Abstract
Ovarian injury can be induced by heat stress. Mice granulosa cells (GCs) are critical for normal ovarian function and they synthesize a variety of growth factors and steroids for the follicle. Furthermore, the growth, differentiation, and maturate of theca cells and oocyte are dependent upon the synthesis of GCs. Due to the critical biological functions of GCs, we hypothesized that the apoptosis and dysfunction of GCs could also be induced by heat stress. We analyzed GCs apoptosis and evaluated the expression of apoptosis-related genes (caspase-3, Bax, Bcl-2) after heat treatment. Radio immunity assay was used to measure the secretion of 17β-estradiol (E2) and progesterone (P4). RT-PCR was used to evaluate the expression of steroids-related genes (Star, CYP11A1, CYP19A1). Our data suggested that heat stress inhibited GCs proliferation, induced GCs apoptosis, decreased E2 and P4 secretion, reduced the steroids-related genes mRNA expression. Besides, our results indicated that heat treatment-induced apoptosis of GCs through the mitochondrial pathway, which involved caspase-3 and Bax. The reduction in steroids secretion and mRNA expression of Star, CYP11A1, and CYP19A1 might also play a role in heat-induced GCs apoptosis and ovarian injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There are many different kinds of stress in livestock, among these stresses, environmental stress has the greatest impact and heat stress is the main stress in environmental stress. Heat stress is sum of the animals for non-specific response of thermal environment, when the ambient temperature exceeds the temperature range of animals, they will show some of the stress response. Heat tress reduces reproductive performance, increases incidence and even causes death. With the advance of global warming and intensive farming, heat stress becomes more serious, which will result in a great economic loss. In prior studies, heat stress was shown to cause ovarian injury, resulted in degradation of reproductive performance [1, 2]. It was also shown to lead to ovarian tissue degeneration and necrosis [3–5]. Although heat-induced ovarian injury has been studied by investigators, its mechanism remains poorly understood. Thus, this study was designed to investigate the effect of heat stress on GCs to elucidate the mechanism of heat-induced ovarian injury.
Granulosa cells (GCs) are critical for ovarian function [6]. In an individual follicle, GCs locate outside of the zona pellucida. These cells maintain physical, physiological connection with the oocyte through gap junctions and play a very important role in nourishing oocyte and regulating oocyte development [7]. In the process of follicular development, GCs regulate the growth, differentiation, and maturation of theca cells as well as the oocyte by synthesizing a variety of growth factors and hormones. GCs are the main source of estrogen and progesterone [8–10]. Gap junctions that exist between GCs and oocytes enable small molecules (such as amino acids, ions and metabolites, etc.) to transfer to the oocyte, which can provide up to 85 % of oocyte metabolic needs. This conduit of nutrient delivery is indispensable for oocyte development [11, 12]. In addition, GCs can regulate the transcriptional activity of oocyte through these gap junctions and facilitate the post-transcriptional modification of several oocyte proteins [13].
Bax is one of the pro-apoptotic signals in mitochondrial pathway and it can promote follicular atresia [14]. Another member of the Bcl-2 family, Bcl-2, has an opposing function to Bax in regulating cell apoptosis [15–17]. In apoptotic cells, Bax is transferred from the cytoplasm to the mitochondria. It accelerates the opening of the mitochondrial ion voltage channel, which increases mitochondrial membrane permeability. This enables cytochrome C to remove from the mitochondria via ion channels and combine with apoptotic protease-activating factor (apaf21), then pro-caspase-9 and caspase-3 is activated. Caspase-3 executes cell apoptosis. In contrast, Bcl-2 inhibits the release of cytochrome C into the cytoplasm [18]. Bax and Bcl-2 regulate apoptosis by forming homodimer to promote apoptosis or heterodimer to inhibit apoptosis [19]. In earlier studies, the ratio of Bax to Bcl-2 was shown to provide the ‘molecule switch’ of cell apoptosis. It was also an indicator to determine whether a cancer cell is drug resistant or recurrent [20, 21].
