Abstract
Purpose
The main purpose of this meta-analysis was to evaluate the impact of beta-thalassemia major (BTM) on the health-related quality of life as assessed by the medical outcomes short-form-36 questionnaire (SF-36).
Methods
A systematic literature search was performed on Cochrane library, Web of Science, Scopus, Science Direct, ProQues, Medline/PubMed, Scholar Google until March 17, 2017 to obtain eligible studies. A fixed effect model was applied to summarize the scores of each domain. The radar chart was used to compare the scores of BTM patients with other health conditions. Spearman’s correlation analysis and meta-regression were used to explore the related factors.
Results
26 studies were included in this study, which were all reliable to summarize the scores of the SF36. Pooled mean scores of the physical health domains ranged from 52.74 to 74.5, with the GH and PF domains being the lowest and the highest, respectively. Further, the pooled mean scores of the mental health domains varied between 59.6 and 71.11, with the (MH-VT) and SF domains being the maximum and the minimum, respectively. Patients with BTM had a substantially compromised HRQoL in comparison with the general population.
Conclusion
BTM could adversely affect the HRQoL of patients. Measuring HRQoL should be considered as an essential part of the overall assessment of health status of BTM patients, which would provide valuable clues for improving the management of disease and making decisions on the treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Beta-thalassemia major (β-TM) is considered as an inherited hemoglobinopathy which can be passed to a person following mutations in the genes constructing the beta-globin chain [1]. Given the fact that the thalassemia heterozygous form is resistant to malaria, the prevalence of thalassemia minor in malaria-prone areas is reported to be high and thus the thalassemia gene can be transmitted to next generations [2, 3]. The thalassemia belt regions also encompass the Mediterranean nations (Southern European countries including Italy, Greece, and Cyprus), the Arabian Peninsula, as well as parts of Africa, Turkey, Iran, India, South-east Asia especially Thailand, Cambodia, and Southern China. The prevalence rates of thalassemia-related genetic disorders in these areas are estimated to be 2.5–15%. In general, about 3% of the world’s population is known to be carriers of beta-thalassemia gene [4]. According to the recent statistics from Iran, 18,600 are affected by β-TM in this country. Meanwhile, the Thalassemia Prevention Program was conducted as a pilot study in Iran in 1991, and then was implemented nationwide in 1997 [5]. Further, this program has been able to prevent 80% of the births of β-TM neonates and 53% of the two carriers of the thalassemia genes [6]. Furthermore, it has played an important role in diminishing 65% of β-TM births in thalassemia belt regions [7].
The β-TM typically occurs at the second six months of life in the form of severe and advanced hemolytic anemia. The most common therapeutic method is regular blood transfusion. However, this treatment is likely to increase iron overload where theexcess iron cannot naturally excrete from the body. In response, this accumulation of iron can cause delays in growth and sexual maturation of myocardial infarction, arrhythmia, liver damage (fibrosis and cirrhosis), diabetes, hypogonadism, hyperparathyroidism, and thyroid failure. On the other hand, the complications of blood transfusion such as the transmission of hepatitis B and C virus infection can be raised as another problem confronted by β-TM patients [8]. Estimating serum ferritin levels is also known as one of the main tests for increased iron overload in patients with β-TM in which levels of serum ferritin usually reach 1000 mg/l after 10–12 times of blood transfusion which can be a starting point for chelating agents [9]. There are three iron chelators: deferoxamine (DFO) which requires subcutaneous or intravenous injection [10]: it can be time-consuming and painful and should be injected within 8–12 h, and sometimes leads to treatment noncompliance [11]; The other options are deferiprone (DFP) and deferasirox (DFX) as oral chelating agents which can be absorbed in the gastrointestinal tract [10]. It seems that advances in the implementation of iron chelation therapy (ICT) and the introduction of edible chelators as well as the comfort and tolerance of these medications have led to patient satisfaction with ICT as well as improved health-related quality of life (HRQoL) [12]. In this respect, the study by Abdul-Zahra et al. (2016) in Iraq showed that DFX could have a significant impact on enhancing HRQoL in patients with β-TM compared to DFO and combined therapy (DFO + DFX) [13].
The need for regular blood transfusions as well as difficult regimens for subcutaneous injection of chelating agents have also created an overwhelming situation for patients with thalassemia and their families. Compared with their healthy peers, these patients suffer from lower HRQoL. The duration of the disease and the length of the course of treatment, frequent hospitalizations, increased medical costs, along with mental-psychological and social damage can significantly trigger stress in these patients and their families. Over the past 30 years, the introduction of procedures such as regular blood transfusion and the use of oral chelating agents have considerably improved the HRQoL in these patients [14].
A mutual relationship can be also observed between the disease and the HRQoL particularly in individuals with chronic illnesses who are affected by multiple problems throughout their life. Under such situations, physical differences and symptoms can have a direct effect on all dimensions of HRQoL. The β-TM can similarly make constant changes in a person’s life (need for self-care, regular and continuous blood transfusions, daily intake of chelators, regular monitoring of serum ferritin levels as well as other specialized tests, etc.) and thus have an impact on HRQoL.
