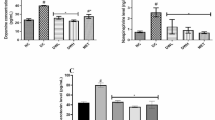
On samples of the mitochondria and synaptic membranes isolated from rat brains using differential centrifugation, we tried to evaluate the neuroprotective efficacy of a combination of mitochondriaspecific antioxidants, acetyl-L-carnitine (ALC) and α-lipoic acid (LA), with nicotinamide (NAm), against diabetes-induced disorders in the CNS. Three groups of adult male Wistar rats were examined; these were control intact rats (group C), animals with experimental streptozotocin (STZ)-induced diabetes (group D; 6 weeks after STZ injections), and diabetic rats treated during the two final weeks of the above period by a combination of ALC, LA, and NAm (separate daily injections; doses 100, 50, and 100 mg/kg body mass, respectively; group D+T). At the day of preparation of the organelle samples, the mean blood glucose levels in groups C, D, and D+T were 4.8, 20.3, and 15.4 mM, respectively. The intensity of reactive oxygen species (ROS) production in the brain mitochondria from rats of group D measured by fluorescent analyses using 2’,7’-dichlorofluorescein diacetate was, on average, 37.2% greater than that in group C. Co-treatment provided a significant decrease in the above index in group D+T (27.8% in comparison with group D). Diabetes led to dramatic intensification of the CYP2E1 protein level in the liver of group D animals (242% vs. group C). In group D+T, this index was 33.1% lower than that in group D.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
S. T. Jerram and R. D. Leslie, “The genetic architecture of type 1 diabetes,” Genes, 8, No. 8, 209 (2017), doi:https://doi.org/10.3390/genes8080209.
M. Rewers, H. Hyöty, A. Lernmark, et al., “The environmental determinants of diabetes in the young (TEDDY) study: 2018 update,” Curr. Diab. Rep., 18, No. 12, 136 (2018).
K. A. Adeshara, A. G. Diwan, and R. S. Tupe, “Diabetes and complications: Cellular signaling pathways, current understanding and targeted therapies,” Curr. Drug Targets, 17, No. 11, 1309–1328 (2016).
V.R. Drel, P. Pacher, R. Stavniichuk, et al., “Poly(ADPribose) polymerase inhibition counteracts renal hypertrophy and multiple manifestations of peripheral neuropathy in diabetic Akita mice,” Int. J. Mol. Med., 28, No. 4, 629–635 (2011).
W. Li, E. Huang, and S. Gao, “Type 1 diabetes mellitus and cognitive impairments: A systematic review,” J. Alzheimers Dis., 57, No. 1, 29–36 (2017).
S. A. Hamed, “Brain injury with diabetes mellitus: evidence, mechanisms and treatment implications,” Expert Rev. Clin. Pharmacol., 10, No. 4, 409–428 (2017).
I. Trikash, V. Gumenyuk, and T. Kuchmerovska, “Diabetes-induced impairments of the exocytosis process and effect of gabapentin: the link with cholesterol level in neuronal plasma membranes,” Neurochem. Res., 40, No. 4, 723–732 (2015).
E. Blázquez, E. Velázquez, V. Hurtado-Carneiro, and J.M. Ruiz-Albusac, “Insulin in the brain: its pathophysiological implications for states related with central insulin resistance, type 2 diabetes and Alzheimer’s disease,” Front. Endocrinol., 5, 161 (2014), doi:https://doi.org/10.3389/fendo.2014.00161.
J. M. Forbes and M. E. Cooper, “Mechanisms of diabetic complications,” Physiol. Rev., 93, No. 1, 137–188 (2013).
S. Dewanjee, S. Das, A.K. Das, et al., “Molecular mechanism of diabetic neuropathy and its pharmacotherapeutic targets,” Eur. J. Pharmacol., 833, 472–523 (2018).
A. Verkhratsky, M. Trebak, F. Perocchi, et al., “Crosslink between calcium and sodium signaling,” Exp. Physiol., 103, No. 2, 157–169 (2018).
