Abstract
A single transcription factor is known to coordinate expression of a set of metabolites in a biochemical pathway; its use therefore can be a functional strategy in generating plants with desired traits. Triticum aestivum Dof1 (TaDof1) transcription factor is mainly associated with improved nitrogen assimilation in plants. In the current research, the transgenic wheat overexpressing TaDof1 transcription factor, under a constitutive promoter, was developed by Agrobacterium-mediated transformation. The two elite wheat cultivars (Galaxy and Faisalabad-2008) were selected for transformation study. The T0 plants were subjected to screening using selection medium containing herbicide BASTA. PCR results confirmed that only 8 out of 31 plants possessed the complete TaDof1 cassette. A transformation efficiency of 0.46% for Galaxy and 0.08% for Faisalaad-2008 was obtained. The quantitative RT-PCR was performed on T1 plants grown under nitrogen-limiting conditions. A substantial rise in the expression of citrate synthase (CS), isocitrate dehydrogenase (ICDH), phosphoenolpyruvate carboxylase (PEPC), and pyruvate kinase (PK) genes regulated by TaDof1 was observed after 4 weeks of nitrogen stress in T1 plants. The maximum fold increase of 464 was recorded for ICDH. Our findings indicate a cooperative modification of nitrogen and carbon metabolisms since they are intimately linked together. Overexpression of TaDof1 in wheat resulted in a significant increase in various agronomic traits. Furthermore, various physiological and biochemical markers (chlorophyll, protein, and soluble sugar contents) exhibited a profound change in TaDof1 transgenic plants in comparison with wild type plants. The results clearly depict the merits of employing transcription factors in engineering plant metabolisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nitrogen is a crucial macronutrient needed by the plants for their growth (Kumar et al. 2009). Plants need nitrogen in order to synthesize chlorophyll, amino acids, proteins, lipids, nucleic acids, and other metabolites (Liu et al. 2018). A large amount of nitrogen fertilizer is used to increase crop yield (Nosengo 2003). In the last 4 decades, the amount of nitrogen-containing fertilizers applied to crops has consistently increased (12 Tg/year to 104 Tg/year) resulting in better crop yield but with adverse effects on the environment (Mulvaney et al. 2009). According to an estimate only 40% of the applied nitrogen is available to the plant under optimum agronomic conditions, the rest is volatilized, denitrified, or immobilized in case of alkaline soils (Liu et al. 2014). Moreover, nitrogen fertilizers severely pollute the environment, especially the aquatic ecosystem (Nosengo 2003). The adverse effects of nitrogen in the environment include algal blooms (Vitousek et al. 2009; Wuebbles 2009), stratospheric ozone depletion, and global warming (Coruzzi and Zhou 2001). The increasing nitrogen fertilizer cost has resulted in demand for more nitrogen use efficient crops, i.e., crops that are better able to uptake, assimilate, and remobilize the nitrogen available to them without adding much fertilizer in the soil. In order to improve nitrogenous fertilizer use efficiency in crops, biotechnological methods hold a great promise for developing genetically engineered crops altered to enhance the desired agronomic traits (Sparks and Jones 2014).
Nitrogen and carbon assimilation utilizes dozens of different proteins and their coordinated expression within the roots and the entire plant vascular system. Often, changing the expression of a single gene in the nitrogen assimilation pathway does not significantly impact nitrogen use efficiency. Overproduction of glutamine synthetase (GS) in shoots of transgenic Lotus corniculatus L. (Vincent et al. 1997) and asparagine synthase in genetically modified tobacco (Brears et al. 1993) resulted in changes in ammonium assimilation in plants but did not increase the amino acid biosynthesis required for nitrogen utilization. Nitrogen assimilation, in addition to requiring inorganic nitrogen, also needs a carbon skeleton (2-oxoglutarate; 2-OG). The carbon compound, 2-OG, is the product of a series of reactions from photo-assimilated carbohydrates (Yanagisawa et al. 2004). The amounts of carbon and nitrogen-containing compounds mutually affect each other, suggesting an intimate link between carbon and nitrogen metabolisms (Stitt 1999; Coruzzi and Zhou 2001). Thus, manipulation of both carbon and nitrogen skeletons might be a functional strategy to enhance nitrogen utilization in plants (Yanagisawa and Sheen 1998; Kurai et al. 2011).
A single transcription factor may regulate several genes in a metabolic pathway; therefore, it is possible to modulate multiple gene expression using transcription factors (Yanagisawa et al. 2004). Overexpression of WRKY transcription factor from wheat in tobacco conferred resistance to various abiotic stresses (Wang et al. 2013a, b). In transgenic Arabidopsis, wheat MYB transcription factor (TaMYB56-B) induced resistance to freezing and salt stress (Yu et al. 2017). Furthermore, the genetically modified wheat overproducing TiMYB2R-1 developed tolerance against take-all disease which is the most destructive root disease of wheat (Liu et al. 2013). Similarly, TiERF1transcription factor conferred resistance in transgenic wheat lines against sharp eyespot (Chen et al. 2008).
