Abstract
Common carp (Cyprinus carpio, carp) are a widespread and ecologically destructive invasive fish species. Carp management is critical for maintaining healthy aquatic ecosystems, and many control options are available, but most have proven to be ineffective. Carp abundances have increased at The Nature Conservancy’s Emiquon Preserve, Illinois, since its restoration in 2007 despite management efforts to suppress this species. We conducted a comparative diet study in Illinois, Tennessee, and Wisconsin to test whether bowfin (Amia calva), spotted gar (Lepisosteus oculatus, gar), and largemouth bass (Micropterus salmoides) commonly preyed upon carp. We focused on bowfin and gar because they are hypoxia-tolerant, similar to carp. We also assessed whether specific fish community characteristics were correlated with carp relative abundances. We found no evidence that bowfin, gar, and bass consumed large numbers of carp. However, carp may be limited in some ecosystems (e.g., Reelfoot Lake, Tennessee) through alternative mechanisms associated with bowfin, gar, bass, and bluegill (Lepomis macrochirus) included in a diverse native fish community.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nonnative species have caused major economic and ecological losses (Nentwig, 2007). Invasive species are the second largest threat to terrestrial and freshwater biodiversity and have cost $125 billion annually in the USA alone for control efforts (Tockner & Stanford, 2002; Allendorf & Lundquist, 2003). About a half million invasive species have been introduced worldwide, 50,000 of which have been introduced to the USA, including at least 138 fishes (Nentwig, 2007). Invasive fishes have negatively affected aquatic ecosystems and cost USA fisheries about $5 billion in damages annually (Nentwig, 2007). Following colonization, control or eradication of invasive species has often proven costly or ineffective (Homans & Smith, 2013; Simberloff et al., 2013; Gaeta et al., 2015). Thus, preventing invasive species introductions is paramount for conserving native ecosystems and biodiversity (Vander Zanden et al., 2010; Gaeta et al., 2015).
Common carp (Cyprinus carpio, carp) are one of the most widespread and destructive invasive species in the world (Weber & Brown, 2011). Carp are native to the Ponto-Caspian region of Eurasia, were introduced to North America during the late 1800s, and are established on every continent except Antarctica and in every state in the USA except Alaska (Penne & Pierce, 2008; Bajer et al., 2009). Due to their tolerance of a wide range of temperature, salinity, and dissolved oxygen concentrations, carp are adapted to a variety of habitats (Bajer & Sorensen, 2010). Carp have been directly implicated in degrading aquatic ecosystems due to their benthic foraging behavior, which uproots aquatic macrophytes and suspends sediments and nutrients into the water column. For example, Hennepin and Hopper lakes, Illinois experienced major declines in aquatic macrophytes, and waterfowl use shortly after restoration, due to rapid population growth of carp (Bajer et al., 2009). Carp may also indirectly decrease water clarity by reducing macroinvertebrate and zooplankton abundances causing an increase in phytoplankton (Bajer & Sorensen, 2010; Weber et al., 2010). Carp have also reduced native fish abundances through the destruction of spawning and nursery habitats and through competition for food resources (Weber & Brown, 2011).
Management of carp is often critical for maintaining healthy aquatic ecosystems and many control options are available. Rotenone has often been used as a management tool to poison carp but has proven to be ineffective (Bajer et al., 2009; VanMiddlesworth et al., 2014). For example, carp survived a rotenone application in a floodplain restoration project at Hennepin and Hopper lakes, Illinois (Bajer et al., 2009). Other control options for carp have included baiting, barriers, water drawdown, electrofishing, and netting but have also proven to be ineffective (Weber & Brown, 2009).
One of the largest floodplain restoration projects in the USA, The Nature Conservancy’s Emiquon Preserve (EP), is located along the Illinois River in west-central Illinois. The EP was disconnected from the Illinois River by levees, drained, and used for agriculture during the early 1920s. The Nature Conservancy purchased this property in 2000, and aquatic restoration began in 2007. Rotenone was used to remove invasive fishes such as carp and grass carp (Ctenopharyngodon idella). Precipitation then naturally filled the area. Native fishes were stocked during 2007–2011 based on historical documentations and included piscivores such as bowfin (Amia calva), largemouth bass (Micropterus salmoides, bass), longnose gar (Lepisosteus osseus), and spotted gar (L. oculatus, gar) (Havera et al., 2003; VanMiddlesworth et al., 2016). Bass were stocked at high abundances (>1.2 million fry and brood stock), with the goal of inhibiting establishment of any nonnative fishes that may have survived the rotenone application through predation. Diet analyses of bass showed that they did not select for carp but instead selected more available and energetically profitable prey items such as other fishes, terrestrial invertebrates, and odonates (Michaels, 2011; VanMiddlesworth et al., 2016). Despite piscivore stockings, and although total catch of carp has remained low relative to native fish abundances, relative abundance of carp has increased during 2007–2014 at the EP (VanMiddlesworth et al., 2014, 2016).
