Abstract
Background and Aim
The purpose of this study was to estimate the sex- and age-specific incidence rates of inflammatory bowel diseases (IBD) in Taiwan. Site-specific cancer occurred in patients with IBD would be reported, too.
Methods
A retrospective study by analyzing the data from the National Health Insurance Research Database of Taiwan.
Results
Between 2000 and 2010, the overall incidence rate of Crohn’s disease (CD) and ulcerative colitis (UC) was 0.208 and 0.838 per 100,000 person-years. For male, the incidence rate of CD was 0.195 (95 % CI 0.113–0.276) per 100,000 persons in 2000 and increased to 0.318 (95 % CI 0.216–0.421) per 100,000 persons in 2010. For female, the incidence rate of CD was 0.092 (95 % CI 0.035–0.149) per 100,000 persons in 2000 and increased to 0.210 (95 % CI 0.128–0.293) per 100,000 persons in 2010. For male, the incidence rate of UC was 0.690 (95 % CI 0.537–0.843) per 100,000 persons in 2000 and increased to 1.351 (95 % CI, 1.140–1.562) per 100,000 persons in 2010. For female, the incidence rate of UC was 0.386 (95 % CI 0.269–0.503) per 100,000 persons in 2000 and increased to 0.858 (95 % CI 0.691–1.024) per 100,000 persons in 2010. Among the CD patients, 0.19 % had colorectal cancers (1/519). Among the UC patients, 0.24 % had colorectal cancers (5/2098).
Conclusions
This nationwide population-based longitudinal epidemiological study of IBD in Taiwan provides data for future global comparisons.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Inflammatory bowel disease (IBD) is characterized by chronic and/or relapsing immune activation and inflammation within the gastrointestinal tract. Crohn’s disease (CD) and ulcerative colitis (UC) are the two major forms. It was believed that IBD is caused by a combination of genetic predisposition, immune dysregulation, and environmental factors [1].
The prevalence and incidence rates of IBD vary geographically. IBD has its highest prevalence and incidence rates in Western countries such as Europe and North America [2]. However, the case numbers of IBD are raising in Asia [3–5]. Westernization of lifestyle, improved sanitation, and industrialization might lead to the increase in the disease [4].
The sex- and age-specific incidence rates of IBD are useful for the study of their etiology and planning for health resources. In 1995, Taiwan established a compulsory National Health Insurance (NHI) system, and coverage had reached 99 % of the population by the end of 2004. As of April 2010, there were 23,023,449 residents enrolled in the system [6].
The new technique of capsule endoscopy, which was introduced in 2000, and double-balloon enteroscopy, which was introduced in 2001 by Yamamoto, had made it possible to explore the entire small intestine and carry out endoscopic interventions. Improved diagnostic methods make physician more aware of IBD, which has historically been rare in Taiwan.
In this study, we estimated the sex- and age-specific incidence rate of IBD in Taiwan using the National Health Insurance Research Dataset (NHIRD) between January 2000 through and December 2010. Site-specific cancer occurred in patients with IBD would be reported, too.
Materials and Methods
Study Design
The primary data source was the Taiwan National Health Insurance Research Dataset (NHIRD). The NHIRD contains the registration files and original claims data for reimbursement. The NHI system is a single-payer social health insurance system. All residents of Taiwan are required to participate. There are approximately 23 million individuals in this registry.
Insured citizens with IBD are eligible to apply for a catastrophic illness certificate. Issuance of a catastrophic illness certificate to patients with IBD requires comprehensive review of clinical data, pathologic finding, image result, and endoscopic pictures by gastrointestinologist commissioned by the Bureau of National Health Insurance (BNHI). We identified the patients who received catastrophic illness certificates for IBD by their associated International Classification of Diseases, 9th revision (ICD-9) codes. The following ICD-9, Clinical Modification (ICD-9-CM) codes were used to identify patient records in this study: 555.x for CD and 556.x for UC.
