Abstract
Purpose
Exercise has numerous benefits for cancer survivors, but very limited research to date has exclusively examined brain cancer patients, specifically those diagnosed with high-grade glioma (HGG). This study examined (1) the feasibility of recruiting HGG patients to an exercise-based study and performing fitness assessments; (2) exercise counseling and programming preferences; and (3) associations between fitness, physical activity (PA), and quality of life (QOL).
Methods
Participants completed assessments prior to starting Temozolamide chemotherapy with radiation (T1), at 2 months and 8 months. Fitness was measured with an incremental cycling exercise test to volitional exhaustion (VO2peak) and hand grip dynamometry. The Godin leisure time questionnaire measured PA and the functional assessment for cancer therapy, brain cancer module (FACT-Br) measured QOL.
Results
Of the 35 approached, N = 16 participated. Due to safety concerns, the aerobic fitness test protocol was altered. Participants preferred to exercise during treatment, alone and unsupervised, at home, and at a moderate intensity. Few participants (<25%) met guidelines for PA at any time point. At T1, aerobic capacity was associated with the FACT Trial Outcome Index (TOI) (r = 0.619, p < 0.05). At 2 months, PA minutes were associated with FACT-TOI (r = 0.653, p = 0.057), FACT-G (r = 0.711, p < 0.05), and FACT-Br scores (r = 0.722, p < 0.05).
Conclusions
Recruitment rate was similar to a previous study in HGG populations, but study completion rate was lower. Most exercise counseling and programming preferences were similar to previous brain cancer patients. Assessing aerobic fitness to VO2peak was not feasible. Aerobic fitness and PA were positively associated with QOL.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
High-grade gliomas (HGG) are aggressive tumors of the brain that include anaplastic lesions and glioblastoma multiformes (GBM). These tumors, particularly GBMs, are associated with a median survival of approximately 4 months in the absence of treatment, improving to nearly 15 months with current therapy [22]. However, a recent review of local data in Calgary, Alberta suggests that the current median survival for patients who have undergone treatment is likely an underestimation of the actual potential survival due to treatment benefit. After reviewing 93 HGG patients treated at the Tom Baker Cancer Centre in Calgary, Alberta, it was observed that 25% of patients (N = 26) failed to complete treatment, with the most common reason being treatment toxicity. In the absence of new therapies, techniques must be identified that will decrease toxicity and allow more patients to complete treatments and therefore potentially improve long-term survival and quality of life (QOL).
One intervention that holds significant promise is the incorporation of individualized exercise therapy for HGG patients [3]. The benefits of exercise for cancer survivors in general have been well documented; however, only a few studies to date have looked exclusively at the brain cancer population [1, 12–14, 17, 19, 20]. Given the evidence linking fitness and exercise with decreased chemotherapy-induced toxicity [2], improved chemotherapy completion rate [4], and improved survival [7, 15, 18, 20], more exploratory studies examining the role of exercise in HGG patients undergoing treatment are warranted. Only one previous study has tested the feasibility of accruing and retaining HGG patients in an exercise-based study and examined associations between PA levels, fitness, and QOL over time [14]. Before large-scale exercise trials can be conducted with HGG patients, more studies of this nature are needed.
Based on the limited research to date in this population, our research questions for this study were (1) is it feasible to accrue newly diagnosed HGG patients into an exercise study and assess fitness parameters? (2) What are the exercise counseling and programming preferences of HGG patients? (3) Is there a relationship between fitness parameters and QOL and PA levels and QOL, and does this change over the course of standard treatment?
It was hypothesized that accrual, interest, and participation in fitness assessments would be feasible. It was also hypothesized that exercise preferences would be similar to that seen in previous literature in other types of cancers—with a preference for home-based or group-supported programming and a desire for education around the role of exercise. Finally, we hypothesized that across the trajectory of treatment, patients would experience a decrease in fitness indices, exercise levels, and QOL.
