Abstract
It has been shown that the circular RNA (circRNA) circPTK2 modulates many types of diseases. However, the possible functions as well as the molecular mechanisms of circPTK2 in preeclampsia (PE) and their effects on trophoblast are unknown. Herein, we obtained the placental tissues from 20 pregnant women with PE who delivered in the Yueyang Maternal Child Medicine Health Hospital between 2019 and 2021 to serve as the PE group, and a normal group was composed of 20 healthy pregnant women with normal prenatal examinations. The circPTK2 level was significantly reduced in tissues from the PE group. The expression and localization of circPTK2 were verified using RT-qPCR. CircPTK2 silencing inhibited HTR-8/SVneo growth and migration in vitro. To investigate the underlying mechanism of circPTK2 in PE progression, dual-luciferase reporter assays were conducted. It was found that circPTK2 and WNT7B could bind directly to miR-619, and that circPTK2 affected WNT7B expression by sponging miR-619. To conclude, this study identified the functions and mechanisms of the circPTK2/miR-619/WNT7B axis in PE progression. In this way, circPTK2 has the potential to be used both in diagnostic and therapeutic settings for PE.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mother and infant health and life safety are seriously compromised by preeclampsia (PE), which is a new type of idiopathic hypertension syndrome occurring after 20 weeks of pregnancy [1]. There are approximately 50 K–60 K stillbirths and premature infant deaths every year in developing countries with its incidence rate ranging from 4%–18% [2]. The PE, which occurs before 34 weeks, is called early-onset PE (EOPE). Researchers have found that early-onset PE has a higher risk of adverse health effects for mothers and babies than later-onset PE in clinical studies [3]. A major factor in the progress of EOPE is shallow placental implantation caused by decreased trophoblast invasion [3]. The main focus of this study was to examine the pathogenesis of EOPE [4].
Circular RNAs (circRNAs) are noncoding RNA molecules found in living cells with cyclic structure [5]. It has been demonstrated that circular RNAs (circRNAs) play a crucial role in a number of biological processes [5, 6]. The function of CircRNAs is achieved through many mechanisms, including interactions with proteins, microRNAs (miRNAs), and mRNAs, as well as by encoding proteins [7]. There is evidence that circRNA disorders in placental tissues contribute to PE pathogenesis [8]. Based on RNA sequencing, Ou et al. identified 49 aberrantly expressed circRNAs in PE and 3 of them were confirmed to be upregulated by qPCR [9]. It was found that hsa_circ_TNRC18 inhibited the migration of trophoblast cells and EMT by regulating miR-762/Grhl2 axis in preeclampsia 15 [10]. In HTR8-S/Vneo trophoblast cells, silencing of hsa_circ_0088227 impairs the trophoblast cells’ ability to proliferate and invasive by modulating STAT3 [11]. Besides, a number of studies have revealed that plasma circRNAs can serve as early biomarkers for PE. Jiang et al. developed a combined model of PAPP-A and two related circRNA to detect PE before the onset symptoms, which exhibited satisfactory predictive ability [12]. However, it is unclear how circPTK2 (hsa_circ_0005273) functions and what mechanisms are involved in EOPE.
In this study, a distinct downregulation of circPTK2 in EOPE tissues was detected, as well as HTR-8/SVneo cells. And the overexpression of circPTK2 significantly reduced PE growth along with migration. Then, using bioinformatics, miR-619 was predicted as a latent target of circPTK2. Subsequently dual-luciferase reporter assays and WB confirmed that circPTK2 stimulated proliferation and invasion of EOPE by circPTK2/miR-619/WNT7B axis. It has been demonstrated that miR-619 inhibits the growth of tumors in many different types of cancer, including pancreatic cancer [13] and oral squamous cell carcinoma [14]. The role and mechanism of miR-619 in PE have not yet been studied. In addition, WNT7B functions as an oncogene in several cancers, such as colorectal cancer [15] and osteosarcoma [16] while the role of which remains unsolved in EOPE. Our work revealed that EOPE progression is governed by the circularPTK2/miR-619/WNT7B axis, and downregulation of circPTK2 results in decreased trophoblast invasion which contributes to shallow placental implantation, a major factor in the progress of EOPE. Out study may improve EOPE prognosis in clinic.
