Abstract
This study aimed to explore the effects and function of microRNA-101a-3p (miR-101a-3p) in epilepsy. Rat model of pilocarpine-induced epilepsy was established and the seizure frequency was recorded. Expression of miR-101a-3p and c-Fos in hippocampus tissues of Rat models were detected by qRT-PCR and western blot. Besides, we established a hippocampal neuronal culture model of acquired epilepsy using Mg2+ free medium to evaluate the effects of miR-101a-3p and c-Fos in vitro. Cells were transfected with miR-101a-3p mimic, si-c-FOS, miR-101a-3p + c-FOS and its corresponding controls. MTT assay was used to detect cell viability upon transfection. Flow cytometry was performed to determine the apoptosis rate. Western blot was performed to measure the protein expression of apoptosis-related proteins (Bcl-2, Bax, and cleaved caspase 3), autophagy-related proteins (LC3 and Beclin1) and c-FOS. The targeting relationship between miR-101a-3p and c-FOS was predicted and verified by TargetScan software and dual-luciferase reporter assay. The role of miR-101a-3p was validated using epilepsy rat models in vivo. Another Rat models of pilocarpine-induced epilepsy with miR-NC or miR-101a-3p injection were established to evaluate the effect of miR-101a-3p overexpression on epilepsy in vivo. MiR-101a-3p was downregulated while c-FOS was increased in hippocampus tissues of Rat model of pilocarpine-induced epilepsy. Overexpression of miR-101a-3p or c-FOS depletion promoted cell viability, inhibited cell apoptosis and autophagy. C-FOS was a target of miR-101a-3p and miR-101a-3p negatively regulated c-FOS expression to function in epilepsy. Overexpression of miR-101a-3p attenuated pilocarpine-induced epilepsy in Rats in vivo. This study indicated that miR-101a-3p could attenuate pilocarpine-induced epilepsy by repressing c-Fos expression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Epilepsy, one of the most common neurological disorders, is a wide-reaching and complex disease affecting more that 70 million people in worldwide [1,2,3]. Also, people with epilepsy are prone to comorbidities and stigma that can negatively impact their quality of life [2]. The common pathologic hallmarks of epilepsy in humans and animal models were hippocampal neuron loss and morphological changes such as the amygdala and hippocampal formation [4]. Currently, 20–30% of patients remain refractory to anti-epileptic drugs (AEDs) treatment. Thus, understanding the process of epilepsy development or epileptogenesis is of great importance to find novel therapeutics for epilepsy.
MicroRNAs (miRNAs) are endogenous, 17∼22 nucleotides, noncoding RNA molecules that function as post-transcriptional regulators of gene expression. MiRNA dysregulation was observed in neurological disorders such as epilepsy [5]. Status epilepticus (SE) can cause damage to hippocampus and result in cognitive deficits and the pathogenesis of epilepsy [6]. Numerous miRNAs were reported to be dysregulated in epileptic brain, which were involved in the pathogenesis and mutagenesis of epilepsy. For example, it has been reported that miR-134 was downregulated in the plasma of patients with epilepsy [7]. MiR-219 was downregulated in epilepsy model and cerebrospinal fluid specimens of epilepsy patients, and silence of miR-219 resulted in seizure behaviors [8]. MiR-101 was a putative amyloid precursor protein (APP) regulator in the rat hippocampus and miR-101a-3p depletion might play a role in the development of Alzheimer disease (AD) [9]. Another research disclosed that miR-101a-3p expression was decreased in Rat model of pilocarpine-induced epilepsy [10]. Besides, a recent transcriptome study suggested that miR-101a-3p was one of the most abundant miRNAs in the amygdala [11], which indicating the pivotal role of miR-101a-3p in neurological disorders. However, the role and underlying mechanism of microRNA-101a-3p (miR-101a-3p) in epilepsy progression was unclear.
C-FOS, an immediate early gene, that the activation of c-fos in the brain has been described initially almost three decades ago [12]. Sufficient research has disclosed the relationship between c-fos and the changes of neuronal activities, including function and structure of neurons in the mammalian nervous system [13]. Expression of c-Fos in individual neurons could be used as a marker of cell activity in neuroendocrine systems [14]. What's more, transient expression of c-fos in the central nervous system was first observed after seizure activity. C-fos protein immunocytochemistry was used as a metabolic marker for tracing neuroanatomical connections, seizure pathways and sites of action of neuroactive drugs [15, 16]. Besides, it has been confirmed that c-FOS could promote epileptogenesis [17]. Thus, targeting c-FOS expression is a potential therapeutic strategy for epilepsy. It is widely known that miRNAs could function by base-pairing to the mRNA 3′-untranslated regions to repress protein synthesis. Wu et al. reported that miR-129 regulated c-FOS to modulate cell proliferation and apoptosis in hippocampal neurons in rats with epilepsy [18]. A recent research suggested that miR-101a-3p negatively regulated FOS expression in neonatal mice cardiomyocytes and myocardial infarction [19]. Thus, we wondered if miR-101a-3p regulate the expression of c-FOS to participate in the regulation of epilepsy.
