Abstract
Changes in the fatty acid (FA) composition of total lipids of Pinus sylvestris needles at different pollution levels caused by emissions from a large aluminum smelter (BrAS) have been studied. In the needles of trees from unpolluted (background) territories, the FA spectrum is represented by 24 acids with prevalence of unsaturated FAs (71.6%). The main unsaturated FA are represented by oleic (C18: 1ω9), linoleic (C18: 2ω6), and α-linolenic (C18: 3ω3) acids. Under the influence of BrAS emissions, the total amount of identified FAs in the needles and the proportion of unsaturated FAs decrease, while the fraction of saturated FAs, on the contrary, increases from 25.4% in unpolluted needles to 33.2% in polluted ones. The content of palmitic FA (C16:0) in the needles exceeds background values by 1.5 times, behenic acid (C22:0) – by 1.6–2.5 times, arachidic acid (C20:0) – by 1.5 times, palmitic margaric acid (C17:0) – by 1.5–2.3 times. These FAs play the important role in the protection of plant membranes from the effects of abiotic stress factors, making them less permeable. The sum of short-chain saturated FAs (C12:0, C14:0, C15:0) increase by 4.8 times in needles of trees that are highly polluted. Pentadecanoic (C15:0) acid is found in the needles only in the background areas and at the low pollution level. With a more severe pollution, C15:0 is not identified, but lauric acid with the cis-configuration of double bonds in the structure (izo-C12:0) appears. The presence of “relict” ∆5-polymethylene FAs in the composition of pine needle membrane lipids is determined. In the background areas, they account for 12.9% of the total FAs. With the industrial pollution intensification, their total content increases and reaches 14.1%. ∆5-polymethylene FAs are also able to protect membranes against negative influences. Thus, changes in the quantitative and qualitative FA composition of pine needle total lipids indicate the activation of the stabilization mechanisms of membrane lipids due to their tight packing in a bilayer. It is one of the adaptive reactions of Pinus sylvestris in response to the impact of the aluminum industry emissions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In recent decades, industrial pollution is one of the immediate anthropogenic factors that have a negative impact on forest ecosystems. The viability of woody plants under technogenic pollution is largely determined by their protective and adaptive capabilities existing at different organization levels. At the cellular level, the maintenance of plant homeostasis is regulated by changes in the content of a number of low molecular weight compounds (Blokhina et al. 2003; Foyer and Noctor 2015), by the activity of antioxidant enzymes and the rearrangement of their isozyme composition (Zhang and Kirkham 1994; Weil and Schaub 1999; Singh et al. 2018). The role of membrane lipids is also significant in the processes of adaptation and resistance of a plant organism to industrial pollution and other unfavorable environmental factors (Yoshida and Sakai 1974; Zielinska et al. 2000; Tarchevsky 2002; Wang et al. 2006; Munnik and Testerink 2009; Moellering et al. 2010; Okazaki et al. 2013; Hou et al. 2016; Kondratenko et al. 2017). The synthesis of unique lipids was shown to sharply increase under stress conditions, while they are present in trace amounts under normal growth conditions (Okazaki and Saito 2014). The regulation of the functionally state of membranes was determined to depend on the ratio of phospho- and galactolipids (Sopin and Trunova 1991; Wang and Benning 2012; Kobayashi, Wada (2016)).
In lipids, the main role in protective-adaptive responses is played by fatty acids. They are able to maintain the fluidity of cell membranes at the functional level as a result of synthesis and decomposition, as well as changes in the saturation degree (Lyons 1973; Ramos et al. 1995; Los’ 2005; Catalá (2006); Taarit et al. 2010). In such a case, desaturase enzymes involved in the formation of double bonds in the hydrocarbon chains of lipid fatty acids play an important role (Ohlrogge and Browse 1995; Il’inskaya and Ozeretskovskaya 1998; Los ‘and Murata 1998, Los’ 2005). The melting temperature of the corresponding fatty acid is known to decrease with an increase in the number of double bonds and the length of the hydrocarbon chain. That leads to an increase in the lipid bilayer plasticity (Arisawa et al. 2016).
As shown by a number of studies, the qualitative and quantitative composition of lipids and fatty acids in plants change under the influence of pollutants, in particular heavy metals (Ouariti et al. 1997; Jemal et al. 2000; Nouairi et al. 2006; Chaffai et al. 2007; Ammar et al. 2008). These changes can be caused by the biosynthesis inhibition and the breakdown of unsaturated fatty acids. The nature of changes in the lipid content depends on plant species characteristics, their age, the nature of a metal-pollutant, its concentration, and the exposure duration (Tukendorf 1993; Drazkiewicz et al. 2003; Nesterov et al. 2009). Most fully, the effect of individual pollutants-elements has been studied on herbaceous plants under experimental conditions. Woody plants, in particular conifers, have been poorly studied in this respect. We found information on the role of fatty acids of pine needle lipids in the cryoresistance formation (Romanova et al. 2016; Nokhsorov et al. 2019). Apparently, the change in the quantitative and qualitative composition of fatty acids in membrane lipids is one of the universal adaptive mechanisms that provide resistance under the influence of various stress factors. In this regard, our scientific interest was to study the changes in the fatty acid composition of the membrane lipids in pine needles under the effect of such a severe negative factor as technogenic emissions.
Such studies are highly relevant for the Irkutsk region (Eastern Siberia, Russia). On the one hand, it is a large industrially developed region, where large industrial facilities of heat-power engineering, fuel, chemical, petrochemical and aluminum industries are concentrated. On the other hand, large areas in the region are occupied by coniferous forests, which are known to be more sensitive to the impact of industrial pollution than deciduous tree species (Rozhkov and Mikhailova 1993).
The aim of this research is to study quantitative and qualitative changes in the composition of fatty acids of membrane lipids and their significance for the adaptation of Pinus sylvestris L. needles to the effect of highly toxic emissions from the aluminum smelter.
Materials and methods
Study area
The research was carried out in the northwestern part of the Irkutsk region, in the vicinity of the city of Bratsk, a large industrial center in Russia. It has been included in the Priority list of cities with the highest level of air pollution (State report “On the State and Environmental Protection of the Irkutsk Region in 2018” 2019). Many stationary industrial facilities are concentrated within the city, of which the largest are non-ferrous metallurgy, heat and power industry, and wood processing industry. In addition, road transport contributes greatly to environmental pollution. The main source of pollution in Bratsk is the Bratsk Aluminum Smelter (BrAS). This is the largest primary aluminum industry enterprise not only in Russia, but also in the world (https://journal.open-broker.ru). The most toxic emission components are fluorinated compounds, in particular hydrogen fluoride and solid fluorides; as well as sulfur dioxide, carbon monoxide, polycyclic aromatic hydrocarbons (PAHs), solid aerosol, which contains a high proportion of heavy metals(State report “On the State and Environmental Protection of the Irkutsk Region in 2018” 2019).
