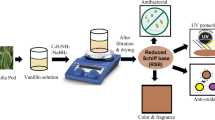
Abstract
Recently plant extracts with intrinsic properties of green, safe and eco-friendly were more often integrated with textiles to endow the fabric with multi-functions such as anti-microbial, anti-oxidation and anti-ultraviolet. Therefore, it is worthwhile to study the role of active constituents in plant extracts on the functionalization of textiles. Herein, Scutellaria baicalensis (chinese skullcap) extracts (SBE) and its main active ingredient, baicalin, were effectively combined with linen fabric by means of impregnation and ultrasonic treatment respectively. The treated linen fabrics were characterized by SEM, FTIR, XRD, TGA, DTG analysis and the relevant functions such as anti-microbial activity, antioxidant activity and ultraviolet protection ability as well as durability of functionalities were also comparatively assessed. It was found that even with the same content of baicalin, the corresponding functions of the linen fabric treated by SBE are obviously better than treated by baicalin. The advantages of the SBE over just baicalin on the functionalization of linen fabric was analyzed and verified by experiments. The conclusion of this work will guide the quantitative addition of plant extracts on textile functional treatment.
Graphic abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plant extracts are never ending natural resources derived from plants which have a complex structure of small molecular organic compounds and change significantly according to plant species and separation processes (Islam and Mohammad 2015; Balandrin et al. 1985). The essence of functional properties of plant extracts is secondary metabolites in plant active ingredients. Compared with chemical synthesis reagents, the plant active ingredients are environmentally-friendly and renewable functional agents. A diverse range of plant extracts and live plants had been successfully ultilized in synthesis (Küp et al. 2020), medication (Sotomil et al. 2019), cosmetics (Hughes et al. 2019) and food industry (Li et al. 2020). Recently more studies were focused on the functionalization of textiles using multifunctional plant extracts (Adeel et al. 2019b; Ticha et al. 2017; Sheikh and Bramhecha 2019; Sheikh et al. 2019). Sheikh et al. (2019) demonstrated the linen fabric had a good antibacterial activity, antioxidant activity and UV protection ability treated by dyeing with plant extracts from Kigelia Africana flowers. Sheikh and Bramhecha (2019) indicated that the linen fabric with a chitosan-based formulation coating could be utilized as a backbone for in situ synthesis of silver nanoparticles using tamarind seed coat extract to help in getting functionalization of linen.
As one kind of plant extracts, Scutellaria baicalensis extracts (SBE) is derived from the dry root of Scutellaria baicalensis Georgi (Lamiaceae family). The main active ingredient of SBE is the baicalin (C21H18O11, 5,6,7-trihydroxyflavone-7-O-glucuronide) (Guo et al. 2013; Zhang et al. 2014), which was proven to have antimicrobial (Wu et al. 2018; Luo et al. 2017), anti-inflammatory (Yang et al. 2016), antioxidant (Gao et al. 2001), anticancer (Li Weber 2009), antiviral (Nayak et al. 2014), anxiolytic (Liao et al. 2003) and other activities. Flavonoids as the major phytochemicals of baicalin existing in SBE (Zhang et al. 2019), are polyphenolic compounds which generally appear in the form of glycosylated derivatives. Flavonoids can inhibit the growth of microorganisms (Rauha et al. 2000; Cushnie and Lamb 2005) and they are also the major compounds in terms of radical scavenging activity (Miliauskas et al. 2004) and UV protection (Mota et al. 2019). Therefore, researches were concentrated on functional treatment of textiles with plant extracts rich in flavonoids such as Scutellaria baicalensis and cactus. Zhou et al. (2016) combined the baicalin with silk fabric through the electrostatic interactions between the ionized carboxyl groups in baicalin and the positively charged amino groups of protein macromolecule of silk, the treated silk fabric exhibited good UV protection. Xu et al. (2017) demonstrated that the silk fibroin grafted carboxylic cotton fabric treated by cactus flavonoid extracts can achieve a highly inhibitory effect against both Staphylococcus aureus and Escherichia coli.
Linen, made from the fiber from flax plants is inherently with good hygroscopicity, moisture conductivity and faint antibacterial properties. However, its vulnerability to damp-breeding moulds and lack of UV protection against sunburn limit its further development as functional textiles (Sheikh and Bramhecha 2018). The integration of plant extracts with the linen fiber can be functionalized on the basis of their advantageous properties. However, most species of plant extracts were less applied to the linen fibers based fabric functionalization rather than to proteinaceous fibers-based fabric (wool, silk) (Adeel et al. 2019a, b; Ticha et al. 2017; Shabbir et al. 2018) because of its high degree of inter-molecular binding and dense structure (Arik 2020). Fabric treatment by ultrasound is an ecofriendly and energy-efficient method to improve the integration of plant extracts with the linen fabric (Ma et al. 2019; Abral et al. 2019; Ticha et al. 2017), because ultrasonic treatment destroys the inter-fiber bonds among the linen molecular layers, resulting in “cracking” or “erosion” on the surface of linen fibers (Wong et al. 2009). Moreover, ultrasound-assistance also accelerates the homogenization of SBE to reduce aggregation of bulky molecules which could increase more activated hydrophilic groups between molecules of SBE and linen fibers (Abou-Okeil et al. 2010). As a result the interaction between plant extracts and macromolecules of linen can be improved. Another challenge is the lack of criteria to assess the inter-relationship between plant extracts and their active ingredients in textile functional treatment due to the great difference of active ingredient in plant origin and taxa even in same species, which will affect the quantitative addition and waste of plant extracts on the functionalization of textiles. The mentioned problems also hinder the development of the plant extracts as a potential perspective in functional applications and energy conservation.