Therefore, in this study, Rat model of pilocarpine-induced epilepsy and hippocampal neuronal culture model of acquired epilepsy were employed to investigate the function of miR-101a-3p and its relationship with c-FOS in epilepsy.
Materials and Methods
Animal Modeling and Treatment
All animal use procedures were in strict accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and approved by the Animal Care and Use Committee of the First Affiliated Hospital of Zhengzhou University. Sprague–Dawley (SD) rats were purchased from Beijing Huafukang Bioscience co.inc (Beijing, China).
Pilocarpine Rat Model of Temporal Lobe Epilepsy was established as previously described [20, 21]. The rats were used in two experiments: 1. the expression of miR-101a-3p and c-FOS in epilepsy modeling; 2. the effects of miR-101a-3p overexpression on epilepsy modeling. In experiment 1, the modeled rats were divided into control group (N = 8) and epilepsy group (N = 8). The rats in the epilepsy group were intraperitoneally injected with 125 mg/kg lithium chloride (LiCl, Sigma, USA) for 18–20 h, then 30 mg/kg of pilocarpine (Sigma, USA) was given intraperitoneally. The control group was intraperitoneally injected with same volume of saline. Then, 10 mg/kg pilocarpine was injected into the rats every 30 min until the rats developed seizures. The grade of convulsions was evaluated by Racine’s classification [22]. Only animals reaching higher than stage 3 were used for present study. The seizure frequency was also recorded. One day post pilocarpine injection, hippocampal tissues were obtained from rats.
In experiment 2, SD rats were divided into two groups: miR-NC (N = 8) group and miR-101a-3p mimic (N = 8) group. Rats in each group were injected with 1 nmol of miR-NC and miR-101a-3p mimic, respectively, into the dorsal hippocampus as described previously [23]. After 3 days injection, all the rats were treated with pilocarpine to produce an epilepsy model. Then, the seizure frequency was recorded and all rats were sacrificed by decapitation after injected with pilocarpine for one day. Then hippocampus tissues were obtained from rats to detect the related genes expression.
Isolation, Culture and Epileptiform Activity Induction of Hippocampus Neurons
Newborn rats were anesthetized and the brain tissue was exposed. Then hippocampus were dissected using an anatomic microscope (Olympus, Japan). The hippocampus was then digested by 0.25% trypsin at 37 °C for 25 min. DMEM (Thermo Fisher Scientific, Waltham, MA, USA) containing 10% fetal bovine serum (FBS, HyClone, Logan, UT, USA) was added to terminate the digestion. Then, the tissue was filtered and plated onto a poly-D-lysine-treated (0.2 mg/mL, Sigma, St. Louis, MO, USA) cell plates at a concentration of 5 × 105 cells/ml for 24 h and washed with phosphate-buffered saline (PBS). After dried in the air, the neurons were attached. Finally, Cells were cultured in the neurobasal medium (Invitrogen, Carlsbad, CA, USA) with 2% B27 (Neurobasal/B27) supplement (Invitrogen) and 10% FBS (HyClone) at 37 °C with 5% CO2. After 3 days of plating, the neurons were treated with cytosine arabinoside (5 μmol/L, Sigma) to inhibit astrocyte proliferation and medium was refreshed every 3 days. Then, a hippocampal neuronal culture model of acquired epilepsy was established using Mg2+ free media as previously described [20, 24, 25]. In brief, cell were cultured in magnesium-free media containing 145 mM NaCl, 2.5 mM KCl, 10 mM HEPES, 2 mM CaCl2, 10 mM glucose, and 0.002 mM glycine (pH 7.3, adjusted to 325 mOsm with sucrose) for 3 h and epileptiform activity was recorded.
Reagent and Cell Transfection
MiR-101a-3p mimic (miR-101a-3p) and the negative control mimic (miR-NC), miR-101a-3p inhibitor (anti-miR-101a-3p) and the control (anti-miR-NC), small interfering RNA against c-FOS (si-c-FOS) and the control (si-NC), pcDNA3.0 vector (pcDNA), or pcDNA c-FOS (c-FOS) were transfected into hippocampus neurons using Lipofectamine3000 (Thermo Fisher Scientific) according to the instructions.
