Abstract
Public concerns about cyanotoxins production in water and its detrimental impacts on human and animal health are growing primarily due to the widespread eutrophication observed in aquatic ecosystems. A review of relevant literature was done to determine the degree of cyanotoxin occurrence and its harmful effects in African waterbodies. Data were extracted from 64 published studies from 1990 to 2022 that quantified the concentration of cyanotoxins in African aquatic ecosystems. Cyanotoxins have been reported in 95 waterbodies (29 lakes, 41 reservoirs, 10 ponds, 9 rivers, 5 coastal waters, and 1 irrigation canal) from 15 African countries. Cyanotoxins were documented in all the regions of Africa except the central region. Microcystins have been reported in nearly all waterbodies (98.9%), but anatoxin-a (5.3%), cylindrospermopsin (2.1%), nodularins (2.1%), homoanatoxin-a (1.1%), and β-N-methylamino-l-alanine (1.1%) were encountered in a small number of water ecosystems, homoanatoxin-a and β-N-methylamino-l-alanine each occurred in one waterbody. The largest concentrations of microcystins and nodularins were reported in South African Lakes Nhlanganzwani (49,410 μg L−1) and Zeekoevlei (347,000 μg g−1). Microcystin concentrations exceeding the WHO guideline for lifetime drinking water (1 μg L−1) were reported in 63% of the aquatic ecosystems surveyed. The most frequently reported toxin-producing cyanobacteria genus is Microcystis spp. (73.7%), followed by Oscillatoria spp. (35.8%) and Dolichospermum spp. (33.7%). Cyanotoxin-related animal mortality and human illness were reported in the continent. Consequently, it is necessary to regularly monitor the level of nutrients, cyanobacteria, and cyanotoxins in African waterbodies in an integrated manner to devise a sustainable water resources management.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
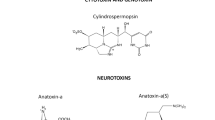
The formation of cyanobacterial blooms, which are usually linked to the development of cyanotoxins, is favored by excessive nutrient enrichment, water stillness, and favorable light and temperature conditions (Merel et al., 2013; Paerl, 2014). Cyanotoxins are commonly alkaloids of low molecular weight or cyclic peptides that cause harm to organisms, including humans (Carmichael et al., 2001; Metcalf & Codd, 2012). They are secondary metabolites, the organism does not utilize them for its primary metabolism and either retains them in producer cells or releases them into the water (Metcalf & Codd, 2012). Cyanotoxins comprise over a hundred different chemicals, each with a unique chemical structure and toxicity (Manning & Nobles, 2017).
Many authors have linked the poisoning and death of animals to cyanotoxins (Carmichael et al., 2001; Chellappa et al., 2008; Harding & Paxton, 2001; Masango et al., 2010; Scott et al., 1981; Shaw et al., 2002; Soll & Williams, 1985; Steyn, 1943; Van Halderen et al., 1995). Microcystins (MCs) are the most commonly reported cyanotoxins (Dawson, 1998; Gupta et al., 2003), and are known to cause cell necrosis, massive hemorrhage, and death (Hooser, 2000; Kuiper-Goodman et al., 1999). Carmichael et al. (2001), Funari and Testai (2008), and Buratti et al. (2017) documented several cases of human intoxication associated with cyanotoxins. Exposure to cyanotoxin-contaminated waters is known to cause skin irritations, conjunctivitis, earaches, gastroenteritis, respiratory diseases, allergic responses, and liver damage (Dietrich & Hoeger, 2005; Vidal et al., 2017).
Nearly every nation in the world has reported the occurrence of cyanobacteria and cyanotoxins (Fristachi et al., 2008), Microcystis and MCs being predominant in many countries (Harke et al., 2016; Ndlela et al., 2016). MCs are the most often reported cyanotoxins worldwide, followed by cylindrospermopsin (CYN), anatoxin-a (ATX-a), saxitoxins (STX), and nodularins (NOD) (Pelaez et al., 2010; Svirčev et al., 2019). In several cases, the levels of MCs, CYN, and STX have exceeded the provisional standards for drinking and recreational water around the world (Loftin et al., 2016; Roegner et al., 2020; Szlag et al., 2015). The most commonly reported cyanobacterial genera found worldwide with cyanotoxins in environmental samples are Microcystis spp., Dolichospermum (Formerly: Anabaena) spp., Aphanizomenon spp., Planktothrix spp., and Oscillatoria spp. (Ibelings et al., 2021; Svirčev et al., 2019).
There are numerous reports of cyanotoxin production in the waterbodies of Africa and its adverse effects. Animal illness and mass deaths have been documented for decades in the aquatic ecosystems of South Africa, Morocco, and Kenya (Krienitz et al., 2005; Oberholster et al., 2009; Oudra et al., 2001, 2002; Scott, 1991; Steyn, 1945). Many Africans have been experiencing broad toxicities resulting in itching, sore eyes, gastroenteritis, and skin rashes (Zilberg, 1966). The problem of eutrophication and algal blooming in Africa is exacerbated due to rapid population growth, urbanization, intensive agriculture, and industrialization (Giri, 2021; Juma et al., 2014).
There is paucity of compiled information on the presence and quantification of cyanotoxins and cyanobacteria in African waterbodies. This could probably be due to lack of resource, skilled technicians and analytical instruments to detect and quantify cyanotoxins. Therefore, this review aims to thoroughly examine the research articles about the prevalence of cyanotoxins and their adverse effects in African waters. This review on several cyanotoxins research aspects will serve as a foundation for ongoing research, monitoring, and management of cyanobacterial blooms in various water resources of Africa.
Research approach: inclusion and exclusion of literature
Articles, books, book chapters, and brief communications that addressed the occurrence, toxic effects, and health risk of cyanotoxins to humans and animals in African aquatic ecosystems was gathered from the Scopus, Web of Science, PubMed, and Science Direct databases. Articles that were published from 1990 to 2022 was searched using a combination of relevant key words/phrases that include “cyanotoxins,” “microcystin,” “cyanobacteria,” “mortality,” “adverse effects” “African waterbodies,” “Eutrophication,” “Southern African waterbodies,” “Eastern African aquatic ecosystems,” “North African waters,” “West African water resources,” and “Central Africa water systems.” Key words/phrases were combined using conjunctions like AND, AND NOT, NOT, and OR.
Inclusion and exclusion of literature was conducted based on the preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines checklist (Salameh et al., 2020) that include journal ranking, impact factor, the relevance of the article to the research theme, reproducibility, and novelty. Abstracts and full-text contents were thoroughly assessed to check the quality and relevance of literature to study objectives. Consequently, 32 publications out of a total of 172 assessed literature found were rejected for not meeting the minimum set requirements (Fig. 1). Numerical data of cyanotoxin concentrations were extracted from only 64 articles studied in various African aquatic ecosystems.
Lake Victoria was classified as a three-ecosystem since it was regarded as a water resource for Kenya, Tanzania, and Uganda. The cyanotoxin concentrations are presented as maximum content in two different units (µg L–1 and µg g–1). Toxin concentrations that were reported in µg L−1 only were compared with the WHO recommended values of drinking and recreational water. The analysis did not include studies conducted on tap water and experimental research involving cyanotoxins.
The role of eutrophication in the occurrence of cyanotoxins in African water bodies
In many African countries, industrialization and urbanization coupled with intensive agricultural practices driven by rapid population growth led to land degradation with knock-on effect on excess nutrient enrichment into aquatic systems (Fetahi, 2019; Giri, 2021; Zinabu et al., 2018). Domestic and municipal wastewater, increased applications of chemical fertilizers, and discharging industrial effluents into nearby aquatic systems without adequate treatment have led to increased nutrient loads into surface water resulting in eutrophication (Akale et al., 2018; Lanckriet et al., 2017). About 28% of African waterbodies are seriously impaired by eutrophication due to increased developmental activities with some sort of production work (Nyenje et al., 2010). The concentration of total phosphorus greater than 0.01 mg L−1 and total nitrogen above 0.3 mg L−1 are enough to cause eutrophication (Tibebe et al., 2022).
In many African countries, recent agricultural intensification with crop–livestock production has led to excess applications of chemical fertilizers along the catchment (Fetahi, 2019; Nyenje et al., 2010; Van Ginkel, 2011). Consequently, runoff from agricultural land washing fertilizers and organic-rich surface soil accelerates the eutrophication of aquatic systems (Lijklema, 1995; Van Ginkel, 2011). Fertilizers applied on farmlands are not completely utilized, a rather large quantity of fertilizer either remain in the soil or gradually washed into aquatic systems due to poor farming practice and reduced water and soil conservation measures (Buratti et al., 2017; Lijklema, 1995). It has been acknowledged that eutrophication is the main factor favoring cyanotoxin production (Chorus et al., 2021; Dolman et al., 2012; Sasner et al., 1981; Tanvir et al., 2021). In various African waterbodies, there are numerous examples of cyanotoxins presence in high concentrations in eutrophic and hypertrophic aquatic systems (Eguzozie et al., 2016; Harding et al., 1995; Masango et al., 2010; Ndlela et al., 2016; Scott et al., 1981, 2018).
