Abstract
The type II class of sex pheromones found in moths is composed of polyene hydrocarbons and their epoxides. Analysis of Utetheisa ornatrix females by gas chromatography-mass spectrometry and measurement of responses of male moths by coupled gas chromatography-electroantennogram detection confirmed the presence of large amounts of (Z,Z,Z)-1,3,6,9-heneicosatetraene (1,3,6,9-21:Hy) and smaller amounts of (Z,Z,Z)-3,6,9-heneicosatriene (3,6,9-21:Hy). Both compounds were detected in pheromone glands of newly emerged adults, with low amounts found in the late pupal stage, indicating that sex pheromone biosynthesis started in the late pupal stage. In our population of females (several hundred sampled), approximately 90% produced the tetraene, 1,3,6,9-21:Hy, as the major component, while the other 10% produced only a large amount (1500–2000 ng) of 3,6,9-21:Hy, with no detectable amount of the tetraene. This result could indicate that two distinct populations are present in our original collection site in Florida. Decapitated female moths accumulated 3,6,9-21:Hy and 1,3,6,9-21:Hy compared to the same age normal females, indicating that female moths continuously produce pheromone. A pheromone biosynthesis activating neuropeptide (PBAN)-like neuropeptide did not affect sex pheromone production as indicated by injection of synthetic PBAN and decapitation of U. ornatrix female adults. When the labeled precursor, D4-9,12,15-18:acid, was injected into the early pupal stage, the most abundantly labeled hydrocarbons were 3,6,9-21:Hy and 1,3,6,9-21:Hy in the female adults. This result indicated that 3,6,9-21:Hy could be biosynthesized from linolenic acid through chain elongation and decarboxylation. To determine how 1,3,6,9-21:Hy is produced, D4-3,6,9-21:Hy was injected into pupae and monitored for incorporation of label. No label was incorporated into 1,3,6,9-21:Hy, although a large amount of triene, 3,6,9-21:Hy, was recovered in the pheromone gland. This indicates that U. ornatrix females do not use 3,6,9-21:Hy to produce 1,3,6,9-21:Hy, and the terminal double bond is introduced earlier in the biosynthetic pathway.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
One major group (type I) of moth sex pheromones is composed of linear fatty acid-derived compounds, 12–18 carbons in chain length, with an oxygenated functional group and one to three double bonds. A second major class (type 2) of sex pheromones is based on hydrocarbons with a polyene and/or epoxide functional group. This class of pheromones is typically found in four large moth families, the Geometridae, Noctuidae, Arctiidae, and Lymantridae (Byer 2006; Witzgall et al. 2004). The polyene type is biosynthesized from diet-derived linoleic or linolenic acids and characterized by 17–23 carbon chains with 1–4 double bonds, and 0–2 epoxides (Millar 2000).
Typically, type I sex pheromones are biosynthesized by specific enzymes in the pheromone gland and released by calling females (Jurenka 2003). In contrast, biosynthesis of type 2 occurs in specialized cells, oenocytes, that are associated with epidermal cells (Diehl 1975). The hydrocarbon pheromone of the arctiid moth, Holomelina aurantiaca, is biosynthesized in oenocytes and transported through the hemolymph to the pheromone gland for storage and release (Schal et al. 1998). In the lymantriid moth, Lymantria dispar, biosynthesis of the alkene precursor of the epoxide sex pheromone, occurs in oenocyte cells by chain elongation, desaturation, and decarboxylation. The alkene is transported to the pheromone gland and converted to (+)-disparlure by epoxidation (Jurenka et al. 2003). Polyene hydrocarbon precursors are present in the hemolymph of geometrid and arctiid moths, with the pheromone epoxide made in the pheromone gland (Wei et al. 2003, 2004).
Utetheisa ornatrix is broadly distributed over North America east of the Rockies and extends into South America, including Brazil, Argentina, and Chile (Pease 1968). Like many arctiid moths, U. ornatrix females produce a hydrocarbon pheromone and release it during the scotophase (Conner et al. 1980). The major sex pheromone component of U. ornatrix was identified as (Z,Z,Z)-3,6,9-heneicosatriene (3,6,9-21:Hy). This component attracted male moths in the field (Conner et al. 1980). Subsequently, however, a different composition of sex attractant was found in this species, varying geographically within populations, and from year to year. This blend consisted of 3,6,9-21:Hy along with (Z,Z,Z)-1,3,6,9-heneicosatetraene (1,3,6,9-21:Hy) as a minor component (Huang et al. 1983; Jain et al. 1983). Recently, (Z,Z,Z)-3,6,9-eicosatriene 3,6,9-20:Hy has also been identified as a minor component produced by females of this species (Lim et al. 2007).