Herein, the SBE and baicalin were effectively combined with the linen fabric by means of impregnation and ultrasonic treatment respectively. The treated linen fabrics were characterized by SEM, FTIR, XRD, TGA, DTG analysis, and their multi-functions as anti-bacterial, anti-mould, anti-oxidant and ultraviolet protection abilities were comparatively assessed to find out the inter-relationship between SBE and baicalin in textile functional treatment. Our work will develop a new production for regulating equivalence dosage of plant extracts and plant-derived chemicals for control of plant extracts additions on functionalization of textiles without contamination and toxicity. In the future, biologically active plant-derived chemicals with particular functions can be expected to play an increasingly significant role in the commercial development on textile functionalization to reduce unnecessary waste in mass production.
Materials and methods
Materials
Ready-for-dyeing bleached linen fabric (EPI = 50, PPI = 48, GSM = 167) was obtained from Jin Tai Co. Ltd. CHN. Baicalin sample with the purity of 95 wt% was supplied by Shanghai Aladdin Biochemical Technology Co., Ltd. CHN, and the SBE was supplied by Lin Cao science Co., Ltd. CHN. The bacterial strains and fungal strains were obtained from Fu Chi science Co., Ltd. CHN. The DPPH (1,1-diphenyl-2-picrylhydrazyl, C18H12N5O6, ≥ 97.0 wt%) was supplied by Phygene Biotechnology Co., Ltd. CHN. All other auxiliary chemical reagents were supplied by National Pharmaceutical Group Chemical Testing Co., Ltd CHN and were used as received.
Measurement for baicalin content of SBE
The baicalin content of SBE were measured by high-performance liquid chromatographic (HPLC) method using Agilent 1100 HPLC analyzer (USA). The baicalin content in SBE can be calculated by the ratio of the absorption peak area at 277 nm wavelength compared with baicalin sample with the purity of 95 wt%. The baicalin content of SBE used in this work was 5.32 wt% (Support Information S1).
Treatment of linen fabrics
As shown in Scheme 1, firstly the bleached linen fabric as control sample (s0) was impregnated with deionized water for 30 min assisted by ultrasonic treatment and dried as the pre-treated sample (s1) for use. 300 g SBE and baicalin aqueous solution with same baicalin contents of 0.27 wt%, 0.53 wt%, 0.80 wt%, 1.06 wt% marked B2 to B9 were prepared using SBE and baicalin sample as solutes respectively. Then the pre-treated linen fabric (s1) was impregnated as prepared solution (B2–B9) respectively and followed by 60 min ultrasonic treatment (40 Hz, 240 W) at different temperatures from 40 to 100 °C with interval 10 °C to find the optimal treatment temperature using ultrasonic cleaner machine (Jie Meng Cleaning Equipment Co., Ltd. CHN, Support Information S2). At last treated linen fabrics marked s2 to s9 were rinsed repeatedly with deionized water to remove the unabsorbed SBE or baicalin and dried at 80 °C for 10 min. The un-treated bleached linen fabric marked control sample (s0) was used for comparison (Table 1, Support Information S3).
Characteristics of treated linen fabrics
The surface morphologies of the treated linen fabric s2 (by SBE) and s6 (by baicalin) were studied by scanning electron microscope (SEM Vega3tescan, CZ). The molecular structures and crystallographic structures of s2 and s6 were analyzed by FT-IR spectrophotometer (Thermo Scientific Nicolet iS50, USA) using KBr disk method in the range from 4000 to 400 cm−1 at a resolution of 2 cm−1. The crystallographic structures of s2 and s6 were characterized by X-ray diffraction diffractometer (XRD Rigaku Smartlab Horiba Co. JPN) with a CuKα radiation (λ = 0.15405 nm) with a scanning rate of 2° min−1 at 40 kV and 40 mA. The un-treated linen fabric (s0) was also analyzed for comparison.
Measurement for the physical properties of treated linen fabrics
The thermal stabilities of treated linen fabrics s2 and s6 were determined by thermo-gravimetric analyzer (TGA, TG209F3, GER) with a heating rate of 10 °C min−1 under N2 environment from 40 °C to 900 °C as the test temperature range. The mechanical properties of treated linen fabrics s2 to s9 were tested by using Tensile Testing Machine (Hd026pc-500 Hong Da Inc. CHN) according to China National Standard (GB/T 3923.1-2013) with loading speed of 20 mm min−1 and each sample was tested three times to obtain the average value. The un-treated linen fabric (s0) was also measured for comparison.
Measurement for the multifunction of treated linen fabrics
The antibacterial activities of the untreated linen fabric s0, the pre-treated linen fabric s1 and treated linen fabrics s2 to s9 as well as their after-washing samples were quantitatively evaluated by shake flask method according to China National Standard (GB/T 20944.3-2008). Qualitative analysis was measured by agar diffusion plate method according to China National Standard (GB/T 20944.1-2007) to determine the distinct bacterial inhibition zone of sample s0, s2 to s9 (Support Information S4).
The anti-mould activities of the pre-treated linen fabric (s1) and treated linen fabrics s2 to s9 were quantitatively evaluated by petri dish test method according to China national standard (GB/T 24346-2009), and the un-treated linen fabric (s0) was also measured for comparison (Support Information S5).
