Abstract
A study was conducted to investigate the occurrence and behavior of six endocrine-disrupting chemicals (EDCs) in sewage, river water, and seawater from the Pearl River Delta (PRD). The six EDCs under study were 4-nonylphenol (NP), bisphenol A (BPA), 17α-ethynylestradiol (EE2), estrone (E2), 17β-estradiol (E2), and estriol (E3). These EDCs, predominated by BPA, were found in high levels in the influents and the effluents of sewage treatment plants in the area. The relatively high concentrations (0.23–625 ng/L) of the EDCs detected in the receiving river water suggested that the untreated sewage discharge was a major contributor. The EDCs detected in eight outlets of the Pear River and the Pear River Estuary were in the ranges of 1.2–234 and 0.2–178 ng/L, respectively. The estrogen equivalents in the aquatic environments under study ranged from 0.08 to 4.5 ng/L, with E1 and EE2 being the two predominant contributors. As the fluxes of the EDCs from the PRD region to the nearby ocean are over 500 tons each year, the results of this study point to the potential that Pearl River is a significant source of the EDCs to the local environment there.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In recent years, exposure to endocrine-disrupting chemicals (EDCs) in the environment has become a major concern worldwide. Studies showed that EDCs could cause infertility, feminization, and other adverse effects in animals (Colborn et al. 1993; Irwin et al. 2001; Jobling et al. 1996; Levy et al. 2004). Fish were also shown to have extremely high EDCs in plasma after they had been caged briefly in sewage treatment plant (STP) effluents (Purdom et al. 1994). A recent study suggested that in addition to their effects on sexual differentiation and reproduction, sex hormones appeared to influence the human immune system (Bouman et al. 2005). EDCs include a wide variety of chemicals, such as natural estrogens, phytoestrogens, industrial chemicals, and pesticides. Among them, the natural estrogens 17α-ethynylestradiol (EE2), estrone (E1), and 17β-estradiol (E2) are the major contributors of estrogenic activity in the environment. Other industrial chemicals carrying a phenolic group, such as bisphenol A (BPA) and nonylphenol (NP), are also suspected to disrupt the endocrine system in animals. For example, the main endocrine effects of BPA are vitellogenin induction in goldfish (Li et al. 2012).
The major environmental sources of EDCs are from effluents of STPs and direct urban or industrial discharges (Petrovic et al. 2002; Zhang et al. 2013). A number of studies showed that EDCs could not be completely removed by present treatment processes to be directly discharged into the receiving river water and other water bodies (Bertanza et al. 2010; Hamid and Eskicioglu 2012; Maletz et al. 2013; Reddy and Brownawell 2005). Many studies also focused on the occurrence of EDCs in the natural environments (Ferguson et al. 2001; Gong et al. 2009; Rocha et al. 2013; Sumpter and Jobling 2013). For natural estrogens, their contents were usually rather low in surface water, often found down to the ng/L level. However, relatively high concentrations were detected in stream or river water in China and the USA, with a maximum of 112 ng/L for E1 and a maximum of 200 ng/L for E2 (Kolpin et al. 2002; Lei et al. 2009). EDCs in sediments and suspended particulates were generally much higher than in soluble concentrations, likely due to their low water solubility or high capacity of sorption (Froehner et al. 2012; Galluba and Oehlmann 2012; Gong et al. 2012).
It has been documented (Wang et al. 2007) that riverine runoff is the most important contributor of contaminants in the coastal zones of Pearl River Delta (PRD), which is one of the most developed and urbanized areas in China while having a total area of 41,700 km2 and a population of 41 million. This area receives 64 % of industrial sewage and 74 % of domestic sewage in Guangdong Province (Ma et al. 2005). It was estimated that about 1.73 × 1010 metric tons of wastewater were discharged from the PRD into the Pearl River Estuary (PRE) and the South China Sea (SCS) per year (He et al. 2005). Therefore, it is necessary to conduct a comprehensive study to evaluate both the impacts of riverine discharges on transfers of contaminants in the PRD and the increased anthropogenic activities on the local coastal environments. In this study, six natural and synthetic EDCs were selected for monitoring their occurrence and removal in the STPs and the receiving river water, and for determining their residues in the riverine runoffs from the Pearl River and the PRE. Additionally, this study had a special twofold interest in quantifying the mass EDCs loadings from PRD to the nearby ocean and in assessing their potential health impacts on aquatic biota.
