Abstract
Background Performance status (PS) scoring systems are tools of immense clinical importance in the management of patients with malignant disease but these tools are subjective and do not provide an objective evaluation of physical functioning. We conducted a pilot study to explore the feasibility and clinical utility of functional capacity testing to assess physical functioning in recurrent primary malignant glioma patients. Patients and methods Using a cross-sectional design, consecutive patients with recurrent glioma performed a six minute walk (6MW) test to assess functional capacity. Performance status was assessed using Karnofsky Performance Status (KPS) scoring system. QOL was assessed by the Functional Assessment of Cancer Therapy-Brain scale. Self-reported exercise behavior was assessed using the Godin Leisure Time Exercise Questionnaire (GLTEQ). Results A total of 171 patients were recruited and tested. Seventy percent were diagnosed with glioblastoma multiforme (WHO grade IV) and 85% were undergoing therapy. Median KPS was 90% (range, 70–100%). Median 6MW distance was 400 m (range, 102–630 m), equivalent to 56 ± 13% (range, 14–87%) of that predicted for age and sex. KPS, self-reported exercise, and QOL increased across 6MW distance quartiles (P < 0.05) although there was considerable variation within each category. 6MW distance and KPS were significantly correlated with each other (r = 0.34, P < 0.01) and several QOL domains (range, r = −0.43 to 0.46, P < 0.05). Conclusions 6MW distance is a clinically feasible tool that provides an objective measure of physical functioning in select patients with recurrent glioma. Further research is required to investigate the prognostic value of these tests in patients with advanced malignancy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Performance status (PS) scoring systems are tools of immense clinical importance in the management of patients with newly diagnosed or recurrent primary high grade glioma [1–3]. PS involves the subjective evaluation of patients’ global physical functioning and capability of independent living and self-care as determined by oncology professionals. Using scoring systems such as KPS, PS has been demonstrated to be a strong, independent predictor of survival and QOL with higher scores favoring better prognosis [1–7]. Indeed, KPS is a central component, together with other factors (e.g., age, extent of resection, etc.), of the established recursive partitioning analysis (RPA) undertaken by the Radiation Therapy Oncology Group (RTOG) [2, 3] as well as a newly derived prognosis prediction nomogram for newly diagnosed glioblastoma [1]. Both tools can be used to classify patients into prognostic risk categories and are routinely used clinically in the planning, randomization, eligibility, and evaluation of clinical trials as well as in decisions regarding the ‘optimal’ therapeutic approach in clinical practice.
Despite widespread clinical use, PS scoring systems are subjective, have poor inter-rate reliability, and are weak predictors of prognosis when PS is ‘good’ (i.e., KPS, ≥70%; ECOG, 0-1) [8–11]. Further, high grade glioma patients receive a broad of range of concurrent or sequential aggressive therapies that profoundly impact patients’ physical functioning and QOL [12–14]; QOL and functional health status (assessed via self-report) are independent predictors of survival in primary glioma [15–18]. Thus, alternative methods that provide an objective assessment of physical functioning may allow for more accurate prognostication and personalized patient care.
Functional capacity testing is widely used in numerous clinical settings and provides a wealth of prognostic and decision-making information [19]. Among the wide number of tests available, a six minute walk test (6MWT) has been demonstrated to be a simple and clinically feasible test to evaluate functional capacity, and a robust predictor of mortality in numerous clinical settings [20–24]. The 6MWT was originally developed for patients with severe cardiac or pulmonary disease; the utility of this test to assess functional capacity among individuals with neurological impairments, poor PS and life expectancy, and comorbid disease such as recurrent glioma patients is unknown. Accordingly, we conducted a pilot study to explore the feasibility and clinical utility of the 6MWT to assess physical functioning in 171 patients with recurrent glioma. We also examined the correlation between 6MW distance, KPS, QOL, and other clinical variables. We hypothesized that a 6MWT would be a feasible and valid tool to assess physical functioning, and correlated with KPS, QOL, and several clinical variables.
