Abstract
Aims
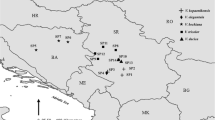
Based on a recent taxonomic revision of the genus Odontarrhena in Albania, our work aimed at comparing the taxa and populations of this genus in terms of nickel accumulation and levels of other metals in relation to their concentrations in the soil at distinct Albanian ultramafic outcrops.
Methods
Several populations of different Odontarrhena species were sampled from 20 different outcrops across the entire Albanian territory. Concentrations of Ni, Co, Cr, Mg, Ca, K, Fe and Mn were determined in soil, roots and shoots of all specimens sampled.
Results
Ultramafic soils across Albania showed variable metal composition, with significant differences between sites. Large differences in mineral element concentrations were also found between the plants which were not related to species identity or geographical distribution of their populations.
Conclusions
Shoot Ni concentrations in Albanian Odontarrhena taxa depend on soil Ni concentrations but not on species identity. For O. chalcidica, the more widely distributed species, the environmental fingerprint for shoot metal composition was found not only for Ni, but also for Ca and Mg, the latter showing a positive linear relationship between soil and plant concentrations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ultramafic (‘serpentine’) soils, derived from ultramafic rocks, present unique geochemical characteristics such as high levels of Ni, Co and Cr, low levels of macronutrients N, P, K and Ca, and have a high Mg/Ca quotient (Brooks 1987; Gonnelli and Renella 2012). However, their elemental composition varies widely on a global scale since these soils are derived from rocks that occur under a broad range of climatic regimes, from temperate to tropical (Kruckeberg 2002). Consequently, ‘serpentine’ soils are not only “ecological islands” (Lefèbvre and Vernet 1990) in the “sea” of normal soils, but also each “island” can be considered as exclusive, since diversity in metal concentrations has been demonstrated at even a very local scale (Galardi et al. 2007; van der Ent et al. 2018).
The chemical and physical properties of ultramafic soils render them uninhabitable by most plant species and, at the same time, give them a major role in the evolutionary adaptation of plants, so that these soils often support distinct communities including rare and endemic taxa (Kruckeberg 1954; Kruckeberg and Kruckeberg 1990). Among the most specialized serpentinophytes there are the so-called Ni-hyperaccumulating plants. The term was coined by Brooks et al. (1977) to define plants with Ni concentrations >1000 μg g−1 DW in their above-ground parts. Such a level of Ni concentrations is extraordinarily high, being 100–1000 times higher than that generally present in plants on non-ultramafic soils, and 10–100 times higher than found in normal plants on Ni-rich ultramafic soils (van der Ent et al. 2013a). Globally, nearly 500 plant species have been reported to possess Ni-hyperaccumulation ability, thus representing the largest group among all the metal-hyperaccumulators, probably due to the large number of ultramafic outcrops worldwide (van der Ent et al. 2013a; Reeves et al. 2018). In the last decades, Ni-hyperaccumulators have attracted much interest for scientific research, conservation purposes, ecological restoration and practical applications of agromining (Wójcik et al. 2017, Whiting et al. 2004, van der Ent et al. 2015).
Nickel hyperaccumulation was first reported by Minguzzi and Vergnano (1948) who discovered extraordinarily high levels of this metal in the shoots of the Tuscan endemic Odontarrhena bertolonii (formerly known as Alyssum bertolonii; Španiel et al. 2015). Later studies showed that Ni concentrations were very variable among the O. bertolonii populations and representative of the degree of mineralization of the soil, along with Co concentration (Galardi et al. 2007). Indeed, O. bertolonii is only one of the many members of this genus that grow on the ultramafics of the Euro-Mediterranean region and Western Asia and accumulate Ni (Cecchi et al. 2010). Currently, several of these taxa are used as model systems to investigate the physiological and molecular mechanisms of Ni accumulation (e.g. Krämer et al. 1996; Ingle et al. 2005; Verbruggen et al. 2009, Deng et al. 2018) and their responses in technological applications of phytoremediation and agromining (Robinson et al. 1997; Chaney et al. 2005; Bani et al. 2015; Kidd et al. 2018). In temperate regions, most promising species for Ni agromining range across the Balkans, where large ultramafic outcrops occur from Serbia and Bosnia at the north to Greece at the south (Salihaj et al. 2018). Albania hosts a large proportion of these outcrops (Brooks 1987; Tatic and Veljovic 1992) and extensive Odontarrhena populations of different species. However, in spite of such importance, the identity and limits of these taxa have remained confused until recently, so preventing their correct characterization in terms of plant metal concentrations in field-collected samples and metal composition of the corresponding soils. Previous studies (Shallari et al. 1998; Tumi et al. 2012; Bani et al. 2013, 2015) focused on the O. muralis s.L. complex and provide useful data on metal accumulation in samples of species of this polymorphic taxon without considering other less widely distributed species that are therefore still unknown. Recently, the taxonomy of this critical group was disentangled by a thorough revision, which led to the recognition of seven taxa (Cecchi et al. 2018). One of these is endemic to calcareous soils south of Lake Ohrid (O. albiflora), two are facultative serpentinophytes (O. chalcidica and O. sibirica) and four are obligate serpentine endemics (O. decipiens, O. moravensis, O. rigida and O. smolikana subsp. glabra). The other subspecies of O. smolikana (O. smolikana subsp. smolikana), also tetraploid, is endemic to the high serpentine massifs of the northern Pindus in Greece (Mt. Smolikas and Vasilitsas) and is characterized by the densely pubescent silicular valves (Hartvig 2002). One major point that emerged was the absence of genuine O. muralis, which is a non-serpentine species distributed in the south-eastern Balkans and Anatolia (Hartvig 2002). Hence all previous reports of this species from Albania are to be referred to other similar taxa of this group, most probably O. chalcidica and/or O. decipiens.
