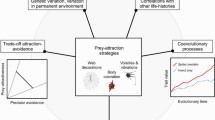
Abstract
A variety of traits is available for predators to distinguish unpalatable prey from palatable Batesian mimics. Among them, body size has received little attention as a possible mimetic trait. Size should influence predator behaviour if it shows variation between models and mimics, is detectable by the predator in question, and is not overshadowed by other traits more salient to the predator. Simple predictions within mimetic populations are that perfect mimics receive the lowest predation rate. However, prey body size is typically tightly linked to the nutritional yield and handling time for a successful predator, as well as likely being correlated with a model’s levels of defence. In certain circumstances, these confounding factors might mean that (a) selection pressures on a mimic’s size either side of the model’s phenotype are not symmetrical, (b) the optimal body size for a mimic is not necessarily equal to that of the model, and/or (c) for predators, attacking better mimics of a model’s body size more readily is adaptive. I discuss promising avenues for improving our understanding of body size as a mimetic trait, including the importance of treatments that range in both directions from the model’s size. Further work is required to understand how body size ranks in saliency against other mimetic traits such as pattern. Comparative studies could investigate whether mimics are limited to resembling only models that are already similar in size.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Predators encountering potential prey may be faced with discriminating genuinely defended items (models) from deceptive ones (Batesian mimics; Bates 1862), a task which strongly depends on the degree of resemblance between the two prey types. In studying mimicry, we often decompose that resemblance into a few distinct traits. These are known to vary in their degree of salience, with predators prioritising information from certain traits (Kazemi et al. 2014; Sherratt et al. 2015; Corral-Lopez et al. 2021). Mimicry of visual traits, often subdivided into colour, pattern and shape (e.g. Kazemi et al. 2014), has received a great deal of attention, and mimicry can operate in other sensory modalities including sound (Barber and Conner 2007) and olfaction (Czaplicki et al. 1975). Body size is another trait that contributes to mimic-model resemblance, but has received little attention in the context of mimicry. Crucially, we know little about how it affects a predator’s behaviour towards models and mimics.
There are several noteworthy properties of body size that set it apart from other mimetic traits. Most models are aposematic, meaning colour and pattern features have evolved as a signal to predators, and mimics must emulate that signal to fool the same predators (Jamie 2017). Body size does not act directly as an aposematic signal, but could still be used incidentally to discriminate models and mimics, and therefore should be classed as a cue rather than a signal per Jamie (2017). However, body size is likely to covary closely with signals such as colour patch size and therefore is indirectly linked to the strength of an aposematic signal (Remmel and Tammaru 2011).
Furthermore, size has consequences for many aspects of a prey organism’s biology, such as levels of defence, handling time and nutritional value, which are of relevance to a potential predator. A final unique feature is that, although here I focus primarily on visual detection of body size, it is also detectable in other sensory modalities such as sound frequencies (Bowling et al. 2017). For these reasons, it is not trivial to predict how selection might act on a mimic’s size relative to its models, and we will need to adapt existing predictions regarding mimicry of other traits.
Consider the giant horntail Urocerus gigas, a black and yellow banded sawfly which bears some resemblance to a wasp or hornet and can reach 4 cm in length (Benson 1952). If we assume it is palatable as prey (it does not sting, but presence of chemical defence is not known), then it might gain from Batesian mimicry of wasps. How would its large size relative to all putative model species impact the effectiveness of that mimicry? In theory, if it is possible to judge the size accurately, its size relative to a wasp might expose it as a mimic. A predator might, if uncertain, be inclined to take the risk of attacking on the basis that the nutritional reward would be especially large. On the other hand, a predator (like many humans, I suspect) might extrapolate from the similarity to wasps and the large size that the horntail is even more highly defended, and respond with great caution.
In this paper, I ask whether, and in what way, size contributes to predator foraging decisions when discriminating models and their Batesian mimics. I present several factors which could influence a predator’s response to mimic size, with associated predictions, and discuss existing evidence. I discuss the importance of evolutionary context in determining how mimicry of size and other traits may coevolve. Finally, I suggest future work which could help to clarify the contribution of size to mimicry.
