Abstract
The effect of nine insecticides used in tomato production was evaluated on adults of two Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae) populations from Rive and Afonso Cláudio, Espírito Santo State, Brazil. The experiment was developed in an acclimatized chamber at 25 ± 1°C, 70 ± 10% relative humidity and 14 h photophase. Eggs of Anagasta kuehniella (Lepidoptera: Pyralidae), previously immersed in insecticides solutions were offered to females of both T. pretiosum populations. Bacillus thuringiensis, lufenuron and triflumuron had lowest negative effects on parasitism and viability of individuals of these populations; however, abamectin and pyrethroids (betacyflurin 50 and 125 g/l and esfenvalerate) insecticides reduced parasitism rates. T. pretiosum emerged from A. kuehniella eggs treated with esfenvalerate but were not able to parasitize non treated eggs of this host. B. thuringiensis, lufenuron and triflumuron may be used in integrated pest management programs to control tomato pests, because they have moderated negative effect on parasitoid wasps.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Parasitoids of the genus Trichogramma are important for the biological control in fruit (Pratissoli et al. 2004a, 2005) forest (Oliveira et al. 2000; Soares et al. 2007) and tomato (Miranda et al. 1998) plantations including Tuta absoluta (Meyrick) (Lepidoptera: Pyralidae) in Brazil (Pratissoli and Parra 2001). The chemical control of that tomato pest (Pratissoli et al. 2003) and plant resistance (Leite et al. 2001) are used. Natural enemies of the genus Trichogramma are an alternative for pest control and to reduce populations of T. absoluta, the most important pest of this culture in Brazil, and insecticide applications in the tomato culture (Pratissoli and Parra 2001). However, chemical control is, still, largely used if the biological control fails and therefore is necessary studies about the associated use of insecticides with parasitoids (Pratissoli et al. 2003) and predators (Zanuncio et al. 1998, 2003, 2005).
Methods to test the side effects of pesticides have been developed as a function of the beneficial arthropods and pesticides studied (Desneux et al. 2007). Negative effects of insecticides on populations of the parasitoid Trichogramma have been reported (Suh et al. 2000; Brunner et al. 2001; Takada et al. 2001; Vieira et al. 2001), whereas some studies show that insecticides may increase the performance of natural enemies (Komeza et al. 2001; Rafalimanana et al. 2002; Delpuech et al. 2005).
Pesticides may interfere with the feeding behavior by repellent, feeding inhibitor, or reducing olfactory capacity effects (Desneux et al. 2007). Mortality frequently is the only effect that insecticides are screened for, while more conspicuous sublethal effects in beneficial insects, such as altered behavior, reduced reproduction, and reduced longevity, are overlooked. However, studies showed that sublethal effects can severely reduce the performance of biological control agents (Stapel et al. 2000; Desneux et al. 2007).
Insects are affected in different ways when exposed to insecticides and it is important to assess the impact of these chemicals on parasitoid species for its use in integrate pest management programs (Elzen et al. 2000; Hill and Foster 2000; Zehnder et al. 2007). Many organisms react differently when in contact with certain compounds what makes necessary to understand the impact of insecticides on parasitoids to preserve these biological control agents (Elzen et al. 2000; Hill and Foster 2000). In this sense, some insecticides have residual effects on natural enemies affecting the reproductive potential of their offspring (Godoy et al. 2004; Jenkins and Isaacs 2007; Moscardini et al. 2008). Thus, the objective of this study was to determine the effect of nine insecticides, used in the control of T. absoluta in tomato plantations, on the emergence and parasitic efficiency of two populations of the parasitoid Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae) and verify the effect on the second generation of parasitoids.