Dof (DNA binding with one finger) proteins are members of a family of transcription factors that are plant-specific (Kang et al. 2016). Dof proteins are found in both dicots and monocots (Yanagisawa 2002) and induce multiple genes expression associated with organic acid metabolism (Yanagisawa et al. 2004). The unique members of Dof transcription gene family known as Dof1 act like master regulators by concurrently regulating the genes associated with nitrogen and carbon metabolism (Yanagisawa et al. 2004; Kurai et al. 2011). Dof1 proteins recognize a sequence of 5′-AAAG-3′ in the promoter regions of genes (Yanagisawa and Sheen 1998). The expression of various genes involved in carbon and nitrogen skeleton production was upregulated in Arabidopsis (Yanagisawa et al. 2004) and rice (Kurai et al. 2011) expressing Dof1. The genes including phosphoenolpyruvate carboxylase (PEPC), pyruvate kinase (PK), citrate synthase (CS), and isocitrate dehydrogenase (ICDH) were triggered by Dof1 in the transgenic lines of Arabidopsis. In maize, Dof1 enhanced the C4 phosphoenol pyruvate carboxylase (PEPC) activity by interacting with it that led to a light-regulated expression pattern according to Dof1 activity (Yanagisawa 2000).
Under nitrogen-limiting conditions, Dof1 plants exhibited a substantial rise in rate of photosynthesis, root biomass, and root nitrogen content (Rueda-Lopez et al. 2017; Pena et al. 2017). It has been observed that Triticum aestivum Dof1 (TaDof1) transcription factor is mainly associated with improved nitrogen assimilation in plants (Kumar et al. 2009).
Wheat (Triticum aestivum L.) is playing an essential role in fulfilling the nutritional needs of human diet (Contardo-Jara et al. 2018). It is grown in almost 100 countries across the globe including Pakistan. Wheat provides 19% of human calories and is cultivated in approximately 17% of the world’s cultivatable area (over 200 million hectares) (Jones et al. 2005; Atchison et al. 2010). In order to meet the increasing need of the crop on yearly basis, wheat production should be raised to an annual rate of 2% (Sparks et al. 2014). In order to make the wheat production sustainable, it is important to reduce the cost of production by increasing fertilizer use efficiency. Hence, one route to increase wheat yield via improvement in nitrogen use efficiency is to engineer nitrogen assimilation pathway by using biotechnological approaches.
The present study was aimed to transform TaDof1 transcription factor in two elite wheat cultivars to develop transgenic wheat lines with enhanced nitrogen and carbon assimilation under nitrogen-limiting conditions. Previous studies revealed that Dof1 expression induced under nitrogen-deficient conditions plays a role in regulating nitrogen and carbon metabolisms in different species like Arabidopsis, maize, and rice (Kurai et al. 2011; Wang et al. 2013a, b; Pena et al. 2017). However, the role of wheat Dof1 (TaDof1) with respect to nitrogen stress has not been studied. The current investigation included the expression analysis of TaDof1 and the genes directly regulated by TaDof1 in transgenic lines. Chlorophyll, total protein, and soluble sugar contents were also estimated in genetically modified wheat lines.
Materials and Methods
Plant Material
Two elite wheat cultivars (Galaxy and FSD-2008) were selected based on their characteristic features like high yield potential, disease resistance, and stress tolerance. Seeds were obtained from Wheat Research Institute (WRI), Ayub Agricultural Research Institute (AARI), Faisalabad, Pakistan. The seeds were sown on the agricultural fields available on the campus. These were sown at different times so that immature embryos for transformation were available for a longer period.
Vector Construction and Transformation in Bacterial Strains
The plant expression vector pSB219 was obtained from Leibniz Institute of Plant Genetics and Crop Plant Research, Germany, and was maintained in DH10α strain of Escherichia coli. The TaDof1 construct was developed in pSB219 in the multiple cloning site (MCS) region between left and right T-DNA borders. The TaDof1 cassette was cotransformed with pAL154 in AGLI strain of Agrobacterium tumefaciens having C58 chromosome background. The pAL154 is involved in providing replication function in trans to pSB219. Moreover, it has a 15 kb Komari fragment with additional virulence genes (virB, virC, and virG542) for efficient plant transformation (Wu et al. 2008). A. tumefaciens and the pAL 154 were kindly provided by Ms. Caroline Sparks, Rothamsted Research, UK. A. tumefaciens harboring the complete TaDof1 construct was used for wheat transformation.
Retrieval of Gene, Promoter, and Terminator Sequences
The TaDof1 gene consists of 876 bp encoding 291 amino acids. Its nucleotide sequence was retrieved from GenBank with accession number AY955493.2. The gene was got synthesized from Operon Technologies, USA, after adding appropriate restriction sites upstream and downstream of the start and stop codons of the gene. For promoter and terminator amplification, already available vectors were used. CaMV35S promoter with accession number KT600155.1 contains 433 bp and Nos terminator with accession number LC375953.1 contains 261 bp (Fig. S1). The TaDof1 cassette was cloned in the vector using restriction enzymes HindIII, AscI, Eco81I, and SdaI that were present in the MCS of the vector. The promoter-gene-terminator cassette of the vector pSB219 is depicted in Fig. 1.
