Abstract
Macrocystis pyrifera (L.) C. Agardh is a species that forms extensive underwater forests along the coastline of the ecoregion of Magellan. There, this alga is exposed to marked variation in photoperiod, temperature, and irradiance, which are modulated by daily and seasonal climatic variations. This study aims to understand the ecophysiological behavior of M. pyrifera, in a natural forest localized in Puerto del Hambre, Magellan Region of Chile, along spatial (depth) and temporal (season) gradients of physical drivers by analyzing algal responses in terms of the photosynthetic pigments and fluorescence yield. In the apical, middle, and basal fronds, the following photosynthetic parameters were seasonally measured: electron transport efficiency (α), maximum relative rate of electron transport (rETRmax), saturation point (E k ), and pigments such as chlorophyll a (Chl a), chlorophyll c (Chl c), and fucoxanthin. Both seasonal and stratified variations were observed. In autumn, α was decreased in the middle fronds (0.136 ± 0.030 (μmol photons m−2 s−1)−1) with respect to apical and basal fronds of autumn. For parameters such as E k , this decrease was observed relative to the depth gradient, with significant differences (p < 0.05) between distinct fronds. rETRmax was high in the apical fronds in spring, autumn, and winter. High Chl a concentration was maintained in all seasons, while the concentration of Chl c in the apical fronds tended to be lower. The concentration of fucoxanthin remained stable without significant differences between dissimilar types of fronds, for seasons (spring, summer, autumn, and winter). The Chl a/Chl c ratio increased with depth, while the Chl a/fucoxanthin ratio varies seasonally. Variations of light intensity in a natural population in a depth gradient and pigment variations in M. pyrifera with the depth stratification reveal the behavior of these algae in the ecoregion of Magellan where Chl a, through the apical fronds, could be regulating the photosynthetic activity of the plants at stratification level. Furthermore, the increase in Chl c and fucoxanthin towards the middle and basal fronds showed similar trends as those measured for α, thus signifying higher photosynthetic efficiency at greater depth. Overall, our results indicate marked seasonal and depth acclimation to different environmental conditions. This study is the first of its kind for the ecoregion of Magellan in which M. pyrifera represents a keystone species of utmost ecological significance.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Understanding species ecology and evolutionary biology is key for explaining the mechanisms that limit the geographical distribution of macroalgal species (Sexton et al. 2009). It is generally accepted that multiple biotic and abiotic drivers act synergistically to cause the limitations of the respective distributional ranges (Bozinovic et al. 2011). It is, thus, crucial to understand better how environmental factors affect fitness, growth, survival, and reproduction in species over space and time (Sexton et al. 2009). According to Graham et al. (2007), temporal and spatial variability in the structure and dynamics of marine ecosystems seems to be driving the processes that regulate the distribution, abundance, and productivity of the giant kelp Macrocystis pyrifera (L.) C. Agardh. M. pyrifera occurs in both hemispheres; in Chile, it stretches over the latitudinal gradient between Valparaíso and Cape Horn (Macaya and Zucarello 2010).
The photosynthetic light-harvesting mechanisms in macroalgae require constant adjustments to the prevailing environmental conditions (Koch et al. 2016). Adjustments in chlorophyll a + c concentration reflect modulations in the in situ radiation climate and are applied in order to balance light harvesting and photoprotection. Furthermore, in brown macroalgae, carotenoids in the photosynthetic mechanisms may function as accessory and also photoprotective pigments (Frank et al. 1997; Griffiths et al. 2016), and such pigments have been classified as powerful antioxidants and natural antitumor drugs (Pangestuti and Kim 2011; Quitral et al. 2012). Because of the significant presence of fucoxanthin, M. pyrifera potentially is a commercially interesting species (Gupta and Abu-Ghannam 2011). For this reason, in-depth knowledge on the factors that influence the physiological features of M. pyrifera with respect to photoacclimation and the expression of pigment profiles in relation to their success in acclimation is crucial (Delgado-Vargas et al. 2000; Meléndez-Martínez et al. 2007).