Methods
The insecticides: abamectin (1.8%; Syngenta Proteção de Cultivos Ltda; São Paulo; Brazil), Bacillus thuringiensis (25,000 IU/mg; Syngenta Proteção de Cultivos Ltda; São Paulo; Brazil), betacyflurin (50 g/l and 125 g/l; Bayer S.A.; São Paulo; Brazil), esfenvalerate (25 g/l; Sumitomo Chemical do Brasil Repres. Ltda; São Paulo; Brazil), lufenuron (50 g/l; Syngenta Proteção de Cultivos Ltda; São Paulo; Brazil), methoxyfenozide (240 g/l; Dow AgroSciences Industrial Ltda; São Paulo; Brazil), tebufenozide (240 g/l; Dow AgroSciences Industrial Ltda; São Paulo; Brazil) and triflumuron (250 g/kg; Bayer S.A.; São Paulo; Brazil) (numbers in parentheses is concentration of active ingredient in commercial product) were evaluated on two T. pretiosum populations. Individuals of these two populations were collected in tomato crops in Rive and Afonso Cláudio, Espírito Santo State, Brazil. The populations were obtained from the different localities. Afonso Cláudio has extensive tomato plantations and its T. pretiosum population is exposed to more insecticides than the Rive’s population. The bioassays were conducted with recently emerged females from two T. pretiosum populations of the same generation. The control treatment had only distilled water. The insecticides evaluated were those used to control tomato crop pests. These are the most used products to control tomato crop pests in Espírito Santo State according to the “Research Institute and Technical Attendance and Rural Extension (INCAPER)”.
Insects rearing
Host
Anagasta kuehniella (Zeller) (Lepidoptera: Pyralidae) was chosen as host because its eggs have been used for mass rearing and field release of parasitoid in Brazil. This host is easy to rear and to multiply the parasitoids for mass release in Brazil. A. kuehniella was reared with a diet of integral wheat (67%) and corn (30%) flours and beer yeast (3%). Eggs of A. kuehniella (0.4 g) were randomly distributed over the diet. Adults of A. kuehniella were daily collected during 5 days with a vacuum cleaner and transferred to PVC tubes (diameter: 200 mm, and length: 25 cm). This rearing system was maintained in an acclimatized chamber (25 ± 1°C, 70 ± 10% relative humidity (rh) and 14:10 L:D).
Parasitoid
Both T. pretiosum populations were collected in commercial tomato plantations using 8.0 × 2.5 cm cardboard strips with 5 cm2 central area containing sterile A. kuehniella eggs previously exposed to ultraviolet light for 50 min. Each population was multiplied and maintained with sterile A. kuehniella eggs glued onto blue rectangle cardboards (2.5 × 10 cm) with 10% Arabic gum in an acclimatized chamber (25 ± 1°C, 70 ± 10% rh and 14:10 L:D) in glass containers (14 × 7 cm) containing honey droplets (as food). These containers were covered with microperforated PVC film.
Experimental procedure
First generation
From 25 pairs of each T. pretiosum population up to 24 h after emergence, females were individualized in tubes (3.0 × 0.5 cm) with a honey droplet (as food) in its internal wall. Forty sterile A. kuehniella eggs (50 min exposed to germicide lamp) were glued on a blue cardboard strip (2.5 × 0.5 cm) and immersed for 5 s in solutions of each insecticide in the commercial concentrations indicate by the manufacturer for the control of T. absoluta in tomato plantations and maintained at room temperature for 1 h to allow drying. Control eggs were immersed in distilled water. The treatment cardboard strips were then exposed to parasitism by T. pretiosum in tubes (3.0 × 0.5 cm) for 24 h in an acclimatized chamber (25 ± 1°C, 70 ± 10% rh and 14:10 L:D). After this period, the cardboards were transferred to glasses tubes (2.5 × 10 cm) and kept in an acclimatized chamber until emergence of adults.
Second generation
Twenty-five females of T. pretiosum obtained from the eggs exposed to the different insecticides were individually placed into the tubes (3.0 × 0.5 cm) and fed with a honey droplet. One cardboard (2.5 × 0.5 cm) with forty sterilized A. kuehniella eggs was offered per female parasitoid (for a 24 h period).
Statistical analysis
The parameters evaluated were percentage of parasitism and emergence in both generations of T. pretiosum populations. Data were transformed to arc sin\( \sqrt {x + 1}, \) submitted to variance analysis, and means were compared using a Scott–Knott test at 5% probability (Scott and Knott 1974).