Wheat Transformation
A complete gene construct in pSB219 having synthetic TaDof1 gene and bar gene as a selectable marker were subjected to wheat transformation. Immature seeds of wheat varieties (FSD-2008 and Galaxy) were collected at 15 days postanthesis (DPA). The immature embryos were dissected aseptically from seeds and placed on callus induction medium (CIM) containing 2,4-dichlorophenoxyacetic acid (2,4-D) for callus formation. The embryogenic calli were selected and used for Agrobacterium-mediated transformation of wheat following the protocol of Jones et al. (2005) with slight modifications. The putative transgenic plants were initially screened by shifting them on regeneration (MSK) medium supplemented with kinetin hormone (1 mg/L), timentin (160 mg/L) and herbicide BASTA (2 mg/L) for 2 weeks followed by another round of selection with 3 mg/L of BASTA in MSK medium for another 2 weeks (Abid et al. 2014). The putative transgenic T0 plants were shifted in pots having peat moss, perlite, and vermiculite in 2:1:1 ratio respectively in climate control room where temperature was maintained at ± 25 °C. During winter season, the T1 plants were grown in sand in a controlled environment at ± 25 °C.
Confirmation of Transgenic Plants by PCR and BASTA Leaf Paint Assay
CTAB method was used to isolate total genomic DNA from T0 plants (Murray and Thompson 1980). Conventional PCR was run to confirm the insertion of complete TaDof1 cassette using the promoter-cds and cds-terminator junction primers (Table 1). PCR was run with a negative and a positive control in which total genomic DNA extracted from control plants was taken as a negative control while TaDof1 vector construct was used as a positive control. The T0 seeds were collected and sown in sand to obtain T1 plants. The T1 plants were screened by applying BASTA leaf paint assay. At the five-leaf plant stage, resistance on the basis of bar gene expression was examined by painting BASTA solution (0.005%) to a specified part on the second or third plantlet leaf. The resistance to herbicide was recorded after 14 days of BASTA leaf paint.
Nitrogen Stress
In order to remove all the nutrients, plants were subjected to leaching after 14 days of germination by maintaining an optimal leaching fraction (17%) (Qiu et al. 2017). After leaching of nutrients, the T1 plants were given nitrogen stress by providing a modified Hoagland nutrient solution (Hoagland and Arnon 1950). In modified Hoagland solution, inorganic nitrogen concentration was reduced from 10 mM in full strength solution to 5 mM. The modified Hoagland solution was given to plants after every 1 week.
Expression Analysis
Expression analysis of TaDof1 and the genes regulated by TaDof1 was done by extracting RNA at two different stages of nitrogen stress, i.e., after 2 weeks of stress and after 4 weeks of stress. Total RNA was isolated from leaves of transgenic wheat plants using Invitrogen Plant RNA Purification Reagent (Cat#12322-012) following the manufacturer’s protocol. Reverse transcription of total RNA (1.2 μg) was done with oligo-dT primers using RevertAidTM H minus first-strand cDNA synthesis kit (Thermo Scientific, USA) as per manufacturer’s instructions. To determine the expression of TaDof1 and other genes in the wild type and transgenic plants, RT-PCR was performed followed by quantitative RT-PCR. As an internal control, actin gene was used for template equalization. TaDofF and TaDofR were used for TaDof1 amplification and ActWhtF and ActWhtR primers (Abid et al. 2017) were used for actin amplification. For quantifying the expression of citrate synthase (CS), isocitrate dehydrogenase (ICDH), phosphoenolpyruvate carboxylase (PEPC), and pyruvate kinase (PK), CSF and CSR, ICDHF and ICDHR, PEPCF and PEPCR, and PKF and PKR primer pairs were used (Table 2).
Morphological Analysis
Various agronomic traits of T1 plants were observed after 4 weeks of nitrogen stress. Among morphological characters, plant height (cm), spike length (cm), number of spikes plant−1, number of grains spike−1, and grain weight (g) were observed.
Physiological and Biochemical Analysis
Estimation of Chlorophyll Content
Arnon (1949) method was employed to estimate the chlorophyll content in leaf samples. Fresh leaf samples were weighed (0.2 g) and ground with 10 ml of 80% acetone followed by centrifugation of the extract at 2500 rpm for 10 min. After transferring the supernatant to test tubes, the volume was raised to 10 ml with 80% acetone. Absorbance was recorded at 645 nm and 663 nm. Chlorophyll contents (chlorophyll a, b and total chlorophyll) were measured as follows:
Estimation of Soluble Proteins
Bradford method (Bradford 1976) was used for determining protein concentration with slight modifications. For this purpose, phosphate buffer solution was used to homogenize 0.2 g of fresh leaf material. The centrifugation of the leaf extract was done at 5000 rpm for 10 min. The supernatant was treated with Coomassie blue reagent. After 5 min of incubation, the absorbance of samples was read at 595 nm. Protein content in the leaf samples was determined using BSA as a standard.
Estimation of Soluble Sugars
Malik and Srivastava (1982) method was used for the estimation of soluble sugar contents. The dry leaf material was ground in 2 ml of 80% acetone and incubated in a shaking incubator overnight. The centrifugation of the extract was done at 2900g. The supernatant was transferred to collection tubes and treated with 5 ml of anthrone reagent. Total soluble sugars were determined by taking absorbance at 625 nm with d-glucose as a standard:
Statistical Analysis
ANOVA in SPSS version 17.0 was used for data analyses and means were analyzed following the Dunnett test. In the Dunnett test, general linear model was used to compare transgenic lines with control plants where F ratios were significant, i.e., p˂0.05. For each sample, three independent replicates were included in the analysis.
Results
Analysis of TaDof1 Cassette in pSB219
In the construct, the TaDof1 gene cassette comprised of 1602 bp. The cassette included CaMV35 promoter, synthetic TaDof1 gene with the stop codon, and a Nos terminator cloned downstream to the TaDof1 gene (Fig. 1, Fig. S1). The integration of complete TaDof1 cassette in pSB219 was confirmed by restriction analysis followed by DNA sequencing analysis.