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR) Assay
Total RNAs in hippocampus tissues of rats in the normal group and the model group and hippocampal neuronal culture model of acquired epilepsy were extracted using Trizol reagent (Invitrogen). For quantification of miR-101a-3p expression, Taqman MicroRNA Assay Kit (Biosystems, Foster City, CA, USA) was used. For c-FOS expression, reverse transcription was performed using Prime Script™ RT reagent kit (Takara, Shiga, Japan). qPCR was performed using the TaqMan® Universal PCR Master Mix II (Biosystems). The primers sequences were listed as follows: miR-101a-3p forward primer, 5′-GCCGCCACCATGGTGAGCAAGG-3′, miR-101a-3p reverse primer, 5′-AATTGAAAAAAGTGATTTAATTT-3′; U6 forward primer, 5′-ATTGGAACGATACAGAGAAGATT-3′, U6 reverse primer, 5′-GGAACGCTTCACGAATTTG-3′; c-FOS forward primer, 5′-GTGCAGCACGGCTTCACCGA-3′, c-FOS reverse primer, 5′-TTGAGCTGCGCCGTTGGAGG-3; β-actin forward primer, 5′- CGAGCAGGAGATGGGAACC-3′, β-actin reverse primer 5′-CAACGGAAACGCTCATTGC-3′. MiR-101a-3p and c-FOS relative expression were calculated with 2−∆∆Ct method. U6 and β-actin were served as the internal controls, respectively.
Cell Viability Assay
The cells were seeded into 96-well plates with 100 μL in each well and incubated in 5% CO2 at 37 °C. After incubation for 48 h, the culture plates were taken out and each well was added with 10 μL of MTT formazan solution (Sigma-Aldrich, St. Louis, MO, USA) (5 mg/mL) and incubated for another 4 h. Optical density (OD) value was determined using an automatic Microplate Reader (BIO-RAD, Cal, New York, USA).
Cell Apoptosis Assay
The Annexin V-FITC/propidium iodide (PI) cell apoptosis detection kit (Sigma) was performed to analyze hippocampus neurons apoptosis. 48 h after the transfection, cells were collected and digest with 0.25% trypsin. After being washed twice with PBS, they were centrifuged and resuspended in 200 μL of binding buffer. Annexin V-FITC (5 μL) and 5 μL of propidium iodide (PI) were added and mixed gently with the cells, and incubated in the dark room for 15 min. Binding buffer (250 μL) was added, and then the wavelength of 490 nm was activated using a flow cytometer FACS Calibur (BD Biosciences, San Jose, CA, USA) to determine the apoptotic rate.
Western Blot Assay
The total proteins of rat hippocampus tissues and hippocampus neurons were extracted, and then lysed with RIPA (Millipore, Billerica, MA, USA) supplemented with the mixture of protease inhibitors 0.1 mmol/L PMSF (Beyotime, shanghai, China). Protein was quantified using the Enhanced BCA Protein Assay Kit (Beyotime). Equal amounts of proteins (45 µg) were loaded and separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis gels (10%–15% SDS-PAGE, Beyotime). Then the blots were electrophoretically transferred to polyvinylidene fluoride (PVDF) membranes (Millipore). The membranes were blocked with skim milk (5%) in phosphate buffered saline with PBST containing 0.05% Tween 20 for 1 h and incubated with corresponding primary antibodies overnight at 4 °C, followed by incubating with HRP-conjugated secondary antibody for 1 h at room temperature. The protein bands were visualized using an enhanced chemiluminescence (ECL) detection kit (Beyotime). The following primary antibodies were used: anti-c-FOS, anti-LC3, anti-Beclin1, anti-Bcl-2, anti-Bax, anti-cleaved caspase 3 and internal control β-actin were purchased from Cell Signaling Technology. Relative protein level was quantified using the Quantity One software (Bio-Rad, USA) by comparison the gray values with β-actin.