The DPPH radical scavenging activities of treated linen fabrics s2 to s9 were measured by testing its corresponding treatment solution B2 to B9 according to the method as described by Gao et al. (1999) and calculated the value according to the method described by Hong KH et al. (Hong et al. 2012) (Support Information S6).
The ultraviolet protection abilities of the pre-treated linen fabric s1, treated linen fabrics s2 to s9 as well as the after washing samples were evaluated according to China National Standard (GB/T 18830-2009), and the un-treated linen fabric (s0) was also measured for comparison (Support Information S7).
The durability of functionalities were evaluated by the antibacterial activity and the ultraviolet protection ability before and after washing according to Part 10.1 of China National Standard (GB/T 20944.3-2008) (Support Information S8).
Results and discussion
Characterization of treated linen fabrics
Compared with the untreated linen fabric, “cracking” or “erosion” can be clearly seen on the surface of fibers after ultrasonic treatment, which indicated ultrasonic treatment can destroy the dense surface structure of the linen fiber (Fig. 1a–d). After subsequent impregnation with SBE and baicalin aqueous solution, it can be clearly seen that the particulate SBE and baicalin were closely deposited on the surface of linen fibers (Fig. 1e–h), which demonstrated the existing interaction between SBE/baicalin and the linen fiber most likely by hydrogen bonding through their reactive groups (e.g. hydroxyl, carboxyl) (Chung et al. 2004; Li et al. 2017). Since the SBE contains other chemical components than baicalin (e.g. baicalein, wogonoside) which have a large particle size and interact with each other to cause agglomeration, so the particle size of SBE are bigger than baicalin sample (Fig. 1i, j) (Zhou et al. 2018; Chirikova et al. 2010).
SEM images of a, b the twining fiber in the untreated linen fabric and its high magnification, c, d the twining fiber in the pre-treated linen fabric and its high magnification, e, f the twining fiber in the linen fabric treated by SBE and its high magnification, g, h the twining fiber in the treated linen fabric by baicalin and its high magnification, i, j the particle size of SBE and baicalin sample
FT-IR spectra of the untreated linen fabric (s0), baicalin sample and the treated linen fabric by baicalin (s6) were described in Fig. 2. A broad peak centered 3300 cm−1 assigned to hydrogen-bonded (O–H) stretching and a broad peak around at 2800–3000 cm−1 assigned to (C–H) stretching are related to characteristic peak of cellulose macromolecules for the untreated linen fabric (Chung et al. 2004). A peak at 3491 cm−1 and 3552 cm−1 assigned to phenolic (–OH), a peak at 1660 cm−1 assigned to the carbonyl (C=O) stretching and a peak at 1726 cm−1 assigned to carboxyl (–COOH) are related to characteristic peak of baicalin (Looker and Hanneman 1962). The diminished peaks at 3491 cm−1 and 3552 cm−1 and detectable peaks at 1660 cm−1 and 1726 cm−1 of s6, which indicated baicalin can combined with the linen fabric by formation of inter-molecular hydrogen bonds between reactive groups in baicalin (e.g. hydroxy, carboxyl) and the hydroxyl groups in the macromolecule of linen fibers (Li et al. 2017). Moreover, in weakly acidic baicalin solution (pH 6.8–7.0, Table 1), baicalin can also combine with negative zeta potential of cellulose macromolecules through electrostatic interaction (Muderrisoglu and Yesil-Celiktas 2019; Rekaby et al. 2013).
The crystalline structures of the untreated linen fabric (s0) and treated linen fabrics (s2, s6) were determined by X-ray diffraction patterns. Figure 3 exhibited the peaks of (1-10), (110) and (200) that correspond to the characteristic diffraction peaks of cellulose I. The peaks appearing at the same position of s2 and s6 indicated that the crystallization characteristics of cellulose I were almost reserved whether treated by SBE or baicalin solution. Besides, the peak widths at half maximum height (pwhm) of s2 or s6 all slightly increased compared with the pwhm of s0, which meant the crystallinity index of s2 or s6 was slightly smaller than s0 according to Scherrer equation (Ling et al. 2019). The result of XRD demonstrated that short-time ultrasonic treatment played a positive effect on reducing crystallinity of cellulose fiber and improvement of combination between SBE/baicalin with the linen fabric (Cui et al. 2016; French and Cintrón 2013; Tang et al. 2014).
The thermal stability and mechanical property of treated linen fabrics
The thermo-stabilities of the untreated linen fabric (s0) and treated linen fabrics (s2, s6) were evaluated by the thermogravimetry analysis (TGA) and differential thermogravimetry (DTG). As shown in Fig. 4a, b, the initial decomposition temperature of s2 decreased from 230 °C of s0 to 200 °C, and the maximum decomposition temperature also decreased from 365 °C of s0 to 361 °C, which indicated that the thermal stability of linen fabric decreased due to the slight reduction in crystallinity of linen fiber after ultrasonic treatment (Liu et al. 2015). In addition, the destruction of the network structure between pectin and linen and the elimination of other non-cellulosic constituents like waxes in the ultrasonic treatment may determine the treated linen fabric will be more exposed to the thermal degradation than the untreated linen fabric, which can also lead to a decrease of the decomposition temperature (Dochia et al. 2018). While the decomposition temperature and the maximum decomposition temperature of s2 were higher than s6 maybe because other compounds (e.g. baicalein, tannins) in SBE tend to establish more hydrogen bonds with the linen fiber macromolecules resulting in higher thermostability (Chirikova et al. 2010; Teli and Pandit 2017).
