Abstract
Background
Long noncoding RNA colon cancer-associated transcript 1 (LncRNA CCAT1) is highly expressed in gastric cancer tissues and plays a role in autophagy. However, the underlying mechanism still needs to be further clarified.
Objective
To study the role of LncRNA CCAT1 in regulating autophagy of gastric cancer cells, analyze its downstream targets, and elucidate the mechanism.
Methods
qPCR detected the expression of LncRNA CCAT1 in gastric cancer cells. The proliferation, migration, and invasion ability of LncRNA CCAT1 and the expression level of autophagy-related proteins in gastric cancer cells were detected. Bioinformatics method predicted the downstream targets of LncRNA CCAT1, and they were verified by dual-luciferase assay. The relationship between LncRNA CCAT1, miR-140, and ATG5 was verified by co-transfection, and the expression levels of ATG5 and ATG5–ATG12 complex proteins were detected. Finally, the role of LncRNA CCAT1 in vivo was confirmed by gastric cancer transplantation model.
Results
LncRNA CCAT1 was highly expressed in gastric cancer cells. LncRNA CCAT1 can promote the proliferation, migration, invasion, and autophagy activity of gastric cancer cells. LncRNA CCAT1 can bind to miR-140-3p and regulate its expression, while miR-140-3p further regulates the expression of ATG5. Overexpression of LncRNA CCAT1 can promote tumor growth in nude mice. After LncRNA CCAT1 silencing, the positive expression rate of ATG5 in nude mice was low.
Conclusion
LncRNA CCAT1 may inhibit the expression of miR-140-3p by sponge adsorption, thus weakening its inhibitory effect on ATG5. Eventually, gastric cancer cells were more prone to autophagy under the pressure of stress.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer (GC), as one of the five significant malignancies in the world, ranks second in the global cancer mortality rate and is also one of the most common malignancies in China at present [1, 2]. With the exploration of the molecular mechanism of gastric cancer, an increasing number of studies have revealed the critical role of autophagy in GC development [3, 4]. Autophagy maintains mitochondrial function and energy balance in cancer cells by degrading aging organelles and proteins under hypoxia or nutrient deficiency conditions. Besides, autophagy can act as a promoter of gastric cancer progression. In contrast, inhibition autophagy can increase the sensitivity of human GC cells to cisplatin [5]. Therefore, elucidation of the autophagy mechanism will help provide a new therapeutic target for gastric cancer treatment.
Long noncoding RNA (referred to as LncRNA) is a class of RNA molecules whose transcriptional length exceeds 200 nt but does not have translation ability [6]. LncRNA has essential biological functions [7, 8]. For example, LncRNA can participate in many critical signal transduction regulatory processes such as genomic imprinting and chromatin modification [9, 10], transcriptional activation post-transcriptional regulation, and protein function regulation [8]. These regulatory roles of LncRNA in physiological and disease processes, especially in tumor occurrence and development, have gradually attracted wide-spread attention [11]. But in the case of GC, the research is at the beginning. Colon cancer-associated transcriptome 1 (CCAT1) is a nuclear-restricted LncRNA that regulates various gastrointestinal cancers. Yang’s study indicated that LncRNA CCAT1 gene expression was significantly upregulated in gastric and colon cancer tissues, and LncRNA CCAT1 can be activated by c-myc which promotes the occurrence and metastasis of colon cancer [12]. However, whether LncRNA CCAT1 is involved in autophagy regulation of gastric cancer has not been discussed.
This project research significance in further understanding of LncRNA in the progression of GC illustrated its function and regulation mechanism, which would deepen our understanding of the pathophysiology of gastric cancer and provide new candidate targets for diagnosis and treatment of GC.
Materials and Methods
Cell Culture and Transfection
Cell lines AGS, MKN-45, and normal gastric epithelial cells GES-1 (Zhejiang Ruyao Biotechnology Co., Ltd., Zhejiang, China) were cultured in RPMI-1640 medium with 10% fetal bovine serum (FBS) and incubated in a CO2 incubator at 37 °C. Lentivirus for overexpression and silence of LncRNA CCAT1 was obtained from Zhejiang Ruyao Biotechnology Co., Ltd. (Zhejiang, China), and overexpressed and silenced LncRNA CCAT1 cell line was named as pCDH-CCAT1 and pLKO-CCAT1, respectively. MiR-140 mimic and negative control miR-NC were synthesized by Genewiz Biotechnology Co., Ltd. (Jiangsu, China), and GC cells were transfected with Lipofectamine 3000 transfection reagent (Thermo Fisher, USA). The kit instructions for cell transfection were followed. Primer sequences for overexpression of LncRNA CCAT1 were as follows: LncRNA CCAT1 forward 5′-cgg GAT CCT TTA AAT CAT ACC AAT TG-3′, LncRNA CCAT1 reverse 5′-TCC CCC GGG CAA TTG GTA TGA TTT AAA AAC-3′. The sequences of shRNA primers used to knock out LncRNA CCAT1 were as follows: Oligo1 forward 5′-CCG GTT TCA CAA GTC ACA CAA TAT TCT CGA GAA TAT TGT GTG ACT TGT GAA ATT TTT G-3′, Oligo1 reverse 5′-AAT TCA AAA ATT TCA CAA GTC ACA CAA TAT TCT CGA GAA TAT TGT GTG ACT TGT GAA A-3′; Oligo2 forward 5′-CCG GCC ACT GCC CTG AAA CTT ATA TCT CGA GAT ATA AGT TTC AGG GCA GTG GTT TTT G-3′, Oligo2 reverse 5′-AAT TCA AAA ACC ACT GCC CTG AAA CTT ATA TCT CGA GAT ATA AGT TTC AGG GCA GTG G-3′; Oligo3 forward 5′-CCG GTT AAC TGC ATG CTC CTA TTT ACT CGA GTA AAT AGG AGC ATG CAG TTA ATT TTT G-3′, Oligo3 reverse 5′-AAT TCA AAA ATT AAC TGC ATG CTC CTA TTT ACT CGA GTA AAT AGG AGC ATG CAG TTA A-3′; ShRNA control: 5′-CCG GCA ACA AGA TGA GCA CCA ACT CGA GTT GGT GCT CTT CAT CTT GTT GTT TTT G-3′.