Materials and methods
Clinical samples
In our study, we selected 20 cases of EOPE puerpera with 20 healthy puerpera as the normal group (n = 20) following the diagnosis criteria endorsed by the International Society for the Study of Hypertension in Pregnancy (ISSHP) (Table 1) [17]. During pregnancy, all puerpera received perinatal care and were delivered via cesarean section in the Yueyang Maternal Child Medicine Health Hospital. Following placenta delivery, placental tissues were collected within 15 min and RNA later is immediately used for storage (− 80 ℃). The Medical Ethics Committee of the Yueyang Maternal Child Medicine Health Hospital approved this study in accordance with the Declaration of Helsinki. Every patient who took part in the research provided informed consent.
Cell lines culture and transfection
HTR-8/SVneo trophoblasts were obtained from the ATCC for this experiment. RPMI-1640 medium (US Gibco) containing 10% fetal bovine serum (US Corning) was used for cell culture at 37 °C and 5% CO2. Cell lines were authenticated before being used through DNA fingerprinting. Transfection was conducted using logarithmic cells inoculated into a 6-well plate (2 × 105 cells/well) with Lipofectamine 3000 system (US Invitrogen). siRNA targeting circPTK2, miR-619 mimics and inhibitors were provided by GeneCopoeia (US). Experiments following transfection were conducted 48 h later.
Quantitative reverse transcription PCR
Total RNA was isolated from placental tissues and cells with TRIzol (US Invitrogen) according to the manufacturer's instructions. PARIS™ Kit (Invitrogen, US) was used to purify cytoplasmic and nuclear RNA. By utilizing a reverse transcription (Takara, Japan), total RNA was reverse transcribed into cDNA, and then, qRT-PCR was performed using SYBR premix ex taq kit (Takara) and the CFX96 Real-Time PCR Detection System. circPTK2, circPTK2, Forward (5’-3’), CGTCTAATCCGACAGCAACA, Reverse (5’-3’), GGGCTGGGATAAAATCCTTC; WNT7B, Forward (5’-3’), CACAGAAACTTTCGCAAGTGG, Reverse (5’-3’), GTACTGGCACTCGTTGATGC; 18S, Forward (5’-3’), TTAATTCCGATAACGAACGAGA, Reverse (5’-3’), CGCTGAGCCAGTCAGTGTAG; GAPDH, Forward (5’-3’), GGAGCGAGATCCCTCCAAAAT, Reverse (5’-3’), GGCTGTTGTCATACTTCTCATGG; β-actin, Forward (5’-3’), AGCGAGCATCCCCCAAAGTT, Reverse (5’-3’), GGGCACGAAGGCTCATCATT; si-NC, UUCUCCGAACGUGUCACGUTT; si-circPTK2, GAAAGATTTCTGCCCAGCAGA.
Cell counting kit-8 (CCK-8) assay
Briefly, inoculating 4000 cells into each well of a 96-well plate 48 h after transfection and incubating them for 24 h. To each well being tested, 10µL of CCK-8 solution was added. A microtiter plate reader (Bio-Tek, USA) was used to measure absorbance at 450 nM after 2 h of incubation at 37 ℃.
Transwell assay
HTR-8/SVneo cells were diluted to a serum-free cellular suspension of 2 × 105/mL after transfection. 200ul of cellular suspension was added to the upper chambers, while the lower ones were filled with RPMI-1640 medium (US Gibco) containing 20% FBS. After 36 h incubation at 37 ℃, for crystal violet staining of the cells, the chamber was taken out, washed with PBS twice, and the cells were fixed in methanol for 20 min. A light microscope was used to observe and count cells in random fields.
Glucose and lactate assay
The glucose assay was carried out as described by Glucose Assay Kit (Abcam, USA, ab102517). After 48 h of transfection, HTR-8/SVneo cells were harvested for lactate production measurements using L-Lactate Assay kit (Abcam, USA, ab65330).
Luciferase reporter assay
We constructed luciferase reporter vectors containing dormant miR-619 binding sites in WNT7B's 3'UTR and circPTK2’s 3’UTR. The Fast Site-Directed Mutagenesis Kit (TIANGEN, China) was used to mutate the corresponding sequence as a control. Using the dual-luciferase enzyme reporter assay system kit (Promega, USA) as instructed by the manufacturer, we evaluated the relative activity of the luciferase reporter.
