Abstract
Acute myeloid leukaemia (AML), chronic lymphocytic leukaemia (CLL), and multiple myeloma (MM) are age-related haematological malignancies with defined precursor states termed myelodysplastic syndrome (MDS), monoclonal B-cell lymphocytosis (MBL), and monoclonal gammopathy of undetermined significance (MGUS), respectively. While the progression from asymptomatic precursor states to malignancy is widely considered to be mediated by the accumulation of genetic mutations in neoplastic haematopoietic cell clones, recent studies suggest that intrinsic genetic changes, alone, may be insufficient to drive the progression to overt malignancy. Notably, studies suggest that extrinsic, microenvironmental changes in the bone marrow (BM) may also promote the transition from these precursor states to active disease. There is now enhanced focus on extrinsic, age-related changes in the BM microenvironment that accompany the development of AML, CLL, and MM. One of the most prominent changes associated with ageing is the accumulation of senescent mesenchymal stromal cells within tissues and organs. In comparison with proliferating cells, senescent cells display an altered profile of secreted factors (secretome), termed the senescence-associated-secretory phenotype (SASP), comprising proteases, inflammatory cytokines, and growth factors that may render the local microenvironment favourable for cancer growth. It is well established that BM mesenchymal stromal cells (BM-MSCs) are key regulators of haematopoietic stem cell maintenance and fate determination. Moreover, there is emerging evidence that BM-MSC senescence may contribute to age-related haematopoietic decline and cancer development. This review explores the association between BM-MSC senescence and the development of haematological malignancies, and the functional role of senescent BM-MSCs in the development of these cancers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
All cellular components of the blood arise from haematopoietic stem cells (HSCs) via a process called haematopoiesis. HSCs, present within the bone marrow (BM), possess the capacity for self-renewal and multilineage differentiation ensuring a lifetime supply of HSCs and various mature blood cell types found within the peripheral blood and tissues. Healthy haematopoiesis is polyclonal whereby it is maintained by a large pool of individual HSC clones with equal proliferative capacity in the absence of clonal dominance. However, chronological ageing can result in cellular acquisition of genetic abnormalities over time [1], including specific somatic mutations within individual HSC clones. This can provide a certain clone with a proliferative advantage over others, thereby driving disproportional clonal expansion, leading to clonal dominance and clonal haematopoiesis of indeterminate potential (CHIP) [2] also known as age-related clonal haematopoiesis (ARCH) [3]. Next-generation sequencing studies have shown that somatic mutations in HSC clones, which predispose individuals to CHIP, are practically universal in healthy 50–70-year-old individuals. These mutated clones generally remain stable for many years, without the development of clinical consequences [3, 4]. Nevertheless, 0.5–1% of individuals with CHIP are at risk of malignant transformation to haematological cancer per year [3]. Most frequently, the accumulation of mutations in HSCs or downstream myeloid progeny leads to the development of myelodysplastic syndromes (MDS), a precursor condition for acute myeloid leukaemia (AML) [5]. The accumulation of genetic abnormalities within other committed haematopoietic cells also leads to clonal expansion of mature haematopoietic cell subsets. This promotes the development of dyscrasias that are recognised as precursor conditions of specific age-related haematological cancers. For example, clonal expansion of B cells can lead to high-count monoclonal B lymphocytosis (MBL; precursor for chronic lymphocytic leukaemia (CLL)) and clonal expansion of post-germinal centre plasma cells (PCs) can lead to monoclonal gammopathy of undetermined significance (MGUS; precursor to multiple myeloma (MM)). Each of these premalignant conditions possess a low, but significant, risk of progression to malignancy, with high-count MBL progressing to CLL at the rate of 1–2% per year [6] and MGUS progressing to MM at the rate of 1% per year [7].
Although it is currently unclear what drives the emergence of CHIP and progression of MDS, MBL, and MGUS to overt cancers, it is evident that ageing is a critical factor for the development of these conditions. While these premalignant conditions are characterised by different initiating mutations and are genetically diverse, the age-related progression to malignancy and dependence on the BM microenvironment for progression to overt cancer is a unifying feature. For example, the overall incidence of CHIP increases with age from 0.9% in individuals aged < 50 years to 10.4% in individuals > 65 years, which further increases to 16% in individuals aged > 75 years [8]. Specifically, CHIP with mutations in leukaemia driver genes is rarely observed in individuals under the age of 50 (0.7%) but is observed in 5.7% of individuals over the age of 65 years [8]. Likewise, 86% of MDS patients are diagnosed after the age of 60 (median age at diagnosis: 76 years), with only 6% of cases diagnosed in individuals under 50 years of age [9]. Furthermore, the prevalence of MBL increases from 20% in individuals older than 60 years to 75% in individuals over 90 years of age [10]. Similarly, 3–5% of the population over the age of 50 years have MGUS, which increases to 7.5% in individuals over the age of 85 years [11]. Notably, the risk of progression from MGUS to MM significantly increases with age, with a yearly risk of progression of 0.3% per year in MGUS patients less than 60 years of age and 1.1% per year in patients over 60 years [7]. Indeed, AML, CLL, and MM are prevalent with age, with a median diagnosis age of 69, 72, and 69 years of age, respectively [12,13,14].
Computational modelling of the relationship between cell intrinsic and extrinsic processes that govern somatic evolution and fitness dynamics within the HSC pool has revealed that the elevated incidence of haematological cancers in later life may not be exclusively dependent on intrinsic mutations but rather on changes in the microenvironment [15]. In accordance with this, a landmark whole exome sequencing study of paired patient plasma cell samples from MGUS and MM showed that malignant PC clones are already present at the MGUS stage, highlighting the role of the extrinsic BM microenvironment in stochastic PC clone activation and progression to malignant states [16]. Furthermore, it has been shown that genetic abnormalities alone are insufficient in driving the proliferation of haematopoietic malignant clones in the absence of a cancer-fostering microenvironment. For example, PCs from patients with stable MGUS have been shown to possess a similar capacity to form overt BM tumours as PCs from MM patients, following transplantation into humanised mice, highlighting the importance of a conducive microenvironment [17]. In addition, while primary human MDS cells engraft poorly in immunodeficient NOD scid gamma mice, engraftment can be significantly improved by co-transplantation with MDS patient-derived mesenchymal stromal cells [18]. In line with this, BM replacement studies using a NHD13 murine model of MDS have shown that healthy wild-type BM can attenuate MDS transformation to leukaemia and improve overall haematopoietic function [19].
Together with the importance of the BM microenvironment in the development of haematological cancers, the association between benign dyscrasia progression to overt haematological cancer and advancing biological age suggests that age-related changes to the BM microenvironment play a critical role in this process. A hallmark of chronological ageing is the accumulation of senescent cells in various tissues and organs, including the BM [20,21,22]. Notably, it is well documented that senescent stromal cells enhance the neoplastic properties of partially transformed cells and promote the outgrowth of malignant cells in the context of solid cancers [23,24,25,26,27,28,29]. In this review, we discuss the evidence that accumulation of senescent BM mesenchymal stromal cells (BM-MSCs) may promote the progression from premalignant haematological dyscrasias to overt malignancies. For the purposes of this review, BM-MSCs collectively refer to mesenchymal stem cells that possess multilineage potential and characteristic cell surface markers [30] and mesenchymal stromal cell cultures which can include fibroblasts and committed mesenchymal stem cell progeny.
2 The role of the BM mesenchymal stromal microenvironment in normal haematopoiesis and malignancy
The BM microenvironment plays a central role in maintaining the dynamic balance between HSC self-renewal, differentiation, quiescence and proliferation [31]. HSC fate is largely dictated by specific anatomic locations in the BM, termed niches, within which they reside [32]. In addition to extracellular matrix, the niche consists of specialised supporting cells including heterogeneous mesenchymal stromal cells, endothelial cells and osteolineage cells [33]. In normal physiological conditions, the BM niche functions to regulate HSC self-renewal and quiescence, critical to preserving the supply of HSCs throughout life. The BM niche also enables HSCs to exit quiescence under haematopoietic stress, such as BM injury or inflammation, to undergo subsequent proliferation and differentiation in response to the specific stimulus [34]. During homeostasis, HSCs are largely quiescent [35] and localise within perivascular regions, although there is contention whether they specifically reside around the endosteal area around arterioles (endosteal niche) [36] or at sinusoids (vascular niche) [37].
BM-MSCs control HSC fate by providing structural scaffolding as well as secreting soluble factors that regulate HSC proliferation and differentiation. Several BM-MSC populations including nerve/glial antigen 2–expressing (NG2+), leptin receptor-expressing cells (LEPR+) or C-X-C motif chemokine 12 (CXCL12)-abundant reticular (CAR) cells have been shown to play a role in promoting HSC maintenance. Specifically, secretion of CXCL12 or stem cell factor (SCF; also known as KITL) by BM-MSCs has been shown to regulate HSC niche retention, self-renewal, and trafficking [38]. The importance of mesenchymal lineage cells on HSC niche maintenance is exemplified by ablation studies. For example, the ablation of BM stromal cell populations such as CAR cells [39], Nestin+ MSCs [40], LEPR+ cells [41], and NG2+ cells [36] has been shown to induce HSC cycling and their depletion in the BM.
Similar to HSCs, tumour cell interactions with specific BM niche components have been shown to exert an impact on their proliferation. For example, following intravenous injection of the murine MM cell line 5TGM1 in C57BL/KaLwRijHsd mice, dormant tumour cells are found in the BM localised to endosteal bone surfaces [42]. Activation of osteoclastic bone resorption by receptor activator of nuclear factor κB ligand (RANKL) administration decreases dormant MM PC numbers, suggesting that remodelling of BM trabecular architecture may release these cells from dormancy [42]. Similarly, leukaemic stem cells in a CML mouse model have been shown to co-localise to CXCL12-secreting MSCs in the BM, which induces quiescence in the leukaemic cells [43]. Cre-mediated deletion of the CXCL12 gene in mesenchymal progenitors promoted the outgrowth of quiescent CML leukaemic stem cells (LSCs) by downregulating quiescence pathways in LSCs and promoting LSC cycling, thereby releasing them from dormancy in mice [44]. Therefore, modifications to the BM microenvironment can cause a switch between tumour cell dormancy and proliferation.