Can predators use mimic body size as a cue?
A useful starting point is to consider the null prediction that predators do not make any use of body size when selecting between models and mimics. I predict a lack of selectivity when any of the following conditions are met: (1) if size is not informative in selecting palatable prey, (2) if size differences are not reliably detectable by the predator, or (3) if predators prioritise information from other traits over size.
Prey size distributions
If the size distributions of models and mimics are largely overlapping then there is little useful information encoded in the size of the prey (Taylor et al. 2017). This could include cases where intraspecific size variation is very high, so carries a weak signal. An obvious example is where the similarity between a model and mimic is maintained through ontogeny, such that (for example) mimic juveniles are smaller than adult models and vice versa. In such scenarios, predators should give little or no weighting to prey size in their decision-making and should instead use other traits (Kikuchi et al. 2015).
Predator sensory capability
Unlike colour, for which visual systems vary greatly in whether and how it is perceived (Cuthill and Bennett 1993), all but the simplest eyes encode some degree of spatial information, so animals with eyes have the potential to interpret the size of an object from the visual angle occupied. The degree of size difference detectable will depend on the visual acuity of the predator and its viewing distance. Example calculations are shown in Table 1. A subtle size difference of 1 mm (e.g. between a hoverfly and a wasp) would be detectable to a human at a distance of over 3 m, but most species do not reach this level of acuity. Some potential hoverfly predators, such as jumping spiders, should easily be able to approach closely enough to detect differences of 1 mm or less in body size (Table 1). Insectivorous birds (taking the European robin Erithacus rubeculus as an example in the absence of data for a more directly relevant species), if taking the decision to attack from around 1 m away, might only detect differences of 3 mm or more, a substantial difference in the context of insect species. Many invertebrate predators are unlikely to glean any useful size comparison until almost in contact with the prey, which may be of little use to active hunters but could be relevant for an ambush predator. We should note that similar limitations would also be imposed by visual acuity on the ability to assess shapes and patterns of mimics in addition to their size.
A further restriction on precise size estimation is imposed by the confounding effect of distance. Size comparisons are simple if the model and mimic are visible adjacent to each other, for example in the vine Boquila trifoliolata which mimics the leaves of the various tree species on which it grows (Gianoli and Carrasco-Urra 2014). If prey is seen in isolation, an assessment of size relies on being able estimate its distance from the observer. Animals have a wide array of mechanisms for estimating distance, some of which rely on contextual cues, others based on perception of the object itself, such as motion parallax (Howard 2012). Some predators are capable of prey selectivity on the basis of size even without any adjacent objects for comparison (Olberg et al. 2005). However, the indirect method of size estimation comes with a level of uncertainty around the true value, which will place a lower limit on what discrepancy in size would be reliably detectable.
Signal salience
When faced with a diverse range of prey stimuli, predators may need to make simplifications and short-cuts in their decision-making. There is a wide range of evidence that predators do not necessarily make use of all the detectable information when selecting between models and mimics, but instead focus on one or two “salient” elements of the signal (Terhune 1977; Kazemi et al. 2014; Corral-Lopez et al. 2021). In one such experiment, humans paid less attention to size than to colour when discriminating prey, but were able to use size when it was the only informative trait (Kikuchi et al. 2015). Thus, if other traits also differ between models and mimics, size may be overlooked and undergo little selective pressure.
Factors influencing predator responses to mimic size
Suppose then that we have a model and a mimic that differ consistently in their body size, a predator capable of detecting that difference, and that body size is not overshadowed by another more salient trait. Which size(s) of mimics will receive the fewest attacks?
Discrimination
A predator’s ability to discriminate between mimic and model declines with increasing mimetic accuracy, as described by signal detection theory (Oaten et al. 1975; Sherratt 2002). For this reason, within a single population, the least accurate mimics should receive the most attacks, as predators will be less likely to confuse them with their models, and protection should decline regardless of whether the mimic is larger or smaller than the model. This is in line with typical predictions for other mimetic traits such as shape. However, we should note that at a population level, when the cost of attacking models is relatively low or when alternative prey are rare, predators are predicted to become less selective and rates of attack may in fact increase with mimetic accuracy (Oaten et al. 1975). Under the influence of kin selection, this could lead to selection against high levels of accuracy (Johnstone 2002).
