Abstract
Wernicke’s encephalopathy (WE) is a thiamine deficiency-related condition, in which lesions are usually present in the periventricular and subcortical areas of the brain. However, lesions have also been found in atypical areas, such as the cerebral cortex. The present study summarizes the clinical outcomes and radiological features of WE with cortical impairment. We report two cases of cortical involvement in patients with WE, and review 22 similar cases from other reports. Among all 24 cases, 4 patients had a confirmed history of chronic daily alcohol abuse, and 19 of them had an identified causes of thiamine deficiency. 17 cases reported specific clinical information, among which 11 patients had symptoms of cortical impairment. 23 cases reported prognostic information at the end of treatment or at follow-up. The mortality rate was 26.1 % in our review. All patients had abnormal magnetic resonance imaging (MRI) signals or pathological findings in the bilateral cortex. Among patients with available MRI, 89.0 % had banding-like signs along the para-central sulcus. 13 cases underwent follow-up MRI examinations and 76.9 % displayed normal images. We suggest that WE with bilateral cortical involvement may have an acceptable prognosis, but that the mortality rate is higher than that among typical cases, especially if patients are not treated promptly and correctly. We identified the frontal and parietal lobes, especially around the central sulcus, to be the most susceptible areas, and suggest that the banding signs may be characteristic of WE. Persistent hyper-intensity on T2-weighted–fluid-attenuated inversion recovery, or gadolinium enhancement, may predict poor outcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The typical manifestations of Wernicke’s encephalopathy (WE) are subacute onset of confusion, ataxia and diplopia from ophthalmoplegia. This condition most commonly occurs in malnourished individuals, especially in alcoholics and post-surgery patients, due to acute or chronic thiamine deficiency. Prompt administration of intravenous or intramuscular thiamine can reverse the condition, but treatment delay or lack of treatment can lead to irreversible dementia or even death. The periventricular areas around the third ventricle, the fourth ventricle, and the mammillary bodies are most prone to lesions in WE. However, lesions have also been reported in atypical areas, such as the cerebral cortex, the substantia nigra, the red nuclei, and the corpus callosum. Patients who had cortical involvement were reported to have worse prognosis. In this study, we report two non-alcoholic cases of WE with cortical impairment who both recovered. We also review the literature on the atypical features of WE.
Patients and methods
We summarized the clinical, radiological, and prognostic characteristics of the two patients, who were diagnosed in 2013, as well as others from the literature with WE and cortical involvement. To review the literature, we searched the PubMed database with the term ‘Wernicke’s Encephalopathy’ plus the descriptive words ‘cortical’, ‘cortex’, ‘cortices’, ‘lobes’, ‘gyrus’, ‘sulci’, or ‘sulcus’. Patients with comorbidities that might affect cortical function were ruled out. The lists of references quoted in all included studies were also screened. Searches were carried out up to May 31, 2016. Details of age, gender, history, presence of alcohol abuse or causes of thiamine deficiency, clinical manifestations such as seizures and pyramidal signs, time from onset to treatment, prognosis, radiological features such as magnetic resonance imaging (MRI) sequence, distribution of cortical lesions at the onset, and changes in follow-up MRI were collected from every case report.
Result
Case 1
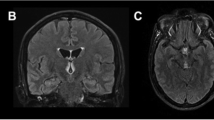
A 57-year-old man was admitted due to progressive confusion accompanied by vomiting, hiccups, and hearing loss in the previous three days. He had been diagnosed with suspected choledocholithiasis and treated with gall bladder excision and a choledocholithotomy 22 days previously. He had no history of alcohol abuse. He had lost 10 kg in weight since the surgery. Positive findings by neurological examination were a confused state, bilateral slow pupillary light reflex, horizontal nystagmus, and absence of deep tendon reflex in all extremities. The Chaddock reflex on the left side was indifferent. An MRI scan showed a hyper-intense signal on T2-weighted images and fluid-attenuated inversion recovery (FLAIR) in the bilateral thalamus, periaqueductal gray matter, and the structures around the third and the fourth ventricles. In addition, signals were observed along the central sulcus of both hemispheres (Fig. 1). Gadolinium enhancement was normal. According to the surgical history, clinical manifestations, and characteristic MRI findings, a diagnosis of WE was made and thiamine therapy commenced. The initial response to thiamine was good, with resolution of all symptoms. A follow-up MRI scan performed 2 weeks after treatment also showed general improvement of signal alterations (Fig. 2).
