Abstract
Cell suspension cultures of Vitis vinifera cv. Vinhão (Vv) were used to study the putative response of V. vinifera to Phaeomoniella chlamydospora (Pc), a fungus frequently associated with esca and grapevine decline. Cells were elicited with a Pc autoclaved biomass extract and methyl jasmonate (MeJ). Phenolic production was evaluated by HPLC-DAD and HPLC-MS/MS. Phenolic production of Vv cells significantly changes after elicitation. Compared to control, Vv cells elicited by Pc extract increase their stilbene production 20-fold and those elicited by MeJ increase stilbenic production 9-fold. In both cases, there is de novo production of viniferin type compounds. We also analyzed the oxidative burst of Vv cells after elicitation with Pc extract and MeJ, using the probe 2′,7′-dichlorodihydrofluorescein diacetate. Adding Pc extract induces an oxidative burst that shows a biphasic pattern in Vv cells. Moreover, the induction of 7 defence-related genes expression in Vv cell cultures upon Pc extract elicitation was investigated employing semi-quantitative RT-PCR. Elicitation increases the expression of class 6 and class 10 pathogenesis-related proteins, β-1,3-glucanase, class III chitinase, lipoxygenase, phenylalanine ammonia lyase and stilbene synthase. Therefore, Vv in vitro cell cultures could be an important tool to study esca disease, since they offer a simple, rapid and selective way to evaluate plant/fungus interactions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Esca is a destructive disease that affects grapevines worldwide. Although esca-like symptoms are reported in ancient Greek and Latin works (Mugnai et al. 1999), only recently has the disease became a major concern due to its dramatic incidence increase. This recent increase has been related to a variety of factors such as changes in the management of vineyards, new cultural practices, introduction of new grapevine cultivars and rootstocks, reduced sanitary care of rootstocks and vineyard propagation material, poor protection of pruning wounds, increasing use of selective fungicides and banishment of arsenite treatment due to its toxicity (Graniti et al. 2000; Surico et al. 2006). Although arsenite treatment did not appear to affect esca-related mortality, the treatments seemed effective in reducing esca symptoms (Di Marco et al. 2000; Fussler et al. 2008). The incidence of esca might also be increased as a result of climatic changes, namely variations in the precipitation (Surico et al. 2006).
The etiology of esca is still not completely understood. Several fungi have been associated with the disease and it is thought that esca is the result of the action of these microorganisms (Mugnai et al. 1999; Graniti et al. 2000). Fungi isolated from grapevines showing esca symptoms commonly include Phaeomoniella chlamydospora, as well as Phaeoacremonium aleophilum and Fomitiporia mediterranea or Fomitiporia punctata (Mugnai et al. 1996; Larignon and Dubos 1997; Mugnai et al. 1999; Chicau et al. 2000; Bruno and Sparapano 2006b). Pathogenicity tests have shown the capacity of P. chlamydospora to produce esca-like symptoms in the laboratory (Tabacchi et al. 2000; Sparapano et al. 2001b; Gubler et al. 2004; Bruno and Sparapano 2006a), as well as in vivo (Sparapano et al. 2001a; Feliciano et al. 2004; Gubler et al. 2004).
Information on the interaction of esca and grapevine is scarce, namely concerning the defence mechanism of the plant against the fungi associated with this disease. A main reason for the lack of information is the difficulty of host-pathogen field studies, due to the inherent complexity of the disease (in which several fungi seem to be involved), and the inconstancy of plant symptoms (which are highly affected by environmental factors). Furthermore, host–pathogen field trials frequently pose some problems: (i) field results can be misleading and they are frequently severely affected by seasonal influences, (ii) they do not separate the effect of the pathogen from effects induced by other biotic and/or abiotic agents present in the environment, and (iii) many field trial assays and breeding programs of woody plants are frequently time consuming and selection of resistant lines may take years (Smalley and Guries 1993). Instead, these authors recommend the use of short term assays in controlled conditions; the gathered information could then be used for in vivo assays in the field. Particularly in the study of esca, field studies may take several years of observations due to discontinuity of disease symptoms (Sparapano et al. 2001a; Marchi et al. 2006). So, in vitro cultures could represent an opportunity to study the putative defence mechanisms against esca disease. Nonetheless, reports on the infection of axenic grapevine plants or cells with esca-associated fungi are few. Some authors have co-cultured grapevine calli with esca-associated fungi resulting in a decrease of calli growth, the appearance of both symptoms and host response in a few weeks, and the distinction of susceptible and less susceptible cultivars (Sparapano et al. 2001b; Santos et al. 2005; Bruno and Sparapano 2006a). However, the few in vitro experiments published are consistent with ex vitro results, suggesting that in vitro assays may replace, at least as a first approach, the ex vitro assays (Santos et al. 2005 and references therein). These systems offer some advantages compared to the in vivo plant-pathogen interaction, which includes the simplicity of manipulation and a better control of external factors that can interfere with the whole-plant tissue metabolic responses, enabling a precise cause-effect determination.
In this study, we report the evaluation of phenolic compounds accumulation and reactive oxygen species production in cell suspension cultures of V. vinifera cv. Vinhão elicited with P. chlamydospora autoclaved biomass extract. We also evaluate the putative role of methyl jasmonate in these mechanisms, since jasmonates have been described as a signal molecules participating in defence responses. Particularly methyl jasmonate has been shown to enhance grapevine defences against fungal infections, and was even suggested as an alternative approach to protect grapevine against powdery mildew (Belhadj et al. 2006). Additionally, we investigate the molecular response of these cell suspension cultures to fungal extract elicitation by studying the activation of 7 defence-related genes transcription.
Materials and methods
In vitro cultures
Calli cultures were established from sterilized leaf segments of V. vinifera cv. Vinhão, collected in May/2003, in a vineyard in Braga - Portugal (Vinho Verde region). These cultures were initiated and maintained on solid medium, constituted by Gamborg B5 macronutrients (Duchefa) and Murashige and Skoog micronutrients (Duchefa) supplemented with 0.8% agar, 2% sucrose, 250 mg/l casein hydrolysate, 0.1 mg/l α-naphthaleneacetic acid and 0.2 mg/l kinetin. Before autoclaving, the pH was adjusted to 6.0 with NaOH 1 M. Cultures were grown at 25 ± 1°C, 16 h light/8 h dark photoperiod illuminated with fluorescent light bulbs (Oshram-Fluora) with a photon flux of 30 μmol/m2s. The calli were routinely subcultured with 4 week intervals. V. vinifera cv. Vinhão cell suspensions were initiated from calli obtained as described above. The cell suspension cultures were grown in 250 ml Erlenmeyer flasks containing 70 ml of liquid culture medium prepared as described above, except agar. The suspension cultures were maintained at the same conditions as calli cultures and shaken at 100 rpm. Subculture occurred every 2 weeks by transferring 10 ml of cell suspension into 70 ml of fresh medium.
