Abstract
This study explored the potential role of agro-ecology in shaping the morphology of Lagune cattle population of Benin. A total of 708 adult Lagune cattle were sampled randomly from nine provenances in two agro-ecological zones (AEZs) and were assessed for eight qualitative and twelve linear body measurements. Data were analyzed using generalized linear model procedures (PROC GLM) followed by the multiple comparison of least square means (LSMEAN) according to the Tukey-Kramer method and multivariate analytical methods, including canonical discrimination analysis (CDA) and hierarchical ascendant classification. Irrespective of AEZ and sex, the body length (102.3 ± 9.31 cm) was greater than the wither height (93.1 ± 7.39 cm) and the body index smaller than 0.85. However, there were significant differences between the two AEZs for most of the measured morphometric and qualitative traits. Moreover, a male-biased sexual size dimorphism was recorded. The CDA based on only four basic body measurements (rump height, body length, heart girth, and ear length) and the calculated Mahalanobis distances suggest that the populations from the two AEZs are distinct and could be further considered ecotypes. Nevertheless, the overall moderate classification rate (70%) of the individual animals into their group of origin indicates interbreeding between the two populations. The pairwise Mahalanobis distances between provenances in the same AEZ were also significant. Together, these results provide supporting evidence for the existence of subdivisions in the Lagune cattle populations from South Benin. The high morphological diversity in the Lagune cattle recorded in the present study could serve as a starting point for the development of efficient selection and sound subpopulation management strategies but also for further phenotypic and genetic characterizations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Lagune cattle are dwarf, shorthorn, and trypanotolerant cattle of the Bos taurus (taurine) subspecies mainly found in coastal areas and near lagoons in West and Central Africa (Rege and Tawah 1999; Felius et al. 2014), where it is known under many local names (Porter 2002). Like all other farm animal genetic resources, the Lagune cattle play important socio-economic and cultural roles and support food security and livelihoods of thousands of smallholders (FAO 2012). But, there is an increasing recognition that this breed is threatened with extinction by indiscriminate crossbreeding and inappropriate husbandry techniques (Rege 1999; Gbangboche et al. 2011; Felius et al. 2014) that could involve an irreversible loss of valuable but untapped genetic diversity necessary to sustain food security (Seré et al. 2008; Hanotte et al. 2010).
The present-day Lagune cattle have evolved in diverse ecological conditions in West Africa. It probably derived from the first waves of migrations of shorthorn Bos taurus into West Africa from Asia about 2750–2500 BC (Epstein 1971). These migrations were followed by ecological adaptation (Felius et al. 2014) leading to several changes in cattle phenotypes (Ajmone-Marsan et al. 2010). As shown in other African indigenous cattle (Kabi et al. 2015; Terefe et al. 2015; Nyamushamba et al. 2017), Lagune cattle populations that have evolved in different ecological conditions may have some adaptive intrinsic characteristics and unique morphological traits that make them different, to some extent, from each other. It is widely accepted that morphological differences in domesticated animal populations of the same species are usually driven by farmer’s selection or by adaptive mechanisms, such as phenotypic plasticity and local adaptation (de Jong and Bijma 2002). Phenotypic plasticity is the ability of a genotype to develop different phenotypes that fit better to the environmental conditions, whereas local adaptation to environmental conditions is a genetic change mainly due to natural selection (Weyl and Coetzee 2016). Hence, the morphological traits of cattle, regardless of its genetic type, are influenced by environment. But in addition, differences between genetic types can vary depending on the environment; that is, the expression of a phenotype is a function of the genotype, the environment, and the genotype by environment interaction (Falconer and Mackay 1996). Knowing and using the best combination of genotype and environment would help to develop efficient breeding strategies for tropically adapted taurine breeds and to improve the sustainability of animal production systems (Burrow 2012).