Western blot analysis
Total proteins were separated through RIPA lysis containing PMSF. Then, we used the SDS-PAGE system for electrophoresis of proteins and then, transferred the proteins onto a PVDF membrane at low temperatures. Following blocking with 5% skim milk for 1 h, anti-WNT7B(Abcam, USA; 1:1200) antibody and anti-GAPDH(Affinity, USA; 1:15,000) antibody were inoculated overnight at 4 ℃. After 1 h at RT, membranes were inoculated with secondary antibody (CST, USA; 1:3000). Detection was carried out using chemiluminescence.
Statistical analysis
The statistical analysis was conducted using SPSS 25.0 software (SPSS, USA). The data are presented as mean ± S.D. Student t-tests were used when comparing groups to determine their significance. Statistical significance was indicated by P < 0.05.
Results
circPTK2 is downregulated in PE
In several diseases, CircPTK2 is thought to be involved in the progression. However, CircPTK2’s function in PE remains unclear. The circPTK2 levels were detected using qRT-PCR in placental tissues from PE and NC groups. Compared to the normal group, the PE group's expression level of circPTK2 was significantly lower (Fig. 1A). Since the expression of circPTK2 remarkably decreases in the PE group, we designed a siRNA to knock down its expression in HTR8/SVneo cells to investigate its role in PE. By using qRT-PCR in HTR8/SVneo cells, si-circPTK2 knockdown was confirmed (Fig. 1B).
Silencing of circPTK2 inhibits HTR8/SVneo cells proliferation, invasion and glucose metabolism
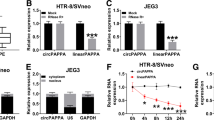
We conducted CCK8 and transwell assays to detect the function of circPTK2 in PE. In the CCK-8 assays, inhibition of circPTK2 remarkably inhibited HTR8/SVneo cell proliferation ability (Fig. 2A). Transwell assays revealed that HTR-8/SVneo cells had significantly fewer invasive cells than NC cells after transfection of si-circPTk2 (Fig. 2B). A significant reduction in glucose uptake and lactate production was observed when circPTK2 was knocked down (Fig. 2C). According to these findings, inhibiting circPTK2 dampens the proliferation, invasion and glucose metabolism of HTR8/SVneo cells.
Silence of circPTK2 inhibits HTR8/SVneo cells proliferation, invasion and glucose metabolism. A After transfection, detect the absorbance at 450 mm by CCK8 assay at 24, 48, and 72 h separately. B Transwell assay was performed on HTR8/SVneo cells to assess its invasive capabilities. C The glucose uptake and lactate production of HTR8/SVneo cells were detected after transfection
circPTK2 acts as a sponge of miR-619 in PE
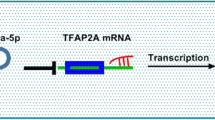
We extracted the cytoplasmic and nuclear RNA to detect the subcellular localization of circPTK2. Based on the results (Fig. 3A), circPTK2 is predominately present in the cytoplasm, implying that it could cross-talk with miRNAs, which are also predominant in the cytoplasm. Circular RNA Interactome was used to predict the probable cross-talk between circPTK2 and miRNAs in order to assess its role in modulating PE progression. As one of the candidates, MiR-619 was predicted to bind to circPTK2 (Fig. 3B). The cross-talking between circPTK2 and miR-619 was further confirmed using a dual-luciferase reporter assay. Significantly lower fluorescence intensities were observed in cells transfected with circPTK2-wt and miR-619 mimics, while transfection with circPTK2-mut did not result in a change in fluorescence intensity (Fig. 3C). According to these findings, circPTK2 acts as a sponge of miR-619. Therefore, we examined the potential association between miR-619 and circPTK2 expression using the placental tissues described above, which showed that miR-619 was notably higher in PE group compared with NC group (Fig. 3D).
