Abstract
Lactic acid ionic liquid [C4mim][Lact] (1-butyl-3-methylimidazolium lactic acid) was synthesized by the neutralization method. Using the solution–reaction isoperibol calorimeter, the molar enthalpies of solution, Δsol H m, with different molalities were determined in the temperature range from (288.15 to 308.15 ± 0.01) K with an interval of 5 K. In terms of Archer’s method, the standard molar enthalpy of solution for [C4mim][Lact], \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \), was obtained, and then, the values of apparent relative molar enthalpy, Φ L, and the Pitzer’s parameters \( \beta_{\text{MX}}^{(0)\text{L}} \) and \( \beta_{\text{MX}}^{(1)\text{L}} \) were determined for [C4mim][Lact]. The plot of \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \) versus (T—298.15) K is a good straight line, and its slope is the standard molar heat capacity of solution, \( \Delta C_{\text{p,m}}^{\theta } = 240\,{\text{J}}\,{\text{K}}^{ - 1} \). According to Glasser’s theory of lattice energy, the hydration enthalpy of cation and anion, (ΔH + + ΔH −) = −471 kJ mol−1, in infinite dilution and the hydration enthalpy of anion, ΔH −([Lact]−) = −257 kJ mol−1, were obtained at 298.15 K. The heat capacity of aqueous [C4mim][Lact], C p(sol), and the apparent molar heat capacity, Φ C p, of various specific molalities were also obtained.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In recent years, ionic liquids (ILs) attach great importance to the scientific community and industry as green solvents and new materials [1–4]. Among them, chiral ionic liquids (CILs) are particularly attractive for their potential applications to chiral discrimination, including asymmetric synthesis and optical resolution of racemates. However, because of their recyclability, ease of synthesis, and high degree of organization, CILs can be viable chiral solvents for effectively transferring their chiralities to reaction products [5, 6]. In fact, several examples of asymmetric induction by CILs have been reported in the literature [7]. This growing interest in the use of CILs as solvents for chiral induction has prompted us to commence the present study on the synthesis and fundamental properties of CILs.
As the CILs, 1-butyl-3-methylimidazolium lactate [C4mim][Lact] possessing chiral anion has been demonstrated to display superior capability in the general field of chiral recognition [8–10]. It is well known that the basic physicochemical data of the ILs such as the molar enthalpies and heat capacity are of great importance for industrial process; however, the thermochemical property of lactate ionic liquid has not been reported.
As a continuation of our previous investigation [11, 12], in this paper we report that (1) the enthalpies of solution for [C4mim][Lact] were measured at (288.15 to 308.15 ± 0.01) K with an interval of 5 K. (2) The values of the standard molar enthalpy, \( \Delta _{\text{sol}} H_{\text {m}}^{\theta } \), and the apparent relative molar enthalpy, Φ L, for [C4mim][Lact] were obtained in terms of Archer’s method [13], and the Pitzer’s parameters \( \beta_{\text{MX}}^{(0)\text{L}} \) and \( \beta_{\text{MX}}^{(1)\text{L}} \) for [C4mim][Lact] were determined according to the Pitzer’s theory. (3) The standard molar heat capacity of solution, \( \Delta C_{\text{p,m}}^{\theta } \), for [C4mim][Lact] was obtained from the slope of the straight line of \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \) versus (T—298.15) K. (4) The heat capacity of aqueous [C4mim][Lact], C p(sol), and the apparent molar heat capacity, Φ C p, of various specific molalities were also obtained.
Experimental
Chemicals
The source, purity, and purification process of the materials are listed in Table 1.