Elicitor preparation
The fungus P. chlamydospora (isolated from V. vinifera plants showing esca symptoms in May/2003, in the same vineyard where plant biological material was collected) was maintained in the dark in solid Potato Dextrose Agar medium at room temperature. To obtain fungal biomass for the elicitor treatments, 1 l Erlenmeyer flasks containing 200 ml of fresh Mathur liquid medium (1 mg/l yeast extract, 1 mg/l bacto peptone, 10 g/l sucrose, 2.5 g/l MgSO47H2O and 2.7 g/l KH2PO4, pH 5.8) were inoculated with plugs of fungus culture and shaken at 250 rpm, at 26°C. After 2 weeks, the mycelium was harvested by filtration, lyophilised for 48 h and ground using mortar and pestle. The fungal elicitor was prepared by resuspending lyophilised fungal biomass in deionized water (40 mg dry weight biomass (dwb)/ml) and autoclaving for 15 min (120°C, 15 Ib/in2).
Determination of cell viability
Samples of cell suspension (150 μl) were mixed with 50 μl of trypan blue 0.4% (w/v) (Sigma) and incubated in the dark for 15 min. Cells were observed under a light microscope; non-viable cells were stained blue.
Elicitation, sample preparation and HPLC analysis
V. vinifera cv. Vinhão cell suspensions were divided into 3 sets: a control group and two elicited groups, one with 100 μM methyl jasmonate (Sigma) and the other one with a P. chlamydospora autoclaved biomass extract (40 mg dwb/culture flask). Elicitation occurred on the 5th culture day. At the 8th culture day the V. vinifera cells were harvested by centrifugation, lyophilized for 48 h and the dried biomass stored at −20°C until further analysis. Dried biomass of V. vinifera cells (0.1–0.2 g) was extracted in the dark with 5 ml of a 70% methanol solution acidified with 0.5% formic acid, during 1 day. The liquid phase was recovered, filtered and samples were stored at 4°C until HPLC analysis. The methanolic extracts were directly subjected to HPLC-DAD analysis: chromatographic separation was carried on a chromatographic Beckman Gold system equipped with a RP18 column (25x0.4 mm, particle size 5 μm, Merck), using water/formic acid (99:1) and methanol as the mobile phases. The elution gradient utilized was 5% of methanol at time 0 min, and 95% methanol at time 45 min. Quantification of stilbene type compounds was made at 306 nm, as resveratrol (Extrasynthese) equivalents, by the external standard method. Phenolic identification was also performed by HPLC–DAD-MSn. Chromatographic separation was carried out on a RP C18 column (25 × 0.4 mm, particle size 5 μm, Merck), using water/formic acid (99:1) and methanol as the mobile phases. Elution was performed as described above. The HPLC system was an Agilent HPLC 1100 instrument series equipped with an Agilent DAD detector (G1315B, Agilent Technologies) and mass detector in series, controlled by software from Agilent Technologies. The mass detector was an ion-trap mass spectrometer (G2445A, Agilent Technologies) equipped with an electrospray ionization (ESI) system and operated as described elsewhere (Silva et al. 2005).
Elicitation, sample preparation and determination of ROS levels
V. vinifera cv. Vinhão cell suspensions were divided into 3 sets: a control group and two elicited groups, one with 100 μM methyl jasmonate (Sigma) and the other with a P. chlamydospora autoclaved biomass extract (40 mg dwb/culture flask). On the 5th culture day, cell suspension cultures were elicited and several 1 ml samples were taken from time 0 (corresponding to the time of elicitation) until 7 h after elicitation. ROS determination procedure was based on previous description (Parsons et al. 1999): after elicitation time course samples (1 ml) were taken; these samples were added to a 200 μM H2DCFDA (Molecular Probes) solution to 2 μM final concentration and quickly vortexed. Samples were incubated in the dark, at room temperature, for 15 min. Samples were quick-spinned and 500 μl of supernatant was collected and diluted with 2.5 ml of ultrapure water. Fluorescence was measured at room temperature in a LS50 Luminescence Spectrometer (Perkin Elmer), with excitation and emission wavelengths of 488 nm and 525 nm, respectively. Fluorescence data was analyzed as previously described (Wang and Joseph 1999). Percentage of fluorescence change was calculated using the formula \( \% fluorescence\;change = \left( {\left( {Fx - F0} \right)/F0} \right) * 100 \); where Fx represents fluorescence at x minutes and F0 represents fluorescence at time 0.