Repeated FLAIR sequence performed 2 weeks after thiamine treatment in Case 1. Compared with Fig. 1, the high-intensity lesions have almost disappeared; simultaneously, the clinical manifestations markedly improved
Case 2
A 45-year-old woman underwent a pancreatico-duodenectomy because of pancreatic carcinoma. After the surgery, she received total parenteral nutrition for about eight weeks. She was transported to the emergency department of our hospital after experiencing vomiting, excessive daytime sleep, and a confused state for 15 days. The patient was uncooperative during the neurological examination. Positive signs were ophthalmoplegia, nystagmus, and neck stiffness. Limbs could move just up from the bed with pain and deep tendon reflexes were present. Bilateral plantar reflexes were normal. Laboratory examinations revealed normocytic anemia (hemoglobin 85 g/L) and a slight increase in alanine aminotransferase (52.3 U/L). Urea, creatinine, lipase and amylase levels, and other tests were normal. The first brain MRI scan, performed on the day of symptom onset, revealed lesions in the dorsal medulla oblongata, the pons tegmentum, bilateral ventral midbrain, bilateral dorsal thalamus, mammillary bodies, and bilateral central sulcus on a T2-FLAIR sequence (Fig. 3). There were no abnormal signals in the cerebral cortex on the first diffusion-weighted imaging (DWI) sequence scan (Fig. 4a, b). A follow-up MRI scan 1 week later showed intense signals in the bilateral central sulcus on a DWI sequence, while lesions in the dorsal thalamus were slightly improved (Fig. 4c, d). Treatment with thiamine commenced after diagnosis of WE. The time between the onset of clinical manifestations and the initiation of thiamine therapy was 17 days. Three days later, the patient was cooperative with a physical examination and could partially answer questions. At the time of discharge, most symptoms had improved remarkably, with the exception of excessive daytime sleep and slight irritability. A follow-up brain MRI performed 17 months later revealed an improvement in the majority of white matter lesions (Fig. 4e, f). A telephone follow-up was performed 20 months later and the patient’s neurological symptoms had partially recovered.
FLAIR sequence performed at onset in Case 2. Hyperintense lesions could be seen in a the bilateral cortex around the central sulcus, b the left frontal lobe, c the bilateral dorsal thalamus, d the bilateral ventral midbrain, superior colliculus, and mammillary bodies, e the pontine tegmentum, and f the dorsal medulla oblongata
MR-DWI scan of Case 2. a and b were produced at the onset of the disease. The cortex was normal while the bilateral thalamus showed higher signals. T2-FLAIR had already showed abnormalities by this point (Fig. 3 a). c and d were produced one week later. Signals in the cortex had become abnormal. e and f were produced 17 months later. The lesions had disappeared in this follow-up MR-DWI
Literature review
We identified an additional 22 patients from 17 reports in the PubMed database (14 in English (Alberti et al. 2012; Cui et al. 2012; D’Aprile et al. 2000; Doss et al. 2003; Fei et al. 2008; Gascon-Bayarri et al. 2011; Liu et al. 2006; Luigetti et al. 2009; Machado et al. 2010; Nolli et al. 2005; Pereira et al. 2011; Sakurai et al. 2009; Yamashita and Yamamoto 1995; Zuccoli et al. 2009) with full text, 3 in other languages (Blanco-Munez et al. 2006; Kinoshita et al. 2001; Kuhn et al. 2004) with Abstracts only). The clinical characteristics of all 24 cases are summarized in Table 1. The patients’ age ranged from 13 to 81 years, with a median age of 52 years. There were 14 men (58.3 %, median age 55 years) and 10 women (41.7 %, median age 46 years). 4 patients (4/24, 16.6 %) had confirmed chronic alcohol abuse, while the other cases denied a history of abuse or it was not specifically mentioned. 19 patients (19/24, 79.2 %) had identified causes of thiamine deficiency. Parenteral nutrition, persistent vomiting, anorexia, or food refusal were the most common causes, which lasted from five days to six months (median time of 4 weeks). 17 cases mentioned specific clinical information, of which 11 patients (11/17, 64.7 %) had symptoms of cortical impairment such as seizures, limb weakness, or pyramidal signs. 23 cases reported the prognosis at the end of treatment or at follow-up visits. Of these, 7 had poor outcome (7/23, 30.4 %), including 6 deaths (mortality 26.1 %) and one permanent vegetative state. Of the deaths, 4 patients deteriorated rapidly and died within two months; and two patients showed slight improvement after treatment but died within six months to two years because of other causes such as respiratory infection. 13 patients (13/23, 56.5 %) had long-term neurological sequelae and seven of these had cognitive disorders. Only 3 patients (3/23, 13.1 %) recovered completely.
