Abstract
Biological neurons keep sensitive to external stimuli and appropriate firing modes can be triggered to give effective response to external chemical and physical signals. A piezoelectric neural circuit can perceive external voice and nonlinear vibration by generating equivalent piezoelectric voltage, which can generate an equivalent trans-membrane current for inducing a variety of firing modes in the neural activities. Biological neurons can receive external stimuli from more ion channels and synapse synchronously, but the further encoding and priority in mode selection are competitive. In particular, noisy disturbance and electromagnetic radiation make it more difficult in signals identification and mode selection in the firing patterns of neurons driven by multi-channel signals. In this paper, two different periodic signals accompanied by noise are used to excite the piezoelectric neural circuit, and the signal processing in the piezoelectric neuron driven by acoustic waves under noise is reproduced and explained. The physical energy of the piezoelectric neural circuit and Hamilton energy in the neuron driven by mixed signals are calculated to explain the biophysical mechanism of auditory neuron when external stimuli are applied. It is found that the neuron prefers to respond to the external stimulus with higher physical energy and the signal which can increase the Hamilton energy of the neuron. For example, stronger inputs used to inject higher energy and it is detected and responded more sensitively. The involvement of noise is helpful to detect the external signal under stochastic resonance, and the additive noise changes the excitability of neuron as the external stimulus. The results indicate that energy controls the firing patterns and mode selection in neurons, and it provides clues to control the neural activities by injecting appropriate energy into the neurons and network.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
A simple neuron model is helpful to discern and predict the dynamical properties of neurons. For better understanding, the biophysical property and mechanism of signal processing, the effect of ion channels, electromagnetic induction, energy flow, synaptic plasticity, and controllability in coupling synapses should be considered in the biological and biophysical neuron models. Artificial neuron models often develop from the neural circuits composed of capacitor, inductor, nonlinear resistor, and specific electric components such as memristors, thermistors, phototubes, Josephson junctions, and piezoelectric ceramics. For example, a piezoelectric ceramic can be connected to a simple neural circuit for discerning external voice and mechanic vibration, and this neural circuit can be mapped into a piezoelectric neuron for exploring the dynamics of auditory neuron. The auditory cortex is the auditory center of the brain. The inner ear sends electrical signals to this area of the brain to receive external sounds. According to the changes of sound environment, the adaptability of neurons can be used to adjust the sound processing process. Even in a noisy environment, the human ear can detect and distinguish the subtle differences of sound, and has the ability to accurately capture a specific sound in complex dialogue [1,2,3,4,5]. Recent research in the field of neuroscience has shown that the brain can decode special auditory signals in complex environments, which is called auditory attention detection (AAD); it opens up new opportunities for the cognitive control of auditory prostheses such as hearing aids and cochlear implants [6,7,8,9]. For example, Mesgarani et al. [1] obtained invasive recordings from the primary and nonprimary auditory cortex (AC) in neurosurgical patients as they listened to multi-talker speech, and they revealed the neural computations underlying the hierarchical formation of auditory objects in human AC during multi-talker speech perception. Xu et al. [10] investigated how sensory-to-category transformation is implemented by cortical neurons during a stimulus categorization task, and their results uncovered a task-dependent dynamic reorganization of cortical response patterns serving as a neural mechanism for sensory-to-category transformation during perceptual decision-making. The detection and discrimination of input spatiotemporal sequence by neurons is the basis of brain function and human brain perception, cognition and motor output. Ref. [11] has realized the simulation of human ear sound azimuth discrimination by constructing a simple dual input/dual output coupled artificial neural network. Li et al. [12] investigated the neural mechanisms of speech comprehension in noise using a functional near-infrared spectroscopy-based inter-brain approach, and they claimed that speaker–listener coupling analysis provides added value and new sight to understand the neural mechanism of speech-in-noise comprehension. From physical aspect, Zhou et al. [13] suggested that a piezoelectric ceramic can be embedded into a simple neural circuit, and then external sound and acoustic wave can be perceived to trigger appropriate firing patterns. In particular, Guo et al. [14] explained the biophysical mechanism for wave filtering of acoustic wave imposed on the piezoelectric neural circuit and auditory neuron. In addition, this kind of frequency selection and wave filtering can be used to understand why human eyes are sensitive to certain frequency band as visible light [15].