qPCR Detection
Total RNA was isolated from cells or tissues using the Trizol method. The microspectrophotometer measured the concentration and quality of RNA at 260/280 nm. Then, the total RNA was reverse-transcribed into cDNA using M-MLV (NEB, UK). The expressions of LncRNA CCAT1 and ATG5 were detected using NovoStart ® SYBR qPCR Supermix Plus (NovoProtein, Shanghai, China) kit with GAPDH as the internal reference. The above methods were also used to detect the expression of miR-140, with U6 as the internal control. Relative expression of LncRNA CCAT1, ATG5, and miR-140 was calculated by 2−ΔΔCt method. The primer sequences involved in this study were listed as follows: Hsa-miR-140-3p 5′-CAC AGG GTA GAA CCA CGG AA-3′, Tiagen miRNA detection reverse primer; U6 forward 5′-CGC TTC GGC AGC ACA TAT AC-3′, U6 reverse 5′-CGA ATT TGC GTG TCA TCC TTG C-3′; LncRNA CCAT1 forward 5′-TTT ATG CTT GAG CCT TGA-3′, LncRNA CCAT1 forward 5′-TTT ATG CTT GAG CCT TGA-3′, LncRNA CCAT1 reverse 5′-CTT GCC TGA AAT ACT TGC-3′; GAPDH forward 5′-CGC TCT CTG CTC CTC CTG TTC-3′, GAPDH reverse 5′-ATC CGT TGA CTC CGA CCT TCA C-3′.
Cell Viability Assay
After transfection for 48 h, cells were inoculated in 96-well plates and cultured in a CO2 incubator for 24, 36, 48, and 72 h. Cell viability was detected by MTT assay. The cells were incubated with MTT solution (0.5 mg/mL) at 37 °C for 4 h, and then, 150 μL dimethyl sulfoxide (DMSO) was added to dissolve the precipitated formazan. After shaking for 5 min, the absorbance of cells in each well was measured at 570 nm with a microplate reader. The larger the OD value, the higher the cell viability.
Scratch Test
The cells were inoculated in a 6-well plate, grew to approximately 100% confluence, and then scratched straight with a 10-μL tip. The cells were washed three times with PBS to remove the cell debris, and the cells were cultured with fresh DMEM containing 1% FBS. The wound healing was observed under a microscope at different time points. Each cell line was photographed from a representative scratch, and cell migration ability was assessed by measuring the wound’s width. Each test was carried out in triplicate in at least two independent experiments.
Transwell Assesses the Ability of Cell Invasion
The lower chamber of the transwell plate was filled with 600 μL fresh culture medium containing 5% FBS. Forty-eight hour after transfection, cells were digested with trypsin, cell density was adjusted at 5.0 × 104/mL, and then, 200 μL suspension cells were inoculated into the upper chamber, which was precoated with Matrigel 24 h before. Cells were cultured at 37 °C in a CO2 incubator for 24 h, the upper section was taken out, the cells on the upper membrane were scraped out with swab, then fixed with 5% paraformaldehyde, stained with crystal violet for 15 min, and observed under an inverted microscope, five random fields were captured, and the invasion cells number was recorded.
Bioinformatics Analysis and Prediction of Target
For prediction of the potential targets of LncRNA CCAT1, bioinformatics Web sites including Target Scan (http://www.targetscan.org), miRDB (http://mirdb.org), and miRanda (http://www.microrna.org) were used, and the results were integrated. Differentially expressed microRNAs in gastric cancer patients were obtained from dbDEMC 2.0 database (http://www.picb.ac.cn/dbDEMC/). The differentially expressed microRNAs of gastric cancer patients were intersected with the predicted miRNAs of LncRNA CCAT1. Target Scan and miRanda further predicted the target genes of miRNA and the binding site.