Radiological findings: All patients had abnormal findings in the bilateral cortex. Details of MRI examinations were reported for 20 patients; 4 mentioned impairment in the cortical regions. All (20/20) had lesions in the frontal and parietal lobes. One patient had an additional impairment in the temporo-occipital cortical gyri (Liu et al. 2006). 18 reports showed MRI images in their article. It is noteworthy that 89 % of patients (16/18) showed a specific band-like signal along the para-central sulcus. One case mentioned dynamic progression over five days. 13 patients underwent a follow-up MRI examination, and of those, 10 (10/13, 76.9 %) had almost normal MRI findings. 2 patients had persistent cortical lesions and 1 case turned to cortical laminar necrosis. The details are shown in Table 2.
Discussion
The classic clinical triad of symptoms observed in WE are a reduced level of consciousness, ataxia, and oculomotor disorders. Impairment of the cortical gyrus is rarely reported and usually results in a worse prognosis or sequelae. In this study, we reported two cases of WE with cortical lesions, both of whom recovered; we also reviewed the data from previous studies (Alberti et al. 2012; Cui et al. 2012; D’Aprile et al. 2000; Doss et al. 2003; Fei et al. 2008; Gascon-Bayarri et al. 2011; Liu et al. 2006; Luigetti et al. 2009; Machado et al. 2010; Nolli et al. 2005; Pereira et al. 2011; Sakurai et al. 2009; Yamashita and Yamamoto 1995; Zuccoli et al. 2009; Blanco-Munez et al. 2006; Kinoshita et al. 2001; Kuhn et al. 2004; Zhong et al. 2005).
The mortality rate of typical WE is approximately 17.0 % (Victor et al. 1971), although it is much higher (6/23, 26.1 %) in WE with cortical impairment, demonstrating that cortical involvement can be an indicator of poor prognosis. However, our two cases, who presented with limb weakness and conscious disturbances, had relatively good outcomes at follow-up. The main explanations are likely to be reversible lesions of the cortex and timely treatment with thiamine. Previous studies have suggested that reversible cytotoxic edema is characteristic of WE and would resolve after thiamine treatment (Zuccoli and Pipitone 2009). A similar process occurs in cells in an ischemic penumbra (Chu et al. 2002). Cortical involvement is not an uncommon pathologic change in post-mortem examination in WE. The histological changes in the motor gyrus are identical to those in other typical WE lesions such as in the mammillary body (Blanco-Munez et al. 2006). Furthermore, cortical laminar necrosis can be found and ascribed to thiamine deficiency (Yamashita and Yamamoto 1995). Therefore, the clinical manifestations and radiological changes in the cortex may resolve due to elimination of cytotoxic edema, as in other areas.
