Abstract
The landscape of the Brazilian Cerrado has been modified by human actions in the last decade with a significant increase in fires. Despite efforts to delimit an area for biodiversity conservation, human pressure on the landscape surrounding these units can affect the local environmental condition. We assessed the effect of landscape factors and fire occurrence on necrophagous Diptera diversity in different vegetation physiognomies of the Cerrado region in Chapada das Mesas National Park. We sampled in 35 sites in five physiognomies of the Cerrado biome (Open Forest or Cerradão, Dense Woodland, Gallery Forest, Park Woodland, and Woodland) using 350 traps to capture the Diptera specimens. We observed lower dipteran abundance and richness in the Gallery Forest compared to other physiognomies, and the species composition differed from all other physiognomies. Specifically, five species of Diptera were predominantly found in Open Forest or Cerradão, two species in Park Woodland, and one in Gallery Forest. The presence of species with generalist habits in forested physiognomies and the low abundance of expected forest-associated species in forest habitats suggests a decline in environmental quality in these areas. This serves as a warning sign for the ecological conditions of the Park’s predominantly forested physiognomies.
Implications for insect conservation
Our results highlight that despite the biodiversity of necrophagous dipterans observed in CMNP, ongoing fire frequency and anthropic activities within the park could severely undermine its ability to preserve Diptera biodiversity. Consequently, environmental agencies must intensify monitoring efforts and implement effective measures to mitigate human-induced impacts in this Brazilian Cerrado conservation unit.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Cerrado biome, covering 25% of Brazil’s territory, stands as one of the world’s most diverse savannas and is considered the second most crucial biome after the Amazon in terms of endemic biodiversity and threatened ecosystems (Durigan and Ratter 2016). It encompasses a wide range of formations and physiognomies, from open fields to dense forests. Notably, 24% of this expanse comprises a mosaic of forest and savannah, with only 4% designated as dry forest (Silva and Bates 2002). This gradient of forest formations can significantly influence the composition and distribution of organisms, including herpetofauna (Nogueira et al. 2005; Barros et al. 2022), insects orders such as Lepidoptera (Pinheiro and Ortiz 1992), dung beetles (Silva et al. 2021), and Diptera (Scherber et al. 2014). For instance, previous studies have documented greater dung beetle richness in savanna areas compared to forest formations (Silva et al. 2021; Barros et al. 2022). The authors suggest that the heterogeneity of habitat within different vegetation formations serves as the primary factor influencing the distribution of organisms across the diverse physiognomies of the Cerrado (Silva et al. 2021).
At present, it is estimated that only 55% (112.6 million hectares) of the native Cerrado coverage remains (Alencar et al. 2020). The primary drivers behind this reduction in coverage are urban expansion, land clearing for agriculture, and cattle ranching (Overbeck et al. 2015). The rapid development of agricultural technology and expansion of cultivated areas, particularly for agro-industrial purposes, have led to significant changes in land use and coverage in the Cerrado. Furthermore, the rates of deforestation, soil erosion, sedimentation, river pollution (Sano et al. 2008), the introduction of exotic plants (grasses), disturbances caused by cattle trampling (soil compaction) (Myers 2006), and increased fire frequency, as fire is used to stimulate grass regrowth for cattle (Ribeiro and Figueira 2011), continue to rise.
Fire is a primary natural disturbance influencing the structure and dynamics of the Cerrado vegetation (Coutinho 1990). Fire occurrences are frequent in savanna-like environments and drive an intricate interplay between climate and vegetation (Miranda et al. 2002). Consequently, the persistent fuel layer is mainly dominated by grasses, which proliferate during the rainy season and become progressively more susceptible to ignition during the dry season, coinciding with the peak of burnings (Van Der Werf et al. 2008). In Brazil, an estimated 3 to 12% of the Cerrado biome is affected by fire annually (Alves and Alvarado 2019). However, human activities have induced uncontrolled, deliberate fires that can severely impact the adaptive capacity and resilience of the region’s flora and fauna (Medeiros and Miranda 2008). For instance, fire promotes herbaceous species adapted to annual burning events at the expense of trees (Pivello 2011). This has negative repercussions for tree-associated invertebrates such as bees, wasps and, tree-dwelling species (Peralta et al. 2017; Vasconcelos et al. 2017), as well as larger, more dispersal-capable vertebrates like certain reptiles and amphibians (Sousa et al. 2015; Abom and Schwarzkopf 2016), and even medium and large mammals (Silveira et al. 1999).
The flies of the families Calliphoridae, Mesembrinellidae, and Sarcophagidae are distributed in various environments and have an essential ecological role in the cycling of nutrients by performing organic matter decomposition (Whitworth and Yussef-Vanegas 2019). Studies on the Diptera of these three families provide relevant information on the human influence on flys communities and are essential as indicators of conservation (Sousa et al. 2020) or degradation of natural environments triggered by human activity (Gadelha et al. 2009). Some Calliphoridae species (e.g. Chrysomya albiceps) and Sarcophagidae (e.g., Tricharaea (Sarcophagula) occidua) are highly synanthropic (Sousa et al. 2020), while Mesembrinellidae species are non-synanthropic, but is abundant and diverse in tropical rain and cloud forests (Whitworth and Yusseff-Vanegas 2019). Studies with necrophagous dipteran communities in different environments, including the cerrado, have highlighted the influence of vegetation type (Sousa et al. 2016) and anthropogenic activities (Sousa et al. 2020) on patterns of abundance, composition, and richness. Regarding vegetation, communities within more structured and heterogeneous vegetation types had higher species richness than those observed in the less complex zones, with the exception of the sarcophagids in the Amazon Forest (Sousa et al. 2016). Nascimento et al. (2021) recorded that sarcophagids, were more abundant in open areas of Cerrado. These findings are consistent with other research, indicating that sarcophagid species are more abundant in open environments, such as pastures, clearings, and savannas, than in adjacent forests (Sousa et al. 2011, 2016; Yepes-Gaurisas et al. 2013).