Method
Study design
This study examined the feasibility (i.e., recruitment and adherence) of an exercise study for HGG brain cancer patients receiving treatment. Exercise assessments included an incremental cycling exercise test to measure aerobic capacity and hand grip strength to measure upper body strength. Participants completed questionnaires to assess exercise counseling and programming preferences, PA levels, and QOL. Fitness assessments, PA, and QOL questionnaires were administered at three time points: (1) immediately prior to starting 6 weeks of concurrent Temozolamide chemotherapy with radiation (T1), (2) during the 4-week off-treatment period after concurrent therapy (i.e., approximately 2 months from T1), and (3) after 6 months of adjuvant Temozolamide (i.e., approximately 8 months from T1). Exercise counseling and programming preferences were assessed at T1 only.
Participants and recruitment
Participants were recruited through the Neuro Oncology group at the Tom Baker Cancer Centre. Approximately 80 HGG patients (55 GBM, 25 anaplastic) are seen each year at this cancer center. The referring oncologist presented the study to potential participants. If interested, the potential participant met with the study coordinator after their medical visit. The study coordinator presented full details of the study to the potential participant, and if interested, obtained consent and set up the initial exercise assessment at the University testing location. Recruitment occurred over a 16-month period. All patients were treated using the standard therapeutic protocol [22]. Eligible participants had to be 18 years or older, able to speak and write English, have a pathological diagnosis of HGG, be postsurgical, and must not have received previous treatment for brain tumors. Informed consent was obtained from all individual participants included in the study. All procedures performed in this study were approved by the University’s research ethics board for human subjects. Participant baseline characteristics are provided in Table 1.
Measures
Fitness parameters
Aerobic capacity was assessed by an incremental exercise test, performed with 12-lead ECG monitoring (Quinton Q-Stress, Cardiac Sciences Corp, Bothell, WA) and expired gas analysis (Parvo Medics TrueOne 2400, East Sandy, UT) on an electrically braked cycle ergometer (Lode Corival, Groningen, NL), using a ramped protocol. During each test, arterial oxygen saturation (Nellcor N-600×, Covidien, Manfield, MA) and heart rate (Quinton Q-Stress) were monitored continuously, while blood pressure (manual sphygmomanometry) and exertional symptoms (rated perceived exertion, dyspnea, leg fatigue) were measured every 2 min. This testing protocol has been tested with advanced stage cancer patients and is considered safe and appropriate for this population with proper monitoring [10]. Grip strength was assessed following the Canadian Physical Activity, Fitness and Lifestyle Approach guidelines, using hand grip dynamometer (Smedley Dynamometer, TTM, Tokyo, Japan) measured to the nearest 1.0 kg.
Questionnaires
Exercise counseling and programming preferences were measured with a series of questions derived from previous cancer and exercise preference literature [9]. Participants indicated their preferences for counseling (i.e., discussion and delivery of information about exercise) and programming (i.e., participation in exercise or physical activity).
PA levels were measured with three items from the Godin Leisure Time Exercise Questionnaire (GLTEQ) [6]. These items measure the frequency and duration of mild, moderate, and strenuous bouts of exercise performed during free time in a typical week. This tool has been successfully used with adult cancer patients and survivors and is considered reliable and valid in comparison to nine other self-report measures of exercise [8]. Total weekly PA minutes were calculated by multiplying the frequency and duration of each mild, moderate, and vigorous activity and adding these together. To determine whether participant reached the prescribed PA guidelines of 150 min of moderate, or 75 min of vigorous activity, total weekly moderate activity was added to two times vigorous activity minutes [21].
QOL was measured with the Functional Assessment for Cancer Therapy–Brain Cancer Module (FACT-Br). The FACT-Br was developed specifically for brain cancer survivors and includes the following subscales of QOL: physical, emotional, social-family, and functional well-being, Trial Outcome Index (TOI) (which is the sum of the physical well-being (PWB), functional well-being (FWB), and “additional concerns” subscales), and FACT general (FACT-G) which is a sum of all subscales except the brain cancer-specific considerations. This tool has been found to be reliable, valid, responsive, brief, and easy to administer in brain cancer survivors [23, 24].