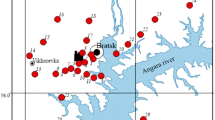
Field surveys were carried out in a forest-covered area within a radius of up to 100 km in different directions from the main source of pollution (BrAS) (Fig. 1). The predominant part of the territory is occupied by Scots pine (Pinus sylvestris L.) and Siberian larch (Larix sibirica Ledeb.) primary forests. Pine stands occupy various habitats: flat, well-warmed areas, elevated landforms, swampy areas. Herbaceous and sedge-herbaceous pine forests predominantly of bonitet class III and sodium carbonate-eluviated soils prevail. The bonitet class was determined according to the scale developed by prof. M.M. Orlov (Forest Encyclopedia et al., (1985)). The choice of pine stands as an object of the present research was due to their wide distribution in the surveyed area.
Sampling methods
The study of pine stands was carried out at 29 sample plots (SP) located in different directions from the aluminum smelter and background natural areas. When choosing a SP, we proceeded from the principle of analogy of landscape and silvicultural characteristics in accordance with the generally accepted recommendations of ICP Forests (Cools, De Vos 2010). The area of each SP was about 0.01 km2. To collect needle samples, 3–5 test sites (TS) were laid on each SP; TS size was approximately 100 m2. Lateral shoots were cut from the southern and south-eastern side of middle part of the crowns of five 40-year-old trees at each TS, using a pruner from Gardena. The needles were selected in the middle of the growing season after the end of the shoot growth phase, in the period from July, 25 to August, 05, 2020. The needles of the second year of life were separated from the shoots, as the most physiologically active. The needles collected from each TS were thoroughly mixed and divided into three parts.
One part of the needles was collected into a Kraft paper bag, it was dried for 48 hours at 60 °С under laboratory conditions, and then it was ground to a powder on a Bosch Electric mills and sifted through a sieve with an aperture diameter of 0.5 mm. The samples were stored in paper bags. Further, this sample portion was used for determining the content of inorganic elements. The qualitative and quantitative composition of fatty acids was determined in the second part of the needles, which was stored in an ultra-low temperature freezer at −83 °C. These needles were delivered to the laboratory from the forest in a Dewar vessel, where there was liquid nitrogen. The third part of needles was packed in aluminum foil and stored in a freezer at −5 °C. This sample portion was used for PAH, hydrogen peroxide and the content of malondialdehyde analysis.
Parameters analyzed
To assess the contamination of needles, the content of fluorine, sulfur, a number of heavy metals (HM), silicon, aluminum, and PAHs was determined. Dry crushed needles were used to determine inorganic contaminants. The fluorine content was determined spectrophotometrically at a wavelength of 540 nm with xylenol orange indicator after dry ashing of the samples and distillation of the resulting ash with water vapor in perchloric acid (Awang et al. 2007). To determine sulfur, aluminum, silicon and HM, needle samples were mineralized in a muffle furnace at 450 °C for three hours. Then, to determine aluminum and HM, the ash was dissolved in 0.1 M nitric acid. To determine silicon, the ash was dissolved in a mixture of concentrated acids (hydrochloric and nitric). To determine sulfur, the ash was dissolved in 2 M hydrochloric acid. The elemental chemical composition in the resulting solutions was determined by atomic absorption spectrophotometry and photocolorimetry (Sanina and Proidakova 2006; Proidakova, Vasileva (2010)) using the instrument park of the Buryat Scientific Center and the Baikal Analytical Center in the Siberian Branch of the Russian Academy of Sciences (Ulan-Ude, Irkutsk). In particular, AAS Vario 6 (Germany) and spectrophotometer UNICO 2100 (USA) were used. PAHs in needles were determined according to A.S. Gorshkov (Gorshkov 2008). Extraction of PAHs from needles was carried out with n-hexane under ultrasonic irradiation. The isolated PAH fraction was analyzed using the Agilent GC System 6890, MSD 5973 chromatomass spectrometer (USA). 16 priority PAHs were identified. They are used by researchers as markers of air pollution by persistent organic pollutants (Boffetta et al. 1997; Vu et al. 2011).
The zoning of the surveyed area depending on the level of industrial pollution was carried out according to the cluster analysis results of data on the content of pollutants in needles. Changes in the elemental composition of needles under the technogenic pollution were characterized using a complex indicator - index of biogeochemical transformation of the elemental composition of needles (Zbt).
where EFl =Cpol/Cb and DFl = Cb/Cpol are local concentration and dispersion coefficients, respectively; Cpol is the element concentration in needles in the polluted area, mg/kg dry weight; Cb is the element concentration in the background area, mg/kg dry weight; n1 and n2 are the numbers of the elements with EFl > 1 and DFl > 1 (Kasimov et al. 2012). The Zbt index has following grades corresponding to low (10–30), moderate (31–50), high (51–80), and critical (>81) disturbance.
The hydrogen peroxide content, an indicator of the oxidative stress development in needles, was determined spectrophotometrically by the formation of a colored complex compound – titanium peroxide from titanium sulfate Ti2(SO4)3 (Brennan and Frenkel 1977). The level of lipid peroxidation (LPO) was determined by the accumulation of the LPO product – malonyldialdehyde (MDA). The MDA content was assessed by the degree of accumulation of the product of its reaction with thiobarbituric acid (Kumar and Knowles 1993).
To study the fatty acid composition, needle samples were fixed in liquid nitrogen and ground in an agate mortar until a homogeneous mass was obtained. Then, lipids were extracted from the mass with a mixture of chloroform-methanol-water solvents (1: 2: 0.8 v/v/v) (Bligh and Dyer 1959). To remove chloroform from lipid extracts, a rotary evaporator RVO-64 (Czech Republic) was used. FA methyl esters were obtained according to the method (Christie, 1993). Their extraction was carried out using the triple treatment with n-hexane (3 × 5 ml). Additional purification from methyl esters was performed with TLC on Sorbfil plates with silica gel. The analysis of methyl esters of fatty acids was carried out by gas-liquid chromatography using the Agilent GC System 6890, MSD 5973 gas chromatograph-mass spectrometer (USA). The detector was a quadrupole mass spectrometer, the ionization method was electron impact (EI), and the ionization energy was 70 eV. The total ion current recording mode was used for the analysis. For separation, the HP-INNOWAX capillary column (30 m × 250 μm × 0.5 μm) was used. The stationary phase was polyethylene glycol. The mobile phase was helium with the gas flow rate 1 ml/min. The evaporator temperature was 250 °C, the ion source −230 °C, the detector −150 °C. The temperature of the line connecting the chromatograph with the mass spectrometer was 280 °C. The scan range was 41–450 a.m.u. The volume of the injected sample was 1 μL, the flow separation was 5:1. The chromatography was carried out in an isothermal mode at 200 °C. Methyl ester standards (Sigma, USA) and mass spectrometry (using the NIST 05 mass spectra library) were used to identify peaks of FA methyl esters (Christie 1993; Wolff and Christie 2002; Torres-Franklin et al. 2009). To assess FA unsaturation in pine needle lipids, the unsaturation coefficient (C) was used. It was calculated as the ratio of the sum of unsaturated FAs to the sum of saturated ones.