The growth and survival of kelp during acclimation processes depends mainly on their ability to deal with fluctuations in the natural environments (e.g., availability of light, temperature, salinity, and nutrients) (Kim and Bhatnagar 2011; Fernandes et al. 2016). The majority of the previous studies on M. pyrifera focused on growth and survival strategies, but recently, more information on how fluctuation in the natural environments can influence the photosynthesis of these organisms has become available (Karsten 2007; Rothäusler et al. 2011a; Karsten 2012; Koch et al. 2016). For example, Gerard (1984) studied light attenuation in the water column within a forest of M. pyrifera, in California, USA, which is largely controlled by self-shading of the individuals, and which requires adjustment of photosynthetic activity with depth. Colombo-Pallotta et al. (2006) studied the photosynthetic efficiency in M. pyrifera with depth: she calculated the light absorption, pigment composition, and thermic dissipation in fronds collected at different depths during spring. For this particular population of M. pyrifera, the authors found that the gradient of light in the water column resulted in high values of ETRmax, Pmax, and E k, in the surface blades, and the opposite in the bottom blades. Another study that shows how photosynthetic activity varies with the depth was conducted by Edwards and Kim (2010), who measured the rates of instantaneous photosynthesis in M. pyrifera at various depths in California, USA, and demonstrated a variation in photosynthetic activity during the day: while the middle and basal fronds exhibited a low photosynthetic rate, without major variations during the day, apical fronds had a high photosynthetic rate, and a variation during the day, demonstrating the acclimatization of these blades to different irradiances.
Rothäusler et al. (2011b) measured the pigment concentration and the photosynthetic activity in floating individuals of M. pyrifera in San Pedro, Chile, and found a high physiological plasticity to different temperatures and UVR as key to enhancing the possibility to persist for long time periods at the sea surface and thus disperse over large distances. Koch et al. (2016) researched the acclimation of M. pyrifera at different depths in central northern Chile by studying the composition of pigments and photosynthetic activity and suggested short-term acclimation between blades of different depths, and a long-term acclimation between different seasons of the year, observing that the xanthophyll cycle pigments, and phlorotannins, are indicators of the different kinds of acclimation processes. These processes of acclimation, in addition to what was described by Macaya and Zucarello (2010) with respect to the genetic diversity found in the population of M. pyrifera located at 42° S, in the Southeast coast of the Pacific, in comparison with the high presence of diversity in the south suggest that the events as the last maximum glacial marked the current diversity in the high seas of the septentrional Patagonia (NPIS, 41°–42° S) (eCOTYPE; sensu Russell 1986). As a result, a high ecophysiological plasticity has developed (Buschmann et al. 2014), where samples of M. pyrifera are capable of enlarging their chlorophyll content, with the purpose of reducing the light saturation, in condition when the nitrogen concentration is high, suggesting that these populations must adjust their physiological performance facing different environmental factors to be able to survive and recuperate the sporophyte population during the following spring. The knowledge about M. pyrifera given by these contributions, in addition to the environmental heterogeneity, and the ecological importance of these wide kelp forests existing in the Magellan region make the development of further studies in order to comprehend the local ecosystem functioning indispensable.
This applies especially when considering that the region of Magellan has the largest sub-Antarctic marine and terrestrial environments on earth, with a heterogeneous setting of fjords and channels (Vergara 2003). These systems show important seasonal variations that affect abiotic parameters such as temperature, salinity, and photoperiod, and these parameters are considered key variables that impact on the survival, reproduction, growth, and ecophysiology of species (Mansilla et al. 2004; Ramlov et al. 2012; Ojeda 2013).
We hypothesize that the variations in light intensity with depth and season will result in adjustments in the photosynthetic parameters and pigment concentrations as a prerequisite of the ecological success of M. pyrifera. We tested these hypotheses in field and laboratory measurements, by measuring the ecophysiological responses of M. pyrifera by (i) measuring the penetration of light intensity in the forest of M. pyrifera and (ii) measuring photosynthetic parameters and pigment concentrations in blades at different depths in the course of the seasons.
Material and methods
Macroalgal material
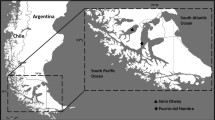
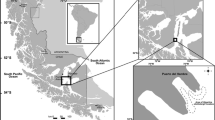
Apical (0 m), middle (1.5–2.0 m), and basal (2.5–3.0 m) fronds from seven young individuals of the giant kelp Macrocystis pyrifera (Ochrophyta, Phaeophyceae) were collected from a population at Puerto del Hambre (53° 36′ S, 70° 55′ W), Strait of Magellan, Chile (Fig. 1), during spring (December), summer (March), autumn (May), and winter (July) in 2014–2015 (Fig. 1). Fronds from different water depths were collected from the same individuals. Additionally, seven fertile sporophylls per individual were sampled in winter.
Right after collecting, the specimens were transferred in a cooler box filled with local seawater (32 PSU) to the laboratory in Punta Arenas, 60 km from the sampling site. In the laboratory, the specimens were carefully cleaned of epiphytes and associated animals. They were maintained for 24 h in a temperature-controlled walk-in cabinet. Both temperatures and photoperiods were adjusted according to the season. This acclimation was carried out to lessen the stress for the specimens that had been caused during transport from the beach to the laboratory.