Elicitation, sample preparation and molecular biology procedures
V. vinifera cv. Vinhão cell suspensions were divided into 2 groups, a control and other elicited with a P. chlamydospora autoclaved biomass extract (0.5 mg dwb/ml—corresponding to 40 mg dwb/culture flask), each one consisting of 2 pooled cell culture flasks. The pooling occurred under sterile conditions on the 5th culture day, prior to elicitation. After elicitation, 15-ml samples were taken 3, 12, 24 and 48 h post-elicitation. Cells were recovered by centrifugation at 4°C, immediately frozen in liquid nitrogen, and stored at −80°C. The frozen biomass was ground to powder with mortar and pestle in the presence of liquid nitrogen. Total RNA was extracted using the RNeasy Plant Mini Kit (QIAGEN GmbH) following supplier’s instructions with a minor alteration, which consisted of addition of a 2% (w/v) polyvinylpyrrolidone (Sigma) solution to supplied buffer RLT. According to the RNeasy Plant Mini Kit manufacturer, the purified RNA can be promptly used in subsequent applications such as cDNA synthesis without the need of DNase digestion, since RNeasy technology efficiently removes most of the DNA. To ensure that RNA extraction process was successful, quantity and quality of RNA were assessed spectrophotometrically in a NanoDrop 1000 Spectrophotometer (Thermo Fisher Scientific). cDNA was synthesized from extracted RNA using the First Strand cDNA Synthesis Kit (Fermentas UAB), according to supplier’s instructions. Quantity and quality of cDNA were determined spectrophotometrically in a NanoDrop 1000 Spectrophotometer (Thermo Fisher Scientific). Primer sequences of V. vinifera actin (ACT), phenylalanine ammonia lyase (PAL), stilbene synthase (STSY), β-1,3-glucanase (GLUC) and class 10 PR protein (PR-10) used were previously described (Bonomelli et al. 2004). Lipoxygenase-9 (LOX), class III chitinase (CH3) and class 6 PR protein (PR-6) primer sequences were designed based on V. vinifera nucleotide sequence deposited in GenBank, using Primer3 (http://fokker.wi.mit.edu/cgi-bin/primer3/primer3_www.cgi). All primer pairs ranged from 19 to 22 bp in length (STAB Vida). A list of primer pairs used, as well as the accession numbers of the nucleotide sequences on which primer design was based, is given in Table 1. The annealing temperature was optimized in a MyCycler™ Thermal Cycler (Bio-Rad) using 40 amplification cycles and a temperature gradient between 35 and 60°C. Amplification mixture consisted of 1X Green Flexi Reaction Buffer (Promega), 2.5 mM MgCl2 (Promega), 1 μM Primer sense, 1 μM Primer antisense, 0.8 mM dNTPs (Fermentas UAB), 5u GoTaq® Flexi DNA Polymerase (Promega) and 2 μl cDNA. PCR products were electrophoresed, alongside with a GeneRuler™ 50 bp DNA Ladder (Fermentas UAB), through a 3% (w/v) agarose (Sigma) gel stained with SYBR Safe™ (Molecular Probes). Images were captured using a ChemiDoc XRS (Bio-Rad). Temperature 60°C was selected for all genes in study to be the optimum temperature that originates a specific PCR product. Following this, cycle number was optimized for each gene in a MyCycler™ Thermal Cycler using a cycle gradient between 26 and 44 cycles in the amplification phase (each amplification cycle being 45 s 95°C, 1 min 60°C and 1 min 72°C). The amplification mixture was that detailed above. PCR products were electrophoresed, as described above. Images were captured using a ChemiDoc XRS and band intensities determined with Quantity One® Software (Bio-Rad). The amount of PCR product over the assayed range of cycles was examined, and an appropriate cycle number falling in the linear range was chosen for each gene. The number of cycles found to be appropriate for STSY, LOX, GLUC and CH3 were 34 cycles and 30 cycles for PAL, PR-6 and PR-10. The amplification of ACT was well within the linear amplification phase at both 30 and 34 cycles. All optimizations were made using pooled cDNA of all samples. PCR reactions were carried in a MyCycler™ Thermal Cycler using the amplification mixture detailed above and the optimized PCR program for each gene. PCR products were electrophoresed, images were captured and band intensities were measured as described above.
Statistical analysis
For HPLC assays, two independent experiments were performed with three independent replicates each. Average and standard deviations were calculated, followed by single factor analysis of variance (ANOVA) and Dunnett’s post-test for comparison of two or more groups with control. For ROS determination assays, three independent experiments with duplicates were performed. Treatment averages and standard deviations were calculated and averages used to determine % of fluorescence change as described above. For gene expression assays, two independent experiments were performed. All samples were normalized by corresponding actin gene expression. Control samples of each time were defined as 1x expression level.
Results
In vitro cultures
V. vinifera cv. Vinhão is an autochthonous recommended red grape variety to produce the wine denominated Vinho Verde. This variety is cultivated in the entire demarcated region due to its quality, being the only regional teinturier variety. Calli cultures were established from leaf explants as described in material and methods. These calli were friable and easily dispersed in liquid medium. After several subcultures (5–7 times) we obtained a homogeneous cell suspension of V. vinifera cv. Vinhão composed mainly by isolated or small cluster of 3–4 cells. These cultures proved to be stable, easy to manipulate, and showed an exponential growth phase between 3rd and 12th day after subculture. These cell suspensions were utilized in the elicitation experiments. Elicitation with P. chlamydospora autoclaved biomass extract induced browning of cultures. However, cells remained viable during the entire period of the experiment and biomass production was similar to that of control condition (data not shown).
Phenolics production
HPLC profiles of V. vinifera cell suspensions methanolic extracts, at 306 nm, are shown in Fig. 1. In control cultures the main compounds produced are the piceid type stilbenes: trans-piceid-glucoside, trans-piceid, trihydroxy-methoxy-stilben-glucoside and cis-piceid, identified in Fig. 1a (numbers 1, 2, 3 and 4, respectively). Compound 1 has a similar UV-spectra of compound 2 and a molecular ion m/z at 551.9 and -MS2 fragments at m/z 388.4 and 226.5 (resveraterol aglycone) corresponding to the sequential loss of glucose residues. Compound 2 and 4 have a similar deprotonated molecular ion m/z at 389.7 and a -MS2 fragment at m/z 226.5 (resveraterol aglycon) corresponding to the loss of a glucose residue. Compound 2 (306, 318 nm) and 4 (284 nm) have a UV-spectrum characteristic of a trans- and cis-piceid, respectively. Compound 3 has a molecular ion m/z at 419.2 and a -MS2 fragment at m/z 257.4 (trihydroxi-methoxi-stilben aglycon) due to a loss of a glucose residue.
HPLC profiles of V. vinifera cultures methanolic extracts, 306 nm, showing significant changes in phenolic production upon elicitation with both fungal extract and methyl jasmonate, including de novo production of viniferin type compounds. a control; b P. chlamydospora autoclaved biomass extract (0.5 mg/ml); c methyl jasmonate (100 μM). 1 - trans-piceid-glucoside; 2 - trans-piceid; 3 – trihydroxy-methoxy-stilben-glucoside; 4 - cis-piceid; 5 - ε-viniferin; 6 – ε-viniferin-diglucoside; 7 – ε-viniferin-glucoside; 8 – ε-viniferin polymer (2 molecules). Black numbers - piceid type compounds. Grey numbers - viniferin type compounds
Elicitation with P. chlamydospora autoclaved biomass extract, as well as elicitation with methyl jasmonate, induces significant changes in phenolic production, when compared to control. Along with the piceid type compounds identified with the numbers 1, 2 and 4, elicitation with P. chlamydospora autoclaved biomass extract also induces de novo production of viniferin type compounds identified in Fig. 1b with the grey numbers 5, 6, 7 and 8. These compounds were later identified by HPLC-DAD-MSn as a ε-viniferin, ε-viniferin-diglucoside, ε-viniferin-glucoside and a 2 molecule ε-viniferin polymer, respectively. Compound 5 has a deprotonated molecular ion m/z at 452.5 (viniferin aglycon). Compound 6 has a deprotonated molecular ion m/z at 776.9 and -MS2 fragments at m/z 614.5 and 452.5, corresponding to the sequential loss of glucose residues. Compound 7 has a deprotonated molecular ion m/z at 615.4 and -MS2 fragment at m/z 452.5 (loss of glucose, vineferin aglycon). Compound 8 has a molecular ion m/z at 907.5 and a -MS2 fragment at m/z 452.5 (viniferin aglycon).