Occurrence of cyanotoxins and cyanobacteria in African waterbodies
We documented cyanotoxins occurrence reports on 95 waterbodies (29 lakes, 41 reservoirs, 10 ponds, 9 rivers, 5 coastal waters, and 1 irrigation canal) in 15 African countries (Fig. 2). Svirčev et al. (2019) reviewed publications until 2018 and indicated the presence of cyanotoxins occurrence in fourteen African countries in 76 freshwater ecosystems. In the present review, almost all aquatic ecosystems (98.9%) contained MCs, while ATX-a (5.3%), CYN (2.1%), NOD (2.1), homoanatoxin-a (HTX) (1.1%), and β-N-methylamino-l-alanine (BMAA) were reported in few waters. MCs are the most frequently reported toxin (60) in the literature surveyed, while other cyanotoxins such as ATX-a (5), NOD (2), CYN (2), HTX (1), and BMAA (1) have minor contributions. ATX-a was quantified in Kenyan ecosystems only. All concentrated extracts of the Ethiopian Rift Valley Lakes had trace amounts of CYN, but none of the concentrations were high enough to be detected by ELISA (Willén et al., 2011). HTX and BMAA, each were reported in one African aquatic system only. The highest amounts of NOD and MCs were 347,000 µg g–1 and 49,410 µg L–1 in lakes Zeekoevlei and Nhlanganzwani, South Africa, respectively (Table 3).
Most of the waterbodies surveyed and reported in μg L−1 (83.6%) contained less than 10 μg L−1 cyanotoxin from 73 waterbodies. The majority of the toxin content reported as µg g–1 (59.1%) was higher than 200 µg g–1. The MCs concentrations in 63% of the water systems were greater than the WHO guideline threshold of lifetime drinking water (1 μg L−1). Twelve waterbodies found in six countries contained MCs concentrations greater than WHO’s provisional guideline values for short-term drinking water (12 μg L−1) and recreational water (24 μg L−1) (Tables 1 and 2). The ATX-a, NOD, CYN, HTX, and BMAA concentrations reported did not exceed threshold values of short-term drinking-water and recreational water. However, fish ponds in Sohag province, Egypt, were reported to have CYN amounts (2.76 µg L−1) above the lifetime drinking-water guideline value set by WHO (0.7 μg L−1).
Aquatic ecosystems studied at least twice such as Hartbeespoort Reservoir (South Africa), Nile River (Egypt), Lake Oubeira (Algeria), and Lake Koka (Ethiopia), showed generally declining trends of cyanotoxins concentrations between 1995 and 2015. Lake Victoria’s MCs concentration has been steadily increasing until 2017 before it decreased to one of the lowest values (0.02 μg L−1). However, Lake Mburo (Uganda), Lebna Reservoir (Tunisia), Ahmadu Bello University (ABU) Reservoir (Nigeria), Lake Bomo (Nigeria), and Lake Makwaye (Nigeria), have showed increments in MCs content between 2003 and 2013. These variations can be linked to management efforts and nutrient enrichment driven by anthropogenic activities.
The most frequently found cyanobacteria with cyanotoxins in water samples in African aquatic ecosystems were Microcystis spp. (73.7%), followed by Oscillatoria spp. (35.8%) and Dolichospermum spp. (33.7%). Other cyanobacterial genera that were present with cyanotoxins in environmental samples in at least 10% of the waterbodies include Merismopedia (24.2%), Pseudanabaena (22.1%), Chroococcus (17.9%), Planktothrix (16.8%), Spirulina (14.7%), Raphidiopsis (9.5%), and Synechococcus (9.5%).
Southern Africa
The occurrence and distribution of cyanotoxins and cyanobacteria have been extensively researched in South African waterbodies. In contrast to NOD, which was only identified in Lake Zeekoevlei, MCs were found in 94.1% of the 17 Southern African aquatic systems surveyed (Table 3). A total of 88.2% of the water systems surveyed had MCs levels over the WHO-recommended limit of 1 μg L−1 for drinking water, and of those, 46.2% had levels above the threshold for both short-term drinking water and recreational water use. In Lake Zeekoevlei, Nhlanganzwane Reservoir, and Kruger National Park Reservoirs (Nhlanganzwani, Mpanamana, Makhohlola, and Sunset), the concentration of cyanotoxins (MCs and NOD) was so high (up to 49,410 μg L−1) that it was linked to serious illness and mortality in domestic and wild animals (Harding et al., 1995; Masango et al., 2010; Oberholster et al., 2009). Between 1984 and 2011, Lake Hartbeespoort's particulate MCs remained stable, while its dissolved MCs decreased between 2005 and 2013. This is consistent with a decline in productivity between 2010 and 2018, which is probably caused by a fall in the water temperature (Ali et al., 2022).
MCs measurements in Botswana, Mozambique, and Zimbabwe waterbodies were less than guideline value for short-term drinking water set by WHO (12 μg L−1), although many of them were higher than the limit set by WHO for drinking water (1 μg L−1) (Table 3). Oreochromis mossambicus and Labeo rosae had estimated daily intake values of MCs concentration in muscle that were 4.1 and 4.6 times, respectively, higher than the recommended tolerable daily intake (TDI) value on 0.04 μg kg−1 of body weight (Nchabeleng et al., 2014). In some Southern African water ecosystems that are sources of drinking water, the presence of MCs at concentrations higher than 1 μg L−1 is especially worrisome (Mhlanga et al., 2006; Pedro et al., 2012; Recknagel et al., 2017).
Microcystis is the most frequently reported (82.3%) cyanobacteria together with the cyanotoxin in Southern African water resources. Some South African waterbodies, including the Vaal Reservoir, Theewaterskloof Reservoir, Hartbeespoort Reservoir, Lake Zeekoevlei, and Orange River, have been found to contain potentially toxin-producing cyanobacteria such as Microcystis aeruginosa, Microcystis flos-aquae, Dolichospermum spp., Oscillatoria spp., Nodularin spumigena and Raphidiopsis raciborskii, Spirulina spp., and Planktothrix (Harding & Paxton, 2001; Scott et al., 1981; Steyn, 1945; Van Halderen et al., 1995).
East Africa
The concentrations of MCs in East African waterbodies ranged from trace to hazardous levels, reaching up to 453.89 μg L−1 (Legedadi Reservoir, Ethiopia) and 19,800 µg g–1 (Lake Baringo, Kenya) (Table 4). From 33 water resources surveyed, the majority of them (57.6%) had MCs concentrations over the WHO standard for drinking water (1 μg L−1). Lake Koka (Ethiopia), Legedadi Reservoir (Ethiopia), and Lake Victoria (Uganda) contained MCs levels that exceed WHO guideline limits of drinking water for short duration and recreational water. Legedadi Reservoir, a major supply of drinking water for Addis Ababa, Ethiopia, had an extremely high MCs concentration (453.89 μg L−1), which could be hazardous to end users' health (Major et al., 2018). Five lakes in Kenya (Lakes Baringo, Simbi, Sonachi, Bogoria, and Nakuru) contain the neurotoxic ATX-a, with concentrations up to 1260 µg g–1 (Lake Baringo).
Lesser Flamingo deaths were associated with the hot spring shore of Lake Bogoria, which had three times more MCs content than the western shore (Ballot et al., 2004; Krienitz et al., 2003). Different MC variants, specifically MC-LR, MC-YR, MC-RR, MC-LA, MC-LF, MC-dmLR, MC-dmRR, LA[NMeSer7]-MC-YR, [Asp3]-MC-RY, [MeAsp3]-MC-RY, and [NMeSer7]-MC-YR), were found in both the water samples and algal seston (Habtemariam et al., 2021; Major et al., 2018; Okello et al., 2010; Olokotum et al., 2022). CYN was found to be the most prevalent cyanotoxin found around the lakeshores in Lake Victoria, Tanzania, where NOD and MCs also co-occurred (Mchau et al., 2021). HTX was also reported in Lake Victoria, Uganda.