The HRQoL is a multi-dimensional concept and is related to the specific impact of a disease or an illness on the quality of life (QoL). HRQoL is also considered as a procedure in which the empirical effect of health is used to estimate QoL. Given the current significant advances in medical science along with the reduction of mortality from chronic diseases such as thalassemia which require a lifelong management, HRQoL has become an important aspect for providing full healthcare in addition to the survival of patients. It can offer valuable information for optimal management of the disease and also play an important role in deciding on treatments for patients with β-TM. Accordingly, the objectives of healthcare services are not only focused on attempts to help patients survive, but also to increase HRQoL as a central goal wherein all attention is paid to the patient’s well-being [15].
Numerous studies have been conducted worldwide to assess HRQoL in β-TM patients. However, HRQoL has been examined only in some of these investigations in a limited context with a small sample size [16] which cannot provide transparent and useful information for large-scale decision-making and policy-making. Therefore, considering the prevalence of this disease for global policy-making, a general estimate of HRQoL in these patients is of utmost importance which can result in a more complete picture of the dimensions of HRQoL in patients suffering from β-TM [17]. In this respect, the HRQoL 36-Item Short-Form Health Survey (SF-36) has been widely used as a general research tool to assess health status. This questionnaire is comprised of 36 items categorized into 8 domains in which the scores obtained can be placed into two general components: physical component summary (PCS) and mental component summary (MCS). The SF-36 is a common tool for assessing the HRQoL which has been employed for both general populations as well as those with various diseases, especially in β-TM patients [18]. This makes it possible to compare HRQoL among people with different illnesses and various health conditions. In this study, attempts were made to use one of the novel and powerful research methods i.e., meta-analysis to explore and combine the results of research studies conducted in the field of HRQoL among β-TM patients. In this regard, combining numerical results from several studies as along with accurate estimation of descriptive indicators can make it possible to explain inconsistencies in the body of research findings. Accordingly, the purpose of the present study was to investigate HRQoL in β-TM patients using a meta-analysis design.
Methods
The SF36 health survey questionnaire
The present study was a meta-analysis of HRQoL and its dimensions based on the results of the SF-36 in patients affected by β-TM. The SF-36 is considered as a standard measurement tool for assessing HRQoL used worldwide, and first developed by Ware and Sherbourne (1992) in the United States. The 36 items in this questionnaire are summarized into 8 domains: (1) physical functioning (PF) with items 3 to 11 associated with ability to perform daily life activities such as caring for individual needs, walking, as well as relaxation; (2) role limitations due to physical problems (RP) including items 12 to 15 related to activities limiting physical abilities and focused on role limitations due to physical problems; (3) bodily pain (BP) (items 20 and 21) assessing the level of pain during the past four weeks and its interference with daily activities; (4) general health (GH) including items 1, 33, 34, 35, 36 assessing general health based on individual perceptions; (5) vitality (VT) with items 22, 26, 28, 30 being concerned with the feeling of agility and energy as well as tiredness; (6) social functioning (SF) encompassing items 19 and 31 concentrating on the effect of physical and mental problems on social activities and relationships with family, friends, and other community members; (7) role limitations due to emotional problems (RE) with items 16, 17, and 18 evaluating the emotional factors interfering with work or other activities and dealing with role limitations due to emotional problems that occur; and (8) mental health (MH) including items 23, 24, 25, 27, 29 measuring feelings of depression and anxiety. To score the items in any domain, the scores of the items are summed up and then converted into a scale from 0 (worst) to 100 (best). The scores in these 8 domains can be reduced to two general components of PCS and MCS [18, 19].
Search strategy
The mentioned questionnaire had been already translated into different languages, whose validity and reliability had been determined across various populations for evaluation and use. In order to achieve the related documents in Persian and in English, a literature search was also performed in the databases of Cochrane Library, Web of Science, Scopus, ScienceDirect, ProQuest, MEDLINE/PubMed, and other sources such as Google Scholar, Islamic World Science Citation Center (ISC), and Scientific Information Database (SID) with no time limits for publication until March 17, 2017. The search strategies in the MEDLINE/PubMed database and the keywords used were as follows:
Quality of Life OR SF-36 OR Short Form-36 OR Medical Outcomes Study Short-Form 36 Health Survey OR Short Form Health Survey 36 OR SF-36 Health Survey OR 36-Item Short-Form Health Survey OR Medical Outcomes Short Form-36 Questionnaire AND Thalassemia OR Thalassemia Major OR Beta Thalassemia OR Beta Thalassemia Major OR β-TM.
Inclusion criteria
The studies examined in this meta-analysis had met the following criteria: they had (1) used a case-control, longitudinal cohort, or cross-sectional research design, (2) investigated HRQoL in β-TM patients via the SF-36, and (3) reported detailed information related to the eight domains mentioned or the two summary scores from the SF-36 in the form of means and standard deviations (SD) or standard errors (SE). On the other hand, studies which had not met the inclusion criteria in the present study as well as those examining HRQoL in patients with β-TM, thalassemia intermedia, and sickle cell anemia with their results reported in combination were excluded.
Selection procedure
Of the total number of 1007 studies searched, 459 cases were omitted due to being duplicates. Then, the titles and abstracts of 548 studies were reviewed and 431 cases were crossed out as they lacked the terms HRQoL and SF-36 in their titles and abstracts. After that, the full texts of 124 articles were examined by two researchers based on the inclusion and exclusion criteria in the present study. Consequently, 85 and 9 cases were removed due to irrelevancy of their results to the SF-36 and the combination of β-TM with sickle cell anemia, respectively. Further, 4 studies were crossed out due to combining β-TM with other types of thalassemia. Finally, 26 articles were selected and introduced into the quality assessment phase (Fig. 1).