Dual-Luciferase Reporter Gene Assay
The wild-type and mutant sequences were chemically synthesized and cloned into a dual-luciferase reporter plasmid. Partial sequences of LncRNA CCAT1 and ATG5 3′-UTR containing wild-type or mutant miR-140-binding sites were cloned into pGL3-basic luciferase vector (Promega, Madison, WI, USA) to generate CCAT1-WT, CCAT1-MUT, and ATG5-3′-UTR-WT (WT) and ATG5-3′-UTR-MUT (MUT), respectively. The constructed luciferase vector was co-transfected with PRL-TK plasmid (Promega) miR-NC and miR-140, respectively, into gastric cancer cells. 48 h after transfection, luciferase activity in cell lysates was measured using the dual-luciferase reporter gene assay system (Promega).
Western Blot
The cells were collected, discarded medium, and washed once with pre-cooled PBS, then the RIPA lysis buffer was added, and the cells were incubated on ice for 10 min. The cell lysates were scraped off with a 1-ml tip, and the DNA was broken with a 1-ml disposable syringe. The cell lysate was centrifuged at 12,000 × g for 10 min, and 5 μL of the supernatant was taken for BCA assay, and the remaining was mixed with 5 × loading buffer and boiled for 5 min. SDS–PAGE was prepared and loaded with 40 μg protein per lane. After electrophoresis, the protein was transferred to the PVDF membrane, blocked with 5% skim milk for 2 h at room temperature. The primary antibody including ATG5, Beclin1, LC3A/B, and ATG12 antibodies was purchased from Wuhan Proteintech Biotechnology Company, ATG5–ATG12 antibodies were bought from CST Companies, and all antibodies were diluted at 1:1000. PVDF membrane was then incubated with primary antibody overnight at 4 °C and then washed three times with TBST for 10 min, and HRP-labeled secondary antibody was added and incubated in a shaker at room temperature for 1.5–2 h. ECL luminescence solution was added, and the images were captured under the protein imager, and the optical density of the strips was quantified with ImageJ software.
Acridine Orange Staining
After digestion, the cells were inoculated into a 6-cm dish with a coverslip pre-placed. After washing with PBS, acridine orange with a final concentration of 1 μg/mL was added, and the cells were incubated at 37 °C for 10 min in the dark. After that, the cells were rinsed once with PBS and mounted with anti-fluorescence quenching solution.
Subcutaneous Tumor Transplantation Experiment in Nude Mice
BalB/C nude mice aged 4 weeks were raised in an SPF environment. After adaptive growth for 1 week, the nude mice were randomly divided into three groups, namely the control group, pCDH-CCAT1 overexpression group, and pLKO-CCAT1 silent expression group. Two hundred microliters of cell suspension containing 5 × 106 cells was implanted in the upper arm of nude mice, and the growth status was observed every 2–3 days after injection. When subcutaneous masses were touchable, the long diameter (A) and short diameter (B) of the tumors were measured with a digital vernier caliper, and the tumor volume (mm3) was calculated with the following formula: 0.5 × a × b2. After 40 days, the nude mice were killed by cervical vertebrae dislocation. The Animal Ethics Committee of Oriental Hepatobiliary Surgery Hospital Affiliated to the Second Military Medical University approved this study’s animal experiment scheme.
HE Staining
The nude mice tumor was stripped, fixed, and embedded in paraffin, and paraffin sections were made at 5 μm thickness. After the tumor tissues in paraffin were dewaxed to water, hematoxylin and eosin staining was performed. After dehydration, the tumor tissues were transparent with xylene, mounted with neutral gum, and observed and photographed under a microscope (Leica, DM500).
Immunohistochemistry
The expression and distribution of ATG5 protein were detected by immunohistochemistry in paraffin sections of each group. Immunohistochemical steps were as follows: After routine paraffin dewaxing to water, antigen repair was performed. Three percentage of hydrogen peroxide solution was added to remove endogenous catalase. One percentage BSA sealing solution was added and sealed for 15 min. After the blocking solution was dumped, the primary antibody ATG5 (1:100 dilution) was added and incubated overnight at 4 °C. Then, it was washed three times with PBST. The secondary antibody (1:100 dilution) was added and incubated for 1 h at room temperature. The DAB was developed for color, and the hematoxylin was redyed for 30 s. It was washed with running water for 1 min. It was treated with 0.1% sodium bicarbonate for blue, subjected to dehydration and xylene transparent, mounted with neutral gum, observed under a microscope, and photographed.
Statistical Methods
All of the above experiments were repeated three times separately, and the data were expressed as mean ± standard deviation. Student’s t test was used to compare two groups, and one-way analysis of variance (ANOVA) was used to analyze the conflict between multiple groups. The other difference was analyzed by post hoc test, and GraphPad Prism 8.0 was used to draw statistical charts. p < 0.05 was considered statistically significant.
Results
Expression Levels of LncRNA CCAT1 and Autophagy-Related Proteins in Gastric Cancer Cells
qPCR results showed that the expression level of the LncRNA CCAT1 gene in gastric cancer cells was significantly higher than that in GES-1 cells (Fig. 1a), and the difference was statistically significant (p < 0.05). As shown in Fig. 1b–c, the gastric cancer cell line was overexpressed, and knockdown of LncRNA CCAT1 was established by lentivirus. After lentivirus infection, qPCR results showed that the overexpression and silencing of LncRNA CCAT1 were achieved in AGS and MKN45. Expression of LncRNA CCAT1 in AGS and MKN45 was upregulated at 7.93 and 6.28 times, respectively. In the three groups of shRNA, Oligo2 has the best silencing efficiency, which abolished the expression of LncRNA CCAT1 in AGS and MKN45 by 89% and 84%, respectively. Thus, No. 2 shRNA was selected for follow-up research and named pLKO-CCAT1.