Nutritional pay-off
Smaller mimics have lower absolute nutritional content, which could make them less valuable as prey. If a predator needs to consume more prey to yield a given nutritional benefit, there are more opportunities to make a mistake and catch a model. This could make a predator more risk-averse when it comes to smaller prey, giving them better protection (Penney et al. 2012).
However, there are other factors that a predator must also consider during prey size selection. In optimal foraging theory, if predators are assumed to optimise their energy intake per unit of time, prey selection will depend not only on size but also on variables such as handling time and encounter rate (Pyke et al. 1977). When handling time is negligible and prey are plentiful, predators may indeed prefer to target larger prey (Quinney and Ankney 1985). For other predators, smaller prey are sufficiently quick to handle that they provide a greater overall payoff to the predator per unit of time (Turesson et al. 2002). In yet other circumstances, predators abandon selective strategies altogether in favour of accepting prey of any size, namely at low encounter rates (Krebs et al. 1977), in the presence of competitors (Barnard and Brown 1981), or when prey profitability is difficult to determine (Houston et al. 1980).
Signal properties
In responding to aposematic prey, birds show stronger innate aversion when prey are larger (Gamberale and Tullberg 1996), or to larger aggregations of prey (Gamberale and Tullberg 1998). These effects are particular to aposematic prey and independent of optimal foraging considerations such as those discussed above: palatable prey did not elicit the same size trend (Gamberale and Tullberg 1996). There is evidence that size of the aposematic signal is more important than overall body size in determining the predator’s response (Remmel and Tammaru 2011). Nonetheless the two are closely correlated with each other in real prey, as larger prey can display a larger signal. Assuming that a mimic’s signal size scales in proportion with its body size, this innate aversion by predators should afford larger mimics increased protection.
Signal receivers may also be biased by a phenomenon known as “peak shift”, whereby the greatest aversion occurs to more extreme trait values than those on which the receiver was trained (Purtle 1973). Thus, having encountered defended prey, chicks generalise their aversive responses towards a larger size than that initially experienced (Gamberale and Tullberg 1996). This bias against large aposematic prey might afford large Batesian mimics extra protection relative to smaller mimics with otherwise similar appearance. However, peak shift occurs in a direction away from the “positive stimulus”, which in the case of mimicry would be undefended prey. Thus, large mimics would have to remain rare or else they would lose their peak shift advantage, since the existence of large, palatable prey would adjust predator generalisation patterns in the opposite direction.
Levels of defence
Innate avoidance of larger aposematic signals may simply result from a cognitive bias eliciting stronger responses, but alternatively a larger individual displaying a larger signal may be an honest indication of a higher level of defence inherent in its size. Where defences involve physical weaponry or armour (Stankowich 2011), it is likely that larger prey will be associated with a larger cost, incurred through increased handling time, damage inflicted or risk of death (Edmunds 1974). Taken to an extreme, large body size itself can be a form of defence (Whitman and Vincent 2008). For chemical defences on the other hand, there does not appear to be a universal rule for the relationship between prey size and toxicity, with some taxa showing a positive association (Whitman and Vincent 2008; Kraemer et al. 2015) and others negative (Cohen 1985). Furthermore, experiments show that smaller prey benefit more from equivalent levels of chemical defence (Smith et al. 2016).
Evidence for size affecting predation on mimics
Experimental
Direct experimental evidence for predators using size as a mimetic cue is hard to find. Mimicry experiments that vary size are already rare, and within those, most do so in only one direction, with imperfect mimics either exclusively larger (Spottiswoode 2013) or smaller (Terhune 1977) than the model. A study by Kikuchi et al. (2015) which used humans foraging for computer-simulated prey did include reciprocal treatments in which sizes of models and mimics were swapped. In the absence of other informative traits, the participants were able to use size to discriminate between models and mimics, and this held regardless of whether rewarding stimuli were larger or smaller.