Balkan species of Odontarrhena are small suffruticose perennials with numerous sterile shoots from a woody base and fertile stems usually appearing for the first time in the spring of the second year. Any literature information on the breeding systems is lacking, but field observations suggest that the bright yellow flowers are visited by a wide variety of pollinating insects including bees, hover flies, flies, wasps and small beetles. Allogamy is the main breeding system (unpubl. data), although selfing may also occur as in the perennial species of Alyssum (Hegi 1986; Rusterholz et al. 2012). All species (except O. sibirica) occur with large populations that often form a major component of the local vegetation, especially the tetraploids O. chalcidica, O. decipiens, and O. smolikana. Population density of synantropic O. chalcidica may be very high in ruderal and heavily disturbed sites, either on serpentine or schist, and this species may become mixed with the other Odontarrhena species at their corresponding sites. The diploids O. rigida and O. moravensis are less dominant in the communities where they occur, though usually well represented. Population genetic structure is unknown but previous molecular work including several Albanian and Balkan populations (Cecchi et al. 2010; Rešetnik et al. 2013) suggested low rates of differentiation possibly due to recent radiation, and do not support genetic drift as a major evolutionary force shaping patterns of phenotypic variation. The clear taxonomic framework at the base of this study, therefore, provides the opportunity to characterize the taxa in terms of plant metal concentrations in relation to soil concentrations in their native habitat of unfertilized serpentine soils. Using a population sampling across all major ultramafic areas of Albania and with multiple sites for each taxon, we tested species identity in relation to soil and shoot Ni concentrations. A further aim of the work was to examine the relationships between the concentrations of different metals in soils and plants. This investigation provides insights into the soil chemistry that could affect shoot Ni concentrations in the taxa studied, which is potentially useful for optimizing the species selection for phytotechnological exploitation of substrata in agromining practices.
Materials and methods
Sampling design for soil and plant materials
Collection of soil and plant materials was performed during four field trips to Albania in 2016 and 2017. Sampling was based on two criteria, geographic and taxonomic. For the former, all major ultramafic areas in the country, as reported in Bani et al. (2013), were systematically visited, from Tropoja at the north to Rehova at the south, as well as a few non-serpentine intervening areas hosting Odontarrhena populations (Fig. 1). Originating in the Cretaceous era, ultramafic soils cover 10% of the Albanian land area (Bani et al. 2013) and include serpentinite and harzburgite (peridotite; Dilek and Furnes 2009). Both types are siliceous rocks with very low silica:magnesium + iron ratios because of the abundance of olivine, pyroxenes and serpentine clays (Salihaj et al. 2018). Based on the taxonomic criterion, we sampled at least two populations of five of the seven native taxa as recently defined in Cecchi et al. (2018), with emphasis on the populations from the type localities of those described from Albania. These were all included in our sampling to establish unambiguous correspondence between taxa and metal concentrations in their shoots and soils. Overall, 24 populations belonging to all five taxa able to grow on serpentine both obligately or facultatively were sampled at 20 sites ranging from 60 to 1250 m above sea level (Table 1; Fig. 1). At three sites, Mt. Shebenik, Pishkash and Mt. Moravë, different taxa were mixed and close to each other in the same loci; O. chalcidica, O. rigida and O. smolikana subsp. glabra in the first, O. chalcidica and O. rigida in the second and O. chalcidica and O. moravensis in the third. The number of populations analyzed per taxon was inevitably uneven because of the different extent of their distributional ranges. The facultative serpentinophyte O. chalcidica, widespread on both serpentine and non-serpentine soils, was sampled from 11 sites, whereas the serpentine obligate endemics O. moravensis, O. rigida and O. smolikana subsp. glabra were all found at only two or three sites. The putative hybrid between the latter and O. chalcidica, corresponding to O. decipiens, was collected from five sites of the northern and central ultramafics. The inter-population distance range (min-max) within each species was 2–220 km for O. chalcidica, 25–180 km for O. decipiens, 10–35 km for O. moravensis, 13–31 km for O. rigida and 32–128 km for O. smolikana. The two taxa not included in the analyses were O. albiflora, only known from a single population on the limestone cliffs of Mali Thatë in the Korçë region south of Lake Ohrid, and O. sibirica collected only once in the extreme south of Albania and not observed since more than a century ago (Cecchi et al. 2018). All the sites visited supported unfertilized serpentine soils, with the exception of one that was on schist.