Methods and materials
Chemicals and materials
All EDCs standards, 4-NP (94 %), BPA, 17α-ethynylestradiol (EE2), estrone (E1), 17β-estradiol (E2), and estriol (E3), were purchased from Sigma-Aldrich Co. (USA). Methanol, acetonitrile (HPLC grade), and methyl tert-butyl ether (MTBE) were obtained from Merck (Darmstadt, Germany). Ultra-pure water was prepared with a Milli-Q water purification system (Millipore, Bedford, MA, USA). Glass fiber filters (0.7 mm) and Oasis HLB cartridges (500 mg, 6 mL) were purchased from Whatman (Maidstone, UK) and Waters (Milfort, USA), respectively. Unless noted otherwise, chemicals used in the analysis were purer than the analytical grade.
Samples collection
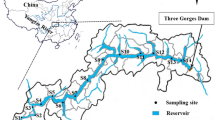
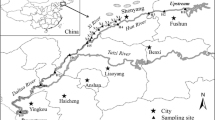
Sewage was collected in a municipal STP in Guangzhou in June 2011. The detailed information of the STP can be found in a related study by Xu et al. (2007). Raw sewage influent and final effluent were collected as 24-h composite samples (each about 20 L, from 7 a.m. to 7 a.m., with three samples combined for analysis). The effluent was directly discharged to the Pearl River, which was the sole receiving river water. A total of six river water samples were collected in the Pearl River after the STP sampling (Fig. 1). A 5-L water sample was collected (~1 m below the surface) at each location using a water grab sampler.
The Pearl River is the largest and most complicated water system in the southern China. Its river water eventually enters the SCS via eight major riverine runoff outlets (Fig. 1): Humen (HM), Jiaomen (JM), Hongqilimen (HQ), Hengmen (HE) on the eastern side (the four eastern outlets); and Modaomen (MD), Jitimen (JT), Hutiaomen (HT), and Yamen (YM) on the western side (the four western outlets). Detailed information of the eight outlets can be found in another related study by Ni et al. (2008). The samplings were carried out in June 2009 and January 2010, the typical month of wet and dry season, respectively. To avoid any potential tide influence, all sampling work was conducted during neap tides (Ni et al. 2008). Hydrologic data were available from the hydrologic stations adjacent to the sampling sites in each outlet (Table S1). At each outlet, three sampling points evenly distributed along the river’s cross section were selected. Water samples (surface and bottom) were collected as 1-h composite samples using a peristaltic pump (BT50b, Prefluid) at the rate of 100 mL/min. Thereafter, the surface and bottom waters at each individual sampling point were combined into one composite sample. Seawater samples from the PRE were also collected during the two sampling campaigns. Seawater samples were collected as 1-h composite samples during neap tides. All water samples including river water and seawater were stored in precleaned 10-L brown glass bottles and kept at 5 °C in a cold storage room.
Samples preparation and extraction
Treatment and analysis of water samples were performed according to Vanderford et al. (2003) based on solid phase extraction. Briefly, 400-mL sewage water and 1,000-mL river water samples were filtered through glass fiber filters and acidified to pH 3.0 using sulfuric acid (18 N). The water was then extracted by an Oasis HLB cartridge that had been previously conditioned using dichloromethane, MTBE, methanol, and water (in that order). The HLB cartridge was then rinsed with 10 mL of ultra-pure water and dried under nitrogen gas for 1 h. Thereafter, the analytes were eluted by 5 mL methanol followed by a mixture of methanol/MTBE (10/90). Extracts were concentrated to a final volume of 1 mL using a gentle stream of nitrogen. Final solutions were filtered through a 0.45-μm filter before analysis. For the recovery experiments, 1 L each of filtered artificial seawater, river water, and sewage water were fortified separately with 100 ng of target analytes and 100 ng of surrogate. The solutions were treated by the same procedure as the field samples described above.
Chemical analysis
Analytes were analyzed using high-performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry (HPLC–MS/MS). HPLC was an HP 1100 LC (Agilent Technologies, Palo Alto, CA, USA) controlled gradient system. An Athena C18-WP (CNW Technologies GmbH, Germany. 4.6 mm × 150 mm i.d., 5.0 μm) chromatograph column was employed. Mass spectrometric measurements were performed on a Sciex API 4000™ (Applied Biosystems, USA) equipped with an electrospray ionization source. The conditions of LC system and mass spectrometric were optimized using the method described in Beck et al. (2005).
Quality assurance and quality control
Quantitative analysis of each compound was performed using two highest characteristic precursor ion/product ion transitions. Along with the retention times, the characteristic ions were used to ensure correct peak assignment and peak purity. In addition, surrogate was added to all the samples to monitor the whole treatment procedure. The limit of quantification (LOQ) was defined as a signal-to-noise ratio of 10. Table S2 shows the chemical characteristics of the target compounds. The mean recoveries for these spiked EDCs ranged from 70 to 104 % in river water, 58 to 112 % in seawater, and 55 to 103 % in sewage water (Table S3). The recoveries of the surrogate ranged from 65 to 92 % with a mean of 80 %. All relative standard deviations were less than 20 %. The LOQs of target compounds were from 0.2 to 15 ng/L.