In a more natural scenario, Marples (1993) presented wild birds with pastry baits of three different sizes, with the medium-sized baits made unpalatable by the addition of quinine. She found that birds were indeed able to avoid the unpalatable prey items with only size to distinguish them. However, less than two-fold differences in size were difficult to distinguish and, depending on prior experience of the predators, in some cases similar protection was evident across the three prey types.
Comparative
In comparative analyses, body size and mimetic accuracy have shown a positive association in hoverflies (Penney et al. 2012; Leavey et al. 2021) and in spiders (Pekár 2022). This is consistent with the hypothesis that larger prey (at least in these taxa) are preferred by predators due to the higher nutritional reward, and hence under stronger selection (Penney et al. 2012). However, an alternative explanation for these results is that in smaller prey there is stronger selection on some other, competing function such as thermoregulation (Taylor et al. 2016).
Other prey defences
While the current evidence pertaining directly to body size Batesian mimicry is limited, we can also make informative comparisons with other, related defensive mechanisms. Deimatic (startle) displays used by harmless prey are more effective when the prey is larger (Kang et al. 2017), and eyespots are only effective in large caterpillars (Hossie et al. 2015). In the latter case, both the size of the signal and of the prey itself influenced predator behaviour, suggesting that large size may benefit prey beyond simply being able to display a signal of greater magnitude, for example implying a greater threat of damage.
Looking at honest signalling, for many taxa aposematism is more prevalent in larger species (Hagman and Forsman 2003; Forthman and Weirauch 2018), although the trend is not universal (Cheney et al. 2014). Even within species, we see examples of aposematism appearing as individuals increase in size through developmental stages (Gaitonde et al. 2018). One possible explanation is that, as discussed above, larger signals elicit stronger aversion from a predator and hence provide a more effective defence. Alternatively, it could be that other defensive strategies are less effective at larger sizes. Larger prey are more easily detectable (Remmel and Tammaru 2009), which makes it harder to adopt crypsis, perhaps promoting the evolution of aposematic displays as an alternative defence. Crucially, both the signal size and crypsis explanations could equally be used to support the evolution of Batesian mimicry in larger prey.
Evolutionary context
So far, I have discussed how the protection experienced by a mimic is influenced by predator behaviour in relation to similarity to the model, nutritional payoff, strength of an aposematic signal, and inferred levels of defence. Body size will also experience selection due to any number of other factors unrelated to defence, such as mechanical limitations (Polilov and Makarova 2017), thermoregulation (Heinrich 1993), and their own feeding strategies (cf. “Nutritional pay-off” above). Furthermore, aspects of prey defence other than mimicry may be under different size-related selection, in some cases from the same predators (e.g. Kang et al. 2017). All of these factors will combine to influence the selective landscape describing the evolutionary pressures on the size of a given Batesian mimic. Those factors unrelated to Batesian mimicry will impose limitations on how far a mimic’s size is free to evolve relative to its model, which may have very different constraints.
As with most theoretical treatments of Batesian mimicry, so far I have largely considered the context of a single mimic species with a single model. The reality is that prey are often members of large mimicry rings which include multiple prey species with varying levels of defence and varying phenotypic similarity to one another. Under these circumstances, predators are forced to rely more heavily on key features of prey signals rather than learning each individual prey type, resulting in weaker selection for accurate mimicry (Beatty et al. 2004; Ihalainen et al. 2012; Kikuchi et al. 2019). A complex community of potential models also means, from the point of view of Batesian mimics, that accuracy depends on the model under consideration, and so there are multiple possible evolutionary trajectories to consider:
-
1.
The mimic could evolve to resemble a model which is already similar in size. It can achieve high accuracy without having to incur costs due to size constraints unrelated to mimicry, but the pool of available models is restricted, and might not include those which, due to levels of defence or abundance, could in theory provide the most protection.
-
2.
The mimic could evolve to match a smaller/larger model in terms of traits such as colour and shape, but remain the same size under the influence of other size constraints. This limits the maximum level of accuracy achievable.