A critical component in efforts to control carp may be realized through decreasing their reproductive output. Bajer & Sorensen (2010) found that carp spawn in shallow Midwestern lakes that experience winter hypoxia, which may reduce predation on eggs and larvae by hypoxia-intolerant native fishes, eventually leading to carp dominance. Thus, we hypothesized that hypoxia-tolerant native fishes such as bowfin and gar may be able to control carp through direct predation. We tested this hypothesis with the prediction that carp would be present in the diets of large-sized individuals of these species. Bowfin and gar are primitive piscivorous fishes that are typically unmanaged, and their ecological role in aquatic ecosystems is relatively understudied (Scarnecchia, 1992). We also assessed whether specific fish community characteristics were correlated with carp relative abundances.
Materials and methods
Study sites
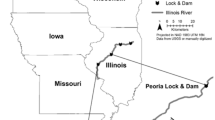
The EP is located along the Illinois River near Lewistown, Fulton County, Illinois and encompasses >2,700 ha (Fig. 1). Two former floodplain lakes were reinundated as the restoration proceeded at the EP, historically referred to as Thompson and Flag lakes. Only Thompson Lake was sampled in our study. Flag Lake was not sampled due to insufficient water depth and high submersed aquatic macrophyte densities that fouled our sampling gears. Bottom substrate was silt/clay with an aquatic macrophyte community composed of emergent, floating-leaved, and primarily submersed species.
Map showing locations of The Nature Conservancy’s Emiquon Preserve, Illinois (N4471044, E749023 UTM Z15); Reelfoot Lake, Tennessee (N4028795, E288768 UTM Z16); Upper Phantom Lake (N4745900, E390809 UTM Z16), Lower Phantom Lake (N4744949, E389638 UTM Z16), Eagle Spring Lake (N4745716, E382753 UTM Z16), and Lulu Lake (N4743277, E381472 UTM Z16), Wisconsin
Reelfoot Lake is located along the Mississippi River near Samburg, Lake and Obion Counties, Tennessee, and has an aquatic surface area of >6,070 ha (Tennessee Wildlife Resources Agency, 2009, 2013) (Fig. 1). Substrate was silt/clay with an aquatic macrophyte community composed of submersed and primarily emergent and floating-leaved species.
Upper Phantom, Lower Phantom, and Eagle Spring lakes are located in Waukesha County, while Lulu Lake is located in adjacent Walworth County and connected to Eagle Spring Lake by a 0.5-km channel (Heussner et al., 2008, 2009) (Fig. 1). All four lakes are located near Mukwonago, Wisconsin. Upper Phantom Lake has an aquatic surface area of 44 ha, Lower Phantom Lake has an aquatic surface area of 150 ha, Eagle Spring Lake has an aquatic surface area of 112 ha, and Lulu Lake has an aquatic surface area of 38 ha (Eagle Spring Lake Management District, 2011; Phantom Lakes Management District, 2012). All four lakes had silt/clay substrates with aquatic macrophyte communities composed of emergent, floating-leaved, and primarily submersed species.
To examine bowfin and gar predation, we conducted a comparative study at the EP, Reelfoot Lake, and four southeastern Wisconsin lakes by assessing the feeding habitats of bowfin, gar, and bass. Reelfoot Lake is similar to the EP: both are shallow, former floodplain lakes that are disconnected from large rivers and have fish communities including carp. The native macrophyte and fish community of Reelfoot Lake appears to have co-existed with carp for >150 years (Tennessee Wildlife Resources Agency, 2009). The Wisconsin lakes differed from the EP and Reelfoot Lake in that they were shallow, upland glacial lakes not associated with a large floodplain river, but carp, bowfin, and bass were members of their fish communities. Similar to Reelfoot Lake, native fish communities in the Wisconsin lakes have co-existed with carp for many years (Heussner et al., 2008, 2009).