Notably, perturbed interactions between HSCs and the BM niche alters normal haematopoiesis and can lead to haematological malignancies. Indeed, studies have shown that haematopoietic malignancies can develop by modulation of supporting cells in the BM microenvironment. For example, constitutive activation of β-catenin in osteoblastic cells can induce MDS and AML [45]. Similarly, the conditional ablation of osteoblasts can accelerate the development of chronic and acute leukaemias in mice [46, 47]. Moreover, conditional deletion of Dicer1 miRNA endonuclease in BM-MSCs is sufficient to distort normal haematopoiesis and result in myelodysplasia which, in some cases, progressed to AML in a murine model [48].
Collectively, these studies demonstrate that malignant cells closely interact with the BM microenvironment to facilitate their proliferation and survival. Given the risk of transition to AML, CLL, and MM increases with age, these studies highlight a potential role for age-related changes in the BM microenvironment in creating a permissive milieu that promotes the early outgrowth of malignant cell clones and the subsequent development of malignancy.
3 Cellular senescence
A hallmark of ageing is the induction of cellular senescence leading to the accumulation of senescent cells within normal tissues. Notably, the accumulation of senescent cells has been linked to the deterioration of tissues including adipose tissue, skeletal muscle, eye tissue and cartilage [20, 21]. Cellular senescence is also associated with various age-related pathologies including glaucoma, cataracts, osteoarthritis, diabetes, and atherosclerosis [49].
Cellular senescence is characterised by irreversible cell cycle arrest, whereby cells cease to divide and differentiate, but remain viable and metabolically active. A feature of cellular ageing is heightened activity of the senescence-associated enzyme β-galactosidase which can be used to identify senescent cells [50]. Another characteristic of senescent cells is an enlarged and flattened cellular morphology that is thought to arise due to endoplasmic reticulum stress and the reorganisation of cytoskeletal scaffolding proteins [51]. Cellular senescence can be induced by various cell-intrinsic effectors including telomere shortening and changes in telomere structure, epigenetic changes, chromatin disorganisation, perturbed proteostasis, DNA damage, and mitochondrial dysfunction [52, 53]. In addition, cellular senescence can also be induced by cell-extrinsic effectors such as oxidative and genotoxic stress, mitogenic signals, inflammation, tissue damage signals, irradiation, chemotherapeutic agents, and nutrient deprivation [52, 53]. Cellular senescence is induced by activation of cyclin-dependent kinase inhibitors, including p21Cip1, p16INK4a, p14ARF, p15INK4b, and p27Kip1, which inhibit cell cycle progression through p53 and RB-dependent and -independent pathways (reviewed in Kumari et al., 2021) [52]. While DNA damage-induced senescence is largely mediated by p21Cip1, age-related or replicative senescence predominantly involves p16INK4a and p14ARF [52, 54, 55].
Cellular senescence can either be acute or chronic in nature. Acute senescence is triggered by a sudden increase in a specific stressor, often during tissue repair, and development and generates a sustained DNA damage response, which activates cell cycle checkpoints that enforce cell cycle arrest. Importantly, cells undergoing acute senescence are efficiently recognised by immune cells, leading to rapid elimination and replacement of senescent cells by healthy cells [56]. In contrast, chronic senescence occurs during ageing due to mild genotoxic stress, such as telomere shortening or DNA damage that accumulates over time, triggering a stochastic switch to senescence [57]. Ultimately, these cells undergo cell cycle arrest and transition into a senescent state, although they remain viable, displaying altered metabolic activity and apoptosis resistance [58]. In addition to greater induction of cellular senescence with age, there is also a defect in clearing these cells due to the concomitant age-related deterioration of the immune system [59]. Moreover, senescent cells have also been reported to recruit immune cells and induce dysfunction, which may contribute to their retention within tissues [60]. Consequently, chronically senescent cells persist in tissues and organs.
The senescence-associated secretory phenotype (SASP) is a classical feature of senescent cells and comprised various cytokines, growth factors, and pro-inflammatory mediators. These include interleukin-1 (IL-1), interleukin-6 (IL-6), C-X-C motif chemokine ligand 1 (CXCL1), C-X-C motif chemokine ligand 8 (CXCL8), C-C motif chemokine ligand 2 (CCL2), C-C motif chemokine ligand 5 (CCL5), C-C motif chemokine ligand 8 (CCL8), granulocyte macrophage colony-stimulating factor (GM-CSF), matrix metalloproteinases (MMP)-1,-3, and insulin growth factor (IGF) family members [61, 62]. Collectively, these factors recruit a plethora of immune cells including macrophages, natural killer (NK) cells, and T lymphocytes, thereby creating a pro-inflammatory tissue state [63]. Consequently, the accumulation of senescent cells is speculated to contribute to chronic inflammation that is observed within tissues with advancing biological age. Chronic inflammation, often accompanied by fibrosis and cell death, has been linked to the development and progression of various age-related conditions such as osteoarthritis, atherosclerosis, Alzheimer’s disease, and cancer [64,65,66]. Moreover, there is also strong evidence to suggest that SASP factors can augment normal haematopoiesis and create a favourable localised microenvironment for cancer, which are discussed in the following sections of this review.
4 BM-MSC senescence and its impact on haematopoiesis
Normal haematopoiesis is disrupted by ageing and is associated with higher frequency of mutations within HSCs (CHIP) [67], myeloid skewing [68], reduced lymphopoiesis [69], reduction in erythropoiesis [70], and decreased BM cellularity [71]. These effects are largely thought to be mediated by increased inflammatory signalling observed with ageing in the BM [72]. Studies suggest that a senescent BM microenvironment plays a critical role in promoting BM inflammation and the dysregulation of haematopoiesis with ageing. For example, it has been shown that myeloid skewing can be alleviated in a murine model when HSCs from aged mice are introduced into a young BM microenvironment. Specifically, transplantation of BM from 17- to 18-month-old mice into 2-month-old recipients revealed that that when ‘aged’ cells were transplanted into ‘young’ animals, fewer myeloid cells were generated compared with ‘aged-to-aged’ transplants [73]. Moreover, induction of BM-MSC senescence by knockdown of the endoribonuclease Dicer1 has been shown to significantly inhibit the ability of BM-MSCs to support HSCs in vitro [74, 75]. Knockout of Dicer1 in BM-MSCs also alters haematopoiesis in vivo by promoting increased apoptosis and proliferation of primitive HSCs, which is accompanied by a reduction in B cells and B cell progenitors and an increased frequency of myeloid cells, resulting in myelodysplasia [48]. In contrast, reversal of BM-MSC senescence by Dicer1 over-expression enhances their stem cell-supporting properties [74].
SASP factors secreted by senescent BM-MSCs are thought to play a central role in ageing-associated haematopoietic decline. For example, the inflammatory cytokine CCL5, characteristic of the SASP profile, is elevated in BM-MSCs of aged mice compared with young controls [73] and has been shown to favour the induction of promyeloid transcription factors in HSCs [73], which may be linked to myeloid skewing of HSCs observed with age [76]. Furthermore, it has been shown that the SASP factor interleukin-6 (IL-6), secreted by aged BM-MSCs, can alter HSC homeostasis in vitro [77]. Specifically, IL-6 secreted by aged BM-MSCs acts as a HSC growth factor and induces rapid HSC expansion, thereby driving clonal outgrowth [77], leading to depletion of the HSC pool and increased risk of genomic aberrations in HSCs [78].
5 MSC senescence as a cancer driver
Senescent MSCs have been shown to facilitate tumour growth in a variety of cancer contexts. For example, co-culture of the breast cancer cell line MDA-MB-231 with senescent fibroblasts increases tumour growth in vitro, when compared with co-culture with non-senescent fibroblasts [79]. In line with this, subcutaneous co-injection of MDA-MB-231 breast cancer cells with senescent fibroblasts leads to earlier onset and more rapid tumour growth when compared with co-injection with non-senescent control fibroblasts in mice [24, 26]. Similarly, subcutaneous growth of the prostate cancer cell line PC3 is increased by co-injection with senescent fibroblasts, compared with non-senescent fibroblasts [27, 28]. Importantly, studies using the fibroblast accelerate stromal supported-tumourigenesis (FASST) mouse model, whereby collagen I promoter-driven Cre recombinase upregulates the CDK-inhibitor p27Kip1 and induces senescence in mesenchymal lineage cells, have shown that senescent mesenchymal cells drive the development of cancer-supportive BM microenvironment. Here, BM tumour growth following intra-cardiac injection of NT2.5 breast cancer cells was significantly greater in FASST mice compared with littermate controls, suggesting that senescent mesenchymal lineage cells promote breast cancer cell homing and/or subsequent proliferation in the BM [29].
SASP factors are thought to play a central role in the cancer-promoting properties of senescent cells. Indeed, studies have shown that treatment of a variety of epithelial cancer cell types (including breast, colorectal and prostate cancer) with conditioned media collected from senescent fibroblasts or MSCs increases cancer cell proliferation compared with conditioned media collected from non-senescent cells in vitro [80,81,82]. In addition to promoting proliferation, SASP factors have also been shown to exert pro-migratory effects on cancer cells. For instance, it has been shown that conditioned medium from senescent fibroblasts significantly increases the migration of MCF7 breast cancer cells in vitro compared with conditioned medium from non-senescent fibroblasts [83]. Furthermore, treatment of non-aggressive human breast cancer cell lines T47D and ZR75.1 with conditioned media collected from senescent fibroblasts induces epithelial-mesenchymal transition and enhanced invasiveness of cancer cells, thereby promoting a malignant cell phenotype [84].