-
3.
The mimic could evolve accurate mimicry of a larger/smaller model in all traits including size. This would maximise the potential benefits from mimicry, but could incur major costs to other functions contributing to fitness.
-
4.
The mimic could evolve an intermediate phenotype that would benefit from similarity to more than one model species (Sherratt 2002). This might reduce selective pressure on size by effectively increasing the degree of variation in the size of the relevant model(s), but could increase vulnerability to some predators, such as specialists (Pekár et al. 2011).
Circumstantial evidence for scenario 1 can be found in certain examples of mimic species that have different models according to age or sex of the individual. In a phenomenon known as transformational mimicry (Mathew 1934), several mimics switch between mimicking different ant species as they progress through developmental instars, with larger instars resembling larger ant species (Jackson and Drummond 1974; Pekár et al. 2020). This strongly suggests these mimics are limited to resembling models of a similar size to their own, and that maintaining the same model throughout all growth stages is ineffective. Note that ant mimics are often seen directly alongside their models (e.g. Mathew 1934), which will make size comparison easier for the predator as it eliminates perceptual uncertainty due to distance (see “Predator sensory capability” above), leading to stronger selection on mimic size.
Similar processes may be at work where species exhibit sexual dimorphism in both size and mimicry. Females of the wasp Chirodamus argentinicus are members of a Müllerian mimicry ring with dark bodies and orange wings, while the smaller, undefended males are Batesian mimics of a very different group of wasps with mostly orange bodies (Evans 1968). Likewise, Motyka et al. (2018) describe several species of net-winged beetle in the genus Dilophotes in which the males and females differ in size and belong to different Müllerian mimicry rings. The authors argue that the mimetic dimorphism is a consequence of the constraints imposed by the size dimorphism (Motyka et al. 2018). The importance of these examples is to illustrate that, even in contexts where size evolution is severely constrained, considerations of size accuracy may shape the evolution of mimicry.
Future directions
One of the aims of this review is to highlight size as a potential mimetic trait, given that it has received very little attention in this context. Many experimental studies that study predator responses to mimics hold size constant (Kauppinen and Mappes 2003; Rashed and Sherratt 2007; Aronsson and Gamberale-Stille 2012; Kazemi et al. 2014). This may be largely a practical decision that avoids potentially confounding factors such as signal size and nutritional value, but it leaves us with an incomplete understanding of how mimic size might be acted upon by natural selection. There is plenty of scope for further studies along the lines of Marples (1993) that explore how size differences on their own can affect a predator’s assessment of a mimetic prey complex, and crucially that include mimics both smaller and larger than the model. Experiments that separate the effects of body size itself from nutritional reward and toxin burdens (e.g. Halpin et al. 2013; Smith et al. 2016) are also very valuable. Artificial, or experimentally manipulated, prey are invaluable in such experiments to allow the separation of size from other variables. However, a useful complement would be the use of more realistic representations of phenotypes from mimicry complexes, as the magnitudes of variation in the various mimetic traits could affect their degree of influence on predator behaviour.
How salient are size differences in comparison to other mimetic traits? Among the various studies of signal salience in mimicry, only one has included size as a treatment, and in that case (with humans as signal receivers) size was less salient than colour (Kikuchi et al. 2015). The experiment by Kikuchi et al. was an important first step, but leaves a number of follow-up questions. How do other traits such as pattern and shape fit into this hierarchy of saliency, and how does it vary among predator species? It is highly likely that the predator’s sensory capabilities will impact the hierarchy, for example with predators that have poor colour vision giving that trait less/no weight. Such questions could be addressed using behavioural experiments like those of Kazemi et al. (2014) and Sherratt et al. (2015), expanded to include size as an extra treatment. What level of colour similarity needs to be reached before other traits such as size become relevant to predation? To date, tests of saliency have mostly involved artificial stimuli designed to show high degrees of contrast in the examined traits. Does, for example, colour still dominate predator decisions when looking at contrasts representative of real mimicry rings, where colour variation might be over a much smaller range? In one recent experiment, Corral-Lopez et al. (2021) compared the contributions of colour and morphology to mimicry of glasswing butterflies by damselflies, and found that here, colour was still the most salient trait to predators. This represents a promising direction for future work, while integrating more mimetic traits including size.
