Abstract
Aims
In tropical rainforests, seedling performance at fine ecological scales has traditionally been explained by seed dispersal limitation and negative density dependence. Soil properties have strong patch structures at fine scales, and we investigated whether such heterogeneity contributes to seedling coexistence.
Methods
From January 2011 to January 2014, in a one-hectare area of tropical rainforest in Southwest China, we sampled seedlings from ninety-nine 2-m2 quadrats, and measured light condition (canopy openness), and the height (>10 cm) of seedlings at bi-monthly intervals. In addition, we measured soil pH, total nitrogen, ammonium, nitrate, and available phosphorus and potassium three times during the survey period. These data were used to examine the effects of soil properties and light on seedling spatial distribution, survival, and growth.
Results
Seedling spatial distribution was significantly associated with light and spatial heterogeneity in all the examined soil properties except available K. These fine scale soil properties and light heterogeneity also promoted seedling growth, whereas they had no discernible effects on seedling survival.
Conclusion
This study highlights the effects of fine scale soil property heterogeneity on seedling spatial distribution and growth, and indicates their importance in the maintenance of seedling biodiversity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The seedling stage is recognized as a bottleneck for plant regeneration (Rodríguez-Pérez and Traveset 2007), community structuring, and maintenance of diversity (Grubb 1977; Johnson et al. 2017). Seedlings experience more size-dependent asymmetric competition for light than adult trees do, and, thus, many studies have concluded that seedling survival and growth are strongly limited by light (Augspurger 1984; Brown et al. 1999; Balderrama and Chazdon 2005). Soil is also considered an important co-limiting factor for seedling recruitment (Palow and Oberbauer 2009). Both field observations and nutrient addition experiments have provided evidence that the addition of nutrients can promote growth and survival (Lawrence 2003; Holste et al. 2011). However, there is still no consensus on the role of soil properties in seedling establishment. For example, Valladares et al. (2016) argued in their review paper the probable existence of a neutral process for seedling establishment in shaded environments. Johnson et al. (2017) found that both canopy openness and soil conditions have no effect on seedling mortality, whereas Uriarte et al. (2018) identified positive effects of solar radiation and soil moisture on seedling regeneration. Thus, there is still a need for field conditions to be explicitly investigated in order to determine the effects of soil heterogeneity on seedling performance (Smith-Martin et al. 2017).
The regeneration niche encompasses not only seedling growth and survival, but also seedling spatial distribution (Grubb 1977). Given their immobility, the spatial distribution of seedlings determines future tree distribution, and thus future community structure. However, relatively few studies have comprehensively investigated whether the spatial distribution of seedlings in forest is caused by the niche process (Collins and Good 1987; Webb and Peart 2000; Norden et al. 2009; Bin et al. 2012; Tian et al. 2013). Moreover, although some of these studies have focused on physical habitat variables (Collins and Good 1987; Webb and Peart 2000; Tian et al. 2013) or used modeling methods, they have generally not included measurements of environment variables (Bin et al. 2012). Only Norden et al. (2009) measured soil properties in a neo-tropical rainforest, including pH and the elemental composition of carbon (C), nitrogen (N), calcium (Ca), magnesium (Mg), potassium (K), aluminum (Al), and iron (Fe), and found that they have weaker effects on seedling distribution than on seedling survival. However, a larger number of studies are needed in order to make generalizations.
Scale is fundamental to ecological processes (Levin 1992), and the mechanisms that contribute to the maintenance of biodiversity are also scale dependent (Ettema and Wardle 2002). Soil heterogeneity, which is critical for species coexistence, occurs at varying scales, including landscape-, meso-, local-, and fine scales (John et al. 2007; Yavitt et al. 2009; Xia et al. 2015; Chadwick and Asner 2016). Many plant environmental association studies are conducted from the landscape scale to meso- and local scales to examine the importance of soil heterogeneity in species diversity (John et al. 2007). However, although often conducted at a specific scale, studies focused on seedling environment association rarely take into consideration of scales effect (Harms et al. 2004). Furthermore, although some studies have investigated soil heterogeneity at the fine scale and strong patch structures were also detected, such studies have rarely evaluated seedling environmental associations at the fine scale (Wang et al. 2007; Xia et al. 2016). One reason for this may be that variations of seedling performance within short distance are strongly affected by negative density dependence, dispersal limitation, or both. A further reason may be that soil properties are expected to vary only slightly at a fine scale, and thus are assumed to have a lesser pronounced effect on seedling performance than other factors. However, soil heterogeneity, with regard to nutrient patch structure, which also occurs at a fine scale, can remain consistent through time (Xia et al. 2016) and the ranges of nutrient variation in a 1 ha area are in the same magnitude as those at the local scale (John et al. 2007; Xia et al. 2015). Given these strong seedling-environmental associations detected at the local scale, we predict that fine scale heterogeneity of soil properties may also affect seedling regeneration.