Studies that had been eliminated due to the combination of the results of β-TM patients with other types of thalassemia included the investigation by Waheed, Dahlui et al., Hajibeigi et al., and Azarkeivan et al. [7, 20,21,22].
Quality assessment
The quality of studies was assessed by Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist designed as a statement to enhance the reporting of observational studies [23].
Data extraction
The search, evaluation, and extraction of the data were performed independently by two researchers. The agreement between the search results as well as any difference of opinions between the two researchers was also investigated by a third person. In this respect, studies whose full texts were not available were retrieved. In case two studies had been conducted in a center, both evaluators and even the third person believed that both articles were required to be included in the present meta-analysis even though the age groups of their participants were reportedly different. The data extracted from each study included the name of the first author, year of publication, country of origin, study design, sample size, average ferritin levels, mean age of patients, gender, and scores of the eight domains of the SF-36 (PF, RP, BP, GH, VT, SF, RE, and MH) and summary scores of the two components of MCS and PCS.
Data synthesis and statistical analysis
The pooled mean scores of the eight domains and the two summary scores were reported with confidence intervals (CI) of 95%. To evaluate the heterogeneity of the given studies, Cochran’s Q test and I2 index were used (I2 Index less than 25% showed low heterogeneity, the value between 25% and 75% indicated moderate heterogeneity, and the index greater than 75% represented high heterogeneity). The I2 values less than 50% also indicated a low variance between studies; thus, fixed effects model and inverse variance method were employed in such cases. Otherwise, instrumental variable (IV) heterogeneity method was used [24]. The effect size with a CI of 95% was also obtained using the metan package of the Stata Software (Version 11.0) [25]. Since the eight domains of the SF-36 represented different dimensions of HRQoL, each domain was separately summarized and analyzed in this meta-analysis. In addition, the two summary scores i.e., MCS and PCS were reported to generate a straightforward understanding of HRQoL.
The radar chart was also utilized to compare the effects of different types of chelating agents including DFP, DFX, DFO, and combination therapy namely DFO + DFP or DFX + DFO on HRQoL. Further, this chart was used to compare the dimensions of HRQoL in β-TM patients with those of other health conditions. For this purpose, after searching in the databases, the researchers extracted the scores of the HRQoL dimensions from the SF-36, derived from studies on populations in Norway and the United Kingdom [26, 27]. On the other hand, since chronic illnesses could influence HRQoL in general populations and the impact of diabetes as a chronic disease on HRQoL had been also confirmed by previous researchers, the effect of type 2 diabetes on HRQoL scores obtained from the SF-36 in the study by Clarke et al. was used for comparisons in this meta-analysis [28]. Previously, Yang et al. [29] employed the scores of HRQoL dimensions obtained from the SF-36 in populations in Norway and the United Kingdom and the scores of type 2 diabetes to compare them with the scores of the HRQoL dimensions resulting from the SF-36 in patients suffering from ankylosing spondylitis [26, 28].
The present study also compared the scores and domains of the SF-36 in the given investigations based on country of origin, year of publication, mean age, sample size, percentage of women, as well as average ferritin levels. Regarding the country of origin, the studies were placed into two sub-groups of upper-middle income and high-income nations according to their national economic level as obtained by the online databases of the World Bank. Considering the year of publication, these studies were categorized into two sub-groups of articles published in 2012 and before that and those published after 2012. The studies were also assigned into two sub-groups based on their sample size i.e., articles with 100 individuals and fewer and those with over 100 individuals.
Results
Characteristics of the studies
Among the 26 articles published between 2008 and 2016, a total number of 2961 β-TM patients had been evaluated in terms of HRQoL including 1603 and 1269 female and male patients, respectively. The number of women and men had not been reported only in three studies; Zarea, Baraz, and Zahmatkeshan [30,31,32]. The mean age of the patients recruited in these 26 studies was 21.79 years and the interquartile range (IQR) was equal to 19.25–24.63. Table 1 outlines some data derived from the studies included in the present meta-analysis.
Overall HRQoL of β-TM patients
The results of I2 index suggested weak heterogeneity of the studies included in this meta-analysis, where the I2 index for each domain of SF-36 and MCS was 0%, which is less than 5.1% for PCS. Also, the final score of the HRQOL was equal to 15.3% as reported in Table 2. Hence, the fixed effect model was used. Table 2 provides the results of the pooled mean scores with the CI of 95% for eight domains of the SF-36 as well as summary scores of the two components of MCS and PCS.
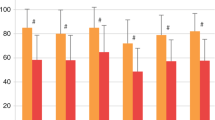
The pooled mean score of the PCS including PF, RP, BP, and GH domains was 56.77. The pooled mean scores of the PCS also ranged from 52.74 to 74.5 with the minimum and maximum scores belonging to the GH and the PF domains, respectively. Further, the pooled mean score for the MCS with the domains of VT, SF, RE, and MH was 56.57. The pooled mean score for the MCS also varied from 59.6 to 71.11. The minimum scores belonged to MH and VT, while the maximum value was observed for SF. Figure 2 demonstrates the pooled mean scores for the SF-36, MCS, and PCS.