Data analysis
Descriptive analyses explored and described sample characteristics, exercise counseling, and programming preferences. Spearman’s Rho nonparametric correlations examined relationships between aerobic fitness, PA levels, QOL, and medical information at baseline and 2 months post-baseline. Changes in the fitness and self-report measures over time were calculated, but due to small sample sizes, and the exploratory nature of a feasibility study, statistical significance is not reported.
Results
Feasibility
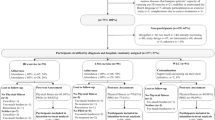
A total of 51 patients met the initial study criteria and were deemed eligible to participate. Out of 51 eligible patients, 35 were approached. Those who were not approached either lived outside of the study area or were not interested in meeting with study recruitment staff. Sixteen of the 35 patients approached agreed to participate in the study, resulting in a recruitment rate of 45.7%. Reasons for declining to participate in the study included the following: living outside of the study area, being too overwhelmed by their diagnosis, or feeling too unwell and not interested in participating. All study participants were diagnosed with stage/grade IV HGG. Of the 16 participants who completed baseline assessments, 10 completed 2-month assessments (62.5%), and 3 (18.8%) completed 8-month assessments. Participant attrition from baseline assessment to 2-month and 8-month assessment is shown in Fig. 1.
Feasibility of fitness assessments
The planned testing protocol for aerobic fitness was to end the test at volitional exhaustion (VO2peak). However, the physicians supervising these exercise tests had concerns regarding the safety of this protocol for participants. Therefore, instead, participants were stopped after reaching the second ventilatory threshold (VT2). At T1, 12 participants (75%) achieved VT2, one stopped due to shortness of breath, one due to discomfort, and two were stopped by the physician in charge of the test due to high blood pressure. At the 2-month assessment, 7 participants (70%) achieved VT2, 3 stopped due to leg fatigue or unable to maintain workload. At the 8-month assessment, the test was terminated by all three participants before VT2 for various reasons, including leg discomfort, dry mouth and the inability to breathe out of the nose, and cadence dropped significantly. Grip strength was well tolerated by participants.
Fitness outcomes
Fitness outcomes are presented in Table 2. The participants (N = 3) who performed assessments at all three time points were representative of all participants (N = 16). At T1, their final work rate was 120 ± 15 watts (vs 120 ± 10 watts for the whole group), their dyspnea and leg discomfort peaked at 2.8 ± 0.9 and 3.2 ± 0.6 (vs 3.3 ± 0.3 and 3.1 ± 0.3, respectively, for the whole group), and their estimated VO2 at the end of the test was 20.4 ± 5.6 ml/kg/min (vs 19.4 ± 1.4 for the whole group). Similar observations can be made for the 10 participants who performed the baseline and 2-month fitness assessments in comparison to all 16 participants who completed T1 assessments.
Exercise preferences
Details regarding exercise counseling and programming preferences are shown in Table 3. When asked about exercise counseling preferences, the majority of participants said they would prefer counseling during treatment (68.8%), in person (62.5%), at a cancer center (56.3%), from an exercise specialist affiliated with a cancer center (62.5%). When asked about exercise programming preferences, most participants would prefer to exercise during treatment (56.3%). They would prefer to be alone (43.8%) and unsupervised (71.4%), and the exercise to take place at home (42.9%), and at a moderate intensity (86.7%). The most preferred type of exercise was walking (56.3%) and recreational (100.0%) versus competitive exercise.
Physical activity levels
Table 4 presents participant PA at each time point. Checks for normality revealed one outlier that reported greater than 2.5 times the standard deviation for 2-month strenuous, moderate, and mild PA duration. The Winsorizing method was used to replace these values with the next highest score that was not an outlier [5]. Average total weekly PA minutes increased slightly at 2-month follow-up, then declined below baseline at 8-month follow-up. PA minutes at guidelines did not meet minimum recommendations (150 min of moderate vigorous) at any time point.
