Abstract
The impact of city effluents on water quality of Indus River was assessed in the southern region of Khyber Pakhtunkhwa, Pakistan. Water samples were collected in dry (DS) and wet (WS) seasons from seven sampling zones along Indus River and the physical, bacteriological, and chemical parameters determining water quality were quantified. There were marked temporal and spatial variations in the water quality of Indus River. The magnitude of pollution was high in WS compared with DS. The quality of water varied across the sampling zones, and it greatly depended upon the nature of effluents entering the river. Water samples exceeded the WHO permissible limits for pH, EC, TDS, TS, TSS, TH, DO, BOD, COD, total coliforms, Escherichia coli, Ca2+, Mg2+, NO3−, and PO42−. Piper analysis indicated that water across the seven sampling zones along Indus River was alkaline in nature. Correlation analyses indicated that EC, TDS, TS, TH, DO, BOD, and COD may be considered as key physical parameters, while Na+, K+, Ca2+, Mg2+, Cl−, F−, NO3−, PO42−, and SO42− as key chemical parameters determining water quality, because they were strongly correlated (r > 0.70) with most of the parameters studied. Cluster analysis indicated that discharge point at Shami Road is the major source of pollution impairing water quality of Indus River. Wastewater treatment plants must be installed at all discharge points along Indus River for protecting the quality of water of this rich freshwater resource in Pakistan.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
River and streams, key sources of freshwater, are continuously being polluted by three major sources of water pollution—agriculture, human settlements, and industries (FAO 2017). This situation has been further aggravated as a result of rapid population growth, increased urbanization and the release of the municipal, domestic, and industrial wastewater into freshwater sources (Iqbal 2013; Khatri and Tyagi 2015). Globally, 80% of untreated wastewater from municipal and industry sources finds its way into freshwater bodies every year (WWAP 2017). According to the United Nations (UN), about 1500 km3 wastewater is produced annually, which is six times more water than exists in all the rivers of the world (WWAP 2003). Moreover, the untreated wastewater discharges entering into water bodies from various toxic sources have been shown to affect aquatic life depending on their chemical specificity, toxicity, bioavailability, and uptake by organisms (Bukola et al. 2015). Furthermore, key water resources are depleting at a threatening rate in many parts of the world mainly due to increase in global temperatures and uncertainties in weather projections (Foster et al. 1999).
Pakistan is a prime example of a country where freshwater resources are sharply diminishing, in quantity and quality, due to low precipitation/higher evaporation caused by climate change and the heavy influx of pollutants (Ahmed and Ali 2000). According to various studies, the water quality of major cities in Pakistan is deteriorating at an alarming rate because of uncontrolled disposal of municipal wastewater into freshwater resources (Bhutta et al. 2002). A study by Shafiq et al. (2011) indicated that large influx of wastewater in the form of human excreta has been added to Indus River contaminating it and leading to spread of various fatal diseases. Similarly, a study by Iqbal et al. (2006) showed that water of Kallar Kahar Lake in Punjab Province, Pakistan, was seriously deteriorating on account of accumulation of different toxic elements above the threshold limits from continuous influxes of wastewater. A study by Shah et al. (2012) revealed existence of high content of cation and trace metals in the surface water (i.e., rivers and streams) of the southern region of the Khyber Pakhtunkhwa (KPK) province. Mohsin et al. (2013) assessed the quality of ground water in Bahawalpur City of Pakistan. Their results indicated significant contamination in ground water with high values of physical-chemical characteristics than the permissible limits. In a more recent study, Zafar et al. (2017) indicated that irrigation water in the southern regions of KPK was not fit for agricultural production on account of accumulation of toxic effluents in the water system.
Water pollution in Pakistan is a serious issue that needs proper monitoring and assessment studies. A significant fraction of the population living in rural areas use natural water sources for their daily water needs both for human and livestock. Direct or indirect usage of polluted water is a main cause of the spread of skin diseases, eye infections, hair loss, etc. (Westcot 1997). Every year, the number of patients with water-borne diseases is increasing at disturbing rates; this may be linked to unsafe drinking water as well as unhealthy water environments due to improper domestic waste disposal (Hashmi and Shahab 1999; Jurate and Sillanpä 2006). According to World Health Organization (WHO 2000), more than 80% of diseases are water-borne in humans. About 40% of deaths are linked to water-borne diseases in Pakistan as reported by Pakistan National Conservation Strategy (SDPI 1993).
The Indus River is a major river with a total length of 3180 km making it one of longest rivers in Asia. It flows through China (western Tibet), India (Jammu and Kashmir), and Pakistan. It can be rightly called the “life line” of Pakistan as it accounts for > 80% of Pakistan’s agricultural production (Meadows 1999; Rasheed 2013). After originating in the highlands of Kailash Mountains of Tibetan Plateau, it runs from north to south through the entire length of the Pakistan and finally empties into the Arabian Sea. The Indus River and its tributaries are flanked by one of the world’s most fertile and populous regions of Pakistan. Therefore, ensuring the bio-safety of this crucial freshwater resource require regular monitoring of water quality.
The physical, bacteriological, and chemical parameters of water play a significant role in classifying and assessing water quality. However, studies aimed at monitoring the water quality as well as assessing the extent of pollution of freshwater sources in Pakistan particularly the Indus River are scarce (Shafiq et al. 2011; Shah et al. 2012). The present study is, therefore, a part of a comprehensive effort conducted to assess the hazardous effects of city effluents entering into Indus River and to quantify the important physical, chemical, and bacteriological parameters which affect water quality of Indus River. Our approach will help determining the extent of pollution impairing water quality of Indus River. The results of this study will help in devising sustainable strategies for protecting the quality of water and aquatic biota of this crucial freshwater resource.
Materials and methods
Study area
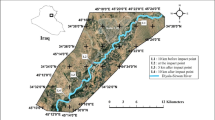
The study was conducted in Dera Ismail Khan (D.I.Khan) areas adjoining Indus River (Fig. 1). D.I.Khan is the southernmost district of province KPK bounded by the Indus River to the east and the Suleman Mountains in the west. D.I.Khan has extreme conditions being semi-arid to subtropical with an annual rainfall ranging from 180 to 300 mm (Khan et al. 2010). The total population of D.I.Khan is approximately 1.01 million (Mehdi et al. 2009) and about 80% of the population live in rural areas. The communities stretched along the Indus River extensively use its water for drinking, irrigation and other domestic purposes without any treatment. The study area of the river started where the major discharges of domestic sewage and industrial effluents from sugar, soap, textile, and oil milling are the main sources of pollutants in the Indus River. In addition, there is no proper sewerage system in the whole city (UNICEF 2010) and large fluxes of pollutants in the form of municipal wastewater are thrown directly into Indus River through seven main discharge points stretched along the river area in D.I.Khan (Shafiq et al. 2011). The study area was divided into seven sampling zones representing the whole system and covering the total area of 32.4 km shown in Fig. 1. Each sampling zone was assigned a code (i.e., zones A through G). Further geographic details about the sampling zones are presented in Table 1. The average distance between any two zones was 5.23 km.