Moreover, senescent cells have also been implicated in driving neoplastic transformation of pre-neoplastic cells. For example, conditioned medium from senescent fibroblasts significantly enhances in vitro migratory and invasive properties of a partially transformed ovarian epithelial cell line compared with conditioned medium from non-senescent fibroblasts [23]. Similarly, senescent fibroblasts have been shown to promote the outgrowth of immortalised epithelial cell lines HaCAT, HMT-3522 S1, and SCp2 in a co-culture setting compared with non-senescent fibroblasts [24]. In addition, while HaCAT and SCp2 cell lines do not form tumours in immunocompromised mice when injected alone, co-injection of these cells with senescent ovarian fibroblasts leads to increased incidence and earlier onset of tumour development, which is not observed with co-injection of non-senescent fibroblasts [24]. In line with this, co-injection of weakly tumourigenic EpH4-v mouse mammary epithelial cells with irradiation-induced senescent murine mammary fibroblasts into mammary fat pads of female nu/nu mice significantly promotes tumour formation and vascularisation relative to co-injection with non-senescent fibroblasts or EpH4-v cells alone [25]. Notably, studies have also shown that senescent fibroblasts are unable to induce neoplastic properties in normal, untransformed epithelial cells [24].
Collectively, these studies support the notion that senescent MSCs provide a localised microenvironment that supports the outgrowth of malignant cells and pre-malignant cells harbouring cancer-initiating mutations. Moreover, there is evidence to suggest that the accumulation of senescent cells specifically in the BM may create a cancer-permissive niche that enables the outgrowth of malignant cell clones in the BM. As senescent cells accumulate with age, this may contribute to age-related progression from cancer precursor conditions to overt cancers, including in the context of haematological cancers.
6 BM-MSC senescence in haematological malignancies
In addition to playing a role in solid cancers and aberrant haematopoiesis, there is increasing evidence that MSC senescence contributes to the development and progression of haematological cancers. As the growth of transformed pre-neoplastic and neoplastic cells can be influenced by SASP factors, it is possible that age-related accumulation of senescent BM-MSCs and SASP leads to clonal outgrowth of transformed cells and malignancy (Fig. 1). In the following sections, we discuss evidence for the role of senescent BM-MSCs in blood disorder precursor conditions and haematological malignancies, as the prevalence of these dyscrasias dramatically increases with age. An overview of evidence that BM-MSC senescence is a common feature of age-related haematological cancers and disorders is provided in Table 1. Additionally, a summary of common SASP factors that may play a role in haematological dysfunction are listed in Table 2.
Schematic model depicting how senescent BM-MSCs may promote the progression of haematological malignancies. The acquisition of genetic abnormalities with ageing within individual haematopoietic stem cell clones and committed haematopoietic clones predisposes them to disproportional clonal expansion. These clones reside or home to the BM, where they remain stable for long periods of time. Concurrently, senescent BM-MSCs accumulate with age and constitute SASP factor-rich activating niches, resulting in the outgrowth of transformed malignant clones and progression to active malignancy
6.1 Myelodysplastic syndromes (MDS) and acute myeloid leukaemia (AML)
MDS are a group of asymptomatic disorders characterised by ineffective haematopoiesis with varying degrees of cytopenias and dysplasia. MDS is diagnosed based on the presence of one or more cytopenias (anaemia, neutropenia, thrombocytopenia), ≥ 10% morphologically dysplastic nucleated cells of one lineage and < 20% blasts in peripheral blood and BM [85]. MDS can transform into AML, with the diagnosis of AML established by ≥ 20% blood and BM involvement of leukaemic blasts, presence of cytopenias and in some cases the presence of genetic abnormalities including t(8;21), inv(16) or t(16;16) [85].
Numerous studies have shown that BM-MSCs from MDS and AML patients display senescent characteristics, including flattened polygonal morphology, a reduced expression of Dicer1 and proliferative and osteogenic potential, while displaying increased β-galactosidase activity and p16INK4a and p21Cip1 expression [74, 86,87,88,89,90,91,92,93,94,95]. BM-MSCs from MDS patients have been shown to express increased levels of SASP factors, including IL-6 and tumour necrosis factor α (TNF-α), compared with those of healthy controls [96, 97]. Further, BM-MSCs from AML patients express increased levels of SASP factors IL-6 [98], interleukin-7 (IL-7) [99], and CXCL8 [100] compared with healthy controls.
Notably, increased expression of SASP factors in the BM and blood plasma of MDS patients is predictive of overall survival and progression to AML, supporting a potential role for senescence and SASP factors in MDS-to-AML transition. For example, several studies have demonstrated that SASP components such as IL-6, IL-7, CXCL8, and TNF-α are significantly elevated in MDS patient BM and peripheral blood plasma compared with healthy controls [101, 102]. In addition, plasma IL-6 levels in MDS patients are an independent predictor of overall survival and risk of progression to AML [101]. In fact, BM-MSC gene expression of IL-6 gradually increases with progression from MDS to AML [98]. Moreover, blood plasma IL-7 levels have been identified as independent prognostic factors for overall survival in MDS patients [101]. Furthermore, decreased proliferative rate of BM-MSCs isolated from AML patients was found to be independently associated with poor patient outcomes such as treatment failure and early relapse [103].
Beyond the association between BM-MSC senescence and MDS to AML transition, there is also evidence that BM-MSC senescence may play an active role in driving progression from MDS to AML. Indeed, senescent BM-MSCs from MDS patients exhibit an enhanced ability to increase survival of leukaemic cells and decreased capacity to support normal haematopoiesis [74, 86, 92, 96]. Moreover, transwell co-culture of mouse myeloid leukaemic cells with senescent BM-MSCs significantly increases leukaemic cell proliferation in comparison to co-culture with non-senescent BM-MSCs [104], suggesting that senescent BM-MSCs promote leukaemic cell proliferation via secreted factors. Importantly, the expression of SASP factors IL-6 and CXCL8 by BM-MSCs has been implicated in promoting leukaemic cell growth. For example, BM-MSCs from MDS patients exhibit an increased expression of IL-6 compared with healthy BM-MSC controls, with IL-6 previously reported to promote the growth of AML cell line HL-60 [105]. In line with this, the administration of an anti-IL-6 receptor blocking antibody was shown to inhibit the proliferation of AML cell lines as well as the clonogenic growth of primary AML cells in vitro [106]. Knockdown of CXCL8 in BM-MSCs has also been shown to significantly inhibit the ability of BM-MSCs to promote the survival of primary AML cells in a co-culture setting [107]. The role of CXCL8 in BM-MSC-mediated promotion of AML cell proliferation has also been explored by blocking the CXCL8 receptor (CXCR2) on CXCL8-knockdown HL-60 and THP1 AML cell lines. In this study, treating co-cultures of AML patient-derived BM-MSCs and CXCL8-knockdown AML cells with CXCR2 inhibitor SB225002 decreased AML cell line proliferation and colony-forming ability, induced cell cycle arrest, and induced AML cell apoptosis in a dose-dependent manner, when compared with vehicle treated controls [100].
The functional role of senescent BM-MSCs in AML progression has been directly explored in vivo using a p16-3MR mouse model [104]. These mice have been engineered to express the p16INK4a promoter coupled with thymidine kinase, enabling the selective ablation of senescent BM-MSCs by administration of ganciclovir. Strikingly, ablation of p16INK4a-expressing senescent BM-MSCs following the engraftment with mouse myeloid leukaemia cells significantly decreased subsequent tumour growth and increased mouse survival, when compared with controls [104], demonstrating the role of senescent BM-MSCs in AML disease progression.
As BM-MSC senescence and secretion of pro-leukaemic SASP factors, including IL-6 and CXCL8, is evident at the MDS stage, and elevated levels of SASP factors are predictive of MDS-to-AML progression, it is possible that the age-related accumulation of senescent BM-MSCs at MDS provides a permissive, SASP-factor rich growth- activating niche for leukaemic cells which facilitates MDS-to-AML progression. Taken together, these studies support the hypothesis that accumulation of senescent BM-MSCs with age in MDS patients may increase the proliferation of the myeloid lineages, increasing the rate of progression to AML in these patients.
6.2 Monoclonal B cell lymphocytosis (MBL) and chronic lymphocytic leukaemia (CLL)
MBL is an asymptomatic haematological dyscrasia characterised by the presence of clonal B cell populations in the blood and no other sign of lymphoproliferative disorder. MBL can be further categorised into low and high count MBL, based on whether the B cell count in the peripheral blood is above or below 0.5 × 109/L [108]. High count MBL has an increased risk of progression to CLL, defined by peripheral blood B cell count of greater than > 5 × 109/L [108].
Studies characterising BM-MSCs isolated from CLL patients have revealed that CLL BM-MSCs, similar to those of patients with MDS and AML, display senescent characteristics such as decreased CFU-F frequency [109], decreased growth rate and growth arrest [109], an enlarged polygonal morphology [110], increased β-galactosidase staining [110], and increased p16INK4a mRNA expression [110] compared with BM-MSCs from healthy, age-matched individuals. Additionally, BM-MSCs from CLL patients display elevated gene and protein expression of several SASP factors, including IL-6, CXCL8, and vascular endothelial growth factor (VEGF), compared with BM-MSCs from healthy individuals [110, 111]. Peripheral blood levels of SASP factors including IL-6 [112], VEGF [113], and CXCL8 [114] are also increased in patients with CLL compared with healthy controls. Importantly, CLL patients with elevated serum levels of IL-6, TNF-α, and CXCL8 exhibit poorer overall survival compared with patients with low serum levels of these SASP factors [112, 114,115,116,117]. Additionally, increased serum levels of VEGF have been shown to be indicative of shorter progression-free survival in early B cell CLL patients [113]. While it is unclear whether increased levels of these cytokines is due to age-related accumulation of senescent BM-MSCs, it is notable that peripheral blood levels of SASP factors IL-6, CXCL8, and TNF-α positively correlate with patient age in CLL patients [115, 116].
