Abstract
Headwater streams export organisms and other materials to receiving streams, and macroinvertebrate drift can shape colonization dynamics in downstream reaches while providing food for downstream consumers. Spring-time drift and organic matter export was measured once monthly (February–May) over a 24-h period near the outlets of 12 eastern Kentucky (USA) streams to document and explore factors governing downstream transport. We compared drift measures as loads (day−1) and concentrations (volume−1) including drift density, biomass, richness, composition, and particulate organic matter across catchment area, month, reach scale factors, and network proximity. Aquatic invertebrate drift densities were roughly 10 times greater than terrestrial invertebrate densities; aquatic richness ranged from 18 to 45 taxa with Ephemeroptera, Plecoptera, Trichoptera, and Diptera genera dominating drift sample richness and abundance. Ordination revealed that assemblages clustered by month and catchment area; organic matter exports (loads or concentrations) also varied by month and catchment area factors. While drift measures were correlated with catchment area and sample date, local factors (e.g., substrate composition, riffle length, channel slope, and network proximity) were generally non-influential. The findings can be used to inform preservation and restoration strategies where headwater streams serve as sources of colonizers and provide food subsidies to receiving streams.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Invertebrate drift (water column entry of benthic invertebrates) is ubiquitous, playing a large role in the colonization dynamics of many species in streams (Brittain & Eikeland, 1988). Drift occurs both behaviorally (e.g., seeking food and avoiding predators) and accidentally (e.g., detached from substrate in swift current) and can vary with abiotic and biotic factors. A rich literature exists where drift studies have been conducted in multiple biomes and encompassing diel and seasonal patterns (Elliott, 1970; Hansen & Closs, 2007; Neale et al., 2008), drift distance (Elliott, 1973; Lancaster et al., 1996), abiotic and biotic controls (O’Hop & Wallace, 1983; Poff & Ward, 1991), and as food subsidies for drift-feeding fishes (Wipfli & Gregovich, 2002; Romaniszyn et al., 2007). Importantly, drift forms a critical biological connection linking upstream–downstream reaches in river networks (Waters, 1964; Townsend & Hildrew, 1976; Altermatt, 2013).
Knowledge of invertebrate transport and dispersal within and between streams is lacking in the Central Appalachians but could be a key controlling factor in determining rates of recovery after disturbance and restoration (e.g., Sundermann et al., 2011; Merovich et al., 2013; Tonkin et al., 2014). In this region, headwater streams harbor diverse and sensitive macroinvertebrates that are vulnerable to extirpation from regional land uses such as large-scale surface mining and streamside residential development (Pond et al., 2008; Pond, 2010; Bernhardt et al., 2012; Cormier et al., 2013; Merriam et al., 2013). In bioassessment, some evidence suggests that intact tributary streams likely contribute sensitive drifting larval colonists to impaired downstream reaches thereby increasing biodiversity and potentially lessening the perceived severity of impairment in receiving streams (Pond et al., 2014; Orlinskiy et al., 2015); others suggest that knowledge of dispersal constraints (e.g., fragmentation and lack of source populations) are critical in understanding appropriate stream restoration designs and predicting subsequent success (Parkyn & Smith, 2011; Sundermann et al., 2011; Tonkin et al., 2014). Support for these dispersal-based linkages was shown by Campbell Grant (2011) who simulated pruned (e.g., loss of headwaters through burial) versus fractal networks; when dispersal was downstream biased (drift), time to metapopulation extinction was shorter for the pruned stream network (conditions often found in upland areas of urbanized and mined landscapes) compared to a more branched, fractal network. This simulation suggests that leaving few or no headwater tributaries would have downstream consequences on the persistence of stream populations. Thus, intact headwaters are needed to counteract downstream extinction rates by providing constant supplies of drifting sensitive colonizers; however when present, these drifters could also affect bioassessment results by masking the detection of unabated stressors to a resident downstream assemblage especially since the fate of these sensitive colonizers is unknown (Pond et al., 2014). These tributaries are also important sites for organic matter inputs and transformation, where these products are ultimately exported to downstream consumers (Gomi et al., 2002; Wipfli & Gregovich, 2002; Meyer et al., 2007).
Because drift can potentially counteract local extinctions and aid in recovery following stream remediation (Parkyn & Smith, 2011), our study documents the quantity of invertebrates (and organic matter) exported from tributary outlets within a Central Appalachian stream network. Our aim was to assess the role of various spatial and temporal factors governing downstream transport from tributaries by analyzing patterns of multiple drift measures between tributary size, sample month, reach scale habitat factors, and network position. We used catchment area as a general proxy for stream size, flow duration and overall stream energy, whereas sample month was used to track phenological shifts in taxon distributions. We hypothesized that drift quantities (abundance, richness, and biomass) and drift community structure are mainly shaped by catchment area and sample month, but these relationships are likely moderated by local habitat and inter-site proximity differences. Therefore, we quantified and compared monthly invertebrate and organic matter exports (as total daily loadings and concentrations), focusing efforts during the spring bioassessment index period (February–May; Kentucky Department for Environmental Protection, KYDEP, 2011), a time when these headwaters have higher baseflows and occupancy rates by a diverse benthic assemblage but tend to lose surface flow in summer and autumn. Purposely, February–May represents a regulatory timeframe in context for headwater bioassessment in the region, and for measuring stream restoration (e.g., natural channel design) success where macroinvertebrates are used as biological indicators.