Spectroradiometric measurements
The spectral composition of the underwater light regimes inside and outside of a M. pyrifera forest at Fuerte Bulnes (53.62° S, 70.92° W), ∼1.1 km off the sampling site in Puerto del Hambre, was measured using a UV-VIS hyper-spectral radiometer with a 256-channel photodiode array and a 2π cosine-corrected collector type (RAMSES ACC; TriOS Mess- und Datentechnik GmbH, Rastede, Germany) in March 2005 (Rautenberger et al. 2009). Underwater irradiances were scanned between 280.1 and 700.7 nm and extracted by the operating software (MSDA_XE, v7.0, TriOS Mess- und Datentechnik GmbH). Prior to all scans, the instrument was warmed up for some minutes and adjusted to the surrounding air and seawater temperature. The hyper-spectral radiometer was carried to the position of interest before six scans per water depth were performed (n = 6). Scans outside the giant kelp forest were carried out at 1.4 and 2.8 m depth, but the spectral composition is very similar. Inside the M. pyrifera forest measurements were performed at 1, 2, and 4 m depth. Underwater irradiances were calculated as described by Rautenberger and Bischof (2016).
Pigment analysis
Both photosynthetic and major accessory pigments were extracted from the specimens directly after the photosynthetic performances were measured according to Mansilla et al. (2004). Prior to pigment extraction with dimethyl sulfoxide (DMSO), the fronds were dried with absorbent paper. Macroalgal fragments of 0.125 g dry weight (DW) were incubated in 1 mL of ≥99% DMSO for 15 min. Chlorophyll a (Chl a), chlorophyll c (Chl c) and fucoxanthin were analyzed photometrically (Genesys UV) at room temperature. Readings at 750.0 nm were used as a correction factor for scattered light. Both Chl a (1), Chl c (2), and fucoxanthin (Fucox) (3) contents (mg g−1 DW) were calculated from measured absorbances at 480.0 nm (A480), 582.0 nm (A582), 631.0 nm (A631), and 665.0 nm (A665) after Seely et al. (1972):
Measurements of photosynthesis
The photosynthetic activities of all fronds throughout the year were measured using a DIVING-PAM chlorophyll fluorometer (Walz GmbH, Germany). Fluorescence was measured by applying a short saturation pulse (>9000 μmol photons m−2 s−1, 0.8 s) in addition to a weak measuring light. Subsequently, the specimens were exposed to a range of actinic light intensities (E: 7–2950 μmol photons m−2 s−1), which increased incrementally after short saturation pulses (≥9000 μmol photons m−2 s−1, 0.8 s) every 30 s. The effective PSII-quantum yield (φPSII) was calculated from maximal (Fm′) and terminal (Ft) fluorescence at each actinic light intensity. The relative photosynthetic electron transport rates (rETR) were calculated by multiplying φPSII with the corresponding actinic light intensity and plotted against the incident actinic light intensities. These hyperbolic rETR vs E curves were fitted according to the model of Platt et al. (1980) (4) using KaleidaGraph v4.0 (Synergy Software, USA):
Anderson et al. (1995) describes this equation as follows: P S is the light-saturated rate of photosynthesis, α is the light-limited rate of photosynthesis, and β represents the extent of photoinhibition.
In order to perform correct measurements of the rapid light curves (RLC) in the measured samples, we take the considerations of Beer et al. (2014).
Data analysis
Means and standard deviations (SDs) were calculated from seven replicates per measurement (n = 7). To analyze the data on pigment concentration, we applied a two-way factorial PERMANOVA. For univariate variables, the Euclidean distance between pairs of observations was calculated (Claudet et al. 2006). The PERMANOVA analysis was performed using the R package “vegan.” For the photosynthetic parameter data, these were analyzed using a two-way ANOVA (factors: seasons, and depth, 2 × 2), Post hoc comparisons of means were assessed with Tukey HSD; these statistical analyses were performed using JMP 12.2.0 software (SAS Institute Inc., USA).