Quality of life
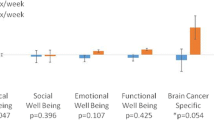
QOL was measured at all three time points, and results are summarized in Table 5. Overall, QOL scores using the FACT-G and FACT-Br declined from baseline to 2-month assessment (6.9 and 11.5 points, respectively) and continued to decline from 2 to 8 months post-baseline (3.9 and 1.5 points, respectively). The decline of 6.9 points on the FACT-G from baseline to 2 months represents a clinically significant decrease in QOL (minimally important is 4–7 points) [25]. Declines in all four subdomains (physical (PWB), emotional (EWB), social-family (SWB), and functional well-being (FWB)) were noted over time, representing a small to moderate effect size [16].
Correlations
Spearman’s Rho nonparametric correlations assessed relationships between QOL, fitness parameters, and PA. At baseline, a positive association existed between aerobic capacity and the FACT TOI (r = 0.619, p < 0.05). Two months after, there were positive associations between total weekly PA minutes and FACT-TOI (r = 0.653, p = 0.057), FACT-G (r = 0.711, p < 0.05), and FACT-Br scores (r = 0.722, p < 0.05). Similar associations after 2 months were found for PA minutes at recommended levels and FACT-TOI (r = 0.718, p < 0.05), FACT-G (r = 0.665, p = 0.05), and FACT-Br scores (r = 0.739, p = 0.05).
Discussion
Brain cancer remains a relatively understudied patient population in the cancer and exercise literature, particularly those diagnosed with HGG. The aims of the present investigation were to (1) determine the feasibility of recruiting HGG patients to an exercise study and performing fitness assessments in the HGG brain tumor patient population, (2) examine exercise counseling and programming preferences in this population, and (3) explore associations between fitness, PA levels, and QOL across the treatment trajectory. The main findings are that (i) recruitment rate was similar to previous studies in other cancer patient populations, (ii) assessing aerobic fitness with an incremental cycling test to VO2peak was not feasible in HGG patients, and (iii) most exercise counseling and programming preferences were similar to previous brain cancer patients; however, some differences were noted (iv) in general, PA and QOL declined over time, and QOL was associated with fitness at baseline and with PA at 2 months.
Feasibility
Recruitment of participants from the eligible patient population approached was 46%, which is similar to a previous study that reported a recruitment rate of 51% [11]. The brain tumor population, and in particular the HGG group, is hard to reach demographic. While this group is often younger and may appear less ill at the start of treatment compared to other tumor groups, the reality of their illness, prognosis, and the anticipated treatment is often overwhelming.
The attrition rate at each time point of this study was higher than expected. Two-month assessments were completed by 62.5% of patients, and only 18.8% completed 8-month assessments. This attrition was higher than a previous study of HGG patients that had 80% of participants complete 6-week assessment and 60% of participants complete 24-week assessments [14]. Our higher dropout rate could be attributed to the poorer health of our sample; in the current study, patients were all stage/grade IV, whereas to be included in the previous study, patients had to have a relatively good prognosis (i.e., estimated life expectancy of ≥6 months, and a Karnofsky performance status of ≥70%). In general, however, attrition rates in this population are expected to be lower than found in other tumor groups, due to very harsh treatments, numerous negative side effects, and the severity of disease progression. The accelerated disease progression in HGG patients highlights the necessity for any intervention work to be done early along the treatment trajectory. Unlike many other tumor groups, where the norm has been to focus on interventions well into cancer survivorship, work within the specific HGG brain tumor patient population should consider early intervention for the potential to use exercise to address treatment-related symptoms and toxicities.
Feasibility of aerobic fitness assessment
In contrast to previous studies in brain tumor and advanced cancer patients [10, 11, 14], the present study found that assessing aerobic fitness with an exercise test to volitional fatigue (VO2peak) in HGG patients was not feasible. Due to safety concerns indicated by the supervising physicians, the testing protocol had to be adapted to stop participants after VT2 rather than allowing them to reach volitional exhaustion. Most participants in the present study (70 to 75%) were able to reach VT2; thus, VT2 threshold could be used as an alternative protocol to assess aerobic fitness and to individualize exercise training intensity when maximal testing may be contraindicated.