Metal concentrations in soil and plant samples
At each location, ten bulk soil samples of ca. 20 g were randomly collected with a shovel at ca.1–15 cm depth, and then pooled together to obtain a single bulk soil sample (ca. 200 g). Each sample was then dried at room temperature, sieved with a 2 mm mesh stainless steel sieve and then fully dried in an oven at 50 °C for 7 days (Pignattelli et al. 2012). From each bulk sample, five subsamples of about 0.5 g were digested using 10 mL of 69% HNO3 in a microwave digestion system (Mars 6, CEM). Elemental concentrations in the digests were determined by atomic absorption spectroscopy (AAnalyst 200 B315–0060, Perkin Elmer, with deuterium background correction) and used to calculate the pseudo-total metal concentrations in the soils. We did not attempt to determine bioavailable element concentrations in this study.
At each site, five adult plants spaced at least 20 m apart from each other and of comparable size and in vegetative phase, were randomly dug up to provide the complete root system. When two or more species were present at the same site, a sample collection was made for all of them. A more intensive sampling (e.g. collection of more than five individual specimens) was avoided in order not to cause damage to these wild populations. Shoots and roots were carefully washed three times with deionized water, dried at 50 °C for 48 h (Selvi et al. 2017), and then about 0.05 g subsamples of dried materials were analyzed in the same way as described for soils. Each plant sample analysis was replicated three times.
Statistical analyses
Ordination by Principal Components Analysis (PCA) was first used to detect the main sources of variation within both the soil and plant analysis matrices and to summarize relationships between, sites/populations/taxa and metals. Eigenvalues were extracted from the correlation matrix. One-way ANOVA followed by an a posteriori Tukey’s test was used to compare the individual means of soil (mean of five analytical replicates of the bulk soil) and plant (mean of five values, each one derived from the mean of three analytical replicates per each plant) metal values, after checking for data for normality using the Shapiro-Wilk test for p < 0.05. ANOVA was performed separately for shoots and roots. Correlation analysis was performed for the elemental concentrations in the soils using Pearson’s chi-squared method. Linear regression analyses were also performed to investigate the relationships between metal concentrations in soils and plants. Univariate statistical analyses were made using GraphPad Prism 7 (GraphPad Software, San Diego, CA) and SigmaPlot 12.0, whereas the ordination was performed using PAST vs. 2.17 software (Hammer, 2012).
Results
Element concentrations in soils
The first two PCA components extracted from the soil metal concentration data set explained 59.3% of the total variance (42.1% and 17.2%, respectively). The resulting scattergram (Fig. 2a) showed broad separation between the 20 sampling sites but no clear grouping patterns. Hence, the observed variations in metal concentrations between sites were not apparently dependent on geographical, altitudinal or other site factors as determined here. Nickel was the most strongly correlated variable on the positive part of the first component (r = 0.89) and Ca/Mg quotient was the major driver on the negative part (r = −0.73). Chromium concentration was negatively correlated with the second component (r = −0.63).
As expected for ultramafic soils, concentrations of Ni, Co, Cr, Mg, Fe and Mn were elevated but low for Ca; except for S.4, the Ca/Mg quotient was <1 and the pH values ranged from 6.33 (S.5) to 7.36 (S.10) (Tables 2 and S1). Positive relationships were apparent between Ni and Co (r = 0.80**), Ni and Cr (r = 0.45*), Ni and Mg (r = 0.44*), Ni and Fe (r = 0.60**), Co and Cr (r = 0.51*), Co and Fe (r = 0.67**), Co and Mn (r = 0.58**), Cr and Fe (r = 0.59**) and Fe and Mn (r = 0.45*).
Significant differences between localities emerged from the ANOVA (Tables 2, S2). Nickel, Cr and Co were highest in S.3 and lowest in S.13; elevated concentrations of some of the latter elements were found in S.12, S.7. S.15 and S.20. Calcium was highest in S.13 and lowest in S.5 and S.20; Mg was highest in S.6 and S.15 and lowest in S.1 and S.13 (excluding S.4). The Ca/Mg quotient was highest in S.13 and lowest in S.20. Potassium was highest in S.1 and S.8, lowest in S.7.
Element concentrations in plants
The first two PCA components accounted for 65.2% of the total variation (43.6% and 21.6%, respectively) in the plant metal data set. The resulting scattergram (Fig. 2b) showed broad separation of the 24 populations due to significant variations in both root and shoot metal concentrations (Tables 3, S4). However, no clear groupings emerged in relation to species identity or geographical origin. In addition, patterns of population distribution in the PCA scattergram were apparently not consistent with the pattern of site distribution in Fig. 2a. Magnesium was the more strongly correlated variable on the positive part of the first component (r = 0.88), but the Ca/Mg quotient was again the major driver on the negative part (r = −0.90). Chromium was negatively correlated with the second component (r = −0.86).
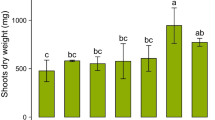
Nickel concentration was invariably higher in shoots than in roots. The highest root Ni concentrations were in two O. chalcidica populations, Oc.6 and Oc.8 (ca. 3.7 and 3.5 mg g−1 DW, respectively; Table 3a), although some samples of this species (Oc.10) contained only 0.5 mg g−1 DW. The minimum Ni concentration (ca. 0.4 mg g−1 DW) was found in O. decipiens (Od.3), but this population was not significantly different from some others. A similar pattern was found for shoots (Table 3b), with the highest concentrations occurring in some O. chalcidica populations (Oc.6 and Oc.8, ca. 19.2 mg g−1 DW). Similarly, Oc.10 showed the lowest Ni concentration among the O. chalcidica populations, although not significantly different from the population showing the lowest Ni in the shoots, Od.3 (ca. 3.4 mg g−1 DW). The Ni shoot/root values ranged from 4 (Od.1) to 15 (Om.2), whereas the bioconcentration factor was generally ≤1 for roots, and widely variable for shoots (from 1.7 in Od.1 to 25 in Od.2; Table S5).