Results
Underwater spectral composition
The spectral composition of the underwater irradiances outside and inside of a forest of M. pyrifera was significantly different (Fig. 2). The spectral composition outside the giant kelp forest resembles the coastal Jerlov water types 1 to 3 where the violet-blue region of the spectrum (380–480 nm) is strongly attenuated by the absorption of Chl a from phytoplankton in the water column. The seawater itself attenuates light >550 nm with a strongest contribution in the red range (620–700 nm) of the visible spectrum. Consequently, the remaining underwater light spectrum outside the forest dominated in the green range between 480 and 570 nm. Therefore, M. pyrifera can only use light of this wavelength range for photosynthesis by its photosynthetic and accessory pigments. Inside the giant kelp forest, the spectral composition of the highest underwater irradiance at 1 m depth was similar to those at 2 and 4 m depth, but different to that outside the forest. The attenuation of the wavelength range between 340 and 480 nm can be ascribed to the presence of UV-absorbing polyphenolic compounds (e.g., phlorotannins) and the absorbance by Chl a, Chl c, and fucoxanthin in M. pyrifera. The remaining green wavelength range (490–580 nm) can be little absorbed by the macroalga’s pigments. At 2 and 4 m depth, the spectrum of the underwater light is similar to that at 1 m due to further absorption by phlorotannins and pigments. However, it is striking that the irradiance below 2 m depth did not further decrease due to either the scarce presence or the absence of macroalgal blades.
Pigment composition
The pigment concentration in M. pyrifera reaches significantly different levels throughout the four seasons and among frond types (apical, middle, and basal). The concentration of Chl a in apical fronds was high in all seasons.
For the pigment Chl a, significant differences (PERMANOVA, p < 0.05) were observed when comparing fronds of the same type, between the analyzed seasons, the apical fronds of the winter season differing from the apical fronds of the spring seasons, summer, and autumn (Table 1), and the same occurred in the middle and basal fronds. Furthermore, other significant differences (PERMANOVA, p < 0.05) were observed when analyzing between different types of fronds, in summer seasons, and winter.
The Chl c concentration was similar in spring and summer (Fig. 3). This pigment concentration is the second highest in comparison with Chl a, showing a decreasing tendency with increasing depth, while being more similar across frond types in autumn and winter. Chl c decreases in winter, especially in the basal fronds (0.024 ± 0.013 mg g−1 DW).
For the pigment Chl c, significant differences were observed between apical fronds of winter with that from the other seasons (PERMANOVA, p < 0.05; Table 1). The same was observed for the basal fronds. Other significant differences were observed in the analysis (PERMANOVA, p < 0.05, Table 1) when comparing the different types of fronds at each season; finally, significant differences were observed.
The concentration of fucoxanthin among frond types remains similar among spring, summer, and autumn; winter was the season with the lowest concentration especially in apical fronds: 0.049 ± 0.023 mg g−1 DW (Fig. 3). With respect to fucoxanthin, significant differences were observed when comparing apical fronds in different seasons (all p values below 0.05, PERMANOVA; Table 1); the same occurred in middle and basal fronds. However, no significant differences were observed between different types of fronds for each sampled season.
The Chl a/Chl c ratio increases with depth (Fig. 4). This situation occurs in spring, summer, and autumn, while in winter the lowest ratio occurs in the middle fronds and the highest ratio in the apical fronds. The values of the Chl a/fucoxanthin ratio do not vary among spring, summer, autumn (Fig. 4), while in winter the ratio increases, with the middle fronds having the highest values (4:5 pigment ratio; Fig. 4).
Photosynthetic parameters
The electron transport efficiency (α), relative rate of electron transport (rETRmax), and saturation point (E k ) are shown in Figs. 5, 6, and 7. The initial slopes of the ETR-E curves (i.e., α values) do not show significant differences among fronds of the same type in different seasons, but do so among frond types (between apical and middle in autumn, and between apical with middle and basal fronds in summer (two-way ANOVA test, all p values below 0.05; Table 2). The middle fronds had the lowest α values, in particular in autumn (α = 0.136 ± 0.030 (μmol photons m−2 s−1)−1).
The rETRmax values were higher in spring than in the other seasons across all frond types. The apical fronds had higher rETRmax values in spring, autumn, and winter (Fig. 6), which range between 98.0 ± 40.6 and 32.0 ± 12.4 in spring and summer, respectively. Seasonal differences were recorded in apical fronds (two-way ANOVA test, p < 0.05), mainly the spring season with summer and autumn seasons (Table 2); but the analyses between different types of fronds per season do not show significant differences.
The apical fronds display higher E k parameter in spring and winter (E k in spring = 521.3 ± 305.5 μmol photons m−2 s−1; Fig. 7), with E k decreasing towards the basal fronds, whereas the middle fronds record their highest E k values in spring and autumn, thus reaching 338.8.0 ± 102.3 μmol photons m−2 s−1 (Fig. 7). Seasonal differences were recorded in apical fronds (p < 0.05) between all seasons sampled (Table 2), and only in summer were significant differences between types of fronds (middle-basal) found (two-way ANOVA test, p < 0.05).