Synthesis of IL [C4mim][Lact]
[C4mim]Br (1-butyl-3-methylimidazolium bromide) was synthesized according to the literature [14], and [C4mim][Lact] was prepared by a neutralization method according to Fukumoto et al. [4]. The structure was confirmed by 1H NMR spectroscopy (see Figure S1 of Supporting Information) and the differential scanning calorimetry (DSC) measurement (see Figure S2 of Supporting Information). The DSC spectroscopy showed that [C4mim][Lact] had no obvious melting point. Silver nitrate test does not show any precipitation, and pH-metric titration of aqueous solution of the IL showed no free OH− ions. The purity of the synthesized IL [C4mim][Lact] is greater than 0.99 mass fractions. The measurement of water content for IL [C4mim][Lact] is less than 0.8 mass% by Karl Fischer moisture titrator (ZSD-2 type).
Determination of enthalpy of aqueous solution
A solution–reaction isoperibol calorimeter was used to determine the molar enthalpies of solution for the ILs as reports [15–17]. The calorimeter was calibrated with KCl in water and THAM [tris-(hydroxymethyl) aminomethane] in 0.1 mol dm−3 HCl (aq) at 298.15 K as Rychly et al.’s [18] and Montgomery et al.’s [19] report. The results showed that the molar solution enthalpy Δsol H m = (17.5427 ± 0.0197) kJ mol−1, for KCl, and Δsol H m = (−29.7893 ± 0.0331) kJ mol−1, for THAM, which are in good agreement with the corresponding data [18, 19] and within experimental error. It means that the calorimeter can be used and ensured the precision for our experiment. Therefore, the molar enthalpies of solution for [C4mim][Lact] with different molalities, Δsol H m, were measured in water in the temperature range from (288.15 to 308.15 ± 0.01) K with an interval of 5 K, and the data are listed in Tables 2, 3, 4, 5, and 6. These tables show that the values of Δsol H m are negative, that is, the dissolution for [C4mim][Lact] is a typical exothermal process.
Results and discussion
The standard molar enthalpy of solution, \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \), and the apparent relative molar enthalpy, Φ L for [C4mim][Lact]
According to Archer et al. [13], in terms of a Debye–Hückel limiting equation, standard molar enthalpy of solution for [C4mim][Lact], \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \), can be obtained at the given temperature in following equation:
where Y is the extrapolation function calculated from experimental data, A H is the Debye–Hückel parameter for enthalpy, and its value at different temperatures was taken from the literature [20], b is 1.2 [20], I is ionic strength (I = m for the 1:1 electrolyte ILs), β is empirical constant, m is molality. Plotting Y versus m, a series of straight lines were obtained. The intercepts of lines are \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \), and the slopes are β; these values are listed in Table 7. The value of the standard molar enthalpy, \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } = - 41.06\,{\text{kJ}}\,{\text{mol}}^{ - 1} \), was obtained at 298.15 K.
The apparent relative molar enthalpy, Φ L, for [C4mim][Lact] may be described in following equation:
The values of Φ L were calculated and are also listed in Tables 2–6, respectively.
According to Pitzer’s theory:
The parameters \( \beta_{\text{MX}}^{(0)\text{L}} \) and \( \beta_{\text{MX}}^{(1)\text{L}} \) account for various types of short-range interactions between M and X; \( C_{\text{MX}}^{\emptyset \text{L}} \) is for triple-ionic interaction and is important only at high concentration. However, the concentration of [C4mim][Lact] was low in the present work, such that the parameter \( C_{\text{MX}}^{\emptyset \text{L}} \) is negligible.
Substituting Eq. (4) into Eq. (3), a working equation for determining Pitzer’s parameters is as follows:
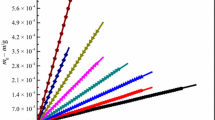
Figure 1 shows the plot of Y P against y′ for [C4mim][Lact]. The values of Pitzer’s parameters: \( \beta_{\text{MX}}^{(0)\text{L}} \) and \( \beta_{\text{MX}}^{(1)\text{L}} \) were obtained from the intercepts and the slopes of the linear regressions, respectively. The correlation coefficients, r, and the standard deviations, s, of the regressions were also obtained. All the above parameters are also listed in Table 8.
