded depressions are annuals, and they rely on regular disturbance by soil tillage (Online Resource 1). Setting aside these areas would lead to a rapid change in the species composition towards perennial communities suppressing rare plants (Hoffmann 1996). Furthermore, more restrictive applications of herbicides and fertiliser will improve the diversity of temporarily flooded depressions (Altenfelder et al. 2016). These conservation measures would provide suitable habitat conditions for different plant communities along a flooding gradient. They could enable the whole set of endangered species and rare communities to establish over a sequence of wet and dry years at the same location.
References
Albrecht H (1999) Vergesellschaftung, Standorteigenschaften und Populationsökologie von Arten der Klasse Isoëto-Nanojuncetea auf Ackerflächen. Mitt bad Landesver Naturkunde u Naturschutz NF 17:403–417
Altenfelder S, Raabe U, Albrecht H (2014) Effects of water regime and agricultural land use on diversity and species composition of vascular plants inhabiting temporary ponds in northeastern Germany. Tuexenia 34:145–162
Altenfelder S, Kollmann J, Albrecht H (2016) Effects of farming practice on populations of threatened amphibious plant species in temporarily flooded arable fields: implications for conservation management. Agric Ecosyst Environ 222:30–37
Aponte C, Kazakis G, Ghosn D, Papanastias VP (2010) Characteristics of the soil seed bank in Mediterranean temporary ponds and its role in ecosystem dynamics. Wetl Ecol Manag 18:243–253
Baskin CC, Baskin JM (2014) Seeds: ecology, biogeography, and evolution of dormancy and germination, 2nd edn. Academic Press, San Diego
Bekker RM, Verweij GL, Bakker JP, Fresco LFM (2000) Soil seed bank dynamics in hayfield succession. J Ecol 88:594–607. doi:10.1046/j.1365-2745.2000.00485.x
Bell DM, Hunter JT, Montgomery L (2012) Ephemeral wetlands of the Pilliga Outwash, northwest NSW. Cunninghamia 12:181–190. doi:10.7751/cunninghamia.2012.12.015
Bernhardt KG, Koch M, Ulbel E, Webhofer J (2004) The soil seed bank as a resource for in situ and ex situ conservation of extinct species. In: Robbrecht E, Bogaerts A (eds) EuroGard III: papers from the third European botanic gardens congress and the second European botanic gardens education congress (BEDUCO II). Scripta Botanica Belgica Series, vol 29. National Botanic Garden, Meise, pp 135–139
Bing (2012) Parstein, Germany. 52.936391 N, 14.024835 E. HERE 2015
Bissels S, Donath TW, Hölzel N, Otte A (2005) Ephemeral wetland vegetation in irregularly flooded arable fields along the northern Upper Rhine: the importance of persistent seedbanks. Phytocoenologia 2–3:469–488
Bliss SA, Zedler PH (1998) The germination process in vernal pools: sensitivity to environmental conditions and effects on community structure. Oecologia 113:67–73
Blümel C, Raabe U (2004) Vorläufige Checkliste der Characeen Deutschlands. Rostocker Meeresbiologische Beiträge 13:9–26
Bock CE, Jones ZF, Bock JH (2007) Relationships between species richness, evenness, and abundance in a southwestern Savanna. Ecology 88:1322–1327. doi:10.1890/06-0654
Brock MA (2011) Persistence of seed banks in Australian temporary wetlands: persistence of seed banks in Australian temporary wetlands. Freshw Biol 56:1312–1327. doi:10.1111/j.1365-2427.2010.02570.x
Burrichter E, Hüppe J, Pott R (1993) Agrarwirtschaftlich bedingte Vegetationsanreicherung und—verarmung in historischer Zeit. Phytocoenologia 23:427–447
Casanova MT (2012) Does cereal crop agriculture in dry swamps damage aquatic plant communities? Aquat Bot 103:54–59. doi:10.1016/j.aquabot.2012.06.002
Casanova MT, Brock MA (1990) Charophyte germination and establishment from the seed bank of an Australian temporary lake. Aquat Bot 36:247–254
Casanova MT, Brock MA (2000) How do depth, duration and frequency of flooding influence the establishment of wetland plant communities? Plant Ecol 147:237–250
Collins DP, Conway WC, Mason CD, Gunnels JW (2013) Seed bank potential of moist-soil managed wetlands in east-central Texas. Wetl Ecol Manag 21:353–366. doi:10.1007/s11273-013-9307-5
Day P, Deadman AJ, Greenwood BD, Greenwood EF (1982) A floristic appraisal of marl pits in parts of north-western England and northern Wales. Watsonia 14:153–165
de Mendiburu F (2014) agricolae: statistical procedures for agricultural research. R package version 1.1-7
De Steven D, Sharitz RR, Singer JH, Barton CD (2006) Testing a passive revegetation approach for restoring coastal plain depression wetlands. Restor Ecol 14:452–460. doi:10.1111/j.1526-100X.2006.00153.x