Statistics and data analysis
The results were summarized into a multielemental database using Excels. As data were not normally distributed in the Pearl River, the distribution of EDCs in concentrations was illustrated using SPSS 12.0 statistical software. All the data of EDCs concentrations were not corrected with recovery values.
Results and discussion
Occurrence and removal of the EDCs at the STP
Table 1 summarizes the concentration profiles and removal rates of the EDCs at the STP. The six EDCs were all detected in the influent with concentrations ranging from 45 to 1,050 ng/L. They were comparable with those found in other STPs in China (Liu et al. 2012; Yu et al. 2011; Zhou et al. 2010), but much higher than those in Brazil, Italy, Germany, and other countries (Baronti et al. 2000; Cargouet et al. 2004; Ying et al. 2009). This difference in EDC concentrations could be due to the difference in contraceptive behavior between people living in China and in these other countries. BPA was the most abundant compound found. This compound is widely used in the production of epoxy resins and polycarbonates, with a consumption of more than 206,000 tons each year in China (Peng et al. 2007), which may explain for its high content found in the influent. The average concentrations of NP and EE2 in the influent were 872 and 94 ng/L, respectively. NP in the sewage was generally at the same level as those found in other studies (Baronti et al. 2000; Cargouet et al. 2004; Ying et al. 2009). In the case of natural estrogens, E1 and E3 were most abundant in the influent with average concentrations of 152 and 157 ng/L, respectively, which were consistent with previous findings (Liu et al. 2012; Yu et al. 2011; Zhou et al. 2010). These high levels of E1 and E3 found in the influent appeared to be consistent with the fact that they are the major excretion products of estrogens from human and wildlife. In Guangzhou, 28.2 % of the total population (12.7 million) are women of childbearing age at 15–49 (National Bureau of Statistics 2011). The E1 concentrations were reportedly found in the range of 5–20 ng/L in wastewater, whereas those of E2 varied from 1 to 10 ng/L (Baronti et al. 2000; Belfroid et al. 1999; Carballa et al. 2004; Johnson et al. 2000; Vethaak et al. 2005).
Except for E3, all the EDCs were still detectable in the effluents after treatment, with mean concentrations ranging from 2.0 to 93 ng/L. The average removal rate reached at 92 %, with the highest being 100 % for E3 (Fig. S1). In particular, EE2 and BPA were removed from the aqueous phase at the rate of 94 and 92 %, respectively. The removal rate for E1 was, however, lower under current treatment (with a mean rate of 83 %), which was consistent with those found in other studies (Baronti et al. 2000; Zhang et al. 2012b). For E2 and E3, their removal rates (96 and 100 % on average) were close to those (90 and 100 % on average) reported in Japan (Nakada et al. 2006) and higher than those (68 and 64 % on average) reported in Germany (Ternes et al. 1999). Zhou et al. (2010) observed that biological treatment was the predominant removal means for natural estrogens, which accounted for 68.4 and 90.9 % of the removals of E2 and E3, respectively. Therefore, the removals of E2 and E3 in this study were likely via biodegradation processes. In addition, advanced oxidation could increase the elimination rates of EDCs in sewage (Bertanza et al. 2010).
Occurrence of the EDCs in the receiving river water
The EDCs, predominated by BPA, were detected in the river water at concentrations ranging from 0.23 to 625 ng/L (Table 1). As for natural estrogens, E1 was the predominant compound with an average concentration of 9.2 ng/L. E2 and E3 were only present in three water samples, all with rather low concentrations. Previous studies from the United States and some European countries supported the findings from the present study that E1 was the only or the most frequently detected natural estrogen (Baronti et al. 2000; Belfroid et al. 1999; Noppe et al. 2007; Rocha et al. 2013; Soto et al. 2004; Williams et al. 2003). None of the EDCs exhibited distinct spatial distribution. The maximum values of BPA and E1 were detected at P3, which suggested that untreated wastewater outfalls might exist around the sampling site. Table S4 lists the selected EDCs in aquatic and coastal environments from different locations. The levels of natural estrogens measured in the Pearl River were generally comparable with those found in other aquatic environments in China and some Asian or European countries (Peng et al. 2008; Zhao et al. 2011a, b). In contrast, the levels of other EDCs measured in the present study were generally much higher than those reported in other countries (Kuch and Ballschmiter 2001; Peng et al. 2007).