The increase in human activity in natural environments makes it necessary to have a biodiversity refuge with available habitat that allows the occurrence of species (Haddad et al. 2015). The consolidation of the Brazilian System of Conservation Units (SNUC) was an important instrument for advancing the protection and preservation of biodiversity and natural habitats and biodiversity conservation (Law nº. 9.985/2000). However, despite efforts to delimit an area for biodiversity conservation, human pressure on the landscape surrounding these units can affect the local environmental condition (Pinheiro and Durigan 2009). In this context, our aim is to assess the effect of landscape factors and fire occurrence on necrophagous Diptera diversity (Calliphoridae, Mesembrinellidae, and Sarcophagidae) in different vegetation physiognomies of the Cerrado region in Chapada das Mesas National Park (CMNP). We expect that: (a) The environmental factors differ among the five physiognomies of the Cerrado, and that more open formations, such as Park Woodland (PW) and Woodland (WOO), exhibit a higher frequency of fires than forest physiognomies; (b) The patterns of richness, abundance, and composition of necrophagous dipteran species in the CMNP are influenced by the types of vegetation physiognomies in the Cerrado; (c) Necrophagous dipteran species are associated with different physiognomies of the Cerrado, with some species adapted to more open formations (Park Woodland - PW and Woodland - WOO), while others are associated with physiognomies characterized by denser vegetation (Open Forest or Cerradão - OF.C, Dense Woodland – DW, and Gallery Forest – GL).
Methods
Study area
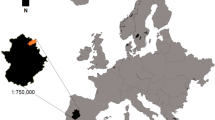
This study was carried out in 35 sites situated within the Chapada das Mesas National Park (CMNP) unit conservation, spanning an expanse of 160,046 hectares distributed in the southern state of Maranhão, Brazil (Fig. 1). The topography of CMNP primarily consists of flat to undulating terrain, featuring plateaus at a basal elevation of around 620 m. The climate is classified as tropical wet, with an average annual temperature of 26 °C and yearly precipitation ranging from 1250 to 1500 mm (Souza et al. 2015). There are two well-defined seasons: a dry season from May to October and the wet season from November to April (Carvalho et al. 2023).
Geographic locations of the 35 sampled sites within in the Chapada das Mesas National Park (CMNP), Maranhão, Brazil. These sites were strategically positioned across five distinct Cerrado physiognomies: Open Forest or Cerradão (OF.C), Dense Woodland (DW), Gallery Forest (GL), Park Woodland (PW) and Woodland (WOO).
The sites were distributed in five physiognomies types of the Cerrado: Open Forest or Cerradão (OF.C, five sites), Dense Woodland (DW, six), Gallery Forest (GF, eight), Park Woodland (PW, six) and Woodland (WOO, 10) (see Ribeiro and Walter 2008 for more details), with a minimum distance of 1 km between sites. Open Forest or Cerradão features a forest physiognomy with trees reaching up to 15 m high, creating a continuous canopy without grass (Juhász et al. 2006). It displays xeromorphic characteristics with tree cover ranging from 50 to 90% (Lima-Ribeiro 2008). Cerradão soils are typically deep and well-drained, with fertility levels varying from medium to low. They are slightly acidic, classified as Dark Red Latosol, Red Yellow Latosol, or Purple Latosol (Ribeiro and Walter 2008). In Dense Woodland, the vegetation is predominantly arboreal, covering 50–70%, with an average tree height ranging from five to eight meters. The shrubby and herbaceous layers are thinner, likely due to shading caused by the higher tree density. The soil predominantly consists of Purple, Dark-Red, Red-Yellow Latosols, and Cambisols. The Gallery Forest is found along small rivers and streams (up to the third or fourth order), creating enclosed vegetation corridors (galleries) over the watercourse (Roque et al. 2013). The tree layer’s average height varies from 20 to 30 m, with overlapping canopies providing tree coverage of 70 to 95%. Park Woodland exhibits savannah formation marked by grouped trees in small, sometimes imperceptible, land elevations called “murundus” or “monchões”. These trees have an average height of three to six meters, forming a tree cover of 5–20%. The soils are hydromorphic, and the murundus offer better drainage than the adjacent flat areas (Ribeiro and Walter 2008). Woodland is primarily tree-shrub, with tree coverage ranging from 20 to 50% and an average tree height of three to six meters. It represents a common and intermediate form between Dense Cerrado and Sparse Cerrado, occurring in soils such as Oxissolos Verm Dark-Elho, Red-Yellow, Cambisols, Quartz Sands, Lithologic or Concretionary soils (Ribeiro and Walter 2008).
Environmental factors
The occurrence of fire within various vegetation physiognomies of the Cerrado was determined through the reconstruction of the burning regime within CMNP, achieved by delimiting burn scars. The identification and demarcation of these burn scars were carried out manually through visual interpretation of false color compositions derived from Landsat satellite images spanning a 28-years period (1990 to 2017), which corresponded to the annual burning season between May and November each year. Subsequently, we conducted a comparative analysis between the location of the sampling sites and the documented fire to ascertain the frequency of burning across the sampling sites over the years leading up to the most recent fire event. The burn scars identified within the satellite images were further verified during field sampling.
The Normalized Difference Vegetation Index (NDVI) is derived from the ratio between the difference in reflectance of Near Infrared and Red bands, divided by the sum of the same variables, as illustrated in the following equation: NDVI = (NIR – R) / (NIR + R) (Eq. 1), wherein NIR signifies Near Infrared and R stands for Red. The resulting values are within the range of -1 to + 1, with values closer to + 1 indicating higher vegetation density (e.g., Carvalho et al. 2023). Although close to 0, positive values are associated with exposed soil or sparse vegetation, while negative values correspond to water bodies. We calculated the NDVI utilizing a Landsat 8 satellite image captured by the OLI sensor, specifically orbit 222, row 65 of June 2017. These Landsat images were generously provided by the United States Geological Survey (USGS), and are accessible at http://earthexplorer.usgs.gov/. For the calculation, we utilized the image’s 4 band (Red) and 5 (NIR) band, processed using QGIS 3.4.4 software. The classification intervals for NDVI classes were established employing the discrete method of cumulative cut-off and count, resulting in five classes of equal intervals. Additionally, we employed the extract by point procedure, utilizing vectorial data in shapefile format containing sampling points, to extract the pixel values and incorporate them into the attribute table.
We manually assessed the distance between the sampled sites and the pasture areas using the field calculator tool in QGIS 3.4.4. This tool determined the direct distance from a point within the sampled area to the nearest border of a pasture area, providing an estimate of the actual distance between the two areas. The data were projected onto the UTM system, Zone 23 S, with the SIRGAS 2000 datum. The pasture areas were demarcated through visual interpretation of Landsat 8 satellite images and subsequently validated through field observations. Based on these procedures, four environmental factors were calculated: Distance between the sampled sites and pasture areas (DSPA), Burning frequency (BF), Year of the last fire (YLF) and Normalized Difference Vegetation Index (NDVI). Correlations among these environmental characteristics consistently exhibited low to moderate values (pairwise Pearson correlation r consistently ≤ 0.7, as shown in Supplementary Figure S1), thus allowing for the inclusion of all of them in subsequent analyses.