M. W. Jann and J. H. Slade, “Antidepressant agents for the treatment of chronic pain and depression,” Pharmacotherapy, 27, 1571–1587 (2007).
P. Kumar, G. N. Rao, B. B. Pal, and A. Pal, “Hyperglycemia-induced oxidative stress induces apoptosis by inhibiting PI3-kinase/Akt and ERK1/2 MAPK mediated signaling pathway causing downregulation of 8-oxoGDNA glycosylase levels in glial cells,” Int. J. Biochem. Cell. Biol., 53, 302–319 (2014).
F. J. Gonzalez, “Role of cytochromes P450 in chemical toxicity and oxidative stress: studies with CYP2E1,” Mutat. Res., 569, Nos. 1/2, 101–110 (2005).
S. V. Bhagwat, M. R. Boyd, and V. Ravindranath, “Multiple forms of cytochrome P450 and associated monooxygenase activities in human brain mitochondria,” Biochem. Pharmacol., 59, No. 5, 573–582 (2000).
F. Seifar, M. Khalili, H. Khaledyan, et al., “α-Lipoic acid, functional fatty acid, as a novel therapeutic alternative for central nervous system diseases: A review,” Nutr. Neurosci., 22, No. 5, 306–316 (2019), doi:https://doi.org/10.1080/1028415X.2017.1386755. Epub 2017 Nov 29.
G. Sergi, S. Pizzato, F. Piovesan, et al., “Effects of acetyl-L-carnitine in diabetic neuropathy and other geriatric disorders,” Aging. Clin. Exp. Res., 30, No. 2, 133–138 (2018).
B. Picconi, I. Barone, A. Pisani, et al., “Acetyl-L-carnitine protects striatal neurons against in vitro ischemia: the role of endogenous acetylcholine,” Neuropharmacology, 50, No. 8, 917–923 (2006).
N. Braidy, R. Grant, and P. S. Sachdev, “Nicotinamide adenine dinucleotide and its related precursors for the treatment of Alzheimer’s disease,” Curr. Opin. Psychiat., 31, No. 2, 160–166 (2018).
S. Nesci, F. Trombetti, V. Ventrella, et al., “The inhibition of the mitochondrial F1FO-ATPase activity when activated by Ca2+ opens new regulatory roles for NAD,” Biol. Chem., 399, No. 2, 197–202 (2018).
T. Kuchmerovska, I. Shymanskyy, S. Chlopicki, and A. Klimenko, “1-Methylnicotinamide (MNA) in prevention of diabetes-associated brain disorders,” Neurochem. Int., 56, 221–228 (2010).
I. P. Abita, R. Chicheportiche, and M. Schweitz, “Effect of neurotoxins (veratridine, sea anemone toxin, tetrodotoxin) on transmitter accumulation. Release by nerve terminals in vitro,” Biochemistry, 16, 1838–1864 (1977).
C. P. LeBel, H. Ischiropoulos, and S. C. Bondy, “Evaluation of the probe 2’,7’-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress,” Chem. Res. Toxicol., 5, 227–231 (1992).
A. Baracca, G. Sgarbi, G. Solaini, and G. Lenaz, “Rhodamine 123 as a probe of mitochondrial membrane potential: evaluation of proton flux through F0 during ATP synthesis,” Biochim. Biophys. Acta., 1606, Nos. 1/3, 137-146 (2003).
W. B. Rathbun and M. V. Betlach, “Estimation of enzymatically produced orthophosphate in the presence of cysteine and adenosine triphosphate,” Anal. Biochem., 28, 436–445 (1969).
H. U. Bergmeyer, Methods of Enzymatic Analysis, Academic Press, New York, London, Vol. 4 (1974).
O. W. Lowry, N. J. Rosebrough, R. L. Farr, and R. J. Randall, “Protein measurement with the Folin reagent,” J. Biol. Chem., 193, 265–275 (1951).