circPTK2 acts as a sponge of miR-619 in PE. A The subcellular localization of circPTK2 was detected by qRT-PCR. B MiR-619 binding sequence on the circPTK2 was predicted using CircInteractome. C Dual-luciferase reporter assay displayed the luciferase activity of HTR8/SVneo cells co-transfection with wt/mut reporter vector and miR-619/miR-mimics. D The relative expression of miR-619 in the preeclampsia (PE) and normal group (n = 20)
circPTK2 inhibits PE progression through miR-619/WNT7B axis
We adopted TargetScan to screen miR-619 downstream candidate target gene. WNT7B, a signal protein essential for developmental processes like placental formation, was identified as a target gene of miR-619 (Fig. 4A). Transfecting miR-619 mimics into HTR8/SVneo cells led to a significant reduction in WNT7B mRNA expression, while the effect of which can be reversed by adding LNA-miR-619 (Fig. 4B). With a dual-luciferase reporter assay, the combination of miR-619 and WNT7B was demonstrated in HTR8/SVneo cells. Significantly lower fluorescence intensities were observed in cells transfected with WNT7B-wt and miR-619 mimics, while transfection with WNT7B-mut did not result in a change in fluorescence intensity (Fig. 4C). Further verification of the binding of circPTK2, miR-619, and WNT7B was accomplished with Ago2-RIP detection in HTR8/SVneo cells. The RIP experiment demonstrated that CircPTK2, WNT7B, and miR-619 were all enriched in Ago2 complexes. Furthermore, transfection of si-circPTK2 significantly decreased the enrichment of circPTK2 in the Ago2 complex, while increasing WNT7B enrichment (Fig. 4D). This led us to investigate WNT7B expression in HTR8/SVneo cells transfected with si-circPTK2 and discovered it was decreased (Fig. 4E).
circPTK2 inhibits PE progression through miR-619/WNT7B axis. A MiR-619 binding sequence on the WNT7B 3’UTR was predicted using TargetScan. B qRT-PCR was used to detect the expression level of WNT7B after transfection. C Dual-luciferase reporter assay displayed the luciferase activity of HTR8/SVneo cells co-transfection with wt/mut reporter vector and miR-619/miR-mimics. D Anti-Argonaute 2 (Ago2) complex RNA immunoprecipitation (RIP) enriched for circPTK2, miR-619, and WNT7B (left); Using Ago2-RIP, relative enrichment of WNT7B and circPLK2 was detected. E WB was used to detect the expression level of WNT7B after transfection
Discussion
EOPE, a placental disease, mainly results from insufficient infiltration of extravillous trophoblasts, maternal placenta abnormally construction and recasting of vessels [18]. As a result, the mother–fetal interface is ischemic and hypoxic, triggering oxidative stress and endoplasmic reticulum stress, which eventually affects fetal blood supply [1]. The study of early-onset PE has gradually extended to the non-coding RNA family, and circRNA plays a key role [19]. In recent years, CircRNAs have emerged as powerful modulators of many biological processes, including PE onset and progression [8, 20].
In this study, a notable difference was observed between the PE group and the NC group, when it came to expression levels of circPTK2. This result showed that circPTK2 appeared to play a role in PE onset, the role of which in cancer has been extensively studied and the results of these studies are somewhat contradictory. Yang et al. proved that circPTK2 exerted an oncogene function in colorectal cancer to promote tumor growth and metastasis [21]. However, according to a study in gastric cancer, circPTK2 significantly dampened gastric cancer cells’ ability to proliferation and invasion [21]. Although there is some contradiction between the role of circPTK2 in different cancers, it has yet to be reported on its involvement in PE. Our results are consistent with the former: inhibition of circPTK2 inhibits HTR8/SVneo cells proliferation and invasion. Although the function of promoting cell proliferation and invasion is the same, it plays a pro-cancer role in colorectal cancer while inhibits disease progression in PE. Besides, we found that circPTK2 promotes glucose uptake and lactate production, the underlined mechanism awaits to be elucidated.
In order to figure out circPTK2’s molecular mechanism in PE, we conducted a series of assays. ceRNA mechanisms are becoming more well understood in recent years, with circRNAs, lncRNAs, and pseudogenes being found to sponge miRNAs to their targets [22]. Our research also starts from the mechanism of ceRNA; upon further bioinformatic analysis, miR-619 docking site has been identified in circPTK2, the investigated circRNA. Following that, cellular assay was conducted to uncover the competitive nature of miR-619 and circPTK2. Research has demonstrated the proliferation suppressor properties of miR-619 in several diseases. Zhou et al. found that lncRNA PVT1 accelerates pancreatic cancer resistance to gemcitabine by sponging miR-619 [13]. Besides, miR-619 is also proved to inhibited OSCC cells proliferative and invasive abilities [14]. circ-FAT1 binds to miR-619 to elevate FOSL2 expression, which accelerates the progression of colorectal cancer [23]. In addition, by competing miR-619 binding sites and upregulating HDAC3, lncRNA SBF2AS1 promotes CRC proliferation and invasion [24]. We exhibited that miR-619 was notably higher in PE group compared with NC group, which was just the opposite of circPTK2. miR-619 binding sites were found in circPTK2 sequences, and dual-luciferase reporter assays along with RIP assays demonstrated that the circPTK2 is capable of directly absorbing miR-619.