Fitness outcomes
Similar to a previous study in brain tumor patients [14], the present study found that aerobic fitness parameters remained relatively stable across the treatment trajectory. Baseline (T1) cardiorespiratory fitness in this study was higher than levels in a previous study of pre-treatment HGG patients [14] (previous study reported VO2peak of 13.1 ± 4.2 ml/kg/min, and peak heart rate of 120 ± 22 bpm (M age 50.6 years), and the current study found that estimated VO2peak levels were 19.4 ± 1.4 ml/kg/min, and peak HR was 141 ± 4.5 bpm (M age 50.6 years)). The higher aerobic fitness level in the current study sample may be related to greater baseline PA minutes per week reported (155.8 ± 108.1 min per week vs 79 ± 121 min per week in the previous study).
Exercise preferences
Earlier work done in the area of exercise preferences by Jones et al. examined a cross-section of brain tumor patients, varying in brain tumor type and treatment status (on or off treatment) but did not separate exercise preferences for only HGG patients [13]. Our current findings both support previous findings and indicate some potential important population differences in the current sample of HGG patients. For counseling preferences, both the current sample and the previous study by Jones et al. found patients undergoing treatment preferred to receive exercise information in-person [13]. For programming preferences, however, the current study found that most participants preferred to exercise during treatment, which is in contrast to the Jones et al. study, which found that participants were more interested in participating in an exercise program following adjuvant treatment [13]. This contrast should be further explored in future intervention studies, given the potential for exercise to improve treatment completion and tolerance in HGG patients. Both the current study and Jones et al. found that most participants preferred to exercise at home, with walking as their choice of exercise modality [13]. However, Jones et al. found that most participants would prefer to exercise with their spouse or other family members, whereas most participants in this study preferred to exercise alone [13]. This contrast should be noted, since the questionnaire used in the present study did not include the option for family or spouse, which may be an important source of exercise support for cancer patients.
Physical activity
In our study, PA minutes at guidelines were below minimum recommended guidelines at all three time points, which is consistent to previous data reported for brain tumor patients [12]. Total weekly minutes of PA increased slightly from T1 to 2-month assessment, then declined below baseline at the 8-month assessment. A previous study of HGG patients also found weekly PA minutes increased from baseline to 6-week assessment, but then found that PA continued to increase at the 24-week assessment [14]. Both the current and previous studies used the Godin Leisure Time Questionnaire to measure PA, but these inconsistent results indicate the need for future studies to utilize objective measures of PA in order to get a more accurate picture of PA levels in HGG patients prior to, during, and after treatment. Physical activity has previously been found to be an independent predictor of survival in brain cancer (recurrent malignant glioma) patients for those reporting ≥9 MET-h per week [20], and future studies utilizing objective measure of PA are needed to determine whether these benefits can be accrued with the accumulation of longer duration and more frequent mild intensity physical activities, or if higher intensity PA is necessary in an HGG population.
Quality of life
In general, QOL parameters declined across the treatment trajectory, which was not surprising given the nature of treatment and disease progression. This trend was similar to the QOL changes in a previous study of brain tumor patients [14].
Correlations between fitness, PA, and QOL
Positive associations were found between PA and QOL and fitness and QOL. This is consistent with previous findings in HGG populations [11, 14]. Positive correlations between PA and QOL at the 2-month assessment have potential clinical relevance for recommending that HGG patients engage in activity throughout the course of treatment. Though causation for PA and fitness improving QOL cannot be determined based on univariate correlations, these associations also corroborate with (1) an exercise trial in brain cancer patients that found post-intervention improvement in overall well-being [1] and (2) case studies in two brain cancer patients that found QOL life was improved following 12 weeks of supervised exercise [17]. These findings indicate the need to further investigate the relationships among aerobic fitness, physical activity, and QOL in a large prospective study or randomized controlled trial.