Devictor V, Moret J, Machon N (2007) Impact of ploughing on soil seed bank dynamics in temporary pools. Plant Ecol 192:45–53
DWD (2014) Download of the long-term mean of temperature and precipitation (1981–2010). http://www.dwd.de. Accessed 27 Jan 2014
Ellenberg H, Weber HE, Düll R, Wirth V, Werner W (2001) Zeigerwerte von Pflanzen in Mitteleuropa. Scr Geobot 18:1–262
Faist AM, Ferrenberg S, Collinge SK (2013) Banking on the past: seed banks as a reservoir for rare and native species in restored vernal pools. Aob Plants 5:plt043. doi:10.1093/aobpla/plt043
Galatowitsch SM, van der Valk AG (1996) The vegetation of restored and natural prairie wetlands. Ecol Appl 6:102–112. doi:10.2307/2269557
Ghosn D, Vogiatzakis IN, Kazakis G, Dimitriou E, Moussoulis E, Maliaka V, Zacharias I (2010) Ecological changes in the highest temporary pond of western Crete (Greece): past, present and future. Hydrobiologia 648:3–18. doi:10.1007/s10750-010-0143-9
Grime JP (1979) Plant strategies and vegetation processes. Wiley, New York
Gruber S, Buehler A, Moehring J, Claupein W (2010) Sleepers in the soil—vertical distribution by tillage and long-term survival of oilseed rape seeds compared with plastic pellets. Eur J Agron 33:81–88. doi:10.1016/j.eja.2010.03.003
Harper JL (1987) Population Biology of Plants, 7th edn. Academic Press, London
Hill M, Gauch H (1980) Detrended correspondence-analysis—an improved ordination technique. Vegetatio 42:47–58. doi:10.1007/BF00048870
Hoffmann J (1996) Zwei Vorkommen von Schoenoplectus supinus (L.) Palla in Ostbrandenburg. Verh Bot Ver Berlin Brandenburg 129:85–96
Hölzel N, Otte A (2004) Inter-annual variation in the soil seed bank of flood-meadows over two years with different flooding patterns. Plant Ecol 174:279–291. doi:10.1023/B:VEGE.0000049108.04955.e2
IPCC (2007) Climate Change 2007: Synthesis Report. Contribution of Working Groups I, II and III to the Fourth Assessment Report of the intergovernmental panel on climate change. Geneva
Kabus T, Mauersberger R (2011) Liste und Rote Liste der Armleuchteralgen (Characeae) des Landes Brandenburg. Natschutz Landschpfl Brandenburg 20:1–32
Kappelle M, Van Vuuren MMI, Baas P (1999) Effects of climate change on biodiversity: a review and identification of key research issues. Biodivers Conserv 8:1383–1397. doi:10.1023/A:1008934324223
Keddy P, Ellis T (1985) Seedling recruitment of 11 wetland plant-species along a water level gradient—shared or distinct responses. Can J Bot-Rev Can Bot 63:1876–1879
Keddy PA, Reznicek AA (1986) Great-lakes vegetation dynamics—the role of fluctuating water levels and buried seeds. J Great Lakes Res 12:25–36
Klafs G, Jeschke L, Schmidt H (1973) Genese und Systematik wasserführender Ackerhohlformen in den Nordbezirken der DDR. Arch Naturschutz Landschaftsforsch 13:287–302
Kwon Y-S et al (2013) Evaluation of global warming effects on the geographical distribution of weeds in paddy fields by characterizing germination time and morphological factors. Ecol Inform 17:94–103. doi:10.1016/j.ecoinf.2013.06.007
Landesumweltamt BS (2006) Liste und Rote Liste der etablierten Gefäßpflanzen Brandenburg. Natschutz Landschpfl Brandenburg 15:1–163
Lansdown RV (2014) The IUCN Red List of threatened species. Version 2015.2. www.iucnredlist.org. Accessed 08 July 2015
Lentz KA, Dunson WA (1999) Distinguishing characteristics of temporary pond habitat of endangered northeastern bulrush, Scirpus ancistrochaetus. Wetlands 19:162–167. doi:10.1007/BF03161745
Li H-L, Wang Y-Y, Zhang Q, Wang P, Zhang M-X, Yu F-H (2015) Vegetative propagule pressure and water depth affect biomass and evenness of submerged macrophyte communities. PloS One 10:e0142586–e0142586. doi:10.1371/journal.pone.0142586
Ludwig G, Schnittler M (1996) Rote Liste gefährdeter Pflanzen Deutschlands. Schriftenreihe Vegetationskunde 28:1–744
Lukacs BA, Sramko G, Molnar A (2013) Plant diversity and conservation value of continental temporary pools. Biol Conserv 158:393–400. doi:10.1016/j.biocon.2012.08.024
Ma M (2005) Species richness vs evenness: independent relationship and different responses to edaphic factors. Oikos 111:192–198. doi:10.1111/j.0030-1299.2005.13049.x
Madsen H, Arnbjerg-Nielsen K, Mikkelsen PS (2009) Update of regional intensity-duration-frequency curves in Denmark: tendency towards increased storm intensities. Atmos Res 92:343–349. doi:10.1016/j.atmosres.2009.01.013
Mann H, Raju MVS (2002) First report of the rare charophyte Nitella macounii (T. F. Allen) T. F. Allen in Saskatchewan and western Canada. Can Field-Nat 116:559–570