From a functional perspective, studies have shown that BM-MSCs isolated from CLL patients exhibit an increased capacity to support the proliferation of normal peripheral blood B cells, when compared with BM-MSCs from healthy donors [109]. In addition, BM-MSCs support in vivo engraftment and ex vivo expansion of primary CLL cells [111, 118,119,120,121]. As BM-MSCs isolated from CLL patients exhibit a senescent phenotype, these pro-proliferative effects may, in part, be due to secretion of pro-proliferative SASP factors. Indeed, blocking BM-MSC protein secretion using the exocytosis inhibitor brefeldin A greatly reduces the pro-survival effects BM-MSCs exert in direct co-culture with CLL cells [122]. Specifically, BM-MSC-derived IL-6 has been suggested to increase primary CLL cell survival in vitro, with the addition of an IL-6 receptor antagonist to co-cultures of BM-MSCs and primary CLL cells significantly attenuating CLL cell survival [118]. Moreover, BM-MSCs have been shown to support the engraftment of CLL cells in vivo via an IL-6-mediated mechanism. Here, NOD scid gamma mice reconstituted with peripheral blood mononuclear cells (PBMCs) from CLL patients in combination with BM-MSCs or supernatants of BM-MSCs displayed superior CLL engraftment compared with PBMC injection alone. Furthermore, the administration of tocilizumab, an IL-6 receptor antagonist, abrogated the supportive effect of BM-MSCs and BM-MSC supernatant on CLL engraftment [118]. BM-MSC-derived VEGF has also been shown to act as a pro-survival factor of primary CLL cells and CLL cell lines in vitro [122, 123]. Indeed, the addition of VEGF neutralising antibody, mAb293, abolished the BM-MSC co-culture-mediated survival of CLL cells, while having no effect on CLL survival in monoculture [122]. Similarly, VEGF inhibitors vatalanib and pazopanib significantly reduce tumour growth of JVM-3 CLL-like cells in vivo [124], although it cannot be ruled out that an anti-angiogenic mechanism may also contribute to the anti-tumour response. Exogenous CXCL8 has also been shown to prolong ex vivo survival of leukaemic cells by upregulating the expression of anti-apoptotic proteins in B-CLL cells [125]. As IL-6, VEGF, and CXCL8 are expressed as part of the SASP milieu, it is plausible that the age-related accumulation of senescent BM-MSCs may provide a CLL growth permissive microenvironment via paracrine proliferative and survival factors.
While it remains to be determined whether senescent BM-MSCs actively promote clonal B cell outgrowth in MBL and CLL, serum levels of SASP factors IL-6, CXCL8, and TNF-α are significantly increased in the peripheral blood of MBL patients compared with healthy individuals [126, 127]. Further studies are required to determine whether BM-MSC senescence is evident at the MBL stage and whether BM-MSC senescence is associated with, and plays an active role in, MBL-to-CLL progression.
6.3 Monoclonal gammopathy of undetermined significance (MGUS) and multiple myeloma (MM)
MGUS is an asymptomatic PC dyscrasia, characterised by the clonal expansion of post-germinal centre PCs in the BM. MGUS is defined by the presence of < 30 g/L serum monoclonal protein (M protein), < 10% monoclonal PCs in the BM and absence of end organ damage (CRAB features: hypercalcaemia, renal failure, anaemia, and bone lesions) [128]. MGUS is the precursor condition to MM. MM is characterised by ≥ 30 g/L M protein and/or ≥ 10% monoclonal PCs in the BM, usually accompanied by CRAB features.
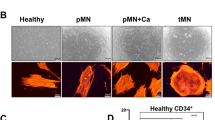
There is mounting evidence that BM-MSCs from MM patients have a senescent phenotype in culture and that BM-MSC senescence is associated with poorer MM patient outcomes. Compared with ex vivo-cultured BM-MSCs from healthy controls, BM-MSCs from MM patients are larger, more irregular in shape, display reduced proliferative potential, reduced osteogenic differentiation potential, increased p16INK4a and p21Cip1 expression and β-galactosidase activity, and decreased expression of Dicer1 and cell-cycle related genes [75, 93, 129,130,131,132,133,134,135,136]. MM BM-MSCs also display an altered secretory profile compared with that of healthy controls, closely resembling that of senescent BM-MSCs [129, 137, 138]. Specifically, BM-MSCs from MM patients exhibit significantly higher gene expression of SASP factors including IL-6, TNF-α, growth differentiation factor 15 (GDF15) and CXCL8, relative to BM-MSCs from age-matched healthy controls [75, 129, 132, 136, 138]. Furthermore, BM-MSCs from MM patients secrete significantly higher IL-6 and GDF15 protein compared with age-matched controls [130, 132, 139, 140].
Notably, serum levels of canonical SASP factors in the peripheral blood of MM patients have been shown to correlate with MM disease severity and progression. For example, serum levels of IL-6 [141,142,143,144,145], TNF-α [146], and GDF15 [145, 147,148,149] are elevated in MM patients compared with healthy individuals. Moreover, serum levels of IL-6 [141,142,143,144], GDF15 [145, 147, 148], and TNF-α [146, 150] positively correlate with more advanced stage of the disease according to both the Durie-Salmon and ISS classification systems. In addition, serum levels of SASP factors have been shown to negatively correlate with MM patient survival. For example, elevated serum levels of IL-6 and GDF15 are associated with significantly shorter overall survival times in newly diagnosed MM patients [140, 142, 151]. Although it is unclear whether increases in the levels of these cytokines are due to the accumulation of senescent BM-MSCs, there is evidence to suggest an association between BM-MSC senescence and MM disease outcomes. Notably, increased numbers of β-galactosidase-positive senescent cells in ex vivo-cultured BM-MSCs from patients are associated with a shorter progression free survival, compared with patients with low numbers of senescent BM-MSCs [75]. Taken together, these findings support the association between increased BM-MSC senescence, increased SASP factors production and disease progression in MM.
Functionally, MM MSCs have been shown to significantly promote the adherence and proliferation of human MM cell lines compared with BM-MSCs from healthy individuals [132, 152,153,154]. While it is unclear if this is attributable to the senescent properties of MM BM-MSCs, the genetic induction of BM-MSC senescence has been shown to promote MM tumour cell growth in a co-culture setting. To this end, direct co-culture of the NCI-H929 MM cell line with senescent BM-MSCs (induced by Dicer1 knockdown) significantly enhanced MM cell growth relative to co-culture with non-senescent BM-MSCs in vitro [75]. Moreover, an association between the induction of BM-MSC senescence by over-expressing the lysophosphatidic acid receptor 1 (LPAR1) and enhanced MM tumour growth has been observed in vivo. Specifically, co-transplantation of IM-9 MM cells with LPAR1-over-expressing senescent BM-MSCs significantly promoted tumour growth in BALB/c-nu/nu mice in comparison to co-transplantation with control BM-MSCs [155]. In line with this, several canonical SASP factors have been shown to promote the proliferation of MM PCs. For example, BM-MSC-derived IL-6 is a potent inducer of MM PC growth and survival [132, 156,157,158,159]. The survival and proliferation of patient-derived MM PCs in short-term cultures is increased with recombinant IL-6 [158, 159] and is abrogated with the addition of anti-IL-6 monoclonal antibodies [158]. Similarly, the growth promoting effects of BM-MSCs on MM cell lines can be abrogated with the addition of an anti-IL-6 monoclonal antibody [132, 160]. Likewise, IL-6 siRNA-mediated knockdown in healthy BM-MSCs significantly reduces the proliferation of the U266 MM cell line both in vitro and in an in vivo subcutaneous xenograft model compared with normal BM-MSCs [161], suggesting that BM-MSC-derived IL-6 mediates a favourable microenvironment for MM cell growth. Notably, there is also a significant positive correlation between MM patient serum IL-6 levels and the proliferation of primary MM BM PCs, as assessed by PC labelling index or Ki67 staining of tumour cells in patient biopsies [141, 144]. Interestingly, anti-IL-6 monoclonal antibodies have been assessed in phase II clinical trials in patients with relapsed or refractory MM [162,163,164] and high-risk smouldering MM (patients at high risk of progressing to overt MM within 2 years) [165]. While anti-IL-6 alone was insufficient to induce anti-tumour responses in heavily pre-treated MM patients [164], there is some evidence that anti-IL-6 therapy may delay disease progression in SMM patients [165].
Several other canonical SASP factors that are secreted by MM BM-MSCs have also been suggested to increase MM PC proliferation in vitro. For example, GDF15 is a MM BM-MSC-derived SASP factor shown to increase MM cell growth and survival in vitro [147]. Supplementation with GDF15 promotes the survival and clonogenic growth of human MM cell lines and primary human MM cells in vitro [140]. Furthermore, pre-treatment of NCI-H929 cells with GDF15 increases their ability to develop MM tumours in vivo [140]. Moreover, survival of mice injected with Vk*Myc MM cells was significantly longer in Gdf15-knockout mice, compared with wild-type controls, strongly supporting the role of microenvironmental GDF15 in myeloma disease progression in vivo [140]. TNF-α is another SASP factor produced by MM BM-MSCs that has been shown to promote survival and proliferation of MM cell lines in vitro. Exogenous supplementation of TNF-α has been shown to reduce apoptosis and increase the percentage of MM cells in S and G2/M cell-cycle phases: an effect that can be reversed by supplementation with an anti-TNF-α antibody [166].
Although the precise role that accumulating senescent BM-MSCs play in MGUS-to-MM progression is currently unknown, emerging evidence suggests an association between BM-MSC senescence and MM disease progression. Analysis of serum cytokine profiles of MGUS patients has revealed that MGUS is associated with a shift towards a pro-inflammatory phenotype compared with healthy individuals [167]. In particular, it has been shown that serum protein levels of canonical SASP factor IL-6 is elevated in MGUS and SMM patients compared with healthy individuals [141, 168, 169]. Furthermore, IL-6 has been shown to be upregulated in BM-MSC culture supernatants from MGUS patients, while it was undetectable cultures from normal donors [170]. However, whether increased IL-6 is a consequence of BM-MSC senescence is currently unknown.
Collectively, as the risk of MGUS-to-MM progression increases age, it is plausible that the accumulation of senescent BM-MSCs with advancing age during MGUS can contribute to creating a niche conducive to the outgrowth of MM PCs, leading to transition to overt MM. Further studies are required to characterise BM-MSCs from MGUS patients in the context of senescence, and to establish whether these cells play an active role in the transition from MGUS to MM.