Biologic sampling
At each site, we employed specific traps adapted by Sousa et al. (2020), each containing 50 g of bovine lung as an attractive bait suspended on tree branches 40 centimeters above the ground. Ten traps were randomly distributed at each site, maintaining a minimum distance of 50 m between them, and a designated separation of 1 km between the replicate sites, totaling 350 traps. The traps were exposed for a period ranging from 24 to 36 h. All fieldwork was conducted during the dry season of October 2015, March and September 2016, April and October 2017, November 2018, and July 2019.
The flies sampled were identified at the species level using the specialized key of Mello (2003), González et al. (2017), Whitworth and Yusseff-Vanegas (2019), Lopes and Tibana (1987), Buenaventura and Pape (2013) and Guimarães (2004). The identification of the Sarcophagidae family is based mainly on the male genitalia, and due to this limitation of taxonomic knowledge, our study identified only male specimens. All individuals are preserved in 70% ethanol and stored in the Laboratory of Environmental Sciences and Biodiversity of the Universidade Estadual do Maranhão, São Luís, Brazil.
Statistical analysis
Each site was considered as an independent sample unit in the analyses. To assess our first hypothesis - The environmental factors differ among the five physiognomies of the Cerrado, and that more open formations, such as Park Woodland (PW) and Woodland (WOO), exhibit a higher frequency of fires than forest physiognomies - we employed Principal Components Analysis (PCA). The PCA utilized Euclidean distance with standardized environmental factors. Axes selected for interpretation were based on the broken-stick criteria (Legendre and Legendre 2012). The significance of the PCA was assessed using Permutational Multivariate Analysis of Variance (PERMANOVA, Anderson 2001), with Euclidean distance and 10,000 permutations. Pairwise comparisons between each pair of Cerrado physiognomies were conducted to interpret the results.
To test our second hypothesis - The patterns of richness, abundance and composition of necrophagous dipteran species in the CMNP are influenced by the types of vegetation physiognomies in the Cerrado - we used the composition, richness, and abundance of species as response variables. Differences in species composition between the physiognomies were assessed using Permutational Multivariate Analysis of Variance (PERMANOVA, Anderson 2001), with pairwise comparisons between each pair of physiognomies. The PERMANOVA analysis utilized Bray-Curtis distance matrix calculated from log-transformed composition data. The null hypothesis of no differences between sites was tested using a Monte Carlo method with 10,000 random permutations, and p-values ≤ 0.05 were considered statistically significant. Principal Coordinates Analysis (PCoA) was employed to visualize differences in species composition among physiognomies. Additionally, differences in species richness of necrophagous Diptera among the physiognomies types were assessed using analysis of variance (ANOVA, Zar 2010), followed by a Tukey’s post hoc test. Necrophagous Diptera abundance was analyzed using a Kruskall-Wallis non-parametric test (Zar 2010).
To test our third hypothesis - Necrophagous dipteran species are associated with different physiognomies of the Cerrado, with some species adapted to more open formations (Park Woodland - PW and Woodland - WOO), while others are related to physiognomies characterized by denser vegetation (Open Forest or Cerradão - OF.C, Dense Woodland – DW, and Gallery Forest – GL - we used an Indicator Species Analysis (Dufrêne and Legendre 1997). This analysis calculates an indicator value (IV) for each species, ranging from 0 to 100, based on the relative abundance and frequencies of each species in different physiognomies. An IV of 0 indicates that a species is entirely absent from a specific physiognomy, while an IV of 100 signifies a complete association, where all individuals of a species were exclusively found in a particular physiognomy. The statistical significance of IVs (α = 0.05) was tested through 10,000 randomizations of the individuals between sites.
We employed Partial Redundancy Analysis (RDA) and Generalized Linear Models (GLM, Zuur et al. 2009) to assess the relationship between species composition, richness, and abundance of individuals with environmental factors. Prior to conducting the RDA analysis, abundance data underwent a Hellinger transformation (the square root of relative abundance), and environmental factors were standardized (Legendre and Gallagher 2001). The statistical significance of the RDA was evaluated using an ANOVA. In the GLM, we used negative binomial adjustment for the richness and abundance of necrophagous flies, and model validated was performed using residuals for adjusted linear mixed models (DHARMa, Hatig 2022). All analyses were conducted in R version 3.4.1 (R Core Team 2016) using the “mgcv” (Wood 2006) and “pairwiseAdonis” (Arbizu 2019) packages for multivariate analyses.
Results
Environmental factors
Environmental conditions varied among the Cerrado vegetation types sampled in our study (pseudo-F = 7.757, p < 0.01). In the comparisons, the Woodland (WOO) physiognomy differed from Open Forest or Cerradão (OF.C), Dense Woodland (DW), and Gallery Forest (GF) (Table 1; Fig. 2). The areas of Park Woodland (PW) and Woodland (WOO) exhibited a higher frequency of fires, while OF.C and GF showed greater vegetation cover, as represented by the Normalized Difference Vegetation Index (NDVI) (Fig. 3). Furthermore, OF.C and GF had the lowest frequency of fires occurrence, with the last record averaging 21 years ago. The Dense Woodland (DW) physiognomy areas were the most distant from the areas used for pastures (Fig. 2).
Ordination derived from Principal Component Analysis (PCA) performed of environmental factors of the sites sampled in Chapada das Mesas National Park (CMNP), Maranhão, Brazil. Physiognomies of the Cerrado: OF.C = Open Forest or Cerradão; DW = Dense Woodland; GF = Gallery Forest; PW = Park Woodland; WOO = Woodland. Environmental factors: DSPA = Distance between sampled sites and pasture areas; BF = Burning frequency; YLF = Year of the last fire; NDVI = Normalized Difference Vegetation Index
Diversity of necrophagous Diptera
We sampled 18,303 necrophagous Diptera individuals distributed in three families and identified 34 species (Table S1). The most abundant species were Chrysomya albiceps and Tricharaea (Sarcophagula) occidua. Richness (F = 5.93; p < 0.01), abundance (X2 = 15.02; p < 0.01) and composition (pseudo-F = 9.97; p < 0 0.01) differ among the physiognomies sampled in the study. Considering the abundance, only two physiognomies were different, with Gallery Forest having a lower abundance than Park Woodland (Fig. 4A, Table S2A). Species richness was lower in Gallery Forest than in the other four physiognomies (Open Forest or Cerradão, Dense Woodland, Woodland, and Park Woodland (Fig. 4B, Table S2B). Environmental factors did not influence the richness and abundance of necrophagous Diptera (Table S4).