Methods
Study area
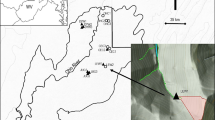
The 12 study streams were headwater tributaries of Clemons Fork (catchment area 15 km2), a state reference condition catchment within the University of Kentucky’s Robinson Research Forest (37°27′N, 83°08′W) in eastern Kentucky, USA. The catchment lies within the Dissected Appalachian Plateau of the Central Appalachian Mountain ecoregion (ecoregion 69d; Woods et al., 2002). Annual precipitation ranges from 100 to 125 cm, and mean annual air temperature is 12°C (Owenby et al., 2001). Prior studies (Pond & McMurray, 2002; Grubbs, 2011; Pond & North, 2013) confirmed that benthic macroinvertebrate assemblages within Robinson Research Forest are similar to other intact headwater streams throughout this ecoregion. Mainstem Clemons Fork is a third order, moderately high-gradient stream draining intact second-growth upland hardwood and mixed-mesophytic forest (Phillippi & Boebinger, 1986) and has relatively low land-use pressures (the catchment has unimproved roads with areas of past and recent silviculture). The network position of the study tributaries (first and second order) were evenly distributed along the length of the Clemons Fork mainstem (Fig. 1). Tributary catchments ranged from 15 to 190 ha and drained 90–100% forested land cover. Study reaches (~50 m) near the mouths of each tributary had relatively high gradients (~2–21% slope) and were fully canopied with extensive riparian forest (Table 1).
Maps showing location of Clemons Fork catchment (square) in eastern Kentucky and the 12 tributary sites (circles) within the study catchment. The Level III Ecoregions (after Woods et al., 2002) are delineated on the Kentucky map, including the location of the Central Appalachians Ecoregion (69)
Sampling and processing
We collected monthly 24-h drift samples from the 12 tributaries in February, March, April, and May 2014 (approximate baseflow, each ~30 days apart). Low, interstitial flow conditions in May precluded sampling with drift nets at four of the smallest sites. Table 1 lists mean and range of physical variables across study sites. Depending on stream width, we secured one or two driftnets (0.45-m wide by 1-m long, 250-µm mesh; Aquatic Research Instruments, Hope, ID) with rebar on the channel bottom across the wetted width. Each net was fitted with PVC collection buckets with 250-µm mesh. Our aim was to capture >90% of wetted width (and thus flow) at the base of riffles near the tributary mouths under non-storm flow conditions (approximate seasonal baseflows); in wider tributaries, this was accomplished by setting two nets side-by-side in riffles with naturally narrowed channel dimensions (see Online Resource 1 for examples). Net discharge was measured using depth and velocity readings at four equidistant locations across each net opening. On retrieval, nets were removed and the contents washed into a 250-µm sieve; all material was preserved in containers containing 90% ethyl alcohol. We observed little or no clogging of nets throughout the study duration.
On each occasion, we measured pH, temperature, specific conductance, dissolved oxygen concentration (DO), and % DO saturation with a multi-probe sonde (YSI, Yellow Springs Instruments, OH). Local habitat was recorded on the first and last occasions (February and May), and included mean length of the immediate upstream riffle (below pool), mean riffle width, mean water depth, and mean substrate composition (%). For each site, we used ArcGIS® 10.1 (ESRI, Redlands, CA) and ArcHydro® tools to calculate catchment area, land cover types (deciduous, coniferous, or mixed forest; from 2011 National Land Cover Dataset, US Geological Survey), mean valley slope, reach slope (lower 50 m above mouth), valley aspect, and fluvial distance between sites.
In the laboratory, high volume samples were split with a Folsom plankton splitter to obtain subsamples of 1/2 to 1/8 of the entire amount (by volume). Lower volume samples were processed in their entirety. All aquatic and terrestrial organisms were sorted from debris under a microscope. Our target number of subsampled organisms ranged from 200 to 300 to help reduce time and effort, but mainly to standardize richness and composition measures across samples (Vinson & Hawkins, 1996); in some cases, we sorted additional subsampled material to reach this target. Additionally, the remaining portion of the sample was scanned for 15 min to search for large/rare taxa; these taxa were only used for richness metrics and not included in density or biomass estimates. Organisms were identified to the lowest possible taxon (usually genus for aquatic; order or family for terrestrial), counted, and body lengths were measured to the nearest 1 mm. Length measurements were used to estimate the ash-free dry mass (AFDM) of drifting organisms using length–weight equations found in the literature (e.g., Sample et al., 1993; Benke et al., 1999; Sabo et al., 2002). Adults of aquatic taxa (e.g., Diptera, Plecoptera) were treated as terrestrial drift. The total number of organisms was extrapolated based on proportion of subsample and discharge measures were used to calculate drift density (individuals m−3 day−1), richness concentrations (no. of taxa m−3 day−1), and animal biomass (mg AFDM m−3 day−1). After sorting organisms from subsamples all debris (particulate detritus, inorganic sediments and remaining organisms) was dried at 50° C for 48 h, weighed, and then ashed at 550°C and re-weighed to determine the transported organic matter (AFDM). The proportion of invertebrate biomass not sorted and identified from subsampling was calculated and subtracted to determine the total particulate organic matter (POM >250 µm).