Fish collection
We collected all fishes using a pulsed-DC boom electrofishing boat using protocols based on Gutreuter et al. (1995). Electrofishing protocols used 15-min electrofishing runs and a standardized power goal based upon surface water temperature (°C) and specific conductivity (µS). We sampled the EP fish community at 26, 89, and 53 random and fixed sites monthly during April–October 2010–2012. We sampled the Reelfoot Lake fish community in a variety of habitats at 50 fixed sites during the 1st week annually in June 2011 and 2012. We sampled the four Wisconsin lake fish communities at 4, 5, 4, and 6 fixed sites for a two-day period during July 2012. All collected fishes were identified and enumerated. Total length (mm) and weight (g) were recorded for all fish collected at the EP; these metrics were only recorded for bowfin, gar, bass, and carp at Reelfoot Lake and the four Wisconsin lakes. Gar were not present in any of the Wisconsin lakes we sampled, while carp were present, but not collected in Upper Phantom Lake.
Diet collection
We collected diets from bowfin, gar, and bass in the study lakes. At the EP, we collected stomach contents from ≥15 bass with lengths ≥150 mm, all bowfin with lengths ≥100 mm, and all gar with lengths ≥100 mm monthly during April–October 2010–2012 using gastric lavage (Seaburg, 1957). For each diet sample, we inserted the plastic nozzle of a garden sprayer into the esophagus of each fish and used water pressure to flush stomach contents into a mesh filter. Diet contents were preserved in 95% ethanol, and prey items were later identified to the lowest taxonomic group possible. All fish were returned to the water unharmed when gastric lavage was used to collect diet contents. Gastric lavage was also used for all bass and bowfin collected from the four Wisconsin lakes and all bass collected from Reelfoot Lake. All bowfin and gar ≥100 mm collected from Reelfoot Lake were euthanized, the entire gut was removed, diet contents were preserved in diluted formalin, and the gut was dissected in the laboratory to identify prey items.
We identified and separated each nonfish prey item in each sample into major taxonomic groups. We identified fish prey using guides for the identification of fish species based on nondigestible parts such as cleithra, vertebrae, ribs, and scales (Oates et al., 1993; Traynor et al., 2010). Next, we enumerated and transferred prey groups into preweighed drying boats. Each drying boat was placed into an oven at 80°C for ≥48 h depending upon the size of the prey items to ensure complete desiccation. We weighed each drying boat to the nearest 0.0001 g to determine the dry biomass of each prey group in each sample.
Analysis
We calculated % biomass for each prey group for each fish species as follows:
We also calculated species richness, Shannon’s equitability (E H ), and Shannon–Weiner Diversity indices (H) for each fish community sampled as follows (Beals et al., 2000; Gregory, 2013):
where S is species richness and p i is the proportion of species i relative to the total number of species.
Results
Emiquon preserve: diets
Twenty-three bowfin, seven gar, and 797 bass diets were collected from the EP. Diet contents in bass (23 taxa) exhibited greater richness than those of bowfin (10 taxa) and gar (2 taxa).
Bowfin prey use was dominated by bluegill (Lepomis macrochirus) and gizzard shad (Dorosoma cepedianum). Odonates occurred most frequently, bluegill dominated the total dry biomass (g), and about 35% of bowfin stomachs were empty (Fig. 2). No carp were found in bowfin diets, and cannibalism was not observed.
Only pumpkinseed (Lepomis gibbosus) and unidentified centrarchids were observed in gar diets. Frequency of occurrence was greatest for unidentified centrarchids, pumpkinseed dominated the total dry biomass (g), and about 57% of gar stomachs were empty. No carp were observed in gar diets, and cannibalism was not observed.
Bass prey use was dominated by gizzard shad and bluegill. Odonates occurred most frequently, gizzard shad dominated the total dry biomass (g), and about 37% of bass stomachs were empty (Fig. 2). Cannibalism was observed, and six young-of-the-year (YOY) (<100 mm) carp were observed in bass diets.
Reelfoot lake: diets
A total of 251 bowfin, 131 gar, and 67 bass diets were collected from Reelfoot Lake. Bowfin diets had the greatest diet richness (18 taxa), while gar and bass diet richness was equal (15 taxa).
Bowfin prey use was dominated by crayfish and bluegill. Crayfish occurred most frequently and dominated total dry biomass (g) (Fig. 3). About 12% of bowfin stomachs were empty. Cannibalism was detected, and no carp were observed in bowfin diets.
Gar prey use was dominated by crayfish, bluegill, and gizzard shad. Crayfish occurred most frequently and dominated the total dry biomass (g) (Fig. 3). About 45% of gar stomachs were empty. Cannibalism was detected and no carp were observed in gar diets.
Bass prey use was dominated by gizzard shad, bluegill, and crayfish. Bluegill also occurred most frequently. Gizzard shad dominated the total dry biomass (g), and about 37% of bass stomachs were empty (Fig. 3). Cannibalism was detected, and no carp were observed in bass diets.
Wisconsin lakes: diets
Twenty-five bowfin and seven bass diets were collected from Lower Phantom Lake during July 2012. Seven and four prey taxa were identified in bowfin and bass diets, respectively.