Results
Impact on the first generation
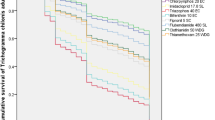
The percentage of parasitism of A. kuehniella by T. pretiosum Rive population was similar in untreated control and with use of B. thuringiensis, lufenuron and triflumuron insecticides. The methoxifenozide and tebufenozide significantly reduced the percentage of parasitism by about 30%, while abamectin, betacyflutin (50 and 125 g/l) and the esfenvalerate insecticides drastically reduced the parasitism rate of this population (Table 1). The parasitism percentage on Afonso Cláudio population was similar to the Rive population in the treatments using esfenvalerate, lufenuron, methoxifenozide and triflumuron and different to the abamectin, B. thuringiensis, betacyflutrin and tebufenozide insecticides. The Rive population showed higher parasitism than Afonso Cláudio population in eggs without treatment with insecticide (Table 1).
The percentage of emergence of T. pretiosum from Afonso Cláudio population was lower with esfenvalerate, whereas Rive population presented lower values with abamectin. The results of Rive population using B. thuringiensis were similar to those of the control. The percentage of emergence of adults from Rive population was lower with abamectin, beacyflutrin (125 g/l), methoxifenozide and triflumuron than those of Afonso Cláudio population. However, this population had lower emergence of adults than that of Rive when betacyflutrin (50 g/l) was used (Table 2).
Impact on the second generation
The population from Afonso Cláudio had lower parasitism with the insecticides abamectin, betacyflutrin, methoxifenozide, tebufenozide and triflumuron in the second generation when compared with control group. Similar results were found for the Rive’s population exposed to abamectin and betacyflutrin (125 g/l) such as found with the abamectin and betacyflutrin (125 g/l) for the population of Rive. The parasitism rate of this population was lower with the methoxifenozide, tebufenozide, triflumuron and the control than those of the Rive population in the second generation (Table 3).
The emergence of individuals of both T. pretiosum populations was not affected by the insecticides in the first generation, except for the esfenvalerate that prevented parasitism in the first generation and for that reason had no emergence of adults in the second generation (Table 4).
Discussion
The results showed that the parasitic efficiency and adult emergence of T. pretiosum were affected for the insecticide used and for the population origin of the parasitoid. Within the insecticides studied, pyrethoids and abamectin decrease the parasitism and adult emergence in both generations of the parasitoid. The insecticides acting as growth regulator and B. thuringiensis can be used in integrated pest management programs, since they have lowest effect on the natural enemy T. pretiosum.
The reduced parasitism of individuals of both T. pretiosum populations with the pyrethroids (betacyflutrin (50 and 125 g/l) and esfenvalerate) and abamectin might have been caused by the high toxicity of these products and their repellent action on T. pretiosum females such as reported for other parasitoids (Jacobs et al. 1984; Singh and Varma 1986), since parasitoids can be olfactory orientation affected by insecticides resulting in a low capacity to found their hosts (Desneux et al. 2004a).
A higher mortality of T. pretiosum adults of populations from Rive and Afonso Cláudio with the pyrethroids and abamectin can also explain the lower parasitism by individuals of these populations. The high mortality of Trichogramma cacoeciae Marchal and T. pretiosum was also reported with insecticides of these chemical groups (Youssef et al. 2004; Rocha and Carvalho 2004), and pyrethroids are more toxic in case of aphid parasitoids when compared with carbamate and carbamyltriazole (Desneux et al. 2004b). The parasitism rate of individuals (T. pretiosum) from Rive’s population was similar with the growth regulators lufenuron and triflumuron, while it was higher with lufenuron in case of Afonso Cláudio population. It shows that these growth regulators can be used in integrated management programs of Lepidoptera pests aiming to conserve Trichogramma species (Suh et al. 2000; Brunner et al. 2001; Hewa-Kapuge et al. 2003; Pratissoli et al. 2004b, c; Moura et al. 2005) The moderate reduction on parasitism rate for Rive’s population of this parasitoid with the methoxifenozide and tebufenozide shows that these insecticides could be used, in integrated pest management programs, but pending restrictions rules.
The higher impact of some insecticides on the emergence of T. pretiosum from Rive than those of Afonso Cláudio population corroborates reports of biological differences among Trichogramma species populations, which can be related to biotic (adaptability and intrinsic capacity of the population in a specific environment) and abiotic (environmental conditions) factors of their origin place (Pratissoli et al. 2003; Moura et al. 2005). Thus, the population from Afonso Cláudio may be resistant or tolerant to insecticides (Croft 1990; Li et al. 2007), since this region has large tomato plantations resulting in a higher use of insecticides than Rive region. In addition difference between the two populations can be also due to stimulus-independent behavioristic resistance as reported in some insects by Georghiou (1972).