Data analysis
Tributary catchment area and sample month were the key factors we used in our sampling design to characterize the export of invertebrates and organic matter. We contrasted monthly metric values as loads (totals per day) and concentrations (total m−3 day−1) using general linear models (GLMs; Systat v. 13) to detect differences (α = 0.05) in slope equality among month classes across a gradient of catchment area. In addition to drift density and total abundance, we focused on invertebrate metrics commonly used in bioassessments in the Central Appalachian region and these metrics included: total aquatic richness, Ephemeroptera + Plecoptera + Trichoptera (EPT) richness, Ephemeroptera and Plecoptera richness and relative abundance (%). Further, we compared monthly exported biomass (invertebrate and POM). Data were checked for normality (Shapiro–Wilks test) and transformed (log, arcsin sqrt) where necessary to meet the assumptions of the GLM. To explore relationships between abiotic factors and community structure, monthly drift assemblages (aquatic taxa) were ordinated using non-metric multidimensional scaling (NMDS; PC-ORD v. 6, Gleneden Beach, OR). NMDS used Bray–Curtis (BC) distances and log-transformed abundances, omitting rare taxa (observed in <5% of samples) (McCune et al., 2002). We investigated the relationship between drift metrics (concentrations and relative abundance) and reach scale habitat variables using Spearman correlation on mean values (averaged over month classes). To evaluate the role of spatial proximity on community structure and turnover, we evaluated community drift data using both BC dissimilarity (for quantitative data) and Jaccard’s dissimilarity indices (J; for binary data) (Anderson et al., 2011). BC was calculated from averaged relative abundances among February–April samples (May was excluded because four of the smallest tributaries had no surface flow) whereas J was calculated similarly but using presence/absence. Here, we tested whether community similarity (based on composition and abundance) was related with how close sites were located in network space. We used channel-connected network distance because it is likely more appropriate given that our sites were located near tributary mouths, and since many aquatic invertebrates disperse within stream corridors, rather than overland (Griffith et al., 1998). For all pairwise BC and J calculations, we used the upper Clemons Fork site as an anchor site (it was the most physically distant site along the channel network; see Fig. 1) so that all dissimilarity values were relative to this site. We used linear regression to determine the relationship between both BC and J (as response variables) with spatial distance (i.e., distance–decay relationship), and with catchment area for comparison.
Results
Invertebrate drift composition
A total of 12,508 subsampled invertebrates were processed from 44 driftnet samples (only 8 samples in May). Aquatic invertebrates made up 89% of all drifting invertebrates collected over the duration of the study and ranged in numbers of individuals between 180 and 2,661 per day (corrected for any subsampling). A total of 208 distinct taxa were identified (167 aquatic, 41 terrestrial). Among the aquatic taxa, Diptera had the most genera (70; 40 were Chironomidae) followed by Plecoptera (23), Ephemeroptera (21), and Trichoptera (15). Combined, these four insect orders contributed most (>95%) of the total relative abundance of aquatic invertebrates exported: Diptera (37%), Ephemeroptera (36.6%), Plecoptera (18.3%), and Trichoptera (3.4%). Collembola (4 families) comprised the majority of all terrestrial drift (55%) followed by the hemlock woolly adelgids (Adelges tsugae Annand, 1924, 23%) and adult Diptera (14.7%). Monthly proportions of aquatic and terrestrial individuals and biomass are shown in Fig. 2. A list of common aquatic taxa and their frequency of occurrence (>10%) across all study sites is presented in Online Resource 2.
Comparing invertebrate and organic matter export with catchment area and month
Across all tributaries, aquatic and terrestrial invertebrate drift density and biomass varied by catchment area and sample month (Table 2). Given the much greater dominance by aquatic invertebrates and our study objectives, we focused the remaining analysis on these organisms. Relationships between monthly richness loadings (A; per day) and concentrations (B; per volume) versus catchment area are shown in Fig. 3. Richness loadings increased significantly with catchment area except for Plecoptera. Ephemeroptera richness showed the strongest response across month and catchment area based on GLMs (Table 3). An interaction between month and catchment area was found with all richness metrics (except Plecoptera) indicating that monthly differences in richness loadings were reliant on catchment area. For concentrations (richness m−3 day−1), opposite trends were found and indicated that smaller streams tended to have more drifting taxa per volume, but this was dependent on sample month. The number of individuals, biomass, and POM increased significantly with increasing catchment area (Fig. 4A) though when standardized by discharge (Fig. 4B), the catchment area effect was reduced (Table 3); however differences between monthly concentrations were significant. Monthly POM concentrations differed markedly among smaller tributaries but not the larger tributaries (i.e., >100 ha, Fig. 4B).
Community structure also varied significantly by month and catchment area. A two dimensional NMDS ordination for the aquatic invertebrate composition was produced (stress = 0.13) with axis 1 and 2 representing 62 and 14% of the explained variance, respectively (Fig. 5). Sites exhibited relatively distinct clustering among month classes, but the invertebrate composition from smaller sites appeared to vary more among streams and across months than that of larger sites (as indicated by position in relation to catchment area vector). Tighter clustering was observed for larger sites. Sample date (and corresponding stream temperature and DO) appeared to drive the ordering of sites in space along axis 1 (|r| > 0.54), whereas axis 2 was negatively correlated with measures related to catchment area, net velocity, and export of POM (|r| > 0.50). Genera contributing to the spread of sites along axes 1 and 2 (highest correlations) included the mayflies Ephemerella Walsh, 1862, Baetis Leach, 1815, and Paraleptophlebia Lestage, 1917 and blackflies Prosimulium Roubaud, 1906 (all |r| > 0.70). Notably, ephemerellid mayflies decreased with increasing sample month, while baetid mayflies increased, peaking in May (Online Resource 3). Similarly, the drift densities of simuliid blackflies was highest in February and March but declined in April and May. Although individual genera of Chironomidae exhibited monthly patterns, the family as a whole did not vary in monthly relative abundances. A list of the top 20 taxa (expressed as drift densities) among sample month is provided in Online Resource 3.