Expression levels of LncRNA CCAT1 and autophagy-related proteins in gastric cancer cells. a LncRNA CCAT1 gene expression was detected by qPCR. b The gene expression level of LncRNA CCAT1 in AGS cells with overexpression and silencing of LncRNA CCAT1 was detected by qPCR. c The expression level of LncRNA CCAT1 gene in MKN-45 cells with overexpression and silencing of LncRNA CCAT1 was detected by qPCR. “*” represents a statistically significant difference compared with the control group, p < 0.05
Effect of LncRNA CCAT1 Gene on the Function of Gastric Cancer Cells
The MTT detected the proliferation activity of gastric cancer cells. As shown in Fig. 2a–b, after LncRNA CCAT1 overexpression, the cell proliferation rate was fastest than the control group (p < 0.05), and when the LncRNA CCAT1 was silenced, the proliferation rate slowed down significantly (p < 0.05), suggesting that LncRNA CCAT1 can promote cell proliferation. After overexpression of LncRNA CCAT1, cell scratch assay results (Fig. 2c, e) showed that the migration distance of AGS and MKN-45 cells was 147 ± 32 μm and 202 ± 25 μm, respectively, compared with 107 ± 12 μm and 108 ± 12 μm in the control group, respectively. The difference was statistically significant (p < 0.05), and the cell migration distance decreased to 64 ± 15 μm and 51 ± 14 μm, respectively, after silencing LncRNA CCAT1, which was statistically significant compared with the control group (p < 0.05). The results of cell invasion assay showed that the number of invaded cells of AGS and MKN-45 in the control group was 71 ± 4 and 45 ± 5, respectively. In contrast, after overexpression of LncRNA CCAT1, the number of invaded cells increased to 136 ± 6 and 124 ± 7 cells (p < 0.05), respectively. The LncRNA CCAT1 silence group dropped to 24 ± 4 and 18 ± 3 (p < 0.05), respectively. The above results suggest that LncRNA CCAT1 can significantly promote the proliferation, migration, and invasion in gastric cancer cells.
Effects of LncRNA CCAT1 gene on the function of gastric cancer cells. a MTT assay was used for AGS cell activity. b MTT was used to detect the movement of MKN-45 cells. c Cell migration was detected by cell scratch assay (scale bar 75 μm). d Cell invasion was detected by transwell assay (scale bar 75 μm). e The results of cell scratch assay were quantified. f Statistical quantification of the results of the transwell experiment. “*” represents a statistically significant difference compared with the control group, p < 0.05
LncRNA CCAT1 Affects the Autophagy Level of Gastric Cancer Cells
After overexpression and silencing of LncRNA CCAT1, the expression of autophagy-related proteins, including ATG5, ATG12, LC3A/B, and Beclin1, was detected by western blot. As shown in Fig. 3a–d, autophagy-related proteins were upregulated significantly compared with the control group, and the difference was statistically significant (p < 0.05). When LncRNA CCAT1 was silenced, the above autophagy-related proteins were downregulated by varying degrees, compared with the control group, and the difference was statistically significant (p < 0.05).
The expression level of LncRNA CCAT1 gene affected the autophagy level of gastric cancer cells. a The expression level of autophagy-related proteins of LncRNA CCAT1 cell line AGS was overexpressed and silenced by western blotting. b The expression level of autophagy-associated proteins in overexpressed and muted LncRNA CCAT1 cell line MKN-45 was detected by western blot. c The statistical result of quantification of the gray value of the western blotting result band A. d The statistical result of quantification of the gray value of the western blotting result band B. “*” represents a statistically significant difference compared with the control group, p < 0.05
LncRNA CCAT1 Affects the Activity and Autophagy of Gastric Cancer Cells by Regulating the Expression Level of miR-140
Bioinformatics analysis suggested that LncRNA CCAT1 might interact with miR-140. MiR-140 expression levels in AGS, MKN-45, and GES-1 were detected by qPCR. As shown in Fig. 4a, the expression level of miR-140 in GES-1 cells was 6.25 and 4.8 times higher than that in AGS and MKN-45 cells, respectively. Meanwhile, expression of miR-140 in the LncRNA CCAT1 overexpression and silencing group was detected, and as shown in Fig. 4b, the expression level of miR-140 in pCDH-CCAT1 overexpression group was inhibited significantly (p < 0.05). In contrast, the expression level of miR-140 in the pLKO-CCAT1 silenced group was increased significantly (p < 0.05). Further, the binding site between LncRNA CCAT1 and miR-140 is shown in Fig. 4c, and the mutation sequence of LncRNA CCAT1 was shown either. Figure 4d shows the detection results of dual-luciferase activity assay, which showed a significant decrease in the fluorescence intensity value between wild-type LncRNA CCAT1 and miR-140 compared with the miR-NC group. The difference was statistically significant (p < 0.05), and the fluorescence intensity of mutant LncRNA CCAT1 was not especially changed after co-transfection with miR-140 mimic (p > 0.05). The autophagy-related proteins Beclin1 and ATG5 were detected by western blot. The results showed a significant increase in the level of Beclin1 and ATG5 after overexpression of LncRNA CCAT1 compared with control groups, the difference was statistically significant (p < 0.05), and miR-140 mimic could effectively reverse the increase of autophagy protein and significantly reduce the expression of the autophagy-related protein (p < 0.05). Western blotting was performed on gastric cancer cells silenced with LncRNA CCAT1, and the results showed significant inhibition of expression of autophagy-related proteins after silencing with LncRNA CCAT1. The difference was statistically significant (p < 0.05), and transfection of miR-140 mimic intensified the inhibition effect (Fig. 5a–d). After transfection with miR-140 mimic, the cell viability of LncRNA CCAT1-overexpressed and silenced cells was detected by MTT assay, which showed that overexpression of LncRNA CCAT1 could significantly improve the activity of gastric cancer cells. After adding miR-140 mimic, it reversed the crude proliferation activity of LncRNA CCAT1, and the difference between the two groups was statistically significant (p < 0.05). Compared with the control group, silencing LncRNA CCAT1 significantly reduced gastric cancer cells’ activity, and the difference was statistically significant (p < 0.05). Simultaneously, after miR-140 mimic, the cell activity was lower than that of the silent group (Fig. 4e, f).