The activity of acyl-lipid ω9–, ω6– and ω3-membrane desaturases involved in the biosynthesis of oleic, linoleic, and α-linolenic acids was determined from the percentage of oleic, linoleic and α-linolenic acids as stearoyl-(SDR), oleyl-(ODR), linoleyl-(LDR) desaturatic relations, according to the formulae (Mazliak 1994; Cartea et al. 1998):
Statistical analysis
The content of inorganic element pollutants in needles was expressed in % of dry weight; the PAH content was expressed in ng/g; hydrogen peroxide concentration was estimated in mM/1 g wet weight; MDA was expressed in mM/g × 10−1 wet weight. The relative content of FAs was determined in weight percent of their total content in the composition of needle lipids. The indicators were determined in three-five biological (3–5 TS on each SP) and three analytical replicates. Statistical data processing was carried out using the MS Excel 2010 software package. Cluster analysis was performed using the STATISTICA 8.0 software package. The figures and tables show the mean value parameters for one SP and their standard deviation (SD). The obtained results were checked for normality (Shapiro–Wilk’s test) and equality of variance (Levene’s test). Differences in the measured variables were estimated using the single-factor analysis of variance (one-way ANOVA). Differences significant at Р ≤ 0.05 are discussed. To assess the interrelation between the parameters, the Pearson correlation coefficient was calculated.
Results and discussion
High levels of direct correlation between the content of the main pollutants in pine needles and their concentration in emissions from the Bratsk aluminum smelter have been revealed (Table 1). That indicated the reliability of industrial pollution of forest stands by BrAS emissions.
The highest levels of inorganic pollutants and PAHs in needles are found within a 3 km radius of the smelter. In the industrial zone, within a 0.5 km radius of the smelter, their concentration reaches maximum values. As the distance from BrAS increases, the level of polluting elements in the needles decreases. However, in directions with the prevailing atmospheric transport of emissions (north-east and south-east), the amount of fluorine and PAHs remains higher than background values at a distance of up to 60 km from the smelter; sulfur—up to 40 km, and heavy metals—up to 35–50 km. The entire territory under study was zoned in accordance with the pollution level. The level was determined using the cluster analysis of the obtained data on the content of pollutants in pine needles. The resulting dendrogram showed clusters corresponding to low (cluster D), moderate (cluster C), high (cluster B) and critical (cluster A) pollution levels of pine stands. The background cluster (cluster E) was a separate one formed in SPs (1, 34, 2, 3, 29) in unpolluted areas (Fig. 2).
Different levels of pollution are characterized by different concentration coefficients (EF) of pollutants in needles and different values of the biogeochemical transformation index of the needle elemental composition (Zbt), calculated according to Eq. 1 (Table 2). The maximum concentrations of pollutant elements in the needles and the highest Zbt index values are characteristic of the critical level of pine stand pollution and are detected at SP located in the industrial zone of the smelter at a distance of 0.5 km. Pine stands on SP, located at a distance of 1–10 km from the BrAS, united into a cluster with the high level of pollution; 11–40 km from BrAS—into a cluster with the moderate level of pollution; 41–60 km—into a cluster with the low level of pollution. SP located at a distance of more than 60 from the aluminum smelter are characterized by the minimum concentrations of pollutant elements in the needles and the Zbt index and correspond to the previously established background values (Mikhailova et al. 2017).
Technogenic pollution is known to cause the oxidative stress in plant assimilatory organs. It is indicated by the excessive generation of hydrogen peroxide, a product of free radical reactions, in cells (Scandalios 2002; Cheeseman 2007). Hence, the content of substances indicative of the development of oxidative stress in pine needles from SPs with different pollution levels was determined. First of all, the concentration of H2O2 in the needles increased with an increase in its pollution level (Fig. 3). In the case of low pollution, the amount of peroxide exceeds the background values by 1.5 times; in the case of moderate pollution – by 2.5 times; in the case of high pollution – by 4.6 times; and in the case of critical pollution – by 4.4 times. Membrane structures are one of the main targets for the action of excessive hydrogen peroxide amounts, as well as other reactive oxygen species (ROS). When membranes are damaged, peroxidation of unsaturated fatty acids of membrane lipids occurs. The activity of lipid peroxidation (LPO) is shown by an increase in the formation of malondialdehyde (MDA) (Krishnamurthy and Rathinasabapathi 2013; Demidchik 2015). According to the data obtained, when pine needles are polluted with BrAS emissions, the concentration of MDA increases from 1.5 to 3.0 times as compared with the background values (see Fig. 3). The fact that the increase in LPO is the result of the intense hydrogen peroxide generation is confirmed by the high correlation coefficient calculated between these parameters (r = 0.87, P ≤ 0.05, n = 29).
The next logical step was to elucidate changes in membrane lipids, in particular, changes in the composition of fatty acids (FA) since these compounds play an important role in adaptation and formation of membrane resistance. Differences in the quantitative composition of FAs in pine needles were identified. The largest number was identified in the background territory – 24. In the case of low industrial load they numbered 23; in the case of moderate and high industrial load – 21; and in the case of critical industrial load – 18 (Table 3).
When considering the qualitative fatty acid composition of needle lipids both in the background areas and under the influence of industrial emissions, saturated FAs were identified, among which palmitic (C16: 0) and stearic (C18: 0) acids predominated. Their relative content in the needles of the background tree stands was 12.97% and 8.69%, respectively (Fig. 4).
Under the influence of BrAS emissions, a multidirectional change in the content of saturated fatty acids was revealed. Thus, as the industrial pollution intensified, the amount of palmitic acid in the total lipids of needles increased up to 1.5 times. On the contrary, stearic acid decreased up to 2.9 times. According to a number of researchers (Zhukov 2015), palmitic FA (C16: 0) plays an important role in the protection of plant membranes from the effects of abiotic stress factors. That is probably what determines its increased level. At the same time, a decrease in the level of stearic acid (C18: 0), on the one hand, can be associated with its elongation with the formation of long-chain saturated acids. On the other hand, the decrease can be associated with stearic acid desaturation with stearoyl-ACP ∆9-desaturase with the formation of oleic acid (Schultz et al. 2000; Ramli et al. 2007). Among saturated FAs, the proportion of margaric acid in needle lipids increases as industrial pollution intensifies up to a high level. The amount of this acid with a normal structure (C17:0) increases 1.7–2.3 times; with the cis-configuration (izo-С17: 0) – 1.1–2.2 times. At the same time, when the pollution level is critical, margaric acid is not identified in needle lipids.