Microcystis dominated the freshwater bodies of East Africa, which occurred in 81.8% of the surveyed waterbodies. The cyanobacterial genera Merismopedia, Dolichospermum, Arthrospira, Pseudanabaena, and Anabaenopsis are present in at least five water ecosystems of East Africa in the habitats that exhibited detectable cyanotoxins. Arthrospira fusiformis dominated three of the five water resources that contained ATX-a (Lakes Simbi, Sonachi, and Bogoria). ATX-a and MC-YR were produced by A. fusiformis monocyanobacterial strain that was isolated from Lake Sonachi (Ballot et al., 2005). The mcyE gene, a member of the microcystin synthesis gene cluster, in field samples taken from Lake Naivasha in Kenya, and the mcyB genotype of Microcystis and Planktothrix species in freshwater lakes in Uganda were identified to produce cyanotoxins (Krienitz et al., 2013; Okello et al., 2010).
North Africa
MCs occurred in all of the North African waterbodies surveyed, with many congeners detected from various ecosystems (up to 21 in Lake des Oiseaux) (Bouhaddada et al., 2016). Of the 19 aquatic ecosystems documented, 63.2% had MCs concentrations higher than the WHO's provisional recommendation limit of 1 μg L−1 for drinking water (Table 5). Fish ponds in the Sohag province (Egypt), Lalla Takerkoust Reservoir (Morocco), and Lake Oubeira (Algeria) have cyanotoxins levels beyond the WHO guidelines for short-term drinking water use and recreational water. In the province of Sohag, a fish pond with a high concentration of CYN (2.76 µg L−1) may cause the accumulation of the toxin in fish, putting consumers at risk. The main sources of drinking water, Lake Oubeira and Lalla Takerkoust Reservoir contained extremely high levels of MCs up to 29,163 μg L−1. The presence of high concentrations of MCs was linked to turtle deaths that occurred during a Microcystis spp. bloom in Lake Oubeira (Nasri et al., 2008). Tomato plants exposed to a crude extract of a harmful cyanobacterial bloom from Lalla Takerkoust Reservoir experienced leaf tissue necrosis (El Khalloufi et al., 2012).
There are a few instances in Egypt and elsewhere where water treatment plants not only failed to completely remove cyanobacterial cells and degrade toxins, but also lysed the cyanobacteria's cells and released significant amounts of MCs (Eynard et al., 2000; Ling, 2000; Mohamed et al., 2015; Mohamed, 2016a). The final treated water's MCs concentration was 3.8 µg L−1, much higher than the quantity found in the untreated water source from Egypt's Nile River (Mohamed, 2016a). Fishponds in Egypt contained considerably high concentrations of MCs and CYN that exceed WHO guideline values of 1 μg L−1. The muscle of common carp from Lake Oubeira exceeded the WHO lifetime limit for the tolerated daily consumption of MCs (Amrani et al., 2014).
Oscillatoria was the most frequently reported (84.2%) cyanobacteria found with the cyanotoxins in water samples from North African waterbodies surveyed. Other cyanobacteria with a smaller frequency of occurrence include Microcystis (73.7%), Pseudanabaena (57.9%), Merismopedia (57.9%), Chroococcus (47.4%), Dolichospermum (36.8%), and Planktothrix (31.6%). The cyanobacterial cell density of some North African water systems positively correlated with MCs contents (Mohamed & Bakr, 2018; Mohamed et al., 2015; Nasri et al., 2007). Anabaena affinis, Planktothrix agardhii, Raphidiopsis catemaco, and Capillaria philippinensis were assigned as CYN producers for the first time in Egyptian fishponds (Mohamed & Bakr, 2018). The presence of the MCs synthetase genes (mcyA, -B, -C, -D, -E, and -G) in the Microcystis spp. isolates indicate that these species are responsible for the MCs production in a few North African water ecosystems (El Herry et al., 2008; Fathalli et al., 2011).
West Africa
All of the 26 waterbodies of three surveyed West African nations (Ghana, Nigeria, and Zimbabwe) contained MCs (Table 6). Major congeners of MCs such as MC-LR, MC-LF, MC-RR, and MC-YR were reported (Addico et al., 2017; Chia & Kwaghe, 2015). The WHO's guideline limit of drinking water 1 μg L−1 for MCs concentration was exceeded in 76.9% of the aquatic ecosystems. None of the water bodies’ MCs levels, however, rose above the WHO threshold limits for recreation usage and drinking water for short periods. The Owabi Reservoir in Ghana, which provides drinking water to the Kumasi metropolitan area, had the highest MCs content (8.73 µg L–1) of all the West African waters examined (Addico et al., 2017). The presence of MCs levels exceeding 1 μg L−1 in about ten coastal ecosystems in Nigeria shows that cyanotoxins are a concern that is not just present in inland waters. In Nigeria, Kwaru stream irrigated vegetables and the fish from some fish ponds in Zaria contained MCs values that exceed WHO recommended total daily intake value (0.04 μg kg−1 body weight) (Abdullahi et al., 2022; Chia et al., 2021).
Dolichospermum is the most frequently found (61.5%) cyanobacteria in the surveyed West African waterbodies where cyantoxins were measured. Other cyanobacterial genera found in at least 5 ecosystems examined include Microcystis (57.7%), Oscillatoria (57.7%), Spirulina (26.9%), Chroococcus (26.9%), Marssoniella (26.9%), Planktothrix (23%), Trichodesmium (23.1%), and Merismopedia(19.2%). All of the samples examined from 11 Nigerian coastal waters that contained the mcyE gene of the MC synthetase (mcy) cluster showed the presence of toxic strains of cyanobacteria (Kadiri et al., 2020). Many West African water systems showed a substantial positive association between MCs concentration and cyanobacterial density (Addico et al., 2017; Chia & Kwaghe, 2015; Chia et al., 2021).
Adverse effects of cyanotoxins in Africa
Research on the Vaal Reservoir, South Africa is the earliest known study on animal toxicity caused by algal blooms in Africa (Steyn, 1945). According to Steyn (1945), the algal-infested water was poisonous, and the degree of poisoning depended upon the number of algae consumed. Since then, many scholars have linked the poisoning of humans and animals in Africa to cyanotoxins (Harding et al., 1995; Krienitz et al., 2005; Nasri et al., 2008; Scott et al., 1981; Soll & Williams, 1985; Van Halderen et al., 1995; Zilberg, 1966).
In South Africa, between 1973 and 1974, cattle kills around Hartbeespoort Reservoir was linked to M. aeruginosa toxicity (Scott et al., 1981). In 1979, three white rhinoceroses (Ceratotherium simum) died related to Microcystis toxicity from Klipvoor Reservoir, Barakologadi Game Reserve (Soll & Williams, 1985). Postmortem macroscopic and microscopic pathology revealed acute hepatotoxicity, hepatomegaly with hemorrhage, severe liver necrosis, numerous ecchymoses, and petechiae. In 1994, a dog died after drinking water from Lake Zeekoevlei. Histopathological examination of the liver revealed periacinar fibrosis with duplication of the central veins, degeneration of hepatocytes, and the distention of the bile canuliculi with bile pigment (Van Halderen et al., 1995). The water contained cyanobacterial bloom of Nodularia spumigena (95%) and Microcystis aeruginosa that produced 3479 µg g–1 nodularin.
Oberholster et al. (2009) associated the possible cause of the mortalities of the white rhinoceroses, zebra, wildebeest, and other wild animals in Nhlanganzwane Reservoir, South Africa with acute exposure to MCs toxins. Similarly, the wildlife mortality near four South African reservoirs (Nhlanganzwani, Mpanamana, Makhohlola, and Sunset) in Kruger National Park was also associated with Microcystis blooms, which were confirmed to be toxic using mouse and fish (Clarias gariepinus) bioassays (Masango et al., 2010). Animal (e.g., fish, giraffes, cows, sheep, etc.) poisoning and deaths around some South African waterbodies, including the Vaal Reservoir, Eastern Transvaal Reservoir, Bloemh Reservoir, Klipdrif Reservoir, Theewaterskloof Reservoir, Erfenis Reservoir, and Orange River, were linked to the toxicity of cyanotoxins (Harding, 1997; Harding & Paxton, 2001; Scott, 1991; Theron, 1990a, 1990b; Van Ginkel & Hohls, 1999).
Cyanotoxins (MCs and ATX-a) ingestion was associated with the mass mortality of lesser flamingos at Lake Bogoria, Kenya (Krienitz et al., 2003, 2005). Death of lesser flamingos related to cyanotoxins is also reported in Lakes Big and Manyara, Tanzania (Lugomela et al., 2006). The mortality of freshwater turtles (Emys orbicularis and Mauremys leprosa) was associated with MCs in Lake Oubeira, Algeria (Nasri et al., 2008). Similarly, deaths and illnesses in wild and domestic animals in the Ethiopian Rift Valley lake areas were also linked to cyanotoxins (Willén et al., 2011). Moreover, Zilberg (1966) related gastroenteritis, skin rashes, itching, and sore eyes of children to the production of algal toxins from Lake Mcllwaine, Salisbury (now Harare).