Differences between populations were also found for the other metals (Tables 3 and S3). Concentrations of Co, Cr, Mg and Ca were always higher in shoots than in roots, whereas the two organs showed quite comparable concentrations of K, Fe and Mn. In comparison to soil, shoot concentrations were higher for Ni, Ca and K. The range of Ca/Mg quotients in roots was relatively wide (from 0.4 in Os.1 to 6.0 in Oc.4), and this was >1 in almost half of the populations. In shoots, it was always >1, ranging from 1.2 (Oc.6) to 16.2 (Oc.10).
When considering the overall population sampling, a positive relationship emerged between shoot Ni and Mg concentrations (r = 0.57*) and a negative one between Ni and Ca (r = −0.41*). Restricting the analysis to the 11 populations of O. chalcidica, these relationships were confirmed and others detected. Shoot Ni and Mg concentrations were positively related (r = 0.84**), whereas Ca, K and the Ca/Mg quotient decreased with increasing Ni (respectively: r = −0.69*; r = −0.67*; r = −0.83**). Concentrations in shoots and roots were positively related for Ni (r = 0.92**), Mg (r = 0.71**) and the Ca/Mg quotient (r = 0.75**).
Soil-plant metal relationships
Relationships between soil and shoot concentrations were mostly non-significant when considering the pooled sample of plant populations. A remarkable exception was for Ni, whose concentration in plant samples increased significantly with concentration in the soil (r = 0.47*), regardless of population species identity (Fig. 3). Furthermore, if the S.4 sample (schist) was excluded from the analysis, plant Ni concentration was negatively related to soil pH (r = −0.44* and r = −0.53* for roots and shoots respectively). By restricting the analysis to the O. chalcidica populations, the plant-soil concentration relationship was again positive for Ni (r = 0.66*), Mg (r = 0.63*) and Ca (r = 0.65*). Calcium in the plant tended to decrease with increasing Ni in the soil (r = −0.61*), and vice versa (Casoil-Niplant: r = −0.65*); the relationship Casoil-Mgplant was also negative (r = −0.62*). Plant Ni concentration was inversely related to the Ca/Mg quotient in the soil (r = −0.67*), and the same negative relationship held for Mgplant and Ca/Mgsoil (r = −0.70*), when excluding the S.4 sample from the analysis.
The above relationships were not due to soil shoot contamination, as ratios between the elements were very different in plants and soils. Furthermore, the very low Cr, Fe and Mn concentrations in plants, despite their high levels in the soil, supports the view that contamination of the plant samples was not significant.
Discussion
Element concentration in the Albanian serpentine outcrops
Nickel concentrations in the Albanian ultramafic soils investigated here were widely variable (500–5000 μg g−1 DW), with more than half of the sites showing pseudo-total concentrations above the mean value of 2.0 mg Ni g−1 DW reported for serpentine outcrops (Brooks 1987). In 17 of the twenty localities Ni concentration was >1.0 mg g−1 DW, the minimum threshold value to select sites potentially suitable for Ni agromining practices (van der Ent et al. 2013b, 2015; Nkrumah et al. 2016). Hence, the current use of this practice in the serpentine area of the Pogradec district (Kidd et al. 2018) can apparently be extended to most of the ultramafic marginal lands in the country. Similarly to Ni, the other metals and the Ca/Mg quotient showed a relatively wide range of variation, more or less scattered around the mean values reported for serpentine soils (Brooks 1987). It is notable that the schist soil sample from the Drin valley (S.4) had a Ca/Mg quotient >1, which indicated a non-serpentine nature, but showed unexpectedly high concentrations of trace metals probably due to its colluvial origin. Other studies on trace elements in Albanian outcrops (Shallari et al. 1998, Bani et al. 2009, 2010 and 2013) also showed variable metal concentrations among the different localities, with concentrations within the range of values reported here.
Ultramafic soils from different world regions are well known to differ in their cation concentrations (Kruckeberg 2002). However, significant local-scale heterogeneity in metal concentrations in soils from outcrops that mostly share a common geologic origin has been rarely demonstrated, for example in the sites of O. bertolonii in central Italy (Galardi et al. 2007). This can be associated with differences in both local rock composition, geomorphological factors and microclimatic conditions influencing rock weathering, although our multivariate analyses did not suggest any clear groupings of sites according to their geographic location or altitude.
Correlation analysis revealed that soils with the highest Ni concentrations were also characterized by the highest values for Co and Cr. Such a relationship between the three ‘serpentine’ metals could be determined by their substituting and associated elements, Mg and Fe, that are usually present in the mafic materials (Brooks 1987). Their proportion was variable, as inferred from the observed differences in Mg and Fe concentrations that were in turn positively related to those of Ni.