Several comparative analyses have already examined the relationship between size and mimetic accuracy, usually revealing weak positive associations (Penney et al. 2012; Wilson et al. 2013; Leavey et al. 2021), but these studies calculate mimetic accuracy to one or a few model species. Although challenging, it would be very interesting to compare mimics to a wide range of potential sympatric models. This would enable testing of whether accuracy in size correlates with mimetic accuracy in other traits. Do mimics tend to resemble models that are also similar in size?
To know the accuracy with which predators can estimate traits including size, we need to know not only which species are typically preying on a mimic, but also in what context the predation decision is carried out. For example, how far away is the predator when it makes the decision to attack, and where are prey situated at the time? A detailed knowledge of natural history provides context which is often crucial to understanding mimicry.
References
Aronsson M, Gamberale-Stille G (2012) Colour and pattern similarity in mimicry: evidence for a hierarchical discriminative learning of different components. Anim Behav 84(4):881–887
Barber JR, Conner WE (2007) Acoustic mimicry in a predator–prey interaction. PNAS 104(22):9331–9334
Barnard CJ, Brown CAJ (1981) Prey size selection and competition in the common shrew (Sorex araneus L.). Behav Ecol Sociobiol 8(3):239–243
Bates HW (1862) XXXII. Contributions to an insect fauna of the Amazon Valley. Lepidoptera: Heliconidæ. Trans Linn Soc Lond 23(3):495–566
Beatty CD, Beirinckx K, Sherratt TN (2004) The evolution of müllerian mimicry in multispecies communities. Nature 431(7004):63–66
Benson RB (1952) Handbooks for the identification of British insects. Hymenoptera, Symphyta, vol 6, Section 2(a–c). Royal Entomological Society, London
Bowling DL, Garcia M, Dunn JC, Ruprecht R, Stewart A, Frommolt KH, Fitch WT (2017) Body size and vocalization in primates and carnivores. Sci Rep 7(1):41070
Caves EM, Brandley NC, Johnsen S (2018) Visual acuity and the evolution of signals. Trends Ecol Evol 33(5):358–372
Cheney KL, Cortesi F, How MJ, Wilson NG, Blomberg SP, Winters AE, Umanzör S, Marshall NJ (2014) Conspicuous visual signals do not coevolve with increased body size in marine sea slugs. J Evol Biol 27(4):676–687
Cohen JA (1985) Differences and similarities in cardenolide contents of queen and monarch butterflies in Florida and their ecological and evolutionary implications. J Chem Ecol 11(1):85–103
Corral-Lopez A, Varg JE, Cano-Cobos YP, Losada R, Realpe E, Outomuro D (2021) Field evidence for colour mimicry overshadowing morphological mimicry. J Anim Ecol 90:698–709
Cuthill IC, Bennett ATD (1993) Mimicry and the eye of the beholder. Proc R Soc B 253(1337):203–204
Czaplicki JA, Porter RH, Wilcoxon HC (1975) Olfactory mimicry involving garter snakes and artificial models and mimics. Behaviour 54(1–2):60–71
Edmunds M (1974) Defense in animals. Longman, Harlow
Evans HE (1968) Studies on neotropical Pompilidae (Hymenoptera) IV. Examples of dual sex-limited mimicry in Chirodamus. Psyche 75(1):1–22
Forthman M, Weirauch C (2018) Phylogenetic comparative analysis supports aposematic colouration–body size association in millipede assassins (Hemiptera: Reduviidae: Ectrichodiinae). J Evol Biol 31(7):1071–1078
Gaitonde N, Joshi J, Kunte K (2018) Evolution of ontogenic change in color defenses of swallowtail butterflies. Ecol Evol 8(19):9751–9763