Awareness, efforts of control, and possible solutions
Several efforts have been made to lessen eutrophication in South Africa since the 1970s (Van Ginkel, 2010, 2011). As a result, many eutrophication control strategies were put into practice, including biomanipulation, lowering detergent P levels, predicting cyanobacterial blooms, and disrupting epilimnion by laminar flow (Van Ginkel, 2011). Lake McIlwaine (now Lake Chivero) in Zimbabwe is an example of a successful eutrophication recovery. The long-term research by the Hydrobiology Research Unit of the University of Rhodesia (now the University of Zimbabwe) since 1967 was combined with intensive monitoring efforts by the City of Salisbury. These efforts included diverting nutrients to pasture irrigation schemes, extensive publicity and developing water pollution control legislation and control of the water hyacinth. As a result, the lake was changed to mesotrophy by the end of the 1970s from hypereutrophic condition in 1971 (Thornton, 1982).
Many African countries have legislations that set standards for effluent discharges including the Republic of South Africa Water Act; Zimbabwe Water Act; Kenyan Water Act; Malawian Water Resources Act; Zambian Natural Resources Conservation Act and Water Pollution Control Act (Brown et al., 2020; Thornton & Boddington, 1989). For instance, South Africa limits ammonia release to water resources to less than 6 mg L−1, whereas Zimbabwe limits nitrate, ammonia, and phosphorus concentrations to under 10, 0.5, and 1 mg L−1, respectively (McKendrick, 1982; NWA, 1998; Wiechers & Heynike, 1986). The Ethiopian Environmental Protection Authority has set the concentrations of ammonia, nitrate, and dissolved phosphorus to be less than 5, 20, and 5 mg L–1, respectively, before discharge to inland waters (EEPA, 2011; FDRE, 2002). Additionally, numerous measures for soil and water conservation have been put into place, but it is important to regularly examine how they will affect the improvement of water quality (Reij, 1991).
Different approaches are used worldwide to control cyanotoxin concentration below acceptable drinking and recreational water limits. These approaches prevent bloom occurrence and eradicate cyanobacterial blooms (Merel et al., 2013). To prevent cyanobacterial blooms occurrence, monitoring the quantity of nutrients (C, N, and P), sediment dredging, sediment inactivation (usually by using aluminum sulfate), dosing with aluminum sulfate (to bind P and make it limiting for cyanobacterial growth), biomanipulation of the food web (using microorganisms such as viruses, fungi, bacteria, zooplankton, and algae), accurate prediction of cyanobacterial growth cycles, and mechanical disturbance of the epilimnion are essential (Harding & Paxton, 2001; Merel et al., 2013; Van Ginkel, 2011). On the other hand, eradication of occurring cyanobacterial blooms requires applying an algaecide, usually copper sulfate (Hrudey et al., 1999). However, this technique should be applied with caution since it releases intracellular toxins as a result of cell lysis (Jones & Orr, 1994; Peterson et al., 1995) and induce Cu2+ accumulation on the sediment causing copper toxicity that allows cyanobacterial blooms a few weeks later (Merel et al., 2013).
Regular monitoring of nutrient, cyanobacteria and cyanotoxin load is required in African waterbodies. Waterbody management, watershed management, and land-use planning are necessary to reduce nutrient enrichment of water ecosystems (Chorus & Welker, 2021; WHO, 2016). Controlling human exposure is the only remaining option for management if control measures on these levels are absent or fall short of their goals(Chorus & Welker, 2021). Options for addressing this range from educating users—that is, raising knowledge and guiding personal reactions to bloom situations—to temporarily limiting access to aquatic ecosystems during the bloom (Chorus, 2005; Stauffer et al., 2019).
Contrary to freshwaters, the public is generally well aware about algal toxins for the marine environment (Chorus & Welker, 2021). Even though scientific understanding has been expanding in Africa for decades (Harding & Paxton, 2001), the general public does not seem to be sufficiently informed about cyanotoxins (Harding et al., 2009; Mohamed, 2016a). Therefore, public gatherings, radio broadcasts, warning signs near impacted waterbodies and the dispersal of information booklets must be used to distribute pertinent information.
Conclusions
This meta-analysis review documented occurrences of cyanobacteria and cyanotoxins among African waterbodies. It showed how widely cyanotoxins have spread throughout African aquatic ecosystems and the resulting health dangers to people and animals. MCs were reported in at least 95 African water resources. ATX-a, CYN, NOD, and HTX were quantified in few aquatic ecosystems. Microcystis spp. dominated the East and Southern African water systems with cyanotoxins in water samples, while Oscillatoria spp. and Dolichospermum spp. were most frequently found in North and West African waters. Extremely high concentrations of MCs are reported from African aquatic resources and large concentrations of cyanotoxins have been linked to animal mass mortalities. Most of the water resources examined had MCs concentrations higher than the WHO recommendation of drinking water (1 μg L−1). This indicates a high potential risk to public health when exposed to the toxins during recreational, domestic, and daily activities. Therefore, it is crucial to regularly monitor cyanotoxins in fish, treatment centers, and drinking water sources. Watershed management activities and point-source pollution control are essential for reducing the excessive levels of nutrients loading and eutrophication into the African waterbodies. Furthermore, the standards set in relation to nutrients’ concentration for African waterbodies should be revised to adopt the international standards.
Availability of data and materials
Not applicable.
References
Abdullahi, H., Tanimu, Y., Akinyemi, S. A., do Carmo Bittencourt-Oliveira, M., & Chia, M. A. (2022). Assessment of microcystins in surface water and irrigated vegetables in Kwaru stream, Hayin Danmani, Kaduna-Nigeria. Environmental Science and Pollution Research. https://doi.org/10.1007/s11356-022-21381-w
Addico, G. N. D., Hardege, J. D., Kohoutek, J., deGraft-Johnson, K. A. A., & Babica, P. (2017). Cyanobacteria and microcystin contamination in untreated and treated drinking water in Ghana. Advances in Oceanography and Limnology, 8(1), 92–106. https://doi.org/10.4081/aiol.2017.6323
Akale, A. T., Moges, M. A., Dagnew, D. C., Tilahun, S. A., & Steenhuis, T. S. (2018). Assessment of nitrate in wells and springs in the north central Ethiopian highlands. Water, 10(4), 476.
Ali, K., Abiye, T., & Adam, E. (2022). Integrating in situ and current generation satellite data for temporal and spatial analysis of harmful algal blooms in the Hartbeespoort Dam, Crocodile River Basin, South Africa. Remote Sensing, 14(17), 4277.
Amrani, A., Nasri, H., Azzouz, A., Kadi, Y., & Bouaïcha, N. (2014). Variation in cyanobacterial hepatotoxin (microcystin) content of water samples and two species of fishes collected from a shallow lake in Algeria. Archives of Environmental Contamination and Toxicology, 66(3), 379–389.
Ballot, A., Krienitz, L., Kotut, K., Wiegand, C., Metcalf, J. S., Codd, G. A., & Pflugmacher, S. (2004). Cyanobacteria and cyanobacterial toxins in three alkaline Rift Valley lakes of Kenya—Lakes Bogoria, Nakuru and Elmenteita. Journal of Plankton Research, 26(8), 925–935.
Ballot, A., Krienitz, L., Kotut, K., Wiegand, C., & Pflugmacher, S. (2005). Cyanobacteria and cyanobacterial toxins in the alkaline crater lakes Sonachi and Simbi, Kenya. Harmful Algae, 4(1), 139–150.
Ballot, A., Pflugmacher, S., Wiegand, C., Kotut, K., & Krienitz, L. (2003). Cyanobacterial toxins in lake Baringo, Kenya. Limnologica, 33(1), 2–9.
Ballot, A., Sandvik, M., Rundberget, T., Botha, C. J., & Miles, C. O. (2013). Diversity of cyanobacteria and cyanotoxins in Hartbeespoort Dam, South Africa. Marine and Freshwater Research, 65(2), 175–189.
Boufligha, K., et al. (2021). Dynamic of microcystin-LR-producing cyanobacteria in a drinking water supply: Guenitra dam (North-East of Algeria). Egyptian Journal of Aquatic Biology and Fisheries, 25(6), 377–395.
Bouhaddada, R., Nélieu, S., Nasri, H., Delarue, G., & Bouaïcha, N. (2016). High diversity of microcystins in a microcystis bloom from an Algerian lake. Environmental Pollution, 216, 836–844.