Box plot of the abundance (A) and richness (B) of necrophagous Diptera in the Open Forest or Cerradão (OF.C) sites, Dense Woodland (DW), Gallery Forest (GF), Park Woodland (PW) and Woodland (WOO). The horizontal line represents the mean; the box represents the interquartile range (IQR), and the whiskers represent values up to +/−1.5×IQR from the 25th and 75th percentiles, respectively
The species composition of the Gallery Forest differs from all other physiognomies (Fig. 5). Although the composition of Open Forest or Cerradão is similar to Dense Woodland, the species that occur in Open Forest or Cerradão differ from Gallery Forest, Park Woodland, and Woodland (Table S3). When evaluating the association of species with the physiognomies of the Cerrado, we observed that C. albiceps, Lucilia eximia, Peckia (Euboetcheria) anguilla, Peckia (Peckia) pexata, Peckia (Sarcodexia) lambens were associated with Open Forest or Cerradão (Table 2). Only Mesembrinella bicolor was associated with Gallery Forest and Oxysarcodexia aura, and T. occidua with Park Woodland (Table 2). Environmental factors were important predictors for species composition of necrophagous Diptera (F = 4.09; p < 0.01). Two species of Sarcophagidae (P. pexata and P. lambens) and one species of Calliphoridae (C. albiceps), were more associated with the highest values of normalized difference vegetation index (NDVI), year of the last fire (YLF), and Distance between sampled sites and pasture areas (DSPA) (Fig. 5B).
(A) Ordination derived from Principal Coordinates Analysis (PCoA) and (B) Redundancy Analysis (RDA) performed of composition species and environmental factors measured in the sites sampled in Chapada das Mesas National Park (CMNP), Maranhão, Brazil. Physiognomies of the Cerrado: OF.C = Open Forest or Cerradão; DW = Dense Woodland; GF = Gallery Forest; PW = Park Woodland; WOO = Woodland. Environmental factors: BF = Burning frequency, NDVI = Normalized Difference Vegetation Index, YLF = Year of the last fire and DSPA = Distance between sampled sites and pasture areas
Discussion
Our hypothesis, proposing that environmental factors differ among the five physiognomies of the Cerrado, and that more open formations, such as Park Woodland (PW) and Woodland (WOO), exhibit a higher frequency of fires than forest physiognomies, was corroborated. Fire is a crucial factor in many global ecosystems, influencing the distribution, abundance, and structural form of certain plant species and communities (Bond et al. 2005), as well as animals (Frizzo et al. 2011; Watson et al. 2012; Novoa et al. 2021). In the Cerrado biome, fire, coupled with the seasonality of rainfall and nutrient-poor soils, is considered a determinant of vegetation structure. When the timing and frequency of occurrence define a burning regime, organisms adapt to this disturbance (Miranda et al. 2010). However, the intensity and frequency of fire can play a critical role in transforming the environment (Wu et al. 2015) if it occurs disorderly. Forest physiognomies are less susceptible to natural fires than savannas and grasslands, with Gallery Forests being less adapted to fire, followed by Riparian Forests, Dry Forests and Cerradão. Thus, while the flora of savannas and grasslands includes numerous species with morphological and physiological characteristics adapted to fire (pyrophytic species), the flora of forests is proportionally less adapted. Nevertheless, the humidity and shading of the forest limit the spread of fire (Walter and Ribeiro 2010).
In the Cerrado biome, the selection pressure to withstand recurrent fires in forest areas was significantly lower than in savannah environments. Consequently, species from forest formations did not need to develop mechanisms to adapt to fire, as many species have efficiently done in savannas and grasslands (Silva et al. 2005). As a result, fire events, whether of natural or anthropic origin, can be devastating for numerous species or their populations when they penetrate forest formations (Silva et al. 2005). Based on our results, we propose continuous monitoring of the fire brigade teams in the Chapada das Mesas National Park, particularly in the Open Forest or Cerradão and Gallery Forest physiognomies, as they exhibit the highest vegetation indices (NDVI) and there have been no records of fires on average for 21 years. The occurrence of fire in these physiognomies can pose serious risks to fauna and flora that are not adapted to severe environmental stress caused by fire.
The second hypothesis that the patterns of richness, abundance, and composition of necrophagous dipteran species in the CMNP are influenced by the types of vegetation physiognomies in the Cerrado, was corroborated, as evidenced by differences in richness, abundance and composition between the analyzed physiognomies (Fig. 4). The Cerrado is a highly heterogeneous biome, encompassing forests, pastures and grasslands in the same region (Oliveira and Marquis 2002). Thus, studies on animal communities, such as insects, also reflect the heterogeneity of the Cerrado given their direct susceptibility with vegetation types (Pacheco and Vasconcelos 2012; Mata and Tidon 2013) and regions within the biome (Ferro and Diniz 2007).
The lower richness observed in the Gallery Forest in our study may be attributed to the predominant presence of species from the families Calliphoridae and Sarcophagidae, which generally demonstrate a high degree of adaptation to less forested environments. It is noteworthy that only 8.5% of the total captured species belong to the fly genera Hemilucilia Brauer, 1985 (H. benoisti, H. segmentaria, and H. semidiaphana) and Mesembrinella (M. bicolor). Flies from the genus Hemilucilia have been documented in the literature as typical inhabitants of forested areas (D’Almeida & Lopes 1983). In various regions of Brazil, they are consistently associated with forest environments (Sousa et al. 2010, 2016; Esposito et al. 2010; Ferraz et al. 2010). The species M. bicolor, along with other mesembrinellids, are considered exclusive to forested habitats, being consistently found only in such areas (Guimarães 1977; Ferreira 1978; Whitworth and Yusseff-Vanegas 2019). The families Calliphoridae and Sarcophagidae include species associated with forest environments as well as more open vegetation, rural, and urban environments (Dufek et al. 2016), suggesting flexibility in the range of niches occupied by these species (e.g., Majer 1987). Studies on Sarcophagidae communities in other locations have shown similar patterns to those found in the CMNP (Sousa et al. 2010, 2011, 2021), indicating that these flies are better adapted to more anthropogenic habitats with reduced vegetation cover. According to Sousa et al. (2011), the resources available in these environments tend to be more ephemeral, implying a competitive advantage for rapidly developing species, such as sarcophagids, which are ovoviviparous.