Map of Dera Ismail Khan (D. I. Khan), a southern region of Khyber Pakhtunkhwa, Pakistan (indicating seven sampling zones along the Indus River. Red marks with numbers 1 through 7 indicate the seven sampling zones: Himmat, Kokaar, Thoyal Faazal, Shami Road, Army Camp, Ghafar Tee, and Darya Khan, respectively. This figure has been reproduced from Mehdi et al. (2009)
Sample collection
The weather profile of the study location can be classified into two distinct seasons—(1) winter season locally called as (Rabi) from October–March with mean maximum air temperature (24 ± 1.9 °C) and (2) summer season locally named as (Kharif) from April–September with mean maximum temperatures (36 ± 1.4 °C) (Fig. 2). Of the two seasons, Rabi season has a total rainfall (327.8 mm) of about 40% of the annual total with a mean rainfall of 18.6 ± 8.5 mm, whereas Kharif season has the maximum (60%) share in the total rainfall with a mean rainfall of 36.1 ± 14.7 mm. Hence, the two seasons can be referred to as dry and wet seasons (i.e., DS and WS), respectively. The rate of inflow of toxic effluents from city municipalities and industries becomes higher in WS as compared to DS due to maximum pressure in the discharge channels directed into Indus River (Iqbal 2013). Water samples were collected in the first week of January and July 2015 representing DS and WS, respectively. Representative water samples were taken in the morning (between 07:00 to 10:00 AM) from each of the seven sampling zones in two seasons (DS and WS). Single grab samples of water were obtained from the river at distance 5 m from the point of discharge and at a depth of half a meter from the surface at each sampling zone in a ten-replicate form resulting in a total of 70 samples (n = 70) per each season (DS and WS) and a total of 140 (70 × 2) samples. All samples were collected in 1.5-l sterilized polypropylene bottles first washed with 5% HNO3 and then rinsed with deionized water. After sample collection, bottles were tightly closed, stored at 4 °C and transported to the laboratory within 3 h for the analysis. Recommended standard protocols and guidelines of WHO ( 2011a) were used for sample collection to avoid any kind of contamination. Water samples collected were used to analyze the range of physicochemical and bacteriological parameters determining water quality.
Analytical procedures
Physical and chemical parameters (i.e., pH, electrical conductivity (EC), total dissolved solids (TDS), dissolved oxygen (DO), and biological dissolved oxygen (BOD)) were determined directly at the sampling site using portable measuring devices (cf. Table 2).
Values for remaining parameters (i.e., total solids (TS), total suspended solids (TSS), total hardness (TH), chemical dissolved oxygen (COD), cations (sodium (Na+), potassium (K+), lithium (Li+), calcium (Ca2+), and magnesium (Mg2+), anions (chloride (Cl−), fluoride (F−), bicarbonate (HCO3−), nitrate (NO3−), phosphate (PO42−), and sulfate (SO42−), were determined in the laboratory following standard methods (APHA 1998; ASTM 1998; AOAC 2000, 2002). Details of the apparatus used and standard methods followed are summarized in Tables 2 and 3.
Water samples were assessed for total coliforms and Escherichia coli (E. coli) within 24 h of sample collection. The samples were processed in a laminar flow hood using sterilized culture media (USEPA 1999). The bacterial load in 100 mL water samples was estimated by most probable number (MPN) technique (Eaton et al. 1995; Benson 1998; APHA 2005).
The basic chemistry of water samples across all the seven sampling zones were assessed by constructing a Piper diagram (Piper 1944). A Piper diagram consists of a combination of triangles and a diamond-shaped quadrilateral. The relative abundance of cations in (% mg L−1) is plotted on left cations’ triangle, while the relative concentration of anions, also in % mg L−1, is simultaneously plotted on right anions’ triangle and the resultant points from the cation and anion triangular plots are placed over the inner diamond-like quadrilateral structure. Water types are designated according to the zones in which these points fall on the middle quadrilateral plot. Each point represents one water type.
Statistical analyses
Descriptive statistics were performed to study the extent of variation between two sampling seasons for various parameters. Combined analysis of variance across the seasons was performed to test the significance and extent of differences among the sampling zones. Pearson correlations were estimated across seven sampling zones and two seasons to assess the inter-relationships among the physicochemical parameters of water. Finally, a cluster analysis based on Euclidean distance matrix was performed to identify and determine spatial patterns among the seven sources of effluents (i.e., sampling zones) stretched along Indus River. All statistical analyses were performed in Genstat (Payne et al. 2009).
Results
Assessing temporal variations in water quality of Indus River
Different seasons had a significant (P < 0.01) effect on physical, bacteriological, and chemical parameters of water. The variation (mean, standard error, minimum, maximum values, and ranges) of different parameters across seven sampling zones for two sampling seasons (i.e., DS and WS) are summarized in Tables 4 and 5. Both seasons showed wide ranges of variation in the physicochemical characteristics and bacteriological assessment of water across the seven sampling zones. However, the extent of variation among zones was comparatively higher in the case of WS than DS. This was evident from higher ranges of values in WS for most of the parameters studied. The results are further elaborately discussed in following subsections.
Temporal variations in the physical parameters of water
The results indicated that the water samples were slightly acidic to moderately alkaline (6.38–8.72) and (6.88–8.90) in both the seasons DS and WS, respectively (Table 4). Mean pH values exceeded the permissible limit (6.5) of drinking water standards of WHO ( 2011a) in both seasons. Data further illustrated that mean value of pH in WS (8.01 ± 0.06) was higher than DS (7.56 ± 0.17) and indicated moderately alkaline nature of water in the wet season. Mean EC values exceeded the permissible limit (250 μS cm−1) of drinking water standards of WHO ( 2011a) in both the seasons (Table 4). EC values were comparatively high in WS (575.00 ± 49.14 μS cm−1) than DS (556.60 ± 54.88 μS cm−1). Mean values of TDS, TS, TSS, and TH in DS ranged from 171.00–660.50, 304.0–841.0, 85.50–180.60, and 142.50–325.50 mg L−1, respectively (Table 4). In WS, values of these parameters ranged from 220.00–710.50, 351.00–895.00, 128.00–225.00, and 202.00–392.00 mg L−1, respectively (Table 4). Values of these parameters surpassed the WHO ( 2011a) permissible limits in both the seasons. However, mean values were much higher in WS. Furthermore, mean values of BOD and COD exceeded the WHO ( 2011a) permissible limits (5 and 10 mg L−1, respectively) in both the seasons (Table 4). Data further indicated increasing trends in mean DO and COD values with lower BOD values in DS. Almost opposite trends were observed in WS (Table 4).