Previously, we conducted an extensively studied of the spatial variations in fine scale soil properties in a 1 ha area of rainforest in South Asia, and found that soil properties including pH, total N, ammonium-N (NH4-N), nitrate-N (NO3-N), and available P and K all had significant patch-specific structures at patch sizes of 10–30 m (Xia et al. 2015), and that the patch structures of these measured soil properties were highly or moderately consistent through time (Xia et al. 2016). In the present study, we sought to examine whether soil heterogeneity has a notable effect on local seedling spatial distribution, survival, and growth at such a fine scale. To this end, we surveyed seedling spatial location, survival, and growth dynamics for 3 years, and also monitored the nutrients and light conditions of each quadrat. We proposed a hypothesis that soil resources are an important component of seedling niches in terms of seedling spatial distribution, growth and survival.
Methods
Study site
This study was conducted in a tropical seasonal rainforest in Yunnan, Southwest China, situated in a 20-ha dynamic forest plot (21°37′08″N, 101°35′07″E), belonging to the Center for Tropical Forest Science – Forest Global Earth Observatory Network (Xia et al. 2015). The canopy of the forest dominated by Parashorea chinensis, whereas Pittosporopsis kerrii is the dominant understory species (Cao et al. 2008). This area receives a mean annual rainfall of 1532 mm and has an annual mean temperature of 21.0 °C (Zhu 2006), with a rainy season from May to October. The soil is lateritic, which has developed from siliceous rocks (Cao et al. 2006).
Data collection
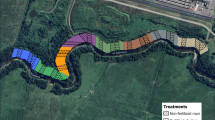
On the 1 ha study site, 99 quadrats were set with a mean distance of 10 m between the adjacent quadrats (Fig. 1a). Within each quadrat, a central 1 m2 area was set as the soil sampling cell, and two 1 × 1 m seedling monitoring cells were set close to this area (Fig. 1b). The soil data collection procedure used in this study followed a previously established protocol (Xia et al. 2015, 2016). In summary, the soil properties assessed in April 2011, August 2011, and March 2012 were pH, total N, NH4-N, NO3-N, and available P and K from the soil collected at a depth of 0–10 cm from each quadrat (for basic soil properties information at each sampling time, please see Xia et al. 2016; for a summary soil properties variation during the entire sampling period, please see Suppl. Table 1). From January 2011 to January 2014, we conducted a bi-monthly seedling assessment (19 census measurements) in which all seedlings with diameter at breast height < 1 cm and height ≥ 10 cm were identified to the species level and their heights were measured. During each seedling census, we also monitored canopy openness using a hemispheric camera placed 1 m aboveground level in each seedling monitoring cell. The canopy photos were analyzed using the Gap Light Analyzer software (Frazer et al. 1999) to obtain values of the percentage (%) canopy openness.
Statistical analysis
In total, 2442 individual seedlings of 157 species were monitored and identified during the 3 years of investigation. We selected a subset of species with >40 individuals recorded during the survey period to perform seedling niche, survival, and growth analyses using R (R Core Team 2017) as detailed below.
We initially performed niche analysis, in which all examined soil properties and canopy openness were selected as environmental variables. We assessed the significance of each species’ niche differentiation in terms of the examined variables by employing outlying mean index (OMI) analysis (Doledéc et al. 2000) in the ade4 package using niche function (Dray and Dufour 2007), where OMI measures the distance between the mean resource conditions used by target species (species centroid) and the mean resource conditions of all species, and OMI analysis places species along resource conditions (Doledéc et al. 2000). We then conducted a species ordination analysis using the vegan package (Oksanen et al. 2015) to investigate the patterns of species sorting in terms of soil properties and canopy openness.