There are two main pathways of ovarian granulosa cells apoptosis: death receptor pathway and mitochondrial pathway [22]. In death receptor pathway, death receptors combine their specific death ligands and activate caspase cascade [23]. In mitochondrial pathway, cell apoptosis is initiated by pro-apoptotic signals (Bax, etc.). Pro-apoptotic signals promote the release of cytochrome C from the mitochondria and into the cytoplasm. Subsequently, cytochrome C combines with apoptotic protease-activating factor (Apaf-1). Then pro-caspase-9 and caspase-3 is activated [24]. Caspase-3 is involved in both of the death receptor and mitochondrial pathway, moreover, it is the final executor of cell apoptosis and the common effect section of various apoptotic pathways downstream [25, 26]. Besides, many hormones and growth factors regulate GCs apoptosis by inhibiting or activating caspase activities. Therefore, the expression of caspase-3 can reflect the overall degree of cell apoptosis. Moreover, Pro-caspase-3 is shown as a PAC-1–sensitive dormant single-chain precursor with an N-terminal prodomain (Pro). During apoptosis, caspase-3 assembles as an active p17–p12 heterotetramer after proteolytic processing between the p17 and p12 subunits (at Asp175) and removal of the prodomain. PAC-1 is proposed to regulate the Asp–Asp–Asp (DDD) safety catch at amino acids 179–181 in pro-caspase-3, consequently inducing a conformational change that leads to proteolytic processing into the active p17 and p12 subunits [27].
In addition to GCs, E2 and P4 are also very important to maintain ovarian function. They regulate GCs apoptosis, follicular development and ovarian atresia, E2 inhibits GCs apoptosis by inhibiting endogenous endonuclease activation and promoting the division and growth of GCs [28]. It regulates the development of follicles by promoting the expression of steroid hormone synthesis genes and gonadotropin receptors. It also facilitates ovulation, and induces the LH surge before ovulation. In vitro experiments, the release of E2 is an important indicator to assess the adequacy of follicular development [29]. Estrogen deficiency or over-activity can cause ovarian tissue aging or tumorigenesis [30]. P4 is similarly critical to ovarian development. P4 maintains the estrous cycle and pregnancy of ovary. It is synthesized mainly in the GCs of the corpus luteum and placental syncytiotrophoblast. In earlier studies, progesterone was shown to regulate the apoptosis of GCs via a protein kinase G-dependent pathway [31].
In earlier studies, steroid-producing enzymes are critical for steroid synthesis. The process is as following: cholesterol is transported from the outer mitochondrial membrane to the inner membrane by steroidogenic acute regulatory protein (StAR), and then it is synthesized to pregnenolone by P450scc (cholesterol-side-chain cleavage enzyme). After that, pregnenolone is catalyzed to progesterone and then synthesized to androgen in thecal cell. At last, androgen is synthesized to estrogen by P450arom (aromatase cytochrome P450) in GCs. StAR is encoded by Star and p450scc is encoded by CYP11A1. P450arom is encoded by CYP19A1 and it is the rate-limiting enzyme for androgens conversing into estrogens [32, 33]. There is a lot of reaction substrate- cholesterol in the mitochondrial outer membrane cytoplasm while there is very few in thecal cells, however, the essential reaction that cholesterol conversing to pregnenolone takes place in the inner mitochondrial membrane [34], therefore, the step that cholesterol is transported from the outer to the inner mitochondrial membrane by StAR is the rate-limiting step in steroid hormone synthesis. After the transfer of cholesterol, P450scc role in the formation of progesterone, so StAR and P450scc are the rate-limiting enzymes for progesterone synthesis [35].
Methods
Primary culture of GCs
In this study, female Kunming mice (21 days old, 15 ± 2 g) were obtained from the Nanjing Qinglongshan Experimental Animal Factory and reared in the same cage. Mice were ad libitum under 25 °C and 12–12 h light dark cycles. All experiments were carried out in accordance with the National Institutes of Health guidelines. Each mouse was injected with PMSG (10 IU) and subsequently the anesthetized with pentobarbital sodium (4 mg/kg body weight) and euthanized by cervical vertebra dislocation after 48 h. Ovary samples were collected in petri dishes (35 × 15 mm) under aseptic conditions. 1 mL syringe needles were used to prick the follicles and release the pellets. The cell suspensions were cultured in F12/DMEM supplemented with 15 % FBS and 5 % Penicillin–Streptomycin in 6-well plates at 37 °C in the CO2 incubator. Cell attachment or not was observed and confirmed 24 h after incubation. After 48 h of culture, the medium was replaced to remove unattached cells and this procedure was repeated every 48 h.