Statistical Analysis
The prevalence rate of IBD was computed by year. Incidence rate was calculated as the number of new cases between 2000 and 2010 divided by the total number of person-years at risk. The person-years at risk was defined as the sum of patients from year 2000 to the diagnosis of IBD, dropout from the national health insurance program, death, or December 31, 2010, whichever came first. The 95 % confidence intervals (CI) for incidence rate were calculated assuming a Poisson distribution. All statistical analyses were performed using SAS statistical software, version 9.1 (SAS Institute, Cary, NC, USA), and the significance level was set at p ≤ 0.05.
Results
Incidence
Between 2000 and 2010, there were 526 (363 males, 163 females) incident cases of CD and 2125 (1289 males, 836 females) incident cases of UC, corresponding to an overall incidence rate of 0.208 and 0.838 per 100,000 person-years for CD and UC, respectively.
In 2000, the incidence rate of CD was 0.195 (95 % CI 0.113–0.276) per 100,000 persons for male and 0.092 (95 % CI 0.035–0.149) per 100,000 persons for female. In 2010, the incidence rate of CD increased to 0.318 (95 % CI 0.216–0.421) per 100,000 persons for male and 0.210 (95 % CI 0.128–0.293) per 100,000 persons for female, respectively (Fig. 1).
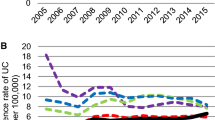
In 2000, the incidence rate of UC was 0.690 (95 % CI 0.537–0.843) per 100,000 persons for male and 0.386 (95 % CI 0.269–0.503) per 100,000 persons for female. In 2010, the incidence rate of UC was increased to 1.351 (95 % CI 1.140–1.562) per 100,000 persons for male and 0.858 (95 % CI 0.691–1.024) per 100,000 persons for female (Fig. 2).
Prevalence
The prevalence rates of CD for male and female increased from 0.441 (95 % CI 0.328–0.580) and 0.216 (95 % CI 0.138–0.321) in 2000 to 1.949 (95 % CI 1.705–2.219) and 0.883 (95 % CI 0.723–1.067) per 100,000 persons in 2010, respectively (Fig. 3).
The prevalence rates of UC for male and female increased from 1.436 (95 % CI 1.225–1.671) and 0.891 (95 % CI 0.724–1.084) in 2000 to 7.610 (95 % CI 7.119–8.125) and 4.77 (95 % CI 4.388–5.178) per 100,000 persons in 2010, respectively (Fig. 4).
Age
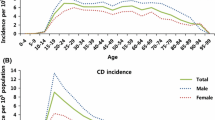
For CD, the male incidence rate peaked at age 20–29 and dropped thereafter with a second smaller peak at age 70–79, while the incidence rate for females remained low at young age and increased after age 60–69 (Fig. 5; Table 1) For UC, the male incidence rate rose at age 20–29, peaked at age 50–59, and dropped thereafter, while the female incidence rate rose at age 20–29 and peaked at age 70–79 (Fig. 6; Table 2).
Cancer in Patients with Inflammatory Bowel Disease
The confirmation of malignant cancer (ICD-9-CM code 140–195 and 200–208) events was based on the registry of Catastrophic Illness Patient Database. We excluded patients with a history of malignant cancer diagnosed before the diagnosis of IBD.
Among 519 patients with CD, six cancers were observed (one colorectal cancer, one lung cancer, and one bladder cancer in male; one breast cancer, one ovary cancer, and one thyroid cancer in female; Table 3).
Among 2098 patients with UC, 37 cancers were observed (two head and neck cancers, one gastric cancer, four colorectal cancers, two liver tumors, two cholangiocarcinomas, five lung cancers, two prostate cancers, four bladder cancers, four kidney cancers in male; one gastric cancer, one colorectal cancer, one liver tumor, one lung cancer, four breast cancers, one uterus and corpus cancer, two kidney cancers in female; Table 4).
Among the CD patients, 0.19 % had colorectal cancers (1/519). Among the UC patients, 0.24 % had colorectal cancers (5/2098). Most of them were male.
Age-standardized incidence rate of colorectal cancer is 34.75 per 100,000 (2000–2006) in Taiwan [7]. Further prospective study and standardized surveillance program are needed for the clarification of cancer risk in patient with IBD.