Spearman’s rank-order correlation analysis and meta-regression results
Table 2 presents the results of Spearman’s rank-order correlation. According to this table, the overall score of the SF-36 was not significantly correlated with the economic conditions of the countries (r = − 0.2, P = 0.34). Moreover, the results of the independent t test indicated that the HRQoL pooled mean scores in upper-middle-income and high-income nations were 64.92 ± 7 and 57.71 ± 12.56; respectively. The mean age in countries with upper-middle income and high income was 20.15 ± 3.1 and 30.39 ± 9.17, respectively. According to the results of the independent t test, the overall score obtained from the SF-36 in Iran was not statistically and significantly different compared to those in other countries (r = − 0.2, P = 0.26). The pooled mean scores of HRQoL in Iran and other countries were found to be 64.64 ± 7.15 and 60.44 ± 11.42, respectively. The mean age of patients in Iran was 21 ± 2.15 years, while that of other countries was 26.02 ± 10 years .
Furthermore, the results of Pearson’s product-moment analysis for six studies reporting serum ferritin levels indicated a strongly negative relationship between the PCS and serum ferritin levels (r = − 0.864, P = 0.027). Also, the results of univariate meta-regression analysis revealed the effect of increased serum ferritin levels on PCS (Fig. 3).
Relative HRQoL condition of β-TM patients
The radar chart (Fig. 4a) compares the HRQoL dimensions in β-TM patients with general populations in Norway and the United Kingdom and those with type 2 diabetes (in the United Kingdom). In each of the 8 dimensions, patients with thalassemia were closer to the center compared to the general populations showing poorer HRQoL status in patients with β-TM compared to that in general populations. The results of the independent t test to compare β-TM patients and general population in Norway in terms of HRQoL and its dimensions between showed p-values of 0.049, 0.14, 0.13, 0.005, 0.75, 0.01, 0.017, 0.005, 0.018, 0.013, and 0.012 for MCS, PCS, PF, RP, BP, GH, VT, SF, RE, MH; respectively. There was also a significant difference in the PF, GH, SF, RE, MH, PCS, and MCS domains as well as the final score of HRQOL.
a Radar chart to compare the HRQoL dimensions in β-TM patients with general populations in Norway and the United Kingdom and patients with type 2 diabetes (in the UK). b Radar chart to compare the effect of chelating agents on HRQoL dimensions in β-TM patients. Deferoxamine (DFO; Desferal). Deferasirox (DFX; Exjade). Deferiprone (DFP,L1). Combination Therapy (DFO + DFP) or (DFX + DFO)
The radar chart also (Fig. 4b) displays the HRQoL dimensions in patients with thalassemia using chelation therapies (DFO, DFX, combination therapy) based on three studies in this field [13, 16, 47]. The HRQoL status of the consumers of DFO and combination therapy across all 8 domains was much closer to the center than that of DFX, indicating poorer HRQoL status with all its dimensions in consumers of DFO and combination therapy compared to DFX users. The mean scores of the HRQoL in DFO, combination therapy, and DFX consumers were reported as 56.93, 61.04, and 70.63, respectively.
Publication bias assessment
Begg’s test and Egger’s test were proposed to examine publication bias. The power rate of the Egger’s test has been reported to be greater than that of the Begg’s test such that it can highlight the possibility of publication bias faster. So, the publication bias in the present study was assessed using the Egger’s test. In all cases, except for GH, MH, PCS, and HRQOL, the p-value was more than 0.05. In the above cases, this value was 0.002, 0.01, 0.002, and 0.0001 respectively suggesting a potential publication bias. Further, graphical funnel plots were analogous in other domains and showed no evidence of bias (Fig. 5).
Discussion
The present meta-analysis aimed to systematically evaluate the effect of β-TM on HRQoL using the SF-36 and also identify factors affecting HRQoL in these patients. Examining the heterogeneity of the studies included in the present met-analysis showed the lowest levels of heterogeneity. Investigating the eight dimensions of HRQoL similarly revealed that β-TM had a negative effect on all dimensions. Although several plans have been proposed for the care and treatment of these patients across all countries located in the thalassemia belt, the low scores of HRQoL compared to those of general populations suggested that further planning is required to focus on HRQoL promotion in these patients.
Further analysis on the economic status of the countries showed a contradiction. The HRQoL scores in upper-middle-income countries were reported to be higher than those in high-income counterparts. However, the mean age of these patients in high-income countries was reported to be approximately 10 years older than that of individuals in upper-middle-income nations. The overall score of HRQoL obtained in Iran was similarly higher compared to that in other countries, but the mean age of patients in Iran was 5 years lower than that in other nations.