Identification of GCs
Microscopic examination and immunohistochemistry for the follicle-stimulating hormone receptor (FSHR) was utilized to identify GCs within cultured cells. The morphological characteristics were determined using an inverted phase contrast microscope. The procedure of immunohistochemistry for the follicle-stimulating hormone receptor (FSHR), in brief, cell samples were fixed in 4 % paraformaldehyde for 20 min, air-dried for 5 min, and incubated in a 3 % H2O2 and pure methanol (1:4) for 30 min to quench endogenous cell peroxidase. Subsequently a blocking buffer was added to the slides. The primary antibody utilized was rabbit polyclonal FSHR (1:200) (Boster Biological Technology, China, diluted 1:200). Immunoreactivity was detected using goat anti-rabbit IgG secondary antibody (Beyotime Biotechnologies, China, diluted 1:50) marked by horseradish peroxidase (HRP). Diaminobenzidine tetrahydrochloride (DAB) was used as the substrate. Then the slides were counterstained with haematoxylin.
Determination of cell growth curve
GCs were cultured in 96-well plates and their optical density (OD) was assessed using the CCK-8 cell viability assay kit (Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions every 24 h until the 8th day. In brief, 10 μL CCK-8 was added to each well and incubated for 4 h at 37 °C. The plate was then scanned by a spectrophotometer (wavelength of 450 nm). The mean absorbance of each well was plotted against time to produce the cell growth curve.
GCs heat treatment
The plates containing cultivated cells were placed in the CO2 incubator at 40 °C or 43 °C with a paraffin membrane. Subsequently, the dishes were immediately returned to the 37 °C CO2 incubator for 6 h. The cell cultures were then terminated for analysis.
Analysis of cell viability
The cells were cultured in 96-well plates and their viability was assessed by utilizing CCK-8 cell viability assay kit (Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions after heat treatment at 40 °C or 43 °C. The cell viability was expressed as the proportion of absorbance values compared to the control.
Analysis of GC apoptosis by flow cytometry (FCM)
GCs were prepared by enzymatic digestion and washed with preheated phosphate buffered saline (PBS). Then the GCs were measured through APC-annexin V/PI double staining by utilizing the Annexin V Apoptosis Detection Kit APC (eBioscience) according to the manufacturer’s instructions before being analyzed by flow cytometry.
Activated-caspase-3 activity assay of GCs
Caspase-3 colorimetric assay kit (KeyGEN BioTECH, Nanjing, China) was used to determine activated-caspase-3 activity after collection and treated cells as previously described. Briefly, 50 μL of lysate was incubated in a 96-well plate with 50 μL of 2 × reaction buffer containing 0.5 μL of DTT. Then they were added with 5 μL caspase-3 substrate. After that the plate was incubated at 37 °C for 4 h and then read on an ELSA microplate reader at 405 nm wavelength. The level of activated-caspase-3 in the lysates was direct proportion to the color reaction. Therefore, the fold increase of activated-caspase-3 protease activity was determined by comparing absorbance from the treated samples with the controls.
RNA extraction and qualitative real-time PCR analysis
Qualitative real-time PCR analysis was used to evaluate the level of mRNAs and was performed using the ABI Prism Sequence Detection System (Applied Biosystems). Total RNA of control groups and treated groups were extracted using the Total RNA Isolation Kit (Takara). An aliquot of the total RNA was treated with DNase I and reverse-transcribed into cDNA with PrimeScriptTM RT Master Mix (TaKaRa). The resulting cDNA was used for SYBR Green (Applied Biosystems) real-time PCR amplification with the SYBR Green Master (Roche). Mouse GAPDH was used as an endogenous control. The sequences of the specific primer pairs for caspase-3, Bax, Bcl-2, Star, CYP11A1, CYP19A1, and GAPDH are presented in Table 1. The relative mRNA expression level from real-time PCR was calculated using the formula \(2^{{ - \Delta \Delta C_{\text{t}} }}\) method, as described previously [36, 37]. In all runs, the differences in mRNA expression of samples were compared to the internal control sample and the mRNA analyses were performed in triplicate.