Brown, C., Campher, D., & King, J. (2020). Status and trends in EFlows in southern Africa. Natural Resources Forum, 44, 66–88.
Buratti, F. M., Manganelli, M., Vichi, S., Stefanelli, M., Scardala, S., Testai, E., & Funari, E. (2017). Cyanotoxins: Producing organisms, occurrence, toxicity, mechanism of action and human health toxicological risk evaluation. Archives of Toxicology, 91(3), 1049–1130.
Carmichael, W. W., Azevedo, S., An, J. S., Molica, R., Jochimsen, E. M., Lau, S., Rinehart, K. L., Shaw, G. R., & Eaglesham, G. K. (2001). Human fatalities from cyanobacteria: Chemical and biological evidence for cyanotoxins. Environmental Health Perspectives, 109(7), 663–668.
Chellappa, N. T., Chellappa, S. L., & Chellappa, S. (2008). Harmful phytoplankton blooms and fish mortality in a eutrophicated reservoir of Northeast Brazil. Brazilian Archives of Biology and Technology, 51(4), 633–641.
Chia, A., Abolude, D., Ladan, Z., Akanbi, O., & Kalaboms, A. (2009). The presence of microcystins in aquatic ecosystems in Northern Nigeria: Zaria as a case study. Research Journal of Environmental Toxicology, 3(4), 170–178.
Chia, M. A., Abdulwahab, R., Ameh, I., Balogun, J. K., & Auta, J. (2021). Farmed tilapia as an exposure route to microcystins in Zaria-Nigeria: A seasonal investigation. Environmental Pollution, 271, 116366.
Chia, M. A., & Kwaghe, M. J. (2015). Microcystins contamination of surface water supply sources in Zaria-Nigeria. Environmental Monitoring and Assessment, 187(10), 1–12.
Chorus, I. (2005). Water safety plans. A better regulatory approach to prevent human exposure to harmful cyanobacteria. In P. M. V. Jefhuisman & H. C. P. Matthijs (Eds.), Harmful cyanobacteria (Vol. 3, p. 249). Springer.
Chorus, I., Fastner, J., & Welker, M. (2021). Cyanobacteria and cyanotoxins in a changing environment: Concepts, controversies, challenges. Water, 13(18), 2463.
Chorus, I., & Welker, M. (2021). Toxic cyanobacteria in water: A guide to their public health consequences, monitoring and management. Taylor & Francis.
Conradie, K. R., & Barnard, S. (2012). The dynamics of toxic Microcystis strains and microcystin production in two hypertrofic South African reservoirs. Harmful Algae, 20, 1–10.
Dawson, R. (1998). The toxicology of microcystins. Toxicon, 36(7), 953–962.
Dietrich, D., & Hoeger, S. (2005). Guidance values for microcystins in water and cyanobacterial supplement products (blue-green algal supplements): A reasonable or misguided approach? Toxicology and Applied Pharmacology, 203(3), 273–289.
Dolman, A. M., Rücker, J., Pick, F. R., Fastner, J., Rohrlack, T., Mischke, U., & Wiedner, C. (2012). Cyanobacteria and cyanotoxins: The influence of nitrogen versus phosphorus. PLoS ONE, 7(6), e38757. https://doi.org/10.1371/journal.pone.0038757
Douma, M., Ouahid, Y., Del Campo, F., Loudiki, M., Mouhri, K., & Oudra, B. (2010). Identification and quantification of cyanobacterial toxins (microcystins) in two Moroccan drinking-water reservoirs (Mansour Eddahbi, Almassira). Environmental Monitoring and Assessment, 160(1), 439–450.
EEPA. (2011). Environmental standards for industrial pollution control in Ethiopia. Ethiopian Environmental Protection Authority Retrieved September 1 from https://plasticsdb.surrey.ac.uk/documents/Ethiopia/Republic%20of%20Ethiopia%20%282011%29%20Environmental%20standards%20for%20industrial%20pollution%20control%20in%20Ethiopia.pdf
Eguzozie, K., Mavumengwana, V., Nkosi, D., Kayitesi, E., & Nnabuo-Eguzozie, E. C. (2016). Bioaccumulation and quantitative variations of microcystins in the Swartspruit River, South Africa. Archives of Environmental Contamination and Toxicology, 71(2), 286–296. https://doi.org/10.1007/s00244-016-0269-5
El Herry, S., Bouaïcha, N., Jenhani-Ben Rejeb, A., & Romdhane, M. (2007). First observation of microcystins in Tunisian inland waters: A threat to river mouths and lagoon ecosystems. Transitional Waters Bulletin, 1(2), 73–82.
El Herry, S., Fathalli, A., Rejeb, A.J.-B., & Bouaïcha, N. (2008). Seasonal occurrence and toxicity of Microcystis spp. and Oscillatoria tenuis in the Lebna Dam, Tunisia. Water Research, 42(4–5), 1263–1273.
El Khalloufi, F., El Ghazali, I., Saqrane, S., Oufdou, K., Vasconcelos, V., & Oudra, B. (2012). Phytotoxic effects of a natural bloom extract containing microcystins on Lycopersicon esculentum. Ecotoxicology and Environmental Safety, 79, 199–205.
Eynard, F., Mez, K., & Walther, J.-L. (2000). Risk of cyanobacterial toxins in Riga waters (Latvia). Water Research, 34(11), 2979–2988.
Fathalli, A., Jenhani, A. B. R., Moreira, C., Welker, M., Romdhane, M., Antunes, A., & Vasconcelos, V. (2011). Molecular and phylogenetic characterization of potentially toxic cyanobacteria in Tunisian freshwaters. Systematic and Applied Microbiology, 34(4), 303–310.
FDRE. (2002). Environmental pollution control proclamation No. 300/2002. Federal Democratic Republic of Ethiopia (FDRE). Retrieved September 1 from https://urbanlex.unhabitat.org/sites/default/files/faolex//eth44282.pdf
Fetahi, T. (2019). Eutrophication of Ethiopian water bodies: A serious threat to water quality, biodiversity and public health. African Journal of Aquatic Science, 44(4), 303–312.
Fristachi, A., Sinclair, J. L., Hall, S., Berkman, J. A. H., Boyer, G., Burkholder, J., Burns, J., Carmichael, W., DuFour, A., & Frazier, W. (2008). Occurrence of cyanobacterial harmful algal blooms workgroup report. In H. K. Hudnell (Ed.), Cyanobacterial harmful algal blooms: State of the science and research needs (pp. 45–103). Springer.
Funari, E., & Testai, E. (2008). Human health risk assessment related to cyanotoxins exposure. Critical Reviews in Toxicology, 38(2), 97–125.
Giri, S. (2021). Water quality prospective in Twenty First Century: Status of water quality in major river basins, contemporary strategies and impediments: A review. Environmental Pollution, 271, 116332.
Gupta, N., Pant, S., Vijayaraghavan, R., & Rao, P. L. (2003). Comparative toxicity evaluation of cyanobacterial cyclic peptide toxin microcystin variants (LR, RR, YR) in mice. Toxicology, 188(2–3), 285–296.
Habtemariam, H., Kifle, D., Leta, S., Beekman, W., & Lürling, M. (2021). Cyanotoxins in drinking water supply reservoir (Legedadi, Central Ethiopia): Implications for public health safety. SN Applied Sciences, 3(3), 1–10.
Harding, W. R., & Paxton, B. R. (2001). Cyanobacteria in South Africa: A review. Water Research Commission Pretoria.
Harding, W. (1997). New regional emergence of cyanobacterial toxicosis. Harmful Algae News, 16, 12–13.
Harding, W., Rowe, N., Wessels, J., Beattie, K., & Codd, G. (1995). Death of a dog attributed to the cyanobacterial (blue-green algal) hepatotoxin nodularin in South Africa. Journal of the South African Veterinary Association, 66(4), 256–259.
Harding, W. R., Downing, T. G., Van Ginkel, C. E., & Moolman, A. P. (2009). An overview of cyanobacterial research and management in South Africa post-2000. Water SA, 35(4), 479–484.
Harke, M. J., Steffen, M. M., Gobler, C. J., Otten, T. G., Wilhelm, S. W., Wood, S. A., & Paerl, H. W. (2016). A review of the global ecology, genomics, and biogeography of the toxic cyanobacterium, Microcystis spp. Harmful Algae, 54, 4–20.
Hooser, S. B. (2000). Fulminant hepatocyte apoptosis in vivo following microcystin-LR administration to rats. Toxicologic Pathology, 28(5), 726–733.