The male sex pheromone and related mating behavior of U. ornatrix has been studied comprehensively. After being attracted to the female, through the long distance sex pheromone, the male courts the female by releasing hydroxydanaidal, derived from host plant pyrrolizidine alkaloids, obtained during larval feeding on seeds of Crotalaria spp. (Eisner and Meinwald 1995). The alkaloids are toxic, and female U. ornatrix may derive protection from predators by receiving these additional alkaloids during mating (Conner and Weller 2004). Because a female transfers some proportion of these alkaloids to her eggs, her offspring may also benefit (Iyengar and Eisner 1999; del Campo et al. 2005). Recent observations indicate that females appear to signal more vigorously in the presence of signaling neighbors, and they time their signaling in relation to their neighbors’ signaling activity. This phenomenon has been described as “female pheromonal chorusing” (Lim and Greenfield 2007). This behavior indicates that females detect female-produced pheromone, as previously demonstrated by gas chromatography-electroantennographic detection (GC-EAD) studies (Grant and O’Connell 2000; Lim et al. 2007).
In the present study, by using gas chromatography-mass spectrometry (GC-MS) and GC-EAD analyses, we determined that 1,3,6,9-21:Hy is the major pheromone component in most females of a U. ornatrix colony originally collected from a population in Florida. However, about 10% of females in our colony produced large amounts of 3,6,9-21:Hy and no C-21 tetraene. By using deuterium-labeled fatty acid and hydrocarbon precursors, we determined that biosynthesis of 1,3,6,9-21:Hy occurred by chain elongation of linolenic acid. We further determined that a brain factor, such as the pheromone biosynthesis activating neuropeptide (PBAN), is not directly involved in regulating pheromone production.
Methods and Materials
Insects
Colonies of U. ornatrix (Lepidoptera, Artiidae) were collected at the Archbold Biological Station (Highlands County, Florida, US). Larvae were reared on a pinto-bean diet mixed with seeds of the host plant, Crotalaria mucronata (Leguminoseae), as described (Dussourd et al. 1991) in an environmental chamber under a L:D14:10 at 26°C at the University of Kansas. Pupae were shipped to Iowa State University, where they were sexed and allowed to emerge separately. A sucrose solution (10%) was provided to adults. Virgin female adults or pupae were used throughout this study.
Chemicals
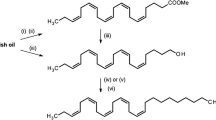
(6,6,7,7-D4)-9,12,15-octadecatrienoic (linolenic) acid (D4-9,12,15-18:acid) was synthesized as described (Rakoff 1988). (12,12,13,13-D4)-(Z,Z,Z)-3,6,9-heneicosatriene (D4-3,6,9-21:Hy) was synthesized from D4-9,12,15-18:acid, following procedures reported in Dogra et al. (1989). 1H-NMR: δ 0.88 (3H, t, J = 6.8 Hz, CH3, C21), 0.98 (3 H, t, J = 7.4 Hz, CH3, C1), 1.26–1.36 (18 H, m, CH2, C12–20), 2.04 (2 H, d, J = 6.8 Hz, CH2–C=, C11), 2.08 (2 H, m, CH2–C=, C2), 2.80 (4 H, t, J = 6.0 Hz, =C–CH2–C=, C5,8), 5.33–5.41 (6 H, m, CH=). 13C-NMR: δ 14.11, 14.26, 20.54, 22.69, 25.52, 25.60, 27.00, 29.30, 29.35, 29.60, 29.64, 29.65, 29.66, 29.68, 31.93, 127.12, 127.56, 128.23, 128.30, 130.42, 131.94. EI-MS m/z (%): 294 [M]+ (4), 265 (1), 238 (22), 184 (2), 170 (2), 149 (4), 135 (10), 121 (10), 108 (95), 95 (48), 93 (48), 79 (100), 67 (52), 55 (32). Synthetic Helicoverpa zea PBAN (Peninsula Laboratories, Belmont, CA, USA) was dissolved in an insect saline (21 mM KCl, 12 mM NaCl, 3 mM CaCl2, 18 mM MgCl2, 170 mM glucose, and 5 mM pipes buffer at pH 6.6 using KOH) before injection.