The environmental factors measured in this study did not appear to influence the richness and abundance of necrophagous dipterans, likely due to the constant activities of fire and grazing in the CMNP over an extended period. Consequently, the species we encountered may have already been filtered by these factors, with the most resistant and potentially sensitive species having become locally extinct. A recent study conducted at CMNP, mapping fire dynamics over 28 years, revealed that the burned area did not change significantly over time (Carvalho et al. 2022). This study also highlighted that despite being a protected area with strict rules governing access and landscape use, there are still human dwellings and cattle raising within the park’s boundaries. Moreover, some necrophagous dipteran species exhibit greater adaptive plasticity, enabling them to navigate through a broader range of environmental variations (Mello et al. 2007; Cabrini et al. 2013). However, a study in different phytogeographic regions of Maranhão (Cerrado, Amazon Forest, Palm Forest, flooded fields, and mangroves) demonstrated that other environmental factors associated with microhabitat conditions, such as litter depth, tree/shrub density, vegetation height, and temperature, positively or negatively influenced the patterns of richness and abundance of necrophagous species (Sousa et al. 2021).
The lower species richness contributed to the dissimilarity in species composition of necrophagous Diptera from the gallery forest compared to other Cerrado physiognomies (Fig. 5). A study on necrophagous dipteran assemblages in the Cerrado showed that conserved forests harbor unique dipteran assemblages, characterized by Neotropical species with a more restricted to habitats in forested areas. Conversely, savannas and disturbed forests host more generalist and even exotic species (Mata and Tidon 2013). Furthermore, savannas feature open areas with intense and direct sunlight, generally attracting sarcophagus flies due to their cuticles having high thermal reflectance and their active thermoregulatory behavior (Willmer 1982; Mulieri et al. 2011). The heliophilous characteristic of these flies may provide an adaptive advantage, allowing them to exploit resources in open areas (Dufek et al. 2020).
Our hypothesis that necrophagous dipteran species are associated with different physiognomies of the Cerrado, with some species adapted to more open formations (Park Woodland - PW and Woodland - WOO), while others are associated with physiognomies characterized by denser vegetation (Open Forest or Cerradão - OF.C, Dense Woodland – DW, and Gallery Forest – GL), was corroborated. We identified five species associated with Dense Forest or Cerradão, one with Gallery Forest and two with Park Woodland. We observed the association of the species P. (Peckia) pexata, P. (Sarcodexia) lambens), P. (Euboettcheria) anguila, C. albiceps and L. eximia) with the Open Forest or Cerradão physiognomy (Table 2). Due to the generalist habit of these species (Mulieri et al. 2008; Sousa et al. 2021), it was expected that they would be associated with the more open physiognomies of the Cerrado, as already registered in another study (Sousa et al. 2021).
The exotic C. albiceps is considered characteristic of anthropogenic environments, such as urban centers (Baumgartner and Greenberg 1984). The native species L. eximia is common in forests and rural areas in Brazil (Linhares 1981), Peru (Baumgartner and Greenberg 1984), and Argentina (Mariluis and Schnack 1989). It should be noted that this Open Forest or Cerradão physiognomy was the area farthest from the pasture areas, with good vegetation cover, as evidenced by the high NDVI values recorded in the study. Additionally, they also presented the lowest incidence of fire records, with the last record being more than 20 years old. Thus, the association of these species with these environments in the Chapada das Mesas National Park raises a warning sign and concerns regarding the environmental integrity of these areas, as these three species are indicators of anthropic environments, given the preference for open areas (Sousa et al. 2021). The species O. aura and T. occidua associated with Park Woodland, and M. bicolor, linked to the Gallery Forest physiognomy, are consistent with results found in other studies (Mulieri et al. 2011; Sousa et al. 2021). This correlation associates these two Sarcophagidae species with anthropogenic areas and M. bicolor with preserved areas.
Our study reveals that more open Cerrado formations, such as Park Woodland (PW) and Woodland (WOO), experience a higher frequency of fires compared to forest formations. We recommend implementing monitoring measures for these physiognomies within Chapada das Mesas National Park (CMNP) to prevent significant environmental damage that could endanger the region’s biodiversity conservation efforts. Furthermore, our research demonstrates a relationship between the distribution of some necrophagous species in CMNP and the various Cerrado physiognomies. The presence of species with generalist habits within forested physiognomy, such as Open Forest or Cerradão, and the low abundance of expected forest-associated species in forest habitats or at least in Gallery Forests, indicates a decline in environmental quality, serving as a warning sign for the ecological condition of CMNP’s predominantly forested areas. Despite the biodiversity of necrophagous dipterans observed in CMNP, continued burning of frequency and anthropic activities within CMNP could seriously compromise the conservation unit’s capacity to protect Diptera biodiversity. Therefore, environmental agencies must enhance monitoring efforts and establish effective measures to mitigate human-induced impacts in this Brazilian Cerrado conservation unit.
References
Abom R, Schwarzkopf L (2016) Short-term responses of reptile assemblages to ire in native and weedy tropical savannah. Glob Ecol Conserv 6:58–66. https://doi.org/10.1016/j.gecco.2016.02.002