Hrudey, M. B., Drikas, M., & Gregory, R. (1999). Remedial Measures. In I. Chorus & J. Bartram (Eds.), Toxic cyanobacteria in water: A guide to their public health consequences, monitoring and management. E & FN Spon.
Ibelings, B. W., Kurmayer, R., Azevedo, S. M., Wood, S. A., Chorus, I., & Welker, M. (2021). Understanding the occurrence of cyanobacteria and cyanotoxins. In I. Chorus & M. Welker (Eds.), Toxic cyanobacteria in water (pp. 213–294). CRC Press.
Jenhani, A. B., Bouaïcha, N., El Herry, S., Fathalli, A., Zekri, I., Zekri, S. H., Limam, A., Alouini, S., & Romdhane, M. S. (2006). Cyanobacteria and their toxic potential in dam water content in Northern Tunisia. Archives De L’institut Pasteur De Tunis, 83(1–4), 71–81.
Jones, G. J., & Orr, P. T. (1994). Release and degradation of microcystin following algicide treatment of a Microcystis aeruginosa bloom in a recreational lake, as determined by HPLC and protein phosphatase inhibition assay. Water Research, 28(4), 871–876.
Juma, D. W., Wang, H., & Li, F. (2014). Impacts of population growth and economic development on water quality of a lake: Case study of Lake Victoria Kenya water. Environmental Science and Pollution Research, 21(8), 5737–5746.
Kadiri, M. O., Isagba, S., Ogbebor, J. U., Omoruyi, O. A., Unusiotame-Owolagba, T. E., Lorenzi, A. S., Bittencourt-Oliveira, M. C., & Chia, M. A. (2020). The presence of microcystins in the coastal waters of Nigeria, from the Bights of Bonny and Benin, Gulf of Guinea. Environmental Science and Pollution Research, 27(28), 35284–35293.
Kaggwa, M., Straubinger-Gansberger, N., & Schagerl, M. (2018). Cyanotoxins in small artificial dams in Kenya utilised for cage fish farming–a threat to local people? African Journal of Aquatic Science, 43(2), 123–129.
Kotut, K., Ballot, A., & Krienitz, L. (2006). Toxic cyanobacteria and their toxins in standing waters of Kenya: Implications for water resource use. Journal of Water and Health, 4(2), 233–245.
Kotut, K., Ballot, A., Wiegand, C., & Krienitz, L. (2010). Toxic cyanobacteria at Nakuru sewage oxidation ponds—A potential threat to wildlife. Limnologica, 40(1), 47–53.
Krienitz, L., Ballot, A., Wiegand, C., Kotut, K., Codd, G. A., & Pflugmacher, S. (2002). Cyanotoxin-producing bloom of Anabaena flos-aquae, Anabaena discoidea and Microcystis aeruginosa (Cyanobacteria) in Nyanza Gulf of Lake Victoria, Kenya.
Krienitz, L., Ballot, A., Casper, P., Codd, G., Kotut, K., Metcalf, J., Morrison, L., Pflugmacher, S., & Wiegand, C. (2005). Contribution of toxic cyanobacteria to massive deaths of Lesser Flamingos at saline-alkaline lakes of Kenya. Internationale Vereinigung Für Theoretische Und Angewandte Limnologie: Verhandlungen, 29(2), 783–786.
Krienitz, L., Ballot, A., Kotut, K., Wiegand, C., Pütz, S., Metcalf, J. S., Codd, G. A., & Stephan, P. (2003). Contribution of hot spring cyanobacteria to the mysterious deaths of Lesser Flamingos at Lake Bogoria, Kenya. FEMS Microbiology Ecology, 43(2), 141–148.
Krienitz, L., Dadheech, P. K., Fastner, J., & Kotut, K. (2013). The rise of potentially toxin producing cyanobacteria in Lake Naivasha, Great African Rift Valley, Kenya. Harmful Algae, 27, 42–51.
Kuiper-Goodman, T., Falconer, I., & Fitzgerald, J. (1999). Toxic cyanobacteria in water: A guide to their public health consequences, monitoring and management. In I. Chorus & J. Bartram (Eds.), Human health aspects. E & FN Spon.
Lanckriet, S., Nyssen, J., & Adgo, E. (2017). Environmental change in lake catchments of Ethiopia. In Wiley Online Library (Vol. 28, pp. 1793–1794).
Lijklema, L. (1995). Development and eutrophication: Experiences and perspectives. Water Science and Technology, 31(9), 11–15.
Ling, B. (2000). Health impairments arising from drinking water polluted with domestic sewage and excreta in China. Schriftenreihe Des Vereins Fur Wasser-, Boden-Und Lufthygiene, 105, 43–46.
Loftin, K. A., Graham, J. L., Hilborn, E. D., Lehmann, S. C., Meyer, M. T., Dietze, J. E., & Griffith, C. B. (2016). Cyanotoxins in inland lakes of the United States: Occurrence and potential recreational health risks in the EPA National Lakes Assessment 2007. Harmful Algae, 56, 77–90.
Lugomela, C., Pratap, H. B., & Mgaya, Y. D. (2006). Cyanobacteria blooms—A possible cause of mass mortality of Lesser Flamingos in Lake Manyara and Lake Big Momela. Tanzania. Harmful Algae, 5(5), 534–541.
Major, Y., Kifle, D., Spoof, L., & Meriluoto, J. (2018). Cyanobacteria and microcystins in Koka reservoir (Ethiopia). Environmental Science and Pollution Research, 25(27), 26861–26873.
Mankiewicz-Boczek, J., Gągała, I., Jurczak, T., Urbaniak, M., Negussie, Y. Z., & Zalewski, M. (2015). Incidence of microcystin-producing cyanobacteria in Lake Tana, the largest waterbody in Ethiopia. African Journal of Ecology, 53(1), 54–63.
Manning, S. R., & Nobles, D. R. (2017). Impact of global warming on water toxicity: Cyanotoxins. Current Opinion in Food Science, 18, 14–20.
Masango, M. G., Myburgh, J. G., Labuschagne, L., Govender, D., Bengis, R. G., & Naicker, D. (2010). Assessment of microcystis bloom toxicity associated with wildlife mortality in the Kruger National Park, South Africa. Journal of Wildlife Diseases, 46(1), 95–102. https://doi.org/10.7589/0090-3558-46.1.95
Mbukwa, E. A., Msagati, T. A., & Mamba, B. B. (2012). Quantitative variations of intracellular microcystin-LR,-RR and-YR in samples collected from four locations in Hartbeespoort Dam in North West Province (South Africa) during the 2010/2011 summer season. International Journal of Environmental Research and Public Health, 9(10), 3484–3505.
Mchau, G. J., Machunda, R., Kimanya, M., Makule, E., Gong, Y. Y., Mpolya, E., Meneely, J. P., Elliott, C. T., & Greer, B. (2021). First Report of the co-occurrence of cylindrospermopsin, nodularin and microcystins in the freshwaters of Lake Victoria, Tanzania. Exposure and Health, 13(2), 185–194.
McKendrick, J. (1982). Water supply and sewage treatment in relation to water quality in Lake McIlwaine. In J. A. Thornton & W. K. Nduku (Eds.), Lake McIlwaine (pp. 202–217). Springer.
Merel, S., Walker, D., Chicana, R., Snyder, S., Baurès, E., & Thomas, O. (2013). State of knowledge and concerns on cyanobacterial blooms and cyanotoxins. Environment International, 59, 303–327.
Metcalf, J. S., & Codd, G. A. (2012). Cyanotoxins. In B. A. Whitton (Ed.), Ecology of cyanobacteria II (pp. 651–675). Springer.
Mhlanga, L., Day, J., Cronberg, G., Chimbari, M., Siziba, N., & Annadotter, H. (2006). Cyanobacteria and cyanotoxins in the source water from Lake Chivero, Harare, Zimbabwe, and the presence of cyanotoxins in drinking water. African Journal of Aquatic Science, 31(2), 165–173.
Mohamed, Z., Ahmed, Z., Bakr, A., Hashem, M., & Alamri, S. (2020). Detection of free and bound microcystins in tilapia fish from Egyptian fishpond farms and its related public health risk assessment. Journal of Consumer Protection and Food Safety, 15(1), 37–47.
Mohamed, Z., & Carmichael, W. (2000). Seasonal variation in microcystin levels of river Nile water at Sohag City, Egypt. Annales De Limnologie-International Journal of Limnology, 36, 227–234.
Mohamed, Z. A. (2016a). Breakthrough of Oscillatoria limnetica and microcystin toxins into drinking water treatment plants-examples from the Nile River, Egypt. Water SA, 42(1), 161–165.