LncRNA CCAT1 affected the activity of gastric cancer cells by regulating the expression level of miR-140. a qPCR was used to detect the relative expression levels of miR-140 in AGS, MKN-45 gastric cancer cells, and GES-1 gastric epithelial cells. b The relative gene expression of miR-140 in AGS and MKN-45 LncRNA CCAT1 cell lines overexpressed and silenced was detected by qPCR. c The targeted binding sequence between LncRNA CCAT1 and miR-140-3p and the mutant sequence of LncRNA CCAT1. d Dual-luciferase activity report. e–f MTT was used to detect the cell activity of LncRNA CCAT1 AGS and MKN-45 cells transfected with miR-NC and miR-140 mimics. “*” represents a statistically significant difference compared with the control group, p < 0.05
LncRNA CCAT1 affected autophagy of gastric cancer cells by regulating the expression level of miR-140. a The expression levels of autophagy-related proteins in overexpressed and silenced LncRNA CCAT1 cell line AGS after transfection with miR-NC and miR-140 mimic were detected by western blot. b Western blotting was used to detect the expression levels of autophagy-related proteins in overexpressed and silenced LncRNA CCAT1 cell line MKN-45 after transfection with miR-NC and miR-140 mimic. c Results A quantitative statistical chart of the gray values of western blotting strips. d Result B quantified statistic chart of gray scale of western blotting strips. “*” represents a statistically significant difference compared with the control group, p < 0.05
MiR-140 Affects the Activity and Autophagy Level of Gastric Cancer Cells Through ATG5
The expression of ATG5 in gastric cancer cells was performed by qPCR. As shown in Fig. 6a, AGS and MKN-45 gastric epithelial cells showed the relatively significantly higher than that in GES-1 cells, and the difference was statistically significant (p < 0.05, Fig. 6a). Subsequently, qPCR was performed on the overexpressed and silenced LncRNA CCAT1 (Fig. 6b, c). The results showed that the expression level of miR-140 was significantly inhibited after the overexpression of LncRNA CCAT1 in gastric cancer cells. The difference was statistically significant (p < 0.05), and the expression level of ATG5 was significantly increased (p < 0.05), and after transfection with miR-140 mimic, the expression level of miR-140 was increased. The gene expression level of ATG5 was decreased considerably, compared with the control group, and the difference was not statistically significant (p < 0.05). However, miR-140 expression was significantly increased in gastric cancer cells silenced by LncRNA CCAT1 (p < 0.05). The expression level of ATG5 was significantly lower than that of control group (p < 0.05). After transfection with miR-140 mimic, the expression level of miR-140 was higher and the expression level of ATG5 gene was lower than that of the silent LncRNA CCAT1 group, and the differences were statistically significant (p < 0.05). Figure 6d shows the binding site sequence and mutation sequence of ATG5 and miR-140-3p, with 7 consecutive paired bases and 11 discontinuous base-binding sites. The results of dual-luciferase activity report (Fig. 6e) showed that there was a targeted binding relationship between wild-type ATG5 and miR-140, showing a significant reduction in the fluorescence intensity after transfection with miR-140, the difference was statistically significant (p < 0.05), and there was no statistically significant difference between miR-140 transfected with mutant ATG5 and miR-NC (p > 0.05). The results were further verified by western blot (Fig. 6f–i). ATG5 protein and ATG5–ATG12 complex protein in gastric cancer cells were significantly downregulated after transfection with a miR-140 mimic (p < 0.05), and after transfection with miR-140 inhibitor, the expression levels of ATG5 protein and ATG5–ATG12 complex protein were specifically upregulated, and the difference was statistically significant compared with the control group (p < 0.05). MTT experiment results showed that compared with the control group, the cell activity of gastric cancer cells transfected with miR-140 mimic was significantly inhibited (p < 0.05), and transfection with miR-140 inhibitor could significantly increase the activity level of gastric cancer cells (p < 0.05). The results of acridine orange staining (Fig. 7c) showed that the orange-red aggregation point was autophagy lysosomes. Compared with the control group, it significantly reduced the number of autophagy lysosomes in gastric cancer cells transfected with miR-140. Autophagosomes were ubiquitous in gastric cancer cells after transfection with miR-140 inhibitor.