When considered the content of long-chain FAs with 20 or more carbon atoms in the chain, an increase in the level of behenic acid (C22:0) by 1.6–2.5 times, and arachidic acid (C20:0) by 1.5 times was revealed. The presence of long-chain FAs in photosynthetic tissues should be noted to be a characteristic feature of gymnosperms, including Pinus sylvestris (Mongrand et al. 2001; Makarenko et al. 2014; Nokhsorov et al. 2019). These FAs are probably irreplaceable participants in many biological processes that cannot proceed with the participation of shorter FAs (Denic, Weissman (2003)). In addition, the increased content of long-chain FAs observed in polluted needles can indicate a decrease in the fluidity of the membrane lipid bilayer (Ivanova et al. 2009). This is probably one of the adaptive mechanisms aimed at regulating membrane microviscosity when exposed to pollutants.
Analysis of the fatty acid composition of needle lipids also revealed significant differences in the amount of short-chain saturated FAs (C12:0 + C14:0 + C15:0) between the background and polluted trees. The sum of these FAs in the background areas was 1.17%. With the pollution load increase, the sum of these FAs also increased reaching the highest values (5.57%) at the high pollution level. The most interesting are the results on the change in the proportion of pentadecanoic (C15:0) acid, which is present in pine needle lipids only in the background areas and at the low pollution level; it probably participates in shoot growth. With a more severe pollution, C15:0 is not identified, but lauric acid with the cis-configuration of double bonds in the structure (izo-C12:0) appears. In addition, a significant increase in the amount of myristic (C14:0) acid in needle lipids of polluted trees should be noted. According to other researchers, the accumulation of short-chain FAs in neutral pine needle lipids occurs when trees adapt to low temperatures and when there is insufficient supply of nutrients (Dorofeeva 2009). The role of short-chain FAs as signaling molecules involved in the gene expression, which regulate cellular functions and maintain homeostasis, was proven (Tan et al. 2014). In addition, there is an assumption about the active participation of C12:0 and C14:0 FAs in the weakening of oxidative stress damage (Zemanova et al. 2015).
Thus, in the composition of pine needle total lipids, the proportion of saturated FAs is 25–34%. The minimum values are found in needles of background area trees, and the maximum values are found in the areas with the high pollution level. According to the researchers (Makarenko et al. 2014), the increase of saturated fatty acids in the total lipids of needles is associated with the rearrangement of the structure of chloroplast membrane. This contributes to a decrease in its elasticity, making the membrane less permeable. This is probably an adaptive response that reduces the intensive input of pollutant elements into the plant organism. At the same time, an increase in the fatty acid saturation can lead to a decrease in the activity of photosystem II due to an increase in the distance between the light-harvesting complex and the reaction center (Mokronosov and Gavrilenko 1992).
The proportion of unsaturated acids decreases from 75% to 66% simultaneously with an increase in the proportion of saturated FAs in polluted needles (see Table 3). Unsaturated FAs are known to add fluidity to cell membranes, which is necessary to maintain their structural and functional state. That is necessary for metabolic processes and ensures the protective functions of pine trees in response to the impact of abiotic stress factors, including industrial pollution (Los’ et al. 2013). The fluidity of cell membranes depends on the ratio of saturated and unsaturated FAs, as well as on the degree of unsaturation of the latter (Los‘ 2001). Our studies revealed a decrease in the unsaturation coefficient (C) of FAs by 15–29% in the needles of polluted trees in comparison with trees on the background territories. The results obtained indicate structural transformations of membranes in the needles of trees under the influence of the aluminum smelter emissions.
The data in Table 3 show that oleic (С18: ω9), linoleic (С18:2ω6), and α-linolenic (С18:3ω3) FAs predominate in the composition of the unsaturated FA fraction. These three components form the basis of mono-, di- and triene classification fractions of unsaturated FAs and largely determine the direction in their quantitative content changes. The amount of oleic and linoleic acids decreases by 29 and 24%, respectively, as industrial pollution intensifies up to the high level. On the contrary, the amount of α-linolenic acid increases by 10% in comparison with the background content. At the critical pollution level, the opposite tendency is revealed: the content of С18:1ω9 and С18:2ω6 fatty acids increases, while the content of С18:3ω3 acid decreases. It is known that α-linolenic acid belongs to the group of triene FAs, which have lower melting points than mono- and diene FAs; that is also important for maintaining the structural and functional stability of plant membranes (Hugly and Somerville 1992; Kodama et al. 1994). At the same time, oleic and linoleic acids serve as substrates for biosynthesis of FAs with a long carbon chain (C ≥ 20) and for Δ5-unsaturated polymethylene FAs in the membranes of assimilation organs of trees, including pine needles (Wolff et al., (1999); Wolff et al., (2001); Mongrand et al. 2001; Sayanova et al. 2007). The tetraene fraction was also identified in the FA composition of pine needle lipids in all the examined SPs. It was only represented by coniferonic acid (C18:4 Δ5,9,12,15), the content of which did not exceed 2.29% of the total FAs. According to the researches (Vetchinnikova et al. 2016), the presence of tetraene FAs in plant glycolipids may indicate the inclusion of biochemical mechanisms of adaptation to the conditions of increased anthropogenic load.
Analysis of the content of unsaturated fatty acids differing in the number of carbon atoms showed that the contribution of these components to the structural organization of total lipids is not the same. C18 FAs are the most abundant in all the surveyed territories; their content varies within 86–89% of the total unsaturated FAs (Fig. 5). The content of C20 FAs is much lower; it varies from 8% in the background areas to 14% in the areas with the high and critical level of industrial pollution. The lowest concentration is typical for C16 FAs. It does not exceed 3% in the background areas and those with the low pollution level. In the areas with the moderate and high pollution level, it does not exceed 1.5%. C16 FAs were not found in the areas with the critical pollution level. Probably, C18 FAs play a decisive role in the structural organization of lipids and in the functional state of cell membranes in general, while C20 FAs are involved in the protection of membranes. They prevent membranes from damage under the conditions of industrial pollution. Other researchers have also reported on the crucial role of C18 unsaturated FAs in plant resistance to unfavorable environmental factors (Demin et al. 2008).