Luciferase Reporter Assay
The target gene of miR-101a-3p was predicted using Targetscan online database, and the hypothesis was confirmed using Dual-luciferase reporter assay. Firstly, luciferase reporter recombinant plasmids inserted with c-FOS 3′-untranslated region (3′UTR) wild type (WT) and mutant type (MUT) were constructed, named as c-FOS 3′UTR-WT and c-FOS 3′UTR-MUT. Cells were inoculated into a 6-well plate for 24 h prior to transfection. Then luciferase reporter plasmids and miR-101a-3p mimic or inhibitor, and their negative controls were co-transfected into hippocampus neurons, respectively. 48 h upon transfection, cells were harvested and lysed and the relative luciferase activity was assessed by using a luciferase reporter assay system (Promega, Mannheim, Germany) in accordance with the manufacturer’s instructions. Renilla luciferase activity was used for normalization of firefly luciferase.
Statistical Analysis
SPSS 20.0 statistical software was used for statistical analysis. Data were expressed as mean ± standard deviation (SD). The comparisons between two groups were tested by Student’s t-test, and comparisons among multiple groups were analyzed using one-way analysis of variance (ANOVA) by Tukey's HSD post hoc multiple comparison test. P < 0.05 was considered statistically significant.
Results
The Expression of miR-101a-3p and c-FOS in Rat Model of Pilocarpine-Induced Epilepsy and Hippocampal Neuronal Culture Model of Acquired Epilepsy
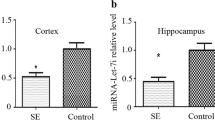
To explore the roles of miR-101a-3p and c-FOS in epilepsy, the rats were divided into two groups: epilepsy (n = 8) group and normal (n = 8) group. We found that the average seizure frequency in Rat model of pilocarpine-induced epilepsy group was markedly higher than normal group (Fig. 1a), suggesting that Rat model of pilocarpine-induced epilepsy was established. Then, the expression of miR-101a-3p and c-FOS in Rat model of pilocarpine-induced epilepsy were detected. The results showed that miR-101a-3p was downregulated in hippocampus tissues of Rat model of pilocarpine-induced epilepsy group (Fig. 1b), while c-FOS protein level was increased (Fig. 1c). Besides, hippocampal neuronal culture model of acquired epilepsy was established and the levels of miR-101a-3p and c-FOS were detected. Compared with control group, the expression of miR-101a-3p was decreased (Fig. 1d), while c-FOS was elevated in hippocampal neuronal culture model of acquired epilepsy (Fig. 1e).
The expression of miR-101a-3p and c-FOS in Rat model of pilocarpine-induced epilepsy and hippocampal neuronal culture model of acquired epilepsy. a Rat model of pilocarpine-induced epilepsy were established and the average seizure frequency was recorded. *P < 0.05, compared with the control, n = 8 in each group. b qRT-PCR was performed to measure the expression of miR-101a-3p in hippocampus tissues of Rats after pilocarpine-induced epilepsy. *P < 0.05, compared with the control, n = 8 in each group. c Western blot was performed to detect the protein level of c-FOS in hippocampus tissues of Rats after pilocarpine-induced epilepsy. *P < 0.05, compared with the control, n = 8 in each group. d The expression of miR-101a-3p in hippocampal neuronal culture model of acquired epilepsy was detected by qRT-PCR. *P < 0.05, compared with the control, n = 3 in each group. e Western blot was performed to measure the protein expression of c-FOS in hippocampal neuronal culture model of acquired epilepsy. *P < 0.05, compared with the control, n = 3 in each group
Overexpression of miR-101a-3p Enhanced Cell Viability, and Inhibited Apoptosis and Autophagy of Hippocampal Neuronal Culture Model of Acquired Epilepsy
To investigate the effect of miR-101a-3p on epilepsy, hippocampal neuronal culture model of acquired epilepsy were transfected with miR-NC or miR-101a-3p. Our data showed that miR-101a-3p was downregulated in epilepsy group, which was reversed by miR-101a-3p overexpression (Fig. 2a). As shown in Fig. 2b, cell viability was suppressed in epilepsy group, while miR-101a-3p overexpression reversed this effect. Moreover, cell apoptosis was promoted in epilepsy group, which was blocked by miR-101a-3p upregulation (Fig. 2c, d). Then, we detected apoptosis-related proteins (Bax, Bcl-2 and Cas-3 cleavage) and autophagy-related proteins (LC3 and Beclin1). The results suggested that the expression of Bax, Cas-3 cleavage, Beclin1 and the ratio of LC3II/LC3I of hippocampal neurons was elevated in epilepsy group, while Bcl-2 was repressed. These effects were reversed by miR-101a-3p overexpression (Fig. 2e–k).