Western blot analysis
The expression levels of pro-caspase-3 in GCs were analyzed by Western blot. The proteins from GCs were extracted by radio immunoprecipitation assay (RIPA Lysis Buffer, Beyotime Biotechnologies, Nanjing, China) according to the manufacturer’s instructions. The crude homogenates were centrifuged at 10,000×g for 3 min and the resultant supernatant, which contained the soluble protein fraction, was collected. The protein content was determined by utilizing the BCA Protein Assay Kit (Beyotime Biotechnologies, Nanjing, China) according to the manufacturer’s instructions. The samples were diluted with sample buffer (pH 6.8, 60 m mol/L Tris–Cl, 200 m mol/L dithiothreitol, 2 % sodium dodecyl sulpfate (SDS), 0.1 % bromophenol blue and 25 % glycerol) and heated to 99.9 °C for 8 min to ensure that they contained equal amounts of protein (12 μg). They were subsequently separated by SDS-PAGE in gels containing 10 % acrylamide. The samples were electrically transferred to an immunoblot PVDF membrane (Bio-Rad, Shanghai, China), which was then blocked with 5 % BSA for 1 h at room temperature and incubated with an anti-mouse caspase-3 (diluted 1:500; Boster Biotechnologies, China) or anti-mouse tubulin monoclonal antibody (diluted 1:1000; Boster Biotechnologies, China) at room temperate overnight with constantly shaking. The membranes were then washed in TBST for 10 min and incubated with a secondary antibody, HRP-labeled Goat Anti-Mouse IgG-H + L (diluted 1:1000; Beyotime Biotechnologies, Nanjing, China), for 2 h at room temperature. After the membranes were washed with TBST and incubated with ECL (electrochemiluminescence) for 3–5 min, the signal was visualized.
Determination of E2 and P4 levels
Radio immunoassay (RIA) was used to assay the levels of E2 and P4. The GCs serum-free culture medium was collected and preserved in −20 °C before and after heat treatment and subsequently sent to Nanjing General Hospital of Nanjing Military Command to determine the levels of E2 and P4. Their levels were expressed as the proportion of hormone concentration compared to the control.
Statistical analysis
Every experiment was repeated three times and six wells were included in each treatment. The data were analyzed using one-way ANOVA in SPASS13.0 and expressed as the mean ± standard deviation (SD) with P value < 0.05 considered significantly significant.
Results
Characterization of cultured GCs
The spherical GCs attached to plates and aggregated into clusters within the first 24 h. GCs were polygonal or fusiform. Their nuclei were large, round, and translucent. GCs covered more than 80 % of the wells at 3rd or 4th day. The cell morphology was consistent with the references [38–40] (Fig. 1). As illustrated in the cell growth curve, GCs proliferated as the cultured time progressed. The proliferation reached the peak by 3rd day, after which it began to slow, particularly by day six (Fig. 2). GCs are the only cells that express FSHR among the ovarian cells [41]. The result of the FSHR immunohistochemistry staining confirmed that the cultured cells were GCs (Fig. 3).
Effects of heat treatment on cell viability and GC apoptosis
The data showed that cell viability reduced following heat treatment. 40 °C group (P < 0.05) and 43 °C group (P < 0.01) had lower viability than control group (Fig. 4). Annexin V and PI double staining was used to verify GC apoptosis. Compared to the apoptotic rate of control group, 40 °C group and 43 °C group were increased (P < 0.05; P < 0.01) and the difference between them was significant (P < 0.05; Fig. 5a, b).