For seedling survival rate analysis, if a seedling was still present at the last census in January 2014, then it was coded as having survived and if otherwise, it was coded as dead. We then assessed whether canopy openness and soil properties affected the survival rate using a Bayesian generalized linear mixed effect model with binomial errors in the brms package (Bürkner 2017) to interface with Stan probabilistic programming language (Carpenter et al. 2017). Because there was co-linearity exist between the measured soil properties, the effect of these variables on seedling survival was examined on a one-by-one basis. The matrix of distance between quadrats was added as a random effect to correctly model spatial autocorrelation structure. To consider the effect of seedling size and observation time on their survival rate, the initial height of each seedling and initial survey time were included as varying intercepts random effects. To assess whether species have specific response to each measured variable, we treated species as a random variable with both varying intercept and varying slope model (to determine species with the same or varying response to a measured environmental variable, respectively). We also employed a default flat prior for fixed effects, and the default iteration length of Markov chains was set as 2000 with the first 1000 iteration as warmup. The default number of chains was set to four chains, and all other parameters were set default. The best model was selected based on the WAIC value.
For growth rate analysis, we initially calculated the relative growth rate of each seedling for each investigation interval, and then matched it with the corresponding canopy openness data and nearest time soil survey data. Thereafter, we assessed whether canopy openness and soil properties have effect on the seedling growth rate using a Bayesian generalized linear mixed effect model with zero inflated beta distribution errors in the brms package (Bürkner 2017). All model setting and selection criteria were the same as those used for survival analysis, with the exception of the random effect, for which we removed the initial survey time and added individual ids to correctly estimate time series autocorrelation in repeat height measurements.
Results
For analysis, we selected 14 species (eight tree and six liana species) with abundance greater than 40 (Table 1), which comprised a total of 1495 individuals. OMI analysis showed that among the14 species, P. chinensis (Pa.Ch), P. kerrii (Pi.Ke), Diospyros nigrocortex (Di.Ni), and Acacia delavayi (Ac.De) exhibited significant niche differentiation (Table 2). In addition, Mezzettiopsis creaghii (Me.Cr) showed marginally significant niche differentiation. Pa.Ch-occupied patches had high available P and total N (Fig. 2 and Table 3). Pi.Ke-occupied patches had low pH and high NH4-N values. Di.Ni- and Me.Cr-occupied patches had high pH values but were low in all nutrients, whereas Ac.De-occupied patches characterized by high NO3-N values. The other species occupied similar microhabitats that did not differ significantly from one another.
Canopy openness contributed significantly to niche differentiation of the examined species (Fig. 2 and Suppl. Table 2). With the exception of available K, all measured soil properties contributed to species separation (Fig. 2 and Suppl. Table 2).
Although canopy openness significantly affected seedling growth, it did not significantly affect survival (Tables 4 and 5). Among the measured soil properties, pH, N (NH4-N), and available P and K all significantly affected seedling growth but not survival (Tables 4 and 5).
Although differences in growth rate were detected between species (Table 1, Suppl. Table 3–9), there was little variation in their growth and survival responses to differences in canopy openness and soil properties (Tables 6 and 7), only detected two species (By.Gr and St.Wa) showing more sensitive growth response to soil NH4-N (Table 6, Suppl. Table 6).
Discussion
In this study, we found that heterogeneity in fine scale soil properties is sufficient to cause spatial niche differentiation among species in a tropical rainforest (Fig. 2). Traditionally, seedling distribution patterns at the fine scale have mainly been explained in terms of the Janzen-Connell hypothesis (Lin et al. 2012; Comita et al. 2014) or dispersal limitation (Seidler and Plotkin 2006; Pinto and MacDougall 2010). However, in this study, we revealed a further perspective of fine scale seedling distribution, in which they are also associated with fine scale heterogeneity in soil properties and light.