Bowfin prey use in Lower Phantom Lake was dominated by unidentified snakes and crayfish. Unidentified snake species dominated the total dry biomass (g) and about 52% of the bowfin stomachs were empty (Fig. 4). No carp were observed in bowfin diets, and cannibalism was not observed.
Proportion of total dry biomass for each bowfin (Amia calva) diet prey item collected from Lower Phantom Lake (a) and for each largemouth bass (Micropterus salmoides) diet prey item collected from Lower Phantom Lake (b), Eagle Spring Lake (c), Lulu Lake (d), and Upper Phantom Lake (e), Wisconsin during July 2012
Bass prey use was dominated by bluegill in Lower Phantom Lake. Odonata was the most frequently occurring. Bluegill dominated the total dry biomass (g) and about 29% of the bass stomachs were empty (Fig. 4). No carp were observed in bass diets, and cannibalism was not observed.
Thirty-one bass diets, representing eight prey taxa, were collected from Eagle Spring Lake during July 2012. Bass prey use was dominated by bluegill and largemouth bass. Bluegill occurred most frequently and dominated the total dry biomass (g), and about 32% of the bass stomachs were empty (Fig. 4). Cannibalism was detected, and no carp were observed in bass diets.
Thirty bass diets were collected from Lulu Lake during July 2012, and twelve prey taxa were identified. Bass prey use, frequency of occurrence, and total dry biomass (g) values were dominated by bluegill (Fig. 4). About 17% of bass stomachs were empty. Cannibalism was detected, and no carp were observed in bass diets.
Sixteen bass diets with nine different prey taxa were collected from Upper Phantom Lake during July 2012. Bass prey use was dominated by northern pike (Esox lucius), golden shiner (Notemigonus crysoleucas), and crayfish. Golden shiner and largemouth bass occurred most frequently. Northern pike dominated the total dry biomass (g) and about 50% of the bass stomachs were empty (Fig. 4). Cannibalism was detected, and no carp were observed in bass diets.
Fish community characteristics
At the EP during 2010–2012, catch per unit effort (CPUE; No. fish/h) of bowfin and gar were always less than those of carp, while CPUE of bass and bluegill were always greater than those of carp (Figs. 5, 6). Mean fish species richness pooled across the 3 years of sampling was 17 (SD = 1.0), evenness was 0.51 (SD = 0.11), and diversity was 1.45 (SD = 0.35).
Reelfoot Lake bowfin CPUE was similar to that of carp in 2011 and greater than carp in 2012. Gar, bass, and bluegill CPUE were always greater than those of carp (Figs. 5, 6). Mean fish species richness pooled across the two years of sampling was 30 (SD = 1.41), evenness was 0.64 (SD = 0.09), and diversity was 2.19 (SD = 0.34).
Lower Phantom Lake bowfin CPUE was greater than that of carp, while CPUE of bass and bluegill were greater than those of carp at Eagle Spring, Lulu, and Upper Phantom lakes during 2012 (Figs. 5, 6). Mean fish species richness pooled across the lakes was 13 (SD = 3.5), evenness was 0.72 (SD = 0.16), and diversity was 1.81 (SD = 0.3).
Discussion
Our study suggests that high abundances of bowfin and bluegill in a fish community may be associated with lower carp abundances, yet we failed to find support for our hypothesis that hypoxia-tolerant native fishes may control carp through direct predation. Bowfin diets were dominated by bluegill, crayfish, odonates, and unidentified snake species across the study systems where bowfin were present. Gar diets were dominated by crayfish, gizzard shad, pumpkinseed, and unidentified centrarchid species across the study systems where gar were present. Bowfin and gar consume a wide variety of prey items based on availability, which have included game and nongame fishes and invertebrates (Lagler & Hubbs, 1940; Toole, 1971; Tyler, 1984; Mundahl et al., 1998; Ashley & Rachels, 1999; Devlin, 1999; Snedden et al., 1999; Robertson et al., 2007; Walker et al., 2013). Our results were similar in that all bowfin and gar seemingly displayed opportunistic feeding behaviors, primarily consuming prey items that appeared to be most available, such as the abundant crayfish observed in the flooded terrestrial vegetation where bowfin and spotted gar were captured in Reelfoot Lake.
Bass diets were dominated by bluegill, crayfish, Diptera, golden shiner, gizzard shad, and odonates across all study systems. Bass are also known to feed on a wide variety of prey items based on availability (Hodgson & Kitchell, 1987; Schindler et al., 1997; Sammons & Maceina, 2006; Ahrenstorrff et al., 2009). Our results supported previous findings in that all bass displayed opportunistic feeding behaviors by consuming prey items that seemed to be most available. Cannibalism by bass was also observed in most of our study systems (Deangelis et al., 1979; Johnson & Post, 1996; Ludsin & Devries, 1997).