Correlation of drift metrics with reach scale factors
We found few significant correlations (P < 0.05) between the means of select drift concentrations or % composition measures and reach scale variable means (Table 4). While physical and chemical measures were correlated with drift, most covaried with sample month (e.g., temperature and DO) or catchment area (e.g., specific conductance) and were excluded from the correlation analysis. Channel slope (ranged from 1.7 to 20.9), the length of the immediate upstream riffle (ranged from 0.6 to 15 m) and substrate composition (i.e., % boulder, cobble, gravel, etc.) were not significantly correlated (P > 0.05) with any drift measures except % Ephemeroptera was negatively correlated with slope (r = −0.60). Net velocity and pH were weakly but positively associated with % Ephemeroptera and negatively for % Plecoptera. By comparison, all mean measures were significantly correlated with catchment area except biomass (m−3 day−1); notably, % Ephemeroptera strongly increased (r = 0.85) with catchment area while mean Plecoptera richness (m−3 day−1) and relative abundance decreased (r = −0.92, −0.75, respectively).
Site proximity and ecological distance
Invertebrate assemblages did not display a distance–decay relationship along the network (spatial distance gradient) nor display any resemblance based on proximity to one another within this network (Fig. 6). Spatial distance of sites and their faunal distance (BC and J dissimilarity) were not related (r 2 = 0.03, P = 0.62 for BC, r 2 = 0.01, P = 0.95 for J). In contrast, a stronger and significant relationship between catchment area and BC (r 2 = 0.58, P = 0.003) and J (r 2 = 0.64, P = 0.006) was observed (Fig. 6).
Distance–decay relationship between average Bray–Curtis (BC) dissimilarity and Jaccard’s dissimilarity (February, March, and April drift samples) with network distance and catchment area (for comparison). Dissimilarities are based on comparisons of each site with upper Clemons Fork site (the largest and most distant site in the network used here as a gradient anchor site). All trend lines based on linear regressions
Discussion
Invertebrate drift is common and widespread in lotic systems and has been considered to be the prevalent mode of dispersal by most stream invertebrates (Waters, 1965). While the pattern of drift and downstream transport of other materials is complex, it is understood that delivery of taxa and materials from upstream sources can contribute to the character of the receiving streams (Gomi et al., 2002). Our hypotheses that drift quantities (abundance, richness, and biomass) and drift community structure would vary mainly by catchment area and sample month were confirmed, but drift metrics or community structure was not strongly related with local habitat or inter-site proximity differences. For most of our drift metrics, we rejected general null hypotheses concerning the roles of tributary catchment area and sample month and rejected the premise that local habitat and network proximity strongly moderated exports or community structure. However, the relationships with catchment area and month varied among drift measures (including data as load or concentration). Whereas larger tributaries individually exported more invertebrates and POM per day, the concentration of invertebrates and POM transported each day from smaller tributaries was often higher (especially April and May).
From the perspective of downstream contributions from headwater streams, our results document that during spring-time baseflow conditions (non-storm), small first–second order tributaries can deliver dozens of genera and hundreds to thousands of organisms on a daily basis to receiving waters. As suspected, the relationship between catchment area, stream width, net discharge, total organism drift and biomass, and organic matter transport were strongly interrelated. When standardizing data by discharge (m−3 day−1), the correlations between some biological endpoints and catchment area were reduced (however, our main interest was to document both total daily loads and concentrations). When standardized by discharge, greater aquatic drift densities and organism biomass tended to occur during lower flows in April and May and might indicate increased behavioral drift in response to declining habitat and food resources as found by others (Minshall & Winger, 1968; Poff & Ward, 1991), or be otherwise related to life history cues. Our daily export values are conservative since typical storm events would lead to export of far more invertebrates than our estimates (O’Hop & Wallace, 1983). In an Oregon headwater stream, Anderson & Lehmkuhl (1968) reported 4-fold increases in drift loads after 1–2 in. storm events (common events for Central Appalachia) that corresponded to a 5-fold increase in discharge. In experimental stream channels, Imbert & Perry (2000) observed 10–33-fold increases in drift abundance in what they deemed both stepwise and abrupt (respectively) non-scouring flows (both incurring ~0.01–0.03 m3 s−1 increases). Our estimates of invertebrate biomass and drift density were roughly similar to other non-storm studies of forested, headwater Appalachian streams (O’Hop & Wallace, 1983; Romaniszyn et al., 2007). Although not studied here, we expect that drift in summer or fall is far less than spring drift (O’Hop & Wallace, 1983) in our streams mainly because of reduced overall daily discharge (Williamson et al., 2015) and occurrence of interstitial flow and non-flowing conditions (personal observation) along many of the headwater tributaries in the catchment. However, summer storm events that increase stream discharge could certainly provide punctuated exports of organisms and organic material to downstream habitats (O’Hop & Wallace, 1983; Corti & Datry, 2012).
Taxonomically, we observed a rich assemblage of aquatic drifters that greatly resembled assemblages present in routine benthic sampling from similar headwater streams in the area (Pond & McMurray, 2002; Grubbs, 2011; Pond & North, 2013). Moreover, many of the common taxa collected in our drift samples (see Online Resources 2 and 3) represent taxa that are frequently lost from coal mining impacts in the region (Pond et al., 2008; Pond, 2010; Bernhardt et al., 2012; Cormier et al., 2013). Overall, EPT dominated the drift, with Ephemeroptera comprising the most richness and abundance in larger tributaries, and Plecoptera often dominating richness and abundance in the smaller tributaries (but this varied by sample month). Since EPT (particularly mayfly richness and abundance) are typically reduced in mining-impacted streams throughout the region (e.g., Pond et al., 2008; Timpano et al., 2015), our finding has potential implications on the importance of intact tributaries to biodiversity and conservation of receiving streams (Pond et al., 2014). Daily export of EPT taxa was strongly related to catchment area and month while the opposite was found for EPT taxon concentrations where smaller steams produced higher monthly richness per volume of water. This was also supported by negative Spearman coefficients for all richness metrics (standardized by daily discharge) confirming that on average, taxa were more concentrated in the water column of smaller tributaries. We also found that catchment area was highly correlated with the relative abundance of drifting Ephemeroptera (positively) and Plecoptera (negatively) indicating unique patterns in the presence (and transport) of these orders based on catchment area.