Both populations of T. pretiosum showed different responses to the insecticides. The Rive’s population had the parasitism more affected negatively, although the Afonso Cláudio population had presented lower parasitism in the control group. These findings corroborates that different populations can be affected in different ways (Pratissoli and Parra 2001) suggesting that many parasitoid’s populations should be used in test of insecticide toxicity.
The two pyrethroid insecticides betacyflutrin and esfenvalerate (beside abamectin) presented great reduction on the parasitism rate and the percentage of emergence of individuals of both T. pretiosum populations, especially those from Rive. B. thuringiensis and the growth regulators (lufenuron and triflumuron) presented the lowest reduction on parasitism and percentage of emergence. It demonstrates that these insecticides can be used in integrated programs of pest management in the tomato crops. However these findings should be tested in field conditions to assess the selectivity of these chemicals into more realistic conditions (in field conditions).
References
Brunner JF, Dunley JE, Doerr MD, Beers EH (2001) Effect of pesticides on Colpoclypeus florus (Hymenoptera: Eulophidae) and Trichogramma platneri (Hymenoptera: Trichogrammatidae), parasitoids of leafrollers in Washington. J Econ Entomol 94:1075–1084
Croft BA (1990) Arthropod biological control agents and pesticides. Wiley, New York, p 723
Delpuech JM, Bardon C, Boulétreau M (2005) Increase of the behavioral response to kairomones by the parasitoid wasp Leptopilina heterotoma surviving insecticides. Arch Environ Contam Toxicol 49:186–191. doi:10.1007/s00244-004-0158-1
Desneux N, Pham-Delegue MH, Kaiser L (2004a) Effects of sublethal and lethal doses of lambda-cyhalothrin on oviposition experience and host searching behaviour of a parasitic wasp, Aphidius ervi. Pest Manag Sci 60:381–389. doi:10.1002/ps.822
Desneux N, Rafalimanana H, Kaiser L (2004b) Dose–response relationship in lethal and behavioural effects of different insecticides on the parasitic wasp Aphidius ervi. Chemosphere 54:619–627. doi:10.1016/j.chemosphere.2003.09.007
Desneux N, Decourtye A, Delpuech JM (2007) The sublethal effects of pesticides on beneficial arthropods. Annu Rev Entomol 52:81–106. doi:10.1146/annurev.ento.52.110405.091440
Elzen GW, Maldonado SN, Rojas MG (2000) Lethal and sublethal effects of selected insecticides and an insect growth regulator on the boll weevil (Coleoptera: Curculionidae) ectoparasitoid Catolaccus grandis (Hymenoptera: Pteromalidae). J Econ Entomol 93:300–303
Georghiou GP (1972) The evolution of resistance to pesticides. Annu Rev Ecol Syst 3:133–168. doi:10.1146/annurev.es.03.110172.001025
Godoy MS, Carvalho GA, Moraes JC, Júnior MG, Morais AA, Cosme LV (2004) Seletividade de inseticidas utilizados na cultura dos citros para ovos e larvas de Chrysoperla externa (Hagen) (Neuroptera: Chrysopidae). Neotrop Entomol 33:639–646
Hewa-Kapuge S, McDougall S, Hoffmann AA (2003) Effects of methoxyfenozide, indoxacarb, and other insecticides on the beneficial egg parasitoid Trichogramma nr. brassicae (Hymenoptera: Trichogrammatidae) under laboratory and field conditions. J Econ Entomol 96:1083–1090
Hill TA, Foster RE (2000) Effect of insecticides on the diamondback moth (Lepidoptera: Plutellidae) and its parasitoid Diadegma insulare (Hymenoptera: Ichneumonidae). J Econ Entomol 93:763–768
Jacobs RJ, Kouskoleskas CA, Gross HR Jr (1984) Responses of Trichogramma pretiosum (Hymenoptera: Trichogrammatidae) to residues of permethrin and endosulfan. Environ Entomol 13:355–358