Herein, TargetScan was used to identify miR-619 response regions within WNT7B mRNA and circPTK2. Then, we used a dual-luciferase reporter assay in addition to a RIP assay to verify their binding. Lastly, we inhibited circKIF4A to reduce STAT3 expression. According to these findings, circPTK2 sponging miR-619 regulates WNT7B expression. WNT pathways play an essential role in placenta and trophoblast development, the proliferation of trophoblast progenitors and stem cells depends on WNT [25]. Sonderegger et al. claimed that the decrease in WNT7b expression from early pregnancy to term suggests its role in trophoblasts [26]. The implantation process may be affected by WNT7b which is induced by estrogen [27]. Placental development can be controlled sequentially via WNT signaling components in mice [28]. Human homologues are likely to contribute to placenta formation and control essential functions, including trophoblast proliferation during the first trimester of pregnancy. For example, knock-out of WNT7B resulted in defects in chorioallantoic fusion [29]. These results are consistent with our experimental results, and circPTK2 regulates PE through modulating WNT7B expression. In addition to their potential role as biomarkers for diagnosis, circRNAs have potential as therapeutic targets [30]. It is expected that the study will identify circPTK2 as a potential biomarker or novel therapeutic target for EOPE based on its proliferation and invasion-promoting effect on extravillous trophoblasts.
Conclusion
In conclusion, our research identified the functions and underlying mechanisms of the circPTK2 in PE progression. By sponging miR-619, circPTK2 regulates HTR-8/SVneo trophoblast proliferation and invasion, as well as modulating WNT7B expression. In this way, circPTK2 is a promising biomarker for the detection of EOPE and a potential therapeutic target.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Chappell LC, Cluver CA, Kingdom J et al (2021) Pre-eclampsia. Lancet 398(10297):341–354
Poon LC, Shennan A, Hyett JA et al (2019) The international federation of gynecology and obstetrics (figo) initiative on pre-eclampsia: a pragmatic guide for first-trimester screening and prevention. Int J Gynaecol Obstet 145(Suppl 1):1–33
Staff AC (2019) The two-stage placental model of preeclampsia: an update. J Reprod Immunol 134–135:1–10
Burton GJ, Redman CW, Roberts JM et al (2019) Pre-eclampsia: pathophysiology and clinical implications. BMJ 366:l2381
Liu P, Wang Z, Ou X et al (2022) The FUS/circEZH2/KLF5/ feedback loop contributes to CXCR4-induced liver metastasis of breast cancer by enhancing epithelial-mesenchymal transition. Mol Cancer 21(1):198
Jia N, Li J (2019) Role of circular RNAs in preeclampsia. Dis Markers 2019:7237495
Liu CX, Chen LL (2022) Circular RNAs: characterization, cellular roles, and applications. Cell 185(12):2016–2034
Sun N, Qin S, Zhang L et al (2021) Roles of noncoding RNAs in preeclampsia. Reprod Biol Endocrinol 19(1):100
Ou Y, Liu M, Zhu L et al (2019) The expression profile of circRNA and its potential regulatory targets in the placentas of severe pre-eclampsia. Taiwan J Obstet Gynecol 58(6):769–777
Shen XY, Zheng LL, Huang J et al (2019) CircTRNC18 inhibits trophoblast cell migration and epithelial-mesenchymal transition by regulating miR-762/Grhl2 pathway in pre-eclampsia. RNA Biol 16(11):1565–1573
Zhou W, Wang H, Yang J et al (2019) Down-regulated circPAPPA suppresses the proliferation and invasion of trophoblast cells via the miR-384/STAT3 pathway. Biosci Rep 39(9):BSR20191965. https://doi.org/10.1042/BSR20191965
Jiang M, Lash GE, Zhao X et al (2018) CircRNA-0004904, CircRNA-0001855, and PAPP-A: potential novel biomarkers for the prediction of preeclampsia. Cell Physiol Biochem 46(6):2576–2586
Zhou C, Yi C, Yi Y et al (2020) LncRNA PVT1 promotes gemcitabine resistance of pancreatic cancer via activating Wnt/β-catenin and autophagy pathway through modulating the miR-619-5p/Pygo2 and miR-619-5p/ATG14 axes. Mol Cancer 19(1):118