The fracture strength and fracture elongation were used to evaluate the mechanical properties of treated linen fabrics (s2 to s9) and the untreated linen fabric (s0) for comparison. As shown in Fig. 5, the average of fracture strength and fracture elongation on dry state were smaller than on wet state, which was consistent with the basic mechanical feature of the linen fiber (Bourmaud et al. 2010). The fracture strength and fracture elongation of s2 to s9 were all smaller than s0 and further reduced with increase of SBE content (s2 to s5) or baicalin content (s6 to s9), which indicated that ultrasonic processing can destroy the dense structure of the linen fiber and more hydrogen bonds between the intra-molecules of the linen fiber were broken with increase of SBE or baicalin contents (Jarvis 2003). Despite of at the same baicalin content, the fracture strength and fracture elongation of linen fabrics treated by SBE (s2 to s5) were slightly lower than those of linen fabrics treated by baicalin (s6 to s9), because the other compounds (e.g. organic acids, phenolic acids, tannins) in SBE can exacerbate inter-molecular hydrogen bonds cleavage between linen fibers and lead to more deterioration of the mechanical properties (Chirikova et al. 2010).
Functionalities of treated linen fabrics
Antimicrobial activity
The mechanism of antibacterial and anti-mould activity is that baicalin or other main phytochemicals as flavonoids contained in SBE can inhibit the activity of enzymes in bacteria and fungi and destroy protein genetic materials such as inhibit DNA or RNA synthesis in Proteus vulgaris and Staphylococcus aureus (Mori et al. 1987), so that further replication of genetic material does not occur to prevent the growth of bacteria and fungi (Yu et al. 2015; Daglia 2012). In addition, flavonoids also can reduce membrane fluidity of various bacteria to inhibit the cytoplasmic membrane function (Mori et al. 1987), and inhibit the energy metabolism to interfere with the life activities of bacteria and fungi (Cushnie and Lamb 2005). Antimicrobial activities were evaluated by two indexes as anti-mould level and antibacterial rate. As shown in Fig. 6 and Tables 2, 3, the antimicrobial activities of treated linen fabrics (s2 to s9) were better than the untreated linen fabric (s0), and due to the synergism of baicalin with other components in SBE the antimicrobial activities of treated linen fabrics by SBE (s2 to s5) were slightly better than treated linen fabrics by baicalin (s6 to s9) with the same baicalin content. Furthermore both SBE and baicalin had better inhibitory effect on S. aureus than on E. coli. The results demonstrated that baicalin was a highly effective antimicrobial agent and can achieve antibacterial rate above 73% and anti-mould level 2 with only 0.27 wt% content. As trends in the demand for antimicrobial textiles continue to rise, the effective control of antimicrobial effect with low dosage and harmless to human body becomes ever more important for the antimicrobial materials. These results may also suggest that SBE and baicalin could be used as green, natural effective antimicrobial agents under low content on textiles functionalization.
Effects of SBE and baicalin on extinction of DPPH radical
DPPH radical is a stable radical which could be quenched by antioxidant molecules causing the purple color to fade or disappear (Support Information S3), resulting in a decrease in absorbance peak at the 517 nm band. Phenolic hydroxyls in baicalin are considered to exhibit high antioxidant activity (Zhou et al. 2018). The flavonoids mainly containing in baicalin as active ingredients of SBE are responsible for the most prominent compound of radical-scavenging capacity due to its structural feature of a diorthohydroxyl group in A-ring (Gao et al. 1999). Moreover tannins are also possible that both of above suggested mechanisms working in tandem as polyphenolic compounds in SBE because of its phenolic hydrogen structure in their molecules (Amarowicz et al. 2004). Figure 7a, b shown the effects of treated linen fabrics by SBE (s2 to s5) and baicalin (s6 to s9) with different absorbance and inhibition rate of DPPH radical. With increase of baicalin content, the extinction of DPPH radical activity increase whether it is treated by SBE or baicalin. While linen fabrics treated by SBE (s2 to s5) have better antioxidant activity than treated by baicalin (s6 to s9) after 30 min reaction because SBE contains other free radical scavengers (e.g. baicalein, wogonoside) than baicalin (Gao et al. 1999).
UV Protection
Flavonoids are known as UV-absorbing compounds and wavelength-selective for UV-B that may prevent the accumulation of UV-B-induced DNA damage, protecting cells against UV damage from UV radiation-induced peroxidation (Dias et al. 2019; Mota et al. 2019; Fernandes et al. 2017; Anouar et al. 2012). Flavonoids as main chemical constituents of baicalin containing in SBE can link to linen macromolecules to provide good UV protection (Chirikova et al. 2010; Teli and Pandit 2017). As shown in Fig. 8, compared with the untreated linen fabric (s0), treated linen fabrics (s2 to s9) exhibited remarkably decreased UV transmittance over the range of 280–400 nm (UVA, UVB), and remarkably increased Ultraviolet Protection Factor (UPF) with increases of SBE and baicalin content. Furthermore, the UPF of linen fabrics treated by SBE (s2 to s5) are better than treated by baicalin (s6 to s9) due to synergistic effect of other flavonoids in SBE (e.g. baicalin, baicalein, wogonoside) (Gao et al. 1999; Mota et al. 2019).