Agrobacterium-Mediated Wheat Transformation and Plant Screening
For Agrobacterium-mediated transformation, around 1499 calli of Galaxy and 1200 of FSD-2008 were inoculated with A. tumefaciens harboring the TaDof1 construct. A total of only 125 plants could be regenerated and shifted to selection medium (3 mg/L of BASTA). In selection medium, 31 putative transgenic plants (24 from Galaxy and 7 from FSD-2008) survived. PCR analysis using two sets of gene junction primers was performed to confirm the integration of TaDof1 cassette in pSB219. PCR results indicated that the complete transgene cassette was integrated in only 8 out of 31 plants while the remaining possessed truncated cassette (Fig. 2). Out of 8 transgenic plants, 7 were of Galaxy while 1 plant was of FSD-2008. The transformation efficiency was calculated for both the varieties and was found to be 0.46% for Galaxy and 0.08% for FSD-2008. After obtaining T1 plants, BASTA leaf paint test was performed on wild type and control plants. Fourteen days after BASTA application, leaflets of transgenic lines displayed resistance to BASTA, while complete necrosis was examined in leaflets of non-transformed and wild type plants. The T1 lines representing six different transgenic events were selected for further analysis.
PCR results using gene junction primers. a PCR results using primers PGF1R1. M, 1 kb Ladder. Lane 1, +ve control. Lane 3–13 PCR, amplified product. Lane 14, negative control. b PCR results using primers GTF2R2. M, 1 kb ladder. Lane 1, +ve control. Lane 3–13, PCR amplified product. Lane 14, negative control
Transgene Analysis on T1 Plants
To induce the expression of TaDof1, six selected T1 wheat lines were subjected to nitrogen stress. The selected T1 lines representing six different transgenic events included F1, G1, G2, G3, G4, and G5.
Quantitative RT-PCR Analysis
The Dof1-binding sites are known to be found in the promoter regions of genes associated with carbon and nitrogen metabolism (Yanagisawa and Schmidt 1999); therefore, the quantitative RT-PCR was performed to determine the expression profiles of TaDof1 and the genes regulated by TaDof1, i.e., CS, ICDH, PEPC, and PK. In order to investigate the temporal effect of nitrogen stress treatment on the expression of TaDof1 and the genes regulated by TaDof1, quantitative RT-PCR was performed after 2 weeks of nitrogen stress and after 4 weeks of stress. The controls (FC and GC) were taken as a reference to measure the relative fold increase in gene expression in transgenic lines under nitrogen-deficient conditions.
After 2 weeks of stress, the TaDof1 plants exhibited an upregulation of TaDof1 expression with transgenic FSD-2008 (F1) exhibiting 5-fold rise in gene expression as compared with control plant. In transgenic lines of Galaxy (G1, G2, G3, G4, and G5), increase in gene expression ranged from 0.70- to 116-fold. However, G3 represented the minimum fold (0.70) increase while G1 showed the maximum fold increase (116) in TaDof1 expression (Fig. 3a). Among different transgenic lines, only a few showed upregulation of the genes affected by TaDof1. The expression levels of CS, ICDH, PEPC, and PK genes ranged from 0.33- to 0.80-fold for F1 and 0.33- to 17-fold for transgenic lines of Galaxy (Fig. 3b-e). However, the expression of TaDof1 and other genes was coordinately activated in all the transgenic plants after 4 weeks of stress. The elevation in TaDof1 expression level was 106 fold in F1 while it ranged from 3 to 36 fold in transgenic Galaxy lines. The minimum rise in TaDof1 was found in G1 (3-fold) while the maximum increase of 36-fold was observed in G4 (Fig. 3a). Similarly, an enhanced expression of CS, ICDH, PEPC, and PK genes was observed in transgenic lines in comparison with wild type plants. For all the genes regulated by TaDof1, the elevated expression profiles ranged from 0.4- to 88-fold for F1 and 0.35–464 for transgenic lines of Galaxy (Fig. 3b-e). After 4 weeks of stress, the maximum fold increase of 464 was found for ICDH gene. Furthermore, the transgenic wheat lines F1, G1, G2, G4, and G5 showed consistency in enhanced expression of all the genes regulated by TaDof1 while G3 exhibited an inconsistent response. The inconsistency may be due to the fact that TaDof1 was not overexpressed in G3 and in turn could not induce the expression of its target genes.