Noise is often applied on nonlinear oscillators and network for estimating the effect of stochastic disturbance and uncertain stimuli. For isolated oscillator, distinct regular oscillation can be developed under nonlinear resonance by taming noise with appropriate intensity, and periodic stimulus can also be effective to control the chaotic oscillation, and regular behavior is obtained due to the effect of forced vibration [16,17,18,19]. Synchronization stability between nonlinear oscillators can be approached under noise even no coupling is applied, which is classified as resonance synchronization [20,21,22,23,24]. For neural circuits and biological neurons, noise is often considered as uncertain disturbance resulting from electromagnetic radiation, and then noisy term can be applied to regulate the membrane potential and channel current as well. Additive noise on membrane potential behaves similar role as external electric forcing, and the excitability of neuron is changed to regulate the firing patterns; also, external energy is injected. One important application of stochastic resonance is its signal detection [25,26,27,28]; maximal information propagation is obtained, and strong subthreshold periodic driving signal can make the system have high peak signal-to-noise ratio (SNR). In the nervous system with multi periodic signal inputs, under certain noise intensity, a single neuron driven by the mixed periodic signal of fundamental frequency and harmonic frequency has the greatest response to the fundamental frequency signal [20, 29]. When noise driving and electromagnetic radiation are imposed on the excitable media and neural circuits synchronously, most auditory neurons still can discern and encode external periodic signals, which makes auditory neurons show regular discharge patterns [30,31,32,33,34]. In practical way, many simple neural circuits can be improved to enhance the potential biophysical functions by incorporating a memristor, Josephson junction, phototube, piezoelectric ceramic, or thermistor into any of the branch circuit. For example, the involvement of memristor into the neural circuit can estimate its effect of electromagnetic induction and build memristive synapse [35,36,37,38,39]. The connection of Josephson junction to neural circuit can discern the effect of external magnetic field [40,41,42]. The phototube activated by external illumination can be used to excite neural circuit and the signal processing of artificial eyes can be reproduced [43,44,45]. The neural circuit coupled by thermistor can perceive the changes of temperature, and thermosensitive neuron can be obtained [46,47,48]. In particular, Yao et al. [49] built a multi-functional neural network composed of piezoelectric neuron, thermosensitive neuron, and light-sensitive neuron, and field coupling is activated to study the collective behaviors for discerning the cooperation mechanism of neurons from different functional regions of the brain. That is, these electric components can perceive external physical signals by converting external stimuli including noise into equivalent currents, and its physical mechanism is considered as energy injection and energy encoding.
In fact, biological neurons and artificial neural circuits can perceive and receive multi-channel inputs, and the competitive cooperation enables appropriate mode selection and firing patterns. For the auditory system, neurons can discern specific frequency band of acoustic waves except those distinct periodic signals even in presence of noise [50]. In this paper, based on the known piezoelectric neuron, more than two periodic signals are imposed on the neural circuit composed of piezoelectric ceramics; the average power of inputs and intrinsic Hamilton energy of the functional neuron are estimated to discuss the response mechanism of neuron driven by multi-channel stimuli in noisy condition. It predicts that the neuron prefers to give smart response to the external stimulus with higher average power, and similar stochastic resonance can be induced when the external noise is tamed carefully. The external signals are considered as periodic signals, chaotic signals for reproducing realistic signals covering acoustic wave within certain frequency band; it seems that the neuron used to respond to the signals injecting more energy, and thus neural activities can be guided and regulated to present most suitable firing patterns. Each biological neuron can be considered as a charged body and external electromagnetic field will change the static distribution of intracellular and extracellular ions, and also the channel current will be regulated as well. When external electromagnetic field is fluctuated, stochastic disturbance will be induced. The neural circuits are often sensitive to external magnetic field. For piezoelectric ceramics, external acoustic wave and mechanic vibration seldom are periodic type and certain stochastic disturbance is accompanied. Therefore, the filtered wave and signals in this neural circuit coupled by piezoelectric ceramics are approached by using periodic signal and certain noise. Additive noise is imposed, and the excitability is changed rather than ion channels, which are sensitive to channel noise.
2 Model and scheme
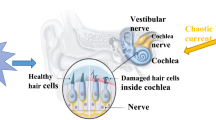
Within artificial neurons and neural circuits, external realistic signals are captured and converted into equivalent current/voltage for exciting the generic neurons, and the firing mode becomes distinct and unique in the firing patterns without discerning the difference in external stimuli. For standard periodic signal and constant signal, most of the firing modes can present distinct characteristics and firing patterns to give response to certain external stimulus. However, the biological neurons can be able to discern the main information and discard the secondary information even in noisy condition when more signal sources are used to excite the neurons. During the electric response, external stimulus can input energy and the inner physical field energy in the neurons can be changed effectively. In fact, the energy absorption of energy in the neuron is nonlinear rather than monotonous way because the external forcing is encoded by a nonlinear processing in the neuron. On the other hand, biological neurons can keep robust to some external stimulus under wave filtering and its firing patterns are dull to the external forcing signals as shown in Fig. 1.
As shown in Fig. 1, the auditory neurons can discern the difference between more than two acoustic waves even in noisy condition by inducing sensitive spatiotemporal patterns and firing modes in the neural activities. Therefore, it is important to explore the potential physical mechanism for response to external stimuli in presence of noise. For simplicity, two external signal sources accompanied by Gaussian white noise are imposed on a piezoelectric neural circuit, which is used to reproduce the biophysical property of auditory neuron and it is shown in Fig. 2.