Jenkins PE, Isaacs R (2007) Reduced-risk insecticides for control of grape berry moth (Lepidoptera: Tortricidae) and conservation of natural enemies. J Econ Entomol 100:855–865. doi:10.1603/0022-0493(2007)100[855:RIFCOG]2.0.CO;2
Komeza N, Fouillet P, Boulétreau M, Delpuech JM (2001) Modification, by the insecticide chlorpyrifos, of the behavioral response to kairomones of a Drosophila parasitoid, Leptopilina boulardi. Arch Environ Contam Toxicol 41:436–442. doi:10.1007/s002440010269
Leite GLD, Picanço M, Guedes RNC, Zanuncio JC (2001) Role of plant age in the resistance of Lycopersicon hirsutum f. glabratum to the tomato leafminer Tuta absoluta (Lepidoptera: Gelechiidae). Sci Hortic (Amsterdam) 89:103–113. doi:10.1016/S0304-4238(00)00224-7
Li XC, Schuler MA, Berenbaum MR (2007) Molecular mechanisms of metabolic resistance to synthetic and natural xenobiotics. Annu Rev Entomol 52:231–253. doi:10.1146/annurev.ento.51.110104.151104
Miranda MMM, Picanço M, Zanuncio JC, Guedes RNC (1998) Ecological life table of Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Biocontrol Sci Technol 8:597–606. doi:10.1080/09583159830117
Moscardini VF, Moura AP, Carvalho GA, Lasmar O (2008) Efeito residual de inseticidas sintéticos sobre Trichogramma pretiosum Riley (Hym. Trichogrammatidae) em diferentes gerações. Acta Sci 30:177–182
Moura AP, Carvalho GA, Rigitano RLO (2005) Toxicidade de inseticidas utilizados na cultura do tomateiro a Trichogramma pretiosum. Pesquisa Agropecu Bras 40:203–210. doi:10.1590/S0100-204X2005000300002
Oliveira HN, Zanuncio JC, Pratissoli D, Cruz I (2000) Parasitism rate and viability of Trichogramma maxacalii (Hym.: Trichogrammatidae) parasitoid of the Eucalyptus defoliator Euselasia apisaon (Lep.: Riodinidae), on eggs of Anagasta kuehniella (Lep.: Pyralidae). For Ecol Manage 130:1–6. doi:10.1016/S0378-1127(99)00172-3
Pratissoli D, Parra JRP (2001) Seleção de linhagens de Trichogramma pretiosum para o controle das traças Tuta absoluta e Phthorimaea operculella. Neotrop Entomol 30:277–282. doi:10.1590/S1519-566X2001000200011
Pratissoli D, Fornazier MJ, Holtz AM, Gonçalves JR, Chioramital AB, Zago HB (2003) Ocorrência de Trichogramma pretiosum em áreas comerciais de tomate, no Espírito Santo, em regiões de diferentes altitudes. Hortic Bras 21:73–76
Pratissoli D, Thuler RT, Pereira FF, Reis EF, Ferreira AT (2004a) Ação transovariana de lufenuron (50 g/l) sobre adultos de Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae) e seu efeito sobre o parasitóide de ovos Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae). Cienc Agrotecnologia 28:1–9
Pratissoli D, Oliveira HN, Gonçalves JR, Zanuncio JC, Holtz AM (2004b) Changes in biological characteristics of Trichogramma pretiosum (Hym.: Trichogrammatidae) reared on eggs of Anagasta kuehniella (Lep.: Pyralidae) for 23 generations. Biocontrol Sci Technol 14:313–319. doi:10.1080/09583150310001639196
Pratissoli D, Fernandes OA, Zanuncio JC, Pastori PL (2004c) Fertility life table of Trichogramma pretiosum and Trichogramma acacioi (Hymenoptera: Trichogrammatidae) on Sitotroga cerealella (Lepidoptera: Gelechiidae) eggs at different constant temperatures. Ann Entomol Soc Am 97:729–731. doi:10.1603/0013-8746(2004)097[0729:FLTOTP]2.0.CO;2
Pratissoli D, Zanuncio JC, Vianna UR, Andrade JS, Zanotti LCM, Silva AF (2005) Biological characteristics of Trichogramma pretiosum and Trichogramma acacioi (Hym.: Trichogrammatidae), parasitoids of the avocado defoliator Nipteria panacea (Lep.: Geometridae), on eggs of Anagasta kuehniella (Lep.: Pyralidae). Braz Arch Biol Technol 48:7–13
Rafalimanana H, Kaiser L, Delpuech JM (2002) Stimulating effects of the insecticide chlorpyrifos on host searching and infestation efficacy of a parasitoid wasp. Pest Manag Sci 58:321–328. doi:10.1002/ps.454