Effects of heat treatment on apoptosis of GCs. a Annexin and PI double staining. The cells were divided into four quadrants as follows: living cells (FITC−, PI−); early apoptotic cells (FITC+, PI−); late apoptotic/necrotic cells (FITC+, PI+). b Statistical chart of apoptosis rate, *P < 0.05 with respect to the control group; **P < 0.01 with respect to the control group; # P < 0.05 with respect to the 40 °C group; ## P < 0.01 with respect to the 40 °C group
Effects of heat treatment on caspase-3 expression in GCs
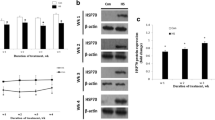
As shown in Fig. 6a, caspase-3 mRNA levels were increased in 40 °C group (P < 0.05) and 43 °C group (P < 0.01) and the difference between 40 °C group and 43 °C group was significant (P < 0.05; Fig. 6a). Procaspase-3 protein levels was also higher than control group after heat treated at 40 °C (P < 0.05) and 43 °C (P < 0.01) (Fig. 6b, c).
Qualitative real-time RT-PCR and Western blot analysis for caspase-3 of GCs in control group and treatment groups. a Relative mRNA expression of caspase-3 in GCs by different treatments. b Western blot analysis of caspase-3 of GCs in control group and treatment groups. c Relative protein expression of caspase-3 in GCs by different treatments, analyzed by Western blotting. d Relative activity of activated-caspase-3. Protein expression is expressed as a percentage of caspase-3 expression versus tubulin expression, and represents the mean ± SD of at least three independent experiments. *P < 0.05 with respect to the control group; **P < 0.01 with respect to the control group; # P < 0.05 with respect to the 40 °C group; ## P < 0.01 with respect to the 40 °C group
In addition, compared to control group, activated-caspase-3 activity was significantly increases in 40 °C group (P < 0.05) as well as 43 °C group (P < 0.01) (Fig. 6d).
Effects of heat treatment on Bax and Bcl-2 expression in GCs
Compared to control group, heat treatment groups contained significantly elevated Bax mRNA levels (Fig. 7a). Interestingly, the level of Bcl-2 mRNA expression was lower after 40 °C heat treatment while it was higher after 43 °C heat treatment. And the Bcl-2 mRNA expression difference between 40 and 43 °C was not statistically significant (Fig. 7b). In addition, the Bax/Bcl-2 ratio was significantly decreased in 40 °C (P < 0.05) group as well as 43 °C group (P < 0.05) compared to the control group (Fig. 7c).
Effect of heat treatment on Bax and Bcl-2 mRNA expression. a Relative mRNA expression of Bax in GCs after heat treatment. b Relative mRNA expression of Bcl-2 in GCs after heat treatment. c Relative mRNA expression of Bcl-2/Bax ratio after heat treatment. *P < 0.05 with respect to the control group; **P < 0.01 with respect to the control group; # P < 0.05 with respect to the 40 °C group; ## P < 0.01 with respect to the 40 °C group
Effects of heat treatment on E2 and P4 secretion of GCs
As shown in Fig. 8, both of E2 and P4 concentration decreased as the temperature increased. The concentration of E2 was not significantly different between 40 °C group and control group, (Fig. 8a) while the concentration of P4 was significantly lower (P < 0.01) (Fig. 8b). In 43 °C group, both of E2 and P4 concentration were significantly lower than control group (P < 0.05; P < 0.01) (Fig. 8). In addition, P4 concentration was significantly different between 40 °C group and 43 °C group (P < 0.05) (Fig. 8b). Star, CYP11A1 and CYP19A1 mRNA levels showed a downward trend as the temperature increased and the difference between 43 °C group and control group was significant (Fig. 9).