Discussion
Classical studies carried out for “kelp” or M. pyrifera showed examples for the variation in pigment composition, indicative for acclimation processes. For example, Wheeler (1980) reported how pigments such as Chl a, Chl c, and fucoxanthin vary in juvenile M. pyrifera, after the plants had been transplanted to different depths. Such variation and acclimation processes have also been reported for other algae such as Sargassum (Gao and Umezaki 1988) or Ascophyllum nodosum (Ramus et al. 1977).
The pigment composition of M. pyrifera reported in this seasonal study showed high concentration of Chl a in all types of fronds, with respect to the other pigments, and in all seasons, but the highest concentration occurred in the apical fronds, during spring, summer, and autumn, while the middle and basal fronds exhibited a lower concentration of Chl a during spring, summer, and winter. Significant differences were registered in the analysis of apical fronds between seasons, and differences in the concentration of Chl a among different types of fronds per season were observed. These results do not coincide with the works of Lichtenthaler and Babani (2004) and Edwards and Kim (2010), who did not find differences in the pigment concentrations of distinct types of frond of M. pyrifera.
The difference in the concentration of Chl a in apical fronds can be explained by the impact of seasonality characteristic of high latitude that affects the region (Ojeda et al. 2014), with marked changes in the photoperiod and the temperature (Gunnarsson and Ingólfsonn 1995), which directly affect the apical fronds found in the canopy. They are further influenced by irradiance, as measured in the M. pyrifera forest, where values differed to those obtained outside the forest. The latter indicate a high irradiance that affects the apical fronds directly. At increasing depths, the irradiance inside the forest diminishes. This decrease has a direct impact on the middle fronds found in the water column and on the basal fronds on the ground. This means that the apical fronds capture most of the light for the photosynthetic process. Colombo-Pallotta et al. (2006) suggest that the apical fronds are better acclimated to high light, while the middle and basal fronds are acclimated to low irradiance. The obtained results show an intra-specific regulation of the plant in regards to its photosynthetic capacity and possibly its surrounding.
This is related with the electron transport efficiency α measured in M. pyrifera, which shows a high α in the apical fronds in the seasons summer and autumn, with variations in the stratification, being the lowest values observed in the middle fronds throughout the seasons. This concurs with the findings of Beer et al. (2014) who report that α is high when the pigment concentration increases. In their study, this was measured in the apical fronds. In species found in more shallow depths, a rapid photosynthetic regeneration occurs, which is explained by the efficient response in the event of environmental factors that cause stress (e.g., high solar radiation, desiccation, and temperatures) (Skene 2004). In contrast, the apical fronds are exposed to a variety of abiotic, stress-generating factors and show therefore a higher α. This could be an acclimation response to these factors. In the case of the Chl a/Chl c ratio of M. pyrifera, the size of the pigment antennae tends to increase with greater depth. This has been observed in spring, summer, and autumn and is due to a decrease in irradiance towards these deeper regions. The increased size of the antennae in relation to greater depth has also been reported for species such as Palmaria decipiens (Becker et al. 2010). This variation in size occurs across the different types of fronds, and has been reported by Koch et al. (2016) as a rapid acclimation to changing light conditions. The size of the antennae in the ratio of Chl a/fucoxanthin only varies in winter and is larger in the middle and apical fronds.
With respect to the accessory pigments such as Chl c and fucoxanthin, these are present in higher concentrations in the middle and basal fronds. They are subjected to lower irradiance levels as observed in the light curves measured inside the M. pyrifera forest. This concurs with findings in the Antarctic region reported by Yokoya et al. (2007) and Castelo et al. (2012); the last authors stated that during a drop in the photon flux conditions the algae change so that they have to capture most of the light. In the reaction centers, accessory pigments therefore are critical in the capture, absorption, and transfer of radiance energy.