Incorporation and Hydrogenation Procedures
The putative pheromone precursors, D4-9,12,15-18:acid and D4-3,6,9-21:Hy, were used to determine the biosynthetic pathway by injection directly into the hemocoel of pupae. Deuterium-labeled compounds were dissolved in vegetable oil and injected into 1- to 2-d-old female pupae. To investigate the effect of PBAN, a saline solution with or without PBAN (10–50 pmol) was injected through abdominal intersegmental membranes into the hemocoel of 1- to 4-d-old decapitated females. Insects were incubated for 1 hr at room temperature, before the pheromone glands were removed, extracted, and analyzed for pheromones as described previously (Choi et al. 2002).
Hydrogenation of the unsaturated hydrocarbon pheromone, extracted from female pheromone glands, was conducted as follows. Hydrocarbon pheromone was purified by silica gel column chromatography. The purified hydrocarbon was dissolved in 100 μl of hexane in a glass bottle containing 10 mg palladium (5% wt on activated carbon; Sigma Chemical Co., St Louis, MO, USA). A hydrogen gas balloon covered the bottle for 2 hrs at room temperature before the hydrocarbon was extracted with hexane (100–200 μl), purified by silica gel column chromatography, and analyzed by gas chromatography (GC) and mass spectrometry (MS) as described below.
GC-EAD Analyses
For GC-EAD analysis, a Hewlett-Packard 5890 Series II GC, equipped with a DB-5 column (30 m × 0.25 mm ID, J & W Scientific, CA, USA) and a 50:50 effluent split, allowed simultaneous flame ionization detection (FID) and EAD of the gland extracts. The oven temperature program used was, 60°C for 1 min and increased to 250°C at 10°C min−1. Helium was the carrier gas. Gland extracts were concentrated to 10–20 μl, under a gentle stream of nitrogen, and 1 μl was injected into the GC column in splitless mode. The outlet for the EAD was supplied with purified, humidified air flowing over the antennal preparation at 600 ml min−1.
Whole-body preparations were used for EAD experiments. A virgin male or female moth was immobilized on a plasticine block with small U-shaped copper wires, and its antennae were restrained with thin copper wires. A glass microelectrode (tip diameter ≈ 1 mm, 0.86 mm I.D., A-M Systems Inc., WA, USA), filled with 0.1 M KCl, was introduced through the cuticle of the distal part of the antenna and served as a recording electrode; another glass microelectrode penetrated the moth’s compound eye to serve as a reference electrode. Silver–silver chloride junctions were used to maintain electrical contact between the electrodes and amplifier. The electroantennogram signal was first amplified with a high-input impedance (>10 Ω) headstage preamplifier (Syntech®, Hilversum, The Netherlands), and further processed with a PC-based signal processing system (Syntech®).
Analyses by GC and GC-MS
Glands were extracted with hexane (100 μl), containing 100 ng octadecane (18:Hy) as internal standard, for 10 min. Extracts were analyzed by GC or selected ion monitoring GC-MS. Incorporation of the labeled precursors into pheromones was determined by GC-MS with a Hewlett-Packard 5890 II mass selective detector coupled with a Hewlett-Packard 5890 GC equipped with a 30-m capillary column, DB-5 (30 m × 0.25 mm; J&W Scientific, CA, USA). The oven was programmed from 50°C for 1 min, to 300°C at 10°C min−1. The mass selective detector was used in the selected ion monitoring mode to detect the following characteristic m/z: 254 for octadecane (18:Hy), 276 for 3,6,9-20:Hy, 296 for heneicosane (21:Hy), 292 for 6,9-heneicoadiene, 290 for 3,6,9,-21:Hy, and 288 for 1,3,6,9-21:Hy. Labeled compounds were detected by using the characteristic ion +4. Adult females were extracted for pheromone and analyzed by GC-MS as described above.
Results and Discussion
Both 1,3,6,9-21:Hy and 3,6,9-21:Hy were detected in pheromone gland extracts of adults of all ages. The amounts of the two components showed a similar pattern with age between scotophase and photophase. Low amounts of both compounds were found in females during the late pupal stage (Fig. 1). The amount of 1,3,6,9-21:Hy varied greatly among individuals. The mean amount found in adult females was similar at all ages measured. However, the amount in 1-d-old females was greater than that in late pupae (Fig. 1B). The amount of 3,6,9-21:Hy (Fig. 1A) was similar for females of different age (including pupae), and roughly 1/30 that of 1,3,6,9-21:Hy (Fig. 1). These results indicate that sex pheromone biosynthesis starts in the late pupal stage, has increased by eclosion, and is maintained over at least 7 days. Thus, female adults are able to start calling and mate soon after emergence. This finding is consistent with previous work demonstrating that female adults are most active with respect to calling and mating at age 1- to 2-day-old (Conner et al. 1980; Itagaki and Conner 1987; Lim and Greenfield 2007).