The shift in dominance (by abundance) over the months studied was related to phenology (e.g., responding to water temperature and increasing photoperiod) as some taxa were either emerging as adults, or were new to the site as early instar hatchlings. Dominance among Ephemeroptera shifted from Ephemerellidae (Ephemerella, Drunella Needham, 1905, Eurylophella Tiensuu, 1935) in February–March, to Baetidae (mostly Baetis, Acentrella Bengtsson 1912, Procloeon Bengtsson 1915) in April–May suggesting shifts in emergence, egg hatch, or larval activity. Another phenological observation was the large numbers of drifting blackflies (Simuliidae, mostly Prosimulium and Stegopterna Enderlein, 1930) found in February and March samples, but significantly declined by April and May. These compositional changes were illustrated in the NMDS ordination where distinctive clusters of site assemblages were arranged by month. Assemblage composition at smaller sites was more variable than at large sites, suggesting that stream size (even at this fine scale) was a key controlling factor on invertebrate drift. Overall, this catchment area influence overrode detection of other factors (i.e., local habitat variables weakly affected drift measures within the range of natural stream types in our study). In a separate study, seasonal and stream size factors in other headwater tributaries of Clemons Fork were found to be important indicators of benthic assemblages (Grubbs, 2011), particularly between the months of February and May.
Spatial proximity of tributaries did not influence overall assemblage structure as commonly found in some distance–decay studies (e.g., Thompson & Townsend, 2006; Rouquette et al., 2013; Cañedo-Argüelles et al., 2015). In Appalachian streams of Maryland, Brown & Swan (2010) compared invertebrate communities from headwater tributary and mainstem sites and found that within the headwater sites, community similarity was mostly driven by local environmental factors, not spatial distance. In contrast, mainstem site similarities exhibited both environmental and distance-related responses, indicating that dispersal (from upstream and downstream sources) also drives community structure in mainstems. Although our study did not compare assemblages in tributaries with those in the mainstem Clemons Fork, but we acknowledge that Brown & Swan (2010) describes a phenomenon that likely occurs in our study area but requires further testing. In large-scale assessments (i.e., across wide geographic distances), proximity is often a strong driver of community structure with high taxa turnover rates over wide spatial gradients (Cañedo-Argüelles et al., 2015). Our study was confined to a relatively small stream network and this may have limited our ability to detect a distance–decay relationship. Further research should be done to integrate drift and benthic datasets to understand metacommunity dynamics.
Implications of drift contributions to receiving streams
Certainly, tributaries (whether polluted or unpolluted) contribute to the structure and function of their receiving waters (Benda et al., 2004; Meyer et al., 2007; Altermatt, 2013). From a vertebrate food resource perspective, the tributaries in the present study provided invertebrate biomass estimates roughly similar to headwater streams in the Southern Appalachians (O’Hop & Wallace, 1983; Siler et al., 2001), Alaska (Wipfli & Gregovich, 2002), and British Columbia (Leung et al., 2009). From a bioassessment perspective, constant localized drift supplies from intervening unimpaired streams at baseflow (and with punctuated increases during storm events) could also confound detection and identification of stressors using invertebrate indicators observed in receiving streams (e.g., where chemically or physically impacted segments temporarily harbor sensitive colonizers that would otherwise be extirpated). These source populations might be transient, where impacted receiving streams exist as sinks supported only by immigrations from unimpacted tributaries (e.g., Pulliam, 1988). Pond et al. (2014) detected improved bioassessment scores when upstream forested tributaries were present, despite ongoing mainstem water quality problems (from coal mining); the authors called for studies to examine the longitudinal influence of intact tributaries on recruitment in degraded receiving streams. From a downstream recruitment perspective, the design of restoration projects should consider connectivity to sources of colonists. In the Appalachian coal mining region where headwater streams are directly damaged, compensatory mitigation is legally required to offset ecological losses of impacted reaches, but little attention has been given to recruitment sources. Parkyn & Smith (2011), Merovich et al. (2013), and Tonkin et al. (2014) each alluded to the importance of spatial proximity and accounting for dispersal constraints in restoration and recovery scenarios.
While our results demonstrate that sensitive invertebrate colonizers are readily exported from unimpaired tributaries, previous studies on the direct influence that tributaries have on receiving streams reported mixed results. In Pennsylvania, the influence by a tributary on large river assemblages was attributed to direct (drift) and indirect (habitat) mass effects (Wilson & McTammany, 2014). In Germany, forested tributaries supplied sensitive taxa to agricultural reaches (although pesticide exposures were high enough to extirpate those taxa downstream) and that the length of forested streams (e.g., catchment area) was a key factor (Orlinskiy et al., 2015). In contrast, assumed inputs from tributaries did not always affect composition of receiving streams in Brazil, although this was highly dependent on the tributary to mainstem size ratio (Milesi & Melo, 2014). Similarly, MacNally et al. (2011) did not find substantial biological difference between tributaries and mainstems in Australia. A limitation of these two no-effect comparisons is that the studies were done in relatively undisturbed catchments (and with a potentially larger species pool); further studies are necessary to determine the influence of dispersing invertebrates from intact tributaries on downstream assemblages versus more degraded tributaries, or where tributaries supply streams of highly different character with novel taxa (e.g., as seen with glacial streams in Knispel & Castella, 2003; limestone streams in Hellmann et al., 2015). While these scenarios suggest inconsistencies on the direct influence of tributary exports, we believe methodological improvements that combine drift, benthic sampling, block-netting, and stable isotope labeling techniques would be insightful.