Matsuo K, Noguchi K, Nara M (1984) Ecological studies on Rorippa islandica (Oeder) Borb. 1. Dormancy and external conditions inducing seed germination. Weed Res (Japan) 29:220–225
Meyer S, Wesche K, Krause B, Leuschner C (2013) Dramatic losses of specialist arable plants in Central Germany since the 1950s/60 s-a cross-regional analysis. Divers Distrib 19:1175–1187. doi:10.1111/ddi.12102
Oksanen J et al. (2013) vegan: Community Ecology Package. R package version 2.0–10
Olano JM, Caballero I, Escudero A (2012) Soil seed bank recovery occurs more rapidly than expected in semi-arid Mediterranean gypsum vegetation. Ann Bot 109:299–307. doi:10.1093/aob/mcr260
Pinke G, Csiky J, Mesterhazy A, Tari L, Pal RW, Botta-Dukat Z, Czucz B (2014) The impact of management on weeds and aquatic plant communities in Hungarian rice crops. Weed Res 54:388–397. doi:10.1111/wre.12084
Pons T, Schröder H (1986) Significance of temperature-fluctuation and oxygen concentration for germination of the rice field weeds Fimbristylis littoralis and Scirpus juncoides. Oecologia 68:315–319. doi:10.1007/BF00384806
Poschlod P (1993) Underground floristics—keimfähige Diasporen im Boden als Beitrag zum floristischen Inventar einer Landschaft am Beispiel der Teichbodenflora. NuL 68:155–159
Poschlod P, Böhringer J, Fennel S, Prume C, Tiekötter A (1999) Aspekte der Biologie und Ökologie von Arten der Zwergbinsenfluren. Mitt bad Landesver naturkunde u Naturschutz 17:219–260
Pukacz A, Pelechaty M, Raabe U (2009) Pierwsze stanowisko Chara baueri (Characeae) w Polsce. Fragm Flor Geobot Pol 16:425–429
R Core Team (2013) A language and environment for statistical computing, 3.0.2 edn. R Foundation for Statistical computing, Vienna
Raabe U (2009) Chara baueri rediscovered in Germany—plus additional notes on Gustav Heinrich Bauer (1794–1888) and his herbarium. ICGC News 20:13–16
Saatkamp A, Poschlod P, Venable DL (2014) The functional role of soil seed banks in natural communities. In: Gallagher RS (ed) Seeds: the ecology of regeneration in plant communities. CABI, Wallingford, pp 263–295
Slivitzky M (2002) Ecological impacts of water use and changes in levels and flows: a literature review. Great Lakes Commission, Ann Arbor
Soininen J, Passy S, Hillebrand H (2012) The relationship between species richness and evenness: a meta-analysis of studies across aquatic ecosystems. Oecologia 169:803–809. doi:10.1007/s00442-011-2236-1
Stirling G, Wilsey B (2001) Empirical relationships between species richness, evenness, and proportional diversity. Am Nat 158:286–299. doi:10.1086/321317
Thompson K, Bakker JP, Bekker RM, Hodgson JG (1998) Ecological correlates of seed persistence in soil in the north-west European flora. J Ecol 86:163–169. doi:10.1046/j.1365-2745.1998.00240.x
van Dijk J, Stroetenga M, van Bodegom PM, Aerts R (2007) The contribution of rewetting to vegetation restoration of degraded peat meadows. Appl Veg Sci 10:315
van Zanten BT et al (2014) European agricultural landscapes, common agricultural policy and ecosystem services: a review. Agron Sustain Dev 34:309–325. doi:10.1007/s13593-013-0183-4
von Lampe M (1996) Wuchsform, Wuchsrhythmus und Verbreitung der Arten der Zwergbinsengesellschaften. Diss Bot 266:1–353
Weiher E, Keddy PA (1999) Relative abundance and evenness patterns along diversity and biomass gradients. Oikos 87:355–361. doi:10.2307/3546751
Wetzel RG (2001) Limnology—lake and river ecosystems, 3rd edn. Academic Press, San Diego
Wisskirchen R, Haeupler H (1998) Standardliste der Farn- und Blütenpflanzen Deutschlands. Ulmer, Stuttgart
Zacharias I, Zamparas M (2010) Mediterranean temporary ponds. A disappearing ecosystem. Biodivers Conserv 19:3827–3834. doi:10.1007/s10531-010-9933-7
Acknowledgments
We thank Philipp Glaab, Ingrid Kapps, Julia Prestele and the staff of the Botanical Garden of the University of Regensburg for help with installing and maintaining the experiment. Uwe Raabe identified the charophytes. Comments on a previous version of this article by three anonymous referees are gratefully acknowledged.
Funding
This work was funded by Deutsche Bundesstiftung Umwelt (Az 29 317–33).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Altenfelder, S., Schmitz, M., Poschlod, P. et al. Managing plant species diversity under fluctuating wetland conditions: the case of temporarily flooded depressions. Wetlands Ecol Manage 24, 597–608 (2016). https://doi.org/10.1007/s11273-016-9490-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-016-9490-2