There are no estimates of the number and effective population size for the Lagune cattle population of Benin. But its drastic regression has been reported owing to indiscriminate slaughter, inappropriate breeding management, strategies and policies and poor marketing management (Gbangboche et al. 2011; Ahozonlin et al. 2019). Compared with other West African cattle breeds, the Lagune cattle have been poorly studied. This breed, which was present throughout all of the southern part of Benin, is now restricted to mainly two agro-ecological zones (DE-MAEP 2001). To our best knowledge, there are no comprehensive studies that have quantified its morphological variation across its agro-ecological range, neither in Benin nor in West Africa. Furthermore, although few phenotypic and genetic diversity studies have been undertaken for West African cattle breeds and have included the Lagune cattle samples from Benin (Meghen et al. 1994; Freeman et al. 2004; Gautier et al. 2009; Koudandé et al. 2009; Traoré et al. 2015; Traoré et al. 2016), their sampling and analyses did not explicitly account for possible variations across and within agro-ecological zones in the country.
The knowledge of the morphological variation among and within the Lagune cattle populations from Benin and of the underlying genetic variation is necessary in developing appropriate management and efficient selection strategies for this underutilized genetic resource. Therefore, the objective of this study was to quantify morphological variations of the Lagune populations across and within two agro-ecological zones in Southern Benin. We hypothesized that the Lagune cattle of Benin are not a homogeneous population and can be subdivided in several sub-populations based on their morphology.
Material and methods
Study area
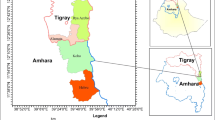
The study was carried out in two agro-ecological zones (Pobe and Oueme Valley) in the Guineo-congolese vegetation zone within the natural distribution area of the Lagune cattle in South Benin (Fig. 1). Both agro-ecological zones share the same subequatorial climatic conditions characterized by a bimodal rainfall regime and an average annual precipitation ranging from 900 to 1300 mm, but differ in terms of averages daily temperature, humidity index, soil characteristics, and predominant vegetation (Table 1).
Sampling procedure
Five municipalities (Bonou, Adjohoun, Dangbo, Zogbodomey, Zè) in the Oueme Valley AEZ and four (Adja-ouere, Akpro-Missereté, Avrankou, Adjarra) in the Pobe AEZ were selected based on their accessibility, the importance of cattle farming as a livelihood strategy, and the effective presence and predominance of Lagune cattle. In each municipality, 10 villages were randomly chosen. Depending on their availability and willingness to participate in the survey, five to ten smallholder farmers keeping at least two cows were surveyed in each village. Each selected farmer was first asked to list the number of cattle breeds present in his/her herd, then to designate at least two mature adult animals of Lagune breed. Only animals (male and/or non-pregnant female) over 3.5 years of age, as confirmed by their dentition, were included in the study. The approximate age of the animals was determined by examining their teeth as described by Poivey et al. (1981), Pace and Wakeman (2003), and Parish and Karisch (2013) and compared with the age given by the farmer. Only animals that possessed four pairs of permanent teeth (3.5 years old and more) were retained and described for various physical traits. A total of 708 adult animals (321 in the Oueme Valley zone and 387 in the Pobe zone), including 585 females and 123 males, were sampled. All individual animals sampled in an agro-ecological zone were considered a population whereas each municipality was considered a provenance.
Data collection procedure
A total of eight physical characteristics and twelve quantitative traits were measured on each sampled animal, restrained on a relatively even surface, according to FAO guidelines (FAO 2011). To avoid biases that might arise from varying enumerators, all measurements were taken by the same team in both AEZs. This team was composed of a researcher and a trained field assistant. The physical characteristics, recorded on the basis of a visual observation, were body hair coat color pattern, body hair coat color, head profile, presence or absence of horns, orientation and horn shape, back profile, and rump profile. The twelve body measurements, described in Table 2, were recorded using a flexible measuring tape and a measuring stick early in the morning before the animals were grazed to avoid biases on certain traits due to feed intake. All linear measurements were taken in centimeters (cm). The geographical positions of the herds in which the animals were sampled were recorded using a Garmin GPS (etrex vista TM) and photographs of each animal measured were taken using a digital camera.