Alencar A, Shimbo J, Lenti F, Marques BC, Zimbres B, Rosa M, Arruda V, Castro I, Ribeiro FMJP, Varela V, Alencar I, Piontekowski V, Ribeiro V, Bustamante MCM, Sano EE, Barroso M (2020) Mapping three decades of changes in the Brazilian savanna native vegetation using landsat data processed in the Google Earth engine platform. Remote Sens 12:924. https://doi.org/10.3390/rs12060924
Alves DB, Alvarado ST (2019) Variação espaço-temporal da ocorrência do fogo nos biomas brasileiros com base na análise de produtos de sensoriamento remoto. Geografia 44:321–345. https://doi.org/10.5016/geografia.v44i2.15119
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46. https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x
Arbizu PM (2019) pairwiseAdonis: Pairwise multilevel comparison using adonis. R package version 0.3
Barros RA, Dorado-Rodrigues TF, Valadão RM, Strüssmann C (2022) Diversity patterns of lizard assemblages from a protected habitat mosaic in the Brazilian Cerrado savanna. J Trop Ecol 38:340–350. https://doi.org/10.1017/S0266467422000244
Baumgartner DL, Greenberg B (1984) The genus Chrysomya (Diptera: Calliphoridae) in the New World. J Med Entomol 21:105–113. https://doi.org/10.1093/jmedent/21.1.105
Bond WJ, Woodward FI, Midgley GF (2005) The global distribution of ecosystems in a world without fire. New Phytol 165:525–538. https://doi.org/10.1111/j.1469-8137.2004.01252.x
Buenaventura E, Pape T (2013) Revision of the New World genus Peckia Robineau-Desvoidy (Diptera: Sarcophagidae). Zootaxa 3622:1–87. https://doi.org/10.11646/zootaxa.3622.1.1
Cabrini I, Grella MD, Andrade CF, Thyssen PJ (2013) Richness and composition of Calliphoridae in an Atlantic Forest fragment: implication for the use of dipteran species as bioindicators. Biodivers Conserv 22:2635–2643. https://doi.org/10.1007/s10531-013-0545-x
Carvalho IS, Alvarado ST, Silva TSF, Cordeiro CLO, Fidelis A, Saraiva RVC, Figueiredo FAMMA, Sousa JRP, Ferraz TM (2023) How changes the fire regime after creating a protected area in the Brazilian Cerrado? J Nat Conserv 71:126318. https://doi.org/10.1016/j.jnc.2022.126318
R Core Team (2016) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL: https://www.R-project.org/
Coutinho LM (1990) Fire in the Ecology of the Brazilian Cerrado. In: Goldmmer DJG (ed) Ecological studies. Springer, Berlin Heidelberg, pp 82–105
D’Almeida JM, Lopes HS (1983) Sinantropia De Dípteros Muscóides (Calliphoridae) no Estado do Rio De Janeiro. Revista Brasileira De Zoociências 6:39–48
Dufek MI, Oscherov EB, Damborsky MP, Mulieri PR (2016) Assessment of the abundance and diversity of Calliphoridae and Sarcophagidae (Diptera) in sites with different degrees of human impact in the Iberá wetlands (Argentina). J Med Entomol 53:827–835. https://doi.org/10.1093/jme/tjw045
Dufek MI, Larrea DD, Damborsky MP, Mulieri PR (2020) The effect of anthropization on Sarcophagidae (Diptera: Calyptratae) community structure: an assessment on different types of habitats in the Humid Chaco ecoregion of Argentina. J Med Entomol 57:1468–1479. https://doi.org/10.1093/jme/tjaa071
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–366. https://doi.org/10.1890/0012-9615(1997)067[0345:SAAIST]2.0.CO;2
Durigan G, Ratter JA (2016) The need for a consistent fire policy for Cerrado conservation. J Appl Ecol 53:11–15. https://doi.org/10.1111/1365-2664.12559
Esposito MC, Sousa JRPD, Carvalho-Filho FDS (2010) Diversidade De Calliphoridae (Insecta: Diptera) na base de extração petrolífera da bacia do Rio Urucu, na Amazônia brasileira. Acta Amazon 40:579–583. https://doi.org/10.1590/S0044-59672010000300018
Ferraz ACP, Gadelha BQ, Queiroz MMC, Moya-Borja GE, Aguiar-Coelho VM (2010) Effects of forest fragmentation on dipterofauna (Calliphoridae) at the Reserva Biológica do Tinguá, Nova Iguaçu, RJ. Braz J Biol 70:55–63. https://doi.org/10.1590/S1519-69842010000100009
Ferreira MJM (1978) Sinantropia de dípteros muscóides de Curitiba, Paraná. I. Calliphoridae. Rev Bras Biol 38:445454
Ferro VG, Diniz IR (2007) Composition of the Arctiidae species (Insecta, Lepidoptera) in Cerrado areas. Rev Bras Zool 24:635–646. https://doi.org/10.1590/S0101-81752007000300015
Frizzo TL, Bonizario C, Borges MP, Vasconcelos H (2011) Uma revisão dos efeitos do fogo sobre a fauna de formações savânicas do Fogo Nas savanas 8 soc. Nat. | Uberlândia, MG | v.35 | e68045 | 2023 | ISSN 1982–4513 Brasil. Oecol Aust 15:365–379. https://doi.org/10.4257/oeco.2011.1502.13
Gadelha BQ, Ferraz ACP, Aguiar-Coelho VM (2009) A importância dos Mesembrinelíneos (Diptera: Calliphoridae) e seu potencial como indicadores de preservação ambiental. Oecologia Brasiliensis 13:661–665. https://doi.org/10.4257/oeco.2009.1304.0
González CR, Llanos L, Oses C, Elgueta M (2017) Calliphoridae from Chile: key to the genera and species (Diptera: Oestroidea). Anales Inst Patagonia 45:19–27. https://doi.org/10.4067/S0718-686X2017000300019
Guimaraes JH (1977) A systematic revision of the Mesembrinellidae, stat. nov. (Diptera, Cyclorrhapha). Arquivos De Zoologia 29(1):1–109
Guimarães HJL (2004) Redescrição dos machos de dez espécies neotropicais de Ravinia Robineau-Desvoidy, 1863 (Diptera, Sarcophagidae). Arch Mus Nac Rio De Janeiro 62:45–66
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Collins CD, Cook WM, Damschen EI, Ewers RM, Foster BL, Jenkins CN, King AJ, Laurance WF, Levey DJ, Margules CR, Melbourne BA, Nicholls AO, Orrock JL, Song DX, Townshend JR (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv 1:e1500052. https://doi.org/10.1126/sciadv.1500052
Hatig F (2022) DHARMa: Residual Diagnostics for Hierarchical (Multi-Level / Mixed) Regression Models. R package version 0.4.5. https://CRAN.Rproject.org/package=DHARMa
Juhász CEP, Cursi PR, Cooper M, Oliveira TC, Rodrigues RR (2006) Dinâmica físico-hídrica de uma toposseqüência de solos sob savana florestada (cerradão) em Assis, SP. Rev Bras Cienc Solo 30:401–412. https://doi.org/10.1590/S0100-06832006000300002
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280. https://doi.org/10.1007/s004420100716
Legendre P, Legendre LF (2012) Numerical ecology. Elsevier