Expression analysis of various genes in transgenic (F1, G1, G2, G3, G4, G5) and control (FC and GC) wheat plants aT. aestivum Dof1 (TaDof1), b citrate synthase (CS), c isocitrate dehydrogenase (ICDH), d phosphoenolpyruvate carboxylase (PEPC), and e pyruvate kinase (PK) of TaDof1 wheat lines and wild type control plants after 2 and 4 weeks of nitrogen stress. Values are represented as the means ± SD of three replicates after normalization with actin expression. Asterisks denote that the difference between the transgenic and control wheat is significant by the Dunnett test (P < 0.05)
Analysis on Agronomic Traits
Various morphological parameters recorded in transgenic and wild type plants after 4 weeks of nitrogen stress are given in Table 3. Means separations were done independently for FC vs F1 and GC vs G1–G5. A remarkable increase in all the traits was observed in the TaDof1 lines in comparison with control plants. A significantly increased height of 64 cm was exhibited by F1 than non-transgenic FSD-2008 (FC) (55 cm). Among transgenic Galaxy lines, the maximum height (65 cm) was shown by G4. Likewise, considerably increased values, i.e., 3 spikes plant−1 and 11 cm of spike length were observed by F1 than FC having 2 spikes plant−1 and 9.5 cm of spike length. The transgenic line G4 exhibited the maximum values (4 spikes plant−1 and 11 cm of spike length) among all the transgenic lines of Galaxy while non-transgenic Galaxy (GC) had 2 spikes plant−1 and 8 cm of spike length. The similar findings were observed for number of grains spike−1 and grain weight where as compared with FC with 15 grains spike−1 and 0.39 g of seed weight F1 possessed 27 number of grains spike−1 and 0.65 g of seed weight. Among transgenic plants of Galaxy, G4 excelled with 35 grains spike−1 and 0.67 g of seed weight as compared with GC (14 grains spike−1 and 0.31 g of seed weight). Nevertheless, an insignificant increment in the values was displayed by G3 for all the characters except for seed weight (Table 3).
Effect of TaDof1 Expression on Physiological and Biochemical Parameters
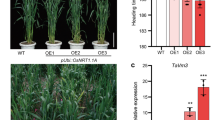
The levels of various physiological and biochemical markers for nitrogen utilization were measured in order to obtain molecular evidence for enhanced growth and nitrogen assimilation in the TaDof1 transgenic plants. Chlorophyll, protein, and soluble sugar contents were measured in the leaves of 4-week old plants grown under low-nitrogen conditions. Significant differences were observed for physiological and biochemical parameters between control and transgenic wheat lines (T1). In comparison with control plants, a substantial increment in chlorophyll a, b and total chlorophyll contents was observed in all the TaDof1 transgenic plants under low-nitrogen conditions (Fig. 4a). In G4, the maximum increase of 31 mg g−1FW, 40 mg g−1FW, and 35 mg g−1FW respectively was found in comparison with wild type plants with 4 mg g−1FW, 6 mg g−1FW, and 5 mg g−1FW of chlorophyll a, b and total chlorophyll contents respectively. For soluble protein content, remarkably increased values were exhibited by 4 out of 6 T1 lines with G4 possessing the highest value of 12 mg g−1FW as compared with control plant (7 mg g−1FW) (Fig. 4b). Similarly, all the T1 lines showed a significant rise in soluble sugar content with G1 exhibiting the maximum rise of 9.9 mg g−1FW in comparison with control plant (4.9 mg g−1FW) (Fig. 4c).
Physiological and biochemical components measured in leaves of transgenic and wild type plants. a Chlorophyll content (mg g−1FW), b protein content (mg g−1FW), c soluble sugars (mg g−1FW). Values are expressed as the means ± SD of three replicates. Asterisks denote that the difference between the transgenic and control wheat is significant by the Dunnett test (P < 0.05)
Discussion
The TaDof1 is a transcription factor that is involved in regulating carbon and nitrogen metabolisms in plants resulting in improved nitrogen utilization (Kumar et al. 2009). The current investigation revealed that the overexpression of TaDof1 transcription factor in wheat led to a coordinated expression of various genes playing role in carbon and nitrogen skeleton production, a remarkable increase in chlorophyll content and an elevation in soluble protein and soluble sugar contents. Since various agronomic traits depicted increased values under low-nitrogen conditions, the TaDof1 wheat plants exhibited better growth than non-transformed controls. The study reveals the improvement in plant growth under nitrogen-limiting conditions due to an efficacious engineering of nitrogen and carbon assimilation pathway. In the nitrogen metabolic pathway, the impact of modifications in the expression of any single enzyme may be masked by concurrent mechanisms in order to maintain homoeostasis. These undesirable consequences can be avoided by engineering the biological mechanisms with transcriptional factors since they have the ability to modify the activity of multiple genes simultaneously. Overproduction of only one enzyme involved in nitrogen assimilation did not increase the biosynthesis of amino acid in genetically modified plants (Brears et al. 1993; Vincent et al. 1997). During photorespiration the recycling of ammonium was improved due to overproduction of glutamine synthetase (GS) that resulted in decreased amounts of ammonia in plants (Oliveira et al. 2002). However, initially the transgenic plants overexpressing GS were grown under nitrogen-sufficient conditions and when the plants were shifted to nitrogen-limiting conditions, they showed enhanced growth (Fuentes et al. 2001). On the contrary, in Dof1 transgenic plants net nitrogen assimilation was improved under low-nitrogen conditions without initial growth of plants under nitrogen-sufficient conditions. In another study, a modified PEPC was overexpressed in potato to generate transgenic potato with elevated levels of amino acids. However, severe growth defects were also observed in genetically modified potato plants as mentioned in the report that organic acid or metabolites imbalance could be the reason of growth defects (Rademacher et al. 2002). These side effects were not found in potato plants overexpressing Dof1. Therefore, the metabolic pathway should be modified to avoid the accumulation of intermediates of different chemical reactions. The modification of nitrogen and carbon assimilation pathway using TaDof1 is a promising approach for coordinated gene expression resulting in enhanced net nitrogen assimilation. A notable improvement in morphological characters coupled with the promotion of various physiological and biochemical parameters is a strong evidence of cooperative modification.