No carp were observed in bowfin or gar diets, while six were observed in bass diets from the EP. Thus, there appears to be a low likelihood that these species can control carp through predation, or we were unable to detect it. If carp were more available than other prey items, perhaps our results would have been different. Devlin (1999) found that carp were the most important prey item in bowfin diets when carp were more abundant than other potential prey items. Carp may also be a less desirable prey type because they are a moderately deep-bodied fish with serrated dorsal and anal spines, which can reduce predator foraging success by increasing handling costs (Sass et al., 2006). Although not specifically tested for in our study, aspects of the enemy release hypothesis and naiveté of native predators to invasive carp as prey may also explain the lack of predation observed in our study and the dominance of carp in some invaded ecosystems (Colautti et al., 2004; Cox & Lima, 2006; Barrio et al., 2010).
Carey & Wahl (2010) found that increasing native fish species diversity reduced the negative effects of carp. In our study, Reelfoot Lake had the greatest native fish species diversity, followed by the four southeastern Wisconsin lakes and then the EP. Carp may also be limited in some aquatic ecosystems through other pathways associated with the presence of bowfin, gar, bass, and other native fishes (e.g., bluegill) as part of the fish species assemblage. Bajer & Sorensen (2010) found that carp spawn in shallow lakes that experience winter hypoxia, presumably to decrease predation risk upon their eggs and larvae. Bajer et al. (2012) also found that YOY carp (<100 mm) were only observed in shallow lakes of the Upper Mississippi River basin that experience winter hypoxia and had low abundances of egg and larval predators. Silbernagel & Sorenson (2013) found that carp recruitment in shallow lakes of the Upper Mississippi River basin was limited by bluegill, which are egg and larval predators. Over 95% of carp eggs found disappeared within 4 days of spawning, which is immediately before the eggs would hatch. Large quantities of carp eggs were found in bluegill diets, and additional laboratory studies found that bluegill sought after and consumed larval carp. Interestingly, bluegill were among the most abundant fish species collected at all six study sites (EP mean bluegill electrofishing CPUE = 47.4/h; Reelfoot = 44.1/h; Wisconsin lakes = 62.5/h). Although we did not examine bluegill diets, it is possible that this species contributes to the biotic resistance of the six ecosystems we examined. Besides bluegill, other native species may also be consuming carp such as piscivorous birds or mammals (Hoffman & Curnow, 1979; Michaels, 2011). A diet study of river otter (Lontra canadensis) at the EP examined scat and found that carp comprised about 70% of their diets (Fretueg, 2012). Our observational results suggest that high abundances of bowfin and bluegill, and to a lesser extent gar and bass, may be essential components of fish communities to limit carp abundances; however, mechanisms limiting carp population growth are not clear and may be direct, indirect, and/or interacting. Thus, additional studies integrating and testing multiple hypotheses (e.g., enemy release hypothesis, native predator naiveté) are critically needed to better understand potential factors limiting carp establishment and dominance (Colautti et al., 2004; Cox & Lima, 2006; Catford et al., 2009; Barrio et al., 2010).
Relatively little is known about the life history and ecology of bowfin and gar. These primitive fishes are often referred to as “rough” or “trash fish” with little value to native aquatic ecosystems or humans (Scarnecchia, 1992). In contrast to this misconception, Devlin (1999) found that bowfin have the potential to consume large quantities of carp if they are the most abundant prey, and our results suggest that high abundances of bowfin may limit carp through unknown mechanisms. Additional research and conservation of bowfin and gar should be promoted to better understand these species and their role in native aquatic ecosystems. Similarly, there is still much to learn about the life history of carp. It is unknown whether the population dynamics of carp in small lakes in the Upper Mississippi River basin translate to other regions or ecosystems where carp are present (Bajer & Sorensen, 2010; Bajer et al., 2012; Silbernagel & Sorenson, 2013). Management practices that increase diversity of native fishes and improve the abundance of bluegill may help create balanced aquatic ecosystems that are more resilient to the negative effects of carp. Further, our research may suggest that fish communities with abundances of bowfin and bluegill exceeding those of carp may impede carp from dominating the fish community. Additional research is critically needed to better understand and sustainably manage carp in native aquatic ecosystems, specifically in systems where bowfin, gar, and carp are present. We specifically recommend that controlled pond studies be conducted to test for individual and interacting effects of bowfin, gar, bass, and bluegill on carp, with particular emphasis on testing the enemy release hypothesis and native predator naiveté toward consumption of carp (Colautti et al., 2004; Cox & Lima, 2006; Barrio et al., 2010). Such studies may be able to elucidate the most important individual or fish community mechanism(s) leading to a native recipe to sustainably control carp.