As with P. chlamydospora autoclaved biomass extract, methyl jasmonate elicitation induces de novo production of the viniferin type compounds 6, 7 and 8, along with the production of piceid type stilbenes 1, 2 and 4 (Fig. 1c).
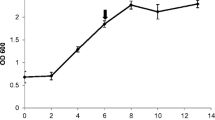
Total phenolic production also shows a significant increase after elicitation (Fig. 2), either with methyl jasmonate (9-fold increase) or, more markedly, with P. chlamydospora autoclaved biomass extract (20-fold increase), comparatively to control conditions. Total piceid production increases from 31.88 μg compound/g dwb in control cultures to 183.90 μg compound/g dwb in cultures elicited with methyl jasmonate and to 456.24 μg compound/g dwb in cultures elicited with P. chlamydospora autoclaved biomass extract. Also, de novo viniferin production occurs at a noteworthy level when cultures are elicited with methyl jasmonate (110.00 μg compound/g dwb) or with P. chlamydospora autoclaved biomass extract (188.55 μg compound/g dwb); the total viniferin production in these two conditions is not significantly different from each other.
Total phenolic production of V. vinifera cv. Vinhão cell cultures showing significant increases of piceid and viniferin production upon elicitation with both fungal extract and methyl jasmonate. C Control; MeJ MeJ (100 μM); Pch P. chlamydospora autoclaved biomass extract (0.5 mg/ml). Results are mean ± standard deviation of three independent replicates. Asterisks indicate significant statistical differences relative to control (* p < 0.05; ** p < 0.01)
ROS production
The kinetics for the accumulation of ROS in V. vinifera cell cultures is shown in Fig. 3. Both crude fluorescence and % of fluorescence change relative to 0 (time of elicitation) are represented (Fig. 3a and b, respectively). By calculating the % of fluorescence change relative to the time of elicitation the data reflects directly the percentage alteration in fluorescence with time since elicitation and once the influence of background fluorescence is removed (Wang and Joseph 1999). ROS levels of cells elicited with methyl jasmonate are similar to those observed in control cells, during the entire experimental period. However, elicitation with P. chlamydospora autoclaved biomass extract induces a biphasic oxidative burst. A swift and smaller burst arises around 25 min after elicitation followed by a second burst, more intense and sustained, peaking around 230 min after fungal elicitor addition.
ROS levels of V. vinifera cv. Vinhão cell cultures after P. chlamydospora elicitation showing that a biphasic oxidative burst is induced by fungal extract elicitation. a ROS levels indicated by crude fluorescence (results are mean ± standard deviation of duplicates); b ROS levels indicated by % of fluorescence change with time. C Control; MeJ MeJ (100 μM); Pch P. chlamydospora autoclaved biomass extract (0.5 mg/ml). Results represent typical data obtained in three independent experiments
Expression of defence-related genes
ACT was checked for its suitability to be the housekeeping gene in this experiment. It was found that ACT gene remains stable throughout the experimental conditions; not showing major differences after elicitation with P. chlamydospora autoclaved biomass extract or along the time, grapevine ACT gene was considered suited to be the housekeeping gene, and was used to normalize gene expression in different samples. Figure 4 shows expression levels of PR-6, PR-10, GLUC, CH3, PAL, STSY and LOX genes; control samples of each time were defined as 1x expression of that time. Although all these genes are shown to be constitutively expressed in V. vinifera cv. Vinhão cell cultures, P. chlamydospora autoclaved biomass extract elicitation increases the transcription of all these genes. Also, the results suggest that these genes are regulated during the time course of infection. PR-6, CH3, PAL, STSY and LOX show a biphasic pattern of induced expression, while PR-10 and GLUC induced expression peaks only once during the time of experiment. PR-6, CH3 and PAL transcripts have a peak at 3 h post elicitation followed by a second stronger increase from 24 h on (Fig. 4a, d and e). STSY and LOX transcripts reach a peak at 12 h post elicitation and show a new increase at 48 h (Fig. 4f and g). PR-10 and GLUC transcripts seem to hit the highest level at 12 h post elicitation, decreasing from then on (Fig. 4b and c).
Gene expression of V. vinifera cv. Vinhão cell cultures at 3, 12, 24 and 48 h after P. chlamydopora addition showing that elicitation increases the transcription of all genes studied in a modulated manner along time course of infection. C Control; Pch P. chlamydospora autoclaved biomass extract (0.5 mg/ml); a PR-6; b PR-10; c GLUC; d CH3; e PAL; f STSY; g LOX. Control samples of each time were defined as 1x expression level. Each sample was normalized by actin gene expression. Results represent typical data obtained in two independent experiments
Discussion
Growth of grapevines is of great economical importance, particularly in countries where its culture is a major component of national agriculture. Esca is one of the major diseases affecting the vine trunk; recently, its incidence has been increasing considerably, particularly in the Mediterranean region (Chiarappa 2000). This disease is characterized by both foliar and wood decay symptoms, which become difficult to study as the foliar symptoms can vary noticeably from year to year, even in the same vines (Mugnai et al. 1999). Along with the complexity of the symptoms, the identity of the causal pathogen is controversial. Together, these factors make the study of the disease in the field particularly difficult. We report here the use of V. vinifera cv. Vinhão cell suspension cultures to study the putative response of Vitis vinifera to P. chlamydospora (a fungus frequently isolated from vines infected with esca). We chose leaf explants to produce cell cultures, since foliar tissue is one where the symptoms of esca are evident in in vivo conditions, and so might originate a better model to study presumed V. vinifera defence mechanisms against fungi associated with this disease. The symptoms in leaves are believed to result from the presence of phytotoxins of fungal origin that are transported to the leaves via xylem sap. Particularly scytalone and isosclerone, both of which can be produced by P. chlamydospora, seem to be very toxic (Tabacchi et al. 2000; Bruno et al. 2007). The cultivar we used (cv. Vinhão) to generate the cell cultures is considered to be less affected by esca in the field, namely not showing external leaf symptoms so frequently, compared with other cultivars in the area. Therefore, some level of resistance/tolerance was suspected which led us to use this variety to investigate putative defence mechanisms against esca-related fungi.