Schematic diagram for piezoelectric neural circuit driven by more than two acoustic waves in presence of Gaussian white noise. NR represents a nonlinear resistor, PC denotes a piezoelectric ceramic, C, L and E describe the capacitor, induction coil, and constant voltage source, respectively. Rs and R are linear resistors and the constant E is used to describe the reversal potential of the ion channel
In generic way, the current iNR across the nonlinear resistor NR is often approached by
where V0, ρ and V denote the cut-off voltage in the i-V curve for NR, normalized parameter for resistance in the linear region, and voltage across the NR, respectively. As reported in Ref. [13], the output voltage VPC from the piezoelectric device can be described by
where F, S, d33, ε, and h represent the external mechanical force, surface area, piezoelectric strain constant, dielectric constant, and thickness of the piezoelectric ceramic, respectively. By applying surface coating, the piezoelectric ceramic is effective to filter and absorb external mechanic vibration and acoustic wave within certain frequency band and amplitudes. As a result, the piezoelectric ceramic behaves as a voltage source for exciting the neural circuit, and a piezoelectric neuron is obtained to simulate the dynamical property and signal processing as auditory neuron effectively after scale transformation. That is, the output voltage across the piezoelectric ceramic is mainly dependent on the external force and the intrinsic parameter μ. When the piezoelectric device is selected, the output voltage is controlled by external acoustic wave and noise as well. According to the known Kirchhoff theorem, the circuit equations for the piezoelectric neural circuit in Fig. 2 can be obtained by:
where the variable V and iL remark the output voltage across the capacitor and channel current across the induction coil, respectively. For further nonlinear analysis on the sampled time series, standard scale transformation is applied on the physical variables and parameters in Eq. (3), and it is defined by:
As a result, the piezoelectric neural circuit for Eq. (3) is replaced by a functional auditory neuron as follows:
where the variable x and y represents the membrane potential and recovery variable for channel current, respectively. Most of the parameters (a, b, c, ξ) are adjustable because they are mapped from the physical parameters for capacitance, inductance, resistance, and constant voltage. Under specific condition, the electric signal converted from acoustic wave via piezoelectric ceramics can be selected with distinct periodic signal or combination of finite periodic stimuli. In noisy condition, the equivalent electric signal ξupc filtered from the acoustic wave can be approached by periodic signals and stochastic disturbance, which can be considered from the disturbance of electric field and imposed on the membrane potential as additive noise. The realistic voice can generate equivalent VPC across the piezoelectric device under piezoelectric conversion and its form is controlled by external physical signal and the material property of the piezoelectric device connected to the neural circuit. The filtered acoustic wave is mapped into equivalent piezoelectric current ξupc, which can be selected as single periodic signal or mixed signals composed of periodical signals and noise, and then the neural activities can be controlled completely.
where the statistical property of the Gaussian white noise ζ(τ) with zero average value can be estimated as < ζ(τ) > = 0.0, < ζ(τ) ζ(s) > = Dδ(τ − s), D is the noise intensity and δ(*) represents Dirac-δ function. A1, A2 and f1, f2 define the amplitude and frequency in the piezoelectric current/voltage after piezoelectric conversion, respectively. When the piezoelectric neuron is driven by a single periodic signal, it presents a variety of firing modes by changing the amplitude or frequency of the external stimulus carefully, and the neuron will present distinct firing patterns when all parameters are fixed. In case of ξupc3, two different periodic signals are imposed synchronously, in which external stimuli are used to excite the neuron from different channels. It is interesting to predict and confirm that the final firing mode and patterns will be dependent on which external stimulus. In addition, this question becomes more interesting in presence of noise.
As is known, the physical field energy EN is kept in the electric components such as capacitor and induction coil, and the similar dimensionless field H also exists in the biological neurons as well.
That is, the field energy in the neural circuit driven by piezoelectric current from the piezoelectric ceramic can be estimated by the equivalent Hamilton energy H, which is dependent on the membrane potential, channel current, and one intrinsic parameter as well. As a result, the firing mode is changed by external forcing and then the Hamilton energy is regulated synchronously. From physical viewpoint, external stimulus and forcing can inject energy into the neuron, and then the firing modes can be controlled effectively. Within an eigencycle T, the average power of a periodic signal is estimated by
where A is the amplitude of the external signal, and the average power becomes a constant. In the presence of noise, the signal-to-noise ratio (SNR) is often estimated to predict the occurrence of stochastic resonance, and it is calculated by applying fast Fourier transform (FFT) on the sampled time series for any variables. The SNR curve can be obtained by changing the noise intensity, and each value for SNR can be obtained by finding the following values (h, ωp, Δω) in the power spectrum for the membrane potentials.
where h means the maximum value of power spectrum peak, and ωp and Δω define the frequency associated with the peak value of power, the half height width of the highest peak of the power spectrum. S and B denote the peak value and fundamental frequency of the power spectrum. Besides the periodic and mixed signals defined in Eq. (6), the realistic acoustic wave can be as signals with wide frequency band, for example, chaotic signals from the Chua system [51, 52] are shown in Eq. (10).
where α, β, γ are normalized parameters, and m0 and m1 are normalized parameters for the conductance in the curve i-v for the Chua diode. The nonlinear function f(x′) describes the dimensionless current across the nonlinear resistor (diode) of the Chua circuit. By setting appropriate parameters, the Chua system can present chaotic state, periodic state, and then the first variable x′ will be used as realistic signal source for checking the sensitivity of the neural circuit. In practical way, surface coating of piezoelectric devices is helpful and effective to filter the realistic acoustic wave like quasi-periodic and chaotic signals, and thus only finite frequency band and amplitude can be converted into equivalent electric signal for exciting the neural circuit, and thus external acoustic wave is detected and discerned effectively.