The dominant canopy and understory species (Pa.Ch and Pi.Ke, respectively) were both found in nutrient rich patches (Fig. 2), and also showed niche separation, suggesting distinct nutrient requirements in canopy and understory species. Di.Ni and Me.Cr occupied in nutrient poor but high pH value areas; however, given that high pH is associated with low NH4-N, it was difficult to distinguish the individual effect of NH4-N and pH. Of the six liana species, only Ac.De showed significant niche separation, which may be because lianas are more likely to be opportunists (Sfair et al. 2018). It is noteworthy that although we could explain the pattern successfully using a niche theory framework, we did not claim to reject the alternative probability that seedling fine scale distribution determined by dispersal limitation (Seidler and Plotkin 2006), negative density dependence (Lin et al. 2012; Comita et al. 2014; Wu et al. 2016), or both, and we concede that these mechanisms may function concomitantly in determining seedling distribution.
We found that canopy openness strongly affected seedling growth (Table 4), thereby indicating that energy limits seedling growth. This result is consistent with the finding of previous studies (Balderrama and Chazdon 2005; Goodale et al. 2014). Among the six soil properties examined, pH, NH4-N, available P and K all significantly affected seedling growth (Table 4), thereby indicating multi-element limitation of seedling growth in the study plot. The effects of soil properties on seedling growth were, however, indistinct, as no clear pattern could be determined. Previously, a multi-site study in a wet tropical rainforest in Costa Rica also found that NH4-N, the sum of base cations, and P have ubiquitous effects on seedling growth (Holste et al. 2011). Whereas in contrast, a survey in a rainforest in Pasoh, Malaysia, found that P but not N, K, Ca, or Mg, affected seedling growth (Juliana et al. 2009). We thus conclude that the effect of soil properties may depend on nutrient limitation condition and many other unmeasured parameters. For instance, soil fertility effects are light dependent and largely disappear under shaded conditions (Peltzer and Wardle 2016). Furthermore, species functional groups, tree size, and neighborhood density may also modify the effect of soil nutrient efficiency (Zhang et al. 2016; Chou et al. 2018).
Among the three forms of N, NH4-N, but neither NO3-N nor total N, was found to affect seedling growth (Table 4). This is perhaps predictable, given that plants growing in wet tropical forest preferentially use NH4-N (Houlton et al. 2007). Because NH4-N is the dominant inorganic N form here (Suppl. Table 1), and plants only absorb a small proportion of organic N with small molecular weight (Jones et al. 2005), and thus detected no significant response to total N variation in the study plot.
A positive but non-significant effect of light on survival was also detected (Table 5), which contrast with the finding of a number of previous studies (Montgomery and Chazdon 2002; Balderrama and Chazdon 2005; Goodale et al. 2014). Only a few studies have identified a similar pattern between light and the survival of most species (Record et al. 2016). We suspect that seedling survival is affected by light but may be less sensitive to light compared with seedling growth. Firstly, the effect of light on seedling survival is already marginal significant (estimated coefficient of 0.21 and 95% CI of −0.03 to 0.43); Secondly, to verify this point, we employed another reasonable but less parsimonious model that let initial height effect nested in species effect in random factors, and then the significance of light on seedling survival was determined (Suppl. Table 10). Thirdly, the detected relationship may be dependent on the degree of the variation in light availability, with small variations have weak effects (Record et al. 2016). In the study plot used in the present study, the mature forest had a closed canopy with mean canopy openness of 2.59 ± 1.71%(SD), whereas both understory forest and gaps have been in included in previous studies in which significant light-related relationships have been detected (Balderrama and Chazdon 2005; Goodale et al. 2014).
Contrary to expectations, none of the soil nutrients examined in the present study had general effects on seedling survival (Table 5). Previously, it has been revealed that a lack of general seedling environmental associations may occur because different species have distinct responses to soil nutrients (Umaña et al. 2017). We considered this factor and employed a varying slope model in the random effect model to evaluate the effects of variations in species response, but were unable to detect any significant varying slope effect (Table 7). Thus, we found no evidence that the lack of a general seedling environment association in the study plot is because of distinct species response. One reason for this observation may be the extreme light limitation in the forest understory, which is a main survival limiting factor when compared with spatial heterogeneity (Beckage and Clark 2003). Other factors not considered in this study include abiotic factors, such as topography and water content, and biotic factors, including negative density dependency and associations with mycorrhiza fungal networks (Battaglia et al. 2000; Gehring 2003; LaManna et al. 2016). These factors may also mask the effect of soil properties on seedling survival, and thus our inability to detect any significant effect of soil properties on seedling survival.