M. M. Bradford, “A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding,” Anal. Biochem., 72, 248–254 (1976).
T. Kuchmerovska, I. Shymanskyy, G. Donchenko, et al., “Poly(ADP-ribosyl)ation enhancement in brain cell nuclei is associated with diabetic neuropathy,” J. Diabetes Complications, 18, No. 4, 198–204 (2004).
M. R. Lakshman, M. Garige, M. A. Gong, et al., “CYP2E1, oxidative stress, post-translational modifications and lipid metabolism,” Subcell. Biochem., 67, 199–233 (2013).
A. S. Pivovarov, F. Calahorro, and R. J. Walker, “Na+/K+-pump and neurotransmitter membrane receptors,” Invert. Neurosci., 19, No. 1, 1 (2019), doi: https://doi.org/10.1007/s10158-018-0221-7
T. Feldmann, M. Shahar, A. Baba, et al., “The Na(+)/Ca(2+)-exchanger: an essential component in the mechanism governing cardiac steroid-induced slow Ca(2+) oscillations,” Cell Calcium, 50, No. 5, 424–432 (2011).
M. M. Guzyk, A. A. Tykhomyrov, V. S. Nedzvetsky, et al., “Poly (ADP-ribose) polymerase-1 (PARP-1) inhibitors reduce reactive gliosis and improve angiostatin levels in retina of diabetic rats,” Neurochem. Res., 41, No. 10, 2526–2537 (2016).
M. M. Guzyk, K. O. Dyakun, L.V. Yanytska, et al., “Inhibitors of poly(ADP-ribose) polymerase-1 as agents providing correction of brain dysfunctions induced by experimental diabetes,” Neurophysiology,49, No. 3, 183–193 (2017).
R. C. Scaduto Jr. and L. W. Grotyohann, “Measurement of mitochondrial membrane potential using fluores-cent rhodamine derivatives,” Biophys. J., 76, No. 1, 469–477 (1999).
M. M. Guzyk, K. O. Dyakun, L. V. Yanitska, and Т. М. Kuchmerovska, “Influence of poly(ADP-ribose) polymerase inhibitors on some parameters of oxidative stress in blood leukocytes of rats with experimental diabetes,” Ukr. Biochem. J., 85, No. 1, 62–70 (2013).
R. Castañeda-Arriaga and J. R. Alvarez-Idaboy, “Lipoic acid and dihydrolipoic acid. A comprehensive theoretical study of their antioxidant activity supported by available experimental kinetic data,” J. Chem. Inform. Model., 54, No. 6, 1642–1652 (2014).
I. C. Fernandez, M. Del Carmen Camberos, G. A. Passicot, et al., “Children at risk of diabetes type 1. Treatment with acetyl-L-carnitine plus nicotinamide – case reports,” J. Pediatr. Endocrinol. Metab., 26, Nos. 3/4, 347–355 (2013).
M. J. Clausen and H. Poulsen, “Sodium/potassium homeostasis in the cell,” Met. Ions Life Sci., 12, 41–67 (2013).
G. E. Torres and S. G. Amara, “Glutamate and monoamine transporters: new visions of form and function,” Curr. Opin. Neurobiol., 17, No. 3, 304–312 (2007).
H. P. Hammes, X. Du, D. Edelstein, et al., “Benfotiamine blocks three major pathways of hyperglycemic damage and prevents experimental diabetic retinopathy,” Nat. Med., 9, No. 3, 294–299 (2003).
T. Kuchmerovska, I. Shymanskyy, L. Bondarenko, and A. Klimenko, “Effects of nicotinamide supplementation on liver and serum contents of amino acids in diabetic rats,” Eur. J. Med. Res., 13, No. 6, 275–280 (2008).
C. C. Shen, H. M. Huang, H. C. Ou, et al., “Protective effect of nicotinamide on neuronal cells under oxygen and glucose deprivation and hypoxia/reoxygenation,” J. Biomed. Sci., 11, No. 4, 472–481 (2004).