Method
Setting and patients
Patients with histologically confirmed, recurrent WHO grade III/IV malignant glioma (i.e., glioblastoma multiforme, anaplastic astrocytoma, or anaplastic oligodendroglioma) presenting at the Preston Robert Tisch Brain Tumor Center (PRT-BTC) at Duke University Medical Center (DUMC). Other major eligibility criteria were: (1) legal age (>18 years old), (2) Karnofsky performance status of at least 70% at study entry, (3) primary attending oncologist approval, (4) ability to read and understand English, and (5) no contraindications to a 6MWT as per American Thoracic Society (ATS) recommendations [25]. Patients who were currently undergoing therapy or off-therapy were eligible for study participation. The DUMC institutional review board approved the study and written informed consent was obtained from all participants prior to initiation of any study procedures.
Procedures
Using an observational, cross-sectional design, potential participants were identified and screened for eligibility via medical record review of patients scheduled for routine follow-up care consultation at the PRT-BTC. Immediately following the consultation and primary attending oncologist approval, potential patients were provided with a thorough review of the study by the study coordinator and asked if they were willing to participate. Consenting patients were then asked to complete a 6MWT and the study questionnaire.
Outcome assessments
Functional capacity
Functional capacity was assessed using a 6MWT [25]. The patient was instructed to walk at their fastest pace and to cover the longest possible distance over 6 min under the supervision of an American College of Sports Medicine (ASCM) certified exercise specialist. All participants were provided with standard phrases of encouragement throughout the assessment in accordance with the ATS guidelines [25]. The distance walked was determined in a measured corridor and performed according to the ATS guidelines [25]. During exercise, oxyhemoglobin saturation (SpO2) and heart rate was monitored continuously using pulse oximetry (BCI, Hand-Held Pulse Oximeter, Waukesha, WN). Age and sex-predicted 6MWD was calculated from the equation of Gibbons et al. [26].
Quality of life
Quality of life (QOL) was assessed using the Functional Assessment of Cancer Therapy-Brain (FACT-BR) scale [27]. The FACT-BR contains subscales for physical (PWB, 7-items), functional (FWB, 7-items), emotional (EWB, 6-items), and social/family (SWB, 7-items) well-being. The four subscales were summed to obtain the FACT-G score (all 27-items). In addition, the FACT-BR contains a 19-item brain cancer subscale (BCS) which assesses symptoms commonly reported by brain cancer patients (e.g., problems concentrating; cognitive function; vision, etc.) [27]. Fatigue was assessed by the 13-item Fatigue Scale (FS) of the FACT measurement system developed specifically for cancer patients [28].
Clinical parameters and performance status
Medical characteristics were abstracted from medical records. Performance status was assessed using the KPS scale and was assessed at the time of study enrollment by the attending oncologist. KPS scores range from 0 (dead) to 100 (normal; no evidence of disease and no physical complaints). Self-reported exercise over the past month was assessed by the Godin Leisure Time Exercise Questionnaire [29, 30].
Statistical analysis
The initial analysis provided descriptive information on clinical parameters and study outcomes. To determine differences on study endpoints based on 6MW distance (m) quartiles (i.e., <320, 320–399.9, 400–463.9, ≥464 m) we used one-way analysis of variance (ANOVA) followed by post-hoc pair-wise comparisons as appropriate. Linear regression analysis was used to determine the univariate association between 6MW distance and KPS, QOL, exercise behavior, and select clinical characteristics. To facilitate interpretation, select clinical characteristics were collapsed into dichotomous variables as follows: age (<50 vs. ≥50 years); sex (male vs. female); histology (WHO grade IV vs. WHO grade III); therapy (on-therapy vs. off-therapy); time since diagnosis (<32 vs. ≥32 months); and decadron use (yes vs. no). Data are presented as mean ± standard deviation. Statistical significance was set at P < 0.05 for all analyses.
Results
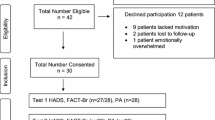
Participant recruitment took place between May 2007 and June 2008. In brief, 567 patients were screened for study eligibility during the study period. Of these, 382 (67%) met inclusion criteria and 171 (45%) agreed to participate and completed all study procedures. Major reasons for non-eligibility were KPS <70% (n = 75), oncologist non-approval (n = 33), and physical impairment (n = 12). Major reasons for study refusal were not interested (n = 54), no time (n = 36), and not feeling well (n = 22).