7 Therapeutic targeting of senescent cells
Recent studies have revealed that senescent cells may represent a novel therapeutic target. For instance, studies in mice expressing the INK-ATTAC transgene enabling the elimination of p16INK4a-expressing senescent cells have shown that global clearance of senescent cells from aged mice is able to delay tumourigenesis and prevent age-related deterioration of organs such as the kidney, heart, and adipose tissues [20]. These findings highlight the possibility that therapeutic agents targeting senescent cells could have anti-tumour effects. In line with this, there is emerging data to suggest that the use of small molecule senolytic agents (drugs intended to induce apoptosis in senescent cells) may represent a novel therapeutic strategy to alleviate various age-related pathologies. Indeed, pharmacological approaches to eliminate senescent cells are increasingly being investigated. For instance, treatment of 24-month-old C57BL/6 mice with the BCL-2 pro-survival protein family inhibitor navitoclax has been shown to reduce the number of senescent cells in the cerebral circulation and improve regional cerebral blood flow, thereby improving memory and age-related cognitive decline in mice [171]. Similarly, treatment of irradiated C57BL/6 mice with navitoclax significantly attenuated the number of senescent alveolar cells in the lungs and reversed persistent pulmonary fibrosis induced by ionising radiation [172]. Administration of navitoclax to naturally aged or total body irradiation-induced prematurely aged C57BL/6J mice has also been shown to effectively clear senescent muscle stem cells and HSCs in vivo. Notably, this selective clearance of senescent cells significantly improved overall HSC and muscle stem cell clonogenicity and long-term BM engraftment ability of HSCs, demonstrating the utility of navitoclax to rejuvenate senescent cell populations [173]. However, while navitoclax has been shown to effectively clear senescent cells in murine [174, 175] and human [176] BM-MSC cultures in vitro, its utility to eliminate senescent BM-MSCs in vivo is yet to be investigated. Importantly, the utility of senolytic agents to eliminate senescent MSCs in the BM has been shown using the medicinal compound tetramethylpyrazine (TMP). In addition to reducing BM-MSC senescence in vitro by suppression of NF-κB signalling [177], local injection of TMP into the BM cavity of 20-month-old C57BL/6 mice has been shown to significantly decrease p16INK4a-positive BM-MSC numbers and decrease protein and mRNA levels of SASP factors including IL-6, TNF-α and IL-1β in the BM of treated mice [178]. TMP administration also improved maintenance of the HSC niche and increased HSC numbers by promoting BM-MSC expression of HSC maintenance and chemo-attraction-related genes such as Cxcl12, Kitlg, Angpt1, Il7, Vcam1 [178].
Additionally, the senolytic agents dasatinib and quercetin have been shown to decrease human senescent cell numbers in vitro [179]. While dasatinib is more effective at clearing senescent adipocytes, quercetin better eliminates senescent BM-MSCs and endothelial cells, with the combination of these agents targeting a broader range of senescent cell types than either agent alone [179]. The effectiveness of dasatinib and quercetin combination therapy at targeting senescent cells has also been explored in mice. Treatment of 20-month-old C57BL/6 mice with both dasatinib and quercetin was reported to significantly lower p16INK4a mRNA expression levels in a population of osteocyte-enriched cells derived from the bones of treated mice and to reduce the percentage of senescent osteocytes, compared with vehicle-treated controls [180]. This attenuated age-related bone loss, as evidenced by improved trabecular bone microarchitecture in the spine and femur compared with vehicle-treated control animals [180]. On this basis, the use of dasatinib and quercetin may be applicable to targeting senescent cells in the BM of humans. Indeed, a Phase 1 pilot study in patients with diabetic kidney disease showed oral administration of dasatinib and quercetin for three days reduced senescent cell burden. Specifically, preliminary analyses demonstrated a significant decrease in the number of p16INK4a and p21Cip1 expressing cells and β-galactosidase positive cells in adipose tissue biopsies and p16INK4a and p21Cip1 expressing epidermal cells in skin biopsies. Notably, the levels of circulating SASP factors in the plasma, namely, IL-1α, IL-6, MMP-9, and MMP-12, were also significantly reduced in drug-treated patients compared with vehicle-treated patients [181]. Furthermore, dasatinib and quercetin have recently been used in the first human open-label pilot study of senolytic agents for the treatment of idiopathic pulmonary fibrosis, a senescence-driven condition [182]. Dasatinib and quercetin treatment significantly improved the physical function of study participants, including chair stand and walking tests, and was accompanied by a trend towards decreased serum SASP factor expression [182]. While preliminary, these studies suggest that the use of senolytic agents is a promising strategy for the treatment of age-related, senescence-driven pathological conditions. Nevertheless, the utility of senolytic agents as a potential treatment or intervention in the context of haematological cancers is yet to be investigated.
8 Concluding remarks
It is clear that the BM microenvironment plays a pivotal role in haematopoiesis and the development and progression of haematological cancers. Evidence suggests that the presence of increased numbers of senescent BM-MSCs is a characteristic feature of a variety of haematological cancers, and these cells secrete an altered cytokine profile that closely resembles the SASP. Indeed, SASP molecules such as IL-6, CXCL8, and GDF15 have been widely shown to be associated with disease progression in patients and to drive haematological cancer cell proliferation in vitro. Together with the age-related emergence of CHIP, MDS, MBL, and MGUS, concurrent age-related increases in BM-MSC senescence may contribute to the progression from these benign states to overt malignancies. In addition to age-related accumulation of senescent BM-MSCs, evidence suggests that BM-MSC senescence can also be induced by haematological cancer cells. For instance, healthy human BM-MSCs co-cultured with primary MM cells or MM cell lines show increased levels of senescence relative to BM-MSCs in monoculture in vitro [75]. Moreover, young adult mice engrafted with primary human AML blasts demonstrate increased levels of BM-MSC senescence compared with mice engrafted with normal human CD34+ HSCs [104]. As such, haematological cancer cells can exacerbate BM-MSC senescence, creating a feed forward loop that further promotes the development of haematological cancers. While there is compelling evidence that BM-MSC senescence promotes MDS-to-AML progression, the role of senescent BM-MSCs in the progression of MBL and MGUS to overt cancer requires further exploration. However, it is clear that SASP factors have pro-proliferative effects on CLL and MM tumour cells, suggesting that age-related increases in BM-MSC senescence may be key drivers of MBL-to-CLL and MGUS-to-MM progression. This is further supported by the observation that SASP factors are detectable at elevated levels in the blood of MBL and MGUS patients and that levels of these factors increase with advancing disease stages. Further studies are required to directly confirm the effects of BM-MSC senescence on haematological tumour outgrowth in vitro and in vivo, as, to date, this has only been explored in the context of AML. There is tremendous scope to investigate the utility of senolytic agents for the treatment of haematological malignancies. As the incidence of haematological malignancies and precursor conditions dramatically increases with age, a better understanding of the relationship between the ageing BM microenvironment and the outgrowth of malignant cells may revolutionise treatment strategies. This may not only alleviate the pro-tumourigenic effects exerted by the SASP, but also improve general haematopoietic function throughout the ageing process.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
References