Discussion
The incidence and prevalence rates of CD and UC are lower in Asia than in the West. However, the incidence and prevalence rates of CD and UC in Asia have been increased rapidly in recent decades. Besides increased awareness of physician and better access to diagnostic facilities, a true increase throughout Asia is considered [2, 4, 5].
There is paucity of study about the incidence and prevalence rates of IBD in Asia. Most of them are based on hospital database. Our present study is derived from population-based database. Recent epidemiologic studies assessing the incidence and prevalence rates of IBD in Asia are summarized in Table 5.
A recent prospective, population-based study of IBD started on April 1, 2011, for 1-year period across nine countries in the Asia–Pacific region revealed that the annual overall incidence rate per 100,000 persons of IBD was 1.37 (95 % CI 1.25–1.51; UC was 0.76, CD was 0.54, and IBD-U was 0.07) in Asia. The incidence rate of IBD is still lower in the West, but it varies throughout Asia. The incidence rate of IBD is highest in Guangzhou (mainland China) (3.14), followed by Hong Kong (2.62) and Macau (2.24), which are highly urbanized [8].
Elderly patients with IBD are defined as patients over 60 years of age [9]. Elderly patients with IBD comprise two different groups: patients with the onset of disease after 60 years of age, and patients with disease onset at <60 years who have lived to an older age. In west countries, approximately 10–15 % of cases of IBD are diagnosed in patients aged >60 years, and 10–30 % of the IBD population are aged >60 years [10]. In our present data, 14.4 % of CD (76/526) and 17.3 % of UC (367/2125) patients had onset of disease after 60 years of age. As the increasing prevalence rate of IBD and the aging of the population, IBD in the elderly is an emerging problem in Taiwan. IBD in the elderly poses challenges in terms of diagnosis, uncertainties regarding therapeutic strategies, risk of drug side effects, and the high rate of comorbidities and polypharmacy [11, 12].
For CD, the peak age at the onset of CD was between 20 and 29 years, with a second peak occurred between 70 and 79 years for male patients, while there was only one peak between 60 and 69 years for female patients. For UC, the male incidence rate rose at age 20–29, peaked at age 50–59, and dropped thereafter, while the female incidence rate rose at age 20–29 and peaked at age 70–79. Elderly-onset IBD patients need to be considered differently than early-onset patients with disease starting at a younger age. In the elderly, IBD may present with atypical symptoms and may be confused with other forms of colitis at the initial presentation, such as ischemic colitis, diverticular disease, and NSAID enterocolitis [13]. The pathophysiology of elderly-onset IBD may be different from young patients. Further investigations regarding genetics, gut microbiota, environmental factors, and host immune are required for better understanding of the pathogenesis.
Population-based studies of site-specific cancer occurred in patients with IBD are lacking in Taiwan. Our present study provided data for further global comparison. IBD may be complicated by intestinal and extra-intestinal cancer, potentially because of local and systemic inflammation [14–16]. The prevalence rate of CRC in IBD patients among Asia was relative low, with a range of 0.3–1.8 %, compared to Western studies of 1.3–5 % [4, 17, 18]. A nationwide study conducted by Korea between 1970 and 2005 revealed that the overall prevalence rate of CRC in patients with UC was 0.37 % (26/7061) [19]. In addition, the estimated cumulative risk of UC-associated CRCs was 0.7 % for patients who had UC for 10 years, 7.9 % for patients who had UC for 20 years, and 33.2 % for patients who had UC for 30 years. The mean age at the time of diagnosis with CRC was 49.6 years, and the mean duration of UC prior to the development of CRC was 11.5 years [19].
As our present data, 0.19 % of CD had incident colorectal cancers and 0.24 % of UC had incident colorectal cancers. Most of them are male in gender. Wei et al. [20] reported that three cases of CRC occurred in 406 Taiwanese UC patients (0.74 %), which is higher incidence compare to our current population-based study, probably because the data were hospital-based from tertiary medical center.
It has been reported that male gender, severity, and long-standing disease are risk factors of UC-associated CRC. In Taiwan, most of the UC patients (72.4 %) could be controlled with 5-aminosalicylic acid alone, which may reflect the different disease severity from Western countries [20].