For examined growth and survival of 14 species that response to six measured soil properties in the present study, only two species were found to have a more sensitive growth response to soil NH4-N (Table 6 and Suppl. Table 6). Thus, we assume that the seedlings in our study plot generally do not show a species-specific response to soil properties in terms of growth or survival. This finding runs counter to our hypothesis, because we expected that different species may be characterized by distinct growth and survival responses to fine scale heterogeneity in soil properties. In this regard, resource co-limitation theory may help us to understand this phenomenon (Bloom et al. 1985; Harpole et al. 2011). Given that light and nutrient limitations occur simultaneously in the understory of tropical rainforests, seedlings are only able to exploit a limited amount of light and soil nutrient resources. Under these multi-resource limited conditions, seedlings growth is restricted, and thus they fail to show any difference of resource utilization strategy. Accordingly, we were unable to observe any species-specific growth response was observed. Given that seedling survival is inherently associated with seedling resource utilizing strategy (Beckage and Clark 2003; Seiwa 2007; Inman-Narahari et al. 2014), the fact that there was no differentiation in such utilization in our study plot, means that we were unable to detect marked differences in seedling survival.
The finding of this study indicates that there is an alternative mechanism whereby biodiversity is maintained. Traditionally, negative density dependence and resource utilization differentiation have been considered the principal mechanisms by which species coexisted. In the present study, we found that for all species examined, fine scale limitations in soil nutrients and light, result in the slow growth of seedlings and non-differential seedling survival, Thereby helpful to maintaining tropical rainforest biodiversity.
Conclusions
The mechanisms that underlie the maintenance of biodiversity are key issues in ecology, and the findings of this study provide new insights on these mechanisms from a seedling regeneration perspective. Our study included scale as a property of heterogeneity, and focused on the relationship of soil heterogeneity and seedling performance at the fine scale. We discovered that fine scale seedling distribution, which is traditionally explained by the Janzen-Connell hypothesis, dispersal limitation, or both, is also associated with the heterogeneity of soil property. The co-occurrence of limitations in soil properties and light conditions, retards growth of the seedling of all species, and it does not significantly influence seedling survival, thereby having a beneficial effect of maintaining seedling diversity.
References
Augspurger CK (1984) Light requirements of neotropical tree seedlings: a comparative study of growth and survival. J Ecol 72:777–795. https://doi.org/10.2307/2259531
Balderrama SIV, Chazdon RL (2005) Light-dependent seedling survival and growth of four tree species in Costa Rican second-growth rain forests. J Trop Ecol 21:383–395. https://doi.org/10.1017/S026646740500235X
Battaglia LL, Fore SA, Sharitz RR (2000) Seedling emergence, survival and size in relation to light and water availability in two bottomland hardwood species. J Ecol 88:1041–1050. https://doi.org/10.1046/j.1365-2745.2000.00518.x
Beckage B, Clark JS (2003) Seedling survival and growth of three forest tree species: the role of spatial heterogeneity. Ecology 84:1849–1861. https://doi.org/10.1890/0012-9658(2003)084[1849:SSAGOT]2.0.CO;2
Bin Y, Lin G, Li B, Wu L, Shen Y, Ye W (2012) Seedling recruitment patterns in a 20 ha subtropical forest plot: hints for niche-based processes and negative density dependence. Eur J For Res 131:453–461. https://doi.org/10.1007/s10342-011-0519-z
Bloom AA, Chapin F, Mooney H (1985) Resource limitation in plants--an economic analogy. Annu Rev Ecol Syst 16:363–392. https://doi.org/10.1146/annurev.ecolsys.16.1.363