Meanwhile, life satisfaction and life expectancy are considered of utmost importance in terms of promoting HRQoL in patients with β-TM, both of which can lead to prolonged patient survival as one of the most important therapeutic goals. It seems that promoting the levels of care and treatment in high-income countries has had an effective role in increasing the life expectancy in β-TM patients. The higher mean age in high-income countries can also indicate these countries’ attention to the issues of tertiary prevention. In this respect, Thalassemia Prevention Program is usually used based on the Clark prevention model in three stages of pre-disease, latent disease, and symptomatic disease [7, 52]. The initial pre-disease prevention interventions aim at reducing the number of cases from the pre-disease to the latent disease stage. Examples of this level of prevention include awareness, health education, genetic screening, and counseling. The secondary prevention also tries to reduce the number of cases from the latent disease to symptomatic disease stage or the birth of a baby affected by β-TM. Examples of the secondary prevention of β-TM include prenatal diagnosis and pre-implantation genetic diagnosis to prevent births of β-TM babies. The tertiary prevention interventions in the symptomatic disease stage also aim at reducing disabilities and facilitating patient rehabilitation and ultimately promoting HRQoL and longevity. Examples of the tertiary prevention include bone marrow transplantation, blood transfusion, chelation therapy, and management of complications, as well as access to medical facilities and equipment [7]. Until a few years ago, patients with β-TM used to die in the second decade of their life. However, fairly recently tremendous transformations have occurred in the treatment of these patients and there are therapeutic advances especially after identifying oral chelators and introducing allogeneic hematopoietic stem cell transplantation (HSCT) [53] along with novel drugs such as Luspatercept [54]. Currently, a relatively long life-span can be expected with these appropriate treatments. At the moment, the HSCT is the only treatment that results in complete remission of thalassemia. The study by Caocci et al. in 2016 also demonstrated that HSCT had led to a return to normal HRQoL as well as physical and mental satisfaction in β-TM patients [53]. Luspatercept (ACE-536) is a modified activin receptor type IIB fusion protein which promotes late-stage erythroid differentiation. The study by Piga et al. in 2016 also demonstrated that Luspatercept was generally safe and well-tolerated at dose levels up to 1.25 mg/kg with no related serious adverse events. Sustained hemoglobin increase in Non-transfusion-dependent (NTD) patients was associated with an improvement in quality of life. Sustained reduction in transfusion burden in Transfusion-dependent (TD) patients was associated with reductions in liver iron concentration (LIC) in patients with elevated baseline LIC [54].
As the most commonly used tool for these patients, since the SF-36 has also been introduced as a golden standard for assessing HRQoL [55], the present meta-analysis compared the HRQoL of patients with β-TM and that of general populations in Norway and the United Kingdom as well as a population with a chronic disease (diabetes) based on the SF-36. The results revealed that patients with β-TM had a lower HRQoL than other groups selected for this comparison. Considering the VT domain, β-TM patients were similar to those under comparison, but such patients with thalassemia had the same situation to that of diabetic individuals in the domains of PF, VT, and BP. The prevalence rate of diabetes mellitus in different communities in patients with thalassemia has been reported as 0–26%. It seems that even the prevalence of impaired glucose tolerance (IGT) is about five times more than that of diabetes. In this regard, two basic mechanisms can contribute to the development of IGT in β-TM patients following excess iron overload including (1) disorder in insulin secretions of pancreatic beta-cells which can be assumed as the major cause of diabetes in such patients and (2) insulin resistance that even occurs in the presence of normal functioning of beta-cells and may be seen before the onset of glucose intolerance or diabetes. Other mechanisms related to iron overload may also be involved in the development of diabetes in patients with thalassemia including reduced serum zinc levels, chronic liver disease, and increased prevalence of hepatitis C or genetic factors in patients with β-TM [56].
Other results which could be interpreted in this meta-analysis were associated with the serious effects of ferritin on diminishing the PCS within HRQoL in these patients. The ferritin levels had been reported in six of studies examined in this meta-analysis. Accordingly, the increase in iron overload induced by regular transfusions could add to the accumulation of iron and result in heightened ferritin levels which could have numerous physical effects as well as possible direct impacts on the PCS [57].
The very impressive effect of the type of chelators on HRQoL and its dimensions was also notable. In patients receiving the DFX, the prevalence of myocardial infarction was lower since the DFX was able to prevent myocardial iron deposition [58]. On the other hand, patient compliance to treatment with the DFX was higher due to satisfaction with the use of this agent [59]. Note that compliance is considered as the most important parameter for successful treatment. Indeed, low compliance in patients to the DFO can be associated with lack of clinical benefits [60] and self-injection difficulties [61]. According to the results of previous studies, the DFO recipients are likely to suffer from depression, fatigue, and decreased physical functioning [62]. On the other hand, the DFO can limit daily exercises, movements, and physical activities and consequently reduce patient self-esteem [17]. These issues can potentially have a negative effect on HRQoL.
In general, this study presented a summary of the quantitative data obtained from HRQoL in a number of β-TM patients across the world via exact reassessment methods methods. However, there were some limitations which warrant a cautious interpretation of the results.
In the present meta-analysis, only the studies published in Persian and in English were included but some investigations had not mentioned the exact sampling methods and also the factors affecting HRQoL had not been highlighted in all these articles. Due to some constraints on the related literature, HRQoL in the given β-TM patients was compared with those in general populations in Norway and the United Kingdom and then with the results of HRQoL in patients suffering from β-TM. Highlighting the importance of investigating ferritin levels in the patient’s conditions, it was found that the ferritin levels had been analyzed and reported in only six studies.
Conclusion
The present study was conducted to investigate the effect of β-TM on HRQoL. The results suggested that β-TM had a significantly negative effect on physical and mental health status, so assessing HRQoL was recognized as an indispensable indicator for a general evaluation of patients with thalassemia through which valuable evidence could be provided to improve treatments and to make effective decisions.
It was assumed that high-income countries had been planning more balanced programs. They had also well managed to increase longevity and life expectancy in these patients via advanced therapies such as the HSCT and access to the novel drug such as luspatercept which have totally revolutionized the treatment with possible improvement of HRQoL. The lower HRQoL in these countries in contradiction with that in other upper-middle-income nations could be due to inappropriate evaluation of HRQoL according to SF-36 in patients with thalassemia. Although many studies had been using this tool for β-TM patients, it seemed that the utilization of a specialized tool for assessing HRQoL in patients with β-TM was necessary.