Temporal variations in the bacteriological parameters of water
Bacteriological analyses result revealed that water samples collected during both seasons were contaminated with coliform and E. coli exceeding the WHO ( 2011a) permissible limits (Table 4). Comparison of both seasons indicated that mean values of total coliforms (74.78 ± 27.09 MPN 100 mL−1) and E. coli (45.94 ± 16.34 MPN 100 mL−1) were high in WS and relatively low in DS for total coliforms (53.53 ± 19.07 MPN 100 mL−1) and E. coli (23.13 ± 7.33 MPN 100 mL−1).
Temporal variations in the chemical parameters of water
Mean concentrations of cations (Na+, K+, Li+, Ca2+, and Mg2+) in DS were 38.00 ± 6.57, 7.00 ± 1.07, 0.989 ± 0.047, 46.86 ± 2.37, and 19.74 ± 1.50 mg L−1, respectively (Table 5). In WS, values of these cations were 42.80 ± 4.37, 7.93 ± 1.09, 1.489 ± 0.056, 50.66 ± 2.43, and 22.74 ± 0.93 mg L−1, respectively (Table 5). Among the cations, mean values of Ca2+ and Mg2+ exceeded the WHO ( 2011a) permissible limits of 60 and 35 mg L−1, respectively in both the seasons. Data of anions (Cl−, F−, HCO3−, NO3−, PO42−, and SO42−) indicated that their mean concentrations were 39.06 ± 5.78, 0.241 ± 0.043, 132.90 ± 8.27, 3.229 ± 0.730, 0.130 ± 0.009, and 86.29 ± 10.70 mg L−1, respectively, in DS (Table 5). In WS, concentrations of these anions were 43.43 ± 5.84, 0.275 ± 0.051, 146.60 ± 8.69, 4.24 ± 0.57, 0.134 ± 0.005, and 91.12 ± 10.91 mg L−1, respectively (Table 5). Concentrations of anions in both seasons were below the WHO ( 2011a) permissible limits in both seasons, except for PO42−, which exceeded the permissible limit of 0.1 mg L−1. It was further noted that maximum values of NO3− exceeded the permissible limit (10 mg L−1) set by PSQC ( 2008) in both seasons (Table 5). The distribution of the concentration of cations and anions in Indus River was observed in following increasing order: Ca2+ > Na+ > Mg2+ > K+ > Li+ and HCO3− > SO42− > Cl− > NO3− > F− > PO42−, respectively, during both seasons.
Assessing spatial variations in water quality of Indus River
A preliminary analysis of the data indicated that trends in water quality determining parameters across sampling zones did not change between the two seasons. Therefore, combined analysis of variance across the seasons was performed to test the significance and extent of differences among the sampling zones. Results indicated that different sampling zones had a significant (P < 0.01) effect on the mean values of physical, bacteriological and chemical parameters studied. The results are elaborately discussed in following subsections.
Spatial variations in the physical parameters of water
Mean pH was high (8.69) in sampling zone G in Darya Khan area followed by pH value of 8.63 recorded in zone F (Ghafar Tee) (Table 6). Minimum pH value (6.98) was recorded in sampling zone E (Army Camp). Data further illustrated that water samples collected from all the seven zones (A to G) exceeded the permissible pH limit (6.5–8.5) of WHO ( 2011a) (Table 6). The pH in most of the sampling zones was above 7 (i.e., alkaline). EC was high (1117.4 μS cm−1) in sampling zone D (Shami Road). Minimum EC value (324.0 μS cm−1) was recorded in water samples taken from sampling zone F (Ghafar Tee). Results further indicated that EC among all the seven sampling zones was above WHO ( 2011a) permissible limit (250 μS cm−1, Table 6), which may be considered alarming. TDS were maximum (652.8 mg L−1) in the water samples collected from sampling zone D (Shami Road) which was well above the permissible limit (500 mg L−1) set by WHO ( 2011a) (Table 6). Minimum and statistically nonsignificant TDS (238.8 and 243.8 mg L−1) were observed in water samples taken from sampling zones B (Kokaar) and E (Army Camp), respectively. TS were below WHO ( 2011a) permissible limit of 500 mg L−1 among all the seven sampling zones except zone D (Shami Road) where maximum TS (825.8 mg L−1) were recorded (Table 6). Minimum TS (344.8 mg L−1) were recorded in water samples taken from sampling zone F (Ghafar Tee). TSS were maximum (194.2 mg L−1) in sampling zone D (Shami Road) among all the zones (Table 6). TSS were minimum (112.2 mg L−1) in water samples obtained from sampling zone G (Darya Khan). TSS levels surpassed the WHO ( 2011a) permissible limit of 150 mg L−1 in most sampling zones (A to F), except zone G (Darya Khan) (Table 6). TH values were high (340.3 mg L−1) in sampling zone D (Shami Road), whereas lowest (190.3 mg L−1) in the sampling zone F (Ghafar Tee). Water samples collected at zone D (Shami Road) exceeded the permissible TH limit (300 mg L−1) of WHO ( 2011a) among all the sampling zones (Table 6). Concentration of DO was high (2.64 mg L−1) in sampling zone F (Ghafar Tee) (Table 6). Minimum DO concentration (2.01 mg L−1) was recorded in water samples taken from sampling zone D (Shami Road). BOD concentration was high (58.36 mg L−1) in sampling zone D (Shami Road) (Table 6). Minimum BOD (25.67 mg L−1) was observed in water samples taken from sampling zone F (Ghafar Tee). Trends of COD across the sampling zones were almost similar as observed for BOD (Table 6). The maximum COD concentration (80.60 mg L−1) was recorded in sampling zone-D (Shami Road), while the lowest concentration (47.91 mg L−1) was recorded in the sampling zone F (Ghafar Tee). Mean concentrations of BOD and COD exceeded the WHO ( 2011a) permissible limits (5 and 10 mg L−1, respectively) across the sampling zones.