GC analysis of pheromone gland extracts showed several peaks, including a very broad peak (Fig. 2B). Analysis indicated that all of these peaks, including the broad part of the chromatogram, had similar mass spectra, with the ion 288 prominent. The first peak was identified as 1,3,6,9-21:Hy based on comparison with published GC-MS data (Jain et al. 1983). This 21-carbon tetraene is unstable and the broad peak and peaks with longer retention times may result from thermolysis of this compound with rearrangement of double bonds into conjugated configurations, such as 2,3,6,9-21:Hy. Hydrogenation of the extract resulted in the detection of only saturated 21:Hy. The areas of the peaks with longer retention times were also reduced if the GC temperatures for the injector and oven were decreased (unpublished data), supporting their formation from thermolysis of 1,3,6,9-21:Hy.
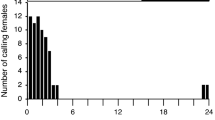
GC-EAD responses of male U. ornatrix to female pheromone gland extracts. Extracts were obtained from a female producing 3,6,9-21:Hy (a) and from one producing 1,3,6,9-21:Hy (b). In both a and b, the top traces are GC-FID signals and bottom traces EAD responses. EAD-active compounds in the extracts were identified by GC and GC-MS
3,6,9-21:Hy, was initially identified as the major pheromone component of U. ornatrix females (Conner et al. 1980). In the present study, however, most females (∼90%) produced about 1/30 the amount of this compound compared to 1,3,6,9-21:Hy. About 10% of female moths analyzed (several hundred) in our colony produced only a large amount (1,500–2,000 ng) of 3,6,9-21:Hy, without any tetraene (Fig. 2A). This result indicates that our colony may consist of two distinct pheromone types and that there may be two distinct populations present in the collection area in Florida. It has been noted previously that different geographical populations of U. ornatrix have different major and minor sex pheromone components (Conner et al. 1980; Huang et al. 1983; Jain et al. 1983). In their natural habitat, U. ornatrix host plants grow in highly populated patches. Presumably because of specificity to their hosts, U. ornatrix populations also show a clumped distribution. Such patchy distributions may allow for the formation of two pheromone types within a broader habitat. Further work is needed to determine the distributions of these two pheromonal types in populations in North and South America (Pease 1968).
Decapitated female moths appeared to accumulate 3,6,9-21:Hy and 1,3,6,9-21:Hy compared to same age normal females. Females, decapitated at 1-d-old, had roughly 3,300 ng of 1,3,6,9-21:Hy when 4-d old (Fig. 3), whereas 4-d-old intact females had roughly 1,000 ng of 1,3,6,9-21:Hy (Fig. 1). This result suggests that decapitated female moths continued to produce pheromone from oenocyte cells for transport to the pheromone gland, but probably did not release pheromone, as did intact females, due to the disconnect of the abdominal ganglia from the brain. The pheromone gland of female U. ornatrix consists of a long pair of slender tubes that extend forward to the sixth abdominal segment (Conner et al. 1980). Control of U. ornatrix rhythmic pheromone gland exposure by the terminal abdominal ganglion has previously been demonstrated (Itagaki and Conner 1987).
Although female U. ornatrix produce relatively large amounts of 1,3,6,9-21:Hy, only trace amounts were detected in the hemolymph during scotophase and photophase (unpublished data). This could be explained in several ways. First, transport of the hydrocarbon from the oenocytes to the pheromone gland may occur at a different time from when the hemolymph was collected. Second, the oenocyte cells are closely associated with the pheromone gland, and thus, transport through the hemolymph may not occur. Third, it is also possible that the pheromone gland directly produces the hydrocarbon pheromones. Further research will deduce which is the case.
A PBAN-like neuropeptide does not appear to influence hydrocarbon sex pheromone production in U. ornatrix, as indicated by similar amounts of pheromone being found in decapitated females injected with either synthetic PBAN or saline (Fig. 3) and the increase in hydrocarbon quantities after decapitation discussed previously. PBAN is also not involved in hydrocarbon pheromone production in the noctuid moth, Anticarsia gemmatalis, (Jeong 2003). In other species of moths producing type 2 pheromones, PBAN regulates epoxidation after the uptake of hydrocarbons in the pheromone gland (Ando et al. 1997; Jurenka et al. 2003). Recently, it was shown that PBAN could regulate the uptake of hydrocarbon into pheromone glands of geometrid moths (Wei et al. 2004).