References
Caocci, G., La Nasa, G., D’Aloja, E., Vacca, A., Piras, E., Pintor, M., et al. (2011). Ethical issues of unrelated hematopoietic stem cell transplantation in adult thalassemia patients. BMC Medical Ethics, 12(1):4.
Kuesap, J., Chaijaroenkul, W., Rungsihirunrat, K., Pongjantharasatien, K., & Na-Bangchang, K. (2015). Coexistence of malaria and thalassemia in malaria endemic areas of Thailand. The Korean Journal of Parasitology, 53(3), 265.
Weatherall, D. (1997). Thalassaemia and malaria, revisited. Annals of Tropical Medicine & Parasitology, 91(7), 885–890.
Vichinsky, E. P., MacKlin, E. A., Waye, J. S., Lorey, F., & Olivieri, N. F. (2005). Changes in the epidemiology of thalassemia in North America: a new minority disease. Pediatrics, 116(6), e818–e825.
Hassanzadeh, J., Mirahmadizadeh, A., Karimi, M., & Rezaeian, S. (2017). Delay in diagnosis of hemoglobulinopathies (thalassemia, sickle cell anemia): A need for management of thalassemia programs. Iranian Journal of Pediatrics, 27(2), e6740.
Safizadeh, H., Farahmandinia, Z., Nejad, S. S., Pourdamghan, N., & Araste, M. (2012). Quality of life in patients with thalassemia major and intermedia in kerman-iran (I.R.). Mediterranean Journal of Hematology & Infectious Diseases, 4(1), e2012058.
Waheed, F. (2015). Thalassaemia prevention in Maldives: Effectiveness of primary, secondary and tertiary prevention interventions. Crawley: The University of Western Australia.
Tari, K., Valizadeh Ardalan, P., Abbaszadehdibavar, M., Atashi, A., Jalili, A., & Gheidishahran, M. (2018). Thalassemia an update: Molecular basis, clinical features and treatment. International Journal of Biomedicine and Public Health, 1(1), 48–58.
Donovan, A., Lima, C. A., Pinkus, J. L., Pinkus, G. S., Zon, L. I., Robine, S., et al. (2005). The iron exporter ferroportin/Slc40a1 is essential for iron homeostasis. Cell Metabolism, 1(3), 191–200.
Rachmilewitz, E. A., & Giardina, P. J. (2011). How I treat thalassemia. Blood, 118(13), 3479–3488.
Nisbet-Brown, E., Olivieri, N. F., Giardina, P. J., Grady, R. W., Neufeld, E. J., Séchaud, R., et al. (2003). Effectiveness and safety of ICL670 in iron-loaded patients with thalassaemia: A randomised, double-blind, placebo-controlled, dose-escalation trial. The Lancet, 361(9369), 1597–1602.
Porter, J., Bowden, D. K., Economou, M., Troncy, J., Ganser, A., Habr, D., et al. (2012). Health-related quality of life, treatment satisfaction, adherence and persistence in β-thalassemia and myelodysplastic syndrome patients with iron overload receiving deferasirox: Results from the EPIC clinical trial. Anemia. https://doi.org/10.1155/2012/297641.
Abdul-Zahra, H. A. I., Hassan, M. K., & Ahmed, B. A. A. H. (2016). Health-related quality of life in children and adolescents with β-thalassemia major on different iron chelators in Basra, Iraq. Journal of Pediatric Hematology/Oncology, 38(7), 503–511.
Telfer, P., Constantinidou, G., Andreou, P., Christou, S., Modell, B., & Angastiniotis, M. (2005). Quality of life in thalassemia. Annals of the New York Academy of Sciences, 1054(1), 273–282.
Klaassen, R., Alibhai, S., & Moreau, K. (2013). Introducing the TranQol: A new disease-specific quality of life measure for children and adults with thalassemia major. Journal of Blood Disorders and Transfusion. https://doi.org/10.4172/2155-9864.1000150.
Senol, S. P., Tiftik, E. N., Unal, S., Akdeniz, A., Tasdelen, B., & Tunctan, B. (2016). Quality of life, clinical effectiveness, and satisfaction in patients with beta thalassemia major and sickle cell anemia receiving deferasirox chelation therapy. Journal of Basic & Clinical Pharmacy, 7(2), 49–59.
Abetz, L., Baladi, J.-F., Jones, P., & Rofail, D. (2006). The impact of iron overload and its treatment on quality of life: Results from a literature review. Health and Quality of Life Outcomes, 4(1), 73.
Hachim, I. Y., Al Nuaimi, Y. E., Abd El Baky, A., Al Hashmi, F., Rahman, A., Mazroei, S., et al. (2014). Quality of life in patients suffering from thalassaemia in Ras Al Khaimah, United Arab Emirates. Hamdan Medical Journal, 212, 1–7.
Ware, J. E. Jr., & Sherbourne, C. D. (1992). The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Medical Care, 30, 473–483.
Azarkeivan, A., Hajibeigi, B., Alavian, S. M., Lankarani, M. M., & Assarf, S. (2009). Associates of poor physical and mental health-related quality of life in beta thalassemia-major/intermedia. Journal of Research in Medical Sciences, 14(6), 349–355.
Dahlui, M., Hishamshah, M. I., Rahman, A. J. A., & Aljunid, S. M. (2009). Quality of life in transfusion-dependent thalassaemia patients on desferrioxamine treatment. Singapore Medical Journal, 50(8), 794–799.