References
Ahrenstorff, T. D., G. G. Sass & M. R. Helmus, 2009. The influence of littoral zone coarse woody habitat on home range size, spatial distribution, and feeding ecology of largemouth bass (Micropterus salmoides). Hydrobiologia 623: 223–233.
Allendorf, F. W. & L. L. Lundquist, 2003. Introduction: population biology, evolution, and control of invasive species. Conservation Biology 17: 24–30.
Ashley, K. W. & R. T. Rachels, 1999. Food habits of bowfin in the Black and Lumber river, North Carolina. Southeastern Association of Fish and Wildlife Agencies 53: 50–60.
Bajer, P. G. & P. W. Sorensen, 2010. Recruitment and abundance of an invasive fish, the common carp, is driven by its propensity to invade and reproduce in basins that experience winter-time hypoxia in interconnected lakes. Biological Invasions 12: 1101–1112.
Bajer, P. G., G. Sullivan & P. W. Sorensen, 2009. Effects of a rapidly increasing population of common carp on vegetative cover and waterfowl in a recently restored Midwestern shallow lake. Hydrobiologia 632: 235–245.
Bajer, P. G., C. J. Chizinski, J. J. Silbernagel & P. W. Sorensen, 2012. Variation in native micro-predator abundance explains recruitment of a mobile invasive fish, the common carp, in a naturally unstable environment. Biological Invasions 14: 1919–1929.
Barrio, I. C., C. G. Bueno, P. B. Banks & F. S. Tortosa, 2010. Prey naiveté in an introduced prey species: the wild rabbit in Australia. Behavioral Ecology 21: 986–991.
Beals, M., L. Gross & S. Harrell, 2000. Diversity indices: Shannon’s H and E. [internet]. Accessed Dec 3, 2013. [available at http://www.tiem.utk.edu/~gross/bioed/bealsmodules/shannonDI.html.]
Carey, M. P. & D. H. Wahl, 2010. Native fish diversity alters the effects of an invasive species on food webs. Ecology 91: 2965–2974.
Catford, J. A., R. Jansson & C. Nilsson, 2009. Reducing redundancy in invasion ecology by integrating hypotheses into a single theoretical framework. Diversity and Distributions 15: 22–40.
Colautti, R. I., A. Ricciardi, I. A. Grigorvich & H. J. MacIsaac, 2004. Is invasion success explained by the enemy release hypothesis? Ecology Letters 7: 721–733.
Cox, J. G. & S. L. Lima, 2006. Naiveté and an aquatic-terrestrial dichotomy in the effects of introduced predators. Trends in Ecology and Evolution 21: 674–680.
Deangelis, D. L., D. K. Cox & C. C. Coutant, 1979. Cannibalism and size dispersal in young-of-the-year largemouth bass: experiment and model. Ecological Modeling 8: 133–148.
Devlin, J. R., 1999. A trend study of common carp in Pentenwell and Castle Rock reservoirs, Wisconsin, and the spring and summer diet of bowfin in the reservoirs. Master’s Thesis. University of Wisconsin-Stevens Point.
Eagle Spring Lake Management District, 2011. A lake management plan for Eagle Spring Lake in Waukesha County. Community Assistance Planning Report 2011 (226)
Fretueg, G. R, 2012. Latrine site selection and diet composition of river otters (Lontra canadensis) in a restored Illinois floodplain. Master’s Thesis. University of Illinois at Springfield.
Gaeta, J. W., T. R. Hrabik, G. G. Sass, B. M. Roth, S. J. Gilbert & M. J. Vander Zanden, 2015. A whole-lake experiment to control invasive rainbow smelt (Actinopterygii, Osmeridae) via overharvest and a food web manipulation. Hydrobiologia 746: 433–444.
Gregory, M. J., 2013. The biology web: Shannon-Weiner Diversity Index. [internet] Accessed Dec 3, 2013. [available at http://faculty.clintoncc.suny.edu/faculty/michael.gregory/files/bio%20206/206%20laboratory/species%20diversity/species_diversity.htm.]
Gutreuter, S., R. Burkhardt & K. Lubinski, 1995. Long Term Resource Monitoring Program Procedures: Fish Monitoring. National Biological Service, Environmental Management Technical Center, Onalaska, WI.