Gamberale G, Tullberg BS (1996) Evidence for a peak-shift in predator generalization among aposematic prey. Proc R Soc B 263(1375):1329–1334
Gamberale G, Tullberg BS (1998) Aposematism and gregariousness: the combined effect of group size and coloration on signal repellence. Proc R Soc B 265(1399):889–894
Gianoli E, Carrasco-Urra F (2014) Leaf mimicry in a climbing plant protects against herbivory. Curr Biol 24(9):984–987
Hagman M, Forsman A (2003) Correlated evolution of conspicuous coloration and body size in poison frogs (Dendrobatidae). Evolution 57(12):2904–2910
Halpin CG, Skelhorn J, Rowe C (2013) Predators’ decisions to eat defended prey depend on the size of undefended prey. Anim Behav 85(6):1315–1321
Heinrich B (1993) The hot-blooded insects. Harvard University Press, Cambridge, MA
Hossie TJ, Skelhorn J, Breinholt JW, Kawahara AY, Sherratt TN (2015) Body size affects the evolution of eyespots in caterpillars. PNAS 112(21):6664–6669
Houston AI, Krebs JR, Jonathan TE (1980) Optimal prey choice and discrimination time in the great tit (Parus major L.). Behav Ecol Sociobiol 6(3):169–175
Howard IP (2012) Perceiving in depth, vol. 3: other mechanisms of depth perception. Oxford University Press, New York
Ihalainen E, Rowland HM, Speed MP, Ruxton GD, Mappes J (2012) Prey community structure affects how predators select for Müllerian mimicry. Proc R Soc B 279(1736):2099–2105
Jackson JF, Drummond BA (1974) A Batesian ant-mimicry complex from the Mountain Pine Ridge of British Honduras, with an example of transformational mimicry. Am Midl Nat 91(1):248–251
Jamie GA (2017) Signals, cues and the nature of mimicry. Proc R Soc B 284(1849):20162080
Johnstone RA (2002) The evolution of inaccurate mimics. Nature 418(6897):524–526
Kang C, Zahiri R, Sherratt TN (2017) Body size affects the evolution of hidden colour signals in moths. Proc R Soc B 284(1861):20171287
Kauppinen J, Mappes J (2003) Why are wasps so intimidating: field experiments on hunting dragonflies (Odonata: Aeshna grandis). Anim Behav 66(3):505–511
Kazemi B, Gamberale-Stille G, Tullberg Birgitta S, Leimar O (2014) Stimulus salience as an explanation for imperfect mimicry. Curr Biol 24(9):965–969
Kikuchi DW, Malick G, Webster RJ, Whissell E, Sherratt TN (2015) An empirical test of 2-dimensional signal detection theory applied to Batesian mimicry. Behav Ecol 26(4):1226–1235
Kikuchi DW, Dornhaus A, Gopeechund V, Sherratt TN (2019) Signal categorization by foraging animals depends on ecological diversity. Elife 8:e43965
Kraemer AC, Serb JM, Adams DC (2015) Model toxin level does not directly influence the evolution of mimicry in the salamander Plethodon cinereus. Evol Ecol 29(4):511–523
Krebs JR, Erichsen JT, Webber MI, Charnov EL (1977) Optimal prey selection in the great tit (Parus major). Anim Behav 25:30–38
Leavey A, Taylor CH, Symonds MRE, Gilbert F, Reader T (2021) Mapping the evolution of accurate Batesian mimicry of social wasps in hoverflies. Evolution 75(11):2802–2815
Marples NM (1993) Do wild birds use size to distinguish palatable and unpalatable prey types? Anim Behav 46(2):347–354
Mathew AP (1934) The life-history of the spider (Myrmarachne plataleoides). J Bombay Nat Hist Soc 37:369–374
Motyka M, Kampova L, Bocak L (2018) Phylogeny and evolution of Müllerian mimicry in aposematic Dilophotes: evidence for advergence and size-constraints in evolution of mimetic sexual dimorphism. Sci Rep 8(1):3744
Oaten A, Pearce CEM, Smyth MEB (1975) Batesian mimicry and signal detection theory. Bull Math Biol 37:367–387