One of the important features characterizing needle lipids is the presence of “relict” ∆5-polymethylene acids (∆5-UPIFA). These fatty acids are characteristic of conifers and some other ancient taxa (Makarenko et al. 2014; Nokhsorov et al. 2019). The ∆5-UPIFA fraction is formed mainly due to pinolenic (C18:3∆5,9,12) and skiadonic (C20:3Δ5,11,14) acids. To a lesser extent, it is formed due to coniferonic (C18:4Δ5,9,12,15) and taxoleic (C18:2Δ5,9) acids. The sum of ∆5-UPIFA was found to be 12.94% of the total FAs in pine needles in the background areas. As industrial pollution intensifies, it increases, reaching 14.05% at the high pollution level. At the critical pollution level, the amount of these FAs decreases to almost the background level. According to the results obtained, the sum of ∆5-polymethylene FAs has a similar change pattern with the content of α-linolenic acid at different levels of industrial pollution. The high content of the sum of ∆5-UPIFA, as well as C18:3ω3, in the lipids of the chloroplast membranes of needles Pinus sylvestris and other coniferous tree species is usually associated with their adaptation to low temperatures (Martz et al. 2006; Torres-Franklin et al. 2009; Roman et al. 2012; Makarenko et al. 2014). Our data on the changes in these acids in the polluted pine needles can probably indicate the occurrence of adaptive reactions to the pressure of low-to-high industrial pollution levels, while a sharp decrease in the acid content under critical pollution indicates the suppression of adaptive reactions.
The biosynthesis of C18 FAs is carried out with the participation of acyl-lipid desaturases (ω9, ω6, ω3), which catalyze the introduction of double bonds into the hydrocarbon chains of oleic (C18:1ω9), linoleic (C18:2ω6) and α-linolenic (C18:3ω3) acids (Nishida and Murata 1996; Los‘ 2001). In this regard, the assessment of the activity of fatty acid desaturases using desaturase ratios (Eqs. 2–4) makes it possible to judge to a certain extent the synthesis mechanisms and the role of the dominant unsaturated FAs in the composition of needle lipids both in the background areas and at different levels of industrial pollution. The values of stearoyl-(SDR), oleyl-(ODR) and linoleyl-(LDR) desaturase ratios calculated for pine needles are given in Table 4.
The minimum values of the SDR ratio are detected in the needles of background pine forests. As the industrial load increases to the high level, the ratio slightly increases by 10%. The genes of acyl-lipid ω9 desaturase, which provides the introduction of the first double bond into C18:0 acid with the formation of oleic acid, are believed to always act at a constant level (Los‘ and Murata 1998). The observed sharp increase in the SDR ratio (by 26% of the background level) at the critical pollution level is apparently due to an increase in the amount of oleic acid and the synthesis of linoleic FA.
The oleil-desaturase ratio (ODR) in the needles of background pine forests is characterized by higher values if compared to the SDR ratio. This indicates an active conversion of oleic acid to linoleic acid by ω6-acyl-lipid desaturase. In the case of industrial pollution, the ODR ratio insignificanlty (less than 7%) differs from the background. That is possibly an indicator of weak dependence of the ω6-desaturase gene on industrial pollution, an indicator of keeping its activity at a constant level.
The linoleyl-desaturase ratio (LDR) in pine needles in the background areas is low. It is associated with a significantly higher proportion of linoleic acid. As the industrial load increases to the high level, LDR increases by 17%. That indicates the expression of the ω3-desaturase genes responsible for the synthesis of α-linolenic acid. As a result, the rate of introduction of the third double bond into α-linolenic acid and its amount increases. At the critical pollution level, LDR decreases to the background value, which indicates suppression of the ω3-acyl-lipid desaturase activity.
Thus, the results obtained indicate that fatty acids of membrane lipids play an important role in the adaptation processes of pine trees during industrial pollution. The changes revealed in the quantitative and qualitative composition of FA lipids of pine needles are aimed at protecting membranes from damage when exposed to the aluminum smelter emissions.
Conclusion
Data on the peculiarities of changes in the fatty acid composition of the total lipids of P. sylvestris needles in the territories polluted by emissions from the large aluminum smelter were obtained. Using the cluster analysis of data on the content of pollutant elements in needles, territories of low, moderate, high and critical levels of pollution, as well as background territories, were identified. It was shown that pollutants initiate the development of oxidative stress in plants and the excessive formation of reactive oxygen species. This is indicated by an increase in the concentration of H2O2 in needles from 1.5 to 4.6 times and an increase in the content of malondialdehyde by 1.5–3.0 times as compared to the background values.
As the industrial pollution intensifies, the total number of FAs in tree needles and the proportion of unsaturated FAs decrease. On the contrary, the fraction of saturated FAs increases. Among saturated FAs, the content of palmitic, myristic, and margaric acids increases. The amount of long-chain (behenic and arachidic) and the amount of short-chain (pentadecanoic, lauric, and myristic) acids increases. The result of these changes is the decrease in the fluidity of the membrane lipid bilayer and the decrease in their permeability. The decrease in the content of unsaturated FAs in needles in the case of pollution caused by industrial emissions is accompanied by the decrease in the unsaturation coefficient, as well as by the change in the ratio of С20, С18, С16 FAs. It was revealed that at the low, moderate and high pollution levels, the content of mono-, di- and tetraenoic acids decreases, and the content of triene acids increases. At the critical pollution level, the opposite tendency was found.
The calculated values of SDR, ODR, and LDR desaturase ratios characterizing the activity of acyl-lipid ω9-, ω6-, ω3-desaturases showed that with the increase in the industrial pollution level from low to high, the activity of all desaturases increases. The activity of oleyl-desaturase, which catalyzes the introduction of the second double bond to oleic acid, increases most significantly. At the critical industrial load level, the activity of ω6 and ω3 desaturases slows down. However, ω9 desaturase, which increases the rate of double bond introduction into stearic acid with the formation of oleic acid, gets a greater activity.
In general, the revealed changes in the FA composition of the total lipids of pine needles indicate the activation of the stabilization mechanisms of membrane lipids in pine needles. They are one of the most important adaptation reactions of Pinus sylvestris in response to the effect of the aluminum smelter emissions.