In the present study, a dilution factor of 1:10 recommended by the U.S. Food and Drug Administration (FDA 1998) was used to extrapolate from the STP effluent data the expected maximum concentrations of the EDCs in the receiving water. The expected maximum concentrations of the EDCs in the Pearl River and the measured concentrations from the sampling site close to the STPs are shown in Fig. 2. The data in the figure reveals that the expected concentrations of E2 and E3 were very close to their measured concentrations. For NP and BPA, however, their measured concentrations were about 30–40 times higher than the expected values; and even the measured concentrations for E1 and EE2 were about 4–5 times higher than the expected. These findings thus strongly implicated that huge amounts of untreated wastewater containing the target compounds had been continuously discharged into the river. These EDCs in the river likely came from either the untreated or non-biologically treated domestic wastewater, rather than from the STP effluent.
The measured and expected concentrations of the EDCs in the Pearl River. Note expected values were derived from the STP effluent data using a dilution factor of 1:10 recommended by U.S. FDA (1998)
Spatial and seasonal variation of the EDCs in the riverine outlets
Table 2 shows the EDCs concentration profiles in different seasons from the eight riverine outlets. E1, the only detected natural estrogen, was present in several water samples. Different from those in the sewage and the Pearl River, the levels of NP were generally higher than those of BPA in the riverine outlets, regardless of dry or wet season. This suggested that additional NP sources might exist near the midstream or down midstream. Spatially, the levels of the detected EDCs in the four eastern outlets were generally higher than those in the western outlets. It should be noted that the four eastern outlets and their upper reaches are all located in the PRD region, where numerous factories and manufactories are present. The PRD region receives 75 % sewage water of the whole Guangdong Province each year. Therefore, the domestic sewage and industrial wastewater in the PRD are likely the main sources of the EDCs in the Pearl River system. Compared with the eastern outlets, poultry breeding farms and fishing ponds are intensively distributed in the areas around the western outlets and their upper reaches. And the natural estrogens found in the western outlets could hence be traced to their consumption in breeding activities.
The levels of these EDCs were significantly (p < 0.05) higher in dry season than in wet season. The strong dilution factor in wet season might be responsible for their low concentrations due to the heavy rainfalls and strong surface runoffs. Statistics from the hydrologic stations showed that the runoff volume in wet season was about 4–8 times larger than in dry season. The study by Liu et al. (2010) revealed that dilution factor could influence the EDCs levels not only in the estuaries but also in coastal water. In addition, biodegradation and photo-degradation might be stronger in summer than in winter due to both the higher microbic activity and the stronger sunlight present in summer months (Zhang et al. 2012a).
Fluxes of EDCs from the river to the ocean
The annual input of the EDCs from the Pearl River to the nearby ocean was calculated according to their concentrations and the flow volumes in different seasons. The annual input from the riverine outlets to the ocean was from 15 to 199 tons with a total loading of 578 tons (Fig. S2). NP and BPA contributed more than 99 % of the total mass loading. The E1 input (1.6 tons) was similar to that of EE2 (1.0 ton). Spatially, the contributions from the eight outlets were consistent with the EDCs levels found. Individually, the HM outlet constituted the largest portion (about 30 %), followed by the JM and HE outlets; this might be attributed to their larger water discharge volumes and the higher concentrations of the EDCs present. It should be noted that the EDCs adsorbed to suspended particles were not included in the flux calculation. Thus, the actual fluxes of EDCs from the eight outlets were likely higher than the values presented here. Nonetheless, the data obtained in this study could help us understand the current situation and the influence of the river discharging to the nearby ocean.
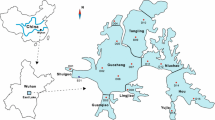
Levels of the EDCs in the PRE
Figure 3 shows the distributions of NP and BPA in the PRE during wet and dry seasons. E1 and EE2 were present at only a few sites. Except for site 16, NP and BPA were found in all the sampling sites. The NP concentrations ranged from 11 to 163 ng/L, with a mean of 45 ng/L. The levels of BPA were from 10 to 178 ng/L, with a mean of 47 ng/L. The spatial distributions of NP and BPA in the PRE together clearly showed that the higher levels all occurred close to the cities or the river outlets. NP and BPA levels in the central locations of the PRE were generally lower than those on both sides of the bank. This finding suggested that municipal sewage and industrial wastewater from the large cities around the PRE, such as Shenzhen and Dongguan, might be the major sources of the EDCs, especially for NP and BPA. In addition, NP and BPA generally showed a decreasing trend from the PRE toward the SCS, confirming that riverine runoff is important for the transport of these contaminants from terrestrial sources to the ocean. It is of note here that NP and BPA were frequently detected at sampling sites that were close to the open sea.