Similarly, rETRmax and E k were higher in the apical fronds during spring and winter while at the same time, the concentration of Chl c was lower. This is in agreement with findings for the same species made by Wheeler (1980), Colombo-Pallotta et al. (2006), and Koch et al. (2016). It demonstrates an intra-specific variation of this alga in terms of its acclimation to varying irradiances as measured inside and outside of the M. pyrifera forest. The photosynthetic efficiency in a single organism is regulated based on depth and variations in irradiance (Colombo-Pallotta et al. 2006). Similarly, E k is intermediate to low saturation levels. This has also been observed by Reiskind et al. (1997) and Colombo-Pallotta et al. (2006). They reported that the photosystem of the basal fronds becomes saturated under lower light intensity, although they point out the low saturation point of the apical fronds in summer when the fronds are subjected to high irradiance. These factors might cause photoinhibition. From a seasonal point of view, the decrease in Chl c and fucoxanthin in winter might be caused by the prevailing environmental conditions as observed in situ, as the cover of the M. pyrifera forest decreases while the water is entirely clear. Therefore, light is able to reach greater depths and thus the middle and basal fronds. The necessity for accessory pigments abates, demonstrating the species’ capacity to acclimate to seasonal changes of light. The acclimation of this species has been studied before; it might happen on a scale of hours, as has been reported by Edwards and Kim (2010) and Cabello-Pasini et al. (2000), or weeks (Ralph and Gademann 2005). Variations in the pigment composition and size of antennae can increase the absorption spectra and promote an optimization of the photosynthetic process. In this way, the range of the species distribution along a certain bathymetric profile is extended (Guimarães 2003).
It should be noted that, due to the photosynthetic parameters, the results for the different fronds of M. pyrifera could show the capacity of this macroalga to transfer organic and inorganic compounds from and to the canopy (Manley 1984). Although the apical fronds have the highest electron transport efficiency, this could be related to a photoprotective specialization and an efficient use of energy. Meanwhile, the middle and basal fronds provide nutrients to the whole plant (Colombo-Pallotta et al. 2006).
In conclusion, our study reveals photo-acclimatory trends in M. pyrifera present in the Magellan ecoregion where light availability along the depth gradient results in an increase of Chl a in apical fronds, a high α and E k , and an increase in the size of the pigment antennae with depth, practically in all the seasons. There is a seasonal acclimation to different environmental conditions, present in the sub-Antarctic region. This can be observed in the variation of Chl c and fucoxanthin over the seasons in middle and basal fronds; this last carotenoid with a high importance for its properties shows photoprotective pigments, related with the increasing size of the pigment antennae in the basal fronds and in the ratio Chl a/Chl c, and the highest electron transport efficiency shown in the middle fronds. The regulation is shown, in the apical fronds for the highest concentration of Chl a, how rETRmax and E k show the importance of the apical fronds in the eco-physiology and of the plants of M. pyrifera. Finally, this study is the first of its kind for the eco-region of Magellan to demonstrate the eco-physiological acclimation patterns which allow the algae to thrive in the particularly sensitive region of Magellan.
References
Anderson JM, Chow WS, Park YI (1995) The grand design of photosynthesis: acclimation of the photosynthetic apparatus to environmental cues. Photosynth Res 46:129–139
Becker S, Graeve M, Bishof K (2010) Photosynthesis and lipid composition of the Antarctic endemic rhodophyte Palmaria decipiens, effects of changing light, and temperature levels. Polar Biol 33:945–955
Beer S, Björk M, Beardall J (2014) Photosynthesis in the marine environment. Wiley, Oxford, pp 61–64
Bozinovic F, Calosi P, Spicer J (2011) Physiological correlates of geographic range in animals. Annu Rev Ecol Evol Syst 42:155–179
Buschmann A, Pereda S, Varela D, Rodriguez J, López A, González L, Schilling M, Henríquez E, Hernández M (2014) Ecophysiological plasticity of annual populations of giant kelp (Macrocystis pyrifera) in a seasonally variable coastal environment in the Northern Patagonian Inner Seas of Southern Chile. J Appl Phycol 26:837–847