Clinical characteristics
Patient characteristics are presented in Table 1. In summary, participants mean age was 49 ± 11 (range, 20–77 years) and 68% were male. Seventy percent were diagnosed with glioblastoma multiforme, median KPS was 90% (ranging from 70 to 100%), and 85% were currently on active therapy. The median number of disease progressions was 1 (range, 1–7). The median time to study enrollment from original diagnosis was 22 months (range, 3–176 months). Median 6MW distance was 400 (range, 102–630 m), equivalent to 44% below that predicted for age and gender. There were no adverse events or abnormal heart rate or SpO2 responses during testing. Participants reported a mean of 164 ± 201 total weekly exercise minutes; 25% were currently meeting national exercise guidelines (i.e., ≥150 min of moderate to vigorous exercise/week) [31], and 26% reported no exercise behavior. Mean overall QOL, as assessed by the FACT-BR, was 132 ± 23 (range, 50–178) while mean Trial Outcome Index (TOI) and fatigue was 91 ± 4 (range, 29–126) and 18 ± 11 (range, 1–49), respectively. Concerning QOL subscales, mean PWB, FWB, SWB, EWB, and BCS was 22 ± 5, 19 ± 6, 19 ± 4, 23 ± 4, and 50 ± 12, respectively.
Univariate analysis: clinical variables associated with walk distance
Univariate associations between 6MW distance, KPS, and clinical variables are presented in Table 2. Variables significantly correlated with 6MW distance were overall QOL, TOI, fatigue, and KPS (range, r = −0.28 to 0.37, P < 0.05). Variables significantly correlated with KPS were age, decadron use, overall QOL, TOI, and fatigue (r = −0.43 to 0.46, P < 0.05). Higher 6MWD and KPS was associated with higher QOL and lower fatigue. Neither 6MWD nor KPS were correlated with self-reported exercise behavior.
Differences between clinical variables based on 6MWD quartiles are presented in Table 3. There was a linear improvement for all clinical variables across 6MW distance quartiles (P < 0.05), except self-reported exercise behavior and percent meeting exercise guidelines. KPS increased across 6MW distance quartiles with the lowest KPS score among patients with a 6MW distance <320 m (KPS = 83 ± 8%) and the highest among those with a 6MW distance ≥464 (KPS = 90 ± 8%). The identical pattern was observed for self-reported exercise behavior and percentage of participants meeting national exercise guidelines. QOL increased across 6MW distance quartiles with the lowest QOL score among patients with a 6MW distance <320 m (QOL = 119 ± 24) and the highest among those with a 6MW distance ≥464 (QOL = 141 ± 23). Identical patterns were observed for all remaining QOL composite scores (i.e., TOI), brain-cancer specific concerns, and fatigue.
Discussion
The principal finding of this study was that the 6MWT is a safe and clinically feasible tool to evaluate functional capacity in select patients with primary recurrent glioma. A second important finding was that recurrent glioma patients have a significant and marked reduction in functional capacity. Finally, higher 6MW distance was associated with higher KPS, self-reported exercise behavior, and sub-domains of QOL. To our knowledge, this is the first study to investigate the clinical utility of a 6MWT to evaluate physical functioning in any cancer population.
The 6MWT was originally developed for patients with severe cardiac disease (i.e., heart failure). Over the past decade, the clinical utility of test has been further established in a wide range of clinical populations including patients with chronic obstructive pulmonary disease (COPD), peripheral artery disease, rheumatoid arthritis, advanced emphysema, pulmonary artery hypertension, and scleroderma. The value of these tests in a population with severe neurological and functional impairments as well as poor life expectancy, such as recurrent glioma patients, is not known. Given the strong prognostic value of subjective measures of physical functioning in recurrent glioma together with the highlighted limitations of these instruments, we felt that the 6MWT may provide a more sensitive but clinically feasible alternative measure of physical functioning. In support of this hypothesis, the results of this study indicate that a 6MWT is a safe and feasible assessment tool to objectively evaluate physical functioning in select patients with recurrent glioma. Indeed, despite poor PS and severe neurological and functional impairments, recurrent glioma patients were able to adequately conduct these tests with no adverse events.