It is critical to understand network–dispersal relationships particularly in the Central Appalachians where large-scale upland land disturbance routinely impacts the biological integrity of downstream waters. Failing restoration attempts (e.g., Palmer & Hondula, 2014) could be tied to a lack of direct sources of drifting colonists from undisturbed tributaries (Parkyn & Smith, 2011), given that adult flight-based dispersal between catchments might not be effective for recolonization (Griffith et al., 1998; Frieden et al., 2014). In a review of stream mitigation/restoration data sets from the mountaintop mining region (KY, VA, WV), Palmer & Hondula (2014) documented poor recovery of macroinvertebrate assemblages tied mostly to lingering poor water quality. A network based meta-analysis on those mitigation projects might be fruitful in discerning whether lack of upstream sources of drifting colonists were partially responsible.
Conclusions
Results presented here provide evidence that small Appalachian tributaries can supply a relatively diverse and abundant suite of taxa to receiving streams during spring-time baseflow conditions. From a regulatory perspective where water quality compliance, biological integrity, and stream restoration are required under environmental laws, we believe that the spatial and temporal contribution of organisms and other materials from intervening tributary streams must be considered. Since catchment area and month structured invertebrate and organic matter exports, these filters should be used as guides to predict tributary influences on exports as they relate to energy subsidies, recolonization of restored areas, maintaining local biodiversity, and explaining bioassessment results in receiving streams. Additional drift and dispersal datasets (in conjunction with benthic data) from the region are necessary to also assist in explaining taxon extirpation risks and to be used in designing successful restoration projects where recruitment is a key limiting factor.
References
Altermatt, F., 2013. Diversity in riverine metacommunities: a network perspective. Aquatic Ecology 47: 365–377.
Anderson, N. H. & D. M. Lehmkuhl, 1968. Catastrophic drift of insects in a woodland stream. Ecology 49: 198–206.
Anderson, M. J., T. O. Crist, J. M. Chase, M. Vellend, B. D. Inouye, A. L. Freestone, N. J. Sanders, H. V. Cornell, L. S. Comita, K. F. Davies, S. P. Harrison, N. J. B. Kraft, J. C. Stegen & N. G. Swenson, 2011. Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecology Letters 14: 19–28.
Benda, L., N. L. Poff, D. Miller, T. Dunne, G. Reeves, G. Pess & M. Pollock, 2004. The network dynamics hypothesis: how channel networks structure riverine habitats. Bioscience 54: 413–427.
Benke, A. C., A. D. Huryn, L. A. Smock & J. B. Wallace, 1999. Length–mass relationships for freshwater macroinvertebrates in North America with particular reference to the southeastern United States. Journal of the North American Benthological Society 18: 308–343.
Bernhardt, E. S., B. D. Lutz, R. S. King, J. P. Fay, C. E. Carter, A. M. Helton & J. Amos, 2012. How many mountains can we mine? Assessing the regional degradation of Central Appalachian rivers by surface coal mining. Environmental Science and Technology 46: 8115–8122.
Brittain, J. E. & T. J. Eikeland, 1988. Invertebrate drift – a review. Hydrobiologia 166: 77–93.
Brown, B. L. & C. M. Swan, 2010. Dendritic network structure constrains metacommunity properties in riverine ecosystems. Journal of Animal Ecology 79: 571–580.
Campbell Grant, E. H., 2011. Structural complexity, movement bias, and metapopulation extinction risk in dendritic ecological networks. Journal of the North American Benthological Society 30: 252–258.
Cañedo-Argüelles, M., K. S. Boersma, M. T. Bogan, J. D. Olden, I. Phillipsen, T. A. Schriever & D. A. Lytle, 2015. Dispersal strength determines meta-community structure in a dendritic riverine network. Journal of Biogeography 42: 778–790.
Cormier, S. M., G. W. Suter & L. Zheng, 2013. Derivation of a benchmark for freshwater ionic strength. Environmental Toxicology and Chemistry 32: 263–271.
Corti, R. & T. Datry, 2012. Invertebrates and sestonic matter in an advancing wetted front travelling down a dry river bed (Albarine, France). Freshwater Science 31: 1187–1201.
Elliott, J. M., 1970. Diel changes in invertebrate drift and the food of trout Salmo trutta L. Journal of Fish Biology 2: 161–165.
Elliott, J. M., 1973. The food of brown and rainbow trout (Salmo trutta and S. gairdneri) in relation to the abundance of drifting invertebrates in a mountain stream. Oecologia 12: 329–347.
Frieden, J. C., E. E. Peterson, J. A. Webb & P. M. Negus, 2014. Improving the predictive power of spatial statistical models of stream macroinvertebrates using weighted autocovariance functions. Environmental Modelling and Software 60: 320–330.
Gomi, T., R. C. Sidle & J. S. Richardson, 2002. Understanding Processes and Downstream Linkages of Headwater Systems Headwaters differ from downstream reaches by their close coupling to hillslope processes, more temporal and spatial variation, and their need for different means of protection from land use. Bioscience 52: 905–916.
Griffith, M. B., E. M. Barrows & S. A. Perry, 1998. Lateral dispersal of adult aquatic insects (Plecoptera, Trichoptera) following emergence from headwater streams in forested Appalachian catchments. Annals of the Entomological Society of America 91: 195–201.