Lima-Ribeiro MS (2008) Efeitos De borda sobre a vegetação e estruturação populacional em fragmentos de Cerradão no Sudoeste Goiano, Brasil. Acta Bot Bras 22:535–545. https://doi.org/10.1590/S0102-33062008000200020
Linhares AX (1981) Synanthropy of Calliphoridae and Sarcophagidae (Diptera) in the city of Campinas, São Paulo, Brasil. Rev Bras Entomol 25:189–215
Lopes HS, Tibana R (1987) On Oxysarcodexia (Diptera, Sarcophagidae), with descriptions of five new species, key, list and geographic distribution of the species. Braz J Biol 47:329–347
Majer JD (1987) Invertebrates as indicators for management. In: DA Saunders, GW Arnold, AA Burbidge, AJM Hopkins (eds.) Nature conservation: the role of remnants of native vegetation. Surrey Beatty and Sons Pty Limited with CSIRO and CALM, New South Wales, Australia
Mariluis JC, Schnack JA (1989) Ecology of the blow flies of an eusynanthropic habitat near Buenos Aires (Diptera, Calliphoridae). Eos 65:93–101
Mata RA, Tidon R (2013) The relative roles of habitat heterogeneity and disturbance in drosophilid assemblages (D iptera, D rosophilidae) in the Cerrado. Insect Conserv Divers 6:663–670. https://doi.org/10.1111/icad.12020
Medeiros MB, Miranda HS (2008) Rebrota E mortalidade pós-fogo em espécies de plantas lenhosas do Cerrado em um período de três anos. Edinb J Bot 65:53–68. https://doi.org/10.1017/S0960428608004708
Mello RP (2003) Chave para a Identificação das Formas Adultas das Espécies Da Família Calliphoridae (Díptera, Brachycera, Cyclorrhapha) Encontradas no Brasil. Entomol Vectores 10:255–268
Mello RS, Queiroz MMC, Aguiar-Coelho VM (2007) Population fluctuations of calliphorid species (Diptera, Calliphoridae) in the Biological Reserve of Tinguá, state of Rio De Janeiro, Brazil. Iheringia Ser Zool 97:481–485. https://doi.org/10.1590/S0073-47212007000400019
Miranda HS, Bustamante MMC, Miranda AC (2002) The fire factor. In: Olivera OS, Marquis RJ (eds) The cerrados of Brazil: ecology and natural history of a neotropical savana. Columbia University, New York, pp 51–68
Miranda HS, Neto WN, Neves BMC (2010) Caracterização das queimadas de Cerrado. In: HS Miranda (ed.). Efeitos do regime de fogo sobre a estrutura de comunidades de Cerrado: Projeto Fogo. – Brasília: Ibama, 2010. 144p
Mulieri PR, Schnack JA, Mariluis JC, Torretta JP (2008) Flesh flies species (Diptera: Sarcophagidae) from a grassland and a woodland in a Nature Reserve of Buenos Aires, Argentina. Rev Biol Trop 56:1287–1294 ISSN 0034-7744
Mulieri PR, Patitucci LD, Schnack JA, Mariluis JC (2011) Diversity and seasonal dynamics of an assemblage of sarcophagid Diptera in a gradient of urbanization. J Insect Sci 11:91. https://doi.org/10.1673/031.011.9101
Myers RL (2006) Living with fire–sustaining ecosystems & livelihoods through Integrated Fire Management. The Nature Conservancy, Tallahassee, FL
Nascimento RFO, Silva JODA, Carvalho-Filho FDS (2021) Scavengers flesh flies (Diptera, Sarcophagidae) from two phytophysiognomies in the state of Maranhão, northeastern of Brazil. https://doi.org/10.1590/1676-0611-BN-2021-1192. Biota Neotrop 21
Nogueira C, Valdujo PH, França FGR (2005) Habitat variation and lizard diversity in a Cerrado area of Central Brazil. Stud Neotrop 40:105–112. https://doi.org/10.1080/01650520500129901
Novoa FJ, Altamirano TA, Bonacic C, Martin K, Ibarra JT (2021) Fire regimes shape biodiversity: responses of avian guilds to burned forests in Andean temperate ecosystems of southern Chile. Avian Conserv Ecol 16:22. https://doi.org/10.5751/ACE-01999-160222
Oliveira PS, Marquis RJ (2002) The Cerrado of Brazil: Ecology and Natural History of a neotropical Savanna. Columbia University, New York
Overbeck GE, Vélez-Martin E, Scarano FR, Lewinsohn TM, Fonseca CR, Meyer ST, Müller SC, Ceotto P, Dadalt L, Durigan G, Ganade G, Gossner MM, Guadagnin DL, Lorenzen K, Jacobi CM, Weisser WW, Pilar VD (2015) Conservation in Brazil needs to include non-forest ecosystems. Divers Distrib 21:1455–1460. https://doi.org/10.1111/ddi.12380
Pacheco R, Vasconcelos HL (2012) Habitat diversity enhances ant diversity in a naturally heterogeneous Brazilian landscape. Biodivers Conserv 21:797–809. https://doi.org/10.1007/s10531-011-0221-y
Peralta G, Stevani EL, Chacoff NP, Dorado J, Vázquez DP (2017) Fire influences the structure of plant–bee networks. J Anim Ecol 86:1372–1379. https://doi.org/10.1111/1365-2656.12731
Pinheiro ES, Durigan G (2009) Dinâmica espaço-temporal (1962–2006) das fitofisionomias em unidade de conservação do Cerrado no sudeste do Brasil. Rev Bras Bot 32:441–454. https://doi.org/10.1590/S0100-84042009000300005
Pinheiro CEG, Ortiz JC (1992) Communities of fruit-feeding butterflies along a vegetational gradient in central Brazil. J Biogeogr 19:505–511. https://doi.org/10.2307/2845769
Pivello VR (2011) The Use of Fire in the Cerrado and amazonian rainforests of Brazil: past and Present. Fire Ecol 7:24–39. https://doi.org/10.4996/fireecology.0701024
Ribeiro MC, Figueira JEC (2011) Uma Abordagem Histórica do Fogo no Parque Nacional Da Serra do Cipó, Minas Gerais – Brasil. Biodiversidade Brasileira 1:212–227
Ribeiro JF, Walter BMT (2008) As principais fitofisionomias do Bioma Cerrado. In: Sano SM, SP Almeida (ed) Cerrado: ecologia e flora. Brasília, Embrapa, pp 153–212
Roque F, Da Mata RA, Tidon R (2013) Temporal and vertical drosophilid (Insecta; Diptera) assemblage fluctuations in a neotropical gallery forest. Biodivers Conserv 22:657–672. https://doi.org/10.1007/s10531-013-0433-4
Sano EE, Rosa R, Brito JLS, Ferreira LG (2008) Mapeamento Semidetalhado do uso da terra do Bioma Cerrado. Pesqui Agropecu Bras 43:153–156. https://doi.org/10.1590/S0100-204X2008000100020
Scherber C, Vockenhuber EA, Stark A, Meyer H, Tscharntke T (2014) Effects of tree and herb biodiversity on Diptera, a hyperdiverse insect order. Oecologia 174:1387–1400. https://doi.org/10.1007/s00442-013-2865-7
Silva JMC, Bates JM (2002) Biogeographic patterns and conservation in the South American Cerrado: a tropical savanna hotspot. Bioscience 52:225–234. https://doi.org/10.1641/0006-3568(2002)052[0225:BPACIT]2.0.CO;2