Spatial variations in the bacteriological parameters of water
The bacterial population was found to be higher than the WHO (2011a) safe limits across the sampling zones (Table 6). The population of total coliforms in water samples ranged from 2.60 to 305.67 MPN 100 mL−1 across seven sampling zones (Table 6). The population of total coliforms was extremely high (305.67 MPN 100 mL−1) in water samples of zone D (Shami Road) followed by 89.50 MPN 100 mL−1 recorded at zone E (Army Camp). The population of total coliforms was lowest (3.20 and 2.60 MPN 100 mL−1) in water samples of zone B (Kokaar) and zone F (Ghafar Tee), respectively. The population of E. coli varied from 2.10 to 157.67 MPN 100 mL−1 across the sampling zones (Table 6). Water samples collected from zone D (Shami Road) indicated the highest population of E. coli (157.67 MPN 100 mL−1) followed by 51.00 MPN 100 mL−1 recorded in water samples of zone E (Army Camp). The lowest population of E. coli was recorded in water samples collected from zone G (Darya Khan), zone F (Ghafar Tee), and zone B (Kokaar) having statistically nonsignificant values of 3.07, 2.20, and 2.10 MPN 100 mL−1, respectively. Overall, sampling zones were ranked based on their level of contamination with total coliforms as zone D > zone E > zone A > zone G > zone C > zone B > zone F. On the basis of contamination with E. coli, sampling zones were ranked as zone D > zone E > zone A > zone C > zone G > zone F > zone B.
Spatial variations in the chemical parameters of water
Analysis of cations showed that mean concentration of Na+ was maximum (106.40 mg L−1) in sampling zone D (Shami Road) and minimum (13.27 mg L−1) in the sampling zone F (Ghafar Tee). However, Na+ concentrations were below the permissible limit (200 mg L−1) of WHO ( 2011a) among all the sampling zones (Table 7). K+ concentration was high (16.47 mg L−1) in sampling zone D (Shami Road) and low (3.82 and 3.46 mg L−1) in sampling zone E (Army Camp) and zone B (Kokaar), respectively (Table 7). As WHO has not established the permissible limit for K+ in freshwater, therefore, comparison with the permissible limit was not made. Li+ content ranged from 0.83 to 1.48 mg L−1 (Table 7). However, Li1+ concentrations were below the permissible limit (2.5 mg L−1) of WHO ( 2011a) among the seven sampling zones (Table 7). Li+ concentrations were comparatively high (1.48 and 1.45 mg L−1) in sampling zone A (Himmat) and zone E (Army Camp), while lower concentration of Li+ (0.83 mg L−1) was recorded in the sampling zone D (Shami Road). Ca2+ concentration was high (69.93 mg L−1) in water samples collected from zone D (Shami Road) which exceeded the WHO ( 2011a) permissible limit of 60 mg L−1. Ca2+ concentration was minimum (37.34 mg L−1) in the sampling zone F (Ghafar Tee) among all the sampling zones (Table 7). Mg2+ concentration was maximum (35.52 mg L−1) in sampling zone D (Shami Road), whereas minimum (18.40–18.51 mg L−1) in sampling zones A (Himmat), E (Army Camp), F (Ghafar Tee), and G (Darya Khan) (Table 7). Mg2+ concentrations were below the permissible limit (35 mg L−1) of WHO ( 2011a) among most of the sampling zones except zone D (Shami Road) where Mg2+ concentration exceeded the permissible limit (Table 7).
Results regarding the analysis of anions indicated that Cl− concentration was maximum (102.18 mg L−1) in sampling zone D (Shami Road), while minimum (20.18 mg L−1) in the sampling zone F (Ghafar Tee) (Table 8). Cl− concentrations were below the permissible limit (250 mg L−1) of WHO ( 2011a) among the seven sampling zones. Concentration of F− was maximum in the sampling zone D (Shami Road), whereas minimum in the sampling zone A (Himmat). None of the samples exceeded the F− permissible limit (1.5 mg L−1) of WHO ( 2011a) among the seven sampling zones (Table 8). HCO3− concentration was maximum (226.10 mg L−1) in sampling zone G (Darya Khan) and minimum (106.58 mg L−1) in the sampling zone B (Kokaar) (Table 8). As WHO has not established the permissible limit for HCO3− in freshwater, therefore, comparison with the permissible limit could not be made. The concentration of NO3− was maximum (10.99 mg L−1) in sampling zone D (Shami Road), whereas minimum concentration of NO3− was recorded in sampling zones B (Kokaar), E (Army Camp), F (Ghafar Tee), and G (Darya Khan) which appeared statistically at par with each other with 1.52, 1.83, 1.81, and 1.62 mg L−1 NO3−, respectively (Table 8). Among the sampling zones, NO3− concentration exceeded the permissible limit of 10 mg L−1 set by PSQCA ( 2008) in sampling zone D (Shami Road). PO42− concentration was maximum (0.228 mg L−1) in sampling zone D (Shami Road), while the minimum concentration was recorded in the sampling zone F (Ghafar Tee). Results further indicated that PO42− concentration exceeded the WHO ( 2011a) permissible limit (0.1 mg L−1) in almost all the seven sampling zones (Table 8). The concentration of SO42− was maximum (172.42 mg L−1) in sampling zone D (Shami Road), while minimum (40.35 mg L−1) in sampling zone F (Ghafar Tee). SO42− concentrations were below the permissible limit (250 mg L−1) of WHO ( 2011a) among the seven sampling zones.
Determination of water type of Indus River
The basic chemistry of water samples across all the seven sampling zones with respect to the presence of some key cations like Na+, K+, Ca2+ and Mg2+ and some chief anions like HCO3−, CO32−, Cl−, and SO42− was determined by constructing a Piper trilinear diagram (Fig. 3). Results indicated that the water in the majority of seven sampling zones along Indus River in D.I.Khan is alkaline in nature. Results further revealed no major change in water types temporally, indicating the impact of rainfall pattern (i.e., DS and WS) on major ion chemistry of water was negligible.
Piper diagram illustrating hydrochemical characterization of Indus River water in dry season (DS) and wet season (WS). Each circle and triangle indicates the mean value of cations and anions in DS and WS, respectively, of the seven sampling zones in Dera Ismail Khan (southern region of Khyber Pakhtunkhwa, Pakistan)
Assessing relationships among physicochemical characteristics of Indus River water
Analysis of correlation coefficients as presented in Table 9 indicated very strong positive correlations (r > 0.70) of some physical parameters (EC, TDS, TS, TH, BOD, and COD) with the rest of physicochemical parameters of water with r values ranging from 0.71 to 0.99. Among the chemical parameters, Na+, K+, Ca2+, Mg2+, Cl−, F−, NO3−, PO42−, and SO42− showed very strong positive correlations (r > 0.70) with most of parameters (r = 0.73–0.99).