Overexpression of miR-101a-3p affected cell viability, apoptosis and autophagy of hippocampal neuronal culture model of acquired epilepsy. a–k Hippocampal neuronal culture model of acquired epilepsy were transfected with miR-NC or miR-101a-3p for 48 h. a The expression of miR-101a-3p was detected by qRT-PCR. b Cell viability was measured by MTT assay. c, d Cell apoptosis was determined by flow cytometry. e–h Western blot was performed to measure the expression of apoptosis-related proteins Bcl-2, Bax, and Cas-3 cleavage. i–k Western blot was performed to measure the levels of autophagy-related protein Beclin1 and LC3. *P < 0.05, compared with the control, n = 3 in each group
c-FOS was a Target of miR-101a-3p
Considering that miR-101a-3p was downregulated, while c-FOS was upregulated in vivo, we further investigated the relationship between miR-101a-3p and c-FOS. We identified c-FOS as a potential target gene of miR-101a-3p using TargetScan online database (Fig. 3a). The data showed that the expression of miR-101a-3p was facilitated by miR-101a-3p mimic, but suppressed by miR-101a-3p inhibitor (Fig. 3b, c). To further confirm the relationship between miR-101a-3p and c-FOS, we analyzed the luciferase activity in hippocampus neurons co-transfected with c-FOS 3′UTR-WT or c-FOS 3′UTR-MUT luciferase reporter and miR-101a-3p mimic, inhibitor, or their negative controls. Our results showed that relative luciferase activity was decreased in miR-101a-3p group, which had no significant change in miR-NC group (Fig. 3d). The relative luciferase activity was increased in anti- miR-101a-3p group, which showed no remarkable change in anti-miR-NC group (Fig. 3e). Moreover, miR-101a-3p decreased c-FOS expression, while miR-101a-3p inhibitor increased c-FOS expression (Fig. 3f).
c-FOS was a target of miR-101a-3p. a The binding sites between miR-101a-3p and c-FOS was predicted by Targetscan online database and the luciferase reporter plasmids containing the wild-type (WT) or mutated (MUT) c-FOS binding sites of miR-101a-3p were established. b and c The expression of miR-101a-3p was detected by qRT-PCR. d and e the luciferase activity was measured in hippocampal neuronal culture model of acquired epilepsy co-transfected with c-FOS 3′UTR-WT or c-FOS 3′UTR-MUT luciferase reporter and miR-101a-3p mimic, inhibitor, or their negative controls. f Expression level of c-FOS in cells transfected with miR-NC, miR-101a-3p, anti-miR-NC, or anti-miR-101a-3p. *P < 0.05, compared with the miR-NC or anti-miR-NC, n = 3 in each group
Depletion of c-FOS Enhanced Cell Viability, but Inhibited Apoptosis and Autophagy of Hippocampal Neuronal Culture Model of Acquired Epilepsy
To determine the effect of c-FOS on hippocampal neuronal culture model of acquired epilepsy, si-NC or si–c-FOS was employed to alter the level of c-FOS prior to detection of cell viability, apoptosis and autophagy. As shown in Fig. 4a, c-FOS protein level was upregulated in epilepsy group, and was decreased by si–c-FOS. Knockdown of c-FOS partly reversed the suppression effect on cell viability (Fig. 4b) and the apoptosis rate (Fig. 4c, d) in epilepsy group. Besides, the decreasing protein level of Bcl-2 and the increasing protein levels of Bax, cas-3 Cleavage, LC3-II and Beclin1 in epilepsy group were reversed by c-FOS depletion (Fig. 4e, k). These results indicating that c-FOS promoted epilepsy seizure in hippocampal neuronal culture model of acquired epilepsy.