Silva VF, Oliveira-Filho AT, Venturin N, Carvalho WAC, Gomes JBV (2005) Impacto do fogo no componente arbóreo de uma floresta estacional semidecídua no município de Ibituruna, MG, Brasil. Acta Bot Brasilica 19:701–716. https://doi.org/10.1590/S0102-33062005000400005
Silva JLD, Silva RJD, Fernandes IM, Sousa WOD, Vaz-de-Mello FZ (2021) Species composition and community structure of dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae) compared among savanna and forest formations in the southwestern Brazilian Cerrado. Zoologia (Curitiba) 37. https://doi.org/10.3897/zoologia.37.e58960
Silveira L, Henrique F, Rodrigues G, Jácomo ATA, Diniz-Filho JAF (1999) Impact of wildfires on the megafauna of Emas National Park, Central Brazil. Oryx 33:108–114. https://doi.org/10.1046/j.13653008.1999.00039.x
Sousa JRPD, Esposito MC, Carvalho Filho FDS (2010) Composição, abundância e riqueza de Calliphoridae (Diptera) das matas e clareiras com diferentes coberturas vegetais Da Base De Extração Petrolífera, bacia do Rio Urucu, Coari, Amazonas. Revista Brasileira De Entomol 54:270–276. https://doi.org/10.1590/S0085-56262010000200010
Sousa JRP, Esposito MC, Carvalho-Filho FS (2011) Composition, abundance and richness of Sarcophagidae (Diptera: Oestroidea) in forests and forest gaps with different vegetation cover. Neotrop Entomol 40:20–27. https://doi.org/10.1590/S1519-566X2011000100003
Sousa HC, Soares AHSB, Costa BM, Pantoja DL, Caetano GH, Queiroz TA, Colli GR (2015) Fire regimes and the demography of the lizard Micrablepharus atticolus (Squamata, Gymnophthalmidae) in a biodiversity hotspot. South Am J Herpetol 10:143–156. https://doi.org/10.2994/SAJH-D-15-00011.1
Sousa JRP, Carvalho-Filho FS, Juen L, Esposito MC (2016) Evaluating the effects of different vegetation types on necrophagous fly communities (Diptera: Calliphoridae; Sarcophagidae): implications for conservation. PLoS ONE 11:10. https://doi.org/10.1371/journal.pone.0164826
Sousa JRP, Carvalho-Filho FS, Juen L, Esposito MC (2020) The efects of cattle ranching on the communities of necrophagous fies (Diptera: Calliphoridae, Mesembrinellidae and Sarcophagidae) in Northeastern Brazil. J Insect Conserv 24:705–717. https://doi.org/10.1007/s10841-020-00246-y
Sousa JRP, Mendes TP, Carvalho-Filho FS, Juen L, Esposito MC (2021) Diversity of Necrophagous flies (Diptera: Calliphoridae, Mesembrinellidae, and Sarcophagidae) in anthropogenic and preserved environments of five different phytophysiognomies in Northeastern Brazil. Neotrop Entomol 50:537–550. https://doi.org/10.1007/s13744-021-00868-0
Souza FT, Koerner TC, Chlad R (2015) A data-based model for predicting wildfires in Chapada Das Mesas National Park in the state of Maranhão. Environ Earth Sci 74:3603–3611. https://doi.org/10.1007/s12665-015-4421-8
Van Der Werf GR, Randerson JT, Giglio L, Gobron N, Dolman AJ (2008) Climate controls on the variability of fires in the tropics and subtropics. Glob Biogeochem Cycles 22:1–13. https://doi.org/10.1029/2007GB003122
Vasconcelos HL, Maravalhas JB, Cornelissen T (2017) Effects of fire disturbance on ant abundance and diversity: a global meta-analysis. Biodivers Conserv 26:177–188. https://doi.org/10.1007/s10531-016-1234-3
Walter BMT, Ribeiro JF (2010) Diversidade fitofisionômica E o papel do fogo no bioma Cerrado. In: HS, Miranda (eds) (Org.) Efeitos do regime de fogo sobre a estrutura de comunidades de Cerrado: Projeto Fogo. Ibama, Brasília, p 144
Watson SJ, Taylor RS, Nimmo DG, Kelly LT, Clarke MF, Bennett AF (2012) The influence of unburnt patches and distance from refuges on post-fire bird communities. Anim Conserv 15:499–507. https://doi.org/10.1111/j.1469-1795.2012.00542.x
Whitworth TL, Yusseff-Vanegas SA (2019) Revision of the genera and species of the neotropical family Mesembrinellidae (Diptera: Oestroidea). Zootaxa 4659:001–146. https://doi.org/10.11646/zootaxa.4659.1.1
Willmer PG (1982) Thermoregulatory mechanisms in Sarcophaga. Oecologia 53:382–385. https://doi.org/10.1007/BF00389018
Wood SN (2006) Generalized additive models: an introduction with R. Chapman and Hall/CRC
Wu Z, He SH, Yang J, Liang Y (2015) Defining fire environment zones in the boreal forests of northeastern China. Sci Total Environ 518–519:106–116. https://doi.org/10.1016/j.scitotenv.2015.02.063
Yepes-Gaurisas D, Sánchez-Rodríguez JD, Mello-Patiu CA, Echeverri MW (2013) Synanthropy of Sarcophagidae (Diptera) in la Pintada, Antioquia-Colombia. Rev Biol Trop 61:1275–1287 ISSN 0034-7744
Zar JH (2010) Biostatistical Analysis. Pearson Prentice-Hall, Upper Saddle River
Zuur AF, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer Science & Business Media
Acknowledgements
LCR would like to thank the Universidade Estadual do Maranhão for the master’s scholarship and the Fundação de Amparo à Pesquisa e ao Desenvolvimento Científico e Tecnológico do Maranhão (FAPEMA) for financing the project research (Universal Public Call/FAPEMA − 40/2015). We appreciate the support of the Pró-Reitoria de Pesquisa e Pós-Graduação da Universidade Estadual do Maranhão (PPG/UEMA) for funding for the revision of language of the article. APJF is grateful to the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for granting a postdoctoral scholarship (Process No. 88887.659930/2021-00) and Fundação de Amparo à Pesquisa do Estado do Piauí (FAPEPI) for providing funding a research Project (Process nº. 00110.000138/2022-85). LJ is thankful to Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for granting research support through productivity grants (Process No. 304710/2019-9).
Author information
Authors and Affiliations
Contributions
All authors contributed to conceive this study: preparation of the material, sampling, and data analysis; writing, supervision, and review of the manuscript, and approval of final version.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there are no conflicts of interest, either financial or not.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rocha, L.C., Faria, A.P.J., Alvarado, S.T. et al. Cerrado physiognomies in a protected area determine the distribution of necrophagous Diptera. J Insect Conserv 28, 749–761 (2024). https://doi.org/10.1007/s10841-024-00596-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-024-00596-x