Clearly, 6MWTs are only appropriate for patients with a ‘good’ PS (i.e., KPS ≥70%) through limiting the generalizability and clinical utility of these tests to all patients with recurrent glioma or advanced malignancy. Indeed, of 567 screened for this study, 67% met inclusion criteria. The major reason for non-eligibility was poor KPS (i.e., <70%). Thus, our present results are likely only generalizable to patients with good KPS, less advanced disease, and experiencing less treatment-related complications. However, we contend that objective measures of physical functioning may be of most value in these patients. For instance, PS scoring systems have been shown to be weak predictors of prognosis when PS is good (i.e., ≥70%). We speculate that among patients with ‘good’ PS, subjective scoring systems are unable to successfully discriminate between patients with a KPS of 70 vs. 80%, 90%, or 100%. This has important implications since KPS is frequently considered in the planning, randomization, eligibility, and evaluation of clinical trials and in decisions regarding the ‘optimal’ therapeutic approach in clinical practice. Indeed, in the majority of trials, a KPS ≥70% is often an eligibility criterion. As demonstrated in this study, 6MW distance varies dramatically among patients within KPS categories of 70 to 100%, suggesting that a 6MWT may provide a more sensitive and ‘real’ evaluation of physical functioning than KPS. However, whether a more sensitive evaluation of physical functioning translates into more accurate prognosis prediction is not known and an important question for future research.
In this study, median 6MW distance was 400 m or 44% below that of age–sex matched comparison data despite a minimum ‘good’ KPS eligibility requirement of ≥70%. The poor functional capacity is lower than in our prior work among patients with inoperable breast and lung cancer [32]. We reported that exercise capacity, as assessed by a cardiopulmonary exercise test to measure peak oxygen consumption (VO2peak), was 33 and 32% below age–sex predicted norms among 46 advanced lung cancer patients and 39 women with metastatic breast cancer receiving palliative chemoradiation [32]. Although 6MWTs are correlated with VO2peak (r = 0.67) [33], such tests are submaximal and underestimate maximal exercise capacity. Nevertheless, such testing provides a robust assessment of functional capacity and is appropriate for frail, elderly, or severely deconditioned clinical populations such as patients with advanced malignancy [24]. Further such tests provide a simple, safe, and cheap objective assessment that can be performed in numerous clinical and research settings.
The clinical importance of poor functional capacity among patients with primary glioma or any other cancer population is not known. 6MW distance is a strong independent predictor of morbidity and mortality in a wide range of clinical populations [20–23]. For example, Lederer and colleagues reported that idiopathic pulmonary fibrosis (IPF) patients with a 6MWD less than 350 m had significantly shorter survival than those with a 6MWD greater than 350 m [34]. A recent updated study from the same group reported an optimal patient stratification of <207 vs. ≥207 m among IPF patients [20]. Other groups have reported similar findings [35]. In this study, 30 and 4% of patients were below the 350 and 207 m cutpoints, respectively. Further studies investigating the prognostic value of objective measures of physical functioning (e.g., 6MWD, VO2peak) among cancer patients, especially those with advanced disease, are required.
A final noteworthy finding of this study was that both 6MWD and KPS were significantly correlated with each other and several clinical endpoints (e.g., QOL, fatigue). These findings corroborate prior reports indicating that KPS is associated with QOL among patients with primary glioma [13, 14], and our prior work indicating that objective measures of physical functioning are associated with QOL in other malignancies with higher scores favoring higher QOL and lower fatigue [36]. Together, these findings provide preliminary evidence that interventions demonstrated to increase 6MWD, may, in turn cause favorable improvements in QOL and fatigue among recurrent glioma patients. Several studies have provided strong evidence that structured exercise training interventions are associated with significant and potential clinically meaningful improvements in QOL across numerous cancer populations at various stages of disease and treatment [37]. Prior studies have not, however, studied the effects of exercise training among cancer patients with neurologic and/or cognitive abnormalities although patients suffering from hemiparetic stroke [38] and Parkinson’s disease [39] derive substantial benefit from exercise rehabilitation. Future studies investigating the safety and efficacy of appropriately prescribed exercise training on QOL and other relevant endpoints among patients with primary glioma are required.