In this study, we show that elicitation of V. vinifera cell cultures with the P. chlamydospora autoclaved biomass extract, as well as elicitation with methyl jasmonate, induces changes in the phenolic production of V. vinifera cell cultures, including de novo production of 3 viniferin type compounds identified as ε-viniferin-diglucoside, ε-viniferin-glucoside and a 2 molecule ε-viniferin polymer. The synthesis of phenolic phytoalexins is a recognized defence mechanism in plants, namely in grapevine. In Vitis spp. these substances accumulate in response to abiotic or biotic elicitors, such as UV irradiation (Langcake and Pryce 1977; Douillet-Breuil et al. 1999; Adrian et al. 2000) or bacterial and fungal pathogens like Pseudomonas syringae, Botrytis cinerea, Rhizopus stolonifer and Plasmopara viticola (Langcake 1981; Dai et al. 1995; Jeandet et al. 1995; Bavaresco et al. 1997; Sarig et al. 1997; Goetz et al. 1999; Robert et al. 2001). In grapevine, resveratrol and related compounds are the major forms of phytoalexins (Langcake 1981; Jeandet et al. 2002). Namely, accumulation of resveratrol, ε-viniferin, α-viniferin and pterostilbene was reported after inoculation of grapevine with the above mentioned pathogens (Langcake 1981; Dai et al. 1995; Jeandet et al. 1995; Bavaresco et al. 1997; Sarig et al. 1997; Robert et al. 2001). Recently, changes in phenolic production of grapevine calli (Bruno and Sparapano 2006a), grape roots (Del Rio et al. 2001) and young V. vinifera plants (Martin et al. 2009) in response to P. chlamydospora infection have been reported.
Therefore, it is possible that the de novo viniferin type compounds produced by our cell cultures might be involved in the defence mechanism against P. chlamydospora, since these compounds have been described as antifungal (Langcake 1981). Essentially, viniferins had been shown to be more fungitoxic than resveratrol. Resveratrol functions as a precursor of viniferins, which are dimers and oligomers of resveratrol formed by an oxidative dehydrogenation condensation process (Amalfitano et al. 2000; Langcake 1981). Furthermore, our results seem to agree with those obtained in vivo, since it was described an increase in the production of trans-resveratrol and ε-viniferin in wood of grapevines showing esca symptoms, as well as an increase in phenolic compounds in esca-diseased leaves (Amalfitano et al. 2000; Agrelli et al. 2009; Lima et al. 2010). Nonetheless, and considering the complexity that characterizes esca (a disease in which additional factors other than fungi may interfere), the possibility that an increase in stilbenic production might be not sufficient to fight the disease cannot be discounted. For example, an increase in phenolic production was detected in esca-affected leaves of V. vinifera cv. Alvarinho, which is another Vinho Verde grapevine variety that is recognizably susceptible towards esca; this suggested that even esca-susceptible varieties could mount a defence response against esca, but probably not in a timely manner that would allow the plant to cope with infection (Lima et al. 2010). Therefore, even if the plants are able to activate phenolic production as a defence response, this may be not sufficient to successfully fight the disease. So, although our cell culture system suggests that the grapevine variety Vinhão may be able to successfully combat the disease, namely because fungal elicitation induced de novo production of likely fungitoxic compounds, this must be confirmed in planta under natural conditions. Nevertheless, our cell culture system also showed that control cultures presented some amount of stilbenic compounds, suggesting that stilbenic production occurs even without elicitation in cv. Vinhão. Given the antifungal nature of these compounds, one can hypothesized that a basal level of preformed stilbenic compounds might be responsible for the apparent tolerance of this grapevine cultivar under natural conditions.
Jasmonates have been proposed as signal molecules involved in several biological processes, including the response to a variety of biotic and abiotic stresses (Creelman and Mullet 1997). For example, exogenous application of methyl jasmonate induces the synthesis of phytoalexins in several plant species, as well as induces de novo transcription of genes, including the gene of phenylalanine ammonia lyase, the key enzyme in the phenylpropanoid pathway (Gundlach et al. 1992). Application of methyl jasmonate to the surface of grapevine leaves results in the formation of lesions that mimic the typical hypersensitive response, and in the activation of associated defence responses like increase of salicylic acid production, accumulation of phenolic compounds and expression of defence-related genes (Repka et al. 2001). Moreover, elicitation of V. vinifera cell suspension cultures with methyl jasmonate results in phytoalexin accumulation, namely stilbene accumulation (Krisa et al. 1999). In this work, methyl jasmonate induced the production of the same phenolics stimulated by P. chlamydospora elicitation. Hence, it is possible that methyl jasmonate is involved in the direct defence mechanisms against P. chlamydospora, possibly as a signal molecule.
We also report that elicitation of V. vinifera cell cultures with the P. chlamydospora autoclaved biomass extract induces a biphasic oxidative burst profile after infection, with a smaller and swift first burst followed by a more intense and sustained second burst, which is typical of incompatible interactions. These results are, therefore, consistent with some level of resistance/tolerance suspected in the grapevine cultivar we used. The rapid generation of ROS is considered one of the earliest events following elicitation, namely, an oxidative burst profile with two phases; generally, compatible interactions induce a single nonspecific burst while incompatible interactions provoke a biphasic oxidative burst, the second burst being correlated to disease resistance (Lamb and Dixon 1997). Nonetheless, the information published in the literature concerning pathogen-induced oxidative burst is very diverse, regarding time of occurrence and intensity, depending on plant system studied or challenging factor used. Usually, ROS accumulation appears sooner in cell cultures, within a few minutes—which is consistent with our results, than in whole tissues or in vivo conditions, in which oxidative burst occurs after several hours (Wojtaszek 1997).