Impacts to the aquatic environments
EDCs have been suspected to be responsible for the estrogenic activities and fish reproductive disorder in rivers (Nakada et al. 2006). Studies revealed that very low concentration of E2 could lead to the induction of plasma vitellogenin (VTG) in male trout (Sumpter and Johnson 2005). It was demonstrated that BPA (22.4 μg/L) significantly increased the number of Xenopus laevis tadpoles with a sex ratio of 70 % when compared to the control tests (Levy et al. 2004). In the present study, the detected EDCs in the river were generally lower than the effective concentrations (Levy et al. 2004). However, it should be noted that the simultaneous occurrence of these compounds in an environment can produce significant joint (e.g., synergistic) effects. The estrogenicity of a compound can be related to that of 17β-estradiol and is often expressed as 17β-estradiol equivalency quotient (EEQ). The EEQ concentrations may be varied according to the estradiol equivalent factor (EEF) obtained from different assays. In the present study, EEQ values were calculated using the following equation:
where Ci is the concentration of estrogenic compound i and EEFi is its respective estradiol equivalent factor. The EEFs of E1, E2, E3, EE2, NP, and BPA were set at 0.1, 1.0, 0.0049, 1.0, 9.0 × 10−4 and 6.0 × 10−5, as those shown in Table S2. As shown in Fig. S3, the average EEQ concentrations in the river water and estuary samples ranged from 0.2 to 4.5 ng/L. EEQ concentrations gradually decreased from the city section to the river outlets and estuary. In the river from the city section, E1, EE2 and NP represented an average of 20, 63, and 8 % of the total in EEQ, respectively. The portions for E1, EE2, and NP in the estuary were 11, 40, and 10 %, respectively. E1 and EE2 apparently were the two predominant contributors to the EEQ concentrations. EE2 is a very potent estrogenic compound, which can significantly elevate the VTG levels in trout even at low concentrations (Purdom et al. 1994). Therefore, EE2 could be a major contributor to the estrogenic response in fish, even though it was present at concentrations lower than those of E1 in the river water. It has been suggested that EEQ concentrations >1 ng/L in aquatic environment could cause reproductive problems in fish (Young et al. 2002). Accordingly, it is reasonable to expect that the EEQ in the Pearl River (city section) can potentially cause endocrine disruption in the aquatic biota there.
Conclusions
The selected EDCs were found to be ubiquitous in sewage, surface water, and seawater from the PRD region; and they could not be completely removed by present treatment techniques used in the STPs. The findings in this study suggested that the untreated municipal sewages and industrial wastewater were the main sources of the EDCs in the Pearl River. The levels of the natural estrogens measured in this study were similar to those detected in other countries. However, the NP and BPA levels measured in this study were in the high range in the world. More than 578 tons of the EDCs per year were discharged into the coastal environment from the PRD region. The concentrations of the EDCs detected in PRD were high enough to raise concerns of the endocrine disruption effects on the aquatic biota living in these waters.
References
Baronti, C., Curini, R., D’Ascenzo, G., Di Corcia, A., Gentili, A., & Samperi, R. (2000). Monitoring natural and synthetic estrogens at activated sludge sewage treatment plants and in a receiving river water. Environmental Science and Technology, 34, 5059–5066.
Beck, I. C., Bruhn, R., Gandrass, J., & Ruck, W. (2005). Liquid chromatography-tandem mass spectrometry analysis of estrogenic compounds in coastal surface water of the Baltic Sea. Journal of Chromatography A, 1090, 98–106.
Belfroid, A. C., Van der Horst, A., Vethaak, A. D., Schafer, A. J., Rijs, G. B. J., Wegener, J., et al. (1999). Analysis and occurrence of estrogenic hormones and their glucuronides in surface water and waste water in The Netherlands. Science of the Total Environment, 225, 101–108.
Bertanza, G., Pedrazzani, R., Zambarda, V., Dal Grande, M., Icarelli, F., & Baldassarre, L. (2010). Removal of endocrine disrupting compounds from wastewater treatment plant effluents by means of advanced oxidation. Water Science and Technology, 61, 1663–1671.
Bouman, A., Heineman, M. J., & Faas, M. M. (2005). Sex hormones and the immune response in humans. Human Reproduction Update, 11, 411–423.
Carballa, M., Omil, F., Lema, J. M., Llompart, M., Garcia-Jares, C., Rodriguez, I., et al. (2004). Behavior of pharmaceuticals, cosmetics and hormones in a sewage treatment plant. Water Research, 38, 2918–2926.