miR-140 targeting binds to ATG5. a qPCR was used to detect the relative expression of Atg5 gene in AGS, MKN-45 gastric cancer cells, and GES-1 gastric epithelial cells. b The expression level of miR-140 gene after transfection with miR-NC and miR-140 mimic in LncRNA CCAT1 cell line AGS. c The expression level of miR-140 gene after transfection with miR-NC and miR-140 mimic in overexpressed and silenced LncRNA CCAT1 cell line MKN-45. d Targeting binding sequence between miR-140-3p and ATG5 and mutation sequence of ATG5. e Dual-luciferase activity report. f Western blot was used to detect the relative expression levels of autophagy-related proteins in transfected miR-NC,miR-140 mimic, and miR-140 inhibitor cell line AGS. g Western blotting was used to detect the relative expression levels of autophagy-related proteins transfected with miR-NC, miR-140 mimic, and miR-140 inhibitor cell line MKN-45. h Results F quantitative statistical chart of the gray values of western blotting strips. i Results G quantified statistical graph of gray scale of western blotting strips. “*” represents a statistically significant difference compared with the control group, p < 0.05
miR-140 affected the activity and autophagy level of gastric cancer cells through ATG5. a MTT assay was used to detect the activity of cells transfected with miR-NC, miR-140 mimic, and miR-140 inhibitor cell line AGS. b Cell activity of miR-NC, miR-140 mimic, and miR-140 inhibitor cell line MKN-45 transfected with MTT was detected. c The transfected miR-NC, miR-140 mimic, and miR-140 inhibitor cell line AGS was stained with acridine orange for autophagy lysosome. d The transfected miR-NC, miR-140 mimic, and miR-140 inhibitor cell line MKN-45 was stained with autophagy lysosome by acridine orange. “*” represents a statistically significant difference compared with the control group, p < 0.05
Effects of Stable Expression and Silencing of LncRNA CCAT1 Gene on Survival Rate and Tumor Changes in Tumor-Bearing Nude Mice
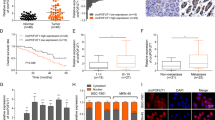
The tumor volume of pLKO-CCAT1 group was the smallest, and the tumor volume of pCDH-CCAT1 group was the largest and fastest by killing the tumor-bearing nude mice by neck removal (Fig. 8a, e). By observing the tumor-bearing survival rate of nude mice, the survival rate of pLKO-CCAT1 group was significantly higher than that of pCDH-CCAT1 group and the control group (Fig. 8d). HE staining (Fig. 8b) and IHC staining (Fig. 8c) showed that pLKO-CCAT1 group had the smallest tumor area, the tiniest blood vessels near the tumor in the whole tissue effectively suppressed the tumor, and the positive rate of ATG5 protein was also the highest.
Effects of stable expression and silencing of LncRNA CCAT1 gene on survival rate and tumor changes in tumor-bearing nude mice. a Tumor map of nude mice bearing tumor. b Results of HE staining of tumor tissue. c IHC staining was used to detect ATG5 protein in tumor tissues. d Survival rate of tumor-bearing nude mice. e Tumor size trends
Discussion
In recent years, there have been many studies on LncRNAs. Downregulation or upregulation of many LncRNAs contributes to tumorigenesis by affecting many cellular processes [13]. LncRNAs contribute to gastric cancer development, including LncRNAs XIST [14], SPRY4-IT1 [15], and ATB [16], which are involved in the pathogenesis of gastric cancer. LncRNA CCAT1 was initially found to be a biomarker for colorectal cancer. LncRNA CCAT1 is upregulated in gastric cancer and acts as a regulator of cell proliferation, invasion, and migration. However, the molecular mechanism of LncRNA CCAT1 in gastric cancer remains unclear.
Previous studies have reported that other ncRNAs regulate autophagy in cancer and can be considered biomarkers for cancer. For example, LncRNA HOTAIR is highly expressed and regulates autophagy by targeting ATG3 and ATG7. LncRNA SnHG11 can also activate the Wnt/β-catenin pathway and promote gastric cancer progression through oncogenic autophagy [17]. Here, we demonstrate that LncRNA CCAT1 is overexpressed in gastric cancer cells compared with normal gastric epithelial cells. Also, the enhancement of LncRNA CCAT1 promoted the activity and autophagy of gastric cancer cell lines. The results suggest that LncRNA CCAT1 may be an essential regulator of gastric cancer progression. Therefore, limiting the biological activity of these LncRNAs may provide a new strategy for targeted therapy of gastric cancer.