The results further indicated moderately strong positive correlations (0.40 < r < 0.70) between pH and HCO3− (r = 0.61); TDS and TSS (r = 0.46); TS and TSS (r = 0.54); TSS and Ca2+, Mg2+, Cl−, F−, NO3−, PO42−, SO42−, BOD, and COD (r = 0.42, 0.64, 0.42, 0.55, 0.59, 0.43, 0.52, 0.53, and 0.46, respectively); K+ and HCO3−, SO42− (r = 0.48); Ca2+ and F− (r = 0.62); SO42− and Cl−, F− (r = 0.69, 62, respectively). There were weak positive correlations (r < 0.40) between pH and K+, Li+, DO (r = 0.24, 0.22, 0.11, respectively); EC and TSS (r = 0.37); TDS and HCO3− (r = 0.10); TSS and Na+, K+, Li+ (r = 0.38, 0.16, 0.23, respectively); TH and Li+ (r = 0.10); Na+ and HCO3− (r = 0.18); Li+ and HCO3− (r = 0.30); DO and Li+ (r = 0.28).
The results indicated very strong negative correlations (r > 0.70) between DO and most of the parameters (r = 0.81–0.99). There were moderately strong negative correlations (0.40 < r < 0.70) between pH and SO42− (r = 0.52); Li+ and Cl−, F− (r = 0.40, 0.44, respectively). There were weak negative correlations (r < 0.40) between pH and most of the physicochemical parameters with r values ranging from 0.04 to 0.16; Li+ and most of the parameters (r = 0.14–0.36); HCO3− and TS, TSS, Ca2+, Mg2+, F−, NO3−, SO42−, DO with r values ranging from 0.01 to 0.38.
Determination of transfer factor(s) of pollutants impairing water quality of Indus River
Cluster analysis was used to identify and determine the key source of pollutants impairing water quality of Indus River in D.I.Khan region. Results showed that seven sampling zones can be classified into three distinct groups on the basis of their extent of variability for physical, bacteriological, and chemical characteristics of water (Fig. 4). Results indicated that sampling zones A (Himmat), B (Kokaar), C (Thoya Faazal), E (Army Camp), and F (Ghafar Tee) were clustered together forming “group 1.” This suggests that these environments were highly correlated and cause little variation in the concentration of various physicochemical parameters determining water quality. On the other hand, sampling zones G (Darya Khan) and D (Shami Road) were not linked to either of the other zone(s) and created two separate groups viz. “group 2” and “group 3,” respectively. This could mean that both these water environments are distinct in their physicochemical makeup.
A schematic representation of cluster analysis for all the parameters under study (cf. “Materials and methods”). The dashed line shows hypothetical intersection of three main clusters for the selection of three distinct environment (sampling area) groups along Indus River, Dera Ismail Khan (southern region of Khyber Pakhtunkhwa, Pakistan). Zone A = Himmat, zone B = Kokaar, zone C = Thoya Faazal, zone D = Shami Road, zone E = Army Camp, zone F = Ghafar Tee, zone G = Darya Khan
Discussion
Results indicated marked temporal variations in physicochemical concentrations and bacteriological parameters. This was evident from the higher values of mean and ranges for physico-chemical parameters like pH, EC, TDS, TS, TSS, TH, BOD, COD, total coliforms, E. coli, cations (Na+, K+, Li+, Ca2+, Mg2+), and anions (Cl−, F−, HCO3−, NO3−, PO42−, SO42−) in the wet season. Results revealed that variations in these parameters may vary with fluctuations in weather conditions determining pollution load. Overall, the magnitude of pollution was high in wet season compared with the dry season. This could be due to the high influx of pollutants in the water system (Carr and Neary 2008; Memon et al. 2011) mainly due to excessive total rainfall (216.5 mm) compared with lower (111.3 mm) rainfall in dry season (Fig. 2). Data also indicated decreasing trends in mean DO and COD values with higher BOD values in wet season. This suggested that significant amounts of organic substances were being released into the water with a high oxygen demand mainly because of increased pollution load during the wet season. During the dry season, the situation improved slightly, perhaps because of low effluent discharge conditions in the water system. The deficiency of the oxygen in the water is a shelter for bacteria and other pathogens, which are anaerobic and injurious to human health (Radha et al. 2007). Almost similar results were reported by Pathak et al. (2012). The bacteriological analyses also revealed similar facts as mean values of total coliforms and E. coli were high in wet season than dry season. This could perhaps be due to the increased accumulation of municipal waste in wet season offering favorable conditions for the organisms to sustain and multiply (Shar et al. 2008). Results concluded that river water quality was highly deteriorated and contaminated by the bacterial population which may pose a potential public health hazard (Usharani et al. 2010). Furthermore, the majority of the parameters exceeded the WHO ( 2011a) safe limits, which clearly indicated the poor suitability of water for drinking and/or irrigation purpose (Shah et al. 2012; Zafar et al. 2017).
There were marked spatial variations in physical, bacteriological, and chemical characteristics of water (Popa et al. 2012). Moreover, values of most of the physical, bacteriological, and chemical parameters revealed an increasing trend in the majority of the sampling zones. One particular location, zone D (Shami Road), excelled in the majority of parameters (i.e., EC, TDS, TS, TSS, TH, BOD, COD, total coliforms, E. coli, Na+, K+, Ca2+, Mg2+, Cl−, F−, NO3−, PO42−, SO42−) which may be considered alarming. For instance, EC is directly related to the concentration of various ionized substances (anions and cations) in the water (Laluraj and Gopinath 2006). The excess of such substances in the water may cause serious health problems in the living population (Borgmann et al. 2005; Javaid et al. 2008). Estimation of TDS provides a qualitative measure of the amount of dissolved ions in water. Elevated levels of TDS may indicate a high concentration of inorganic salts principally Ca2+, Mg2+, K+, Na+, HCO3−, Cl−, and SO42− in water (WHO 2003). Furthermore, increased TDS can result in water having a bitter or salty taste, result in incrustations and corrosion, and reduced efficiency of water filters and equipment (Oram 2012). High values of TS and TSS may indicate high extent of organic particles from decomposing materials (Murphy 2007). High TH values may indicate high concentration of dissolved Ca2+ and Mg2+ ions in water (Wurts and Masser 2004). Increased TH can cause lime buildup in pipes and water heaters, may decrease soap’s cleaning ability and turn white laundry gray (Mechenich and Andrews 2004). Excessive concentrations of NO3− in water may result from sewage disposal into freshwater sources and can be hazardous to health, especially for infants and pregnant women (WHO 2011b; Oram 2012). High concentrations of Cl−, F−, PO42−, and SO42− may indicate the presence of anthropogenic pollutants in wastewater discharges into Indus River (Iscen et al. 2008; Khatri and Tyagi 2015). The increased presence of these ions in natural water may cause serious environmental problems because of its contribution to the eutrophication process (Egemen 2000; Khan et al. 2007).