Our results indicated upregulation of CS, ICDH, PK, and PEPC genes in the TaDof1 transgenic plants under nitrogen-deficient conditions. CaMV35S is a well-known constitutive promoter that confers constitutive expression in a wide array of plants, but in various studies, it has been used for transgene expression under stress conditions. In different studies, the increased expression of transgene has been noticed under stress conditions than control conditions indicating that CaMV35S has some stress-responsive elements responsible for transgene expression under stress conditions (Sharir et al. 2005; Cho and Hong 2006; Sun et al. 2012; Song et al. 2014). In the current study, similar results were obtained that the transgene expression increased after 4 weeks of stress as compared with 2 weeks of stress.
The transgenic lines F1, G1, G4, and G5 showed a response of manifold increase in genes expression (Fig. 3). The transgenic rice expressing Dof1 showed a higher number of PEPC transcripts that led to an enhanced carbon flow towards nitrogen utilization (Kurai et al. 2011). The increased PEPC transcript levels have also been observed in other Dof1 plants (Yanagisawa et al. 2004; Wang et al. 2013a, b; Pena et al. 2017) which suggest that PEPC can be used as a molecular marker for Dof1 plants. Yanagisawa et al. (2004) reported an increased activity of CS, ICDH, and PK genes in transgenic Arabidopsis expressing Dof1. In another related study conducted by Wang et al. (2013a, b), higher PK levels were observed in Dof1 tobacco plants. These reports suggest that in addition to inorganic nitrogen present in soil, carbon skeleton (2-OG) is also required which is produced from metabolic intermediates catalyzed by ICDH, CS, PK, and PEPC genes. Hence, a synchronous activation of genes associated with carbon and nitrogen skeleton production leads to net assimilation of nitrogen in transgenic plants expressing Dof1.
The longer period of nitrogen stress resulted in upregulation of CS, ICDH, PEPC, and PK genes (Fig. 3). This significant rise in expression of the genes after 4 weeks of stress might be due to a continuous increase in TaDof1 transcripts over a time period that led to a high number of TaDof1 residues interacting with Dof1-binding domains in the promoter areas of genes. These findings are consistent with the study conducted by Corrales et al. (2014) in which quantitative RT-PCR expression analyses revealed that Dof genes respond to different abiotic stresses with different timing in Dof1 transgenic plants. They explained that minor levels of Dof genes transcripts were recorded in 4-week old plant leaves while a profound increment in transcripts of Dof genes was observed in the leaves of 8-week old plants. The highest fold increase of 464 was found for ICDH gene (Fig. 3c) indicating its essential role in carbon and nitrogen metabolism pathway (Kurai et al. 2011). The plants selected in the study represent different transgenic events; this is why they show variable response. Further, the variable response of different varieties under nitrogen stress conditions may be due to the random nature of transgene insertion (Pérez-González and Caro 2019) (Fig. 3a). However, among the studied varieties, G4 showed a consistent response of higher expression of TaDof1 and the genes regulated by TaDof1 after 4 weeks of nitrogen stress (Fig. 3).
A marked rise in the morphological traits (plant height (cm), spike length (cm), number of spikes plant−1, number of grains spike−1, and grain weight (g) in TaDof1 wheat lines was observed (Table 3). The comparable results were obtained by Wang et al. (2013a, b) which state that under low-nitrogen conditions, different agronomic characters, e.g., plant length and leaf surface, showed better results in transgenic tobacco lines than non-transformed plants. In addition to this, significant changes in physiological and biochemical parameters were recorded in the TaDof1 transgenic plants. In the present investigation, a profound increase in chlorophyll content, soluble protein, and sugar content was recorded in the TaDof1 transgenic plants than non-transgenic plants (Fig. 3). These results are in line with the previous study conducted by Wang et al. (2013a, b) which reported elevated levels of chlorophyll and soluble protein contents in the Dof1 transgenic tobacco plants than wild type plants. A higher chlorophyll and soluble sugar content was recorded in Arabidopsis expressing Dof1 (Yanagisawa et al. 2004). A full-length finger millet Dof1 gene could regulate expression of genes associated with carbon and nitrogen metabolisms that led to protein accumulation in grain (Kumar et al. 2014). The study conducted by Rueda-Lopez et al. (2017) revealed that Dof1 transgenic poplar trees accumulated considerably more carbohydrates than the wild type plants. Taken together, these findings highlighted that overproduction of TaDof1 transcription factor in wheat generated a broad biochemical modification through intimately linked expression of genes. In future, characterization of plants expressing TaDof1 will further explore the intricate regulation of several metabolites that affect plant growth.