LncRNA CCAT1 has been found to promote tumorigenesis, metastasis, and progression of gastric cancer [18, 19]. Also, it has been reported that LncRNA CCAT1 can promote the expression of ATG7 by inhibiting miR-181, thus promoting autophagy in HCC cells [20]. MiR-140-3p plays a vital role in many cancers, such as breast cancer [21], cervical cancer [22], colorectal cancer [23], and bladder cancer [24]. In our study, LncRNA CCAT1 inhibited the expression of miR-140. Silencing LncRNA CCAT1 significantly reduced the activity and autophagy level of gastric cancer cells. Therefore, we speculated that LncRNA CCAT1 might regulate autophagy through negative regulation of miR-140. This study’s preliminary results showed that LncRNA CCAT1 gene expression level was relatively high in AGS and MKN-45 cells of five human gastric cancer cell lines, namely HGC-27, IM95M, SNU-1, AGS, and MKN-45 (the results shown in supplementary materials). Moreover, it has been documented that the autophagy of AGS and MKN-45 cells can protect mitochondrial function damage and promote cell survival [25, 26]. This indicates that AGS and MKN-45 cells may be more consistent with this study’s content and will be used as the main research target cells.
The autophagy pathway is a highly regulated dynamic process, mainly executed by the autophagy-associated (ATG) gene family. As far as its involvement in autophagosome extension is concerned, ATG5 is a crucial regulator necessary for autophagy [27]. ATG5 is considered a key regulator of autophagy, regulating tumorigenesis in GC through its effect on autophagy [28]. Ge et al. [29] also found that the upregulation of the expression level of ATG5 protein could induce gastric cancer resistance to chemotherapy drugs. In our study, the prediction and validation target of miR-140 was ATG5, which binds to the 3′-UTR of miR-140-3p. Combined with previous studies and this experiment’s results, miR-140 was under-expressed in gastric cancer tissues and cells. MiR-140 mimic could inhibit gastric cancer cells’ activity and promote autophagy, while miR-140 inhibitor showed the opposite effect. The expression level of ATG5 was inhibited after transfection with miR-140 mimic, and the expression level of ATG5–ATG12 complex protein, which plays a role in its downstream, was also inhibited. The activity of gastric cancer cells and the autophagy level were decreased correspondingly, while obtaining the opposite results after transfection with miR-140 inhibitor. Therefore, we infer that miR-140 may regulate autophagy by regulating the expression of ATG5. Besides, it is proved that LncRNA CCAT1 inhibited the expression of miR-140, and ATG5 could be used as a target of miR-140. LncRNA CCAT1-miR-140-ATG5 axis may mediate the occurrence of autophagy in gastric cancer. MiR-140 inhibitor increased ATG5 and ATG5–ATG12 complex proteins’ expression levels, suggesting that miR-140 may regulate autophagy in gastric cancer through the function of ATG5 and its downstream acting proteins.
Although LncRNA CCAT1 is an oncogene, how LncRNA CCAT1 regulates autophagy remains to be studied. Our study proved that LncRNA CCAT1 inhibited ATG5, the target of miR-140-3p, and the regulation of ATG5 expression by miR-140-3p. Meanwhile, after the overexpression of LncRNA CCAT1, the expression of the miR-140-3p gene was decreased and the expression of ATG5 was enhanced. The results showed that LncRNA CCAT1 regulates autophagy through sponge miR-140-3p to increase the expression of ATG5.
In conclusion, this study confirmed that LncRNA CCAT1 could act as a new oncogene by affecting cell activity and autophagy. Besides, LncRNA CCAT1 induces enhanced ATG5 expression and regulates its action by binding LncRNA CCAT1 to miR-140-3p. LncRNA CCAT1-miR-140-ATG5 can be used as a new target pathway for the targeted therapy of gastric cancer.
References
Zhang J, Qiu WQ, Zhu H et al. HOTAIR contributes to the carcinogenesis of gastric cancer via modulating cellular and exosomal miRNAs level. Cell Death Dis. 2020;11:780.
Cao J, Zhang X, Xu P et al. Circular RNA circLMO7 acts as a microRNA-30a-3p sponge to promote gastric cancer progression via the WNT2/β-catenin pathway. J Exp Clin Cancer Res. 2021;40:6.
Peng Q, Zhao P, Shen Y, Cheng M, Wu Y, Zhu Y. Prognostic implication and functional exploration for microRNA-20a as a molecular biomarker of gastrointestinal cancer. BMC Cancer. 2020;20:420.
Zhang Q, Miao Y, Fu Q et al. CircRNACCDC66 regulates cisplatin resistance in gastric cancer via the miR-618/BCL2 axis. Biochem Biophys Res Commun. 2020;526:713–720.
Zhang HQ, He B, Fang N, Lu S, Liao YQ, Wan YY. Autophagy inhibition sensitizes cisplatin cytotoxicity in human gastric cancer cell line SGC7901. Asian Pac J Cancer Prev. 2013;14:4685–4688.
Tsagakis I, Douka K, Birds I, Aspden JL. Long non-coding RNAs in development and disease: conservation to mechanisms. J Pathol. 2020;250:480–495.
Charles Richard JL, Eichhorn P. Platforms for Investigating LncRNA Functions. SLAS Technol. 2018;23:493–506.
Statello L, Guo CJ, Chen LL, Huarte M. Gene regulation by long non-coding RNAs and its biological functions. Nat Rev Mol Cell Biol. 2021;22:96–118.