Effects of heat treatment on the secretion of E2 and P4 in GCs. a Relative concentration of E2 in GCs after different treatments. b Relative concentration of P4 in GCs after different treatments. *P < 0.05 with respect to the control group; **P < 0.01 with respect to the control group; # P < 0.05 with respect to the 40 °C group; ## P < 0.01 with respect to the 40 °C group
Effect of heat treatment on “rate-limiting” genes mRNA expression. a Relative mRNA expression of Star in GCs after heat treatment. b Relative mRNA expression of CYP11A1 in GCs after heat treatment. c Relative mRNA expression of CYP19A1 after heat treatment. *P < 0.05 with respect to the control group; **P < 0.01 with respect to the control group; # P < 0.05 with respect to the 40 °C group; ## P < 0.01 with respect to the 40 °C group
Discussion
It is previously demonstrated that heat stress can cause injury to ovary [4, 5] and GCs are critical for normal ovarian function [6]. In the process of follicular development, GCs regulate the growth, differentiation, and maturation of theca cells as well as the oocyte by synthesizing a variety of critical growth factors and hormones. Moreover, GCs are the main source of estrogen and progesterone [8–10]. Thus, GCs apoptosis will disturb the process of follicular development and the secretion of steroids. Prior investigations have found that E2 and P4 can protect GCs from apoptosis [31]. In the present study, our data suggested that heat treatment could induce GCs apoptosis and disturb the secretion of E2 and P4, indicating that ovarian injury has occurred.
Caspase-3 is involved in two major apoptotic pathways of GCs- death receptor pathway and mitochondrial pathway. Previous studies have found that the proteins involved in the mitochondrial pathway are primarily those belonging to the Bcl-2 family [42], and Bcl-2 gene deletion could cause a reduction of follicles number in mice, while overexpression of Bcl-2 could inhibit GC apoptosis and follicular atresia [19]. After heat treatment, GCs proliferation was inhibited, the expression of caspase-3 were significantly increased. Moreover, the ratio of Bax/Bcl-2 was significantly increased, which indicates that the mitochondrial pathway plays an important role in heat-induced apoptosis of GCs. The expression of Bax mRNA was significantly elevated while the expression of Bcl-2 mRNA was not, which suggested that Bax might play an important role in heat-induced GC apoptosis while Bcl-2 was not as critical as Bax. We hypothesized that the possible mechanistic process was, Bax was transferred from the cytoplasm to the mitochondria and accelerated the opening of mitochondrial ion voltage channel, which increased mitochondrial membrane permeability. Subsequently, cytochrome C was removed from the mitochondria via ion channels and cytochrome C combined with apoptotic protease-activating factor (Apaf-1). Thus, pro-caspase-9 was activated and then caspase-3 was activated, ultimately cell was apoptotic. The apoptosis of cells is co-regulated by multiple genes and proteins so there were many potential mechanisms by which Bcl-2 mRNA expression was increased or decreased. We suspected that the reduction of Bcl-2 mRNA expression after 40 °C heat treatment might due to heat induction. However, the increasing level of Bcl-2 mRNA expression might be the result of heat shock proteins, which was a self-protection mechanism in GCs [43–45]. Meanwhile, our data showed that, after heat treatment, the secretion of E2 and P4 was reduced and the related genes (Star, CYP11A1, CYP19A1) mRNA expression were reduced, which indicated that heat treatment might disturb the secretion of E2 and P4 via inducing GCs apoptosis and reducing the related genes expression. Moreover, the decline of E2 and P4, in turn, enhanced the possibility of GCs apoptosis and disturbed follicular development.
In conclusion, heat-induced ovarian injury is well understood, but the present study highlights a potential mechanism of heat-induced ovarian injury. We found that Bax was involved in the mechanism via mitochondrial pathway. Furthermore, the reduction in steroids secretion (E2, P4) and mRNA expression of Star, CYP11A1, and CYP19A1 might also play an important role in heat-induced GCs apoptosis and ovarian injury (Fig. 10), although the precise mechanism by which this occurs still needs to be clarified.