Element concentration in plants
Shoot Ni, Ca and K concentrations were generally higher than in the corresponding soil, whereas Co, Cr, Mg, Fe and Mn were lower, as expected from the peculiar features of serpentine Ni-accumulating plants in terms of trace elements and macronutrients (see for example Brooks 1987; Kazakou et al. 2008). All elements were more concentrated in shoots than in roots, except for K, Fe and Mn, and all elements displayed a wide range of variation in the taxa, in both roots and shoots. For example, Ni concentration showed a ten-fold spanning interval in roots and a seven-fold one in shoots. Concentrations of the elements observed here were within the range of those reported in previous studies on Balkan plants (Shallari et al. 1998, Bani et al. 2009, 2010 and 2013), considering that taxa originally referred to Alyssum markgrafii and A. bertolonii actually correspond to O. decipiens and O. smolikana (Cecchi et al. 2018).
It is of note that O. chalcidica included the populations with the highest shoot Ni concentration recorded here, ca. 19.2 mg g−1 DW, from the localities of Prrenjas and Pogradec. The latter site has been already selected and used to develop most promising agromining experimental systems with O. chalcidica (Bani et al. 2018).
Despite the low soil Ca concentration, another important feature was the inversion of the soil Ca/Mg quotient in all five taxa, the shoot Ca concentration of our samples always falling within the predicted range for non-serpentine species (Marschner 1985). This confirms the well-known ability of Ni hyperaccumulators to accumulate this macronutrient (Reeves et al. 1997).
Multivariate analysis showed no clear separation of populations in relation to their origin or taxonomic identity, thus suggesting their metal concentration profiles to be neither species-specific nor associated with geographical factors.
Apart from possible environmental constraints driving trace element relationships in O. chalcidica, explored in the following paragraph, we found that shoot Ni concentration was positively correlated with Mg, the macronutrient excluded from the substrate. On the other hand, it was negatively associated with Ca and K, two macronutrients that are accumulated in respect to their levels in the substrate (thus also driving the inverse relation Ni-Ca/Mg). This becomes of particular importance for agromining applications. In addition to high shoot Ni concentrations, low levels of Ca are in fact also desirable during metal recovery processes to make Ni salts with high value (Nkrumah et al. 2018; Simonnot et al. 2018), and these two characteristics appeared to be already associated in the O. chalcidica populations growing on unfertilized serpentine soils and analyzed here. The same comment applies to K, which needs to be removed from the ashes by washing with water before lixiviation with sulphuric acid (Zhang et al. 2016).
Regarding relationships between root and shoot metal concentration in the O. chalcidica populations, Ni and Mg were the only elements whose concentrations in the two organs were positively related. Regardless of the absolute element concentrations, all populations of this species showed a similar allocation pattern not only for Ni accumulation, but also for the exclusion of Mg, whose excess is one of the major edaphic stress factors of the serpentine soils (Brooks 1987).
Unravelling soil-plant relationships in Albanian species of Odontarrhena
Although the identification of unambiguous relationships between elements in soils and hyperaccumulating plants has for many years been an ultimate aim of biogeochemical prospecting, few studies are available to date on this topic, especially at the plant population level (Galardi et al. 2007). In field studies, there are many potentially variable factors affecting the final plant ionome with respect to soil metal concentrations, such as climate, altitude, intrinsic soil physical and chemical properties, as well as intra-specific genetic variability. Moreover, in natural soils all element concentrations vary simultaneously, thus obscuring the effects of competition between the elements. Despite these potential shortcomings, our pooled soil-taxa datasets pointed to a positive significant linear relationship between Ni concentration in soils and shoots of the Odontarrhena species studied here, and to some intriguing features of the element parallel behavior in the O. chalcidica shoot systems from serpentine soil.
To the best of our knowledge, this work is the first report of such a positive Nisoil-Niplant relationship in Albanian Odontarrhena hyperaccumulating taxa collected in their natural habitat, based on a robust population sampling from a broad geographic and taxonomic range. Looking at this relationship, the five taxa appeared interspersed with each other, thus showing that Ni concentration in the Albanian Odontarrhena hyperaccumulators depends mainly on Ni concentration in the substrate, rather than on species identity. This is supported by the finding that shoot Ni concentration was not significantly different in those species populating the same outcrop, often mixed at a very local scale as in the case of site S.6 (O. smolikana, O. rigida, O. chalcidica). Although the possible occurrence of genetic variation in shoot Ni concentration still needs to be assessed in most of the species here studied, the Albanian taxa of Odontarrhena are likely useful indicators of soil Ni concentration. On the other hand, different hyperaccumulator taxa often exhibit similarly high levels of accumulation over a very wide range of soil metal levels (Baker 1981), suggesting the need for experimental studies to better understand soil-plant Ni causal relationships in these species. Moreover, another relevant finding was that, for the soil pH values of the Albanian outcrops here studied, the relationship between this parameter and shoot Ni concentration in Odontarrhena plants was inverse. A possible explanation could be suggested by the work of Kukier et al. (2004), showing that the liming of a natural serpentine soil produced a reduction in Ni accumulation in two Odontarrhena species, likely due to an increased Ni binding by ferric oxide as soil pH raised.