3 Numerical results and discussion
In this section, the standard fourth order Runge–Kutta algorithm is applied to find numerical solutions of the dynamical systems, the transient for calculation is about 2000 time units, and the time step is fixed at 0.01. The initial values for variables in Eq. (4) are fixed at (x0,y0) = (0.2, 0.1), and then initial values (x0′, y0′, z0′) = (0.01, 0.1, 1.0) are used to trigger chaotic series from Chua system shown in Eq. (10). As presented in Eq. (6) and Eq. (8), the average power values for three kinds periodic signals are obtained by < P1 > = 0.0338, < P2 > = 0.04805, < P3 > = 0.57245. In Fig. 3, the evolution of membrane potential and firing patterns, Hamilton energy, and the SNR under noise are calculated, respectively.
Evolution of firing patterns, Hamilton energy, SNR, and ISI bifurcation in the piezoelectric neuron driven by periodic signals. For (a), F1 is applied with A1 = 0.26, f1 = 0.015, then F1 is applied and noise intensity is changed for getting SNR; (b) F2 is applied with A2 = 0.31, f2 = 0.015, then F2 is applied and noise intensity is changed for getting SNR; (c) F3 is applied with A3 = 1.07, f3 = 0.015, then F3 is applied and noise intensity is changed for getting SNR. The parameters are selected as a = 0.7, b = 0.8, c = 0.1, and ξ = 0.175, and the initial values are selected as (0.2, 0.1) in Eq. (5). The inserted subfigure shows the formed attractors under the same parameter setting
The firing patterns are regulated by changing the external stimulus, and the Hamilton energy is also changed. The average Hamilton energy is increased when higher amplitude in the external forcing is applied because more energy can be injected and captured by the neuron. When the firing patterns show distinct periodicity, periodic attractors are formed synchronously. Similar stochastic resonance (SR) is induced and peak value is detected in the power spectrum by changing the noise intensity carefully. In particular, the optimal noise intensity tends to be a lower value for inducting stochastic resonance when the neuron is excited by external stimulus with higher power value, and higher Hamilton energy is obtained as well. It indicates the occurrence of SR becomes easier because injection of more energy into the neuron. That is, distinct periodic signal can be effectively discerned and identified in presence of noise when SR is induced. As a result, stronger noise will induce bursting and even chaotic patterns. It is interesting to discern the main contribution and role in regulating the Hamilton energy and firing mode in the neural activities when two different periodic stimuli are applied for exciting the neuron. Firstly, we consider the case that two external stimuli show slight diversity in the average power, which the external stimulus F is composed of two periodic signals with close average power, and the results are calculated in Fig. 4.
(a) Evolution of membrane potential and Hamilton energy in the neuron driven by two periodic signals (F1 + F2) synchronously in absence of noise. (b) SNR distribution and ISI bifurcation under noise. (c) Firing patterns and Hamilton energy under noise D = 3. The parameters are selected as a = 0.7, b = 0.8, c = 0.1, ξ = 0.175, A1 = 0.26, f1 = 0.015, A2 = 0.31, and f2 = 0.015, and the initial values are selected as (0.2, 0.1) in Eq. (5). The inserted subfigure shows the formed attractors under the same parameters setting
When external forcing F1 and F2 are combined and applied to excite the neuron synchronously, higher Hamilton energy is obtained by capturing more energy from the external stimuli. Similar SR is induced and the peak value for power occurs at lower noise intensity. In case of periodic firing, the attractors become periodic and the involvement of noise will change the profile of the attractors. In case of SR state, the neuron tends to present regular firing and oscillation and average Hamilton energy is obtained. Compared the profile of the attractors, it is found that the neuron driven by combined signals prefers to show similar firing patterns induced by F2 with higher average power. It indicates that the neuron may give response to the external stimulus with higher energy injection and power value. It is worthy of investigating the signal identification of two periodic signals with higher diversity in power, and the results are estimated in Fig. 5.