Proposed mechanism of heat-induced injury in GCs. Bax was involved in the mechanism via mitochondrial pathway. The secretion of E2 and P4 might be disturbed by heat treatment via inducing GCs apoptosis and reducing the related genes expression. Moreover, the decline of E2 and P4, in turn, might enhance the possibility of GCs apoptosis and ovarian injury
References
Marion RS, Spain JN, Spiers DE (1998) Effects of controlled heat stress on ovarian function of dairy cattle. Lactating cows. J Dairy Sci 81:2124–2131
Nivet AL, Vigneault C, Blondin P (2013) Changes in granulosa cells’ gene expression associated with increased oocyte competence in bovine. Reproduction 145:555–565
Guo L, Zhang M (2008) Advances in heat stress on livestock reproductive function and its regulation mechanism. China Anim Husb Vet Med 9:119–122
Zeng JY (2012) Progress on Animal reproductive performance of heat stress. China Animal Husbandry and Veterinary Digest 7
Wu JG, Mao DG (2007) Effect of heat stress on reproductive function in animals and the mechanism. China Anim Husb Vet Dig 4:28–29
Havelock JC, Rainey WE, Carr BR (2004) Ovarian granulosa cell lines. Mol Cell Endocrinol 228:67–78
Jančar N, Kopitar AN, Ihan A (2007) Effect of apoptosis and reactive oxygen species production in human granulosa cells on oocyte fertilization and blastocyst development. J Assist Reprod Genet 24:91–97
Denkova R, Bourneva V, Staneva-Dobrovski L (2004) In vitro effects of inhibin on apoptosis and apoptosis related proteins in human ovarian granulosa cells. Endocr Regul 38:51–55
Wang SJ, Liu WJ, Wu CJ (2012) Melatonin suppresses apoptosis and stimulates progesterone production by bovine granulosa cells via its receptors (MT1 and MT2). Theriogenology 78:1517–1526
Matsuda F, Inoue N, Manabe N (2012) Follicular growth and atresia in mammalian ovaries: regulation by survival and death of granulosa cells. J Reprod Dev 58:44–50
Albertini DF, Combelles CM, Benecchi E (2001) Cellular basis for paracrine regulation of ovarian follicle development. Reproduction 121:647–653
Gilchrist RB, Ritter LJ, Armstrong DT (2004) Oocyte-somatic cell interactions during follicle development in mammals. Anim Reprod Sci 82–83:431–446
Carabatsos MJ, Sellitto C, Goodenough DA (2000) Oocyte–granulosa cell heterologous gap junctions are required for the coordination of nuclear and cytoplasmic meiotic competence. Dev Biol 226:167–179
Renault TT, Floros KV, Elkholi R (2015) Mitochondrial shape governs BAX-induced membrane permeabilization and apoptosis. Mol Cell 57:69–82
Croker BA, O’Donnell JA, Nowell CJ (2011) Fas-mediated neutrophil apoptosis is accelerated by Bid, Bak, and Bax and inhibited by Bcl-2 and Mcl-1. Proc Natl Acad Sci 108:13135–13140
Kamoi K, Iino M, Ishiguro H (2006) Regeneration therapy for oral disease. Hum Cell 19:76–82
Xie X, Clausen OPF, Boysen M (2003) Prognostic value of Bak expression in oral tongue squamous cell carcinomas. Oncol Rep 10:369–374
Chipuk JE, Green DR (2008) How do BCL-2 proteins induce mitochondrial outer membrane permeabilization? Trends Cell Biol 18:157–164
Lalier L, Cartron PF, Juin P (2007) Bax activation and mitochondrial insertion during apoptosis. Apoptosis 12:887–896
Del Poeta G, Venditti A, Del Principe MI (2003) Amount of spontaneous apoptosis detected by Bax/Bcl-2 ratio predicts outcome in acute myeloid leukemia (AML). Blood 101:2125–2131
Oshikawa T, Okamoto M, Ahmed SU (2006) The relationship between gene expression of Bcl-2 and Bax and the therapeutic effect in oral cancer patients. Cancer Chemother 33:1723–1725
Chen W, Woodruff TK, Mayo KE (2000) Activin A-Induced HepG2 Liver Cell Apoptosis: involvement of activin receptors and smad proteins 1. Endocrinology 141:1263–1272
Matsuda-Minehata F, Maeda A, Cheng Y (2008) Regulation of granulosa cell apoptosis by death ligand–receptor signaling. Anim Sci J 79:1–10