In addition, the expression of 7 recognized defence-related genes was investigated in our cell culture system. We report here early results on the activation of a defence response at the molecular level in V. vinifera cell cultures upon elicitation with P. chlamydospora autoclaved biomass extract. Investigation on the role of methyl jasmonate in modulating the transcription of defence-related genes is ongoing. The expression of all genes studied was shown to be enhanced in cell suspensions after fungal elicitor addition. The expression of PR proteins, namely PR-6, PR-10, PR-2 (β-1,3-glucanase - GLUC) and PR-8 (class III chitinase – CH3) was investigated. Expression of genes encoding enzymes of the octadecanoid (lipoxygenase – LOX) and phenylpropanoid (phenylalanine ammonia lyase – PAL – and stilbene synthase – STSY) pathways was also studied. The expression of all these genes was already shown to be induced in Vitis in response to several stresses. However, the timings and patterns of induction vary with experimental system or elicitor used. PR-6, PR-10, PR-2 and PR-3 expression has been shown to increase in grapevine leaves after UV-C irradiation (Bonomelli et al. 2004). Also, PR-6, PR-10, β-1,3-glucanase and class III chitinase transcripts were shown to accumulate in grapevine, and in grapevine cell cultures, in response to infection with several known pathogens, B. cinerea-derived elicitors, yeast extract, salicylic acid or related compounds (Busam et al. 1997; Jacobs et al. 1999; Robert et al. 2001; Bézier et al. 2002; Robert et al. 2002; Kortekamp 2006; Repka 2006; Fung et al. 2008). The importance of LOX in grapevine defence was already suggested by its up-regulation in leaves in response to UV-C irradiation (Bonomelli et al. 2004), as well as in grapevine cells elicited with laminarin or challenged with the B. cinerea-derived elicitors botrycin and cinerein (Aziz et al. 2003; Repka 2006). Because the production of stilbenic phytoalexins is one of the most common defence mechanisms in grapevine, it becomes obvious the importance of PAL and STSY (the enzyme that synthesizes resveratrol using the cinnamic acid p-coumaroyl-CoA derived from the phenylpropanoid pathway) in grapevine defence (Jeandet et al. 2002). In this way, PAL and STSY up-regulation has been reported in grapevine in response to several stresses, such as UV irradiation (Bonomelli et al. 2004; Borie et al. 2004), P. viticola, B. cinerea, P. syringae and E. necator infections (Robert et al. 2001; Bézier et al. 2002; Kortekamp 2006; Fung et al. 2008) and other elicitors including fungal cell walls, methyl jasmonate or B. cinerea-derived elicitors (Melchior and Kindl 1991; Repka et al. 2001; Repka, 2006). STSY was also shown to be induced in grapevine cell cultures challenged with fungal elicitor (Wiese et al. 1994).
Our results point towards the presence of a modulation of expression of the genes under study along the time course of infection, given that a biphasic pattern of induced expression is observed for PR-6, CH3, PAL, STSY and LOX genes, while PR-10 and GLUC genes appear to have only one phase of increased induced expression (at least during the time period investigated). It has been suggested that the biphasic gene accumulation may allow the plant to adapt to a specific attack and modulate the defence response; the biphasic shape results of the consecutive expression of two types of genes: a first type quickly expressed after elicitation with a rapid degrading mRNA, followed by a second slowly expressed gene type (Wiese et al. 1994). Particularly in what concerns increased expression of PAL and STSY after infection, the results here obtained in cell cultures agree with results obtained in vivo in esca-affected grapevines - in which the presence of P. chlamydospora was confirmed (Letousey et al. 2010).
Understanding the infection process involved in esca is an important step in order to develop strategies to fight the disease. However, studies on defence mechanisms of Vitis plants to esca are still scarce, in spite of the well documented presence of the disease in the field, particularly since the eighties of last century (and even before). In this study, V. vinifera cell suspension cultures elicited with both P. chlamydospora autoclaved biomass extract and methyl jasmonate changed their phenolic production when compared to control cultures. The increase in stilbene production after elicitation, specifically the production of viniferin type compounds already described as antifungal, the biphasic oxidative burst and the induction of several defence-related genes expression after elicitation with P. chlamydospora autoclaved biomass extract seem to indicate that V. vinifera cell suspension cultures, could be an important tool to study esca disease, particularly when considering that the results here obtained in vitro agree with observations in vivo in esca-affected plants. Therefore, these V. vinifera cell suspension cultures allow us to selectively study host defence response to esca-associated fungi without interference of external factors in a short period of time (2–3 weeks), leading to reliable results. Also, the experimental system allows the analysis of a large number of plants and/or cultivars. In conclusion, V. vinifera cv. Vinhão cell suspension cultures offer a simple, rapid and selective way to investigate the interaction between Vitis vinifera and esca-associated fungi, therefore representing a valuable model to study esca disease. Data acquired with this in vitro system can be further useful to design and conduct the more expensive and time-consuming field trials.
References
Adrian, M., Jeandet, P., Douillet-Breuil, A.-C., Tesson, L., & Bessis, R. (2000). Stilbene content of mature Vitis vinifera berries in response to UV-C elicitation. Journal of Agricultural and Food Chemistry, 48, 6103–6105.
Agrelli, D., Amalfitano, C., Conte, P., & Mugnai, L. (2009). Chemical and spectroscopic characteristics of the wood of Vitis vinifera Cv. sangiovese affected by esca disease. Journal of Agricultural and Food Chemistry, 57, 11469–11475.
Amalfitano, C., Evidente, A., Surico, G., Tegli, S., Bertelli, E., & Mugnai, L. (2000). Phenols and stilbene polyphenols in the wood of esca-diseased grapevines. Phytopathologia Mediterranea, 39, 178–183.
Aziz, A., Poinssot, B., Daire, X., Adrian, M., Bézier, A., Lambert, B., et al. (2003). Laminarin elicits defense responses in grapevine and induces protection against Botrytis cinerea and Plasmopara viticola. Molecular Plant-Microbe Interactions, 16, 1118–1128.
Bavaresco, L., Petegolli, D., Cantu, E., Fregoni, M., Chiusa, G., & Trevisan, M. (1997). Elicitation and accumulation of stilbene phytoalexin in grapevine berries infected by Botrytis cinerea. Vitis, 36, 77–83.
Belhadj, A., Saigne, C., Telef, N., Cluzet, S., Bouscaut, J., Corio-Costet, M.-F., et al. (2006). Methyl jasmonate induced defense responses in grapevine and triggers protection against Erysiphe necator. Journal of Agricultural and Food Chemistry, 54, 9119–9125.