References
Abid N, Maqbool A, Malik KA (2014) Screening commercial wheat (Triticum aestivum L.) varieties for Agrobacterium-mediated transformation ability. Pak J Agr Sci 51(1):83–89
Abid N, Khatoon A, Maqbool A, Irfan M, Bashir A, Asif I, Shahid M, Saeed A, Brinch-Pedersen H, Malik KA (2017) Transgenic expression of phytase in wheat endosperm increases bioavailability of iron and zinc in grains. Transgenic Res 26(1):109–122
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24(1):1–15
Atchison J, Head L, Gates A (2010) Wheat as food, wheat as industrial substance; comparative geoghraphies of transformation and mobility. Geoforum 41:236–246
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brears T, Liu C, Knight TJ, Coruzzi GM (1993) Ectopic overexpression of asparagine synthetase in transgenic tobacco. Plant Physiol 103(4):1285–1290
Chen L, Zhang Z, Liang H, Liu H, Du L, Xu H, Xin Z (2008) Overexpression of TiERF1 enhances resistance to sharp eyespot in transgenic wheat. J Exp Bot 59(15):4195–4204
Cho EK, Hong CB (2006) Over-expression of tobacco NtHSP70-1 contributes to drought-stress tolerance in plants. Plant Cell Rep 25(4):349–358
Contardo-Jara V, Schwanemann T, Esterhuizen-Londt M, Pflugmacher S (2018) Protein association of β-N-methylamino-L-alanine in Triticum aestivum via irrigation. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 35(4):731–739
Corrales AR, Nebauer SG, Carrillo L, Fernandez-Nohales P, Marques J, Renau-Morata B, Granell A, Pollmann S, Vicente-Carbajosa J, Molina RV, Medina J (2014) Characterization of tomato cycling Dof factors reveals conserved and new functions in the control of flowering time and abiotic stress responses. J Exp Bot 65(4):995–1012
Coruzzi GM, Zhou L (2001) Carbon and nitrogen sensing and signaling in plants: emerging ‘matrix effects’. Curr Opin Plant Biol 4(3):247–253
Fuentes SI, Allen DJ, Ortiz-Lopez A, Hernandez G (2001) Overexpression of cytosolic glutamine synthetase increases photosynthesis and growth at low nitrogen concentrations. J Exp Bot 52(358):1071–1081
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil, vol 347. Agricultural Experiment Station circular, Berkeley
Jones HD, Doherty A, Wu H (2005) Review of methodologies and a protocol for the Agrobacterium-mediated transformation of wheat. Plant Methods 1(1):5
Kang WH, Kim S, Lee HA, Choi D, Yeom SI (2016) Genome-wide analysis of Dof transcription factors reveals functional characteristics during development and response to biotic stresses in pepper. Sci Rep 6:33332
Kumar R, Taware R, Gaur VS, Guru SK, Kumar A (2009) Influence of nitrogen on the expression of TaDof1 transcription factor in wheat and its relationship with photosynthetic and ammonium assimilating efficiency. Mol Biol Rep 36(8):2209–2220
Kumar A, Kanwal P, Gupta AK, Singh BR, Gaur VS (2014) A full-length Dof1 transcription factor of finger millet and its response to a circadian cycle. Plant Mol Biol Rep 32:419–427
Kurai T, Wakayama M, Abiko T, Yanagisawa S, Aoki N, Ohsugi R (2011) Introduction of the ZmDof1 gene into rice enhances carbon and nitrogen assimilation under low-nitrogen conditions. Plant Biotechnol J 9(8):826–837
Liu X, Yang L, Zhou X, Zhou M, Lu Y, Ma L, Ma H, Zhang Z (2013) Transgenic wheat expressing Thinopyrum intermedium MYB transcription factor TiMYB2r-1 shows enhanced resistance to the take-all disease. J Exp Bot 64(8):2243–2253
Liu CW, Sung Y, Chen BC, Lai HY (2014) Effects of nitrogen fertilizers on the growth and nitrate content of lettuce (Lactuca sativa L.). Int J Environ Res Public Health 11(4):4427–4440
Liu XY, Koba K, Koyama LA, Hobbie SE, Weiss MS, Inagaki Y, Shaver GR, Giblin AE, Hobara S, Nadelhoffer KJ, Sommerkorn M, Rastetter EB, Kling GW, Laundre JA, Yano Y, Makabe A, Yano M, Liu CQ (2018) Nitrate is an important nitrogen source for arctic tundra plants. Proc Natl Acad Sci U S A 115(13):3398–3403
Malik CP, Srivastava AK (1982) Text book of plant physiology. Kalyani Publishers, Delhi
Mulvaney RL, Khan SA, Ellsworth TR (2009) Synthetic nitrogen fertilizers deplete soil nitrogen: a global dilemma for sustainable cereal production. J Environ Qual 38(6):2295–2314
Murray MG, Thompson WF (1980) Rapid isolation of higher weight DNA. Nucleic Acids Res 8(19):4321–4325
Nosengo N (2003) Fertilized to death. Nature 425(6961):894–895
Oliveira IC, Brears T, Knight TJ, Clark A, Coruzzi GM (2002) Overexpression of cytosolic glutamine synthetase. Relation to nitrogen, light, and photorespiration. Plant Physiol 129(3):1170–1180
Pena PA, Quach T, Sato S, Ge Z, Nersesian N, Changa T, Dweikat I, Soundararajan M, Clemente TE (2017) Expression of the maize Dof1 transcription factor in wheat and sorghum. Front Plant Sci 8:434
Pérez-González A, Caro E (2019) Benefits of using genomic insulators flanking transgenes to increase expression and avoid positional effects. Sci Rep 9(1):8474
Qiu R, Liu C, Wang Z, Yang Z, Jing Y (2017) Effects of irrigation water salinity on evapotranspiration modified by leaching fractions in hot pepper plants. Sci Rep 7(1):7231