It is clear that early treatment with thiamine supplements will lead to better clinical outcomes. In our study, Case 1 started treatment within 3 days and the response to thiamine was excellent, resulting in significant improvement by the time of discharge and full recovery at follow-up. Case 2 received intravenous thiamine 17 days after onset and focal lesions partially improved. However, she still had mental and emotional dysfunction after 20 months. In our literature review, patients who received intravenous thiamine within a few days tended to recover rapidly with good outcomes, while the others whose treatment was delayed had more severe sequelae. The importance of prompt treatment is further supported by the fact that two cases who did not receive thiamine treatment died promptly (Liu et al. 2006; Blanco-Munez et al. 2006). We conclude that if the patient receives proper treatment at an early stage, the cortical impairment can be reversed, leading to better prognosis.
Cortical impairment with symmetric hyper-intense signals on MRI scans is rare in acute WE. Lesions usually present as a ‘banding sign’ extending along the sides of the central gyrus or as focal swelling of the fronto-parietal cortices. It is worth noting that 89 % of the cases demonstrated this specific signal and that these patients were more likely to have seizures or motor dysfunction. Usually when the lesions disappeared, most clinical symptoms resolved simultaneously. However, patients with extensive cortical impairment (Pereira et al. 2011) and persistent gadolinium enhancement (Nolli et al. 2005) may have poor outcomes. These findings suggest that these distinctive MRI signals may become a diagnostic marker and a prognostic indicator for WE.
Previous studies have demonstrated a dynamic development from vasogenic edema to cytotoxic edema in WE (Liu et al. 2006). In Case 2, high cortical signal intensity in a DWI sequence was not present at the onset of the condition (Fig. 4a), while T2-FLAIR had already shown abnormal lesions. However, abnormalities in the DWI sequence appeared 1 week later, supporting the hypothesis of dynamic progression. The absence of thiamine reduces the efficiency of glucose metabolism, resulting in cerebral lactic acidosis and subsequent cytotoxic edema. On the other hand, dysfunction of the blood-brain barrier induced by thiamine deficiency can result in vasogenic edema (Manzo et al. 2014). Together these may explain the co-existence of concurrent cytotoxic and vasogenic edema, although the precise underlying mechanisms remain unclear.
This study has some limitations. First, although the thiamine level is crucial for WE diagnosis, it can remain within the normal range in some cases (Gascon-Bayarri et al. 2011). We did not determine thiamine levels, but the clinical manifestations, MRI findings, and excellent therapeutic responses supported our diagnoses. Secondly, we could not obtain full information from the three non-English reports for which only Abstracts were available. Moreover, the lack of certain clinical details in some reports may affect the data integrity. Finally, apparent diffusion coefficient sequences were not performed, and dynamic progression from vasogenic to cytotoxic edema was not observed. More evidence regarding these issues.
In summary, cortical impairment in WE is not a common manifestation but leads to a higher mortality rate. In a literature review, the frontal and the parietal lobes were the most susceptible areas, especially the central sulcus. Symmetrical banding signs may be a distinctive characteristic in the early stages of the condition. Although WE with cortical lesions leads to worse outcomes, the condition can be reversed if patients receive timely treatment. Persistent hyper-intensity on T2-FLAIR or gadolinium enhancement predicts poor prognosis.