Durability of functionalities
The durability of functionalized textiles was evaluated by comparing functionalities before and after washing. The antibacterial activities and ultraviolet protection factor (UPF) of treated linen fabrics (s2 to s9) after 10 washing cycles according to China National Standard (GB/T 20944.3-2008) were measured. Compared with the untreated linen fabric (s0), the pre-treated linen fabric (s1), and treated linen fabrics before washing (s2 to s9), although antibacterial activities and ultraviolet protection factor (UPF) of treated linen fabrics (e.g. s3) after 10 washing cycles were visibly reduced from 90.15% to 80.15% (S. aureus), from 87.21% to 76.16% (E. coli), from 22.03 to 14.75 (UPF), it was still 10% higher than the untreated and pre-treated linen fabric (Fig. 9). These results further confirmed the SBE and baicalin can be used to functionalize textiles assisted by ultrasonic treatment because of the considerable durability of functions.
The equivalence relationship between SBE and baicalin on functionalization of textiles
Comparative analysis of the functions of linen fabrics treated by SBE and baicalin with at the corresponding equivalent baicalin contents (0.27 wt%, 0.53 wt%, 0.80 wt%, 1.06 wt%), it was found that under the same baicalin content, the functions of linen fabrics treated by SBE were generally better than by baicalin due to synergistic effects of other components in SBE (e.g. baicalein, wogonoside) on textiles functionalization (Lu et al. 2011; Gao et al. 1999). The equivalent relationship between SBE and baicalin for a certain function can be determined by quantifiable functional indicators such as antibacterial activity rate, DPPH radical inhibition rate and UPF. As shown in Figs. 10, 11 and 12, if at same antibacterial activity rate (S. aureus, E. coli) of 90%, baicalin content 0.7 wt%, 0.86 wt% and SBE content 9.94 wt%, 12.8 wt% (baicalin content 0.53 wt%, 0.68 wt%) are functionally equivalent. If at same DPPH radical inhibition rate of 50% (0 min), 80% (30 min), baicalin content 0.69 wt%, 0.71 wt% and SBE content 10.7 wt%, 9.23 wt% (baicalin content 0.57 wt%, 0.49 wt%) are functionally equivalent. If at same UPF of 30, baicalin content 0.85 wt% and SBE content 15.42 wt% (baicalin content 0.82 wt%) are functionally equivalent. The equivalent relationship between these SBE and baicalin can guide the quantitative addition and waste reduction when functionalizing textiles with SBE.
Experimental verification
In order to further verify the equivalent relationship between SBE and baicalin on the functionalization of textiles. The SBE and baicalin aqueous solution with different baicalin content according to same functions were prepared to treat linen fabrics respectively and the corresponding functions were measured subsequently (Support Information S9). As shown in Table 4, the bacteriostatic rates (S. aureus) of linen fabrics treated by baicalin with baicalin content of 0.71 wt% and by SBE with baicalin content of 0.53 wt% were very close (89.92%, 90.29%), and the inhibition of DPPH (30 min) of linen fabrics treated by baicalin with baicalin content of 0.71wt % and by SBE with baicalin content of 0.49 wt% were very close (80.23%, 80.01%). Meanwhile linen fabrics were treated by baicalin and SBE with almost the same baicalin content (0.71 wt%, 0.68 wt%), the bacteriostatic rates of treated linen fabrics by SBE were significantly better than by baicalin. The same result was obtained by other functional evaluations, which confirmed the credibility of the previously conclusions of equivalent relationship between SBE and baicalin on functionalization of textiles.
Conclusion
Hence, the SBE and its the active ingredients baicalin were effectively combined with linen fabrics by means of impregnation and ultrasonic treatment respectively. Through the comparative analysis of the functions of above-treated linen fabrics, it was also found that linen fabrics treated by SBE exhibited better antimicrobial activity, antioxidant activity and ultraviolet protection ability than treated by just baicalin. Subsequently, the equivalent relationship between SBE and baicalin on functionalization of textiles was further studied and verified experimentally. The conclusion of this work has very practical guiding significance for the quantitative addition and waste reduction of plant extracts on the functionalization of textiles.