Our results also indicated that direct discharge of untreated domestic, municipality, and industrial wastewater into the river was responsible for the high organic pollution that led to high BOD, COD, and low DO values across the sampling zones. Low concentrations of DO may induce a negative impact on the aquatic biota (Helmer and Hespanhol 1997). Results revealed that quality of Indus River water was deteriorated by the presence of bacterial population at multiple locations. Moreover, water samples collected from all the seven zones exceeded the permissible limits of WHO ( 2011a) for the majority of the parameters. This is an indication of potential degradation of the water reflecting the health hazards for human use (Amin et al. 2010).
Assessing the basic chemistry of water samples via Piper trilinear diagram indicated an alkaline nature of Indus River in D.I.Khan. This was also evident from the fact that pH in most of the sampling zones was above 7 (i.e., alkaline), which indicates that water is probably hard and contains Ca2+ and Mg2+ (David 2004). Increased HCO3− ions in water may cause high pH values as are the major contributors in alkalinity development (Mechenich and Andrews 2004). Almost similar results were found by Shah et al. (2012) and Zafar et al. (2017).
The correlation analysis among physicochemical characteristics of Indus River clearly showed that high values of pH might be caused by high concentrations of HCO3− as indicated by their strong positive correlation with each other. Furthermore, it was clearly evident that fluctuations in EC correlated positively with the majority of parameters studied (Sunitha et al. 2005). This suggested that water quality can be assessed with sufficient accuracy just by the measurement of EC alone. This could provide a means for easier and faster monitoring of water quality (Kalyanaraman and Geetha 2005). Another objective of this study was to determine the key parameters determining water quality in the river. Among the large number of parameters studied, EC, TDS, TS, TH, DO, BOD, and COD may be considered as key physical parameters, while Na+, K+, Ca2+, Mg2+, Cl−, F−, NO3−, PO42−, and SO42− as key chemical parameters determining water quality, because they were correlated with most of the parameters (Trivedi et al. 2009). Therefore, such an analysis could be a very useful tool for predicting water quality across the environments. Overall, it was concluded that the extent of pollution in the Indus River water is strongly related to the degree of effluents entering the aquatic environment. Mahajan et al. (2005) and Zafar et al. (2017) also concluded that most of physicochemical parameters of water are correlated indicating their interdependence with each other (Chaubey and Patil 2015). The results proved to be a useful mean for rapid monitoring of water quality with the help of systematic calculations of the correlation coefficient between water quality parameters (Heydari et al. (2013).
We used cluster analysis to determine the transfer factor(s) of pollutants impairing water quality of Indus River. Results indicated that sampling zones- D (Shami Road) and G (Darya Khan) were not related in terms of their water quality. This was also evident from the fact that majority of the physical, bacteriological, and chemical parameters attained their maximum values at sampling zone D (Shami Road) followed by zone E (Army Camp), while sampling area at Darya Khan (zone G) was observed as the less polluted one. This could be related to the nature and extent of municipal wastewater accumulating into these two locations at Indus River, D.I.Khan. Zone D (Shami Road) is located in a densely populated area of the D.I.Khan city and contains the main discharge point receiving contaminants from whole city’s municipal sewage. Generally, levels of measured parameters were low in sampling zone G (Darya Khan) in comparison to other zones. This could be related to the dilution effect on account of its furthest location from the rest of the effluent discharge points. This suggests that extent of hazardous accumulation of certain elements in Indus River water is controlled by both the type and continuous inflow of toxic effluents at various discharge points. These results were in agreement with the study of Tahir and Bhatti (1994) who concluded that direct discharge from untreated wastes is the main cause of high levels of dissolved and suspended solids, hardness, and other chemical and biological properties of water. There are no sewage effluents treatment plants in the whole D.I.Khan region and untreated wastewater are disposed directly into Indus River. This may pose adverse effects on the aquatic biota, besides making the water unfit for domestic and agricultural use. Cluster analysis thus determined the extent of spatial variation on the basis of variations in physical and chemical water characteristics of each sampling zone and indicated a strong association between spatial effects and water quality of Indus River.
Conclusions
In this study, we assessed the hazardous effects of contaminants entering into Indus River and quantified the important physical, bacteriological, and chemical parameters impacting the water quality of the river. There were marked temporal and spatial variations in the water quality of Indus River. However, the interactive impacts of spatio-temporal factors on water quality were conservative. Our results clearly indicated the accumulation of high levels of bacterial population, certain cations as well as anions in freshwaters of Indus River at most of the sampling sites which is impacting the physical characteristics of water as well. The magnitude of pollution was high in wet season compared with the dry season. Overall results suggested that the uncontrolled and continuous discharge of wastewater into the Indus River at seven discharge points has a considerable negative impact on the water quality of the river. Moreover, sewage drains at Shami Road (sampling zone D) are the major source of polluting Indus River in D.I.Khan, a southern region of Khyber Pakhtunkhwa, Pakistan. It is therefore highly recommended that the injudicious disposal of municipal wastewater into the river should be discouraged. Wastewater treatment plants must be installed at all the discharge points along Indus River in order to reduce future hazards in reference to aquatic and human life.
References
Ahmed, K., & Ali, W. (2000). Evaluation of Ravi River water quality. Pakistan Journal of Drainage Water Management, 4, 10–15.
Amin, A., Ahmad, T., Ehsanullah, M., Khatak, M. M., Masror, M., & Khan, M. A. (2010). Evaluation of industrial and city effluent quality using physicochemical and biological parameters. Electronic Journal of Environmental, Agricultural and Food Chemistry, 9(5), 931–939.
AOAC (Association of Official Analytical Chemistry) (2000). Official methods of analysis of the association of official analytical chemists. International 17th ed. Gaithersburg: Association of Analytical Communities.