This study does have limitations. The most important limitation, as previously discussed, is patient selection bias because of the relatively low eligibility rate, the transparent purpose of the investigation, and exclusion of patients with poor KPS (<70%). As such, patients with better KPS, less advanced disease, and experiencing less treatment-related complications were probably more likely to participate in this study. In addition, we only examined the relationship between 6MWD and KPS as rated by only one observer (i.e., the attending oncologist). Future studies are required to investigate the relationship between objective measures of physical functioning and other PS scoring systems used in the oncology setting. Other limitations are the relatively small sample size, particularly those with a KPS of 70%, and the cross-sectional study design. Future large-scale studies are required using prospective and/or repeated measure designs.
In summary, this study provides promising but preliminary evidence for the feasibility of a 6MWT to evaluate functional capacity in select patients with recurrent glioma. Large-scale studies investigating the prognostic value of these tests in patients with advanced malignancy are now required. Identifying new informative predictive parameters that complement existing prognostic risk classification systems can facilitate the paradigm shift towards personalized medicine.
References
Gorlia T, van den Bent MJ, Hegi ME, Mirimanoff RO, Weller M, Cairncross JG et al (2008) Nomograms for predicting survival of patients with newly diagnosed glioblastoma: prognostic factor analysis of EORTC and NCIC trial 26981-22981/CE.3. Lancet Oncol 9(1):29–38. doi:10.1016/S1470-2045(07)70384-4
Curran WJ Jr, Scott CB, Horton J, Nelson JS, Weinstein AS, Fischbach AJ et al (1993) Recursive partitioning analysis of prognostic factors in three Radiation Therapy Oncology Group malignant glioma trials. J Natl Cancer Inst 85(9):704–710. doi:10.1093/jnci/85.9.704
Scott CB, Scarantino C, Urtasun R, Movsas B, Jones CU, Simpson JR et al (1998) Validation and predictive power of Radiation Therapy Oncology Group (RTOG) recursive partitioning analysis classes for malignant glioma patients: a report using RTOG 90-06. Int J Radiat Oncol Biol Phys 40(1):51–55. doi:10.1016/S0360-3016(97)00485-9
Buckner JC (2003) Factors influencing survival in high-grade gliomas. Semin Oncol 30(6 Suppl 19):10–14. doi:10.1053/j.seminoncol.2003.11.031
Filippini G, Falcone C, Boiardi A, Broggi G, Bruzzone MG, Caldiroli D et al (2008) Prognostic factors for survival in 676 consecutive patients with newly diagnosed primary glioblastoma. Neuro Oncol 10(1):79–87. doi:10.1215/15228517-2007-038
Donato V, Papaleo A, Castrichino A, Banelli E, Giangaspero F, Salvati M et al (2007) Prognostic implication of clinical and pathologic features in patients with glioblastoma multiforme treated with concomitant radiation plus temozolomide. Tumori 93(3):248–256
Nieder C, Astner ST, Molls M, Grosu AL (2007) Analysis of long-term survivors of glioblastoma multiforme in a single institution with aggressive local retreatment protocol. Anticancer Res 27(4C):2993–2996
Kaasa T, Wessel J (2001) The Edmonton Functional Assessment Tool: further development and validation for use in palliative care. J Palliat Care 17(1):5–11
Abernethy AP, Shelby-James T, Fazekas BS, Woods D, Currow DC (2005) The Australia-modified Karnofsky Performance Status (AKPS) scale: a revised scale for contemporary palliative care clinical practice ISRCTN81117481. BMC Palliat Care 4:7. doi:10.1186/1472-684X-4-7