J. Luo, A. Y. Nikolaev, S. Imai, et al., “Negative control of p53 by Sir2alpha promotes cell survival under stress,” Cell, 107, No. 2, 137–148 (2001).
M. M. Guzyk, T. M. Tykhonenko, K. O. Dyakun, et al., “Altered sirtuins 1 and 2 expression in the brain of rats induced by experimental diabetes and the ways of its correction,” Ukr. Biochem. J., 91, No. 1, 21–29 (2019).
J. S. Ungerstedt, M. Blomback, T. Soderstrom, “Nicotinamide is a potent inhibitor of proinflammatory cytokines,” Clin. Exp. Immunol., 131, No. 1, 48–52 (2003).
E. Turunc Bayrakdar, Y. Uyanikgil, L. Kanit, et al., “Nicotinamide treatment reduces the levels of oxidative stress, apoptosis, and PARP-1 activity in Aβ(1-42)-induced rat model of Alzheimer’s disease,” Free Radic. Res., 48, No. 2, 146–158 (2014).
G. Traina, “The neurobiology of acetyl-L-carnitine,” Front. Biosci. (Landmark Ed.), 21, 1314–1329 (2016).
D. Ziegler, P. A. Low, and W. J. Litchy, “Efficacy and safety of antioxidant treatment with α-lipoic acid over 4 years in diabetic polyneuropathy: the NATHAN 1 trial,” Diabetes Care, 34, No. 9, 2054–2060 (2011).
T. M. Kuchmerovskaya, P. K. Parhomets, G. V. Chichkovskaya, et al., “Nature of the brain synaptic membranes that bind nicotinamide adenine dinucleotide,” Neurochemistry,4, No. 4, 373–378 (1985).
L. J. Yan, “Redox imbalance stress in diabetes mellitus: Role of the polyol pathway,” Anim. Model Exp. Med., 1, No. 1, 7–13 (2018).
W. H. Tang, K. A. Martin, and J. Hwa, “Aldose reductase, oxidative stress, and diabetes mellitus,” Front. Pharmacol., 3, 87–94 (2012).
G. C. Ferreira and M. C. McKenna, “L-Carnitine and acetyl-L-carnitine roles and neuroprotection in developing brain,” Neurochem. Res., 46, No. 6, 1661–1675 (2017).
T. Aureli, M. E. Di Cocco, C. Puccetti, et al., “Acetyl-Lcarnitine modulates glucose metabolism and stimulates glycogen synthesis in rat brain,” Brain Res., 796, Nos. 1/2, 75–81 (1998).
G. C. Ferreira and M. C. McKenna, “L-Carnitine and acetyl-L-carnitine roles and neuroprotection in developing brain,” Neurochem. Res., 42, No. 6, 1661–1675 (2017).
T. M. Hagen, J. Liu, J. Lykkesfeldt, et al., “Feeding acetyl-L-carnitine and lipoic acid to old rats significantly improves metabolic function while decreasing oxidative stress,” Proc. Natl. Acad. Sci. USA, 99, No. 4, 1870–1875 (2002).
H. Schupke, R. Hempel, G. Peter, et al., “New metabolic pathways of alpha-lipoic acid,” Drug Metab. Dispos., 29, No. 6, 855–862 (2001).
M. Sinha, A. Bir, A. Banerjee, et al., “Multiple mechanisms of age-dependent accumulation of amyloid beta protein in rat brain: Prevention by dietary supplementation with N-acetylcysteine, α-lipoic acid and α-tocopherol,” Neurochem. Int., 95, 92–99 (2016).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuchmerovska, T.M., Dyakun, K.O., Guzyk, M.M. et al. Effects of a Combined Mitochondria-Targeted Treatment on the State of Mitochondria and Synaptic Membranes from the Brains of Diabetic Rats. Neurophysiology (2019). https://doi.org/10.1007/s11062-019-09816-6
Received:
Published:
DOI: https://doi.org/10.1007/s11062-019-09816-6