References
Abou-Okeil A, El-Shafie A, Zawahry MME (2010) Ecofriendly laccase–hydrogen peroxide/ultrasound-assisted bleaching of linen fabrics and its influence on dyeing efficiency. Ultrason Sonochem 17(2):383–390. https://doi.org/10.1016/j.ultsonch.2009.08.007
Abral H, Hartono A, Hafizulhaq F, Handayani D, Sugiarti E, Pradipta O (2019) Characterization of PVA/cassava starch biocomposites fabricated with and without sonication using bacterial cellulose fiber loadings. Carbohyd Polym 206:593–601. https://doi.org/10.1016/j.carbpol.2018.11.054
Adeel S, Rehman FU, Iqbal MU, Habib N, Kiran S, Zuber M, Zia KM, Hameed A (2019a) Ultrasonic assisted sustainable dyeing of mordanted silk fabric using arjun (Terminalia arjuna) bark extracts. Environ Prog Sustain 38(s1):S331–S339. https://doi.org/10.1002/ep.13048
Adeel S, Rehman FU, Zia KM, Azeem M, Kiran S, Zuber M, Irfan M, Qayyum MA (2019b) Microwave-supported green dyeing of mordanted wool fabric with Arjun Bark Extracts. J Nat Fibers. https://doi.org/10.1080/15440478.2019.1612810
Amarowicz R, Pegg RB, Rahimi-Moghaddam P, Barl B, Weil JA (2004) Free-radical scavenging capacity and antioxidant activity of selected plant species from the Canadian prairies. Food Chem 84(4):551–562. https://doi.org/10.1016/s0308-8146(03)00278-4
Anouar EH, Gierschner J, Duroux JL, Trouillas P (2012) UV/Visible spectra of natural polyphenols: a time-dependent density functional theory study. Food Chem 131(1):79–89. https://doi.org/10.1016/j.foodchem.2011.08.034
Arik B (2020) Characterization and wrinkle resistance enhancement by Sol–Gel method of variously pretreated linen fabrics. Fiber Polym 21(1):82–89. https://doi.org/10.1007/s12221-020-9329-6
Balandrin MF, Klocke JA, Wurtele ES, Bollinger WH (1985) Natural plant chemicals: sources of industrial and medicinal materials. Science 228(4704):1154–1160. https://doi.org/10.1126/science.3890182
Bourmaud A, Morvan C, Baley C (2010) Importance of fiber preparation to optimize the surface and mechanical properties of unitary flax fiber. Ind Crop Prod 32(3):662–667. https://doi.org/10.1016/j.indcrop.2010.08.002
Chirikova NK, Olennikov DN, Tankhaeva LM (2010) Pharmacognostic study of aerial parts of Baikal skullcap (Scutellaria baicalensis Georgi). Russ J Bioorg Chem 36(7):909–914. https://doi.org/10.1134/S1068162010070198
Chung C, Lee M, Choe EK (2004) Characterization of cotton fabric scouring by FT-IR ATR spectroscopy. Carbohyd Polym 58(4):417–420. https://doi.org/10.1016/j.carbpol.2004.08.005
Cui S, Zhang S, Ge S, Xiong L, Sun Q (2016) Green preparation and characterization of size-controlled nanocrystalline cellulose via ultrasonic-assisted enzymatic hydrolysis. Ind Crop Prod 83:346–352. https://doi.org/10.1016/j.indcrop.2016.01.019
Cushnie TPT, Lamb AJ (2005) Antimicrobial activity of flavonoids. Int J Antimicrob Ag 26(5):343–356. https://doi.org/10.1016/j.ijantimicag.2005.09.002
Daglia M (2012) Polyphenols as antimicrobial agents. Curr Opin Biotechnol 23(2):174–181. https://doi.org/10.1016/j.copbio.2011.08.007
Dias MC, Pinto DCGA, Freitas H, Santos C, Silva AMS (2019) The antioxidant system in Olea europaea to enhanced UV-B radiation also depends on flavonoids and secoiridoids. Phytochemistry 170:112199. https://doi.org/10.1016/j.phytochem.2019.112199
Dochia M, Chambre D, Gavrilaş S, Moisă S (2018) Characterization of the complexing agents’ influence on bioscouring cotton fabrics by FT-IR and TG/DTG/DTA analysis. J Therm Anal Calorim 132:1489–1498. https://doi.org/10.1007/s10973-018-7089-y
Fernandes AS, Mazzei JL, Oliveira CG, Evangelista H, Marques MRC, Ferraz ERA, Felzenszwalb I (2017) Protection against UV-induced toxicity and lack of mutagenicity of Antarctic sanionia uncinata. Toxicology 376:126–136. https://doi.org/10.1016/j.tox.2016.05.021
French AD, Cintrón MS (2013) Cellulose polymorphy, crystallite size, and the Segal Crystallinity Index. Cellulose 20:583–588. https://doi.org/10.1007/s10570-012-9833-y
Gao Z, Huang K, Yang X, Xu H (1999) Free radical scavenging and antioxidant activities of flavonoids extracted from the radix of Scutellaria baicalensis Georgi. BBA 1472(3):643–650. https://doi.org/10.1016/S0304-4165(99)00152-X
Gao Z, Huang K, Xu H (2001) Protective effects of flavonoids in the roots of Scutellaria baicalensis georgi against hydrogen peroxide-induced oxidative stress in hs-sy5y cells. Pharmacol Res 43(2):173–178. https://doi.org/10.1006/phrs.2000.0761
Guo M, Zhang N, Li D, Liang D, Liu Z, Li F, Fu Y, Cao Y, Deng X, Yang Z (2013) Baicalin plays an anti-inflammatory role through reducing nuclear factor-κb and p38 phosphorylation in S. aureus-induced mastitis. Int Immunopharmacol 16(2):125–130. https://doi.org/10.1016/j.intimp.2013.03.006
Hong KH, Bae JH, Jin SR, Yang JS (2012) Preparation and properties of multi-functionalized cotton fabrics treated by extracts of gromwell and gallnut. Cellulose 19(2):507–515. https://doi.org/10.1007/s10570-011-9613-0