Schag CC, Heinrich RL, Ganz PA (1984) Karnofsky performance status revisited: reliability, validity, and guidelines. J Clin Oncol 2(3):187–193
Yates JW, Chalmer B, McKegney FP (1980) Evaluation of patients with advanced cancer using the Karnofsky performance status. Cancer 45(8):2220–2224. doi:10.1002/1097-0142(19800415)45:8<2220::AID-CNCR2820450835>3.0.CO;2-Q
Whitton AC, Rhydderch H, Furlong W, Feeny D, Barr RD (1997) Self-reported comprehensive health status of adult brain tumor patients using the Health Utilities Index. Cancer 80(2):258–265. doi:10.1002/(SICI)1097-0142(19970715)80:2<258::AID-CNCR14>3.0.CO;2-T
Giovagnoli AR, Tamburini M, Boiardi A (1996) Quality of life in brain tumor patients. J Neurooncol 30(1):71–80. doi:10.1007/BF00177445
Mackworth N, Fobair P, Prados MD (1992) Quality of life self-reports from 200 brain tumor patients: comparisons with Karnofsky performance scores. J Neurooncol 14(3):243–253. doi:10.1007/BF00172600
Mauer M, Stupp R, Taphoorn MJ, Coens C, Osoba D, Marosi C et al (2007) The prognostic value of health-related quality-of-life data in predicting survival in glioblastoma cancer patients: results from an international randomised phase III EORTC Brain Tumour and Radiation Oncology Groups, and NCIC Clinical Trials Group study. Br J Cancer 97(3):302–307. doi:10.1038/sj.bjc.6603876
Mauer ME, Taphoorn MJ, Bottomley A, Coens C, Efficace F, Sanson M et al (2007) Prognostic value of health-related quality-of-life data in predicting survival in patients with anaplastic oligodendrogliomas, from a phase III EORTC brain cancer group study. J Clin Oncol 25(36):5731–5737. doi:10.1200/JCO.2007.11.1476
Taphoorn MJ, van den Bent MJ, Mauer ME, Coens C, Delattre JY, Brandes AA et al (2007) Health-related quality of life in patients treated for anaplastic oligodendroglioma with adjuvant chemotherapy: results of a European Organisation for Research and Treatment of Cancer randomized clinical trial. J Clin Oncol 25(36):5723–5730. doi:10.1200/JCO.2007.12.7514
McCarter H, Furlong W, Whitton AC, Feeny D, DePauw S, Willan AR et al (2006) Health status measurements at diagnosis as predictors of survival among adults with brain tumors. J Clin Oncol 24(22):3636–3643. doi:10.1200/JCO.2006.06.0137
Ross RM (2003) ATS/ACCP Statement on cardiopulmonary exercise testing. Am J Respir Crit Care Med 167(2):211–277. doi:10.1164/rccm.167.2.211
Lederer DJ, Arcasoy SM, Wilt JS, D’Ovidio F, Sonett JR, Kawut SM (2006) Six-minute-walk distance predicts waiting list survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 174(6):659–664. doi:10.1164/rccm.200604-520OC
Lettieri CJ, Nathan SD, Browning RF, Barnett SD, Ahmad S, Shorr AF (2006) The distance-saturation product predicts mortality in idiopathic pulmonary fibrosis. Respir Med 100(10):1734–1741. doi:10.1016/j.rmed.2006.02.004
Flaherty KR, Andrei AC, Murray S, Fraley C, Colby TV, Travis WD et al (2006) Idiopathic pulmonary fibrosis: prognostic value of changes in physiology and six-minute-walk test. Am J Respir Crit Care Med 174(7):803–809. doi:10.1164/rccm.200604-488OC
Cote CG, Pinto-Plata V, Kasprzyk K, Dordelly LJ, Celli BR (2007) The 6-min walk distance, peak oxygen uptake, and mortality in COPD. Chest 132(6):1778–1785. doi:10.1378/chest.07-2050
Jones LW, Eves ND, Haykowsky M, Joy AA, Douglas PS (2008) Cardiorespiratory exercise testing in clinical oncology research: systematic review and practice recommendations. Lancet Oncol 9(8):757–765
ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories (2002) ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med 166(1):111–117