Downregulation of c-FOS regulated the cell viability, apoptosis and autophagy in hippocampal neuronal culture model of acquired epilepsy. a–k Hippocampal neuronal culture model of acquired epilepsy were transfected with si-NC or si-c-FOS. a The protein level of c-FOS in the transfected cells was detected by western blot. b Cell viability of transfected cells was detected by MTT assay. c and d Cell apoptosis of transfected cells was determined by flow cytometry. e–h Western blot was performed to measure the expression of apoptosis-related proteins Bcl-2, Bax, and Cas-3 cleavage in cells upon transfection. i–k Western blot was performed to measure the levels of autophagy-related protein Beclin1 and LC3 in cells upon transfection. *P < 0.05, compared with the control, n = 3 in each group
miR-101a-3p Promoted Cell Viability, and Suppressed Apoptosis and Autophagy in Hippocampal Neuronal Culture Model of Acquired Epilepsy by Targeting c-FOS
To further investigate whether miR-101a-3p regulates autophagy by targeting c-FOS, cells were transfected with negative control mimic, miR-101a-3p mimic, miR-101a-3p mimic + pcDNA 3.0 vector, or miR-101a-3p mimic + pcDNA c-FOS. Western blot assay was performed to detect the protein level of c-FOS and the results demonstrated that the expression of c-FOS was decreased in hippocampal neuronal culture model of acquired epilepsy transfected with miR-101a-3p mimic, which was rescued by pcDNA c-FOS transfection (Fig. 5a). Our data indicated that cell viability was promoted by miR-101a-3p overexpression, which was abolished by c-FOS overexpression (Fig. 5b). The results showed that upregulation of miR-101a-3p decreased cell apoptotic rate, which was attenuated by c-FOS transfection (Fig. 5c and d). To further clarify the molecular mechanism of apoptosis, western blot was performed to evaluate the expression of apoptosis-related protein Bcl-2, Bax, and Cas-3 cleavage. Consistent the results of flow cytometry, upregulation of miR-101a-3p decreased the expression of Cas-3 cleavage and Bax and increased Bcl-2 expression, interestingly, which were also attenuated by c-FOS transfection in hippocampal neuronal culture model of acquired epilepsy (Fig. 5e–h). Then, the expression of autophagy-related protein Beclin1 and LC3 were also evaluated by western blot. Our results demonstrated that miR-101a-3p decreased the expression of Beclin1 and the ratio of LC3II/LC3I, which were attenuated by c-FOS transfection in hippocampal neuronal culture model of acquired epilepsy (Fig. 5i–k).
miR-101a-3p enhanced cell viability, and suppressed apoptosis and autophagy in hippocampal neuronal culture model of acquired epilepsy by targeting c-FOS. Hippocampal neuronal culture model of acquired epilepsy were established and transfected with miR-101a-3p, miR-NC, miR-101a-3p + pcDNA-NC, or miR-101a-3p + c-FOS. a Western blot was performed to measure the expression of c-FOS. b Cell viability was measured by MTT assay. c, d Cell apoptosis was determined by flow cytometry. e–h Western blot was employed to measure the expression of apoptosis-related proteins Bcl-2, Bax, and Cas-3 cleavage. i–k Western blot was performed to measure the expression of autophagy-related proteins Beclin1 and LC3. *P < 0.05, compared with the Control, n = 3 in each group
Overexpression of miR-101a-3p Attenuated Pilocarpine-Induced Epilepsy in Rats
We then further confirmed the role of miR-101a-3p in vivo. The Rat model of pilocarpine-induced epilepsys were divided into two groups: miR-NC group and miR-101a-3p group. We injected the negative control mimic (n = 8) or miR-101a-3p mimic (n = 8) into Rat model of pilocarpine-induced epilepsys. Consistent with in vitro, the average seizure frequency was decreased in Rat model of pilocarpine-induced epilepsys with miR-101a-3p mimic injection compared with miR-NC injection group (Fig. 6a). In addition, we also examined the expression of c-FOS using qRT-PCR and western bolt assays. The results demonstrated that c-FOS expression was also decreased in Rat model of pilocarpine-induced epilepsy with miR-101a-3p mimic injection (Fig. 6b, c).
Effects of miR-101a-3p overexpression on Rat model of pilocarpine-induced epilepsy. Rats were intrahippocampal injected with 1 nM miR-NC or miR-101a-3p mimic for 3 days, prior to inject with pilocarpine. a After the injection of pilocarpine, the average seizure frequency was recorded. *P < 0.05, compared with the miR-NC, n = 8 in each group. b and c 24 h upon injection, the hippocampus tissues were obtained. And the mRNA and protein level of c-FOS in hippocampus tissues of rat was analyzed by western blot. *P < 0.05, compared with the miR-NC, n = 8 in each group
Discussion
Nerve cell injury plays an important role in the development of epilepsy and is partly achieved through apoptosis and autophagy pathway. The changing levels of miRNAs have been identified to shape the development and function of the nervous system [26], which may play irreplaceable roles in the pathogenesis of neurological disorders including epilepsy [27]. In this research, we established Rat model of pilocarpine-induced epilepsy and hippocampal neuronal culture model of acquired epilepsy to investigate the role of miR-101a-3p and c-FOS on epilepsy in vitro and in vivo. In hippocampus tissues of Rat models that we made, miR-101a-3p expression was decreased, while c-FOS protein level was elevated. A similar situation was found in hippocampal neuronal culture model of acquired epilepsy. The abnormal expression of miR-101a-3p and c-FOS in cell model and Rat model of pilocarpine-induced epilepsy indicating the correlation between miR-101a-3p, c-FOS and epilepsy.