Gillies LA, Kuwana T (2014) Apoptosis regulation at the mitochondrial outer membrane. J Cell Biochem 115:632–640
Riedl SJ, Shi Y (2004) Molecular mechanisms of caspase regulation during apoptosis. Nat Rev Mol Cell Biol 5:897–907
Sudo H, Minami A (2010) Regulation of apoptosis in nucleus pulposus cells by optimized exogenous Bcl-2 overexpression. J Orthop Res 28:1608–1613
Porter AG (2006) Flipping the safety catch of procaspase-3. Nat Chem Biol 2:509–510
Chaube SK, Prasad PV, Thakur SC (2005) Estradiol protects clomiphene citrate–induced apoptosis in ovarian follicular cells and ovulated cumulus–oocyte complexes. Fertil Steril 84:1163–1172
Rosenfeld CS, Wagner JS, Roberts RM (2001) Intraovarian actions of oestrogen. Reproduction 122:215–226
Bayne S, Li H, Jones MEE (2011) Estrogen deficiency reversibly induces telomere shortening in mouse granulosa cells and ovarian aging in vivo. Protein Cell 2:333–346
Peluso JJ, Pappalardo A (2004) Progesterone regulates granulosa cell viability through a protein kinase G-dependent mechanism that may involve 14-3-3σ. Biol Reprod 71:1870–1878
Drouilhet L, Taragnat C, Fontaine J (2010) Endocrine characterization of the reproductive axis in highly prolific lacaune sheep homozygous for the FecLL mutation. Biol Reprod 82:815–824
Miller WL, Auchus RJ (2011) The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr Rev 32:81–151
Stocco DM (2001) StAR protein and the regulation of steroid hormone biosynthesis. Annu Rev Physiol 63:193–213
Belin F, Goudet G, Duchamp G (2009) Intrafollicular concentrations of steroids and steroidogenic enzymes in relation to follicular development in the Mare. Biol Reprod 62:1335–1343
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the \(2^{{ - \Delta \Delta {\rm C}_{\text{t}} }}\) method. Methods 25:402–408
Vandesompele J, De Preter K, Pattyn F, (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3: research0034
Minegish T, Nakamura K, Takakura Y (1991) Cloning and sequencing of human FSH receptor cDNA. Biochem Biophys Res Commun 175:1125–1130
Allan CM, Garcia A, Spaliviero J (2006) Maintenance of spermatogenesis by the activated human (Asp 567 Gly) FSH receptor during testicular regression due to hormonal withdrawal. Biol Reprod 74:938–944
Jun JK, Yoon JS, Ku SY (2006) Follicle-stimulating hormone receptor gene polymorphism and ovarian responses to controlled ovarian hyperstimulation for IVF-ET. J Hum Genet 51:665–670
Simoni M, Gromoll J, Nieschlag E (1997) The follicle-stimulating hormone receptor: biochemistry, molecular biology, physiology, and pathophysiology. Endocr Rev 18:739–773
Tischner D, Woess C, Ottina E (2010) Bcl-2-regulated cell death signalling in the prevention of autoimmunity. Cell Death Dis 1:e48
Kim JY, Yenari MA, Lee JE (2015) Regulation of inflammatory transcription factors by heat shock protein 70 in primary cultured astrocytes exposed to oxygen–glucose deprivation. Neuroscience 286:272–280
Tan JGL, Lee YY, Wang T (2015) Heat shock protein 27 overexpression in CHO cells modulates apoptosis pathways and delays activation of caspases to improve recombinant monoclonal antibody titre in fed-batch bioreactors. Biotechnol J 10:790–800
Lee JM, Lee JM, Kim KR (2015) Zinc preconditioning protects against neuronal apoptosis through the mitogen-activated protein kinase-mediated induction of heat shock protein 70. Biochem Biophys Res Commun 459:220–226
Acknowledgments
This research was supported by the National Natural Science Foundation of China No. 31501955, the Science and Technology Sustentation Project of China (2011BAD28B02, 2012BAD12B00).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luo, M., Li, L., Xiao, C. et al. Heat stress impairs mice granulosa cell function by diminishing steroids production and inducing apoptosis. Mol Cell Biochem 412, 81–90 (2016). https://doi.org/10.1007/s11010-015-2610-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2610-0