Data analysis
Five morphological indices (Table 3) were generated from the linear body measurements taken. All statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). Descriptive statistics for the visually measured physical characteristics and for the linear body measurements were performed using the procedures PROC FREQ and PROC UNIVARIATE, respectively. The Least square means (LSMEAN) of the morphometric traits, their standard errors (SE), and the coefficients of variation were calculated for each sex group and agro-ecological zone (AEZ). Subsequently, the general linear model procedure (PROC GLM) and the chi-square (χ2) test were used to analyze the effect of sex group and of AEZ on the morphometric and physical traits of the Lagune cattle respectively. A stepwise discriminant analysis was performed using the PROC STEPDISC to identify the morphometric traits and morphological indices with high discriminating power. The relative discriminatory ability of a variable was assessed using the partial R2, the F-value, and the level of significance (P > F). Subsequently, the PROC CANDISC procedure was used to perform canonical discriminant analyses on the selected variables, to derive canonical functions, linear combinations of the variables that summarize variation between AEZ and between provenances, and to calculate the associated Mahalanobis distances. The PROC DISCRIM procedure was used to measure the ability of the computed canonical functions to assign each individual animal to its a priori AEZ or provenance group. The degree of morphological similarity or dissimilarity between animals from different AEZs and provenances was determined using the hierarchical cluster analysis procedure (PROC CLUSTER). Finally, the PROC TREE procedure was used to build the dendrograms of populations from provenances within each AEZ and between AEZs.
Results
Morphometric trait variation between agro-ecological zones
There were significant differences between agro-ecological zones (Table 4) for most of the measured morphometric traits. Except for hock circumference (HC), horn tip interval (HTI), pelvic width (PW), and body length (BL), Lagune cattle sampled from the Oueme Valley AEZ had significantly larger MC, head length (HL), ear length (EL), heart girth (HG), height at withers (HW), horn length (LH), scapulo-ischial length (SIL), and rump height (HR) than those from the Pobe AEZ. Further, irrespective of AEZ, all the linear body measurements, except MC, HL, EL HTI, and HC, showed significant differences between cows and bulls (Table 5). Irrespective of AEZ and sex, the body index (Bi) values were smaller than 0.85. In contrast, there were significant differences between the populations from the two AEZs for the over increase (OIi) and height slope (HSi) indices.
Physical trait variations
A diversity of horn shape, of coat color and patterns, and of horn orientation was observed in the Lagune populations with significant variations between the two AEZs (Table 6). Patchy/pied coat (48.2%) were the most frequently recorded body hair coat color pattern with a diversity of color, either pied black (50.3%) or black pied (46.0%). It was followed by plain coat (29.8%) with white (51.5%) or black (47.0%) as dominant colors. Spotted coat (22.1%) with dark red, fawn, and brown was also observed in the two zones, but significantly more common in the Oueme Valley AEZ than in the Pobe AEZ.
Multivariate analysis of morphometric traits
The results of the stepwise discriminant analysis (Table 7) indicated that three (HR, Bi, and EL) out of the 10 variables included in the analysis had higher (P < 0.001) partial R2 and F-values implying their greater discriminant power compared with others in assessing the morphometric similarity/dissimilarity between the Lagune cattle from the two AEZs. The use of these three variables in the canonical discriminant analysis generated three significant canonical variables (CAN 1, CAN 2, and CAN 3) as revealed by the standardized coefficients for the discriminant function, the canonical correlation, the eigenvalue, and share of total variance accounted for (Table 8). The first two canonical functions accounted for 72.29% of the total variation. CAN 1 was dominated by a high positive loading of HR and a negative loading of Bi, whereas CAN 2 was dominated by a high positive loading of EL, and CAN 3 by high positive loadings of Bi and HR. The measured squared Mahalanobis distances between the populations from the two AEZs were 19.27 and significant (P ˂ 0.0001). The discriminant functions correctly classified about 70% of the individual cattle (Table 9) in their a priori group. Similarly, the pairwise Mahalanobis distances between provenances within each AEZ were significant (Table 10).
The dendrograms generated using the squared Mahalanobis distances between provenances showed two sub-groups or sub-populations of Lagune within each of the two AEZs (Fig. 2). Further, irrespective of AEZ, the cluster analysis separated the provenances in three homogeneous and distinct groups (Fig. 3).