AOAC (Association of Official Analytical Chemistry) (2002). Official methods of analysis of the association of official analytical chemists. International 17th ed. Gaithersburg: Association of Analytical Communities.
APHA (American Public Health Association). (1998). Standard methods for the examination of water and wastewater (20th ed.). Washington, DC: APHA.
APHA (American Public Health Association). (2005). Standard methods for the examination of water and wastewater (21st ed.). Washington, DC: APHA.
ASTM (American Society for Testing and Materials). (1998). Standard test methods for calcium and magnesium in water. West Conshohocken: ASTM International.
Benson, H.J. (1998). Microbiological applications: Laboratory manual in general microbiology, 7th edition, pp. 208˗211.
Bhutta, M.N., Ramzan, M., & Hafeez, C.A. (2002). Ground water quality and availability in Pakistan. Proceedings of seminar on ‘strategies to address the present and future water quality issues’, March 6–7, 2002. Pakistan Council of Research in Water Resources. Islamabad, Pakistan.
Borgmann, U., Couillard, Y., Doyle, P., & Dixon, D. G. (2005). Toxicity of sixty-three metals and metalloids to Hyalella azteca at two levels of water hardness. Environmental Toxicology and Chemistry, 24, 641–652.
Bukola, D., Zaid, A., Olalekan, E. A., & Falilu, A. (2015). Consequences of anthropogenic activities on fish and the aquatic environment. Poultry, Fisheries & Wildlife Sciences, 3(2), 1–12.
Carr, G.M., & Neary, J.P. (2008). Water quality for ecosystem and human health, 2nd edition. UNEP Global Environmental Monitoring System Water Programme with International Institute, PAS-European Regional Centre for Eco-hydrology UNESCO IAP Water Programme, pp. 9˗17.
Chaubey, S., & Patil, M. K. (2015). Correlation study and regression analysis of water quality assessment of Nagpur City, India. International Journal of Scientific and Research Publications, 5, 753–757.
David, C. (2004). Small water supplies. Trowbridge: Cromwell Press.
Eaton, A. D., Clesceri, L. S., & Berg, A. E. G. (1995). Standard methods for the examination of water and wastewater (19th ed.). Washington: American Public Health Association.
Egemen, Ö. (2000). Environment and water pollution. Ege University, Faculty of Fisheries. No. 42, Izmir, Turkey, pp. 120.
FAO (Food and Agriculture Organization of the United Nations). (2017). Water pollution from agriculture: A global review. Rome: FAO.
Foster, S. S. D., Morris, B. L., Lawrence, A. R., & Chilton, P. J. (1999). Groundwater impacts and issues in developing cities an introductory review. In J. Chilton (Ed.), Groundwater in urban environment: selected city profiles (pp. 3–16). the Netherlands: Balkema.
Hashmi, S.K., & Shahab, S. (1999). The need for water quality guide lines for Pakistan. (In Proceedings: Water resources achievements and issues in 20th century and challenges for next millennium. Pakistan Council of Research in Water Resources, Islamabad, Pakistan.
Helmer, R., Hespanhol, I. 1997. Water pollution control—A guide to the use of water quality management principles. World Health Organization (WHO), pp. 459.
Heydari, M. M., Abbas, A., Rohani, S. M., & Hosseini, S. M. A. (2013). Correlation study and regression analysis of drinking water quality in Kashan City, Iran. Walailak Journal of Science and Technology, 10, 315–324.
Iqbal, A. R. (2013). Environmental issues of Indus River basin: An analysis. ISSRA Papers. Institute for Strategic Studies, Research & Analysis (ISSRA), National Defence University, Islamabad, Pakistan, 5(1), 89–112.
Iqbal, F., Raza, N., Ali, M., & Athar, M. (2006). Contamination of Kallar Kahar Lake by inorganic elements and heavy metals and their temporal variation. Journal of Applied Science and Environmental Management, 10, 95–98.
Iscen, C. F., Emiroğlu, Ö., Ilhan, S., Arslan, N., Yilmaz, V., & Ahiska, S. (2008). Application of multivariate statistical techniques in the assessment of surface water quality in Uluabat Lake, Turkey. Environmental Monitoring and Assessment, 144, 269–276.
Javaid, S., Shah, S. G. S., Chaudhary, A. J., & Khan, M. H. (2008). Assessment of trace metal contamination of drinking water in the Pearl Valley, Azad Jammu and Kashmir. Clean, 36, 216–221.
Jurate, V., & Sillanpä, M. (2006). Chemical evaluation of potable water in Eastern Qinghai Province, China: Human health aspects. Chemical evaluation of potable water in Eastern Qinghai Province, China: Human health aspects. Environment International, 32, 80–86.
Kalyanaraman, S. B., & Geetha, G. (2005). Correlation analysis and prediction of characteristic parameters and water quality index of ground. Water Pollution Research, 24(1), 197–200.
Khan, I., Ullah, H., & Imran, M. (2007). Nitrate and phosphate pollution in surface and ground water in western Malaysia. Journal of Chemical Society of Pakistan, 29, 315–320.
Khan, R. U., Rashid, A., Khan, M. S., & Ozturk, E. (2010). Impact of humic acid and chemical fertilizer application on growth and grain yield of rainfed wheat (Triticum aestivum L.) Pakistan Journal of Agricultural Research, 23, 113–121.
Khatri, N., & Tyagi, S. (2015). Influences of natural and anthropogenic factors on surface and groundwater quality in rural and urban areas. Frontiers in Life Science, 8(1), 23–39.
Laluraj, C. M., & Gopinath, G. (2006). Assessment of seasonal variation of groundwater quality of Pheretic aquifer—A river basin system. Environmental Monitoring and Assessment, 117, 45–57.
Mahajan, S. V., Khare, S., & Shrivastava, V. S. (2005). A correlation and regression study. Indian Journal of Environmental Protection, 25, 254–259.
Meadows, P. (1999). The Indus River: Biodiversity, resource, human kind (1st ed.). Karachi: Oxford University Press.
Mechenich, C., & Andrews, E. (2004). Home water safety: Interpreting drinking water test results. Cooperative Extension Publishing, University of Wisconsin–Extension.
Mehdi, T., Mustafa, T., Chaudhry, R., Butt, S., Hoat, A., Ali, M., Amin, K., & Tariq, S. (2009). Profile of district Dera Ismail Khan with focus on livelihood related issues. South Asia Partnership-Pakistan. Haseeb Memorial Trust Building, Nasirabad, 2 km Raiwind Road, P.O. Box Thokar Niaz Beg, Lahore-53700, Pakistan, pp. 1–39.