(a) Evolution of membrane potential and Hamilton energy in the neuron driven by two periodic signals (F1 + F3) synchronously in absence of noise. (b) SNR distribution and ISI bifurcation under noise. (c) Firing patterns and Hamilton energy under noise D = 17. The parameters are selected as a = 0.7, b = 0.8, c = 0.1, ξ = 0.175, A1 = 0.26, f1 = 0.015, A2 = 1.07, and f2 = 0.015, and the initial values are selected as (0.2, 0.1) in Eq. (5). The inserted subfigure shows the formed attractors under the same parameters setting
Compared with the case for neuron driven by single periodic stimulus, the Hamilton energy is further increased when more periodic stimuli are applied because more energy is injected and encoded. When noise is applied, periodic attractor is corrupted to induce a chaotic attractor. Higher noise intensity is required to induce SR when the periodic stimuli are held with higher power. In case of SR, regular firing patterns are induced and kept in the neural activities. In addition, the firing mode is mainly decided and controlled by the external forcing F3 with higher power because the threshold of noise intensity for inducing SR is the same as the value for neuron driven by single periodic signal F3. That is, the external stimulus with higher power can be identified and perceived more sensitively than other stimuli with lower power values. It is interesting to discuss the response in the neural activity of neuron driven by chaotic signal, which is derived from the Chua circuit and average power value < P4 > = 0.023, and the results are shown in Fig. 6.
Evolution of firing patterns (a); Hamilton energy (b); SNR (c); and ISI bifurcation (d) in the auditory neuron driven by chaotic signals under noise. The parameters are fixed at α = 10, β = 16, γ = 0.01, m0 = − 1.296, m1 = − 0.7364, a = 0.7, b = 0.8, c = 0.1, ξ = 0.175, A1 = 0.26, and f1 = 0.015, and initial values are selected as (0.2, 0.1) in Eq. (5), and (0.01, 0.1, 1.0) in Eq. (10). The inserted subfigure shows the formed attractors under the same parameters setting
It is found that the neuron is forced to present chaotic behavior when external chaotic stimulus is applied, and the average Hamilton energy is decreased to lower value when chaotic attractor is formed. In addition, similar SR is induced in the neural activities when noise is tamed carefully in presence of chaotic driving, and SNR reaches the peak value at noise intensity D = 58. It is interesting to discuss the identification between periodic signal (F1, < P1 > = 0.0338) and chaotic signal (F4, < P4 > = 0.023), and the results are plotted in Fig. 7.
(a) Evolution of membrane potential and Hamilton energy in the neuron driven by periodic and chaotic signals (F1 + F4) synchronously in absence of noise. (b) SNR distribution and ISI bifurcation under noise. (c) Firing patterns and Hamilton energy under noise D = 90. The parameters are fixed at α = 10, β = 16, γ = 0.01, m0 = − 1.296, m1 = − 0.7364, a = 0.7, b = 0.8, c = 0.1, ξ = 0.175, A1 = 0.26, and f2 = 0.015, and initial values are selected as (0.2, 0.1) in Eq. (5), and (0.01, 0.1, 1.0) in Eq. (10). The inserted subfigure shows the formed attractors under the same parameters setting
It is found the final firing mode and patterns are mainly controlled by the chaotic forcing when the average power in the periodic stimulus is lower as the average power in the chaotic driving, and then the firing patterns show distinct chaotic behaviors. When noise intensity is increased, chaos in the formed attractor and firing patterns is further enhanced and the neuron tends to keep lower Hamilton energy. That is, the periodic signal is difficult to be discerned and it is covered by the chaotic forcing and the Hamilton energy in the auditory neuron is further decreased. The SNR reaches a peak value at noise intensity D = 90, the regularity in the firing patterns is enhanced at this noise intensity, and it is similar to the case for neuron driven by single periodic signal F1. The neuron prefers to select the chaotic firing mode induced by F4, and the periodic stimulus is covered by the chaotic forcing. It is interesting to discuss the case when periodic signal with higher power value (F3, < P3 > = 0.572) and chaotic stimulus (F4, < P4 > = 0.023) are applied to excite the neuron, and the signal identification is shown in Fig. 8.
(a) Evolution of membrane potential and Hamilton energy in the neuron driven by periodic and chaotic signals (F3 + F4) synchronously in absence of noise. (b) SNR distribution and ISI bifurcation under noise. (c) Firing patterns and Hamilton energy under noise D = 17. The parameters are fixed at α = 10, β = 16, γ = 0.01, m0 = − 1.296, m1 = − 0.7364, a = 0.7, b = 0.8, c = 0.1, ξ = 0.175, A3 = 1.07, and f2 = 0.015, and initial values are selected as (0.2, 0.1) in Eq. (5), and (0.01, 0.1, 1.0) in Eq. (10). The inserted subfigure shows the formed attractors under the same parameters setting
The involvement of chaotic stimulus can decrease the Hamilton energy in the neuron, and neuron prefers to show similar periodic firing patterns as the case for neuron driven by single periodic signal F3. Similar SR occurs at the noise intensity D = 17, which is also effective to induce SR in the neuron driven by sole periodic signal F3. In particular, the Hamilton energy shows slight increase under the SR state.