Hughes K, Ho R, Butaud JF, Filaire E, Ranouille E, Berthon JY, Raharivelomanana P (2019) A selection of eleven plants used as traditional Polynesian cosmetics and their development potential as anti-ageing ingredients, hair growth promoters and whitening. J Ethnopharmacol 245:112159. https://doi.org/10.1016/j.jep.2019.112159
Islam SU, Mohammad F (2015) Natural colorants in presence of anchors so-called mordants as promising coloring and antimicrobial agents for textile materials. ACS Sustain Chem Eng 3(10):2361–2375. https://doi.org/10.1021/acssuschemeng.5b00537
Jarvis M (2003) Chemistry: cellulose stacks up. Nature 426(6967):611–612. https://doi.org/10.1038/426611a
Küp FÖ, Çoşkunçay S, Duman F (2020) Biosynthesis of silver nanoparticles using leaf extract of Aesculus hippocastanum (horse chestnut): evaluation of their antibacterial, antioxidant and drug release system activities. Mat Sci Eng C-Mater 107:110207. https://doi.org/10.1016/j.msec.2019.110207
Li Weber M (2009) New therapeutic aspects of flavones: the anticancer properties of Scutellaria and its main active constituents Wogonin, Baicalein and Baicalin. Cancer Treat Rev 35(1):57–68. https://doi.org/10.1016/j.ctrv.2008.09.005
Li J, Jiang Q, Deng P, Chen Q, Yu M, Shang J, Li W (2017) The formation of a host-guest inclusion complex system between β-cyclodextrin and baicalin and its dissolution characteristics. J Pharm Pharmacol 69(6):663–674. https://doi.org/10.1111/jphp.12708
Li YX, Zhang C, Pan SY, Chen L, Liu M, Yang KL, Zeng XB, Tian J (2020) Analysis of chemical components and biological activities of essential oils from black and white pepper (Piper nigrum L.) in five provinces of southern China. LWT-Food Sci Technol 117:108644. https://doi.org/10.1016/j.lwt.2019.108644
Liao JF, Hung WY, Chen CF (2003) Anxiolytic-like effects of baicalein and baicalin in the Vogel conflict test in mice. Eur J Pharmacol 464(2–3):141–146. https://doi.org/10.1016/S0014-2999(03)01422-5
Ling Z, Wang T, Makarem M, Cintrón MS, Cheng HN, Kang X, Bacher M, Potthast A, Rosenau T, King H, Delhom CD, Nam S, Edwards JV, Kim SH, Xu F, French AD (2019) Effects of ball milling on the structure of cotton cellulose. Cellulose 26:305–328. https://doi.org/10.1007/s10570-018-02230-x
Liu Y, Hu J, Gao J, Guo H, Chen Y, Cheng Q, Via BK (2015) Wood veneer dyeing enhancement by ultrasonic-assisted treatment. BioResources 10(1):1198–1212. https://doi.org/10.15376/biores.10.1.1198-1212
Looker JH, Hanneman WW (1962) Physical and chemical properties of hydroxyflavones. i. Infrared absorption spectra of monohydroxyflavones and their o-methyl and o-acetyl derivatives1,2. J Org Chem 27(2):381–389. https://doi.org/10.1021/jo01049a010
Lu Y, Joerger R, Wu C (2011) Study of the chemical composition and antimicrobial activities of ethanolic extracts from roots of Scutellaria baicalensis georgi. J Agric Food Chem 59(20):10934–10942. https://doi.org/10.1021/jf202741x
Luo J, Dong B, Wang K, Cai S, Liu T, Cheng X, Lei D, Chen Y, Li Y, Kong J, Chen Y (2017) Baicalin inhibits biofilm formation, attenuates the quorum sensing-controlled virulence and enhances Pseudomonas aeruginosa clearance in a mouse peritoneal implant infection model. PLoS ONE 12(4):e0176883. https://doi.org/10.1371/journal.pone.0176883
Ma X, Wei Y, Wang S, Zuo X, Shen B (2019) Sustainable ultrasound-assisted ultralow liquor ratio dyeing of cotton fabric with natural turmeric dye. Text Res J 90(5–6):685–694. https://doi.org/10.1177/0040517519878793
Miliauskas G, Venskutonis PR, Van Beek TA (2004) Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem 85(2):231–237. https://doi.org/10.1016/j.foodchem.2003.05.007
Mori A, Nishino C, Enoki N, Tawata S (1987) Antibacterial activity and mode of action of plant flavonoids against Proteus vulgaris and Staphylococcus aureus. Phytochemistry 26(8):2231–2234. https://doi.org/10.1016/S0031-9422(00)84689-0
Mota MD, Costa RYS, Guedes AAS, Silva LCRCE, Chinalia FA (2019) Guava-fruit extract can improve the UV-protection efficiency of synthetic filters in sun cream formulations. J Photochem Photobiol, B 201:111639. https://doi.org/10.1016/j.jphotobiol.2019.111639
Muderrisoglu C, Yesil-Celiktas O (2019) High-yield biocatalysis of baicalein 7-O-β-d-glucuronide to baicalein using soluble helix pomatia-derived β-glucuronidase in a chemically defined acidic medium. Catal Lett 149(6):1701–1709. https://doi.org/10.1007/s10562-019-02745-3
Nayak MK, Agrawal AS, Bose S, Naskar S, Bhowmick R, Chakrabarti S, Sarkar S, Chawla-Sarkar M (2014) Antiviral activity of baicalin against influenza virus H1N1-pdm09 is due to modulation of NS1-mediated cellular innate immune responses. J Antimicrob Chemother 69(5):1298–1310. https://doi.org/10.1093/jac/dkt534
Rauha JP, Remes S, Heinonen M, Hopia A, Kähkönen M, Kujala T, Pihlaja K, Vuorela H, Vuorela P (2000) Antimicrobial effects of Finnish plant extracts containing flavonoids and other phenolic compounds. Int J Food Microbiol 56(1):3–12. https://doi.org/10.1016/S0168-1605(00)00218-X