References
Ammar WB, Nouairi I, Zarrouk M, Jernal F (2008) The effect of cadmium on lipid and fatty acid biosynthesis in tomato leaves. Biologia 63:86–93. https://doi.org/10.2478/s11756-008-0002-6
Arisawa K, Mitsudome H, Yoshida K, Sugimoto S, Ishikawa T, Fujiwara Y, Ichi I (2016) Saturated fatty acid in the phospholipid monolayer contributes to the formation of large lipid droplets. Biochem Biophys Res Commun 480(4):641–647. https://doi.org/10.1016/j.bbrc.2016.10.109
Awang MB, Mikhailova TA, Luangjame J et al. (2007) Sub-Manual on forest vegetation monitoring in EANET. Network Center for EANET, Acid Deposition and Oxidant Research Center (ADORC), Niigata
Bao K, Zaccone C, Tao Y, Wang J, Shen J, Zhang Y (2020) Source apportionment of priority PAHs in 11 lake sediment cores from Songnen Plain, Northeast China. Water Res168:115–158. https://doi.org/10.1016/j.watres.2019.115158
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Botany 91:179–194. https://doi.org/10.1093/aob/mcf118
Boffetta P, Jourenkova N, Gustavsson P (1997) Cancer risk from occupational and environmental exposure to polycyclic aromatic hydrocarbons. Cancer Causes Control 8:444–472
Brennan T, Frenkel C (1977) Involvement of hydrogen peroxide in the regulation of senescence in pear. Plant Physiol 59:411–416. https://doi.org/10.1104/pp.59.3.411
Cartea ME, Migdal M, Galle AM, Pelletier G, Guerche P (1998) Comparison of sense and antisense methodologies for modifying the fatty acid composition of Arabidopsis thaliana oilseed. Plant Sci 136:181–194. https://doi.org/10.1016/S0168-9452(98)00089-2
Catalá A (2006) An overview of lipid peroxidation with emphasis in outer segments of photoreceptors and the chemiluminescence assay. Int J Biochem Cell Biol 38:1482–1495. https://doi.org/10.1016/j.biocel.2006.02.010
Chaffai R, Elhammadi MA, Seybou TN, Tekitek A, Marzouk B, Ferjani E (2007) Altered fatty acid profile of polar lipids in maize seedlings in response to excess copper. J Agron Crop Sci 193(3):207–217. https://doi.org/10.1111/j.1439-037X.2007.00252.x
Cheeseman JM (2007) Hydrogen peroxide and plant stress: a challenging relationship. Plant Stress 1(1):4–15
Christie WW (1993) Preparation of ester derivatives of fatty acids for chromatographic analysis. In: Christie WW (ed) Advances in lipid methodology. Two Oily Press, Dundee. The Scottish Crop Research Institute, Invergowrie, Dundee, Scotland DD2 5DA, pp 69–111
Cools N, De Vos B (2010) Manual on methods and criteria for harmonized sampling, assessment, monitoring and analysis of the effects of air pollution on forests, UNECE, ICP Forests - Hamburg. (http://www.icp-forests.org/Manual.htm)
Demidchik V (2015) Mechanisms of oxidative stress in plants: From classical chemistry to cell biology. Environ Exp Bot 109:212–228. https://doi.org/10.1016/j.envexpbot.2014.06.021
Demin IN, Deryabin AN, Sinkevich MS, Trunova TI (2008) Insertion of cyanobacterial desA gene coding for Δ12-acyl-lipid desaturase increases potato plant resistance to oxidative stress induced by hypothermia. Russian. J Plant Physiol 55(5):639–648
Denic V, Weissman JS (2003) A molecular caliper mechanism for determining very long-chain fatty acid length. Cell 130:663–677. https://doi.org/10.1016/j.cell.2007.06.031
Dorofeeva OS (2009) The effect of borane on the physiological and biochemical parameters of seedlings of Scots pine: Abstract of PhD dissertation. Publishing house of the Karelian Scientific Center, Petrozavodsk. 143 p. (in Russian)
Drazkiewicz M, Tukendorf A, Baszynski T (2003) Age-dependent response of maize leaf segments to cadmium treatment: effect on chlorophyll fluorescence and phytochelatin accumulation. J Plant Physiol 160:247–254. https://doi.org/10.1078/0176-1617-00558
Forest Encyclopedia (1985) / Ed. Vorobiev GI. Publishing house Soviet Encyclopedia, Moscow. (in Russian)
Foyer CH, Noctor G (2015) Defining robust redox signalling within the context of the plant cell. Plant Cell Environ 38(2):239. https://doi.org/10.1111/pce.12487
Gorshkov AG (2008) Determination of polycyclic aromatic hydrocarbons in the needles of a Scotch pine (Pinus sylvestris L.) a biomonitor of atmospheric pollution. J Anal Chem 63(8):805–811. https://doi.org/10.1134/S1061934808080169
Hou Q, Ufer G, Bartels D (2016) Lipid signaling in plant responses to abiotic stress. Plant Cell Environ 39(5):1029–1048. https://doi.org/10.1111/pce.12666
Hugly S, Somerville CR (1992) A role of membrane lipid polyunsaturation in chloroplast biogenesis at low temperature. Plant Physiol 99(1):197–202. https://doi.org/10.1104/pp.99.1.197
Il’inskaya LI, Ozeretskovskaya OL (1998) Fatty acids lipoxygenation products as signal molecules inducing the resistance of plants (review). Appl Biochem Microbiol 34(5):425–436
Ivanova TV, Myasoedov NA, Pchelkin VP, Tsydendambaev VD, Vereshchagin AG (2009) Increased content of very-long-chain fatty acids in the lipids of halophyte vegetative organs. Russian J Plant Physiol 56(6):787–794
Jemal F, Zarrouk M, Ghorbal MH (2000) Effect of cadmium on lipid composition of pepper. Biochem Soc Trans 28:907–909. https://doi.org/10.1042/bst0280907
Kasimov NS, Bityukova VR, Kislov AV et al. (2012) Ecogeochemical problems of large cities. Razved okhrana nedr 7:8–13. in Russian
Kobayashi K, Wada H (2016) Role of lipids in chloroplast biogenesis. In: Yuki N, Yonghua L-B (eds) Lipids in plant and algae development. Springer International Publishing, Switzerland, pp 103–125
Kodama H, Hamada T, Horiguchi G, Nishimura M, Iba K (1994) Genetic enhancement of cold tolerance by expression of a gene for chloroplast (omega)-3 fatty acid desaturase in transgenic tobacco. Plant Physiol 105(2):601–605. https://doi.org/10.1104/pp.105.2.601
Kondratenko EP, Soboleva OM, Sukhikh AS (2017) The influence of the microwave electromagnetic field on the fatty acid composition of Hordeum sativum seedlings. Himiya rastitel’nogo syr’ya 3:93–99. https://doi.org/10.14258/jcprm.2017031792. in Russian
Krishnamurthy A, Rathinasabapathi B (2013) Oxidative stress tolerance in plants. Plant Signal Behav 8(10). https://doi.org/10.4161/psb.25761
Kumar GNM, Knowles NR (1993) Changes in lipid peroxidation and lypolytic and free-radical scavenging enzyme activities during aging and sprouting of potato (Solanum tuberosum). Plant Physiol 102:115–124. https://doi.org/10.1104/pp.102.1.115
Los‘ DA (2001) Structure, regulation of expression and functioning of fatty acid desaturases. Uspekhi biologicheskoj himii 41:163–198. in Russian
Los‘ DA (2005) Molecular mechanisms of cold tolerance of plants. Vestnik Rossiiskoi Akademii Nauk 4:338–345. in Russian