Welch, J. S., Ley, T. J., Link, D. C., Miller, C. A., Larson, D. E., Koboldt, D. C., Wartman, L. D., Lamprecht, T. L., Liu, F., Xia, J., Kandoth, C., Fulton, R. S., McLellan, M. D., Dooling, D. J., Wallis, J. W., Chen, K., Harris, C. C., Schmidt, H. K., Kalicki-Veizer, J. M., Lu, C., et al. (2012). The origin and evolution of mutations in acute myeloid leukemia. Cell, 150(2), 264–278. https://doi.org/10.1016/j.cell.2012.06.023
Steensma, D. P., Bejar, R., Jaiswal, S., Lindsley, R. C., Sekeres, M. A., Hasserjian, R. P., & Ebert, B. L. (2015). Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. Blood, 126(1), 9–16. https://doi.org/10.1182/blood-2015-03-631747
Jaiswal, S., Fontanillas, P., Flannick, J., Manning, A., Grauman, P., Mar, B. G., Lindsley, R. C., Mermel, C., Burtt, N., Chavez, A., Higgins, J. M., Moltchanov, V., Kinnunen, L., Koistinen, H., Ladenvall, C., Getz, G., Correa, A., Gabriel, S., Kathiresan, S., Stringham, H., et al. (2014). Clonal hematopoiesis with somatic mutations is a common, age-related condition associated with adverse outcomes. Blood, 124(21), 840. https://doi.org/10.1056/NEJMoa1408617
Young, A. L., Challen, G. A., Birmann, B. M., & Druley, T. E. (2016). Clonal haematopoiesis harbouring AML-associated mutations is ubiquitous in healthy adults. Nature Communucations, 7, 12484. https://doi.org/10.1038/ncomms12484
Veiga, C. B., Lawrence, E. M., Murphy, A. J., Herold, M. J., & Dragoljevic, D. (2021). Myelodysplasia syndrome, clonal hematopoiesis and cardiovascular disease. Cancers, 13(8), 1968. https://doi.org/10.3390/cancers13081968
Strati, P., & Shanafelt, T. D. (2015). Monoclonal B-cell lymphocytosis and early-stage chronic lymphocytic leukemia: Diagnosis, natural history, and risk stratification. Blood, 126(4), 454–462. https://doi.org/10.1182/blood-2015-02-585059
Kyle, R. A., Larson, D. R., Therneau, T. M., Dispenzieri, A., Kumar, S., Cerhan, J. R., & Rajkumar, S. V. (2018). Long-term follow-up of monoclonal gammopathy of undetermined significance. New England Journal of Medicine, 378(3), 241–249. https://doi.org/10.1056/NEJMoa1709974
Genovese, G., Kahler, A. K., Handsaker, R. E., Lindberg, J., Rose, S. A., Bakhoum, S. F., Chambert, K., Mick, E., Neale, B. M., Fromer, M., Purcell, S. M., Svantesson, O., Landen, M., Hoglund, M., Lehmann, S., Gabriel, S. B., Moran, J. L., Lander, E. S., Sullivan, P. F., Sklar, P., et al. (2014). Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. New England Journal of Medicine, 371(26), 2477–2487. https://doi.org/10.1056/NEJMoa1409405
Ma, X., Does, M., Raza, A., & Mayne, S. T. (2007). Myelodysplastic syndromes: Incidence and survival in the United States. Cancer, 109(8), 1536–1542. https://doi.org/10.1002/cncr.22570
Shim, Y. K., Middleton, D. C., Caporaso, N. E., Rachel, J. M., Landgren, O., Abbasi, F., Raveche, E. S., Rawstron, A. C., Orfao, A., Marti, G. E., & Vogt, R. F. (2010). Prevalence of monoclonal B-cell lymphocytosis: a systematic review. Cytometry. Part B: Clinical Cytometry, 78. https://doi.org/10.1002/cyto.b.20538
Kyle, R. A., Therneau, T. M., Rajkumar, S. V., Larson, D. R., Plevak, M. F., Offord, J. R., Dispenzieri, A., Katzmann, J. A., & Melton, L. J., 3rd. (2006). Prevalence of monoclonal gammopathy of undetermined significance. New England Journal of Medicine, 354(13), 1362–1369. https://doi.org/10.1056/NEJMoa054494
Costa, L. J., Brill, I. K., Omel, J., Godby, K., Kumar, S. K., & Brown, E. E. (2017). Recent trends in multiple myeloma incidence and survival by age, race, and ethnicity in the United States. Blood Advances, 1(4), 282–287. https://doi.org/10.1182/bloodadvances.2016002493
Hallek, M., Shanafelt, T. D., & Eichhorst, B. (2018). Chronic lymphocytic leukaemia. Lancet, 391(10129), 1524–1537. https://doi.org/10.1016/S0140-6736(18)30422-7
Oran, B., & Weisdorf, D. J. (2012). Survival for older patients with acute myeloid leukemia: A population-based study. Haematologica, 97(12), 1916–1924. https://doi.org/10.3324/haematol.2012.066100
Rozhok, A. I., Salstrom, J. L., & DeGregori, J. (2014). Stochastic modeling indicates that aging and somatic evolution in the hematopoetic system are driven by non-cell-autonomous processes. Aging, 6(12), 1033–1048. https://doi.org/10.18632/aging.100707
Dutta, A. K., Fink, J. L., Grady, J. P., Morgan, G. J., Mullighan, C. G., To, L. B., Hewett, D. R., & Zannettino, A. C. W. (2019). Subclonal evolution in disease progression from MGUS/SMM to multiple myeloma is characterised by clonal stability. Leukemia, 33(2), 457–468. https://doi.org/10.1038/s41375-018-0206-x
Das, R., Strowig, T., Verma, R., Koduru, S., Hafemann, A., Hopf, S., Kocoglu, M. H., Borsotti, C., Zhang, L., Branagan, A., Eynon, E., Manz, M. G., Flavell, R. A., & Dhodapkar, M. V. (2016). Microenvironment-dependent growth of preneoplastic and malignant plasma cells in humanized mice. Nature Medicine, 22(11), 1351–1357. https://doi.org/10.1038/nm.4202
Medyouf, H., Mossner, M., Jann, J.-C., Nolte, F., Raffel, S., Herrmann, C., Lier, A., Eisen, C., Nowak, V., Zens, B., Müdder, K., Klein, C., Obländer, J., Fey, S., Vogler, J., Fabarius, A., Riedl, E., Roehl, H., Kohlmann, A., Staller, M., et al. (2014). Myelodysplastic cells in patients reprogram mesenchymal stromal cells to establish a transplantable stem cell niche disease unit. Cell Stem Cell, 14(6), 824–837. https://doi.org/10.1016/j.stem.2014.02.014
Balderman, S. R., Li, A. J., Hoffman, C. M., Frisch, B. J., Goodman, A. N., LaMere, M. W., Georger, M. A., Evans, A. G., Liesveld, J. L., Becker, M. W., & Calvi, L. M. (2016). Targeting of the bone marrow microenvironment improves outcome in a murine model of myelodysplastic syndrome. Blood, 127(5), 616–625. https://doi.org/10.1182/blood-2015-06-653113
Baker, D. J., Childs, B. G., Durik, M., Wijers, M. E., Sieben, C. J., Zhong, J., Saltness, A., & R., Jeganathan, K. B., Verzosa, G. C., Pezeshki, A., Khazaie, K., Miller, J. D., & van Deursen, J. M. (2016). Naturally occurring p16Ink4a-positive cells shorten healthy lifespan. Nature, 530, 184. https://doi.org/10.1038/nature16932
Jeon, O. H., Kim, C., Laberge, R. M., Demaria, M., Rathod, S., Vasserot, A. P., Chung, J. W., Kim, D. H., Poon, Y., David, N., Baker, D. J., van Deursen, J. M., Campisi, J., & Elisseeff, J. H. (2017). Local clearance of senescent cells attenuates the development of post-traumatic osteoarthritis and creates a pro-regenerative environment. Nature Medicine, 23(6), 775–781. https://doi.org/10.1038/nm.4324
Farr, J. N., Fraser, D. G., Wang, H., Jaehn, K., Ogrodnik, M. B., Weivoda, M. M., Drake, M. T., Tchkonia, T., LeBrasseur, N. K., Kirkland, J. L., Bonewald, L. F., Pignolo, R. J., Monroe, D. G., & Khosla, S. (2016). Identification of Senescent Cells in the Bone Microenvironment. Journal of Bone and Mineral Research, 31(11), 1920–1929. https://doi.org/10.1002/jbmr.2892
Lawrenson, K., Grun, B., Benjamin, E., Jacobs, I. J., Dafou, D., & Gayther, S. A. (2010). Senescent fibroblasts promote neoplastic transformation of partially transformed ovarian epithelial cells in a three-dimensional model of early stage ovarian cancer. Neoplasia, 12(4), 317–325. https://doi.org/10.1593/neo.91948
Krtolica, A., Parrinello, S., Lockett, S., Desprez, P. Y., & Campisi, J. (2001). Senescent fibroblasts promote epithelial cell growth and tumorigenesis: A link between cancer and aging. Proceedings of the National Academy of Sciences of the United States of America, 98(21), 12072–12077. https://doi.org/10.1073/pnas.211053698
Coppé, J. P., Patil, C. K., Rodier, F., Krtolica, A., Beauséjour, C. M., Parrinello, S., Hodgson, J. G., Chin, K., Desprez, P. Y., & Campisi, J. (2010). A human-like senescence-associated secretory phenotype is conserved in mouse cells dependent on physiological oxygen. PLoS ONE, 5(2), e9188. https://doi.org/10.1371/journal.pone.0009188
Di, G. H., Liu, Y., Lu, Y., Liu, J., Wu, C., & Duan, H. F. (2014). IL-6 secreted from senescent mesenchymal stem cells promotes proliferation and migration of breast cancer cells. PLoS ONE, 9(11), e113572. https://doi.org/10.1371/journal.pone.0113572
Laberge, R. M., Sun, Y., Orjalo, A. V., Patil, C. K., Freund, A., Zhou, L., Curran, S. C., Davalos, A. R., Wilson-Edell, K. A., Liu, S., Limbad, C., Demaria, M., Li, P., Hubbard, G. B., Ikeno, Y., Javors, M., Desprez, P. Y., Benz, C. C., Kapahi, P., Nelson, P. S., et al. (2015). MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nature Cell Biology, 17(8), 1049–1061. https://doi.org/10.1038/ncb3195