Bézier, A., Lambert, B., & Baillieul, F. (2002). Study of defense-related gene expression in grapevine leaves and berries infected with Botrytis cinerea. European Journal of Plant Pathology, 108, 111–120.
Bonomelli, A., Mercier, L., Franchel, J., Baillieul, F., Benizri, E., & Mauro, M.-C. (2004). Response of grapevine defenses to UV-C exposure. American Journal of Enology and Viticulture, 55, 51–59.
Borie, B., Jeandet, P., Parize, A., Bessis, R., & Adrian, M. (2004). Resveratrol and stilbene synthase mRNA production in grapevine leaves treated with biotic and abiotic phytoalexin elicitors. American Journal of Enology and Viticulture, 55, 60–64.
Bruno, G., & Sparapano, L. (2006a). Effects of three-esca associated fungi on Vitis vinifera L.: I. Characterization of secondary metabolites in culture media and host responses to the pathogens in calli. Physiological and Molecular Plant Pathology, 69, 209–223.
Bruno, G., & Sparapano, L. (2006b). Effects of three esca-associated fungi on Vitis vinifera L.: II. Characterization of biomolecules in xylem sap and leaves of healthy and diseased vines. Physiological and Molecular Plant Pathology, 69, 195–208.
Bruno, G., Sparapano, L., & Graniti, A. (2007). Effects of three esca-associated fungi on Vitis vinifera L.: IV. Diffusion through the xylem of metabolites produced by two tracheiphilous fungi in the woody tissue of grapevine leads to esca-like symptoms on leaves and berries. Physiological and Molecular Plant Pathology, 71, 106–124.
Busam, G., Kassemeyer, H.-H., & Matern, U. (1997). Differential expression of chitinases in Vitis vinifera L. responding to systemic acquired resistance activators or fungal challenge. Plant Physiology, 115, 1029–1038.
Chiarappa, L. (2000). Esca (black measles) of grapevine. An overview. Phytopathologia Mediterranea, 39, 11–15.
Chicau, G., Aboim-Inglez, M., Cabral, S., & Cabral, J. (2000). Phaeoacremonium chlamydosporum and Phaeoacremonium angustius associated with esca and grapevine decline in Vinho Verde grapevines in northwest Portugal. Phytopathologia Mediterranea, 39, 80–86.
Creelman, R., & Mullet, J. (1997). Oligosaccharins, brassinolides, and jasmonates: nontraditional regulators of plant growth, development and gene expression. The Plant Cell, 9, 1211–1223.
Dai, G., Andary, C., Mondolot-Cosson, L., & Boubals, D. (1995). Histochemical studies on the interaction between three species of grapevine, Vitis vinifera, V. rupestris and V. rotundifolia and the downy mildew fungus, Plasmopara viticola. Phisiological and Molecular Plant Pathology, 46, 177–188.
Del Rio, J., Gonzalez, A., Fuster, M., Botia, J., Gomez, P., Frias, V., et al. (2001). Tylose formation and changes in phenolic compounds of grape roots infected with Phaeomoniella chlamydospora and Phaeoacremonium species. Phytopathologia Mediterranea, 40, S394–S399.
Di Marco, S., Mazzullo, A., Calzarano, F., & Cesari, A. (2000). The controlo f esca: status and perspectives. Phytopathologia Mediterranea, 39, 232–240.
Douillet-Breuil, A.-C., Jeandet, P., Adrian, M., & Bessis, R. (1999). Changes in the phytoalexin content of various Vitis spp. in response to ultraviolet C elicitation. Journal of Agricultural and Food Chemistry, 47, 4456–4461.
Feliciano, A., Eskalen, A., & Gubler, W. (2004). Differential susceptibility of three grapevine cultivars to Phaeoacremonium aleophilum and Phaeomoniella chlamydospora in California. Phytopathologia Mediterranea, 43, 66–69.
Fung, R., Gonzalo, M., Fekete, C., Kovacs, L., He, Y., Marsh, E., et al. (2008). Powdery mildew induces defense-oriented reprogramming of the transcriptome in a susceptible but not in a resistant grapevine. Plant Physiology, 146, 236–249.
Fussler, L., Kobes, N., Bertrand, F., Maumy, M., Grosman, J., & Savary, S. (2008). A characterization of grapevine trunk diseases in France from data generated by the national grapevine wood diseases survey. Phytopathology, 98, 571–579.
Goetz, G., Fkyerat, A., Métais, N., Kunz, M., Tabacchi, R., Pezet, R., et al. (1999). Resistance factors to grey mould in grape berries: identification of some phenolics inhibitors of Botrytis cinerea stilbene oxidase. Phytochemistry, 52, 759–767.
Graniti, A., Surico, G., & Mugnai, L. (2000). Esca of grapevine: a disease complex or a complex of diseases? Phytopathologia Mediterranea, 39, 16–20.
Gubler, W., Thind, T., Feliciano, A., & Eskalen, A. (2004). Pathogenicity of Phaeoacremonium aleophilum and Phaeomoniella chlamydospora on grape berries in California. Phytopathologia Mediterranea, 43, 70–74.
Gundlach, H., Müller, M., Kutchan, T., & Zenk, M. (1992). Jasmonic acid is a signal transducer in elicitor-induced plant cell cultures. Proceedings of the National Academy of Sciences USA, 89, 2389–2393.
Jacobs, A., Dry, I., & Robinson, S. (1999). Induction of different pathogenesis-related cDNAs in grapevine infected with powdery mildew and treated with ethephon. Plant Pathology, 48, 325–336.
Jeandet, P., Bessis, R., Sbagghi, M., & Meunier, P. (1995). Production of the phytoalexin resveratrol by grapes as a response to Botrytis attack under natural conditions. Journal of Phytopathology, 143, 135–139.
Jeandet, P., Douillet-Breuil, A.-C., Bessis, R., Debord, S., Sbaghi, M., & Adrian, M. (2002). Phytoalexins from the Vitaceae: biosynthesis, phytoalexin gene expression in transgenic plants, antifungal activity and metabolism. Journal of Agricultural and Food Chemistry, 50, 2731–2741.
Kortekamp, A. (2006). Expression analysis of defence-related genes in grapevine leaves after inoculation with a host and a non-host pathogen. Plant Physiology and Biochemistry, 44, 58–67.
Krisa, S., Larronde, F., Budzinsky, H., Decendit, A., Deffieux, G., & Mérillon, J.-M. (1999). Stilbene production by Vitis vinifera cell suspension cultures: methyl jasmonate induction and 13 C biolabeling. Journal of Natural Products, 62, 1688–1690.