In a summary, the external stimuli contain different powers and energy scales and certain energy is injected and encoded by the neuron. The neuron prefers to give sensitive response to the external stimulus containing higher energy or power, and then the energy is absorbed to control the firing modes effectively. In noisy condition, the neuron can be induced with similar SR when two different external stimuli are applied. The neuron often shows lower Hamilton energy when the neuron is forced to present chaotic behaviors, but similar SR can still be induced by applying appropriate noise intensity. When external stimuli are composed of periodic and chaotic signals with close power, the periodic firing mode is corrupted to show chaotic behaviors and identification of weak periodic signal becomes difficult. Above all, we discussed and clarified the potential response criterion for isolated neuron driven by more than two periodic signals, and it is said that neuron gives preference to external inputs with higher power and energy even noise is considered. When two or more neurons are clustered in the same region or area, the energy diversity between neurons will enable the creation of synaptic connections for reaching possible energy balance [53,54,55]. As mentioned in Ref. [56,57,58,59,60], estimation of Hamilton energy and control the energy flow are much effective to control the synchronous behaviors of chaotic systems and neurons. Therefore, dynamical response in the neural activities of neurons is also controlled by the energy injection and absorption, and thus neurons can give appropriate mode selection and response to different external stimuli. Biological neurons show distinct intelligence with handling the signals processing, and each neuron in the neural network can perceive any external stimulus and then its intrinsic energy is regulated to create appropriate synapse connection to adjacent neurons without preferring the energy level in the inputs with higher value. That is, neurons can perceive external stimulus randomly and the increase of intrinsic Hamilton energy will be shared and kept balance when appropriate links are created via chemical or memristive synapse connections to adjacent neurons.
4 Conclusions
A realistic sound signal or acoustic wave often contains distinct wave band, and it can be perceived and discerned by neurons only when it has distinct power value than other signal sources. An external stimulus can inject certain energy into the neuron or neural circuit, and the increase of the Hamilton energy will regulate the dynamics of the forced neural circuit or excited neuron. When two different signal sources are applied to excite the neuron from different channels, the inner field energy of neuron is tamed and the neuron prefers to show firing mode and energy value as the case for neuron controlled by external stimulus with higher power value. That is, among periodic stimuli, the neural activity of neuron will be dominated by the external forcing that can inject more energy into the neuron. In experience, stronger inputs as larger amplitude can induce a prior response than weak disturbance because of higher energy injection hidden in the external stimuli with stronger intensity. Under noisy condition, similar SR can be induced when two or more periodic stimuli are applied. In particular, similar SR also occurs when chaotic signal is used to excite the neuron under appropriate noise intensity. The results meet our experience that a signal processor prefers to receive and encode the external signal with higher energy and then the firing mode is controlled by this external stimulus completely. In noisy condition, the external stimulus can be discerned under SR state.
References
O’Sullivan, J., Herrero, J., Smith, E., et al.: Hierarchical encoding of attended auditory objects in multi-talker speech perception. Neuron 104(6), 1195–1209 (2019)
Shamma, S.A., Micheyl, C.: Behind the scenes of auditory perception. Curr. Opin. Neurobiol. 20(3), 361–366 (2010)
Phillips, D.P., Hall, S.E., Boehnke, S.E.: Central auditory onset responses, and temporal asymmetries in auditory perception. Hear. Res. 167(1–2), 192–205 (2002)
Carcea, I., Insanally, M.N., Froemke, R.C.: Dynamics of auditory cortical activity during behavioural engagement and auditory perception. Nat. Commun. 8, 14412 (2017)
Hsu, A., Woolley, S.M.N., Fremouw, T.E., et al.: Modulation power and phase spectrum of natural sounds enhance neural encoding performed by single auditory neurons. J. Neurosci. 24, 9201–9211 (2004)
Das, N., Biesmans, W., Bertrand, A., et al.: The effect of head-related filtering and ear-specific decoding bias on auditory attention detection. J. Neural Eng. 13(5), 056014 (2016)