Discussion
Our study presents evidence of the existence of a sexual dimorphism in the Lagune cattle, with significantly higher values of most of the body measurements (HW, BL, and HR especially) recorded for males. This result is in line with previous findings by Aboagye et al. (1994) and by Gbangboche et al. (2011). Similar dimorphism was also reported by Mwai et al. (2015), Daikwo et al. (2018), and Yakubu et al. (2018) in the savanna Muturu cattle of Nigeria, a very similar breed to Lagune (Aboagye et al. 1994; Porter et al. 2016).
The body length was greater than the wither height whereas the calculated body index (Bi) was less than 0.85 irrespective of sex and agro-ecological zone. According to Chacón et al. (2011), these results confirm that the Lagune cattle are brevigline animals. Body ratio (over increase index) and height slope are indicated that the two populations were slightly shorter at the withers than at the rump. Our findings further confirm that Lagune cattle have a small and compact body, no hump, a straight back, a broad head, and very short horns. These morphological characteristics are similar to those reported by Aboagye et al. (1994) and Kubkomawa et al. (2015).
The analyses show that, except for hock circumference and pelvis width, Lagune cattle from the Oueme Valley AEZ had significantly larger features than those from the Pobe AEZ. These significant morphological divergences between the two AEZs could be attributed to their differences in terms of geographical position, soil, and vegetation conditions (Desta et al. 2011; Traoré et al. 2015; Tenagne et al. 2016).
Similar to body size, coat color is a key morphological adaptive trait in ruminants (Berihulay et al. 2019; Gaughan et al. 2018) and is controlled by a small number of genes of large effect (Olson 1999). Hence, the high coat color variability observed in Lagune cattle included in our study suggests that the observed morphological divergences among provenances and between the two AEZs could also be of genetic origin, resulting from the adaptation of the Lagune breed to diverse environmental challenges. Indeed, as pointed out by Olson (1999) and Porto-Neto et al. (2014), the color of the coat is directly related to radiation and heat absorption, and therefore affects the animal’s tolerance or adaption to heat stress and water scarcity. The coat color diversity observed in the present study agrees with reports by Rege and Tawah (1999) and by Aboagye et al. (1994).
The results of the discriminant analyses confirm that the Lagune cattle populations from the two AEZs were morphologically distinct. The relatively high morphological distance (19.27) between the two cattle populations coupled with the correct classification rate of 70% of the individual cattle into their area of origin by the discriminant function models further indicates that they represent two different ecotypes. These results suggest a strong influence of the environmental factors on the morphology of the Lagune cattle. Indeed, environmental factors influence the phenotype (Rauw and Gomez-Raya 2015) which is the expression of the genotype in response to various environments (Kolmodin et al. 2003). We further argue that these ecological drivers may have also played a role in changing the genetic composition of the breed through natural selection as the latter allow populations within a breed to adapt to specific environments (Blackburn et al. 2017). Nevertheless, the important overlap between the populations of the two AEZs as shown in Fig. 4 suggests that factors other than environment, such as farmer’s general herd management practices including selection for specific traits and genotype by environment interactions, might affect Lagune cattle morphology. These genotype by environment interactions are of major importance in tropical cattle breeding systems (Burrow 2012) as it can be used to ensure that livestock populations with a small effective population size maintain a high fitness across environments (Kristensen et al. 2015). This might be also of relevance for the Lagune cattle which is considered threatened (Rege 1999; Gbangboche et al. 2011; Felius et al. 2014), although estimates of its effective population size are currently not available due to lack of census. Intermingling of individuals from the two populations, facilitated by their geographical proximity, could also explain the observed overlap.