Considering all populations, no other soil-plant relationships were found for the other metals, probably because the five species behave in a similar way only for Ni, which is invariably hyperaccumulated. Restricting the analysis to the 11 population samples of O. chalcidica, the significantly positive relationship between Nisoil-Nishoot observed here suggested that the wide variation in shoot Ni concentrations is of a predominantly environmental origin. This implies that populations of this species can be a useful tool in mineral nickel prospecting, as previously found for O. bertolonii (Galardi et al. 2007). Nevertheless, further research is necessary to assess a possible role of intra-specific genetic variation in such variability, since, for example, large differences in Ni shoot concentration were reported in more than 100 accessions of O. muralis grown on the same serpentine soil (Li et al. 2003).
Regarding the other elements, Mg and Ca were also accumulated in plant shoots in relation to their concentrations in the soil, although shoot concentrations were lower than the substrate for Mg and higher for Ca. The environmental control on the shoot ionome is therefore likely to also occur for these two elements, without excluding a possible role of genetic variation. Hence, the exclusion of Mg, in any case proportional to its soil concentration, could account for the positive relationship between Ni and Mg concentrations in the shoots, since the two elements were also correlated in the substrate. For Ca, its proportional accumulation with respect to soil concentration, as in the case of Ni, probably contributed to drive the inverse relationship between these two elements in the shoots. Actually, a negative relationship between Casoil-Niplant was already predicted for Balkan populations of O. chalcidica and attributed to competition for Ca (Bani et al. 2009). In our case, the relationship Casoil-Niplant was linear and without a ‘saturating effect’ of the soil Ca on the plant Ni, probably because of our larger data set and unambiguous taxon sampling. This negative relationship could also be one of the factors behind the inverse relationship between Ca/Mgsoil and Niplant in our plant samples from the field.
Conclusions
Albanian ultramafic outcrops, widely distributed across the country, showed a highly variable soil metal composition, with significant differences between the sites. All the Ni-hyperaccumulating taxa of Odontarrhena analyzed showed substantial differences in mineral element concentrations. Despite the large number of uncontrolled factors involved in field studies, our work highlighted, for the first time, a positive and linear relationship between soil Ni concentration and shoot Ni concentration. Remarkably, this relationship was not affected by species identity. Furthermore, new relationships between elements in soils and plants were found in the O. chalcidica populations, with an environmental fingerprint for the shoot metal composition not only for Ni, but also for Ca and Mg.
References
Baker AJM (1981) Accumulators and excluders – strategies in the response of plants to heavy metals. J Plant Nutr 3:643–654
Bani A, Echevarria G, Mullaj A, Reeves RD, Morel JL, Sulçe S (2009) Ni hyperaccumulation by Brassicaceae in serpentine soils of Albania and NW Greece. Northeast Nat 16(Special Issue 5):385–404
Bani A, Pavlova D, Echevarria G, Mullaj A, Reeves RD, Morel JL, Sulçe S (2010) Nickel hyperaccumulation by species of Alyssum and Thlaspi (Brassicaceae) from ultramafic soils of the Balkans. Bot Serbica 34(1):3–14
Bani A, Imeri A, Echevarria G, Pavlova D, Reeves RD, Morel JL, Sulçe S (2013) Nickel hyperaccumulation in the serpentine flora of Albania. Fresenius Environ Bull 22:1792–1801
Bani A, Echevarria G, Zhang X, Benizri E, Laubie B, Morel JL, Simonnot MO (2015) The effect of plant density in nickel-phytomining field experiments with Alyssum murale in Albania. Aust J Bot 63:72–77
Bani A, Echevarria G, Pavlova D, Shallari S, Morel JL, Sulçe S (2018) Element Case Studies: Nickel. In: Van der Ent A, Echevarria G, Baker AJM, Morel JL (eds) Agromining: farming for metals: extracting unconventional resources using plants. Springer International Publishing, Cham, pp 221–232
Brooks RR (1987) Serpentine and its vegetation: a multidisciplinary approach. Dioscorides Press, Portland
Brooks RR, Lee J, Reeves RD, Jaffré T (1977) Detection of nickeliferous rocks by analysis of herbarium specimens of indicator plants. J Geochem Explor 7:49–57
Cecchi L, Gabbrielli R, Arnetoli M, Gonnelli C, Hasko A, Selvi F (2010) Evolutionary lineages of Ni-hyperaccumulation and systematics in European Alysseae (Brassicaceae): evidence from nrDNA sequence data. Ann Bot 106:751–767