Havera, S. P., K. E. Roat & L. L. Anderson, 2003. The Thompson Lake Emiquon story: the biology, drainage, and restoration of an Illinois River bottomland lake. Natural History Survey, Special Publication, Springfield.
Heussner, B., S. Gospodarek & A. Notbohm, 2008. Comprehensive survey report of Eagle Spring Lake-Waukesha County (WBIC 0768600) and Lulu Lake-Walworth County (WBIC 768800). Wisconsin Department of Natural Resources Report.
Heussner, B., S. Gospodarek & A. Notbohm, 2009. Comprehensive survey report of Upper Phantom Lake (WBIC 766000) and Lower Phantom Lake (WBIC 765800). Wisconsin Department of Natural Resources Report.
Hodgson, J. R. & J. F. Kitchell, 1987. Opportunistic foraging by largemouth bass (Micropterus salmoides). American Midland Naturalist 118: 323–336.
Hoffman, R. D. & R. D. Curnow, 1979. Mercury in herons, egrets, and their foods. The Journal of Wildlife Management 43: 85–93.
Homans, F. R. & D. J. Smith, 2013. Evaluating management options for aquatic invasive species: concepts and methods. Biological Invasions 15: 7–16.
Johnson, J. M. & D. M. Post, 1996. Morphological constraints on intracohort cannibalism in age-0 largemouth bass. Transactions of the American Fisheries Society 125: 809–812.
Lagler, K. F. & F. V. Hubbs, 1940. Food of the long-nosed gar (Lepisosteus osseus oxyurus) and the bowfin (Amia calva) in Southern Michigan. American Society of Icthyologists and Herpetologists 4: 239–241.
Ludsin, S. A. & D. R. Devries, 1997. First-year recruitment of largemouth bass: the interdependency of early life stages. Ecological Applications 7: 1024–1038.
Michaels, N. N., 2011. Biomanipulation of the largemouth bass Micropterus salmoides population to control invasive species and eutrophication at The Nature Conservancy’s Emiquon Preserve. Master’s Thesis. Western Illinois University, Macomb.
Mundahl, N. D., C. Mclnytschuk, D. K. Spielman, J. P. Horkins, K. Funk & A. M. Bilicki, 1998. Effectiveness of bowfin as a predator on bluegill in a vegetated lake. North American Journal of Fisheries Management 18: 286–294.
Nentwig, W., 2007. Biological Invasions, Vol. 193. Springer, New York.
Oates, D. W., L. M. Krings & K. L. Ditz, 1993. Field manual for the identification of selected North American freshwater fish by fillets and scales. Other Publications in Wildlife Management. Paper 13. http://digitalcommons.unl.edu/icwdmother/13.
Penne, C. R. & C. L. Pierce, 2008. Seasonal distribution, aggregation, and habitat selection of common carp in Clear Lake, Iowa. Transactions of the American Fisheries Society 137: 1050–1062.
Phantom Lakes Management District, 2012. Upper Phantom Lake and Lower Phantom Lake water report 2012. University of Wisconsin-College of Natural Resources and Center for Wetland Science and Education. Upper and Lower Phantom lakes in Waukesha County.
Robertson, C. R., S. C. Zeug & K. O. Winemiller, 2007. Associations between hydrological connectivity and resource partitioning among sympatric gar species (Lepisosteidae) in a Texas river and associated oxbows. Ecology of Freshwater Fish 17: 119–129.
Sammons, S. M. & M. J. Maceina, 2006. Changes in diet and food consumption of largemouth bass following large-scale hydrilla reduction in Lake Seminole, Georgia. Hydrobiologia 560: 109–120.
Sass, G. G., C. M. Gille, J. T. Hinke & J. F. Kitchell, 2006. Whole-lake influences of littoral structured complexity and prey body morphology on fish predator-prey interactions. Ecology of Freshwater Fish 15: 301–308.
Scarnecchia, D. L., 1992. A reappraisal of gars and bowfins in fishery management. Fisheries 17: 6–12.
Schindler, D. E., J. R. Hodgson & J. F. Kitchell, 1997. Density-dependent changes in individual foraging specialization of largemouth bass. Oecologia 110: 592–600.
Seaburg, K. G., 1957. A stomach sampler for live fish. Progressive Fish-Culturist 19: 137–139.
Silbernagel, J. J. & P. W. Sorenson, 2013. Direct field and laboratory evidence that a combination of egg and larval predation controls recruitment of invasive common carp in many lakes of the Upper Mississippi River Basin. Transactions of the American Fisheries Society 142: 1134–1140.