Sufficient studies have shown that miRNAs reduces the expression level of target genes by base-pairing to the mRNA 3′-untranslated regions. As it has been disclosed that miR-101a-3p negatively regulated FOS expression in neonatal mice cardiomyocytes and myocardial infarction [19]. In consistent with this research, our research disclosed that miR-101a-3p targets to c-FOS and negatively regulated its expression. In hippocampal neuronal culture model of acquired epilepsy, overexpression of miR-101a-3p or knockdown of c-FOS both enhanced cell viability, suppressed apoptosis and autophagy of cells.
Multiple studies have shown that different stimuli such as seizure activity can induce c-FOS expression in hippocampal structures [12, 28]. In an earlier study, it has been confirmed that injection of pentylenetetrazole (PTZ, which induce epileptogenesis in mice) to mice induced c-Fos expression in several brain regions including the cerebral cortex and hippocampus [29]. Research also disclosed that c-FOS was highly expressed in neuronal populations during the development of the SE [30]. Therefore, the induction of c-FOS expression might play an important role in the development of seizures. Interestingly, our research also shown that miR-101a-3p exert its antagonism on epilepsy by suppressing c-FOS expression.
Taken together, these findings indicated that upregulation of miR-101a-3p suppressing c-FOS expression to protect hippocampal neurons against autophagy and apoptosis in hippocampal neuronal culture model of acquired epilepsy. Importantly, we disclosed that miR-101a-3p played neuroprotective role in the hippocampal neuronal culture model of acquired epilepsy and Rat model of pilocarpine-induced epilepsy, which providing a novel therapeutic approach for epilepsy.
References
Singh A, Trevick S (2016) The epidemiology of global epilepsy. Neurol Clin 34:837–847
Trinka E, Kwan P, Lee B, Dash A (2018) Epilepsy in Asia: disease burden, management barriers, and challenges. Epilepsia 60:7
Fauser S, Tumani H (2017) Epilepsy. Handb Clin Neurol 146:259–266
Cendes F, Sakamoto AC, Spreafico R, Bingaman W, Becker AJ (2014) Epilepsies associated with hippocampal sclerosis. Acta Neuropathol 128:21–37
Karnati HK, Panigrahi MK, Gutti RK, Greig NH, Tamargo IA (2015) miRNAs: key players in neurodegenerative disorders and epilepsy. J Alzheimer’s Dis 48:563–580
Kortvelyessy P, Huchtemann T, Heinze HJ, Bittner DM (2017) Progranulin and its related micrornas after status epilepticus: possible mechanisms of neuroprotection. Int J Mol Sci 18:490
Avansini SH, de Sousa Lima BP, Secolin R, Santos ML, Coan AC, Vieira AS, Torres FR, Carvalho BS, Alvim MK, Morita ME, Yasuda CL, Pimentel-Silva LR, Dogini DB, Rogerio F, Cendes F, Lopes-Cendes I (2017) MicroRNA hsa-miR-134 is a circulating biomarker for mesial temporal lobe epilepsy. PLoS ONE 12:e0173060
Zheng H, Tang R, Yao Y, Ji Z, Cao Y, Liu Z, Peng F, Wang W, Can D, Xing H, Bu G, Xu H, Zhang YW, Zheng W (2016) MiR-219 protects against seizure in the kainic acid model of epilepsy. Mol Neurobiol 53:1–7
Vilardo E, Barbato C, Ciotti M, Cogoni C, Ruberti F (2010) MicroRNA-101 regulates amyloid precursor protein expression in hippocampal neurons. J Biol Chem 285:18344–18351
Wu Q, Yi X (2018) Down-regulation of long noncoding RNA MALAT1 protects hippocampal neurons against excessive autophagy and apoptosis via the PI3K/Akt signaling pathway in rats with epilepsy. J Mol Neurosci 65:234–245
Cohen JL, Ata AE, Jackson NL, Rahn EJ, Ramaker RC, Cooper S, Kerman IA, Clinton SM (2017) Differential stress induced c-Fos expression and identification of region-specific miRNA-mRNA networks in the dorsal raphe and amygdala of high-responder/low-responder rats. Behav Brain Res 319:110–123
Herrera DG, Robertson HA (1996) Activation of c-fos in the brain. Prog Neurobiol 50:83–107