A population-based study in Europe with 15-year follow-up revealed that the prevalence rate of intestinal and extra-intestinal cancers was 9.1 %, while the prevalence rate of CRC was 1.3 %, suggesting that no overall increased risk of cancer in patient with IBD [18]. However, a 30-year analysis of a colonoscopic surveillance program for neoplasia in UC shows an overall prevalence rate of 6.3 % for CRC in 600 UC patients [21]. Concerning extra-intestinal cancers, patients with IBD also may be at increased risk of prostate cancer, pulmonary cancer, cervical dysplasia, non-Hodgkin lymphoma, non-melanoma skin cancer (NMSC), and melanoma [22, 23].
Recently, two large population-based studies in Denmark revealed that the relative risk of intestinal malignancy appears to be decreasing over time, without a concomitant increase in the risk of extra-intestinal malignancy, suggesting that immunosuppressive medications reduce the risk of intestinal malignancy by suppressing intestinal inflammation [24, 25]. The association between cancer risk and patient characteristics and history of medical treatment needed further investigation.
Conclusions
This nationwide population-based epidemiological study of IBD in Taiwan provides data for future global comparisons. Differences in gender incidence and the age at the onset may provide clues for the etiology of these diseases. Besides, CRC in patient with IBD was relatively rare in Taiwan, compared to Western countries.
References
Abraham C, Cho Judy H. Inflammatory bowel disease. N Engl J Med. 2009;361:2066–2078.
Molodecky NA, Soon S, Rabi DM, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time based on systematic review. Gastroenterology. 2012;142:46–54.
Economou M, Pappas G. New global map of Crohn’s disease: genetic, environmental, and socioeconomic correlations. Inflamm Bowel Dis. 2008;14:709–720.
Thia KT, Loftus E, Sandborn WJ, Yang SK. An update on the epidemiology of inflammatory bowel disease in Asia. Am J Gastroenterol. 2008;103:3167–3182.
Prideaux L, Kamm MA, De Cruz PP, Chan FK, Ng SC. Inflammatory bowel disease in Asia: a systematic review. J Gastroenterol Hepatol. 2012;27:1266–1280.
Kuo CFSL, Yu KH, Chou IJ, et al. Epidemiology and mortality of systemic sclerosis: a nationwide population study in Taiwan. Scand J Rheumatol. 2011;40:373–378.
Chiang C-J, Chen YC, Chen C-J, You S-L, Lai M-S, Taiwan cancer registry task force. Cancer Trends in Taiwan. Jpn J Clin Oncol. 2010;40:897–904.
Ng SC, Tang W, Ching JY, et al. Incidence and phenotype of inflammatory bowel disease based on results from the Asia-pacific Crohn’s and colitis epidemiology study. Gastroenterology. 2013;145:158–165.
Cosnes J, Gower-Rousseau C, Seksik P, et al. Epidemiology and natural history of inflammatory bowel diseases. Gastroenterology. 2011;140:1785–1794.
Gisbert JP, Chaparro M. Systematic review with meta-analysis: inflammatory bowel disease in the elderly. Alim Pharmacol Ther. 2014;39:459–477.
Joannie R, Ruel CH, Charpentier C, Gower-Rousseau C, Colombel J-F. IBD in the elderly. Inflamm Bowel Dis Monit. 2013;14:1–11.
Ananthakrishnan ANBD. Treatment of ulcerative colitis in the elderly. Dig Dis. 2009;27:327–334.
Katz S, Pardi DS. Inflammatory bowel disease of the elderly: frequently asked questions. Am J Gastroenterol. 2011;106:1889–1897.
Herrinton LJLL, Levin TR, et al. Incidence and mortality of colorectal adenocarcinoma in persons with inflammatory bowel disease from 1998 to 2010. Gastroenterology. 2012;143:382–389.
Pedersen NDD, Elkjaer M, Gamborg M, Munkholm P, Jess T. Risk of extra-intestinal cancer in inflammatory bowel disease: meta-analysis of population-based cohort studies. Am J Gastroenterol. 2010;105:1480–1487.