Simberloff, D., J.-L. Martin, P. Genovesi, V. Maris, P. A. Wardle, J. Aronson, F. Courchamp, B. Galil, E. Garcia-Berthou, M. Pascal, P. Pysek, R. Sousa, E. Tabacchi & M. Vila, 2013. Impacts of biological invasions: what’s what and the way forward. Trends in Ecology and Evolution 28: 58–66.
Snedden, G. A., W. E. Kelso & D. A. Rutherford, 1999. Diel and seasonal patterns of spotted gar movement and habitat use in the lower Atchafalaya River Basin, Louisiana. Transactions of the American Fisheries Society 128: 144–154.
Tennessee Wildlife Resources Agency, 2009. Habitat Enhancement and Monitoring Report.
Tennessee Wildlife Resources Agency, 2013. Reelfoot Lake general fishing information. Web. http://www.reelfoot.com/general_fishing_info.htm.
Tockner, K. & J. A. Stanford, 2002. Riverine flood plains: present state and future trends. Environmental Conservation 29: 308–330.
Toole, J. E., 1971. Food Study of the Bowfin and Gars in Eastern Texas, Vol. 6. Texas Parks and Wildlife Department.
Traynor, D., A. Moerke & R. Greil. 2010. Identification of Michigan Fishes using Cleithra. Great Lakes Fishery Commission Miscellaneous Publication 2010-02.
Tyler, J. D., 1984. Notes on food habits, size, and spawning behavior of spotted gar in Lake Lawtonka, Oklahoma. Proceedings of the Oklahoma Academy of Sciences 64: 8–10.
Vander Zanden, M. J., G. J. A. Hansen, S. N. Higgins & M. S. Kornis, 2010. A pound of prevention, plus a pound of cure: early detection and eradication of invasive species in the Laurentian Great Lakes. Journal of Great Lakes Research 36: 199–205.
VanMiddlesworth, T. D., N. N. Michaels & A.F. Casper, 2014. The Nature Conservancy’s Emiquon Preserve Fish and Aquatic Vegetation Monitoring 6-year (2007–2012) Report. INHS Technical Report (01).
VanMiddlesworth, T. D., N. N. McClelland, G. G. Sass, A. F. Casper, T. W. Spier & M. J. Lemke, 2016. Fish community succession and biomanipulation to control two common aquatic ecosystem stressors during a large-scale floodplain lake restoration. Hydrobiologia. doi:10.1007/s10750-016-2696-8.
Walker, R. H., E. R. Kluender, T. E. Inebnit & S. R. Adams, 2013. Differences in diet and feeding ecology of similar-sized spotted (Lepisosteus oculatus) and shortnose (Lepisosteus platostomus) gars during flooding of a south-eastern US river. Ecology of Freshwater Fish 22: 617–625.
Weber, M. J. & M. L. Brown, 2009. Effects of common carp on aquatic ecosystems 80 years after “carp as a dominant”: ecological insights for fisheries management. Reviews in Fisheries Science 17: 524–537.
Weber, M. J. & M. L. Brown, 2011. Relationships among invasive common carp, native fishes and physicochemical characteristics in upper Midwest (USA) lakes. Ecology of Freshwater Fish 20: 270–278.
Weber, M. J., M. L. Brown & D. W. Willis, 2010. Spatial variability of common carp populations in relation to lake morphology and physicochemical parameters in the upper Midwest United States. Ecology of Freshwater Fish 19: 555–565.
Acknowledgments
We thank The Nature Conservancy (TNC) for support and funding this project. The authors also thank TNC staff members Doug Blodgett, Tharran Hobson, Jason Beverlin, Dr. Maria Lemke, Denim Perry, Sally McClure, Mark Jones, Cammy Smith, and Dr. Jeff Walk for their assistance. We also thank the University of Tennessee at Martin, Reelfoot Lake Biological Station for funding, electrofishing boat use, and lodging. Josh Justice, Chris Bailey, and Cody Johnston provided critical field support for the Reelfoot Lake sampling. Current and former staff members of the Illinois Natural History Survey’s, Illinois River Biological Station, and Wisconsin Department of Natural Resources were also instrumental in completing field portions of this study. The support for Wisconsin Department of Natural Resources personnel was provided in part by the Federal Aid in Sportfish Restoration Program, Project F-95-P, study SSOT.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: Michael J. Lemke, Maria A. Lemke & Jeffery W. Walk / Large-Scale Floodplain Restoration in the Illinois River Valley
Rights and permissions
About this article
Cite this article
VanMiddlesworth, T.D., Sass, G.G., Ray, B.A. et al. Food habits and relative abundances of native piscivores: implications for controlling common carp. Hydrobiologia 804, 89–101 (2017). https://doi.org/10.1007/s10750-016-2866-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-016-2866-8