Cargouet, M., Perdiz, D., Mouatassim-Souali, A., Tamisier-Karolak, S., & Levi, Y. (2004). Assessment of river contamination by estrogenic compounds in Paris area (France). Science of the Total Environment, 324, 55–66.
Colborn, T., Saal, F. S. V., & Soto, A. M. (1993). Developmental effects of endocrine-disrupting chemicals in wildlife and humans. Environmental Health Perspectives, 101, 378–384.
FDA (Federal Drug Administration). (1998). Guidance for industry environmental assessment of human drugs and biologics applications (revision 1). Rockville, MD.
Ferguson, P. L., Iden, C. R., & Brownawell, B. J. (2001). Distribution and fate of neutral alkylphenol ethoxylate metabolites in a sewage-impacted urban estuary. Environmental Science and Technology, 35, 2428–2435.
Froehner, S., Machado, K. S., Stefan, E., Bleninger, T., da Rosa, E. C., & de Castro Martins, C. (2012). Occurrence of selected estrogens in mangrove sediments. Marine Pollution Bulletin, 64, 75–79.
Galluba, S., & Oehlmann, J. (2012). Widespread endocrine activity in river sediments in Hesse, Germany, assessed by a combination of in vitro and in vivo bioassays. Journal of Soils and Sediments, 12, 252–264.
Gong, J., Ran, Y., Chen, D., Yang, Y., & Ma, X. (2009). Occurrence and environmental risk of endocrine-disrupting chemicals in surface waters of the Pearl River, South China. Environmental Monitoring and Assessment, 156, 199–210.
Gong, J., Ran, Y., Chen, D., Yang, Y., & Zeng, E. Y. (2012). Association of endocrine-disrupting chemicals with total organic carbon in riverine water and suspended particulate matter from the Pearl River, China. Environmental Toxicology and Chemistry, 31, 2456–2464.
Hamid, H., & Eskicioglu, C. (2012). Fate of estrogenic hormones in wastewater and sludge treatment: A review of properties and analytical detection techniques in sludge matrix. Water Research, 46, 5813–5833.
He, X. Y., Shi, J. H., Xin, H. H., Wang, B., & He, S. D. (2005). Dynamic changes of land-originated oils from 8 mouths of Pearl River. China Science Technology Information, 21, 84–85.
Irwin, L. K., Gray, S., & Oberdorster, E. (2001). Vitellogenin induction in painted turtle, Chrysemys picta, as a biomarker of exposure to environmental levels of estradiol. Aquatic Toxicology, 55, 49–60.
Jobling, S., Sheahan, D., Osborne, J. A., Matthiessen, P., & Sumpter, J. P. (1996). Inhibition of testicular growth in rainbow trout (Oncorhynchus mykiss) exposed to estrogenic alkylphenolic chemicals. Environmental Toxicology and Chemistry, 15, 194–202.
Johnson, A. C., Belfroid, A., & Di Corcia, A. (2000). Estimating steroid oestrogen inputs into activated sludge treatment works and observations on their removal from the effluent. Science of the Total Environment, 256, 163–173.
Kolpin, D. W., Furlong, E. T., Meyer, M. T., Thurman, E. M., Zaugg, S. D., Barber, L. B., et al. (2002). Pharmaceuticals, hormones, and other organic wastewater contaminants in US streams, 1999–2000: A national reconnaissance. Environmental Science and Technology, 36, 1202–1211.
Kuch, H. M., & Ballschmiter, K. (2001). Determination of endocrine-disrupting phenolic compounds and estrogens in surface and drinking water by HRGC-(NCI)-MS in the picogram per liter range. Environmental Science and Technology, 35, 3201–3206.
Lei, B., Huang, S., Zhou, Y., Wang, D., & Wang, Z. (2009). Levels of six estrogens in water and sediment from three rivers in Tianjin area, China. Chemosphere, 76, 36–42.
Levy, G., Lutz, I., Kruger, A., & Kloas, W. (2004). Bisphenol A induces feminization in Xenopus laevis tadpoles. Environmental Research, 94, 102–111.
Li, Z., Zhang, H., Gibson, M., & Liu, P. (2012). An evaluation of the combined effects of phenolic endocrine disruptors on vitellogenin induction in goldfish Carassius auratus. Ecotoxicology, 21, 1919–1927.
Liu, Y., Guan, Y., Tam, N. F. Y., Mizuno, T., Tsuno, H., & Zhu, W. (2010). Influence of rainfall and basic water quality parameters on the distribution of endocrine-disrupting chemicals in coastal area. Water, Air, and Soil pollution, 209, 333–343.