Jess TRC, Peyrin-Biroulet L. Risk of colorectal cancer in patients with ulcerative colitis: a meta-analysis of population-based cohort studies. Clin Gastroenterol Hepatol. 2012;10:639–645.
Ooi CJ, Fock KM, Makharia GK. The Asia-Pacific consensus on ulcerative colitis. J Gastroenterol Hepatol. 2010;25:453–468.
Katsanos KH, Pedersen N, Shuhaibar M, et al. Cancer in inflammatory bowel disease 15 years aft er diagnosis in a population-based European Collaborative follow-up study. J Crohns Colitis. 2011;5:430–442.
Kim BJYS, Kim JS, Jeen YT, et al. Trends of ulcerative colitis-associated colorectal cancer in Korea: a KASID study. J Gastroenterol Hepatol. 2009;24:667–671.
Wei S-C, Shieh MJ, Chang MC, Chang YT, Wang C-Y, Wong J-M. Long-term follow-up of ulcerative colitis in Taiwan. J Chin Med Assoc. 2012;75:151–155.
Rutter MD, Saunder BP, Wilkinson KH, et al. Thirty year analysis of a colonoscopic surveillance program for neoplasia in ulcerative colitis. Gastroenterology. 2006;130:1030–1038.
Jess T, Horvath-Puho E, Fallingborg J, Rasmussen HH, Jacobsen BA. Cancer risk in inflammatory bowel disease according to patient phenotype and treatment: a danish population-based cohort study. Am J Gastroenterol. 2013;108:1869–1876.
Long MD, Martin CF, Pipkin CA, Herfarth HH, Sandler RS, Kappelman MD. Risk of melanoma and nonmelanoma skin cancer among patients with inflammatory bowel disease. Gastroenterology. 2012;143:390–399.
Jess T, Simensen J, Jørgensen KT, Pedersen BV, Nielsen NM, Frisch M. Decreasing risk of colorectal cancer in patients with inflammatory bowel disease over 30 years. Gastroenterology. 2012;143:375–381.
Kappelman MD, Farkas D, Long MD, et al. Risk of cancer in patients with inflammatory bowel diseases: a nationwide population-based cohort study with 30 years of follow-up evaluation. Clin Gastroenterol Hepatol. 2014;12:265–273.
Zheng JJZX, Huangfu Z, Shi XH, Guo ZR. Prevalence and incidence rates of Crohn’s disease in mainland China: a meta-analysis of 55 years of research. J Dig Dis. 2010;11:161–166.
Asakura KNY, Inoue N, Hibi T, Watanabe M, Takebayashi T. Prevalence of ulcerative colitis and Crohn’s disease in Japan. J Gastroenterol. 2009;44:659–665.
Yao TMT, Hiwatashi N. Crohn’s disease in Japan: diagnostic criteria and epidemiology. Dis Colon Rectum. 2000;43:S85–S93.
Yang SKYS, Kim JH, Park JY, et al. Epidemiology of inflammatory bowel disease in the Songpa-Kangdong district, Seoul, Korea, 1986–2005: a KASID study. Inflamm Bowel Dis. 2008;14:542–549.
Sood AMV, Sood N, Bhatia AS, Avasthi G. Incidence and prevalence of ulcerative colitis in Punjab. North India Gut. 2003;52:1587–1590.
Lee YMFK, See SJ, Ng TM, Khor C, Teo EK. Racial differences in the prevalence of ulcerative colitis and Crohn’s disease in Singapore. J Gastroenterol Hepatol. 2000;15:622–625.
Acknowledgments
CJ Kuo, KH Yu, CT Chiu, MY Su, CM Hsu, CF Kuo, and HW Wang developed the study concept and design and provided the critical revision of the manuscript for important intellectual content. LC See, MJ Chiou, and JR Liu collected and interpreted the data. All authors approved the final version of the manuscript.
Conflict of interest
The authors report no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuo, CJ., Yu, KH., See, LC. et al. The Trend of Inflammatory Bowel Diseases in Taiwan: A Population-Based Study. Dig Dis Sci 60, 2454–2462 (2015). https://doi.org/10.1007/s10620-015-3630-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3630-z