Hajibeigi, B., Azarkeyvan, A., Alavian, S. M., Lankarani, M. M., & Assari, S. (2009). Anxiety and depression affects life and sleep quality in adults with beta-thalassemia. Indian Journal of Hematology and Blood Transfusion, 25(2), 59–65.
Vandenbroucke, J. P., von Elm, E., Altman, D. G., Gotzsche, P. C., Mulrow, C. D., Pocock, S. J., et al. (2007). Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. Annals of Internal Medicine, 147(8), W163–W194.
Higgins, J. P., Thompson, S. G., Deeks, J. J., & Altman, D. G. (2003). Measuring inconsistency in meta-analyses. BMJ, 327(7414), 557–560.
Harris, R., Bradburn, M., Deeks, J., Harbord, R., Altman, D., & Steichen, T. (2010). Module for fixed and random effects meta-analysis. Boston College: Statistical software Components. Department of Economics.
Jenkinson, C., Coulter, A., & Wright, L. (1993). Short form 36 (SF36) health survey questionnaire: Normative data for adults of working age. BMJ: British Medical Journal, 306(6890), 1437–1440.
Ribu, L., Hanestad, B. R., Moum, T., Birkeland, K., & Rustoen, T. (2007). A comparison of the health-related quality of life in patients with diabetic foot ulcers, with a diabetes group and a nondiabetes group from the general population. Quality of Life Research, 16(2), 179–189.
Clarke, P. M., Simon, J., Cull, C. A., & Holman, R. R. (2006). Assessing the impact of visual acuity on quality of life in individuals with type 2 diabetes using the short form-36. Diabetes Care, 29(7), 1506–1511.
Yang, X., Fan, D., Xia, Q., Wang, M., Zhang, X., Li, X., et al. (2016). The health-related quality of life of ankylosing spondylitis patients assessed by SF-36: A systematic review and meta-analysis. Quality of Life Research, 25(11), 2711–2723.
Baraz, S., Miladinia, M., & Mosavinouri, E. (2016). A comparison of quality of life between adolescences with beta thalassemia major and their healthy peers. International Journal of Pediatrics, 4(1), 1195–1204.
Zahmatkeshan, N., Mobasser, N., & Zamanzadeh, V. (2016). Quality of life in thalassemia major patients in an Iranian district. Global Journal of Health Science, 9(5), 266.
Zarea, K., Baraz Pordanjani, S., Pedram, M., & Pakbaz, Z. (2010). Quality of life in children with thalassemia who referred to Thalassemia Center of Shafa Hospital. Jundishapour University of Medical Sciences Journal, 8(4), 455–462.
Emadi, D., Rasooli, L., & Mohammadi Farsani, S. (2016). Quality of life among patients with beta-thalassemia major in Shahrekord city, Iran. International Journal of Epidemiologic Research, 3(4), 324–328.
Adib-Hajbaghery, M., & Ahmadi, M. (2015). Health related quality of life, depression, anxiety and stress in patients with beta-thalassemia major. Iranian Journal of Pediatric Hematology & Oncology, 5(4), 193–205.
Amani, F., Fathi, A., Valizadeh, M., Farzaneh, E., & Fattahzadeh-Ardalani, G. (2015). Quality of life among Ardabil patients with beta-thalassemia major. International Journal of Research in Medical Sciences, 3(11), 3308–3312.
Javanbakht, M., Keshtkaran, A., Shabaninejad, H., Karami, H., Zakerinia, M., & Delavari, S. (2015). Comparison of blood transfusion plus chelation therapy and bone marrow transplantation in patients with beta-thalassemia: Application of SF-36, EQ-5D, and visual analogue scale measures. International Journal of Health Policy and Management, 4(11), 733–740.
Khairkhah, F., Mahmoodi Nesheli, H., Khodabakhsh, E., & Hosseini, S. R. (2015). Evaluation of mental health and quality of life among β-thalassemia major patients. Caspian Journal of Pediatrics, 1(2), 54–59.
Mahmoud, M. A. M. Health related quality of life in Thalassemia patients treated by iron chelation therapy in the United arab Emirates 2015.
Vafaei M., Azad, M., Shiargar, P., & Kazemi Haki, B. (2015). Quality of life in patients with thalassemia major referred to Ardabil Buali Hospital in 2012-13. Medical Sciences Journal, 25(4), 305–310.
Amoudi, A. S., Balkhoyor, A. H., Abulaban, A. A., Azab, A. M., Radi, S. A., Ayoub, M. D., et al. (2014). Quality of life among adults with beta-thalassemia major in western Saudi Arabia. Saudi Medical Journal, 35(8), 882–885.
Klaassen, R. J., Barrowman, N., Merelles-Pulcini, M., Vichinsky, E. P., Sweeters, N., Kirby-Allen, M., et al. (2014). Validation and reliability of a disease-specific quality of life measure (the TranQol) in adults and children with thalassaemia major. British Journal of Haematology, 164(3), 431–437.
Yengil, E., Acipayam, C., Kokacya, M. H., Kurhan, F., Oktay, G., & Ozer, C. (2014). Anxiety, depression and quality of life in patients with beta thalassemia major and their caregivers. International Journal of Clinical and Experimental Medicine, 7(8), 2165–2172.
Gollo, G., Savioli, G., Balocco, M., Venturino, C., Boeri, E., Costantini, M., et al. (2013). Changes in the quality of life of people with thalassemia major between 2001 and 2009. Patient Preference and Adherence, 7, 231–236.