Los‘ DA, Mironov KS, Allakhverdiev SI (2013) Regulatory role of membrane fluidity in gene expression and physiological functions. Ptotosynthesis Res 116(2):489–509. https://doi.org/10.1007/s11120-013-9823-4
Los‘ DA, Murata N (1998) Structure and expression of fatty acid desaturase. Biochem Biophys Acta 1394(1):3–15. https://doi.org/10.1016/S0005-2760(98)00091-5
Lyons JM (1973) Chilling injury in plants. Annu Rev Plant Physiol 24:445–466
Makarenko SP, Konenkina TA, Suvorova GG, Oskorbina MV (2014) Seasonal changes in the fatty acid composition of Pinus sylvestris needle lipids. Russian. J Plant Physiol 61(1):119–123. https://doi.org/10.7868/80015330313050102
Martz F, Kiviniemi S, Pavla TE, Sutinen M-L (2006) Contribution of omega-3 fatty acid desaturase and 3-ketoacyl-ACP synthase II (KASII) genes in the modulation of glycerolipid fatty acid composition during cold acclimation in birch leaves. J Exp Bot 57:897–909. https://doi.org/10.1093/jxb/erj075
Mazliak P (1994) Desaturation processes in fatty acid and acyl lipid biosynthesis. J Plant Physiol 143:399–406. https://doi.org/10.1016/S0176-1617(11)81799-2
McKillup S (2011) Statistics explained. An introductory guide for life scientists. Cambridge University Press, University Printing House, Cambridge CB2 8BS, United Kingdom
Mikhailova TA, Afanasieva LV, Kalugina OV, Shergina OV, Taranenko EN (2017) Changes in nutrition and pigment complex in pine (Pinus sylvestris L.) needles under technogenic pollution in Irkutsk region. Russia. J For Res-JPN 6:386–392. https://doi.org/10.1080/13416979.2017.1386020
Moellering ER, Muthan B, Benning C (2010) Freezing tolerance in plants requires lipid remodeling at the outer chloroplast membrane. Science 330:226–228. https://doi.org/10.1126/science.11918003
Mokronosov AT, Gavrilenko VF (1992) Photosynthesis. Physiological, environmental and biochemical aspects. Publishing House of Moscow State University, Moscow (in Russian)
Mongrand S, Badoc A, Patouille B, Lacomblez C, Chavent M, Cassagne C, Bessoule JJ (2001) Taxonomy of gymnospermae: multivariate analyses of leaf fatty acid composition. Phytochemistry 58:101–115. https://doi.org/10.1016/S0031-9422(01)00139-X
Munnik T, Testerink C (2009) Plant phospholipid signaling: “in a nutshell”. J Lipid Res 50:260–265. https://doi.org/10.1194/jlr.R800098-JLR200
Nesterov VN, Rozentsvet OA, Murzaeva SV (2009) Changes in lipid composition in the tissues of fresh-water plant Hydrilla verticillata induced by accumulation and elimination of heavy metals. Russian. J Plant Physiol 56(1):85–93
Nishida I, Murata N (1996) Chilling sensitivity in plants and Cyanobacteria: the crucial contribution of membrane lipids. Annu Rev Plant Physiol Plant Mol Biol 47:541–568. https://doi.org/10.1146/annurev.arplant.47.1.541
Nokhsorov VV, Dudareva LV, Petrov KA (2019) Content and composition of lipids and their fatty acids in needles of Pinus sylvestris L. and Picea obovata Ledeb. upon cold hardening in the cryolithozone of Yakutia. Russian. J Plant Physiol 66(4):548–555. https://doi.org/10.1134/S0015330319040109
Nouairi I, Ben Ammar W, Ben Youssef NB, Daoud D, Ghorbal MH, Zarrouk M (2006) Comparative study of cadmium effects on membrane lipid composition of Brassica juncea and Brassica napus leaves. Plant Sci 170:511–519. https://doi.org/10.1016/j.plantsci.2005.10.003
Ohlrogge J, Browse J (1995) Lipid biosynthesis. Plant Cell 7:957–970
Okazaki Y, Otsuki H, Narisawa T et al. (2013) A new class of plant lipid is essential for protection against phosphorus depletion. Nat Commun 4:1510. https://doi.org/10.1038/ncomms2512
Okazaki Y, Saito K (2014) Roles of lipids as signaling molecules and mitigators during stress response in plants. Plant J 79:584–596. https://doi.org/10.1111/tpj.12556
Ouariti O, Boussama N, Zarrouk M, Cherif A, Ghorbal MH (1997) Cadmium- and copper-induced changes in tomato membrane lipids. Phytochemistry 45(7):1343–1350. https://doi.org/10.1016/S0031-9422(97)00159-3
Proidakova OA, Vasileva IE (2010) Method to improve schemes of sample preparation and atomic-absorption analysis of geochemical samples. Inorg Mater 46(14):1503–1512. https://doi.org/10.1134/S0020168510140062
Ramli US, Baker DS, Quant PA, Harwood JL (2007) Control analysis of lipid biosynthesis in tissue cultures from oil crops shows that flux control is shared between fatty acid synthesis and lipid assembly. Biochem J 364(2):393–401. https://doi.org/10.1042/bj20010203
Ramos PR, Gieseg SP, Schuster B, Esterbauer H (1995) Effect of temperature and phase transition on oxidation resistance of low density lipoprotein. J Lipid Res 36(10):2113–2128
Roman A, Andreu V, Hernandez ML, Lagunas B, Picorel R, Martinez-Rivas JM, Alfonso M (2012) Contribution of the different omega-3 fatty acid desaturase genes to the cold response in soybean. J Exp Bot 63:4973–4982. https://doi.org/10.1093/jxb/ers174
Romanova IM, Zhivetev MA, Dudareva LV, Graskova IA (2016) Variation of fatty acid composition and activity of acyl-lipid desaturases of Pinus sylvestris L. needles, growing in the Irkutsk region. Himiya rastitel’nogo syr’ya 2:61–66. https://doi.org/10.14258/jcprm.201602732. in Russian
Rozhkov AS, Mikhailova TA (1993) The effect of fluorine-containing emissions on conifers. Springer, Berlin Heidelberg
Sanina NB, Proidakova OA (2006) Heavy metals in soil of the Baikal biosphere reserve (in connection with degradation of fir forests of the northern macroslope of Khamar-Daban Range). Chin J Geochem 25:191–192. https://doi.org/10.1007/BF02840114
Sayanova O, Haslam R, Venegas Caleron M, Napier JA(2007) Cloning and characterization of unusual fatty acid desaturases from Anemone leveillei: identification of an acyl-coenzyme A C20∆5-desaturase responsible for the synthesis of sciadonic acid Plant Physiol 144:445–467. https://doi.org/10.1104/pp.107.098202
Scandalios JG (2002) The rise of ROS. Trends Biochem Sci 27(9):483–486. https://doi.org/10.1016/s0968-0004(02)02170-9
Schultz DJ, Suh MC, Ohlrogge JB (2000) Stearoyl-acyl carrier protein and unusual acyl-acyl carrier protein desaturase activities are differentially influenced by ferredoxin. Plant Physiol 124:681–692. https://doi.org/10.1104/pp.124.2.681
Singh R, Parihar P, Prasad SM (2018) Simultaneous exposure of sulphur and calcium hinder As toxicity: Up-regulation of growth, mineral nutrients uptake and antioxidants system. Ecotoxicol Environ Saf 161:318–331. https://doi.org/10.1016/j.ecoenv.2018.05.060
Sopin AI, Trunova TI (1991) The dynamics of phospholipids and fatty acids in etiolated winter wheat seedlings during hardening to frost. Fiziologiya rastenij 38(1):142–149.