Mohamed, Z. A. (2016b). Harmful cyanobacteria and their cyanotoxins in Egyptian fresh waters—State of knowledge and research needs. African Journal of Aquatic Science, 41(4), 361–368. https://doi.org/10.2989/16085914.2016.1219313
Mohamed, Z. A., & Bakr, A. (2018). Concentrations of cylindrospermopsin toxin in water and tilapia fish of tropical fishponds in Egypt, and assessing their potential risk to human health. Environmental Science and Pollution Research, 25(36), 36287–36297.
Mohamed, Z. A., Carmichael, W. W., & Hussein, A. A. (2003). Estimation of microcystins in the freshwater fish Oreochromis niloticus in an Egyptian fish farm containing a Microcystis bloom. Environmental Toxicology: An International Journal, 18(2), 137–141. https://doi.org/10.1002/tox.10111
Mohamed, Z. A., Deyab, M. A., Abou-Dobara, M. I., El-Sayed, A. K., & El-Raghi, W. M. (2015). Occurrence of cyanobacteria and microcystin toxins in raw and treated waters of the Nile River, Egypt: Implication for water treatment and human health. Environmental Science and Pollution Research, 22(15), 11716–11727.
Mokoena, M. M., & Mukhola, M. S. (2019). Current effects of cyanobacteria toxin in water sources and containers in the Hartbeespoort Dam Area, South Africa. International Journal of Environmental Research and Public Health, 16(22), 4468.
Mwaura, F., Koyo, A. O., & Zech, B. (2004). Cyanobacterial blooms and the presence of cyanotoxins in small high altitude tropical headwater reservoirs in Kenya. Journal of Water and Health, 2(1), 49–57.
Nasri, A.-B., Bouaïcha, N., & Fastner, J. (2004). First report of a microcystin-containing bloom of the cyanobacteria Microcystis spp. in Lake Oubeira, Eastern Algeria. Archives of Environmental Contamination and Toxicology, 46(2), 197–202.
Nasri, H., Bouaïcha, N., & Harche, M. K. (2007). A new morphospecies of Microcystis sp. forming bloom in the Cheffia dam (Algeria): Seasonal variation of microcystin concentrations in raw water and their removal in a full-scale treatment plant. Environmental Toxicology: an International Journal, 22(4), 347–356.
Nasri, H., El Herry, S., & Bouaïcha, N. (2008). First reported case of turtle deaths during a toxic Microcystis spp. bloom in Lake Oubeira Algeria. Ecotoxicology and Environmental Safety, 71(2), 535–544.
Nchabeleng, T., Cheng, P., Oberholster, P. J., Botha, A., Smit, W. J., & Luus-Powell, W. (2014). Microcystin-LR equivalent concentrations in fish tissue during a postbloom Microcystis exposure in Loskop Dam, South Africa. African Journal of Aquatic Science, 39(4), 459–466.
Ndlela, L. L., Oberholster, P. J., Van Wyk, J., & Cheng, P.-H. (2016). An overview of cyanobacterial bloom occurrences and research in Africa over the last decade. Harmful Algae, 60, 11–26.
NWA. (1998). Revision of general authorisations in terms of Section 39 of the National Water Act, 1998 (act no. 36 of 1998). University of Pretoria.
Nyenje, P., Foppen, J., Uhlenbrook, S., Kulabako, R., & Muwanga, A. (2010). Eutrophication and nutrient release in urban areas of sub-Saharan Africa—A review. Science of the Total Environment, 408(3), 447–455. https://doi.org/10.1016/j.scitotenv.2009.10.020
Oberholster, P. J., Myburgh, J. G., Govender, D., Bengis, R., & Botha, A.-M. (2009). Identification of toxigenic Microcystis strains after incidents of wild animal mortalities in the Kruger National Park, South Africa. Ecotoxicology and Environmental Safety, 72(4), 1177–1182.
Okello, W., Ostermaier, V., Portmann, C., Gademann, K., & Kurmayer, R. (2010). Spatial isolation favours the divergence in microcystin net production by Microcystis in Ugandan freshwater lakes. Water Research, 44(9), 2803–2814.
Olokotum, M., Humbert, J.-F., Quiblier, C., Okello, W., Semyalo, R., Troussellier, M., Marie, B., Baumann, K., Kurmayer, R., & Bernard, C. (2022). Characterization of potential threats from cyanobacterial toxins in Lake Victoria embayments and during water treatment. Toxins, 14(10), 664.
Oudra, B., Loudiki, M., Sbiyyaa, B., Martins, R., Vasconcelos, V., & Namikoshi, N. (2001). Isolation, characterization and quantification of microcystins (heptapeptides hepatotoxins) in Microcystis aeruginosa dominated bloom of Lalla Takerkoust lake–reservoir (Morocco). Toxicon, 39(9), 1375–1381.
Oudra, B., Loudiki, M., Vasconcelos, V., Sabour, B., Sbiyyaa, B., Oufdou, K., & Mezrioui, N. (2002). Detection and quantification of microcystins from cyanobacteria strains isolated from reservoirs and ponds in Morocco. Environmental Toxicology: An International Journal, 17(1), 32–39.
Paerl, H. W. (2014). Mitigating harmful cyanobacterial blooms in a human-and climatically-impacted world. Life, 4(4), 988–1012.
Pedro, O., Correia, D., Lie, E., Skåre, J. U., Leão, J., Neves, L., Sandvik, M., & Berdal, K. G. (2011). Polymerase chain reaction (PCR) detection of the predominant microcystin-producing genotype of cyanobacteria in Mozambican lakes. African Journal of Biotechnology, 10(83), 19299–19308.
Pedro, O., Rundberget, T., Lie, E., Correia, D., Skaare, J. U., Berdal, K. G., Neves, L., & Sandvik, M. (2012). Occurrence of microcystins in freshwater bodies in Southern Mozambique. Journal of Research in Environmental Science and Toxicology, 1, 58–65.
Pelaez, M., Antoniou, M. G., He, X., Dionysiou, D. D., Cruz, A. A., Tsimeli, K., Triantis, T., Hiskia, A., Kaloudis, T., & Williams, C. (2010). Sources and occurrence of cyanotoxins worldwide. In D. Fatta-Kassinos, K. Bester, & K. Kümmerer (Eds.), Xenobiotics in the urban water cycle (pp. 101–127). Springer.
Peterson, H. G., Hrudey, S. E., Cantin, I. A., Perley, T. R., & Kenefick, S. L. (1995). Physiological toxicity, cell membrane damage and the release of dissolved organic carbon and geosmin by Aphanizomenon flos-aquae after exposure to water treatment chemicals. Water Research, 29(6), 1515–1523.
Raffoul, M., Enanga, E., Senar, O. E., Creed, I., & Trick, C. (2020). Assessing the potential health risk of cyanobacteria and cyanotoxins in Lake Naivasha, Kenya. Hydrobiologia, 847(4), 1041–1056.
Recknagel, F., Orr, P. T., Bartkow, M., Swanepoel, A., & Cao, H. (2017). Early warning of limit-exceeding concentrations of cyanobacteria and cyanotoxins in drinking water reservoirs by inferential modelling. Harmful Algae, 69, 18–27.
Reij, C. (1991). Indigenous soil and water conservation in Africa. Sustainable Agriculture Programme of the International Institute for Environment and Development.
Roegner, A., Sitoki, L., Weirich, C., Corman, J., Owage, D., Umami, M., Odada, E., Miruka, J., Ogari, Z., & Smith, W. (2020). Harmful algal blooms threaten the health of peri-urban fisher communities: A case study in Kisumu Bay, Lake Victoria, Kenya. Exposure and Health, 12(4), 835–848.
Salameh, J.-P., Bossuyt, P. M., McGrath, T. A., Thombs, B. D., Hyde, C. J., Macaskill, P., Deeks, J. J., Leeflang, M., Korevaar, D. A., Whiting, P., Takwoingi, Y., Reitsma, J. B., Cohen, J. F., Frank, R. A., Hunt, H. A., Hooft, L., Rutjes, A. W. S., Willis, B. H., Gatsonis, C., et al. (2020). Preferred reporting items for systematic review and meta-analysis of diagnostic test accuracy studies (PRISMA-DTA): Explanation, elaboration, and checklist. BMJ, 370, m2632. https://doi.org/10.1136/bmj.m2632
Sasner, J. J., Ikawa, M., Foxall, T. L., & Watson, W. H. (1981). Studies on aphantoxin from aphanizomenon flos-aquae in New Hampshire. In W. W. Carmichael (Ed.), The water environment: Algal toxins and health (pp. 389–403). Boston: Springer. https://doi.org/10.1007/978-1-4613-3267-1_27
Scott, L. L., Downing, S., & Downing, T. (2018). Potential for dietary exposure to β-N-methylamino-l-alanine and microcystin from a freshwater system. Toxicon, 150, 261–266.