Song A, Wu Y, Chu W et al (2021) Involvement of miR-619-5p in resistance to cisplatin by regulating ATXN3 in oral squamous cell carcinoma. Int J Biol Sci 17(2):430–447
Nie X, Liu H, Liu L et al (2020) Emerging roles of Wnt ligands in human colorectal cancer. Front Oncol 10:1341
Matsuoka K, Bakiri L, Wolff LI et al (2020) Wnt signaling and Loxl2 promote aggressive osteosarcoma. Cell Res 30(10):885–901
Brown MA, Magee LA, Kenny LC et al (2018) Hypertensive disorders of pregnancy: ISSHP classification, diagnosis, and management recommendations for international practice. Hypertension 72(1):24–43
Phipps EA, Thadhani R, Benzing T et al (2019) Pre-eclampsia: pathogenesis, novel diagnostics and therapies. Nat Rev Nephrol 15(5):275–289
Munjas J, Sopić M, Stefanović A et al (2021) Non-coding RNAs in preeclampsia-molecular mechanisms and diagnostic potential. Int J Mol Sci 22(19):10652. https://doi.org/10.3390/ijms221910652
Li X, Yang L, Chen LL (2018) The biogenesis, functions, and challenges of circular RNAs. Mol Cell 71(3):428–442
Yang H, Li X, Meng Q et al (2020) CircPTK2 (hsa_circ_0005273) as a novel therapeutic target for metastatic colorectal cancer. Mol Cancer 19(1):13
Tay Y, Rinn J, Pandolfi PP (2014) The multilayered complexity of ceRNA crosstalk and competition. Nature 505(7483):344–352
Ma W, Niu Z, Han D et al (2022) Circ-FAT1 up-regulates FOSL2 expression by sponging miR-619-5p to facilitate colorectal cancer progression. Biochem Genet 60(4):1362–1379
Chen G, Gu Y, Han P et al (2019) Long noncoding RNA SBF2-AS1 promotes colorectal cancer proliferation and invasion by inhibiting miR-619-5p activity and facilitating HDAC3 expression. J Cell Physiol 234(10):18688–18696
Dietrich B, Haider S, Meinhardt G et al (2022) WNT and NOTCH signaling in human trophoblast development and differentiation. Cell Mol Life Sci 79(6):292
Sonderegger S, Husslein H, Leisser C et al (2007) Complex expression pattern of Wnt ligands and frizzled receptors in human placenta and its trophoblast subtypes. Placenta 28(Suppl A):S97-102
Cross JC, Nakano H, Natale DR et al (2006) Branching morphogenesis during development of placental villi. Differentiation 74(7):393–401
Parr BA, Cornish VA, Cybulsky MI et al (2001) Wnt7b regulates placental development in mice. Dev Biol 237(2):324–332
Galceran J, Fariñas I, Depew MJ et al (1999) Wnt3a-/–like phenotype and limb deficiency in Lef1(-/-)Tcf1(-/-) mice. Genes Dev 13(6):709–717
Li D, Li Z, Yang Y et al (2020) Circular RNAs as biomarkers and therapeutic targets in environmental chemical exposure-related diseases. Environ Res 180:108825
Funding
This work was supported by the Research Foundation of Natural Science Foundation of Hunan province (No. 2021JJ40550); Natural Science Foundation of Hunan province (No. 2021JJ30693); National Natural Science Foundation of China(No. 82000421);Guiding project of Hengyang science and Technology Bureau(No. 2021181);Guiding project of Hengyang science and Technology Bureau(No. 201909);Natural Science Foundation of Hunan province (No. 2022JJ40016); Scientific Research Project of Hunan Provincial Health Commission (No.20200929); Clinical Innovation Leading Scientific Research Projects of Hunan Science and Technology Department (No.2020SK52601).
Author information
Authors and Affiliations
Contributions
SW and LL designed and carried out the study, collected and analyzed the data, and wrote the manuscript. SW, TT and JX conducted the cell experiments. XY and JC performed glucose and lactate assay on. ZT and PW reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, S., Liu, L., Tao, T. et al. circPTK2 promotes proliferation, migration and invasion of trophoblast cells through the miR-619/WNT7B pathway in preeclampsia. Mol Cell Biochem 478, 2621–2627 (2023). https://doi.org/10.1007/s11010-023-04688-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-023-04688-1