Liu, S., Ying, G.-G., Zhao, J.-L., Zhou, L.-J., Yang, B., Chen, Z.-F., et al. (2012). Occurrence and fate of androgens, estrogens, glucocorticoids and progestagens in two different types of municipal wastewater treatment plants. Journal of Environmental Monitoring, 14, 482–491.
Ma, Q. J., Hu, M., Zhu, T., Liu, L. L., & Dai, M. H. (2005). Seawater, atmospheric dimethylsulfide and aerosol ions in the Pearl River Estuary and the adjacent northern South China Sea. Journal of Sea Research, 53, 131–145.
Maletz, S., Floehr, T., Beier, S., Kluemper, C., Brouwer, A., Behnisch, P., et al. (2013). In vitro characterization of the effectiveness of enhanced sewage treatment processes to eliminate endocrine activity of hospital effluents. Water Research, 47, 1545–1557.
Nakada, N., Tanishima, T., Shinohara, H., Kiri, K., & Takada, H. (2006). Pharmaceutical chemicals and endocrine disrupters in municipal wastewater in Tokyo and their removal during activated sludge treatment. Water Research, 40, 3297–3303.
National Bureau of Statistics. (2011). The sixth nationwide census. Beijing.
Ni, H.-G., Lu, F.-H., Luo, X.-L., Tian, H.-Y., Wang, J.-Z., Guan, Y.-F., et al. (2008). Assessment of sampling designs to measure riverine fluxes from the Pearl River Delta, China to the South China Sea. Environmental Monitoring and Assessment, 143, 291–301.
Noppe, H., Verslycke, T., De Wulf, E., Verheyden, K., Monteyne, E., Van Caeter, P., et al. (2007). Occurrence of estrogens in the Scheldt estuary: A 2-year survey. Ecotoxicology and Environmental Safety, 66, 1–8.
Peng, X., Wang, Z., Mai, B., Chen, F., Chen, S., Tan, J., et al. (2007). Temporal trends of nonylphenol and bisphenol A contamination in the Pearl River Estuary and the adjacent South China Sea recorded by dated sedimentary cores. Science of the Total Environment, 384, 393–400.
Peng, X., Yu, Y., Tang, C., Tan, J., Huang, Q., & Wang, Z. (2008). Occurrence of steroid estrogens, endocrine-disrupting phenols, and acid pharmaceutical residues in urban riverine water of the Pearl River Delta, South China. Science of the Total Environment, 397, 158–166.
Petrovic, M., Sole, M., de Alda, M. J. L., & Barcelo, D. (2002). Endocrine disruptors in sewage treatment plants, receiving river waters, and sediments: Integration of chemical analysis and biological effects on feral carp. Environmental Toxicology and Chemistry, 21, 2146–2156.
Purdom, C. E., Hardiman, P. A., Bye, V. J., Eno, N. C., Tyler, C. R., & Sumpter, J. P. (1994). Estrogenic effects of effluents from sewage treatment works. Chemistry and Ecology, 8, 275–285.
Reddy, S., & Brownawell, B. J. (2005). Analysis of estrogens in sediment from a sewage-impacted urban estuary using high-performance liquid chromatography/time-of-flight mass spectrometry. Environmental Toxicology and Chemistry, 24, 1041–1047.
Rocha, M. J., Cruzeiro, C., Reis, M., Rocha, E., & Pardal, M. A. (2013). Determination of 17 endocrine disruptor compounds and their spatial and seasonal distribution in the Sado River Estuary (Portugal). Toxicological and Environmental Chemistry, 95, 237–253.
Soto, A. M., Calabro, J. M., Prechtl, N. V., Yau, A. Y., Orlando, E. F., Daxenberger, A., et al. (2004). Androgenic and estrogenic activity in water bodies receiving cattle feedlot effluent in eastern Nebraska, USA. Environmental Health Perspectives, 112, 346–352.
Sumpter, J. P., & Jobling, S. (2013). The occurrence, causes, and consequences of estrogens in the aquatic environment. Environmental Toxicology and Chemistry, 32, 249–251.
Sumpter, J. P., & Johnson, A. C. (2005). Lessons from endocrine disruption and their application to other issues concerning trace organics in the aquatic environment. Environmental Science and Technology, 39, 4321–4332.
Ternes, T. A., Stumpf, M., Mueller, J., Haberer, K., Wilken, R. D., & Servos, M. (1999). Behavior and occurrence of estrogens in municipal sewage treatment plants—I. Investigations in Germany, Canada and Brazil. Science of the Total Environment, 225, 81–90.