Kaya, E.M., Elhilali, M.: Modelling auditory attention. Philos. Transact. Royal Soc. B: Biol. Sci. 372(1714), 20160101 (2017)
Geravanchizadeh, M., Roushan, H.: Dynamic selective auditory attention detection using RNN and reinforcement learning. Sci. Rep. 11(1), 15497 (2021)
Lu, Y., Wang, M., Yao, L., et al.: Auditory attention decoding from electroencephalography based on long short-term memory networks. Biomed. Signal Process. Control 70, 102966 (2021)
Xin, Y., Zhong, L., Zhang, Y., et al.: Sensory-to-category transformation via dynamic reorganization of ensemble structures in mouse auditory cortex. Neuron 103(5), 909–921 (2019)
He, Y., Nie, S., Liu, R., et al.: Spatiotemporal information processing emulated by multiterminal neuro-transistor networks. Adv. Mater. 31(21), 1900903 (2019)
Li, Z., Li, J., Hong, B., et al.: Speaker–listener neural coupling reveals an adaptive mechanism for speech comprehension in a noisy environment. Cereb. Cortex 31(10), 4719–4729 (2021)
Zhou, P., Yao, Z., Ma, J., et al.: A piezoelectric sensing neuron and resonance synchronization between auditory neurons under stimulus. Chaos Solitons Fract. 145, 110751 (2021)
Guo, Y., Zhou, P., Yao, Z., et al.: Biophysical mechanism of signal encoding in an auditory neuron. Nonlinear Dyn. 105, 3603–3614 (2021)
Zhang, X., Ma, J.: Wave filtering and firing modes in a light-sensitive neural circuit. J. Zhejiang Univ.-Sci. A 22(9), 707–720 (2021)
Longtin, A.: Stochastic resonance in neuron models. J. Stat. Phys. 70, 309–327 (1993)
Moss, F., Ward, L.M., Sannita, W.G.: Stochastic resonance and sensory information processing: a tutorial and review of application. Clin. Neurophysiol. 115(2), 267–281 (2004)
Jung, P., Mayer-Kress, G.: Spatiotemporal stochastic resonance in excitable media. Phys. Rev. Lett. 74(11), 2130 (1995)
Harmer, G.P., Davis, B.R., Abbott, D.: A review of stochastic resonance: circuits and measurement. IEEE Trans. Instrum. Meas. 51, 299–309 (2002)
Chialvo, D.R., Calvo, O., Gonzalez, D.L., et al.: Subharmonic stochastic synchronization and resonance in neuronal systems. Phys. Rev. E 65, 050902 (2002)
Zhou, C., Kurths, J.: Noise-induced synchronization and coherence resonance of a Hodgkin-Huxley model of thermally sensitive neurons. Chaos 13, 401–409 (2003)
Huang, P., Guo, Y., Ren, G., et al.: Energy-induced resonance synchronization in neural circuits. Mod. Phys. Lett. B 35, 2150433 (2021)
Nakamura, O., Tateno, K.: Random pulse induced synchronization and resonance in uncoupled non-identical neuron models. Cogn. Neurodyn. 13(3), 303–312 (2019)
Locatelli, N., Mizrahi, A., Accioly, A., et al.: Noise-enhanced synchronization of stochastic magnetic oscillators. Phys. Rev. Appl. 2, 034009 (2014)
Stein, R.B., Gossen, E.R., Jones, K.E.: Neuronal variability: noise or part of the signal? Nat. Rev. Neurosci. 6, 389–397 (2005)
Ma, W.J., Beck, J.M., Latham, P.E., et al.: Bayesian inference with probabilistic population codes. Nat. Neurosci. 9(11), 1432–1438 (2006)
McDonnell, M.D., Ward, L.M.: The benefits of noise in neural systems: bridging theory and experiment. Nat. Rev. Neurosci. 12(7), 415–425 (2011)
Tougaard, J.: Stochastic resonance and signal detection in an energy detector–implications for biological receptor systems. Biol. Cybern. 83(5), 471–480 (2000)
Balenzuela, P., Braun, H., Chialvo, D.R.: The ghost of stochastic resonance: an introductory review. Contemp. Phys. 53, 17–38 (2012)
Wang, G., Ge, M., Lu, L., et al.: Study on propagation efficiency and fidelity of subthreshold signal in feed-forward hybrid neural network under electromagnetic radiation. Nonlinear Dyn. 103, 2627–2643 (2021)
Ge, M.Y., Wang, G.W., Jia, Y.: Influence of the Gaussian colored noise and electromagnetic radiation on the propagation of subthreshold signals in feedforward neural networks. Sci. China Technol. Sci. 64(4), 847–857 (2021)
Liu, Y., Xu, W., Ma, J., et al.: A new photosensitive neuron model and its dynamics. Front. Inform. Technol. Electron. Eng. 21(9), 1387–1396 (2020)
Xu, Y., Liu, M., Zhu, Z., et al.: Dynamics and coherence resonance in a thermosensitive neuron driven by photocurrent. Chin. Phys. B 29(9), 098704 (2020)
Yao, Z., Zhou, P., Zhu, Z., et al.: Phase synchronization between a light-dependent neuron and a thermosensitive neuron. Neurocomputing 423, 518–534 (2021)
Bao, B., Wang, N., Xu, Q., et al.: A simple third-order memristive band pass filter chaotic circuit. IEEE Trans. Circuits Syst. II Express Briefs 64(8), 977–981 (2016)
Bao, B., Xu, L., Wu, Z., et al.: Coexistence of multiple bifurcation modes in memristive diode-bridge-based canonical Chua’s circuit. Int. J. Electron. 105(7), 1159–1169 (2018)
Chen, M., Ren, X., Wu, H.G., et al.: Periodically varied initial offset boosting behaviors in a memristive system with cosine memductance. Front. Inform. Technol. Electron. Eng. 20(12), 1706–1716 (2019)