Plot of Y P [extrapolation function calculated from Eq. (5)], against y′ for [C4mim][Lact] in temperature range of (288.15 to 308.15 ± 0.01) K. Black square box 288.15 K; red circle 293.15 K; green triangle 298.15 K; blue inverted triangle 303. 15 K; turquoise blue left pointed triangle 308.15 K. (Color figure online)
The standard molar heat capacity of solution, \( \Delta C_{\text{p,m}}^{\theta } \), for [C4mim][Lact]
Plotting \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \) against (T—298.15) K for [C4mim][Lact], a good straight line was obtained and is shown in Fig. 2. The linear regression equation is \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } /{\text{kJ}}\,{\text{mol}}^{ - 1} = - 41.01 + 0.240 \) (T—298.15) K with a correlation coefficient, r = 0.995, and standard deviation, s = 0.22. From the linear regression equation, the intercept −41.01 is the standard molar enthalpy of solution \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \), which is in good agreement with the experimental value −41.06 kJ mol−1. The slope is 0.240 which means the standard molar heat capacity of solution, \( \Delta C_{\text{p,m}}^{\theta } = 240\,{\text{J}}\,{\text{K}}^{ - 1} \,{\text{mol}}^{ - 1} \), for [C4mim][Lact].
The estimation of the anion hydration enthalpy, ΔH −([Lact]−), for [C4mim][Lact]
The hydration enthalpy of the [C4mim][Lact] was estimated in terms of the following thermodynamic cycle indicated (see Fig. 3):
where ΔH + is the cation hydration enthalpy, and ΔH − is the anion hydration enthalpy, U POT is lattice energy which can be estimated by Glasser’s theory [21, 22]:
where α and β are fitting coefficients, ionic strength I = 1, according to the suggestion from Gutowski et al. [23], the values of α and β are, respectively, 83.3 nm kJ mol−1 and 157.3 kJ mol−1 for the ILs which are 1:1 salts. According to Fang et al. [24], ρ = 1.1282 g cm−3 for [C4mim][Lact] at 298.15 K; then, the value of molecular volume, V m = 0.3357 nm3, was calculated by using the equation, V m = M/(Nρ). According to Eq. (4), U POT for [C4mim][Lact] is 430 kJ mol−1, was calculated; further, the hydration enthalpy of cation and anion in infinite dilution, (ΔH + + ΔH −) = −471 kJ mol−1, at 298.15 K was obtained. Since the hydration enthalpy of cation, ΔH +([C4mim]+), is −214 kJ mol−1 [25], the hydration enthalpy of the lactic acid anion, ΔH −([Lact]−), is −257 kJ mol−1, was obtained. Compared with ΔH −([Gly]−) = −281 kJ mol−1 [11], the hydration enthalpy of ΔH −([Lact]−) is littler. The mainly reason may be caused by the hydration ability which decreases with the increase in the size of anion volumes [26]. The volume of [Lact]− is larger than that of [Gly]−, so its hydration ability is weaker.
The apparent molar heat capacity, Φ C p, for aqueous [C4mim][Lact]
The apparent molar heat capacity, Φ C p, for aqueous [C4mim][Lact] may be expressed as:
where the molar enthalpy of solution, Δsol H m, for various specific molalities of [C4mim][Lact] can be calculated. According to Paulechka et al. [27], molar heat capacity, C p (ILs), of IL can be estimated with the molar volume (V/cm3 mol−1) through the following empirical equation:
where V is molar volume, V = 202.09 cm−3, which was calculated by using the equation, V = M/ρ, ρ = 1.1282 g cm−3 [24], the value of C p(ILs) for [C4mim][Lact] at 298.15 K obtained from Eq. (9) was 395.60 J K−1 mol−1. The apparent molar heat capacity, Φ C p, of various molalities for [C4mim][Lact] was obtained and is listed in Table 9. From Table 9, we can see the values of Φ C p increase with an increase in the molalities.