State report “On the State and Environmental Protection of the Irkutsk Region in 2018” (2019). Irkutsk, LLC (in Russian)
Suvorova G, Korzukhin M, Ivanova M (2017) Influence of environmental factors on photosynthesis of three coniferous species. Ann Res Rev Biol 12(3):1–14. https://doi.org/10.9734/ARRB/2017/31526
Taarit MB, Msaada K, Hosni K, Marzouk B (2010) Changes in fatty acid and essential oil composition of sage (Salvia officinalis L.) leaves under NaCl stress. Food Chem 119:951–956. https://doi.org/10.1016/j.foodchem.2009.07.055
Tan J, McKenzie C, Potamitis M, Thorburn AN, Mackay CR, Macia L (2014) The role of short-chain fatty acids in health and disease. Adv Immunol 121(1):91–119. https://doi.org/10.1016/B978-0-12-800100-4.00003-9
Tarchevsky IA (2002) Plant Cell Signaling Systems. Nauka, Moscow
Torres-Franklin M-L, Repellin A, Huynh V-B et al. (2009) Omega-3 fatty acid desaturase (FAD3, FAD7, FAD8) gene expression and linolenic acid content in cowpea leaves submitted to drought and after rehydration. Environ Exp Bot 65:162–169. https://doi.org/10.1016/j.envexpbot.2008.12.010
Tukendorf A (1993) The response of spinach plants to excess of copper and cadmium. Photosynthetica 28:573–575
Vetchinnikova LV, Androsova VI, Morozova IV et al. (2016) Patterns of fatty acids composition of lipids in the tallus of a lichen Hypogimnia physodes (L.) NYL. Sciences of Europe 7-1(7):4–9 (in Russian)
Vu VT, Lee BK, Kim JT, Lee CH, Kim IH (2011) Assessment of carcinogenic risk due to inhalation of polycyclic aromatic hydrocarbons in PM10 from an industrial city: a Korean case-study. J Hazard Mater 189:349–356
Wang X, Devaiah SP, Zhang W, Welti R (2006) Signaling functions of phosphatidic acid. Prog Lipid Res 45:250–278. https://doi.org/10.1016/j.plipres.2006.01.005
Wang Z, Benning C (2012) Chloroplast lipid synthesis and lipid trafficking through ER-plastid membrane contact sites. Biochem Soc Trans 40(2):457–463. https://doi.org/10.1042/BST20110752
Weil M, Schaub H (1999) Influence of exhaust gas and ozone on extracellular peroxidase activity of Helianthus annuus L. leaves. J Plant Physiol 154:523–528
Wolff RL, Christie WW (2002) Structure, practical sources (gymnosperm seeds), gas-liquid chromatographic data (equivalent chain lengths), and mass spectrometric characteristics of all-cis ∆5-olefinic acids. Eur J Lipid Sci Technol 104:234–244. 0931-5985/2002/0404-0234
Wolff RL, Christie WW, Pedrono F et al. (1999) Delta5-olefinic acids in the seed lipids from four Ephedra species and their distribution between the alpha and beta positions of triacylglycerols. Characteristics common to coniferophytes and cycadophytes. Lipids 58:101–115
Wolff RL, Lavialle O, Pedrono F et al. (2001) Fatty acid composition of Pinaceae as taxonomic markers. Lipids 36:439–451. https://doi.org/10.1007/s11745-001-0741-5
Yoshida S, Sakai A (1974) Phospholipid degradation in frozen plant cells associated with freezing injury. Plant Physiol 53:509–511
Zemanova V, Pavlik MK, Kyjaková P, Pavlikova D (2015) Fatty acid profiles of ecotypes of hyperaccumulator Noccaea caerulescens growing under cadmium stress. J Plant Physiol 180(1):27–34. https://doi.org/10.1016/j.jplph.2015.02.012
Zhang J, Kirkham MB (1994) Drought-stress induced changes in activities of superoxide dismutase, catalase, and peroxidase in Wheat species. Plant Cell Physiol 35(5):785–791. https://doi.org/10.1093/oxfordjournals.pcp.a078658
Zhukov AV (2015) Palmitic acid and its role in the structure and plant cell membranes. Russian J Plant Physiol 62(5):706–713. https://doi.org/10.7868/S001533031505019X
Zielinska D, Radecka H, Radeski J (2000) Contribution of membrane surface charge in the interaction of lead and tin derivatives with model lipid membrane. Chemosphere 40(3):327–330
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. OVK: conceptualization, methodology, writing original draft, visualization, project administration, funding acquisition, TAM: reviewing and editing, LVA: validation, investigation, VVG: investigation, MVI: data curation, formal analysis. The first draft of the manuscript was written by OVK and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The research was carried out with the financial support of the Russian Foundation for Basic Research and the Government of the Irkutsk Region within the framework of the scientific project No. 20-44-380009.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
The research article ensures objectivity and transparency in research and secures that accepted principles of ethical and professional conduct have been followed. This original research article does not contain any studies with human participants and animals performed by any of the authors.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kalugina, O.V., Mikhailova, T.A., Afanasyeva, L.V. et al. Changes in the fatty acid composition of pine needle lipids under the aluminum smelter emissions. Ecotoxicology 30, 2083–2095 (2021). https://doi.org/10.1007/s10646-021-02479-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-021-02479-2