Rekaby M, Abd-El Thalouth JI, EL-Salam SHA (2013) Improving reactive ink jet printing via cationization of cellulosic linen fabric. Carbohyd Polym 98(2):1371–1376. https://doi.org/10.1016/j.carbpol.2013.07.048
Shabbir M, Rather LJ, Mohammad F (2018) Economically viable UV-protective and antioxidant finishing of wool fabric dyed with Tagetes erecta flower extract: valorization of marigold. Ind Crop Prod 119:277–282. https://doi.org/10.1016/j.indcrop.2018.04.016
Sheikh J, Bramhecha I (2018) Multifunctional modification of linen fabric using chitosan-based formulations. Int J Biol Macromol 118:896–902. https://doi.org/10.1016/j.ijbiomac.2018.06.150
Sheikh J, Bramhecha I (2019) Multi-functionalization of linen fabric using a combination of chitosan, silver nanoparticles and Tamarindus Indica L. seed coat extract. Cellulose 26(16):8895–8905. https://doi.org/10.1007/s10570-019-02684-7
Sheikh J, Singh N, Pinjari D (2019) Sustainable functional coloration of linen fabric using Kigelia Africana flower colorant. J Nat Fibers. https://doi.org/10.1080/15440478.2019.1658260
Sotomil JM, Münchow EA, Pankajakshan D, Spolnik KJ, Ferreira JA, Gregory RL, Bottino MC (2019) Curcumin-A natural medicament for root canal disinfection: effects of irrigation, drug release, and photoactivation. J Endod 45(11):1371–1377. https://doi.org/10.1016/j.joen.2019.08.004
Tang Y, Yang S, Zhang N, Zhang J (2014) Preparation and characterization of nanocrystalline cellulose via low-intensity ultrasonic-assisted sulfuric acid hydrolysis. Cellulose 21(1):335–346. https://doi.org/10.1007/s10570-013-0158-2
Teli MD, Pandit P (2017) A novel natural source sterculia foetida fruit shell waste as colorant and ultraviolet protection for linen. J Nat Fibers 15(3):337–343. https://doi.org/10.1080/15440478.2017.1328327
Ticha MB, Meksi N, Attia HE, Haddar W, Guesmi A, Jannet HB, Mhenni MF (2017) Ultrasonic extraction of Parthenocissus quinquefolia colorants: extract identification by HPLC-MS analysis and cleaner application on the phytodyeing of natural fibres. Dyes Pigments 141:103–111. https://doi.org/10.1016/j.dyepig.2017.02.002
Wong SS, Kasapis S, Tan YM (2009) Bacterial and plant cellulose modification using ultrasound irradiation. Carbohyd Polym 77(2):280–287. https://doi.org/10.1016/j.carbpol.2008.12.038
Wu SC, Chu XL, Su JQ, Cui ZQ, Zhang ZJ et al (2018) Baicalin protects mice against Salmonella typhimurium infection via the modulation of both bacterial virulence and host response. Phytomedicine 48:21–31. https://doi.org/10.1016/j.phymed.2018.04.063
Xu Y, Chen D, Du Z, Li J, Wang Y, Yang Z, Peng F (2017) Structure and properties of silk fibroin grafted carboxylic cotton fabric via amide covalent modification. Carbohyd Polym 161:99–108. https://doi.org/10.1016/j.carbpol.2016.12.071
Yang W, Li H, Cong X, Wang X, Tian W et al (2016) Baicalin attenuates lipopolysaccharide induced inflammation and apoptosis of cow mammary epithelial cells by regulating NF-κB and HSP72. Int Immunopharmacol 40:139–145. https://doi.org/10.1016/j.intimp.2016.08.032
Yu XD, Zheng RB, Xie JH, Su JY, Huang XQ, Wang YH et al (2015) Biological evaluation and molecular docking of baicalin and scutellarin as Helicobacter pylori urease inhibitors. J Ethnopharmacol 162:69–78. https://doi.org/10.1016/j.jep.2014.12.041
Zhang Y, Wang X, Wang L, Yu M, Han X (2014) Interactions of the baicalin and baicalein with bilayer lipid membranes investigated by cyclic voltammetry and UV–Vis spectroscopy. Bioelectrochemistry 95:29–33. https://doi.org/10.1016/j.bioelechem.2013.10.008
Zhang L, Liu X, Yang H, Zhao R, Liu C, Zhang R, Zhang Q (2019) Comparative pharmacokinetic study on phenolic acids and flavonoids in spinal cord injury rats plasma by UPLC-MS/MS after single and combined oral administration of danshen and huangqin extract. J Pharmaceut Biomed 172:103–112. https://doi.org/10.1016/j.jpba.2019.04.024
Zhou Y, Yang ZY, Tang RC (2016) Bioactive and UV protective silk materials containing baicalin—the multifunctional plant extract from Scutellaria baicalensis Georgi. Mater Sci Eng, C 67:336–344. https://doi.org/10.1016/j.msec.2016.05.063
Zhou Y, Yang ZY, Tang RC (2018) Green and facile fabrication of AgNPs@silk for colorful and multifunctional textiles using baicalin as a natural reductant. J Clean Prod 170(1):940–949. https://doi.org/10.1016/j.jclepro.2017.09.213
Acknowledgments
The work were kindly supported by No. 215 Research Institute of China Aerospace Science and Industry Nanjing Chenguang Groups, State Key Laboratory of Bio-Fibers and Eco-textiles of Qingdao University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, H., Li, Z., Liu, Y. et al. Advantages of Scutellaria baicalensis extracts over just baicalin in the ultrasonically assisted multi-functional treatment of linen fabrics. Cellulose 27, 4831–4846 (2020). https://doi.org/10.1007/s10570-020-03109-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-020-03109-6