Fig. 4 shows results obtained from GC-MS analysis of pheromone gland extracts in which females were injected with labeled precursors. When D4-9,12,15-18:acid was injected into the early pupal stage, the most abundantly labeled hydrocarbons in U. ornatrix adults were 3,6,9-21:Hy and 1,3,6,9-21:Hy. This indicates that 3,6,9-21:Hy could be biosynthesized by chain elongation and decarboxylation of linolenic acid. Typically, hydrocarbon pheromone biosynthesis occurs through modification of fatty acid metabolic pathways (Jurenka 2003). Labeling of the major pheromone component, 1,3,6,9-21:Hy, is of greater interest because it could be produced through two possible pathways based on when the terminal desaturation occurs (Fig. 5). To determine which pathway occurs, D4-3,6,9-21:Hy was injected into pupae. No label was incorporated in 1,3,6,9-21:Hy, although a large amount of labeled triene, 3,6,9-21:Hy, was recovered. This suggests that U. ornatrix females do not use 3,6,9-21:Hy to produce 1,3,6,9-21:Hy. To determine when the terminal desaturation occurs in the biosynthetic pathway, additional studies will require the synthesis and injection of various chain-length precursors.
Representative partial mass chromatograms obtained from GC-MS analysis of hexane extracts of pheromone glands of U. ornatrix after injection of 0.5 μl vegetable oil or 0.5 μl vegetable oil containing 50 μg D4-9,12,15-18:acid into female pupae. The top two chromatograms were from females producing 3,6,9-21:Hy, and the bottom two chromatograms were from females producing predominantly 1,3,6,9-21:Hy. The GC-MS was set in the selective ion mode to detect m/z 288, 290, 292, and 294. Ions 292 and 294 are shown in bold and represent the label incorporation from the deuterium-labeled substrates
References
Ando, T., Ohtani, K., Yamamoto, M., Miyamoto, T., Qin, X.-R., and Witjaksono. 1997. Sex pheromone of Japanese giant looper, Ascotis selenaria cretacea: identification and field tests. J. Chem. Ecol. 23:2413–2423.
Byer, J. A. 2006. Pheromone component patterns of moth evolution revealed by computer analysis of the Pherolist. J. Anim. Ecol. 75:399–407.
Choi, M. Y., Han, K. S., Boo, K. S., and Jurenka, R. A. 2002. Pheromone biosynthetic pathways in the moths Helicoverpa zea and Helicoverpa assulta. Insect Biochem. Mol. Biol. 32:1353–1359.
Conner, W. E. and Weller, S. J. 2004. A quest for alkaloids: the curious relationship between tiger moths and plants containing pyrrolizidine alkaloids, pp. 248–282, in R. T. Carde and J. G. Millar (eds.). Advances in Insect Chemical Ecology. Cambridge University Press, Cambridge, UK.
Conner, W. E., Eisner, T. Vander Meer, R. K., Guerrero, A., Ghiringelli, D., and Meinwald, J. 1980. Sex attractant of an arctiid moth (Utetheisa ornatrix): a pulsed chemical signal. Behav. Ecol. Sociobiol. 7:55–63.
Del Campo, M. L. Smedley, S. R., and Eisner, T. 2005. Reproductive benefits derived from defensive plant alkaloid possession in an arctiid moth (Utetheisa ornatrix). Proc. Natl. Acad. Sci. U S A 102:13508–13512.
Diehl, P. A. 1975. Synthesis and release of hydrocarbons by the oenocytes of the desert locust, Schistocera gregaria. J. Insect Physiol. 21:1237–1246.
Dogra, V., Sabharwal, A., Sharma, S., Hug, M. A., Kad, G. L., and Vig, O. P. 1989. Synthesis of highly unsaturated insect sex pheromones. Synthesis of (3Z,6Z,9Z)-3,6,9-nonadecatriene and (3Z,6Z,9Z)-3,6,9-heneicosatriene. J. Indian Chem. Soc. 66:169–171.
Dussourd, D. E., Harvis, C. A., Meinwald, J., and Eisner, T. 1991. Pheromonal advertisement of a nuptial gift by a male moth (Utetheisa ornatrix). Proc. Natl. Acad. Sci. U S A 88:9224–9227.
Eisner, T. and Meinwald, J. 1995. The chemistry of sexual selection. Proc. Natl. Acad. Sci. U S A 92:50–55.