Zhang R, Huang XQ, Jiang YY, Li N, Wang J, Chen SY. LncRNA TUG1 regulates autophagy-mediated endothelial-mesenchymal transition of liver sinusoidal endothelial cells by sponging miR-142-3p. Am J Transl Res. 2020;12:758–772.
Ye M, Ma J, Liu B, Liu X, Ma D, Dong K. Linc01105 acts as an oncogene in the development of neuroblastoma. Oncol Rep. 2019.
Castro-Oropeza R, Melendez-Zajgla J, Maldonado V, Vazquez-Santillan K. The emerging role of lncRNAs in the regulation of cancer stem cells. Cell Oncol (Dordr). 2018;41:585–603.
He X, Tan X, Wang X et al. C-Myc-activated long noncoding RNA CCAT1 promotes colon cancer cell proliferation and invasion. Tumour Biol. 2014;35:12181–12188.
Zhang X, Zhang W, Jiang Y, Liu K, Ran L, Song F. Identification of functional lncRNAs in gastric cancer by integrative analysis of GEO and TCGA data. J Cell Biochem. 2019;120:17898–17911.
Chen DL, Ju HQ, Lu YX et al. Long non-coding RNA XIST regulates gastric cancer progression by acting as a molecular sponge of miR-101 to modulate EZH2 expression. J Exp Clin Cancer Res. 2016;35:142.
Xie M, Nie FQ, Sun M et al. Decreased long noncoding RNA SPRY4-IT1 contributing to gastric cancer cell metastasis partly via affecting epithelial-mesenchymal transition. J Transl Med. 2015;13:250.
Saito T, Kurashige J, Nambara S et al. A Long Non-coding RNA Activated by Transforming Growth Factor-β is an Independent Prognostic Marker of Gastric Cancer. Ann Surg Oncol. 2015;22:S915–S922.
Wu Q, Ma J, Wei J, Meng W, Wang Y, Shi M. lncRNA SNHG11 Promotes Gastric Cancer Progression by Activating the Wnt/β-Catenin Pathway and Oncogenic Autophagy. Mol Ther. 2021;29:1258–1278.
Li Y, Zhu G, Ma Y, Qu H. lncRNA CCAT1 contributes to the growth and invasion of gastric cancer via targeting miR-219-1. J Cell Biochem. 2019;120:19457–19468.
Yang F, Xue X, Bi J et al. Long noncoding RNA CCAT1, which could be activated by c-Myc, promotes the progression of gastric carcinoma. J Cancer Res Clin Oncol. 2013;139:437–445.
Guo J, Ma Y, Peng X, Jin H, Liu J. LncRNA CCAT1 promotes autophagy via regulating ATG7 by sponging miR-181 in hepatocellular carcinoma. J Cell Biochem. 2019;120:17975–17983.
Zhou Y, Wang B, Wang Y, Chen G, Lian Q, Wang H. miR-140-3p inhibits breast cancer proliferation and migration by directly regulating the expression of tripartite motif 28. Oncol Lett. 2019;17:3835–3841.
Ma J, Zhang F, Sun P. miR-140–3p impedes the proliferation of human cervical cancer cells by targeting RRM2 to induce cell-cycle arrest and early apoptosis. Bioorg Med Chem. 2020;28:115283.
Jiang W, Li T, Wang J et al. miR-140-3p Suppresses Cell Growth And Induces Apoptosis In Colorectal Cancer By Targeting PD-L1. Onco Targets Ther. 2019;12:10275–10285.
Wang Y, Chen J, Wang X, Wang K. miR-140-3p inhibits bladder cancer cell proliferation and invasion by targeting FOXQ1. Aging (Albany NY). 2020;12:20366–20379.
Rong L, Li Z, Leng X, et al. Salidroside induces apoptosis and protective autophagy in human gastric cancer AGS cells through the PI3K/Akt/mTOR pathway. Biomed Pharmacother. 2020;122:109726.
Almasi S, Kennedy BE, El-Aghil M et al. TRPM2 channel-mediated regulation of autophagy maintains mitochondrial function and promotes gastric cancer cell survival via the JNK-signaling pathway. J Biol Chem. 2018;293:3637–3650.
Klionsky DJ. Autophagy: from phenomenology to molecular understanding in less than a decade. Nat Rev Mol Cell Biol. 2007;8:931–937.
Zhang Q, Wu S, Zhu J, Chai D, Gan H. Down-regulation of ASIC1 suppressed gastric cancer via inhibiting autophagy. Gene. 2017;608:79–85.
Ge J, Chen Z, Huang J, et al. Upregulation of autophagy-related gene-5 (ATG-5) is associated with chemoresistance in human gastric cancer. PLoS One. 2014;9(10):e110293.
Funding
This study was supported by the subject of Shanghai Municipal Health and Family Planning Commission (general project) (No. 201840070) and the subject of Science and Technology Commission of Jiading District, Shanghai (No. JDKW-2018-W12).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, F., Peng, Zx., Ji, Wd. et al. LncRNA CCAT1 Upregulates ATG5 to Enhance Autophagy and Promote Gastric Cancer Development by Absorbing miR-140-3p. Dig Dis Sci 67, 3725–3741 (2022). https://doi.org/10.1007/s10620-021-07187-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-021-07187-9