The heat capacity of aqueous [C4mim][Lact], C p(sol), can be obtained by the following equation:
where \( C_{\text{p,w}}^{0} \) is the molar heat capacity of water, M w is the molar mass of water, m is the molality. The values of C p(sol) are also listed in Table 9, and the values of C p(sol) also increase with the increase in the molalities.
Conclusions
The standard molar enthalpies, \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \), and the apparent relative molar enthalpies, Φ L, and the parameters \( \beta_{\text{MX}}^{(0){\text{L}}} \) and \( \beta_{\text{MX}}^{(1)\text{L}} \) were obtained for [C4mim][Lact] in the temperature range from (288.15 to 308.15 ± 0.01) K with an interval of 5 K. Then, the standard molar heat capacity of solution, \( \Delta C_{\text{p,m}}^{\theta } \), for [C4mim][Lact] was obtained from the slope of the straight line of \( \Delta _{\text{sol}} H_{\text{m}}^{\theta } \) versus (T—298.15) K. In addition, the hydration enthalpy of the anion, ΔH −([Lact]−), was obtained by Glasser’s theory. Compared with the value of ΔH −([Gly]−) = −281 kJ mol−1, the hydration enthalpy of ΔH −([Lact]−) = −257 kJ mol−1 is littler. The heat capacity of aqueous, C p(sol), and the apparent molar heat capacity, Φ C p, for various molalities of IL [C4mim][Lact] were estimated, and the results show that the values of C p(sol) and Φ C p increase with an increase in molalities.
References
Murillo-Hernández JA, López-Ramírez S, Domínguez JM, Duran-Valencia C, García-Cruz I, González-Guevara JA. Survey on ionic liquids effect based on metal anions over the thermal stability of heavy oil. J Therm Anal Calorim. 2009;95:173–9.
Pawlak K, Szurkowski J, Skrzypczak A, Bialek-Bylka GE. The thermal deactivation of all-trans and 15-cis beta-carotene-excited states in the ionic liquids without and with methylenoxy group. J Therm Anal Calorim. 2015;120:627–32.
Zhang SJ, Wang JJ, Lu XM, Zhou Q. Structures and interactions of ionic liquids. Heidelberg: Springer; 2014. p. 151–97.
Fukumoto K, Yoshizawa M, Ohno H. Room temperature ionic liquids from 20 natural amino acids. J Am Chem Soc. 2005;127:2398–9.
Wagner V, Schulz PS, Wasserscheid P. Asymmetric hydrogenation catalysis via ion-pairing in chiral ionic liquids. J Mol Liq. 2014;192:177–84.
Navarro P, Larriba M, Beigbeder JB. Thermal stability and specific heats of [bpy][BF4] + [bpy][Tf2N] and [bpy][BF4] + [4bmpy][Tf2N] mixed ionic liquid solvents. J Therm Anal Calorim. 2015;119:1235–43.
Schulz PS, Mller N, Bsmann A, Wasserscheid P. Effective chirality transfer in ionic liquids through ion-pairingeffects. Angew Chem. 2007;119:1315–7.
Wu JW, Su P, Huang J, Wang SM, Yang Y. Synthesis of teicoplanin-modified hybrid magnetic mesoporous silica nanoparticles and their application in chiral separation of racemic compounds. J Colloid Interface Sci. 2013;399:107–14.
Baudequin C, Bregeon D, Levillain J, Guillen F, Plaquevent JC, Gaumont AC. Chiral ionic liquids, a renewal for the chemistry of chiral solvents? Design, synthesis and applications for chiral recognition and asymmetric synthesis. Tetrahedron Asymmetry. 2005;16:3921–45.
Kapnissi-Christodoulou CP, Stavrou IJ, Mavroudi MC. Chiral ionic liquids in chromatographic and electrophoretic separations. J Chromatogr A. 2014;1363:2–10.
Guan W, Xue WF, Li N, Tong J. Enthalpy of solution of amino acid ionic liquid 1-butyl-3-methylimidazolium glycine. J Chem Eng Data. 2008;53:1401–3.