Cecchi L, Bettarini I, Colzi I, Coppi A, Echevarria G, Pazzagli L, Bani A, Gonnelli C, Selvi F (2018) The genus Odontarrhena (Brassicaceae) in Albania: taxonomy and nickel accumulation in a critical group of metallophytes from a major serpentine hot-spot. Phytotaxa 351(1):1–28
Chaney RL, Angle JS, McIntosh MS, Reeves RD, Li YM, Brewer EP, Chen KY, Roseberg JR, Perner H, Synkowski EC, Broadhurst CL, Wang S, Baker AJM (2005) Using hyperaccumulator plants to phytoextract soil Ni and cd. Z Naturforsch 60C:190–198
Deng THB, van der Ent A, Tang YT, Sterckeman T, Echevarria G, Morel JL, Qiu RL (2018) Nickel hyperaccumulation mechanisms: a review on the current state of knowledge. Plant Soil 423:1–11
Dilek Y, Furnes H (2009) Structure and geochemistry of Tethyan ophiolites and their petrogenesis in subduction rollback systems. Lithos 113:1–20
Galardi F, Mengoni A, Pucci S, Barletti L, Massi L, Barzanti R, Gabbrielli R, Gonnelli C (2007) Intra-specific differences in mineral element composition in the Ni-hyperaccumulator Alyssum bertolonii: a survey of populations in nature. Environ Exp Bot 60:50–56
Gonnelli C, Renella G (2012) Chromium and nickel. In: Alloway BJ (ed) Heavy metals in soils. Springer, Dordrecht, pp 313–333
Hartvig P (2002) Alyssum. In: Strid A, Tan K (eds) Flora Hellenica 2. Gantner Verlag, Ruggell, pp 199–224
Hegi G (1986) Illustrierte Flora von Mittel-Europa, 3rd rev. ed., 4 (1). Verlag, Berlin-Hamburg, 70 pp
Ingle RA, Mugford ST, Rees JD, Campbell MM, Smith JAC (2005) Constitutively high expression of the histidine biosynthetic pathway contributes to nickel tolerance in hyperaccumulator plants. Plant Cell 17:2089–2106
Kazakou E, Dimitrakopoulos PG, Baker AJM, Reeves RD, Troumbis AY (2008) Hypotheses, mechanisms and trade-offs of tolerance and adaptation to serpentine soils: from species to ecosystem level. Biol Rev 83:495–508
Kidd PS, Bani A, Benizri E, Gonnelli C, Hazotte C, Kisser J, Konstantinou M, Kuppens T, Dimitris K, Laubie B, Malina R, Morel JL, Olcay H, Pardo T, Pons MN, Prieto-Fernández A, Puschenreiter M, Quintela-Sabarís C, Ridard C, Rodríguez-Garrido B, Rosenkranz T, Rozpądek P, Saad R, Selvi F, Simonnot M-O, Tognacchini A, Turnau K, Wazny R, Witters N, Echevarria G (2018) Developing sustainable agromining systems in agricultural ultramafic soils for nickel recovery. Front Environ Sci 6:44
Krämer U, Cotter-Howells JD, Charnock JM, Baker AJM, Smith JAC (1996) Free histidine as a metal chelator in plants that accumulate nickel. Nature 379:635–638
Kruckeberg AR (1954) The ecology of serpentine soils. III. Plant species in relation to serpentine soils. Ecology 35:267–274
Kruckeberg AR (2002) Geology and plant life. University Press, Washington
Kruckeberg AR, Kruckeberg AL (1990) Endemic metallophytes: their taxonomic, genetic and evolutionary attributes. In: Shaw AJ (ed) Heavy metal tolerance in plants: evolutionary aspects. CRC Press Inc, Boca Raton, pp 301–312
Kukier U, Peters CA, Chaney RL, Angle JS, Roseberg RJ (2004) The effect of pH on metal accumulation in two Alyssum species. J Environ Qual 32:2090–2102
Lefèbvre C, Vernet P (1990) Microevolutionary processes on contaminated deposits. In: Shaw AJ (ed) Heavy metal tolerance in plants: evolutionary aspects. CRC Press Inc, Boca Raton, pp 286–297
Li Y-M, Chaney RL, Brewer EP, Angle JS, Nelkin JP (2003) Phytoextraction of nickel and cobalt by hyperaccumulator Alyssum species grown on nickel-contaminated soils. Environ Sci Technol 37:1463–1468
Marschner H (1995) Mineral nutrition of higher plants. Academic Press, London
Minguzzi C, Vergnano O (1948) Il contenuto di nichel nelle ceneri di Alyssum bertolonii. Atti Soc Tosc Sci Nat 55:49–74
Nkrumah PN, Baker AJM, Chaney RL, Erskine PD, Echevarria G, Morel JL, van der Ent A (2016) Current status and challenges in developing nickel phytomining: an agronomic perspective. Plant Soil 406:55–69
Nkrumah PN, Chaney RL, Morel JL (2018) Agronomy of ‘metal crops’ used in agromining. In: Van der Ent A, Echevarria G, Baker AJM, Morel JL (eds) Agromining: farming for metals: extracting unconventional resources using plants. Springer International Publishing, Cham, pp 19–38
Pignattelli S, Colzi I, Buccianti A, Cecchi L, Arnetoli M, Monnanni R, Gabbrielli R, Gonnelli C (2012) Exploring element accumulation patterns of a metal excluder plant naturally colonizing a highly contaminated soil. J Hazard Mater 227-228:362–369
Reeves RD, Baker AJM, Kelepertsis A (1997) The distribution and biogeochemistry of some serpentine plants of Greece. In: Jaffré T, Reeves RD, Becquer T (eds) Ecologie des Milieux sur Roches Ultramafiques et sur Sols Metallifères, ORSTOM, Noumea, Documents Scientifiques et Techniques No. III/2:205–207
Reeves RD, Baker AJM, Jaffré T, Erskine PD, Echevarria G, van der Ent A (2018) A global database for plants that hyperaccumulate metal and metalloid trace elements. New Phytol 218(2):407–411