The observed significant morphological differences between individuals from different provenances within agro-ecological zones further support the hypothesis of the existence of several morphotypes (subgroups) in each of the two identified Lagune cattle ecotypes. In addition, the dendogram generated by the cluster analysis (Fig. 3) distinguished three homogeneous groups of provenances. It separated two provenances (Zè and Zogbodomey) from the rest and indicated that some provenances of different AEZs are morphologically more close to each other than to other provenances from their respective AEZ. Again, these results suggest that the variation of the ecological conditions and fodder resource constraints among provenances of the same AEZ represent also a source of morphological variation. Similar significant morphological differences were observed in the Ankole cattle populations from Uganda by Kabi et al. (2015) who associated it with morphological signatures of selection. These authors reported that in a context of low availability of feed and water resources, indigenous farmers’ selection criteria tend to favor individuals with low body dimensions.
The different provenances and AEZs were successfully separated with only three variables involving four basic body measurements, namely the rump height, body length, heart girth, and ear length. As consequence, taking these basic measurements consistently can be very useful in breed identification and classification as well as in the selection of individuals exhibiting highest potential for traits of interest. The high morphological variability in the Lagune populations recorded in the present study infers a considerable genetic variation, which, if confirmed through molecular analyses, could be used in breed improvement.
Conclusion
This study has investigated the morphological variability in the Lagune cattle populations from South Benin. These populations were morphologically sub-structured according to agro-ecological zones into two ecotypes. Evidence presented herein supports the hypothesis of the existence of several Lagune cattle subpopulations. The high morphological diversity recorded among these subpopulations suggests the existence of high untapped genetic variation that should be exploited for the development and implementation of sound management and genetic improvement of this indigenous cattle breed.
References
Aboagye, G.S., Tawah, C.L. and Rege, J.E.O., 1994. Shorthorn cattle in West and Central Africa III. Physical adaptive and special genetic characteristics. World Animal Review, 78(1), 22–32.
Ahozonlin, C.M., Koura, I.B. and Dossa, L.H., 2019. Determinants of crossbreeding practices by cattle farmers in South Benin, West Africa: implications for the sustainable use of the indigenous Lagune cattle population. Sustainable Agriculture Research 8(2):101–109. DOI:https://doi.org/10.5539/sar.v8n2p101
Ajmone-Marsan, P., Garcia, J.F. and Lenstra, J.A., 2010. On the origin of cattle: how aurochs became cattle and colonized the world. Evolutionary Anthropology, 19, 148–157. https://doi.org/10.1002/evan.20267
Alderson, G.L.H., 1999. The development of a system of linear measurements to provide an assessment of type and function of beef cattle. Animal Genetic Resources Information, 25, 45-55. DOI: https://doi.org/10.1017/S1014233900005782
Banerjee, S., Ahmed, M.B. and Tefere, G., 2014. Studies on morphometrical traits of Boran bulls reared on two feedlots in Southern Ethiopia. Animal Genetic Resources Information, 54, 53–63. doi:https://doi.org/10.1017/S2078633614000095.
Berihulay, H., Abied, A., He, X., Jiang, L. and Ma, Y., 2019. Adaptation Mechanisms of Small Ruminants to Environmental Heat Stress. Animals, 9, 75. doi :https://doi.org/10.3390/ani9030075.
Blackburn, H. D., Krehbiel, B., Ericsson, S. A., Wilson, C., Caetano, A. R. and Paiva, S. R., 2017. A fine structure genetic analysis evaluating ecoregional adaptability of a Bos taurus breed (Hereford). PloS one, 12(5), e0176474. doi: https://doi.org/10.1371/journal.pone.0176474.
Burrow, H. M., 2012. Importance of adaptation and genotype× environment interactions in tropical beef breeding systems. Animal, 6(5), 729-740. DOI: https://doi.org/10.1017/S175173111200002X.