Scott, W. (1991). Occurrence and significance of toxic cyanobacteria in Southern Africa. Water Science and Technology, 23(1–3), 175–180.
Scott, W. E., Barlow, D. J., & Hauman, J. H. (1981). Studies on the ecology, growth and physiology of toxic Microcystis aeruginosa in South Africa. In W. W. Carmichael (Ed.), The water environment: Algal toxins and health (pp. 49–69). Springer.
Semyalo, R., Rohrlack, T., Naggawa, C., & Nyakairu, G. W. (2010). Microcystin concentrations in Nile tilapia (Oreochromis niloticus) caught from Murchison Bay, Lake Victoria and Lake Mburo: Uganda. Hydrobiologia, 638(1), 235–244.
Shaw, G. R., McKenzie, R. A., Wickramasinghe, W. A., Seawright, A. A., Eaglesham, G. K., & Moore, M. R. (2002). Comparative toxicity of the cyanobacterial toxin cylindrospermopsin between mice and cattle: human implications. In K. A. Steidinger, J. H. Landsberg, C. R. Tomas, & G. A. Vargo (Eds.), Harmful algae (pp. 465–467). St. Petersburg: Florida Fish and Wildlife Conservation Commission, Florida Institute of Oceanography, and Intergovernmental Oceanographic Commission of UNESCO.
Simiyu, B. M., Oduor, S. O., Rohrlack, T., Sitoki, L., & Kurmayer, R. (2018). Microcystin content in phytoplankton and in small fish from eutrophic Nyanza Gulf, Lake Victoria, Kenya. Toxins, 10(7), 275.
Singo, A., Myburgh, J. G., Laver, P. N., Venter, E. A., Ferreira, G. C., Rösemann, G. M., & Botha, C. J. (2017). Vertical transmission of microcystins to Nile crocodile (Crocodylus niloticus) eggs. Toxicon, 134, 50–56.
Soll, M., & Williams, M. (1985). Mortality of a white rhinoceros (Ceratotherium simum) suspected to be associated with the blue-green alga Microcystis aeruginosa. Journal of the South African Veterinary Association, 56(1), 49–51.
Stauffer, B. A., Bowers, H. A., Buckley, E., Davis, T. W., Johengen, T. H., Kudela, R., McManus, M. A., Purcell, H., Smith, G. J., & Vander Woude, A. (2019). Considerations in harmful algal bloom research and monitoring: Perspectives from a consensus-building workshop and technology testing. Frontiers in Marine Science, 6, 399.
Steyn, D. (1943). Poisoning of animals by algae on dams and pans. Farming in South Africa, 18, 489–492.
Steyn, D. G. (1945). Poisoning of animals and human beings by algae. South African Journal of Science, 41, 243–244.
Straubinger-Gansberger, N., Kaggwa, M. N., & Schagerl, M. (2014). Phytoplankton patterns along a series of small man-made reservoirs in Kenya. Environmental Monitoring and Assessment, 186(8), 5153–5166.
Svirčev, Z., Lalić, D., Bojadžija Savić, G., Tokodi, N., Drobac Backović, D., Chen, L., Meriluoto, J., & Codd, G. A. (2019). Global geographical and historical overview of cyanotoxin distribution and cyanobacterial poisonings. Archives of Toxicology, 93(9), 2429–2481.
Szlag, D. C., Sinclair, J. L., Southwell, B., & Westrick, J. A. (2015). Cyanobacteria and cyanotoxins occurrence and removal from five high-risk conventional treatment drinking water plants. Toxins, 7(6), 2198–2220.
Tanvir, R. U., Hu, Z., Zhang, Y., & Lu, J. (2021). Cyanobacterial community succession and associated cyanotoxin production in hypereutrophic and eutrophic freshwaters. Environmental Pollution, 290, 118056. https://doi.org/10.1016/j.envpol.2021.118056
Theron, C. P. (1990a). Kameelperdvrektes en die teenwoordigheid van Microcystis in Bloemhof Dam. DWAF-HRI Report N, 4.
Theron, C. P. (1990b). Ondersoek na die voorkoms van Microcystis in Klipdrif Dam.
Thornton, J. A. N. W. K. (1982). Lake McIlwaine : the eutrophication and recovery of a tropical African man-made lake. Dr. W. Junk ; Distributors for the U.S. and Canada, Kluwer Boston.
Thornton, J., & Boddington, G. (1989). A new look at the old problem of eutrophication management in Southern Africa. The Environmentalist, 9(2), 121–129.
Tibebe, D., Zewge, F., Lemma, B., & Kassa, Y. (2022). Assessment of spatio-temporal variations of selected water quality parameters of Lake Ziway, Ethiopia using multivariate techniques. BMC Chemistry, 16(1), 1–18.
Tilahun, S., Kifle, D., Tigist, W. Z., Johansen, J. A., Taye, B. D., & Hansen, J. H. (2019). Temporal dynamics of intra-and extra-cellular microcystins concentrations in Koka reservoir (Ethiopia): Implications for public health risk. Toxicon, 168, 83–92.
Van Ginkel, C. (2010). Investigating the applicability of ecological informatics modelling techniques for predicting harmfull algal blooms in hypertrophic reservoirs of South Africa (WRC Report No. TT 451/10, Issue. W. R. Commission).
Van Ginkel, C. (2011). Eutrophication: Present reality and future challenges for South Africa. Water SA, 37(5), 693–702.
Van Ginkel, C., & Hohls, B. (1999). Toxic algae in Erfenis and Allemanskraal dams. Occasional report by the Institute for Quality Studies, Department of Water Affairs and Forestry.
Van Halderen, A., Harding, W., Wessels, J. C., Schneider, D., Heine, E., Van der Merwe, J., & Fourie, J. (1995). Cyanobacterial (blue-green algae) poisoning of livestock in the western Cape Province of South Africa. Journal of the South African Veterinary Association, 66(4), 260–264.
Vidal, F., Sedan, D., D’Agostino, D., Cavalieri, M. L., Mullen, E., Parot Varela, M. M., Flores, C., Caixach, J., & Andrinolo, D. (2017). Recreational exposure during algal bloom in Carrasco Beach, Uruguay: A liver failure case report. Toxins, 9(9), 267.
WHO. (2016). Protecting surface water for health: Identifying, assessing and managing drinking-water quality risks in surface-water catchments. WHO.
WHO. (2020). Cyanobacterial toxins: Anatoxin-a and analogues. WHO.
Wicks, R. J., & Thiel, P. G. (1990). Environmental factors affecting the production of peptide toxins in floating scums of the cyanobacterium Microcystis aeruginosa in a hypertrophic African reservoir. Environmental Science & Technology, 24(9), 1413–1418.
Wiechers, H., & Heynike, J. (1986). Sources of phosphorus which give rise to eutrophication in South African waters. Water SA, 12(2), 99–102.
Willén, E., Ahlgren, G., Tilahun, G., Spoof, L., Neffling, M.-R., & Meriluoto, J. (2011). Cyanotoxin production in seven Ethiopian Rift Valley lakes. Inland Waters, 1(2), 81–91.
Zewde, T., Kifle, D., Johansen, J., Demissie, T., Hansen, J., & Tadesse, Z. (2020). Cyanobacterial abundance and microcystins in water, seston and fish tissues in Lake Hora-Arsedi (Ethiopia). African Journal of Aquatic Science, 45(4), 475–485.
Zilberg, B. (1966). Gastroenteritis in Salisbury European children—A five-year study. Central African Journal of Medicine, 12(9), 164–168.
Zinabu, E., Kelderman, P., Van Der Kwast, J., & Irvine, K. (2018). Evaluating the effect of diffuse and point source nutrient transfers on water quality in the Kombolcha River Basin, an industrializing Ethiopian catchment. Land Degradation & Development, 29(10), 3366–3378.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the paper conception and design. Material preparation, and data collection and analysis were performed by TM, TF, FE, and AM. The first draft of the manuscript was written by TM, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
All authors want to publish the research paper entitled “Cyanotoxins in African Water Bodies and its Adverse Effects. A Review.” in Environmental Geochemistry and Health journal.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Muluye, T., Fetahi, T., Engdaw, F. et al. Cyanotoxins in African waterbodies: occurrence, adverse effects, and potential risk to animal and human health. Environ Geochem Health 45, 7519–7542 (2023). https://doi.org/10.1007/s10653-023-01724-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-023-01724-3