References
Alberti N, Bocquet J, Molinier S et al (2012) Wernicke’s encephalopathy with atypical cortical damage. Diagn Interv Imaging 93(10):804–807
Blanco-Munez O, Suarez-Gauthier A, Martin-Garcia H et al (2006) Unusual cortical compromise in a case of Wernicke’s encephalopathy. Rev Neurol 42(10):596–599
Chu K, Kang DW, Kim HJ et al (2002) Diffusion-weighted imaging abnormalities in Wernicke encephalopathy: reversible cytotoxic edema? Arch Neurol 59(1):123–127
Cui HW, Zhang BA, Peng T et al (2012) Wernicke’s encephalopathy in a patient with acute pancreatitis: unusual cortical involvement and marvelous prognosis. Neurol Sci 33(3):615–618
D’Aprile P, Tarantino A, Santoro N et al (2000) Wernicke’s encephalopathy induced by total parenteral nutrition in patient with acute leukaemia: unusual involvement of caudate nuclei and cerebral cortex on MRI. Neuroradiology 42(10):781–783
Doss A, Mahad D, Romanowski CA (2003) Wernicke encephalopathy: unusual findings in nonalcoholic patients. J Comput Assist Tomogr 27(2):235–240
Fei GQ, Zhong C, Jin L et al (2008) Clinical characteristics and MR imaging features of nonalcoholic Wernicke encephalopathy. AJNR Am J Neuroradiol 29(1):164–169
Gascon-Bayarri J, Campdelacreu J, Garcia-Carreira MC et al (2011) Wernicke’s encephalopathy in non-alcoholic patients: a series of 8 cases. Neurologia 26(9):540–547
Kinoshita Y, Inoue Y, Tsuru E et al (2001) Unusual MR findings of Wernicke encephalopathy with cortical involvement. Brain Nerve 53(1):65–68
Kuhn J, Friedel V, Knitelius HO et al (2004) Iatrogenic Wernicke-Korsakow syndrome with unusual neurological deficits and MRI lesions. Nervenarzt 75(8):795–800
Liu YT, Fuh JL, Lirng JF et al (2006) Correlation of magnetic resonance images with neuropathology in acute Wernicke’s encephalopathy. Clin Neurol Neurosurg 108(7):682–687
Luigetti M, De Paulis S, Spinelli P et al (2009) Teaching NeuroImages: the full-blown neuroimaging of Wernicke encephalopathy. Neurology 72(22):e115
Machado A, Ribeiro M, Soares-Fernandes J et al (2010) Cortical linear lesions in Wernicke’s encephalopathy: can diffusion-weighted imaging herald prognostic information? J Neuropsychiatr Clin Neurosci 22(1):123–124
Manzo G, De Gennaro A, Cozzolino A et al (2014) MR imaging findings in alcoholic and nonalcoholic acute Wernicke’s encephalopathy: a review. Biomed Res Int 2014:503596
Nolli M, Barbieri A, Pinna C et al (2005) Wernicke’s encephalopathy in a malnourished surgical patient: clinical features and magnetic resonance imaging. Acta Anaesthesiol Scand 49(10):1566–1570
Pereira DB, Pereira ML, Gasparetto EL (2011) Nonalcoholic Wernicke encephalopathy with extensive cortical involvement: cortical laminar necrosis and hemorrhage demonstrated with susceptibility-weighted MR phase images. AJNR Am J Neuroradiol 32(2):E37–E38
Sakurai K, Sasaki S, Hara M et al (2009) Wernicke’s encephalopathy with cortical abnormalities: clinicoradiological features: report of 3 new cases and review of the literature. Eur Neurol 62(5):274–280
Victor M, Adams RD, Collins GH (1971) The Wernicke-Korsakoff syndrome. A clinical and pathological study of 245 patients, 82 with post-mortem examinations. Jama thes 7(3):389–389
Yamashita M, Yamamoto T (1995) Wernicke encephalopathy with symmetric pericentral involvement: MR findings. J Comput Assist Tomogr 19(2):306–308
Zhong C, Jin L, Fei G (2005) MR imaging of nonalcoholic Wernicke encephalopathy: a follow-up study. AJNR Am J Neuroradiol 26(9):2301–2305
Zuccoli G, Pipitone N (2009) Neuroimaging findings in acute Wernicke’s encephalopathy: review of the literature. AJR Am J Roentgenol 192(2):501–508
Zuccoli G, Santa CD, Bertolini M et al (2009) MR imaging findings in 56 patients with Wernicke encephalopathy: nonalcoholics may differ from alcoholics. AJNR Am J Neuroradiol 30(1):171–176
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Informed consent
Informed consent was obtained from the two Cases included in the study.
Additional information
Lei Wu and Di Jin contributed to the work equally.
Rights and permissions
About this article
Cite this article
Wu, L., Jin, D., Sun, X. et al. Cortical damage in Wernicke’s encephalopathy with good prognosis: a report of two cases and literature review. Metab Brain Dis 32, 377–384 (2017). https://doi.org/10.1007/s11011-016-9920-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9920-0