Chen, M., Qi, J.W., Wu, H.G., et al.: Bifurcation analyses and hardware experiments for bursting dynamics in non-autonomous memristive FitzHugh-Nagumo circuit. Sci. China Technol. Sci. 63(6), 1035–1044 (2020)
Bao, B.C., Zhu, Y.X., Ma, J., et al.: Memristive neuron model with an adapting synapse and its hardware experiments. Sci. China Technol. Sci. 64, 1107–1117 (2021)
Zhang, Y., Wang, C.N., Tang, J., et al.: Phase coupling synchronization of FHN neurons connected by a Josephson junction. Sci. China Technol. Sci. 63(11), 2328–2338 (2020)
Zhang, Y., Zhou, P., Tang, J., et al.: Mode selection in a neuron driven by Josephson junction current in presence of magnetic field. Chin. J. Phys. 71, 72–84 (2021)
Zhang, Y., Xu, Y., Yao, Z., et al.: A feasible neuron for estimating the magnetic field effect. Nonlinear Dyn. 102(3), 1849–1867 (2020)
Hussain, I., Jafari, S., Ghosh, D., et al.: Synchronization and chimeras in a network of photosensitive FitzHugh–Nagumo neurons. Nonlinear Dyn. 104, 2711–2721 (2021)
Xie, Y., Yao, Z., Hu, X., et al.: Enhance sensitivity to illumination and synchronization in light-dependent neurons. Chin. Phys. B 30, 120510 (2021)
Xie, Y., Zhu, Z.G., Zhang, X.F., et al.: Control of firing mode in nonlinear neuron circuit driven by photocurrent. Acta Physica Sinica 70(21), 210502 (2021)
Hussain, I., Ghosh, D., Jafari, S.: Chimera states in a thermosensitive FitzHugh-Nagumo neuronal network. Appl. Math. Comput. 410, 126461 (2021)
Tagne, J.F., Edima, H.C., Njitacke, Z.T., et al.: Bifurcations analysis and experimental study of the dynamics of a thermosensitive neuron conducted simultaneously by photocurrent and thermistance. Eur. Phys. J. Spec. Top. 231, 993–1004 (2022). https://doi.org/10.1140/epjs/s11734-021-00311-w
Zhang, X., Yao, Z., Guo, Y., et al.: Target wave in the network coupled by thermistors. Chaos Solitons Fract. 142, 110455 (2021)
Yao, Z., Wang, C.: Control the collective behaviors in a functional neural network. Chaos Solitons Fract. 152, 111361 (2021)
Siveke, I., Myoga, M.H., Grothe, B., et al.: Ambient noise exposure induces long-term adaptations in adult brainstem neurons. Sci. Rep. 11, 5139 (2021)
Muthuswamy, B., Chua, L.O.: Simplest chaotic circuit. Int. J. Bifurcat. Chaos 20(05), 1567–1580 (2010)
Murali, K., Lakshmanan, M., Chua, L.O.: The simplest dissipative nonautonomous chaotic circuit. IEEE Transact. Circ. Syst. I: Fundament. Theory App. 41(6), 462–463 (1994)
Ma, X., Xu, Y.: Taming the hybrid synapse under energy balance between neurons. Chaos Solitons Fract. 159, 112149 (2022)
Zhou, P., Zhang, X., Ma, J.: How to wake up the electric synapse coupling between neurons? Nonlinear Dyn. 108, 1681–2169 (2022)
Xie, Y., Yao, Z., Ma, J.: Phase synchronization and energy balance between neurons. Front. Inform.Technol. Electron. Eng. (2022). https://doi.org/10.1631/FITEE.2100563
Sarasola, C., Torrealdea, F.J., d’Anjou, A., et al.: Energy balance in feedback synchronization of chaotic systems. Phys. Rev. E 69, 011606 (2004)
Hou, Y., Fang, P.: Synchronization and stability of two unbalanced rotors with fast antirotation considering energy balance. Math. Probl. Eng. 2015, 694145 (2015)
Wang, Y., Sun, G., Ren, G.: Diffusive field coupling induced synchronization between neural circuits under energy balance. Chin. Phys. B (2022). https://doi.org/10.1088/1674-1056/ac7bff
Torrealdea, F.J., d’Anjou, A., Graña, M., et al.: Energy aspects of the synchronization of model neurons. Phys. Rev. E 74, 011905 (2006)
Yao, Z., Zhou, P., Alsaedi, A., et al.: Energy flow-guided synchronization between chaotic circuits. Appl. Math. Comput. 374, 124998 (2020)
Acknowledgements
This project is partially supported by the National Natural Science Foundation of China under the Grant Nos. 11765011.
Author information
Authors and Affiliations
Contributions
Ying Xie finished the definition of dynamical model and numerical results and figures; Jun Ma suggested this study, contributed to the writing of the original draft and editing of the final version, and explained the biophysical mechanism and numerical results.
Corresponding author
Ethics declarations
Ethical approval
This is a theoretical study. The Lanzhou University of Technological Research Ethics Committee has confirmed that no ethical approval is required.
Informed consent
All authors (Ying Xie and Jun Ma) agree to submit and publish this work on Journal of Biological Physics.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xie, Y., Ma, J. How to discern external acoustic waves in a piezoelectric neuron under noise?. J Biol Phys 48, 339–353 (2022). https://doi.org/10.1007/s10867-022-09611-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10867-022-09611-1