Memon, M., Soomro, M. S., Akhtar, M. S., & Memon, K. S. (2011). Drinking water quality assessment in Southern Sindh (Pakistan). Environmental Monitoring and Assessment, 177, 39–50.
Mohsin, M., Safdar, S., Asghar, F., & Jamal, F. (2013). Assessment of drinking water quality and its impact on residents health in Bahawalpur City. International Journal of Humanities and Social Science, 3, 114–128.
Murphy, S. (2007). General information on solids. In City of Boulder: USGS Water Quality Monitoring. http://www.bcn.boulder.co.us/basin/data/NEW/info/TSS.html.
Oram, B. (2012). A drinking water guide for Pennsylvania. Water Research Center B.F. Environmental Consultants Inc. 15 Hillcrest Drive, Dallas, PA 18612.
Pathak, H., Pathak, D., & Limaye, S. N. (2012). Studies on the physico-chemical status of two water bodies at Sagar city under anthropogenic influences. Advances in Applied Science Research, 3(1), 31–44.
Payne, R. W., Murra, Y. D. A., Harding, S. A., Baird, D. B., & Soutar, D. M. (2009). GenStat for Windows (12th ed.). Hemel, Hempstead: VSN International.
Piper, M. (1944). A graphic procedure in the geochemical interpretation of water-analyses. EOS, Transactions, American Geophysical Union, 25, 914–928.
Popa, P., Timofti, M., Voiculescu, M., Dragan, S., Trif, C., & Georgescu, L. P. (2012). Study of physico-chemical characteristics of wastewater in an urban agglomeration in Romania. The Scientific World Journal. https://doi.org/10.1100/2012/549028.
PSQCA (Pakistan Standard & Quality Control Authority) (2008). Guidelines and standards for drinking-water quality.
Radha, K. R., Dharmaraj, K., & Kumari, B. D. R. (2007). A comparative study on the physicochemical and bacterial analysis of drinking, borewell and sewage water in the three different places of Sivakasi. Journal of Environmental Biology, 28(1), 105–108.
Rasheed, B. (2013). Impact assessment of hydroclimatic change on water stress in the Indus Basin. M.Sc. Thesis. Massachusetts Institute of Technology, pp. 114.
SDPI (Sustainable Development Policy Institute, Islamabad, Pakistan) (1993). Policy Paper Series No. 3. http://www.sdpi.org.
Shafiq, H. B., Ajaz, M., & Rasool, S. A. (2011). Bacterial and toxic pollutants in lakes of River Indus. Pakistan Journal of Botany, 43, 1765–1772.
Shah, M. T., Alizai, A. H., & Khan, S. D. (2012). Environmental geochemistry of surface and subsurface water from Dera Ismail Khan Division, Khyber Pakhtunkhwa, Pakistan. Journal of Chemical Society of Pakistan, 34, 243–250.
Shar, A. H., Kazi, Y. F., & Soomro, I. H. (2008). Impact of seasonal variation on bacteriological quality of drinking water. Bangladesh Journal of Microbiology, 25, 69–72.
Sunitha, V., Sudharshan, V., & Reddy, B. R. (2005). Hydrogeochemistry of ground water, Gooty area, Anantapur district, AndhraPradesh, India. Pollution Research, 24(1), 217–224.
Tahir, M.A., & Bhatti, M.A. (1994). Survey of drinking water quality in the rural areas of Rawalpindi district. Proceedings of Pakistan Council of Research in Water Resources, Islamabad.
Trivedi, P., Bajpai, A., & Thareja, S. (2009). Evaluation of water quality: Physico-chemical characteristics of Ganga River at Kanpur by using correlation study. Nature and Science, 1, 91–94.
UNICEF (United Nations International Children’s Emergency Fund) (2010). Intervention report on D.I.Khan. http://www.complex.pakresponse.info.
USEPA (United States Environmental Protection Agency Office of Water) (1999). Engineering and Analysis Division1200 Pennsylvania Avenue, NW (4303T) Washington, DC 20460.
Usharani, K., Umarani, K., Ayyasamy, P. M., Shanthi, K., & Lakshmanaperumalsamy, P. (2010). Physico-chemical and bacteriological characteristics of Noyyal River and ground water quality of Perur, India. Journal of Applied Science and Environmental Management, 14(2), 29–35.
Westcot, D.W. (1997). Quality control of wastewater for irrigated crop production. FAO water reports no. 10 Rome, pp. 86.
WHO (World Health Organization) (2000). Global water supply and sanitation assessment report.
WHO (World Health Organization) (2003). Total dissolved solids in drinking-water. Background document for development of WHO “Guidelines for Drinking-water Quality”.
WHO (World Health Organization) (2011a). Guidelines for drinking water quality, 4th Edition, pp. 564.
WHO (World Health Organization) (2011b). Nitrate and nitrite in drinking-water. Background document for development of WHO “Guidelines for Drinking-water Quality”.
Wurts, W.A., & Masser, M.P. (2004). Southern Regional Aquaculture Center, Publication No. 4100.
WWAP (World Water Assessment Programme) (2003). Water for people, water for life (2003): 3rd World Water Forum in Kyoto, Japan. http://www.norman-network.net.
WWAP (World Water Assessment Programme) (2017). The United Nations world water development report: Wastewater, the untapped resource. United Nations Educational, Scientific and Cultural Organization, Paris.
Zafar, S., Khan, A., Ullah, H., Khan, M. S., Khan, I., Hameed, A., Rehman, S. U., & Yasmeen, G. (2017). Assessing impact of effluent discharge on irrigation water quality in southern region of Khyber Pakhtunkhwa, Pakistan. Environmental Monitoring and Assessment, 189, 156.
Acknowledgements
The authors gratefully acknowledge funding from Higher Education Commission (HEC), Government of Pakistan, for conducting the laboratory analysis. We thank the staff of Institute of Chemical Sciences, Gomal University D.I.Khan; National Water Quality Lab. PCRWR, Islamabad, Pakistan; Department of Soil and Environmental Sciences, Faculty of Agriculture, Gomal University D.I.Khan; and Department of Chemistry, Faculty of Physical Sciences, Kohat University of Science and Technology, for their laboratory facilities provided.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khan, I., Khan, A., Khan, M.S. et al. Impact of city effluents on water quality of Indus River: assessment of temporal and spatial variations in the southern region of Khyber Pakhtunkhwa, Pakistan. Environ Monit Assess 190, 267 (2018). https://doi.org/10.1007/s10661-018-6621-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-018-6621-7