Brown N, Press M, Bebber D (1999) Growth and survivorship of dipterocarp seedlings: differences in shade persistence create a special case of dispersal limitation. Philos Trans R Soc B Biol Sci 354:1847–1855. https://doi.org/10.1098/rstb.1999.0526
Bürkner P-C (2017) brms : an R package for Bayesian multilevel models using Stan. J Stat Softw 80. https://doi.org/10.18637/jss.v080.i01
Cao M, Zou X, Warren M, Zhu H (2006) Tropical forests of Xishuangbanna, China. Biotropica 38:306–309. https://doi.org/10.1111/j.1744-7429.2006.00146.x
Cao M, Zhu H, Wang H, et al (2008) Xishuangbanna tropical seasonal rainforest dynamics plot: tree distribution maps, diameter tables and species documentation. Yunnan Science and Technology Press, Kunming
Carpenter B, Gelman A, Hoffman MD, et al (2017) Stan : a probabilistic programming language. J Stat Softw 76. https://doi.org/10.18637/jss.v076.i01
Chadwick KD, Asner GP (2016) Tropical soil nutrient distributions determined by biotic and hillslope processes. Biogeochemistry 127:273–289. https://doi.org/10.1007/s10533-015-0179-z
Chou CB, Hedin LO, Pacala SW (2018) Functional groups, species and light interact with nutrient limitation during tropical rainforest sapling bottleneck. J Ecol 106:157–167. https://doi.org/10.1111/1365-2745.12823
Collins SL, Good RE (1987) The seedling regeneration niche: habitat structure of tree seedlings in an oak-pine forest. Oikos 48:89–98. https://doi.org/10.2307/3565692
Comita LS, Queenborough SA, Murphy SJ, Eck JL, Xu K, Krishnadas M, Beckman N, Zhu Y, Gómez-Aparicio L (2014) Testing predictions of the Janzen-Connell hypothesis: a meta-analysis of experimental evidence for distance- and density-dependent seed and seedling survival. J Ecol 102:845–856. https://doi.org/10.1111/1365-2745.12232
Doledéc S, Chessel D, Gimaret-Carpentier C et al (2000) Niche separation in community analysis: a new method. Ecology 81:2914–2927. https://doi.org/10.1890/0012-9658(2000)081
Dray S, Dufour A-B (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22:1–20
Ettema CH, Wardle DA (2002) Spatial soil ecology. Trends Ecol Evol 17:177–183. https://doi.org/10.1016/S0169-5347(02)02496-5
Frazer GW, Canham CD, Lertzman KP (1999) Gap Light Analyzer (GLA), Version 2.0: imaging software to extract canopy structure and gap light transmission indices from true-colour fisheye photographs, users manual and program documentation. Simon Fraser University, Burnaby, British Columbia, and the Institute of Ecosystem Studies, Millbrook, New York, https://doi.org/10.1017/S1461145799001625
Gehring CA (2003) Growth responses to arbuscular mycorrhizae by rain forest seedlings vary with light intensity and tree species. Plant Ecol 167:127–139. https://doi.org/10.1023/A:1023989610773
Goodale UM, Berlyn GP, Gregoire TG, Tennakoon KU, Ashton MS (2014) Differences in survival and growth among tropical rain forest pioneer tree seedlings in relation to canopy openness and herbivory. Biotropica 46:183–193. https://doi.org/10.1111/btp.12088
Grubb PJ (1977) The maintenance of species richness in plant communities: the importance of the regeneration niche. Biol Rev 52:107–145. https://doi.org/10.1111/j.1469-185X.1977.tb01347.x
Harms KE, Powers JS, Montgomery RA (2004) Variation in small sapling density, understory cover, and resource availability in four Neotropical forests. Biotropica 36:40–51. https://doi.org/10.1111/j.1744-7429.2004.tb00294.x
Harpole WS, Ngai JT, Cleland EE, Seabloom EW, Borer ET, Bracken MES, Elser JJ, Gruner DS, Hillebrand H, Shurin JB, Smith JE (2011) Nutrient co-limitation of primary producer communities. Ecol Lett 14:852–862. https://doi.org/10.1111/j.1461-0248.2011.01651.x
Holste EK, Kobe RK, Vriesendorp CF (2011) Seedling growth responses to soil resources in the understory of a wet tropical forest. Ecology 92:1828–1838. https://doi.org/10.1890/10-1697.1
Houlton BZ, Sigman DM, Schuur EAG, Hedin LO (2007) A climate-driven switch in plant nitrogen acquisition within tropical forest communities. Proc Natl Acad Sci U S A 104:8902–8906. https://doi.org/10.1073/pnas.0609935104