Lamb, C., & Dixon, R. (1997). The oxidative burst in plant disease resistance. Annual Reviews of Plant Physiology and Plant Molecular Biology, 48, 251–275.
Langcake, P. (1981). Disease resistance of Vitis spp. and the production of stress metabolites resveratrol, ε-viniferin, α-viniferin and pterostilbene. Physiological Plant Pathology, 18, 213–226.
Langcake, P., & Pryce, R. (1977). The production of resveratrol and the viniferins by grapevines in response to ultraviolet irradiation. Phytochemistry, 16, 1193–1196.
Larignon, P., & Dubos, B. (1997). Fungi associated with esca disease in grapevine. European Journal of Plant Pathology, 103, 147–157.
Letousey, P., Baillieul, F., Perrot, G., Rabenoelina, F., Boulay, M., Vaillant-Gaveau, N., et al. (2010). Early events prior to visual symptoms in the apoplectic form of grapevine esca disease. Phytopathology, 100, 424–431.
Lima, M., Felgueiras, M., Graça, G., Rodrigues, J., Barros, A., Gil, A., et al. (2010). NMR metabolomics of esca disease-affected Vitis vinifera cv. Alvarinho leaves. Journal of Experimental Botany, 61, 4033–4042.
Marchi, G., Peduto, F., Mugnai, L., Di Marco, S., Calzarano, F., & Surico, G. (2006). Some observations on the relationship of manifest and hidden esca to rainfall. Phytopathologia Mediterranea, 45, S117–S126.
Martin, N., Vesentini, D., Rego, C., Monteiro, S., Oliveira, H., & Ferreira, R. (2009). Phaeomoniella chlamydospora infection induces changes in phenolic compounds content in Vitis vinifera. Phytopathologia Mediterranea, 48, 101–116.
Melchior, F., & Kindl, H. (1991). Coordinate- and elicitor-dependent expression of stilbene synthase and phenylalanine ammonia-lyase genes in Vitis cv. Optima. Archives of Biochemistry and Biophysics, 288, 552–557.
Mugnai, L., Surico, G., & Esposito, A. (1996). Microflora associata al mal dellésca della vite in Toscana. Informatore Fitopatologico, 11, 49–55.
Mugnai, L., Graniti, A., & Surico, G. (1999). Esca (black measles) and brown wood-streaking: two old and elusive diseases of grapevines. Plant Disease, 83, 404–418.
Parsons, H., Yip, J., & Vanlerberghe, G. (1999). Increased respiratory restriction during phosphate-limited growth in transgenic tobacco cells lacking alternative oxidase. Plant Physiology, 121, 1309–1320.
Repka, V. (2006). Early defence responses induced by two distinct elicitors derived from a Botrytis cinerea in grapevine leaves and cell suspensions. Biologia Plantarum, 50, 94–106.
Repka, V., Fischerová, I., & Silhárová, K. (2001). Methyl jasmonate induces a hypersensitive-like response of grapevine in the absence of avirulent pathogens. Vitis, 40, 5–10.
Robert, N., Ferran, J., Breda, C., Coutos-Thévenot, P., Boulay, M., Buffard, D., et al. (2001). Molecular characterization of the incompatible interaction of Vitis vinifera leaves with Pseudomonas syringae pv. pisi: expression of genes coding for stilbene synthase and class 10 PR protein. European Journal of Plant Pathology, 107, 249–261.
Robert, N., Roche, K., Lebeau, Y., Breda, C., Boulay, M., Esnault, R., et al. (2002). Expression of grapevine chitinase genes in berries and leaves infected by fungal or bacterial pathogen. Plant Science, 162, 389–400.
Santos, C., Fragoeiro, S., & Phillips, A. (2005). Physiological response of grapevine cultivars and a rootstock to infection with Phaeoacremonium and Phaeomoniella isolates: an in vitro approach using plants and calluses. Scientia Horticulturae, 103, 187–198.
Sarig, P., Zutkhi, Y., Monjauze, A., Lisker, N., & Ben-Arie, R. (1997). Phytoalexin elicitation in grape berries and their susceptibility to Rhizopus stolonifer. Physiological and Molecular Plant Pathology, 50, 337–347.
Silva, B., Ferreres, F., Malva, J., & Dias, A. (2005). Phytochemical and antioxidant characterization of Hypericum perforatum alcoholic extracts. Food Chemistry, 90, 157–167.
Smalley, E., & Guries, R. (1993). Breeding elms for resistance to Dutch elm diseases. Annual Review of Phytopathology, 31, 325–352.
Sparapano, L., Bruno, G., & Graniti, A. (2001a). Three-year observation of grapevines cross-inoculated with esca-associated fungi. Phytopathologia Mediterranea, 40, S376–S386.
Sparapano, L., De Leonardis, S., Campanella, A., & Bruno, G. (2001b). Interaction between esca-associated fungi, grapevine calli and micropropagated shoot cultures of grapevine. Phytopathologia Mediterranea, 40, S423–S428.
Surico, G., Mugnai, L., & Marchi, G. (2006). Older and more recent observations on esca: a critical overview. Phytopathologia Mediterranea, 45, S68–S86.
Tabacchi, R., Fkyerat, A., Poliart, C., & Dubin, G.-M. (2000). Phytotoxins from fungi of esca of grapevine. Phytopathologia Mediterranea, 39, 156–161.
Wang, H., & Joseph, J. (1999). Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radical Biology & Medicine, 27, 612–616.
Wiese, W., Vornam, B., Krause, E., & Kindl, H. (1994). Structural organization and differential expression of three stilbene synthase genes located on a 13 Kb grapevine DNA fragment. Plant Molecular Biology, 26, 667–677.
Wojtaszek, P. (1997). Oxidative burst: an early plant response to pathogen infection. Biochemical Journal, 322, 681–692.
Acknowledgements
Marta Lima was supported by Fundação para a Ciência e a Tecnologia (FCT) through the PhD grant SFRH/BD/17944/2004.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lima, M.R.M., Ferreres, F. & Dias, A.C.P. Response of Vitis vinifera cell cultures to Phaeomoniella chlamydospora: changes in phenolic production, oxidative state and expression of defence-related genes. Eur J Plant Pathol 132, 133–146 (2012). https://doi.org/10.1007/s10658-011-9857-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-011-9857-4