Watanabe Y, Johnson RS, Butler LS, Binder DK, Spiegelman BM, Papaioannou VE, McNamara JO (1996) Null mutation of c-fos impairs structural and functional plasticities in the kindling model of epilepsy. J Neurosci 16:3827–3836
Hoffman GE, Smith MS, Verbalis JG (1993) c-Fos and related immediate early gene products as markers of activity in neuroendocrine systems. Front Neuroendocrinol 14:173–213
Dragunow M, Faull R (1989) The use of c-fos as a metabolic marker in neuronal pathway tracing. J Neurosci Methods 29:261–265
André V, Pineau N, Motte JE, Marescaux C, Nehlig A (1998) Mapping of neuronal networks underlying generalized seizures induced by increasing doses of pentylenetetrazol in the immature and adult rat: a c-Fos immunohistochemical study. Eur J Neurosci 10:2094–2106
Malhi SM, Jawed H, Hanif F, Ashraf N, Zubair F, Siddiqui BS, Begum S, Kabir N, Simjee SU (2014) Modulation of c-Fos and BDNF protein expression in pentylenetetrazole-kindled mice following the treatment with novel antiepileptic compound HHL-6. Biomed Res Int 2014:876712
Wu DM, Zhang YT, Lu J, Zheng YL (2018) Effects of microRNA-129 and its target gene c-Fos on proliferation and apoptosis of hippocampal neurons in rats with epilepsy via the MAPK signaling pathway. J Cell Physiol 233:6632–6643
Lin B, Xu J, Wang F, Wang J, Zhao H, Feng D (2020) LncRNA XIST promotes myocardial infarction by regulating FOS through targeting miR-101a-3p. Aging 12:7232–7247
Geng JF, Liu X, Zhao HB, Fan WF, Geng JJ, Liu XZ (2018) LncRNA UCA1 inhibits epilepsy and seizure-induced brain injury by regulating miR-495/Nrf2-ARE signal pathway. Int J Biochem Cell Biol 99:133–139
Glien M, Brandt C, Potschka H, Voigt H, Ebert U, Loscher W (2001) Repeated low-dose treatment of rats with pilocarpine: low mortality but high proportion of rats developing epilepsy. Epilepsy Res 46:111–119
Racine RJ (1972) Modification of seizure activity by electrical stimulation. II Motor seizure. Electroencephalogr Clin Neurophysiol 32:281–294
Wang W, Wang X, Chen L, Zhang Y, Xu Z, Liu J, Jiang G, Li J, Zhang X, Wang K, Wang J, Chen G, Luo J (2016) The microRNA miR-124 suppresses seizure activity and regulates CREB1 activity. Expert Rev Mol Med 18:e4
Xiao Z, Peng J, Wu L, Arafat A, Yin F (2017) The effect of IL-1beta on synaptophysin expression and electrophysiology of hippocampal neurons through the PI3K/Akt/mTOR signaling pathway in a rat model of mesial temporal lobe epilepsy. Neurol Res 39:640–648
Xu Z, Zhang J, Lei X, Xu Z, Peng Y, Yao B, Xu P (2013) Effects of valproate sodium on extracellular signal-regulated kinase 1/2 phosphorylation following hippocampal neuronal epileptiform discharge in rats. Exp Ther Med 6:1397–1401
Nowak JS, Michlewski G (2013) miRNAs in development and pathogenesis of the nervous system. Biochem Soc Trans 41:815–820
Li MM, Li XM, Zheng XP, Yu JT, Tan L (2014) MicroRNAs dysregulation in epilepsy. Brain Res 1584:94–104
Schreiber SS, Tocco G, Najm I, Finch CE, Johnson SA, Baudry M (1992) Absence of c-fos induction in neonatal rat brain after seizures. Neurosci Lett 136:31–35
D’Intino G, Vaccari F, Sivilia S, Scagliarini A, Gandini G, Giardino L, Calzà L (2006) A molecular study of hippocampus in dogs with convulsion during canine distemper virus encephalitis. Brain Res 1098:186–195
Sinel'nikova VV, Shubina LV, Gol'tiaev MV, Loseva EV, Kichigina VF (2012) [Detection of c-fos expression in animal brain in a pilocarpine model of temporal lobe epilepsy]. Zhurnal vysshei nervnoi deiatelnosti imeni I P Pavlova 62:497–505
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Geng, J., Zhao, H., Liu, X. et al. MiR-101a-3p Attenuated Pilocarpine-Induced Epilepsy by Downregulating c-FOS. Neurochem Res 46, 1119–1128 (2021). https://doi.org/10.1007/s11064-021-03245-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-021-03245-w