Inman-Narahari F, Ostertag R, Asner GP, Cordell S, Hubbell SP, Sack L (2014) Trade-offs in seedling growth and survival within and across tropical forest microhabitats. Ecol Evol 4:3755–3767. https://doi.org/10.1002/ece3.1196
John R, Dalling JW, Harms KE, Yavitt JB, Stallard RF, Mirabello M, Hubbell SP, Valencia R, Navarrete H, Vallejo M, Foster RB (2007) Soil nutrients influence spatial distributions of tropical tree species. Proc Natl Acad Sci 104:864–869. https://doi.org/10.1073/pnas.0604666104
Johnson DJ, Condit R, Hubbell SP, Comita LS (2017) Abiotic niche partitioning and negative density dependence drive tree seedling survival in a tropical forest. Proc R Soc B Biol Sci 284:20172210. https://doi.org/10.1098/rspb.2017.2210
Jones DL, Healey JR, Willett VB, Farrar JF, Hodge A (2005) Dissolved organic nitrogen uptake by plants - an important N uptake pathway? Soil Biol Biochem 37:413–423. https://doi.org/10.1016/j.soilbio.2004.08.008
Juliana WW, Burslem D, Swaine M (2009) Nutrient limitation of seedling growth on contrasting soil from pasoh forest reserve. J Trop For Sci 21:316–327
LaManna JA, Walton ML, Turner BL, Myers JA (2016) Negative density dependence is stronger in resource-rich environments and diversifies communities when stronger for common but not rare species. Ecol Lett 19:657–667. https://doi.org/10.1111/ele.12603
Lawrence D (2003) The response of tropical tree seedlings to nutrient supply: meta-analysis for understanding a changing tropical landscape. J Trop Ecol 19:239–250. https://doi.org/10.1017/S0266467403003274
Levin SA (1992) The problem of pattern and scale in ecology. Ecology 73:1943–1967. https://doi.org/10.2307/1941447
Lin L, Comita LS, Zheng Z, Cao M (2012) Seasonal differentiation in density-dependent seedling survival in a tropical rain forest. J Ecol 100:905–914. https://doi.org/10.1111/j.1365-2745.2012.01964.x
Montgomery RA, Chazdon RL (2002) Light gradient partitioning by tropical tree seedlings in the absence of canopy gaps. Oecologia 131:165–174. https://doi.org/10.1007/s00442-002-0872-1
Norden N, Chave J, Belbenoit P, Caubère A, Châtelet P, Forget PM, Riéra B, Viers J, Thébaud C (2009) Interspecific variation in seedling responses to seed limitation and habitat conditions for 14 Neotropical woody species. J Ecol 97:186–197. https://doi.org/10.1111/j.1365-2745.2008.01444.x
Oksanen J, Blanchet FG, Kindt R, et al (2015) Vegan: community ecology package. http://cran.r-project.org/package=vegan
Palow DT, Oberbauer SF (2009) Soil type affects seedling shade response at low light for two Inga species from Costa Rica. Plant Soil 319:25–35. https://doi.org/10.1007/s11104-008-9846-3
Peltzer DA, Wardle DA (2016) Soil fertility effects on tree seedling performance are light-dependent: evidence from a long-term soil chronosequence. Oikos 125:1121–1133. https://doi.org/10.1111/oik.02878
Pinto SM, MacDougall AS (2010) Dispersal limitation and environmental structure interact to restrict the occupation of optimal habitat. Am Nat 175:675–686. https://doi.org/10.1086/652467
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://doi.org/10.3390/biomimetics2040019
Record S, Kobe RK, Vriesendorp CF, Finley AO (2016) Seedling survival responses to conspecific density, soil nutrients, and irradiance vary with age in a tropical forest. Ecology 97:2406–2415. https://doi.org/10.1002/ecy.1458
Rodríguez-Pérez J, Traveset A (2007) A multi-scale approach in the study of plant regeneration: finding bottlenecks is not enough. Perspect Plant Ecol Evol Syst 9:1–13. https://doi.org/10.1016/j.ppees.2007.07.003
Seidler TG, Plotkin JB (2006) Seed dispersal and spatial pattern in tropical trees. PLoS Biol 4:2132–2137. https://doi.org/10.1371/journal.pbio.0040344
Seiwa K (2007) Trade-offs between seedling growth and survival in deciduous broadleaved trees in a temperate forest. Ann Bot 99:537–544. https://doi.org/10.1093/aob/mcl283