Rademacher T, Hausler RE, Hirsch HJ, Zhang L, Lipka V, Weier D, Kreuzaler F, Peterhansel C (2002) An engineered phosphoenolpyruvate carboxylase redirects carbon and nitrogen flow in transgenic potato plants. Plant J 32(1):25–39
Rueda-Lopez M, Pascual MB, Pallero M, Henao LM, Lasa B, Jauregui I, Aparicio-Tejo PM, Canovas FM, Avila C (2017) Overexpression of a pine Dof transcription factor in hybrid poplars: a comparative study in trees growing under controlled and natural conditions. PLoS One 12(4):e0174748
Sharir I, Isaacson T, Lurie S, Weiss D (2005) Dual role for tomato heat shock protein 21: protecting photosystem II from oxidative stress and promoting color changes during fruit maturation. Plant Cell 17(6):1829–1838
Song A, Zhu X, Chen F, Gao H, Jiang J, Chen S (2014) A chrysanthemum heat shock protein confers tolerance to abiotic stress. Int J Mol Sci 15(3):5063–5078
Sparks CA, Jones HD (2014) Genetic transformation of wheat via particle bombardment. Methods Mol Biol 1099:201–218
Sparks CA, Doherty A, Jones HD (2014) Genetic transformation of wheat via Agrobacterium-mediated DNA delivery. Methods Mol Biol 1099:235–250
Stitt M (1999) Nitrate regulation of metabolism and growth. Curr Opin Plant Biol 2(3):178–186
Sun L, Liu Y, Kong X, Zhang D, Pan J, Zhou Y, Wang L, Li D, Yang X (2012) ZmHSP16.9, a cytosolic class I small heat shock protein in maize (Zea mays), confers heat tolerance in transgenic tobacco. Plant Cell Rep 31(8):1473–1484
Vincent R, Fraisier V, Chaillou S, Limami MA, Deleens E, Phillipson B, Douat C, Boutin JP, Hirel B (1997) Overexpression of a soybean gene encoding cytosolic glutamine synthetase in shoots of transgenic Lotus corniculatus L. plants triggers changes in ammonium assimilation and plant development. Planta 201(4):424–433
Vitousek PM, Naylor R, Crews T, David MB, Drinkwater LE, Holland E, Johnes PJ, Katzenberger J, Martinelli LA, Matson PA, Nziguheba G, Ojima D, Palm CA, Robertson GP, Sanchez PA, Townsend AR, Zhang FS (2009) Agriculture. Nutrient imbalances in agricultural development. Science 324(5934):1519–1520
Wang C, Deng P, Chen L, Wang X, Ma H, Hu W, Yao N, Feng Y, Chai R, Yang G, He G (2013a) A wheat WRKY transcription factor TaWRKY10 confers tolerance to multiple abiotic stresses in transgenic tobacco. PLoS One 8(6):e65120
Wang Y, Fu B, Pan L, Chen L, Fu X, Li K (2013b) Overexpression of Arabidopsis Dof1, GS1 and GS2 enhanced nitrogen assimilation in transgenic tobacco grown under low-nitrogen conditions. Plant Mol Biol Report 31(4):886–900
Wu H, Doherty A, Jones HD (2008) Efficient and rapid Agrobacterium-mediated genetic transformation of durum wheat (Triticum turgidum L. var. durum) using additional virulence genes. Transgenic Res 17(3):425–436
Wuebbles DJ (2009) Atmosphere. Nitrous oxide: no laughing matter. Science 326(5949):56–57
Yanagisawa S (2000) Dof1 and Dof2 transcription factors are associated with expression of multiple genes involved in carbon metabolism in maize. Plant J 21(3):281–288
Yanagisawa S (2002) The Dof family of plant transcription factors. Trends Plant Sci 7(12):555–560
Yanagisawa S, Schmidt RJ (1999) Diversity and similarity among recognition sequences of Dof transcription factors. Plant J 17:209–214
Yanagisawa S, Sheen J (1998) Involvement of maize Dof zinc finger proteins in tissue-specific and light-regulated gene expression. Plant Cell 10(1):75–89
Yanagisawa S, Akiyama A, Kisaka H, Uchimiya H, Miwa T (2004) Metabolic engineering with Dof1 transcription factor in plants: improved nitrogen assimilation and growth under low-nitrogen conditions. Proc Natl Acad Sci U S A 101(20):7833–7838
Yu Y, Ni Z, Chen Q, Qu Y (2017) The wheat salinity-induced R2R3-MYB transcription factor TaSIM confers salt stress tolerance in Arabidopsis thaliana. Biochem Biophys Res Commun 491(3):642–648
Authors’ Contribution Statement
Ammarah Hasnain conducted the experiments and wrote the manuscript. Asma Maqbool and Kauser Abdulla Malik designed the experiments and analyzed data. Muhammad Irfan and Aftab Bashir contributed new analytical tools. All authors read and approved the manuscript.
Funding
The project was supported by Agricultural Linkages Programme (ALP) under Pakistan Agricultural Research Council (PARC), Pakistan. Project Identification No. CS-313.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Message
• Overexpression of TaDof1 in T1 wheat lines resulted in enhanced expression of genes involved in nitrogen assimilation. Physiological and biochemical parameters analysis also indicated improved nitrogen assimilation in T1 plants
Electronic supplementary material
ESM 1
(DOCX 515 kb)
Rights and permissions
About this article
Cite this article
Hasnain, A., Irfan, M., Bashir, A. et al. Transcription Factor TaDof1 Improves Nitrogen and Carbon Assimilation Under Low-Nitrogen Conditions in Wheat. Plant Mol Biol Rep 38, 441–451 (2020). https://doi.org/10.1007/s11105-020-01208-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-020-01208-z