Rocha LCD, Carvalho GA (2004) Adaptação da metodologia padrão da IOBC para estudos de seletividade com Trichogramma pretiosum Riley, 1879 (Hymenoptera: Trichogrammatidae) em condições de laboratório. Acta Sci 26:315–320
Scott AJ, Knott MA (1974) A cluster analysis method for grouping means in the analysis of variance. Biometrics 30:507–512. doi:10.2307/2529204
Singh PP, Varma GC (1986) Comparative toxicities of some insecticides to Chrysoperla carnea (Chrysopidae: Neuroptera) and Trichogramma brasiliensis (Hymenoptera: Trichogrammatidae), two arthropod natural enemies of cotton pests. Agric Ecosyst Environ 15:23–30. doi:10.1016/0167-8809(86)90110-6
Soares MA, Leite GLD, Zanuncio JC, Rocha SL, Sá VGM, Serrão J (2007) Flight capacity, parasitism and emergence of six Trichogramma (Hymenoptera: Trichogrammatidae) species from reforested areas with eucalyptus in Brazil. Phytoparasitica 35:314–318
Stapel JO, Cortesero AM, Lewis WJ (2000) Disruptive sublethal effects of insecticides on biological control: altered foraging ability and life span of a parasitoid after feeding on extrafloral nectar of cotton treated with systemic insecticides. Biol Control 17:243–249. doi:10.1006/bcon.1999.0795
Suh CPC, Orr DB, Duyn JWV (2000) Effect of insecticides on Trichogramma exigum (Hymenoptera: Trichogrammatidae) preimaginal development and adult survival. J Econ Entomol 93:577–583
Takada Y, Kawamura S, Tanaka T (2001) Effects of various insecticides on the development of the egg parasitoid Trichogramma dendrolimi (Hymenoptera: Trichogrammatidae). J Econ Entomol 94:1340–1343
Vieira A, Oliveira L, Garcia P (2001) Effects of conventional pesticides on the preimaginal developmental stages and on adults of Trichogramma cordubensis (Hymenoptera: Trichogrammatidae). Biocontrol Sci Technol 11:527–534. doi:10.1080/09583150120067553
Youssef AI, Nasr FN, Stefanos SS, Elkhair SSA, Shehata WA, Agamy E, Herz A, Hassan SA (2004) The side-effects of plant protection products used in olive cultivation on the hymenopterous egg parasitoid Trichogramma cacoeciae Marchal. J Appl Entomol 128:593–599. doi:10.1111/j.1439-0418.2004.00892.x
Zanuncio JC, Batalha VC, Guedes RNC, Picanço MC (1998) Insecticide selectivity to Supputius cincticeps (Stal) (Het.: Pentatomidae) and its prey Spodoptera frugiperda (J.E. Smith) (Lep.: Noctuidae). J Appl Entomol 122:457–460
Zanuncio TV, Serrão JE, Zanuncio JC, Guedes RNC (2003) Permethrin-induced hormesis on the predator Supputius cincticeps (Stål, 1860) (Heteroptera: Pentatomidae). Crop Prot 22:941–947. doi:10.1016/S0261-2194(03)00094-2
Zanuncio TV, Zanuncio JC, Serrão JE, Medeiros RS, Pinon TBM, Sediyama CAZ (2005) Life table of the predator Supputius cincticeps (Stål. 1860) (Heteroptera: Pentatomidae) exposed to sublethal doses of permethrin. Biol Res 38:31–39
Zehnder G, Gurr GM, Kuhne S, Wade MR, Wratten SD, Wyss E (2007) Arthropod pest management in organic crops. Annu Rev Entomol 52:57–80. doi:10.1146/annurev.ento.52.110405.091337
Acknowledgments
To “Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq)”, to “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)” and to “Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG)”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vianna, U.R., Pratissoli, D., Zanuncio, J.C. et al. Insecticide toxicity to Trichogramma pretiosum (Hymenoptera: Trichogrammatidae) females and effect on descendant generation. Ecotoxicology 18, 180–186 (2009). https://doi.org/10.1007/s10646-008-0270-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-008-0270-5