Sfair JC, Weiser V d L, Martins FR et al (2018) Species traits and abundance influence the organization of liana-tree antagonistic interaction. Austral Ecol 43:236–241. https://doi.org/10.1111/aec.12560
Smith-Martin CM, Gei MG, Bergstrom E, Becklund KK, Becknell JM, Waring BG, Werden LK, Powers JS (2017) Effects of soil type and light on height growth, biomass partitioning, and nitrogen dynamics on 22 species of tropical dry forest tree seedlings: comparisons between legumes and nonlegumes. Am J Bot 104:399–410. https://doi.org/10.3732/ajb.1600276
Tian K, Chen L, Mi X et al (2013) The effect of habitat filtering on tree seedling distribution in a subtropical evergreen broadleaf forest in China. Chinese Sci Bull (Chinese Version) 58:3561–3569. https://doi.org/10.1360/972013-165
Umaña MN, Zhang C, Cao M et al (2017) A core-transient framework for trait-based community ecology: an example from a tropical tree seedling community. Ecol Lett 619–628. https://doi.org/10.1111/ELE.12760
Uriarte M, Muscarella R, Zimmerman JK (2018) Environmental heterogeneity and biotic interactions mediate climate impacts on tropical forest regeneration. Glob Chang Biol 24:e692–e704. https://doi.org/10.1111/gcb.14000
Valladares F, Laanisto L, Niinemets Ü, Zavala MA (2016) Shedding light on shade: ecological perspectives of understorey plant life. Plant Ecol Divers 9:237–251. https://doi.org/10.1080/17550874.2016.1210262
Wang L, Mou PP, Huang J, Wang J (2007) Spatial heterogeneity of soil nitrogen in a subtropical forest in China. Plant Soil 295:137–150. https://doi.org/10.1007/s11104-007-9271-z
Webb CO, Peart DR (2000) Habitat associations of trees and seedlings in a Bornean rain forest. J Ecol 88:464–478. https://doi.org/10.1046/j.1365-2745.2000.00462.x
Wu J, Swenson NG, Brown C, Zhang C, Yang J, Ci X, Li J, Sha L, Cao M, Lin L (2016) How does habitat filtering affect the detection of conspecific and phylogenetic density dependence? Ecology 97:1182–1193. https://doi.org/10.1890/14-2465
Xia S-W, Chen J, Schaefer D, Detto M (2015) Scale-dependent soil macronutrient heterogeneity reveals effects of litterfall in a tropical rainforest. Plant Soil 391:51–61. https://doi.org/10.1007/s11104-015-2402-z
Xia S-W, Chen J, Schaefer D, Goodale UM (2016) Effect of topography and litterfall input on fine-scale patch consistency of soil chemical properties in a tropical rainforest. Plant Soil 404:385–398. https://doi.org/10.1007/s11104-016-2854-9
Yavitt JB, Harms KE, Garcia MN, Wright SJ, He F, Mirabello MJ (2009) Spatial heterogeneity of soil chemical properties in a lowland tropical moist forest, Panama. Aust J Soil Res 47:674–687. https://doi.org/10.1071/SR08258
Zhang Z, Papaik MJ, Wang X, Hao Z, Ye J, Lin F, Yuan Z (2016) The effect of tree size, neighborhood competition and environment on tree growth in an old-growth temperate forest. J Plant Ecol 10:970–980. https://doi.org/10.1093/jpe/rtw126
Zhu H (2006) Forest vegetation of Xishuangbanna, South China. For Stud China 8:1–27. https://doi.org/10.1007/s11632-006-0014-7
Acknowledgements
This study was founded by National Science Foundation of China (31270566), and also supported by the CAS 135 program (No. 2017XTBG-F01). We thank Lang Ma for his assistance in field, and thank María Natalia Umaña for revision suggestions. We also thank Editage Company for language revision and anonymous reviewers for improving earlier versions of the manuscript. We appreciate the Biogeochemistry Laboratory of the Xishuangbanna Tropical Botanical Garden for nutrient analysis, the Xishuangbanna Station for Tropical Rain Forest Ecosystem Studies (XSTRE) for assistance in the field study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Jennifer Powers.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 37 kb)
Rights and permissions
About this article
Cite this article
Xia, SW., Cao, M., Yang, X. et al. Fine scale heterogeneity of soil properties causes seedling spatial niche separation in a tropical rainforest. Plant Soil 438, 435–445 (2019). https://doi.org/10.1007/s11104-019-04027-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04027-8