Rešetnik I, Satovic Z, Schneeweiss GM, Liber Z (2013) Phylogenetic relationships in Brassicaceae tribe Alysseae inferred from nuclear ribosomal and chloroplast DNA sequence data. Mol Phylogenet Evol 69:772–786
Robinson BH, Chiarucci A, Brooks RR, Petit D, Kirkman JH, Gregg PEH, De Dominicis V (1997) The nickel hyperaccumulator plant Alyssum bertolonii as a potential agent for phytoremediation and phytomining of nickel. J Geochem Explor 59:75–86
Rusterholz HP, Aydin D, Bauer B (2012) Population structure and genetic diversity of relict populations of Alyssum montanum on limestone cliffs in the northern Swiss Jura mountains. Alp Bot 122:109–117
Salihaj M, Bani A, Shahu E, Benizri E, Echevarria G (2018) Metal accumulation by the ultramafic flora of Kosovo. Ecol Res 33(4):687–703
Selvi F, Carrari E, Colzi I, Coppi A, Gonnelli C (2017) Responses of serpentine plants to pine invasion: vegetation diversity and nickel accumulation in species with contrasting adaptive strategies. Sci Total Environ 595:72–80
Shallari S, Schwartz C, Hasko A, Morel JL (1998) Heavy metals in soils and plants of 316 serpentine and industrial sites of Albania. Sci Total Environ 209:133–142
Simonnot M-O, Vaughan J, Laubie B (2018) Processing of bio-ore to products. In: Van der Ent A, Echevarria G, Baker AJM, Morel JL (eds) Agromining: farming for metals: extracting unconventional resources using plants. Springer International Publishing, Cham, pp 39–52
Španiel S, Kempa M, Salmerón-Sánchez E, Fuertes-Aguilar J, Francisco Mota J, Al-Shehbaz IA, German DA, Olšavská K, Šingliarová B, Zozomová-Lihová J, Marhold K (2015) AlyBase – database of names, chromosome numbers, and ploidy levels of Alysseae (Brassicaceae), with a new generic concept of the tribe. Plant Syst Evol 301:2463–2491
Tatic B, Veljovic V (1992) Distribution of serpentinized massives on the Balkan peninsulas and their ecology. In: Roberts BA, Proctor J (eds) In: the ecology of areas with Serpentinized rocks- a world view. Kluwer Academic Publishers, Dordrecht, pp 199–215
Tumi AF, Mihailović N, Gajić BA, Niketić M, Tomović G (2012) Comparative study of hyperaccumulation of nickel by Alyssum murale. Populations from the ultramafics of Serbia. Pol J Environ Stud 21:1855–1866
van der Ent A, Baker AJM, Reeves RD, Pollard AJ, Schat H (2013a) Hyperaccumulators of metal and metalloid trace elements: facts and fiction. Plant Soil 362:319–334
van der Ent A, Baker AJM, van Balgooy MMJ, Tjoa A (2013b) Ultramafic nickel laterites in Indonesia (Sulawesi, Halmahera): mining, nickel hyperaccumulators and opportunities for phytomining. J Geochem Explor 128:72–79
van der Ent A, Baker AJM, Reeves RD, Chaney RL, Anderson CWN, Meech JA, Erskine PD, Simonnot MO, Vaughan J, Morel JL, Echevarria G, Fogliani B, Rongliang Q, Mulligan DR (2015) Agromining: farming for metals in the future? Environ Sci Technol 49:4773–4780
van der Ent A, Cardace D, Tibbett M, Echevarria G (2018) Ecological implications of pedogenesis and geochemistry of ultramafic soils in Kinabalu Park (Malaysia). Catena 160:154–169
Verbruggen N, Hermans C, Schat H (2009) Molecular mechanisms of metal hyperaccumulation in plants. New Phytol 181:759–776
Whiting SN, Reeves RD, Richards D, Johnson MS, Cooke JA, Malaisse F, Paton A, Smith JAC, Angle JS, Chaney RL, Ginocchio R, Jaffré T, Johns R, McIntyre T, Purvis OW, Salt DE, Schat H, Baker AJM (2004) Research priorities for conservation of metallophyte biodiversity and their potential for restoration and site remediation. Restor Ecol 12:106–116
Wójcik M, Gonnelli C, Selvi F, Dresler S, Rostański A, Vangronsveld J (2017) Metallophytes of serpentine and calamine soils - their unique ecophysiology and potential for phytoremediation. Adv Bot Res 83:1–42
Zhang X, Laubie B, Houzelot V, Plasari E, Echevarria G, Simonnot M-O (2016) Increasing purity of ammonium nickel sulfate hexahydrate and production sustainability in a nickel phytomining process. Chem Eng Res Des 106:26–32
Acknowledgments
This study was carried out in the framework of Agronickel (ERA-NET Cofund-supported FACCE SURPLUS project). We thank Professor Alan Baker (Universities of Melbourne and Queensland, Australia) for his help in improving the text of this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Fangjie Zhao.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 45 kb)
Rights and permissions
About this article
Cite this article
Bettarini, I., Colzi, I., Coppi, A. et al. Unravelling soil and plant metal relationships in Albanian nickel hyperaccumulators in the genus Odontarrhena (syn. Alyssum sect. Odontarrhena, Brassicaceae). Plant Soil 440, 135–149 (2019). https://doi.org/10.1007/s11104-019-04077-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04077-y