Strengths and limitations
This study examining the role of fitness PA in brain tumor patients, particularly advanced HGG patients, contributes to a literature base where few studies exist. However, this study was not without limitations. Recruitment rate, though similar to previous studies, was low, attrition rates led to a small sample size at all three time points, and due to the cross-sectional nature of the study, causation cannot be determined.
Conclusions
This study found that recruiting a HGG patient population to an exercise study was feasible, but longitudinal studies may be challenging in this population due to harsh treatments, negative side effects, and disease progression impacting participant attrition. Although performing fitness assessments in patients with aggressive cancer such as HGG might help to develop appropriate and specific exercise recommendations, decisions whether to perform these assessments should be made on an individual basis. Specifically, maximal testing during treatment in HGG patients may not be warranted, and in patients with any aggressive tumor and/or poor prognosis, the risks of fitness testing may outweigh the benefits. The use of submaximal or functional (handgrip, 6-min walk, etc.) testing protocols should be considered as an alternative.
HGG patients undergoing treatment had similar exercise preferences to a larger sample of brain cancer patients, except that HGG patients were more likely to prefer an exercise program during (versus following) treatment. Understanding the exercise counseling and programming preferences of HGG patients are important for the development of exercise protocols during treatment. This study found that fitness and PA levels were low, but remained stable from baseline to 2 months after starting treatment, and that there were positive associations between cardiorespiratory fitness and PA levels with QOL across the treatment trajectory.
The findings from this study, taken together with results from the few other studies examining PA in HGG patients, suggest a positive relationship between PA with QOL. This underscores the need for larger trials to examine PA as a method to improve QOL during and following treatment. Future exercise studies in brain cancer patients should (1) account for HGG diagnoses when determining timing of the intervention (during vs after treatment), (2) explore the option of exercising with a spouse or family member, (3) examine the use of submaximal and functional measures of physical fitness, and (4) employ objective measures of PA to examine the association of PA with QOL.
References
Capozzi LC, Boldt KR, Easaw J, Bultz B, Culos-Reed SN (2015) Evaluating a 12-week exercise program for brain cancer patients Psychooncology
Chicco AJ, Schneider CM, Hayward R (2006) Exercise training attenuates acute doxorubicin-induced cardiac dysfunction. J Cardiovasc Pharmacol 47:182–189
Cormie P, Nowak AK, Chambers SK, Galvao DA, Newton RU (2015) The potential role of exercise in neuro-oncology. Front Oncol 5:85
Courneya KS, Segal RJ, Mackey JR, Gelmon K, Reid RD, Friedenreich CM, Ladha AB, Proulx C, Vallance JK, Lane K, Yasui Y, McKenzie DC (2007) Effects of aerobic and resistance exercise in breast cancer patients receiving adjuvant chemotherapy: a multicenter randomized controlled trial. J Clin Oncol 25:4396–4404
Field A (2013) Discovering statistics using IBM SPSS statistics. SAGE, Thousand Oaks, California
Godin G, Shephard RJ (1985) A simple method to assess exercise behavior in the community. Can J Appl Sport Sci 10:141–146
Holmes MD, Chen WY, Feskanich D, Kroenke CH, Colditz GA (2005) Physical activity and survival after breast cancer diagnosis. JAMA 293:2479–2486
Jacobs DR Jr, Ainsworth BE, Hartman TJ, Leon AS (1993) A simultaneous evaluation of 10 commonly used physical activity questionnaires. Med Sci Sports Exerc 25:81–91
Jones LW, Courneya KS (2002) Exercise counseling and programming preferences of cancer survivors. Cancer Pract 10:208–215