Cabello-Pasini A, Aguirre-von-Wobeser E, Figueroa F (2000) Photoinhibition of photosynthesis in Macrocystis pyrifera (Phaeophyceae), Chondrus crispus (Rhodophyceae) and Ulva lactuca (Chlorophyceae) in outdoor culture systems. J Photochem Photobiol 57:169–178
Castelo D, Trigueiro T, Colepicolo P, Marinho Soriano E (2012) Seasonal changes in the pigment composition of natural population of Gracilaria domingensis (Gracilariales, Rhodophyta). Braz J Pharmacog 22:874–880
Claudet J, Pelletier D, Jouvenel JY, Bachet F, Galzin R (2006) Assessing the effects of marine protected area (mpa) on a reef fish assemblage in a Northwestern Mediterranean marine reserve: identifying community-based indicators. Biol Conserv 130:349–369
Colombo-Pallotta M, García Mendoza E, Ladah L (2006) Photosynthetic performance, light absorption, and pigment composition of Macrocystis pyrifera (Laminariales, Phaeophyceae) blades from different depths. J Phycol 42:1225–1234
Delgado-Vargas F, Jiménez A, Paredes-López O (2000) Natural pigments: carotenoids, anthocyanins, and betalains—characteristics, biosynthesis, processing, and stability. Crit Rev Food Sci Nutr 40:173–289
Edwards M, Kim K (2010) Diurnal variation in relative photosynthetic performance in giant kelp Macrocystis pyrifera (Phaeophyceae, Laminariales) at different depths as estimated using PAM fluorometry. Aquat Bot 92:119–128
Fernandes F, Barbosa M, Oliveira AP, Azevedo IC, Sousa-Pinto I, Valentão P, Andrade PB (2016) The pigments of kelps (Ochrophyta) as part of the flexible response to highly variable marine environments. J Appl Phycol 28:3689–3696
Frank H, Chynwat V, Desamero R, Farhoosh R, Erickson J, Bautista J (1997) On the photophysics and photochemical properties of carotenoids and their role as light–harvesting pigments in photosynthesis. Pure Appl Chem 69:2117–2124
Gao K, Umezaki I (1988) Comparative photosynthetic capacities of the leaves of upper and lower parts of Sargassum plants. Bot Mar 31:231–236
Gerard V (1984) The light environment in a giant kelp forest: influence of Macrocystis pyrifera on spatial and temporal variability. Mar Biol 84:189–195
Graham M, Vasquez J, Buschmann A (2007) Global ecology of the giant kelp Macrocystis: from ecotypes to ecosystems. Oceanogr Mar Biol 45:39–88
Griffiths M, Harrison S, Smit M, Maharajh D (2016) Major commercial products from micro–and macroalgae. In: Bux F, Chisti Y (eds) Algae biotechnology. Springer, Cham, pp 269–300
Guimarães SMPB (2003) Uma análise da diversidade da flora marinha bentônica do estado do Espírito Santo, Brasil. Hoehnea 30:11–19
Gunnarsson K, Ingólfsonn A (1995) Seasonal changes in the abundance of intertidal algae in Southwestern Iceland. Bot Mar 38:69–77
Gupta S, Abu-Ghannam N (2011) Bioactive potential and possible health effects of edible brown seaweeds. Trends Food Sci Technol 22:315–326
Karsten U (2007) Research note: salinity tolerance of Arctic kelps from Spitsbergen. Phycol Res 55:257–262
Karsten U (2012) Seaweed acclimation to salinity and desiccation stress. In: Wiencke C, Bishoff K (eds) Seaweed biology. Springer, Berlin, pp 87–107
Kim S, Bhatnagar I (2011) Physical, chemical, and biological properties of wonder kelp—Laminaria. Adv Food Nutr Res 64:85
Koch K, Thiel M, Hagen W, Graeve M, Gómez I, Jofre D, Hoffman L, Tala F, Bischof K (2016) Short and long term acclimation patterns of the giant kelp Macrocystis pyrifera (Laminariales, Phaeophyceae) along a depth gradient. J Phycol 52:260–273
Lichtenthaler HK, Babani F (2004) Light adaptation and senescence of the photosynthetic apparatus. Changes in pigment composition, chlorophyll fluorescence parameters and photosynthetic activity. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a fluorescence: a signature of photosynthesis. Springer, Dordrecht, pp 713–736
Macaya EC, Zucarello GC (2010) Genetic structure of the giant kelp Macrocystis pyrifera along the southeastern Pacific. Mar Ecol Prog Ser 420:103–112
Manley S (1984) Micronutrient uptake and translocation by Macrocystis pyrifera. J Phycol 20:192–201
Mansilla A, Palacios M, Aguilar S (2004) Efecto de la Salinidad en el Desarrollo Inicial de Sarcothalia Crispata (Bory) Leister (Rhodophyta, Gigartinales) Bajo Condiciones de Laboratorio. An Inst Patagonia 32:13–23
Meléndez-Martínez A, Vicario I, Heredia F (2007) Pigmentos carotenoides: consideraciones estructurales y fisicoquímicas. Arch Latinoam Nutr 57:109–117