Vanderford, B. J., Pearson, R. A., Rexing, D. J., & Snyder, S. A. (2003). Analysis of endocrine disruptors, pharmaceuticals, and personal care products in water using liquid chromatography/tandem mass spectrometry. Analytical Chemistry, 75, 6265–6274.
Vethaak, A. D., Lahr, J., Schrap, S. M., Belfroid, A. C., Rijs, G. B. J., Gerritsen, A., et al. (2005). An integrated assessment of estrogenic contamination and biological effects in the aquatic environment of The Netherlands. Chemosphere, 59, 511–524.
Wang, J. Z., Guan, Y. F., Ni, H. G., Luo, X. L., & Zeng, E. Y. (2007). Polycyclic aromatic hydrocarbons in riverine runoff of the Pearl River Delta (China): Concentrations, fluxes, and fate. Environmental Science and Technology, 41, 5614–5619.
Williams, R. J., Johnson, A. C., Smith, J. J. L., & Kanda, R. (2003). Steroid estrogens profiles along river stretches arising from sewage treatment works discharges. Environmental Science and Technology, 37, 1744–1750.
Xu, W., Zhang, G., Li, X., Zou, S., Li, P., Hu, Z., et al. (2007). Occurrence and elimination of antibiotics at four sewage treatment plants in the Pearl River Delta (PRD), South China. Water Research, 41, 4526–4534.
Ying, G.-G., Kookana, R. S., Kumar, A., & Mortimer, M. (2009). Occurrence and implications of estrogens and xenoestrogens in sewage effluents and receiving waters from South East Queensland. Science of the Total Environment, 407, 5147–5155.
Young, W. F., Whitehouse, P., Johnson, I., & Sorokin, N. (2002). Proposed predicted no effect concentrations (PNECs) for natural and synthetic steroid oestrogens in surface waters. Environment Agency R & D Technical report P2-T04/1. Bristol: England and Wales Environment Agency (pp. 172).
Yu, Y., Huang, Q., Wang, Z., Zhang, K., Tang, C., Cui, J., et al. (2011). Occurrence and behavior of pharmaceuticals, steroid hormones, and endocrine-disrupting personal care products in wastewater and the recipient river water of the Pearl River Delta, South China. Journal of Environmental Monitoring, 13, 871–878.
Zhang, W., Li, Y., Su, Y., Mao, K., & Wang, Q. (2012a). Effect of water composition on TiO2 photocatalytic removal of endocrine disrupting compounds (EDCs) and estrogenic activity from secondary effluent. Journal of Hazardous Materials, 215–216, 252–258.
Zhang, W., Li, Y., Wang, C., Wang, P., & Wang, Q. (2013). Energy recovery during advanced wastewater treatment: Simultaneous estrogenic activity removal and hydrogen production through solar photocatalysis. Water Research, 47, 1480–1490.
Zhang, W., Li, Y., Wu, Q., & Hu, H. (2012b). Removal of endocrine-disrupting compounds, estrogenic activity, and Escherichia coliform from secondary effluents in a TiO2-coated photocatalytic reactor. Environmental Engineering Science, 29, 195–201.
Zhao, J.-L., Ying, G.-G., Chen, F., Liu, Y.-S., Wang, L., Yang, B., et al. (2011a). Estrogenic activity profiles and risks in surface waters and sediments of the Pearl River system in South China assessed by chemical analysis and in vitro bioassay. Journal of Environmental Monitoring, 13, 813–821.
Zhao, J.-L., Ying, G.-G., Yang, B., Liu, S., Zhou, L.-J., Chen, Z.-F., et al. (2011b). Screening of multiple hormonal activities in surface water and sediment from the Pearl River System, South China, using effect-directed in vitro bioassays. Environmental Toxicology and Chemistry, 30, 2208–2215.
Zhou, H., Huang, X., Wang, X., Zhi, X., Yang, C., Wen, X., et al. (2010). Behaviour of selected endocrine-disrupting chemicals in three sewage treatment plants of Beijing, China. Environmental Monitoring and Assessment, 161, 107–121.
Acknowledgments
The authors would like to acknowledge the financial support from NSFC (Nos. 41272390 and 41106104) and the Scientific Frontier Program for Young Talents of the South China Sea Institute of Oceanology, CAS (Grant No. SQ200909). The study was also funded by CAS/SAFEA International Partnership Program for Creative Research Teams (No. KZCX2-YW-T001).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xu, W., Yan, W., Huang, W. et al. Endocrine-disrupting chemicals in the Pearl River Delta and coastal environment: sources, transfer, and implications. Environ Geochem Health 36, 1095–1104 (2014). https://doi.org/10.1007/s10653-014-9618-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-014-9618-3