Chacón, E., Macedo, F., Velázquez, F., Paiva, S.R., Pineda, E. and McManus, C., 2011. Morphological measurements and body indices for Cuban Creole goats and their crossbreds. Revista Brasileira de Zootecnia, 40(8), 1671–1679. https://doi.org/10.1590/S1516-35982011000800007
Daikwo, S.I., Ogah, D.M., Amuda, A.J. and Dike, U.A., 2018. Prediction of Body Weight of Savanna Muturu Cattle (Bos brachyceros). Asian Journal of Research in Animal and Veterinary Sciences, 2(1), 1–6. Doi: https://doi.org/10.9734/AJRAVS/2018/43017.
de Jong, G. and Bijma, P., 2002. Selection and phenotypic plasticity in evolutionary biology and animal breeding. Livestock Production Science, 78(3), 195–214. https://doi.org/10.1016/S0301-6226(02)00096-9
DE-MAEP (Direction de l’Elevage-Ministère de l’Agriculture de l’Elevage et de la Pêche), 2001. Rapport national 2000. Cotonou, République du Bénin
Desta, T.T., Ayalew, W. and Hedge, B.P., 2011. Breed and trait preferences of Sheko cattle keepers in southern Ethiopia. Tropical Animal Health and Production, 43, 851–856. https://doi.org/10.1007/s11250-010-9772-2
Epstein, H., 1971. The origin of the domestic animals of Africa, Vol 1. New York, NY, USA, Pub. Africana.
Falconer, D.S. and Mackay, T.F.C., 1996. Introduction to quantitative genetics. Essex. UK: Longman Group.
FAO, 2011. Guidelines to Phenotypic characterization of Animal Genetic Resources. Food and Agriculture Organization of the United Nations, FAO, Rome.
FAO, 2012. World livestock 2011: Livestock in food security. Food and Agriculture Organization of the United Nations, FAO, Rome.
Felius, M., Beerling, M.L., Buchanan, D.S., Theunissen, B., Koolmees, P.A. and Lenstra, J.A., 2014. On the History of Cattle Genetic Resources. Diversity, 6, 705–750. Doi:https://doi.org/10.3390/d6040705.
Freeman, A.R., Meghen, C.M., Machugh, D.E., Loftus, R.T., Achukwi, M.D., Bado, A., Sauveroche, B. and Bradley, D.G., 2004. Admixture and diversity in West African cattle populations. Molecular Ecology, 13(11), 3477–3487. https://doi.org/10.1111/j.1365-294X.2004.02311.x
Gantner, V., Mijić P., Kuterovac, K., Solićr, D. and Gantner, R., 2011. Temperature-humidity index values and their significance on the daily production of dairy cattle. Mljekarstvo, 61(1), 56–63. Accessed 12 May 2018 at https://hrcak.srce.hr/65205
Gaughan, J.B., Sejian, V., Mader, T.L. and Dunshea, F.R., 2018. Adaptation strategies: ruminants. Animal Frontiers, 9(1), 47–53. https://doi.org/10.1093/af/vfy029
Gautier, M., Flori, L., Riebler, A., Jaffrézic, F., Laloé, D., Gut, I., Moazami-Goudarzi, K. and Foulley, J.L., 2009. A whole genome Bayesian scan for adaptive genetic divergence in West African cattle. B M C genomics, 10(1), 550. https://doi.org/10.1186/1471-2164-10-550
Gbangboche, A.B., Alkoiret, T.I., Chrysostome, C.A.A.M., Salifou, S. and Hornick, J.L., 2011. Preweaning growth performance of Lagune cattle in Benin. International Journal of Livestock Production, 2(10), 159–165.
Hanotte, O., Dessie, T. and Kemp S., 2010. Time to tap Africa's livestock genomes. Washington: Science, 328(5986), 1640–1641. Doi: https://doi.org/10.1126/science.1186254
Kabi, F., Masembe, C., Negrini, R. and Muwanika, V., 2015. Patterns of indigenous female cattle morphometric traits variations in Uganda: evidence for farmers’ selection to enhance agro-ecological fitness. Animal Genetic Resources Information, 56, 79–90. Doi: https://doi.org/10.1017/S2078633614000551.