Grubbs, S. A., 2011. Influence of flow permanence on headwater macroinvertebrate communities in a Cumberland Plateau watershed, USA. Aquatic Ecology 45: 185–195.
Hansen, E. A. & G. P. Closs, 2007. Temporal consistency in the long-term spatial distribution of macroinvertebrate drift along a stream reach. Hydrobiologia 575: 361–371.
Hellmann, J. K., J. S. Erikson & S. A. Queenborough, 2015. Evaluating macroinvertebrate community shifts in the confluence of freestone and limestone streams. Journal of Limnology 74: 64–74.
Imbert, J. B. & J. A. Perry, 2000. Drift and benthic invertebrate responses to stepwise and abrupt increases in non-scouring flow. Hydrobiologia 436: 191–208.
Kentucky Department for Environmental Protection (KYDEP), 2011. Laboratory Procedures for Macroinvertebrate Processing, Taxonomic Identification, and Reporting. Kentucky Energy and Environment Cabinet [available on internet at http://water.ky.gov/Documents/QA/Surface%20Water%20SOPs/BenthicMacroinvertebratesLabProcessingandIdentificationSOP.pdf].
Knispel, S. & E. Castella, 2003. Disruption of a longitudinal pattern in environmental factors and benthic fauna by a glacial tributary. Freshwater Biology 48: 604–618.
Lancaster, J., A. G. Hildrew & C. Gjerlov, 1996. Invertebrate drift and longitudinal transport processes in streams. Canadian Journal of Fisheries and Aquatic Sciences 53: 572–582.
Leung, E. S., J. S. Rosenfeld & J. R. Bernhardt, 2009. Habitat effects on invertebrate drift in a small trout stream: implications for prey availability to drift-feeding fish. Hydrobiologia 623: 113–125.
MacNally, R., E. Wallis & P. S. Lake, 2011. Geometry of biodiversity patterning: assemblages of benthic macroinvertebrates at tributary confluences. Aquatic Ecology 45: 43–54.
McCune, B., J. B. Grace & D. L. Urban, 2002. Analysis of Ecological Communities, Vol. 28. MjM Software Design, Gleneden Beach.
Merovich Jr., G. T., J. T. Petty, M. P. Strager & J. B. Fulton, 2013. Hierarchical classification of stream condition: a house-neighborhood framework for establishing conservation priorities in complex riverscapes. Freshwater Science 32: 874–891.
Merriam, E. R., J. T. Petty, M. P. Strager, A. E. Maxwell & P. F. Ziemkiewicz, 2013. Scenario analysis predicts context-dependent stream response to landuse change in a heavily mined Central Appalachian watershed. Freshwater Science 32: 1246–1259.
Meyer, J. L., D. L. Strayer, J. B. Wallace, S. L. Eggert, G. S. Helfman, N. E. Leonard & M. C. Rains, 2007. The contribution of headwater streams to biodiversity in river networks. Journal of the American Water Resources Association 43: 86–103.
Milesi, S. V. & A. S. Melo, 2014. Conditional effects of aquatic insects of small tributaries on mainstream assemblages: position within drainage network matters. Canadian Journal of Fisheries and Aquatic Sciences 71: 1–9.
Minshall, G. W. & P. V. Winger, 1968. The effect of reduction in stream flow on invertebrate drift. Ecology 49: 580–582.
Neale, M. W., M. J. Dunbar, J. Jones & A. T. Ibbotson, 2008. A comparison of the relative contributions of temporal and spatial variation in the density of drifting invertebrates in a Dorset (UK) chalk stream. Freshwater Biology 53: 1513–1523.
O’Hop, J. & J. B. Wallace, 1983. Invertebrate drift, discharge, and sediment relations in a southern Appalachian headwater stream. Hydrobiologia 98: 71–84.
Orlinskiy, P., R. Münze, M. Beketov, R. Gunold, A. Paschke, S. Knillmann & M. Liess, 2015. Forested headwaters mitigate pesticide effects on macroinvertebrate communities in streams: mechanisms and quantification. Science of the Total Environment 524: 115–123.
Owenby, J., R. Heim, M. Burgin & D. Ezel, 2001. Climatography of the U.S. No. 81 – Supplement #3. Maps of Annual 1961–1990 Normal Temperature, Precipitation and Degree Days. National Climate Data Center, Asheville.
Palmer, M. A. & K. L. Hondula, 2014. Restoration as mitigation: analysis of stream mitigation for coal mining impacts in southern Appalachia. Environmental Science and Technology 48: 10552–10560.
Parkyn, S. M. & B. J. Smith, 2011. Dispersal constraints for stream invertebrates: setting realistic timescales for biodiversity restoration. Environmental Management 48: 602–614.
Phillippi, M. A. & A. Boebinger, 1986. A vegetational analysis of three small watersheds in Robinson Forest, Eastern Kentucky. Castanea 51: 11–30.
Poff, N. L. & J. V. Ward, 1991. Drift responses of benthic invertebrates to experimental streamflow variation in a hydrologically stable stream. Canadian Journal of Fisheries and Aquatic Sciences 48: 1926–1936.
Pond, G. J., 2010. Patterns of Ephemeroptera taxa loss in Appalachian headwater streams (Kentucky, USA). Hydrobiologia 641: 185–201.
Pond, G. J. & S. E. McMurray, 2002. A macroinvertebrate bioassessment index for headwater streams in the eastern coalfield region. Kentucky Department for Environmental Protection, Division of Water, Frankfort.