Gibbons WJ, Fruchter N, Sloan S, Levy RD (2001) Reference values for a multiple repetition 6-minute walk test in healthy adults older than 20 years. J Cardiopulm Rehabil 21(2):87–93. doi:10.1097/00008483-200103000-00005
Weitzner MA, Meyers CA, Gelke CK, Byrne KS, Cella DF, Levin VA (1995) The Functional Assessment of Cancer Therapy (FACT) scale. Development of a brain subscale and revalidation of the general version (FACT-G) in patients with primary brain tumors. Cancer 75(5):1151–1161. doi:10.1002/1097-0142(19950301)75:5<1151::AID-CNCR2820750515>3.0.CO;2-Q
Yellen SB, Cella DF, Webster K, Blendowski C, Kaplan E (1997) Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. J Pain Symptom Manage 13(2):63–74. doi:10.1016/S0885-3924(96)00274-6
Godin G, Jobin J, Bouillon J (1986) Assessment of leisure time exercise behavior by self-report: a concurrent validity study. Can J Public Health 77(5):359–362
Godin G, Shephard RJ (1985) A simple method to assess exercise behavior in the community. Can J Appl Sport Sci 10(3):141–146
American College of Sports Medicine Position Stand (1998) The recommended quantity and quality of exercise for developing and maintaining cardiorespiratory and muscular fitness, and flexibility in healthy adults. Med Sci Sports Exerc 30(6):975–991. doi:10.1097/00005768-199806000-00032
Jones LW, Eves ND, Mackey JR, Peddle CJ, Haykowsky M, Joy AA et al (2007) Safety and feasibility of cardiopulmonary exercise testing in patients with advanced cancer. Lung Cancer 55(2):225–232. doi:10.1016/j.lungcan.2006.10.006
Win T, Jackson A, Groves AM, Sharples LD, Charman SC, Laroche CM (2006) Comparison of shuttle walk with measured peak oxygen consumption in patients with operable lung cancer. Thorax 61(1):57–60. doi:10.1136/thx.2005.043547
Kawut SM, O’Shea MK, Bartels MN, Wilt JS, Sonett JR, Arcasoy SM (2005) Exercise testing determines survival in patients with diffuse parenchymal lung disease evaluated for lung transplantation. Respir Med 99(11):1431–1439. doi:10.1016/j.rmed.2005.03.007
Hallstrand TS, Boitano LJ, Johnson WC, Spada CA, Hayes JG, Raghu G (2005) The timed walk test as a measure of severity and survival in idiopathic pulmonary fibrosis. Eur Respir J 25(1):96–103. doi:10.1183/09031936.04.00137203
Jones LW, Eves ND, Mackey JR, Peddle CJ, Haykowsky M, Joy AA et al (2008) Systemic inflammation, cardiorespiratory fitness, and quality of life in patients with advanced non-small cell lung cancer. J Thorac Oncol 3(2):194–195. doi:10.1097/JTO.0b013e318160f36b
Jones LW, Demark-Wahnefried W (2006) Diet, exercise, and complementary therapies after primary treatment for cancer. Lancet Oncol 7(12):1017–1026. doi:10.1016/S1470-2045(06)70976-7
Dobkin BH (2008) Training and exercise to drive poststroke recovery. Nat Clin Pract Neurol 4(2):76–85. doi:10.1038/ncpneuro0709
Fisher BE, Wu AD, Salem GJ, Song J, Lin CH, Yip J et al (2008) The effect of exercise training in improving motor performance and corticomotor excitability in people with early Parkinson’s disease. Arch Phys Med Rehabil 89(7):1221–1229. doi:10.1016/j.apmr.2008.01.013
Acknowledgements
This study was supported by funds from the Tug McGraw Research Center and the Preston Robert Tisch Brain Tumor Center at Duke.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jones, L.W., Cohen, RR., Mabe, S.K. et al. Assessment of physical functioning in recurrent glioma: preliminary comparison of performance status to functional capacity testing. J Neurooncol 94, 79–85 (2009). https://doi.org/10.1007/s11060-009-9803-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-009-9803-x