Liu, D., & Hornsby, P. J. (2007). Senescent human fibroblasts increase the early growth of xenograft tumors via matrix metalloproteinase secretion. Cancer Research, 67(7), 3117–3126. https://doi.org/10.1158/0008-5472.Can-06-3452
Luo, X., Fu, Y., Loza, A. J., Murali, B., Leahy, K. M., Ruhland, M. K., Gang, M., Su, X., Zamani, A., Shi, Y., Lavine, K. J., Ornitz, D. M., Weilbaecher, K. N., Long, F., Novack, D. V., Faccio, R., Longmore, G. D., & Stewart, S. A. (2016). Stromal-initiated changes in the bone promote metastatic niche development. Cell Reports, 14(1), 82–92. https://doi.org/10.1016/j.celrep.2015.12.016
Dominici, M., Le Blanc, K., Mueller, I., Slaper-Cortenbach, I., Marini, F., Krause, D., Deans, R., Keating, A., Prockop, D., & Horwitz, E. (2006). Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy, 8(4), 315–317. https://doi.org/10.1080/14653240600855905
Goulard, M., Dosquet, C., & Bonnet, D. (2018). Role of the microenvironment in myeloid malignancies. Cellular and Molecular Life Sciences, 75(8), 1377–1391. https://doi.org/10.1007/s00018-017-2725-4
Mendelson, A., & Frenette, P. S. (2014). Hematopoietic stem cell niche maintenance during homeostasis and regeneration. Nature Medicine, 20(8), 833–846. https://doi.org/10.1038/nm.3647
Wei, Q., & Frenette, P. S. (2018). Niches for Hematopoietic Stem Cells and Their Progeny. Immunity, 48(4), 632–648. https://doi.org/10.1016/j.immuni.2018.03.024
Mitroulis, I., Kalafati, L., Bornhauser, M., Hajishengallis, G., & Chavakis, T. (2020). Regulation of the bone marrow niche by inflammation. Frontiers in Immunology, 11, 1540. https://doi.org/10.3389/fimmu.2020.01540
Passegue, E., Wagers, A. J., Giuriato, S., Anderson, W. C., & Weissman, I. L. (2005). Global analysis of proliferation and cell cycle gene expression in the regulation of hematopoietic stem and progenitor cell fates. Journal of Experimental Medicine, 202(11), 1599–1611. https://doi.org/10.1084/jem.20050967
Kunisaki, Y., Bruns, I., Scheiermann, C., Ahmed, J., Pinho, S., Zhang, D., Mizoguchi, T., Wei, Q., Lucas, D., Ito, K., Mar, J. C., Bergman, A., & Frenette, P. S. (2013). Arteriolar niches maintain haematopoietic stem cell quiescence. Nature, 502(7473), 637–643. https://doi.org/10.1038/nature12612
Acar, M., Kocherlakota, K. S., Murphy, M. M., Peyer, J. G., Oguro, H., Inra, C. N., Jaiyeola, C., Zhao, Z., Luby-Phelps, K., & Morrison, S. J. (2015). Deep imaging of bone marrow shows non-dividing stem cells are mainly perisinusoidal. Nature, 526(7571), 126–130. https://doi.org/10.1038/nature15250
Sugiyama, T., Kohara, H., Noda, M., & Nagasawa, T. (2006). Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity, 25(6), 977–988. https://doi.org/10.1016/j.immuni.2006.10.016
Omatsu, Y., Sugiyama, T., Kohara, H., Kondoh, G., Fujii, N., Kohno, K., & Nagasawa, T. (2010). The essential functions of adipo-osteogenic progenitors as the hematopoietic stem and progenitor cell niche. Immunity, 33(3), 387–399. https://doi.org/10.1016/j.immuni.2010.08.017
Mendez-Ferrer, S., Michurina, T. V., Ferraro, F., Mazloom, A. R., Macarthur, B. D., Lira, S. A., Scadden, D. T., Ma’ayan, A., Enikolopov, G. N., & Frenette, P. S. (2010). Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature, 466(7308), 829–834. https://doi.org/10.1038/nature09262
Zhou, B. O., Yue, R., Murphy, M. M., Peyer, J. G., & Morrison, S. J. (2014). Leptin-receptor-expressing mesenchymal stromal cells represent the main source of bone formed by adult bone marrow. Cell Stem Cell, 15(2), 154–168. https://doi.org/10.1016/j.stem.2014.06.008
Lawson, M. A., McDonald, M. M., Kovacic, N., Hua Khoo, W., Terry, R. L., Down, J., Kaplan, W., Paton-Hough, J., Fellows, C., Pettitt, J. A., Neil Dear, T., Van Valckenborgh, E., Baldock, P. A., Rogers, M. J., Eaton, C. L., Vanderkerken, K., Pettit, A. R., Quinn, J. M., Zannettino, A. C., Phan, T. G., et al. (2015). Osteoclasts control reactivation of dormant myeloma cells by remodelling the endosteal niche. Nature Communications, 6, 8983. https://doi.org/10.1038/ncomms9983
Agarwal, P., & Bhatia, R. (2015). Influence of bone marrow microenvironment on leukemic stem cells: Breaking up an intimate relationship. Advances in Cancer Research, 127, 227–252. https://doi.org/10.1016/bs.acr.2015.04.007
Agarwal, P., Isringhausen, S., Li, H., Paterson, A. J., He, J., Gomariz, A., Nagasawa, T., Nombela-Arrieta, C., & Bhatia, R. (2019). Mesenchymal niche-specific expression of Cxcl12 controls quiescence of treatment-resistant leukemia stem cells. Cell Stem Cell, 24(5), 769–784 e766. https://doi.org/10.1016/j.stem.2019.02.018
Kode, A., Manavalan, J. S., Mosialou, I., Bhagat, G., Rathinam, C. V., Luo, N., Khiabanian, H., Lee, A., Murty, V. V., Friedman, R., Brum, A., Park, D., Galili, N., Mukherjee, S., Teruya-Feldstein, J., Raza, A., Rabadan, R., Berman, E., & Kousteni, S. (2014). Leukaemogenesis induced by an activating beta-catenin mutation in osteoblasts. Nature, 506(7487), 240–244. https://doi.org/10.1038/nature12883
Krevvata, M., Silva, B. C., Manavalan, J. S., Galan-Diez, M., Kode, A., Matthews, B. G., Park, D., Zhang, C. A., Galili, N., Nickolas, T. L., Dempster, D. W., Dougall, W., Teruya-Feldstein, J., Economides, A. N., Kalajzic, I., Raza, A., Berman, E., Mukherjee, S., Bhagat, G., & Kousteni, S. (2014). Inhibition of leukemia cell engraftment and disease progression in mice by osteoblasts. Blood, 124(18), 2834–2846. https://doi.org/10.1182/blood-2013-07-517219
Bowers, M., Zhang, B., Ho, Y., Agarwal, P., Chen, C. C., & Bhatia, R. (2015). Osteoblast ablation reduces normal long-term hematopoietic stem cell self-renewal but accelerates leukemia development. Blood, 125(17), 2678–2688. https://doi.org/10.1182/blood-2014-06-582924
Raaijmakers, M. H., Mukherjee, S., Guo, S., Zhang, S., Kobayashi, T., Schoonmaker, J. A., Ebert, B. L., Al-Shahrour, F., Hasserjian, R. P., Scadden, E. O., Aung, Z., Matza, M., Merkenschlager, M., Lin, C., Rommens, J. M., & Scadden, D. T. (2010). Bone progenitor dysfunction induces myelodysplasia and secondary leukaemia. Nature, 464(7290), 852–857. https://doi.org/10.1038/nature08851
Childs, B. G., Durik, M., Baker, D. J., & van Deursen, J. M. (2015). Cellular senescence in aging and age-related disease: From mechanisms to therapy. Nature Medicine, 21(12), 1424–1435. https://doi.org/10.1038/nm.4000
Dimri, G. P., Lee, X., Basile, G., Acosta, M., Scott, G., Roskelley, C., Medrano, E. E., Linskens, M., Rubelj, I., Pereira-Smith, O., &, et al. (1995). A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proceedings of the National Academy of Sciences of the United States of America, 92(20), 9363–9367. https://doi.org/10.1073/pnas.92.20.9363
Mammoto, T., Torisawa, Y. S., Muyleart, M., Hendee, K., Anugwom, C., Gutterman, D., & Mammoto, A. (2019). Effects of age-dependent changes in cell size on endothelial cell proliferation and senescence through YAP1. Aging, 11(17), 7051–7069. https://doi.org/10.18632/aging.102236
Kumari, R., & Jat, P. (2021). Mechanisms of cellular senescence: Cell cycle arrest and senescence associated secretory phenotype. Frontiers in Cell and Developmental Biology, 9, 645593. https://doi.org/10.3389/fcell.2021.645593
Mirzayans, R., Andrais, B., Hansen, G., & Murray, D. (2012). Role of p16(INK4A) in Replicative Senescence and DNA Damage-induced premature senescence in p53-deficient human cells. Biochemistry Research International, 2012, 951574. https://doi.org/10.1155/2012/951574
Petrova, N. V., Velichko, A. K., Razin, S. V., & Kantidze, O. L. (2016). Small molecule compounds that induce cellular senescence. Aging Cell, 15(6), 999–1017. https://doi.org/10.1111/acel.12518
Krishnamurthy, J., Torrice, C., Ramsey, M. R., Kovalev, G. I., Al-Regaiey, K., Su, L., & Sharpless, N. E. (2004). Ink4a/Arf expression is a biomarker of aging. Journal of Clinical Investigation, 114(9), 1299–1307. https://doi.org/10.1172/jci22475
Roninson, I. B. (2003). Tumor cell senescence in cancer treatment. Cancer Research, 63(11), 2705–2715.
van Deursen, J. M. (2014). The role of senescent cells in ageing. Nature, 509(7501), 439–446. https://doi.org/10.1038/nature13193
Wang, E. (1995). Senescent human fibroblasts resist programmed cell death, and failure to suppress bcl2 is involved. Cancer Research, 55(11), 2284.