Kolmodin, R., Strandberg, E., Jorjani, H. and Danell, B., 2003. Selection in the presence of genotype by environmental interaction: Response in environmental sensitivity. Journal of Animal Science, 76, 375-385. DOI: https://doi.org/10.1017/S1357729800058604
Koudandé, O.D., Dossou-Gbété, G., Mujibi, F., Kibogo, H., Mburu, D., Mensah, G.A., Hanotte, O. and Van Arendonk, J.A.M., 2009. Genetic diversity and zebu genes introgression in cattle population along the coastal region of the Bight of Benin. Animal Genetic Resources Information, 44, 45-55. DOI: https://doi.org/10.1017/S1014233900002856
Kristensen, T. N., Hoffmann, A. A., Pertoldi, C. and Stronen, A. V., 2015. What can livestock breeders learn from conservation genetics and vice versa?. Frontiers in genetics, 6, 38. https://doi.org/10.3389/fgene.2015.00038
Kubkomawa, I.H., Krumah, L.J., Etuk, E.B. and Okoli, I.C., 2015. Current pastoral cattle production situation in West Africa: A review. Dynamic Journal of Animal Science and Technology, 1(1), 1–17., http://www.journaldynamics.org/djast. Accessed 28 Nov 2016.
Meghen, C., MacHugh, D.E. and Bradley, D.G., 1994. Genetic characterization and West African cattle. World Animal Review, 78(1), 59–66.
Mwai, O., Hanotte, O., Kwon, Y. and Cho, S., 2015. African indigenous cattle: unique genetic resources in a rapidly changing world. Asian Australasian Journal of Animal Science, 28(7), 911–921. https://doi.org/10.5713/ajas.15.0002R.
Nyamushamba, G.B., Mapiye, C., Tada, O., Halimani, T.E. and Muchenje, V., 2017. Conservation of indigenous cattle genetic resources in Southern Africa’s smallholder areas: turning threats into opportunities - A review. Asian-Australasian Journal of Animal Sciences, 30(5), 603–621. https://doi.org/10.5713/ajas.16.0024
Olson, T. A., 1999. Genetics of color variation. In: The genetics of cattle. Eds: Fries R and Ruvinsky A. Wallingford: CAB International, pp. 33-53.
Pace, J.E. and Wakeman, D.L., 2003. Determining the age of cattle by their teeth. CIR253. Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences. University of Florida, Gainesville, FL.
Parish, J.A. and Karisch, B.B., 2013. Estimating cattle age using dentition. Mississippi Extension Service of Department of Agriculture Mississippi State University, cooperating with U.S.
Poivey, J.P., Landais, E., Seitz, J.L. and Kouyaté, M., 1981. Détermination de l'âge des bovins par l'examen de la dentition. Revue d'Élevage et de Médecine Vétérinaire des Pays Tropicaux, 34, 55–62.
Porter, V., 2002. Mason's world dictionary of livestock breeds, types and varieties. 5th Edition, CABI Publishing, Wallingford.
Porter, V., Alderson, L., Hall, S.J. and Sponenberg, D.P., 2016. Mason's World Encyclopedia of Livestock Breeds and Breeding, 2 Volume Pack. CABI Publishing, Wallingford.
Porto-Neto, L.R., Reverter, A., Prayaga, K.C., Chan, E.K.F., Johnston, D.J., Hawken, R.J. and Barendse, W., 2014. The genetic architecture of climatic adaptation of tropical cattle. PLoS ONE, 9(11), e113284. https://doi.org/10.1371/journal.pone.0113284
Rauw, W.M. and Gomez-Raya, L., 2015. Genotype by environment interaction and breeding for robustness in livestock. Frontiers in genetics, 6, 310, https://doi.org/10.3389/fgene.2015.00310
Rege, J.E.O., 1999. The state of African cattle genetic resources I. Classification framework and identification of threatened and extinct breeds. Animal Genetic Resources Information, 25, 1-26. DOI: https://doi.org/10.1017/S1014233900003448
Rege, J.E.O. and Tawah, C.L., 1999. The state of African cattle genetic resources II: geographical distribution, characteristics and uses of present-day breeds and strains. Animal Genetic Resources Information, 26, 1-25. DOI: https://doi.org/10.1017/S1014233900001152
Salako, A.E., 2006. Application of morphological indices in the assessment of type and function in Sheep. International Journal of Morphology, 24(1), 13–18.