Haghpanah, S., Nasirabadi, S., Ghaffarpasand, F., Karami, R., Mahmood, M., Parand, S., et al. (2013). Quality of life among iranian patients with beta-thalassemia major using the SF-36 questionnaire. Sao Paulo Medical Journal, 131(3), 166–172.
Imani, E., Asadi Nooghabi, F., Hosseini Teshnizi, S., Yosefi, P., & Salari, F. (2013). Comparison quality of life in patients with thalassemia major based on participating in group activities, Bandar Abbas. Scientific Journal of Iran Blood Transfus Organ, 10(2), 198–206.
La Nasa, G., Caocci, G., Efficace, F., Dessi, C., Vacca, A., Piras, E., et al. (2013). Long-term health-related quality of life evaluated more than 20 years after hematopoietic stem cell transplantation for thalassemia. Blood, 122(13), 2262–2270.
Goulas, V., Kourakli-Symeonidis, A., & Camoutsis, C. (2012). Comparative effects of three iron chelation therapies on the quality of life of greek patients with homozygous transfusion-dependent Beta-thalassemia. Isrn Hematology Print, 2012, 139862.
Khani, H., Majdi, M. R., Azad Marzabadi, E., Montazeri, A., Ghorbani, A., & Ramezani, M. (2012). Quality of life of iranian β-thalassaemia major patients living on the southern coast of the caspian sea. Eastern Mediterranean Health Journal, 18(5), 539–545.
Musallam, K. M., Khoury, B., Abi-Habib, R., Bazzi, L., Succar, J., Halawi, R., et al. (2011). Health-related quality of life in adults with transfusion-independent thalassaemia intermedia compared to regularly transfused thalassaemia major: New insights. European Journal of Haematology, 87(1), 73–79.
Hadi, N., Karami, N., & Montazeri, A. (2009). Health-related quality of life in Major thalassemic patients. Payesh, 8(4), 387–393.
Messina, G., Colombo, E., Cassinerio, E., Ferri, F., Curti, R., Altamura, C., et al. (2008). Psychosocial aspects and psychiatric disorders in young adult with thalassemia major. Internal and Emergency Medicine, 3(4), 339–343.
Leavell, H. R. (1956). Applied preventive medicine (rheumatic fever and rheumatic heart disease). Annual Review of Medicine, 7(1), 489–498.
Caocci, G., Vacca, A., Piras, E., Serreli, V., Dessi, C., Marcias, M., et al. (2016). Return to normal life after hematopoietic stem cell transplantation for thalassemia: A study of patients transplanted from matched sibling donors. Bone Marrow Transplantation, 51(12), 1640.
Piga, A. G., Tartaglione, I., Gamberini, R., Voskaridou, E., Melpignano, A., Ricchi, P., et al. (2016). Luspatercept increases hemoglobin, decreases transfusion burden and improves iron overload in adults with beta-thalassemia. Blood, 128(22), 851.
Ware, J. E. (2000). Jr. SF-36 health survey update. Spine, 25(24), 3130–3139.
Bonakdaran, S., Khajeh-Dalouie, M., & Jalili-Shahri, J. (2010). Correlation between serum zinc level with impaired glucose tolerance and insulin resistance in major thalassemic patients. Iranian Journal of Endocrinology and Metabolism, 11(6), 667–672, 735.
Arian, M., Memarian, R., Vakilian, F., & Badiee, Z. (2013). Impact of an 8-week walking program on quality of life in patients with thalassemia major. Feyz Journal of Kashan University of Medical Sciences, 17(5), 463–470.
Pathare, A., Taher, A., & Daar, S. (2010). Deferasirox (Exjade®) significantly improves cardiac T2* in heavily iron-overloaded patients with β-thalassemia major. Annals of Hematology, 89(4), 405–409.
Taher, A., Al Jefri, A., Elalfy, M. S., Al Zir, K., Daar, S., Rofail, D., et al. (2010). Improved treatment satisfaction and convenience with deferasirox in iron-overloaded patients with β-thalassemia: results from the ESCALATOR trial. Acta Haematologica, 123(4), 220–225.
Borgna-Pignatti, C., Rugolotto, S., De Stefano, P., Zhao, H., Cappellini, M. D., Del Vecchio, G. C., et al. (2004). Survival and complications in patients with thalassemia major treated with transfusion and deferoxamine. Haematologica, 89(10), 1187–1193.
Pepe, A., Meloni, A., Capra, M., Cianciulli, P., Prossomariti, L., Malaventura, C., et al. (2010). Deferasirox, deferiprone and desferrioxamine treatment in thalassemia major patients: Cardiac iron and function comparison determined by quantitative magnetic resonance imaging. Haematologica, 2009, 019042.
Ismail, A., Campbell, M. J., Ibrahim, H. M., & Jones, G. L. (2006). Health related quality of life in Malaysian children with thalassaemia. Health and Quality of Life Outcomes, 4(1), 39.
Acknowledgements
The authors of this study express their gratitude and appreciate the support of the Vice-Chancellor’s Office for Research at Semnan University of Medical Sciences.
Funding
This study was conducted with the financial and spiritual support of Semnan University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors of this study declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Arian, M., Mirmohammadkhani, M., Ghorbani, R. et al. Health-related quality of life (HRQoL) in beta-thalassemia major (β-TM) patients assessed by 36-item short form health survey (SF-36): a meta-analysis. Qual Life Res 28, 321–334 (2019). https://doi.org/10.1007/s11136-018-1986-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-018-1986-1