Guan W, Li L, Wang H, Tong J, Yang JZ. study on thermochemical properties of ionic liquids based on transition metal. J Therm Anal Calorim. 2008;94(2):507–10.
Archer DG, Widegren JA, Kirklin DR, Magee JW. Enthalpy of solution of 1-octyl-3-methylimidazolium tetrafluoroborate in water and in aqueous sodium fluoride. J Chem Eng Data. 2005;50:1484–91.
Wilkes JS, Levisky JA, Wilson RA, Hussey CL. Dialkylimidazolium chloroaluminate melts: a new class of room-temperature ionic liquids for electrochemistry. Inorg Chem. 1982;21:1263–8.
Di YY, Qu SS, Liu Y, Wen DC, Tang H, Li LW. A thermochemical study of the solid-state coordination reactions of two α-amino acids with copper(II) acetate. J Thermochim Acta. 2002;387:115–9.
Liu JG, Xue WF, Qin Y, Yan CW. Enthalpy of solution for anhydrous VOSO4 and estimated enthalpy of reaction for formation of the ion pair [VOSO4]0. J Chem Eng Data. 2009;54:1938–41.
Ji M, Liu MY, Gao SL, Shi QZ. A new microcalorimeter for measuring thermal effects. Instrum Sci Technol. 2001;29:53–7.
Montgomery RL, Melaugh RA, Lau CC, Meier GH, Chan HH, Rossini FD. Determination of the energy equivalent of a water solution calorimeter with a standard substance. J Chem Thermodyn. 1977;9:915–36.
Rychly R, Pekarek V. The use of potassium chloride and tris(hydroxymethyl)aminomethane as standard substances for solution calorimetry. J Chem Thermodyn. 1977;9:391–6.
Pitzer KS. In activity coefficients in electrolyte solution, Chapter 3. Boca Raton, FL: CRC Press; 1991.
Glasser L. Lattice and phase transition thermodynamics of ionic liquids. Thermochim Acta. 2004;421:87–93.
Glasser L, Jenkins HDB. Volume-based thermodynamics: a prescription for its application and usage in approximation and prediction of thermodynamic data. J Chem Eng Data. 2011;56:874–80.
Gutowski KE, Rogers RD, Dixon DA. Accurate thermochemical properties for energetic materials applications. II. Heats of formation of imidazolium-, 1,2,4-triazolium-, and tetrazolium-based energetic salts from isodesmic and lattice energy calculations. J Phys Chem B. 2007;111:4788–800.
Fang DW, Tong J, Guan W, Wang H, Yang JZ. Prediction of the thermodynamic properties of 1-alkyl-3-methylimidazolium lactate ionic liquids [Cnmim][Lact](n = 2, 3, 4, 5, and 6) by parachor. Sci Sin Chim. 2010;40(9):1339–47.
Guan W, Yang JZ, Li L, Wang H, Zhang QG. Thermochemical properties of aqueous solution containing ionic liquids. 1. The heat of reaction mixed 1-methyl-3-butylimidazolium chloride with InCl3. Fluid Phase Equilib. 2006;239:161–75.
Liang XY, Zhang X, Wang SG, Li W, Rong H. A study on molar enthalpy of solution in amino acid tetrafluoroborate ionic liquids. J Beijing Inst Petrochem Technol. 2011;19:50–3.
Paulechka YU, Kabo AG, Blokhin AV, Kabo GJ, Shevelyova MP. Heat capacity of ionic liquids: experimental determination and correlations with molar volume. J Chem Eng Data. 2010;55:2719–24.
Acknowledgements
This project was supported by the National Natural Science Foundation of China (21173107) and Liaoning Excellent Talents in University (2015025).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zheng, L., Bu, XX., Fan, BH. et al. Study on thermodynamic property for ionic liquid [C4mim][Lact](1-butyl-3-methylimidazolium lactic acid). J Therm Anal Calorim 123, 1619–1625 (2016). https://doi.org/10.1007/s10973-015-5051-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-5051-9