Ojeda J (2013) Dinámica estacional de macroalgas y moluscos intermareales y su relación con el conocimiento tradicional ecológico yagán, en canales subantárticos del Cabo de Hornos: una aproximación biocultural desde la filosofía ambiental de campo. Postgraduate Thesis, Universidad de Magellan, Punta Arenas, Chile, 145 pp
Ojeda J, Rosenfeld S, Marambio J, Rozzi R, Mansilla A (2014) Patrones estacionales y espaciales de la diversidad de moluscos intermareales de Bahía Róbalo, canal Beagle, Reserva de la Biosfera Cabo de Hornos, Chile. Rev Biol Mar Ocean 49(3):493–509
Pangestuti R, Kim S (2011) Biological activities and health benefit effects of natural pigments derived from marine algae. J Funct Foods 3:255–266
Platt T, Gallegos CL, Harrison WG (1980) Photoinhibition of photosynthesis in natural assemblages of marine phytoplankton. J Mar Res 38:687–701
Quitral V, Morales C, Sepúlveda M, Schwartz M (2012) Propiedades nutritivas y saludables de algas marinas y su potencialidad como ingrediente funcional. Rev Chil Nutr 39:196–202
Ralph PJ, Gademann R (2005) Rapid light curves: a powerful tool to assess photosynthetic activity. Aquat Bot 82:222–237
Ramlov F, de Souza J, Farias A, Maraschin M, Horta P, Yokoya N (2012) Effects of temperature, salinity, irradiance, and nutrients on the development of carposporelings and tetrasporophytes in Gracilaria domingensis (Kütz.) Sonder ex Dickie (Rhodophyta, Gracilariales). Bot Mar 55:253–259
Ramus J, Lemons F, Zimmerman C (1977) Adaptation of light-harvesting pigments to downwelling light and the consequent photosynthetic performance of the eulittoral rockweeds Ascophyllum nodosum and Fucus vesiculosus. Mar Biol 42:293–293
Rautenberger R, Bischof K (2016) Dynamic summer solar radiation in Antarctic coastal ecosystems and its effects on photosynthesis of the endemic Antarctic brown macroalga Desmarestia menziesii (Phaeophyceae). Algol Stud 151-152:123–150
Rautenberger R, Mansilla A, Gómez I, Wiencke C, Bischof K (2009) Photosynthetic responses to UV- radiation of intertidal macroalgae from the Strait of Magellan (Chile). Rev Chil Hist Nat 82:43–61
Reiskind JB, Madsen TV, Van Ginkel LC, Bowes G (1997) Evidence that inducible C4-type photosynthesis is a chloroplastic CO2- concentrating mechanism in Hydrilla, a submersed monocot. Plant Cell Environ 20:211–220
Rothäusler E, Gómez I, Hinojosa I, Karsten U, Tala F, Thiel M (2011a) Physiological performance of floating giant kelp Macrocystis pyrifera (Phaeophyceae): latitudinal variability in the effects of temperature and grazing1. J Phycol 47:269–281
Rothäusler E, Gómez I, Karsten U, Tala F, Thiel M (2011b) Physiological acclimation of floating Macrocystis pyrifera to temperature and irradiance ensures long–term persistence at the sea surface at mid–latitudes. J Exp Mar Biol Ecol 405:33–41
Russell G (1986) Variation and natural selection in marine macroalgae. Oceanogr Mar Biol Annu Rev 24:309–377
Seely GR, Duncan MJ, Vidaver WE (1972) Preparative and analytical extraction of pigments from brown algae with dimethyl sulfoxide. Mar Biol 12:184–188
Sexton J, McIntyre P, Angert RK (2009) Evolution and ecology of species range limits. Annu Rev Ecol Evol Syst 40:415–436
Skene K (2004) Key differences in photosynthetic characteristics of nine species of intertidal macroalgae are related to their position on the shore. Can J Bot 82:177–184
Vergara J (2003) Tipología y clasificación de fiordos y piedmonts submarinos de Magellan. Chile Invest Geogr Chile 37:21
Wheeler WN (1980) Pigment content and photosynthetic of the frond of Macrocystis pyrifera. Mar Biol 56:97–102
Yokoya NS, Necchi O Jr, Martins AP, Gonzalez SF, Plastino EM (2007) Growth responses and photosynthetic characteristics of wild and phycoerythrin-deficient strains of Hypnea musciformis (Rhodophyta). J Appl Phycol 19:197–205
Acknowledgements
The authors wish to thank to the project FONDECYT 1140940 “Macroalgal adaptive radiation: potential links to ecological niche diversity in the ecoregion of Magallanes and Chilean Antarctic.” We are thankful for the graduate scholarships by the Institute of Ecology and Biodiversity granted to FM (ICM, P05-002) and JPR (ICM, P05-002) and to JM (PFB-23-2008) and SR (ICM P05-002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marambio, J., Rodriguez, J.P., Mendez, F. et al. Photosynthetic performance and pigment composition of Macrocystis pyrifera (Laminariales, Phaeophyceae) along a gradient of depth and seasonality in the ecoregion of Magellan, Chile. J Appl Phycol 29, 2575–2585 (2017). https://doi.org/10.1007/s10811-017-1136-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-017-1136-0