Pond, G. J. & S. H. North, 2013. Application of a benthic observed/expected-type model for assessing Central Appalachian streams influenced by regional stressors in West Virginia and Kentucky. Environmental Monitoring and Assessment 185: 9299–9320.
Pond, G. J., M. E. Passmore, F. A. Borsuk, L. Reynolds & C. J. Rose, 2008. Downstream effects of mountaintop coal mining: comparing biological conditions using family-and genus-level macroinvertebrate bioassessment tools. Journal of the North American Benthological Society 27: 717–737.
Pond, G. J., M. E. Passmore, N. D. Pointon, J. K. Felbinger, C. A. Walker, K. G. Krock, J. B. Fulton & W. L. Nash, 2014. Long-term impacts on macroinvertebrates downstream of reclaimed mountaintop mining valley fills in Central Appalachia. Environmental Management 54: 919–933.
Pulliam, H. R., 1988. Sources, sinks, and population regulation. American Naturalist 132: 652–661.
Romaniszyn, E. D., J. J. Hutchens & J. B. Wallace, 2007. Aquatic and terrestrial invertebrate drift in southern Appalachian Mountain streams: implications for trout food resources. Freshwater Biology 52: 1–11.
Rouquette, J. R., M. Dallimer, P. R. Armsworth, K. J. Gaston, L. Maltby & P. H. Warren, 2013. Species turnover and geographic distance in an urban river network. Diversity and Distributions 19: 1429–1439.
Sabo, J. L., J. L. Bastow & M. E. Power, 2002. Length–mass relationships for adult aquatic and terrestrial invertebrates in a California watershed. Journal of the North American Benthological Society 21: 336–343.
Sample, B. E., R. J. Cooper, R. D. Greer & R. C. Whitmore, 1993. Estimation of insect biomass by length and width. American Midland Naturalist 1993: 234–240.
Siler, E. R., J. B. Wallace & S. L. Eggert, 2001. Long-term effects of resource limitation on stream invertebrate drift. Canadian Journal of Fisheries and Aquatic Sciences 58: 1624–1637.
Sundermann, A., S. Stoll & P. Haase, 2011. River restoration success depends on the species pool of the immediate surroundings. Ecological Applications 21: 1962–1971.
Thompson, R. & C. Townsend, 2006. A truce with neutral theory: local deterministic factors, species traits and dispersal limitation together determine patterns of diversity in stream invertebrates. Journal of Animal Ecology 75: 476–484.
Timpano, A. J., S. H. Schoenholtz, D. J. Soucek & C. E. Zipper, 2015. Salinity as a limiting factor for biological condition in mining-influenced Central Appalachian headwater streams. Journal of the American Water Resources Association 51: 240–250.
Tonkin, J. D., S. Stoll, A. Sundermann & P. Haase, 2014. Dispersal distance and the pool of taxa, but not barriers, determine the colonisation of restored river reaches by benthic invertebrates. Freshwater Biology 59: 1843–1855.
Townsend, C. R. & A. G. Hildrew, 1976. Field experiments on the drifting, colonization and continuous redistribution of stream benthos. The Journal of Animal Ecology 45: 759–772.
Vinson, M. R. & C. P. Hawkins, 1996. Effects of sampling area and subsampling procedure on comparisons of taxa richness among streams. Journal of the North American Benthological Society 15: 392–399.
Waters, T. F., 1964. Recolonization of denuded stream bottom areas by drift. Transactions of the American Fisheries Society 93: 311–315.
Waters, T. F., 1965. Interpretation of invertebrate drift in streams. Ecology 46: 327–334.
Williamson, T. N., C. T. Agouridis, C. D. Barton, J. A. Villines & J. G. Lant, 2015. Classification of ephemeral, intermittent, and perennial stream reaches using a TOPMODEL-based approach. Journal of the American Water Resources Association 51: 1739–1759.
Wilson, M. J. & M. E. McTammany, 2014. Tributary and mainstem benthic macroinvertebrate communities linked by direct dispersal and indirect habitat alteration. Hydrobiologia 738: 75–85.
Wipfli, M. S. & D. P. Gregovich, 2002. Export of invertebrates and detritus from fishless headwater streams in southeastern Alaska: implications for downstream salmonid production. Freshwater Biology 47: 957–969.
Woods, A. J., J. M. Omernik, W. H. Martin, G. J. Pond, W. M. Andrews, S. M. Call, J. A. Comstock & D. D. Taylor, 2002. Ecoregions of Kentucky (2 Sided Color Poster with Map, Descriptive Text, Summary Tables, and Photographs, Map Scale 1:1,000,000). US Geological Survey, Reston.
Acknowledgments
This study was conducted as part of EPA’s Regional Research Partnership Program. We thank R. Pomponio and J. Forren (US EPA Region 3) and M. Bagley (US EPA ORD, Cincinnati) for programmatic support. E. D’Amico (Dynamac, Inc.) provided GIS support. Previous versions of the manuscript were improved by L. Reynolds and K. Krock (US EPA Region 3) and two anonymous reviewers. We would also like to thank C. Barton and the University of Kentucky’s Robinson Forest Research Committee for logistical support, and field assistance from M. Compton, M. Vogel, K. Howard, A. Rogers, S. Stiles, and J. Stermer. Although this research was supported by EPA, the views and opinions expressed in this article are those of the authors and do not necessarily represent the official views or positions of the EPA or the US government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Additional information
Handling editor: Marcelo S. Moretti
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pond, G.J., Fritz, K.M. & Johnson, B.R. Macroinvertebrate and organic matter export from headwater tributaries of a Central Appalachian stream. Hydrobiologia 779, 75–91 (2016). https://doi.org/10.1007/s10750-016-2800-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-016-2800-0