Grant, A. J., and O’connell, R. J. 2000. Responses of olfactory receptor neurons in Utetheisa ornatrix to gender-specific odors. J. Comp. Physiol. A 186:535–542.
Huang, W., Pulaski, S. P., and Meinwald, J. 1983. Synthesis of highly unsaturated insect pheromones: (Z,Z,Z)-1,3,6,9-heneicosatetraene and (Z,Z,Z)-1,3,6,9-nonadecatetraene. J. Org. Chem. 48:2270–2274.
Itagaki, H. and Conner, W. E. 1987. Neural control of rhythmic pheromone gland exposure in Utetheisa ornatrix (Lepidoptera: Arctiidae). J. Insect Physiol. 33:177–181.
Iyengar, V. K. and Eisner, T. 1999. Female choice increases offspring fitness in an arctiid moth (Utetheisa ornatrix). Proc. Natl. Acad. Sci. U S A 96:15013–15016.
Jain, S. C., Dussourd, D. E., Conner, W. E., Eisner, T. Guerrero, A., and Meinwald, J. 1983. Polyene pheromone components from an arctiid moth (Utetheisa ornatrix): characterization and synthesis. J. Org. Chem. 48:2266–2270.
Jeong, H.-S. 2003. Hydrocarbon pheromone biosynthesis of female velvetbean caterpillar moth, Anticarsia gemmatalis. MS dissertation. Iowa State University, Ames.
Jurenka, R. A. 2003. Biochemistry of female moth sex pheromones, pp. 53–80, in G. Blomquist and R. Vogt (eds.). Insect Pheromone Biochemistry and Molecular Biology. Academic Press, New York.
Jurenka, R. A., Subchev, M., Abad, J. L., Choi, M. Y., and Fabrias, G. 2003. Sex pheromone biosynthetic pathway for disparlure in the gypsy moth, Lymantria dispar. Proc. Natl. Acad. Sci. U S A 100:809–814.
Lim, H. and Greenfield, M. D. 2007. Female pheromonal chorusing in an arctiid moth, Utetheisa ornatrix. Behav. Ecol. 18:165–173.
Lim, H., Park, K. C., Baker, T. C., and Greenfield, M. D. 2007. Perception of conspecific female pheromone stimulates female calling in an arctiid moth, Utetheisa ornatrix. J. Chem. Ecol. (in press).
Millar, J. G. 2000. Polyene hydrocarbons and epoxides: a second major class of lepidopteran sex attractant pheromones. Annu. Rev. Entomol. 45:575–604.
Pease, R. W. J. 1968. Evolution and hybridization in the Utetheisa ornatrix complex (Lepidoptera: Arctiidae). I. Inter- and intra-population variation and its relation to hybridization. Evolution 22:719–735.
Rakoff, H. 1988. Preparation of methyl cis-9,cis-12,cis-15-octadecatrienoate-15,16-D2 and methyl cis-9,cis-12,cis-15-octadecatrienoate-6,6,7,7-D4. Lipids 23:280–285.
Schal, C., Sevala, V., and Carde, R. T. 1998. Novel and highly specific transport of a volatile sex pheromone by hemolymph lipophorin in moths. Naturwissenschaften 85:339–342.
Wei, W., Miyamoto, T., Endo, M., Murakawa, T., Pu, G.-Q., and Ando, T. 2003. Polyunsaturated hydrocarbons in the hemolymph: biosynthetic precursors of epoxy pheromones of geometrid and arctiid moths. Insect Biochem. Mol. Biol. 33:397–405.
Wei, W., Yamamoto, M., Asato, T., Fujii, T., Pu, G.-Q., and Ando, T. 2004. Selectivity and neuroendocrine regulation of the precursor uptake by pheromone glands from hemolymph in geometrid female moths, which secrete expoxyalkenyl sex pheromones. Insect Biochem. Mol. Biol. 34:1215–1224.
Witzgall, P., Lindblom, T. Bengtsson, M., and Toth, M. 2004. The Pherolist. ( http://www-pherolist.slu.se/pherolist.php ).
Acknowledgements
This research was, in part, supported by the Iowa Agriculture and Home Economics Experiment Station, Ames, Iowa, project 6698.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, MY., Lim, H., Park, K.C. et al. Identification and Biosynthetic Studies of the Hydrocarbon Sex Pheromone in Utetheisa ornatrix . J Chem Ecol 33, 1336–1345 (2007). https://doi.org/10.1007/s10886-007-9306-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-007-9306-1