Jones LW, Eves ND, Mackey JR, Peddle CJ, Haykowsky M, Joy AA, Courneya KS, Tankel K, Spratlin J, Reiman T (2007) Safety and feasibility of cardiopulmonary exercise testing in patients with advanced cancer. Lung Cancer 55:225–232
Jones LW, Friedman AH, West MJ, Mabe SK, Fraser J, Kraus WE, Friedman HS, Tresch MI, Major N, Reardon DA (2010) Quantitative assessment of cardiorespiratory fitness, skeletal muscle function, and body composition in adults with primary malignant glioma. Cancer 116:695–704
Jones LW, Guill B, Keir ST, Carter BSK, Friedman HS, Bigner DD, Reardon DA (2006) Patterns of exercise across the cancer trajectory in brain tumor patients. Cancer 106:2224–2232
Jones LW, Guill B, Keir ST, Carter K, Friedman HS, Bigner DD, Reardon DA (2007) Exercise interest and preferences among patients diagnosed with primary brain cancer. Support Care Cancer 15:47–55
Jones LW, Mourtzakis M, Peters KB, Friedman AH, West MJ, Mabe SK, Kraus WE, Friedman HS, Reardon DA (2010) Changes in functional performance measures in adults undergoing chemoradiation for primary malignant glioma: a feasibility study. Oncologist 15:636–647
Jones LW, Watson D, Herndon JE 2nd, Eves ND, Haithcock BE, Loewen G, Kohman L (2010) Peak oxygen consumption and long-term all-cause mortality in nonsmall cell lung cancer. Cancer 116:4825–4832
King MT, Cella D, Osoba D, Stockler M, Eton D, Thompson J, Eisenstein A (2010) Meta-analysis provides evidence-based interpretation guidelines for the clinical significance of mean differences for the FACT-G, a cancer-specific quality of life questionnaire. Patient Relat Outcome Meas 1:119–126
Levin GT, Greenwood KM, Singh F, Tsoi D, Newton RU (2015) Exercise improves physical function and mental health of brain cancer survivors: two exploratory case studies integrative cancer therapies
Meyerhardt JA, Giovannucci EL, Holmes MD, Chan AT, Chan JA, Colditz GA, Fuchs CS (2006) Physical activity and survival after colorectal cancer diagnosis. J Clin Oncol 24:3527–3534
Peters KB, West MJ, Hornsby WE, Waner E, Coan AD, McSherry F, Herndon JE 2nd, Friedman HS, Desjardins A, Jones LW (2014) Impact of health-related quality of life and fatigue on survival of recurrent high-grade glioma patients. J Neuro-Oncol 120:499–506
Ruden E, Reardon DA, Coan AD, Herndon JE 2nd, Hornsby WE, West M, Fels DR, Desjardins A, Vredenburgh JJ, Waner E, Friedman AH, Friedman HS, Peters KB, Jones LW (2011) Exercise behavior, functional capacity, and survival in adults with malignant recurrent glioma. J Clin Oncol 29:2918–2923
Schmitz KH, Courneya KS, Matthews C, Demark-Wahnefried W, Galvao DA, Pinto BM, Irwin ML, Wolin KY, Segal RJ, Lucia A, Schneider CM, von Gruenigen VE, Schwartz AL, American College of Sports M (2010) American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc 42:1409–1426
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for R, Treatment of Cancer Brain T, Radiotherapy G, National Cancer Institute of Canada Clinical Trials G (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Thavarajah N, Bedard G, Zhang L, Cella D, Beaumont JL, Tsao M, Barnes E, Danjoux C, Sahgal A, Soliman H, Chow E (2014) Psychometric validation of the functional assessment of cancer therapy--brain (FACT-Br) for assessing quality of life in patients with brain metastases. Support Care Cancer 22:1017–1028
Weitzner MA, Meyers CA, Gelke CK, Byrne KS, Cella DF, Levin VA (1995) The functional assessment of cancer therapy (FACT) scale. Development of a brain subscale and revalidation of the general version (FACT-G) in patients with primary brain tumors. Cancer 75:1151–1161
Yost KJ, Eton DT (2005) Combining distribution- and anchor-based approaches to determine minimally important differences: the FACIT experience. Eval Health Prof 28:172–191
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Nicole Culos-Reed, S., Leach, H.J., Capozzi, L.C. et al. Exercise preferences and associations between fitness parameters, physical activity, and quality of life in high-grade glioma patients. Support Care Cancer 25, 1237–1246 (2017). https://doi.org/10.1007/s00520-016-3516-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-016-3516-4