Olberg RM, Worthington AH, Fox JL, Bessette CE, Loosemore MP (2005) Prey size selection and distance estimation in foraging adult dragonflies. J Comp Physiol A 191(9):791–797
Pekár S (2022) New drivers of the evolution of mimetic accuracy in Batesian ant-mimics: size, habitat and latitude. J Biogeogr 49(1):14–21
Pekár S, Jarab M, Fromhage L, Herberstein ME (2011) Is the evolution of inaccurate mimicry a result of selection by a suite of predators? A case study using myrmecomorphic spiders. Am Nat 178(1):124–134
Pekár S, Tsai Y-Y, Michalko R (2020) Transformational mimicry in a myrmecomorphic spider. Am Nat 196(2):216–226
Penney HD, Hassall C, Skevington JH, Abbott KR, Sherratt TN (2012) A comparative analysis of the evolution of imperfect mimicry. Nature 483(7390):461–464
Polilov AA, Makarova AA (2017) The scaling and allometry of organ size associated with miniaturization in insects: a case study for Coleoptera and Hymenoptera. Sci Rep 7(1):43095
Purtle RB (1973) Peak shift: a review. Psychol Bull 80(5):408–421
Pyke GH, Pulliam HR, Charnov E (1977) Optimal foraging: a selective review of theory and tests. Q Rev Biol 52:137–154
Quinney TE, Ankney CD (1985) Prey size selection by tree swallows. Auk 102(2):245–250
Rashed A, Sherratt TN (2007) Mimicry in hoverflies (Diptera: Syrphidae): a field test of the competitive mimicry hypothesis. Behav Ecol 18(2):337–344
Remmel T, Tammaru T (2009) Size-dependent predation risk in tree-feeding insects with different colouration strategies: a field experiment. J Anim Ecol 78(5):973–980
Remmel T, Tammaru T (2011) Evidence for the higher importance of signal size over body size in aposematic signaling in insects. J Insect Sci 11(1):1–11
Sherratt TN (2002) The evolution of imperfect mimicry. Behav Ecol 13(6):821–826
Sherratt TN, Whissell E, Webster R, Kikuchi DW (2015) Hierarchical overshadowing of stimuli and its role in mimicry evolution. Anim Behav 108:73–79
Smith KE, Halpin CG, Rowe C (2016) The benefits of being toxic to deter predators depends on prey body size. Behav Ecol 27(6):1650–1655
Spottiswoode CN (2013) A brood parasite selects for its own egg traits. Biol Lett 9(5):20130573
Stankowich T (2011) Armed and dangerous: predicting the presence and function of defensive weaponry in mammals. Adapt Behav 20(1):32–43
Taylor CH, Reader T, Gilbert F (2016) Why many Batesian mimics are inaccurate: evidence from hoverfly color patterns. Proc R Soc B 283:20161585
Taylor CH, Warrin J, Gilbert F, Reader T (2017) Which traits do observers use to distinguish Batesian mimics from their models? Behav Ecol 28(2):460–470
Terhune EC (1977) Components of a visual stimulus used by scrub jays to discriminate a Batesian model. Am Nat 111(979):435–451
Turesson H, Persson A, Brönmark C (2002) Prey size selection in piscivorous pikeperch (Stizostedion lucioperca) includes active prey choice. Ecol Freshw Fish 11(4):223–233
Whitman DW, Vincent S (2008) Large size as an antipredator defense in an insect. J Orthoptera Res 17(2):353–371
Wilson JS, Jahner JP, Williams KA, Forister ML (2013) Ecological and evolutionary processes drive the origin and maintenance of imperfect mimicry. PLoS ONE 8(4):e61610
Acknowledgements
I would like to thank Tom Reader, Francis Gilbert and two anonymous reviewers for their valuable advice in preparing this manuscript.
Funding
CHT is funded by the Natural Environment Research Council, Grant NE/S000623/1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Taylor, C.H. Body size in Batesian mimicry. Evol Ecol 37, 233–243 (2023). https://doi.org/10.1007/s10682-022-10204-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-022-10204-6