Segnalini, M., Nardone, A., Bernabucci, U., Vitali, A., Ronchi, B. and Lacetera, N., 2011. Dynamics of the temperature-humidity index in the Mediterranean basin. International Journal of Biometeorology, 55, 253–263. https://doi.org/10.1007/s00484-010-0331-3
Segnalini, M., Bernabucci, U., Vitali, A., Nardone, A. and Lacetera, N., 2013. Temperature humidity index scenarios in the Mediterranean basin. International Journal of Biometeorology, 57, 451–458, Doi https://doi.org/10.1007/s00484-012-0571-5.
Seré, C., van der Zijpp, A., Persley, G. and Rege, E., 2008. Dynamics of livestock production systems, drivers of change and prospects for animal genetic resources. Animal Genetic Resources Information, 42, 3-24. DOI: https://doi.org/10.1017/S1014233900002510
Sinsin, B., Kampmann D., (eds)., 2010: Biodiversity Atlas of West Africa, Volume I: Benin. Cotonou and Frankfurt/Main
Szabolcs, B., Nagy, B., Nagy, L. and Kiss, B., 2007. Comparison of body measurements of beef cows of different breeds. Archiv fur Tierzucht, Dummerstorf, 50(4), 363–3. https://doi.org/10.5194/aab-50-363-2007.
Tenagne, A., Mekuriaw, G. and Kumar, D., 2016. Phenotypic Characterization of Indigenous Cattle Populations in West Gojjam Administrative Zones, Amhara National Regional State. Ethiopia. Journal of Life Science and Biomedicine, 6(6), 127–138. www.jlsb.science-line.com. Accessed 24 Feb 2017.
Terefe, E., Haile, A., Mulatu, W., Dessie, T. and Mwai O., 2015. Phenotypic characteristics and trypanosome prevalence of Mursi cattle breed in the Bodiand Mursi districts of South Omo Zone, southwest Ethiopia. Tropical Animal Health and Production, 47, 485–493. https://doi.org/10.1007/s11250-014-0746-7.
Traoré, A., Koudandé, D.O., Fernandez, I., Soudré, A., Granda, V., Alvarez, I., Diarra, S., Diarra, F., Kaboré, A., Sanou, M., Tamboura, H.H. and Goyache, F., 2015. Geographical assessment of body measurements and qualitative traits in West African cattle. Tropical Animal Health and Production, 47(8), 1505–1513. Doi: https://doi.org/10.1007/s11250-015-0891-7
Traoré, A., Koudandé, O.D., Fernández, I., Soudré, A., Álvarez, I., Diarra, S., Diarra, F., Kaboré, A., Sanou, M., Tamboura, H.H. and Goyache, H., 2016. Multivariate characterization of morphological traits in West African cattle sires. Archives Animal Breeding, 59, 337–344. Doi: https://doi.org/10.5194/aab-59-337-2016.
Weyl, P.S.R. and Coetzee, J.A., 2016. Morphological variations in southern African populations of Myriophyllum spicatum: Phenotypic plasticity or local adaptation? South African Journal of Botany, 103, 241–246. https://doi.org/10.1016/j.sajb.2015.07.016
Yakubu, A., Hingir, A.V. and Abdullah, A.R., 2018. Multivariate analysis of sexual dimorphism in the morphometric traits of Muturu cattle in North Central Nigeria. Nigerian Journal of Genetics, 32, 8–15.
Acknowledgments
The authors are grateful to all the cattle farmers for their valuable help and collaboration during the field work.
Funding
This study was funded by the German Volkswagen Foundation (grant number Az 89367).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical standards
The manuscript does not contain clinical studies or patient data.
Informed consent
Informed consent was obtained from all individual participant Lagune Cattle farmers included in this study.
Statement of animal rights
Body measurements and morphological description were obtained with the permission of the farmers. The measured animals were managed by the farmers themselves. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ahozonlin, M.C., Dossa, L.H., Dahouda, M. et al. Morphological divergence in the West African shorthorn Lagune cattle populations from Benin. Trop Anim Health Prod 52, 803–814 (2020). https://doi.org/10.1007/s11250-019-02071-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-019-02071-1