Ray, D., & Yung, R. (2018). Immune senescence, epigenetics and autoimmunity. Clinical Immunology, 196, 59–63. https://doi.org/10.1016/j.clim.2018.04.002
Prata, L., Ovsyannikova, I. G., Tchkonia, T., & Kirkland, J. L. (2018). Senescent cell clearance by the immune system: Emerging therapeutic opportunities. Seminars in Immunology, 40, 101275. https://doi.org/10.1016/j.smim.2019.04.003
Coppe, J. P., Desprez, P. Y., Krtolica, A., & Campisi, J. (2010). The senescence-associated secretory phenotype: The dark side of tumor suppression. Annual Review of Pathology, 5, 99–118. https://doi.org/10.1146/annurev-pathol-121808-102144
Freund, A., Orjalo, A. V., Desprez, P.-Y., & Campisi, J. (2010). Inflammatory networks during cellular senescence: Causes and consequences. Trends in Molecular Medicine, 16(5), 238–246. https://doi.org/10.1016/j.molmed.2010.03.003
Kale, A., Sharma, A., Stolzing, A., Desprez, P.-Y., & Campisi, J. (2020). Role of immune cells in the removal of deleterious senescent cells. Immunity & Ageing, 17(1), 16. https://doi.org/10.1186/s12979-020-00187-9
Brennan, F. M., Maini, R. N., & Feldmann, M. (1995). Cytokine expression in chronic inflammatory disease. British Medical Bulletin, 51(2), 368–384. https://doi.org/10.1093/oxfordjournals.bmb.a072967
Caruso, C., Lio, D., Cavallone, L., & Franceschi, C. (2004). Aging, longevity, inflammation, and cancer. Annals of the New York Academy of Sciences, 1028, 1–13. https://doi.org/10.1196/annals.1322.001
Brod, S. A. (2000). Unregulated inflammation shortens human functional longevity. Inflammation Research, 49(11), 561–570. https://doi.org/10.1007/s000110050632
Buscarlet, M., Provost, S., Zada, Y. F., Barhdadi, A., Bourgoin, V., Lepine, G., Mollica, L., Szuber, N., Dube, M. P., & Busque, L. (2017). DNMT3A and TET2 dominate clonal hematopoiesis and demonstrate benign phenotypes and different genetic predispositions. Blood, 130(6), 753–762. https://doi.org/10.1182/blood-2017-04-777029
Beerman, I., Bhattacharya, D., Zandi, S., Sigvardsson, M., Weissman, I. L., Bryder, D., & Rossi, D. J. (2010). Functionally distinct hematopoietic stem cells modulate hematopoietic lineage potential during aging by a mechanism of clonal expansion. Proceedings of the National Academy of Sciences of the United States of America, 107(12), 5465–5470. https://doi.org/10.1073/pnas.1000834107
Linton, P. J., & Dorshkind, K. (2004). Age-related changes in lymphocyte development and function. Nature Immunology, 5(2), 133–139. https://doi.org/10.1038/ni1033
Guralnik, J. M., Eisenstaedt, R. S., Ferrucci, L., Klein, H. G., & Woodman, R. C. (2004). Prevalence of anemia in persons 65 years and older in the United States: Evidence for a high rate of unexplained anemia. Blood, 104(8), 2263–2268. https://doi.org/10.1182/blood-2004-05-1812
Ogawa, T., Kitagawa, M., & Hirokawa, K. (2000). Age-related changes of human bone marrow: A histometric estimation of proliferative cells, apoptotic cells, T cells, B cells and macrophages. Mechanisms of Ageing and Development, 117(1–3), 57–68. https://doi.org/10.1016/s0047-6374(00)00137-8
Bousounis, P., Bergo, V., & Trompouki, E. (2021). Inflammation, aging and hematopoiesis: a complex relationship. Cells, 10(6), 1386. https://doi.org/10.3390/cells10061386
Ergen, A. V., Boles, N. C., & Goodell, M. A. (2012). Rantes/Ccl5 influences hematopoietic stem cell subtypes and causes myeloid skewing. Blood, 119(11), 2500–2509. https://doi.org/10.1182/blood-2011-11-391730
Zhao, Y., Wu, D., Fei, C., Guo, J., Gu, S., Zhu, Y., Xu, F., Zhang, Z., Wu, L., Li, X., & Chang, C. (2015). Down-regulation of Dicer1 promotes cellular senescence and decreases the differentiation and stem cell-supporting capacities of mesenchymal stromal cells in patients with myelodysplastic syndrome. Haematologica, 100(2), 194–204. https://doi.org/10.3324/haematol.2014.109769
Guo, J., Zhao, Y., Fei, C., Zhao, S., Zheng, Q., Su, J., Wu, D., Li, X., & Chang, C. (2018). Dicer1 downregulation by multiple myeloma cells promotes the senescence and tumor-supporting capacity and decreases the differentiation potential of mesenchymal stem cells. Cell Death & Disease, 9(5), 512. https://doi.org/10.1038/s41419-018-0545-6
Pang, W. W., Price, E. A., Sahoo, D., Beerman, I., Maloney, W. J., Rossi, D. J., Schrier, S. L., & Weissman, I. L. (2011). Human bone marrow hematopoietic stem cells are increased in frequency and myeloid-biased with age. Proceedings of the National Academy of Sciences of the United States of America, 108(50), 20012–20017. https://doi.org/10.1073/pnas.1116110108
O’Hagan-Wong, K., Nadeau, S., Carrier-Leclerc, A., Apablaza, F., Hamdy, R., Shum-Tim, D., Rodier, F., & Colmegna, I. (2016). Increased IL-6 secretion by aged human mesenchymal stromal cells disrupts hematopoietic stem and progenitor cells’ homeostasis. Oncotarget, 7(12), 13285–13296. https://doi.org/10.18632/oncotarget.7690
Adams, P. D., Jasper, H., & Rudolph, K. L. (2015). Aging-induced stem cell mutations as drivers for disease and cancer. Cell Stem Cell, 16, 601–612. https://doi.org/10.1016/j.stem.2015.05.002
Tsai, K. K., Chuang, E. Y., Little, J. B., & Yuan, Z. M. (2005). Cellular mechanisms for low-dose ionizing radiation-induced perturbation of the breast tissue microenvironment. Cancer Research, 65(15), 6734–6744. https://doi.org/10.1158/0008-5472.CAN-05-0703
Bavik, C., Coleman, I., Dean, J. P., Knudsen, B., Plymate, S., & Nelson, P. S. (2006). The gene expression program of prostate fibroblast senescence modulates neoplastic epithelial cell proliferation through paracrine mechanisms. Cancer Research, 66(2), 794–802. https://doi.org/10.1158/0008-5472.CAN-05-1716
Takasugi, M., Okada, R., Takahashi, A., Virya Chen, D., Watanabe, S., & Hara, E. (2017). Small extracellular vesicles secreted from senescent cells promote cancer cell proliferation through EphA2. Nature Communications, 8(1), 15729. https://doi.org/10.1038/ncomms15728
Li, Y., Xu, X., Wang, L., Liu, G., Li, Y., Wu, X., Jing, Y., Li, H., & Wang, G. (2015). Senescent mesenchymal stem cells promote colorectal cancer cells growth via galectin-3 expression. Cell & Bioscience, 5, 21. https://doi.org/10.1186/s13578-015-0012-3
Capell, B. C., Drake, A. M., Zhu, J., Shah, P. P., Dou, Z., Dorsey, J., Simola, D. F., Donahue, G., Sammons, M., Rai, T. S., Natale, C., Ridky, T. W., Adams, P. D., & Berger, S. L. (2016). MLL1 is essential for the senescence-associated secretory phenotype. Genes and Development, 30(3), 321–336. https://doi.org/10.1101/gad.271882.115
Coppe, J. P., Patil, C. K., Rodier, F., Sun, Y., Munoz, D. P., Goldstein, J., Nelson, P. S., Desprez, P. Y., & Campisi, J. (2008). Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biology, 6(12), 2853–2868. https://doi.org/10.1371/journal.pbio.0060301
Krause, J. R. (2009). WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues: An Overview. Critical Values, 2(2), 30–32. https://doi.org/10.1093/criticalvalues/2.2.30
Geyh, S., Oz, S., Cadeddu, R. P., Frobel, J., Bruckner, B., Kundgen, A., Fenk, R., Bruns, I., Zilkens, C., Hermsen, D., Gattermann, N., Kobbe, G., Germing, U., Lyko, F., Haas, R., & Schroeder, T. (2013). Insufficient stromal support in MDS results from molecular and functional deficits of mesenchymal stromal cells. Leukemia, 27(9), 1841–1851. https://doi.org/10.1038/leu.2013.193
Poloni, A., Maurizi, G., Mattiucci, D., Amatori, S., Fogliardi, B., Costantini, B., Mariani, M., Mancini, S., Olivieri, A., Fanelli, M., & Leoni, P. (2014). Overexpression of CDKN2B (p15INK4B) and altered global DNA methylation status in mesenchymal stem cells of high-risk myelodysplastic syndromes. Leukemia, 28(11), 2241–2244. https://doi.org/10.1038/leu.2014.197
Lopez-Villar, O., Garcia, J. L., Sanchez-Guijo, F. M., Robledo, C., Villaron, E. M., Hernández-Campo, P., Lopez-Holgado, N., Diez-Campelo, M., Barbado, M. V., Perez-Simon, J. A., Hernández-Rivas, J. M., San-Miguel, J. F., & del Cañizo, M. C. (2009). Both expanded and uncultured mesenchymal stem cells from MDS patients are genomically abnormal, showing a specific genetic profile for the 5q− syndrome. Leukemia, 23, 664. https://doi.org/10.1038/leu.2008.361
Aanei, C. M., Flandrin, P., Eloae, F. Z., Carasevici, E., Guyotat, D., Wattel, E., & Campos, L. (2012). Intrinsic growth deficiencies of mesenchymal stromal cells in myelodysplastic syndromes. Stem Cells and Development, 21(10), 1604–1615. https://doi.org/10.1089/scd.2011.0390
Fei, C., Zhao, Y., Guo, J., Gu, S., Li, X., & Chang, C. (2014). Senescence of bone marrow mesenchymal stromal cells is accompanied by activation of p53/p21 pathway in myelodysplastic syndromes. European Journal of Haematology, 93(6), 476–486. https://doi.org/10.1111/ejh.12385
Kornblau, S. M., Ruvolo, P. P., Wang, R. Y., Battula, V. L., Shpall, E. J., Ruvolo, V. R., McQueen, T., Qui, Y., Zeng, Z., Pierce, S., Jacamo, R., Yoo, S. Y., Le, P. M., Sun, J., Hail, N., Jr., Konopleva, M., & Andreeff, M. (2018). Distinct protein signatures of acute myeloid leukemia bone marrow-derived stromal cells are prognostic for patient survival. Haematologica, 103(5), 810–821. https://doi.org/10.3324/haematol.2017.172429
Chandran, P., Le, Y., Li, Y., Sabloff, M., Mehic, J., Rosu-Myles, M., & Allan, D. S. (2015). Mesenchymal stromal cells from patients with acute myeloid leukemia have altered capacity to expand differentiated hematopoietic progenitors. Leukemia Research, 39(4), 486–493. https://doi.org/10.1016/j.leukres.2015.01.013
Choi, H., Kim, Y., Kang, D., Kwon, A., Kim, J., Min Kim, J., Park, S. S., Kim, Y. J., Min, C. K., & Kim, M. (2020). Common and different alterations of bone marrow mesenchymal stromal cells in myelodysplastic syndrome and multiple myeloma. Cell Proliferation, 53(5), e12819. https://doi.org/10.1111/cpr.12819
Geyh, S., Rodríguez-Paredes, M., Jäger, P., Khandanpour, C., Cadeddu, R. P., Gutekunst, J., Wilk, C. M., Fenk, R., Zilkens, C., Hermsen, D., Germing, U., Kobbe, G., Lyko, F., Haas, R., & Schroeder, T. (2016). Functional inhibition of mesenchymal stromal cells in acute myeloid leukemia. Leukemia, 30(3), 683–691. https://doi.org/10.1038/leu.2015.325
Falconi, G., Fabiani, E., Fianchi, L., Criscuolo, M., Raffaelli, C. S., Bellesi, S., Hohaus, S., Voso, M. T., D'Alo, F., & Leone, G. (2016). Impairment of PI3K/AKT and WNT/beta-catenin pathways in bone marrow mesenchymal stem cells isolated from patients with myelodysplastic syndromes. Experimental Hematology, 44(1), 75–83 e71–74, https://doi.org/10.1016/j.exphem.2015.10.005
Poon, Z., Dighe, N., Venkatesan, S. S., Cheung, A. M. S., Fan, X., Bari, S., Hota, M., Ghosh, S., & Hwang, W. Y. K. (2019). Bone marrow MSCs in MDS: Contribution towards dysfunctional hematopoiesis and potential targets for disease response to hypomethylating therapy. Leukemia, 33(6), 1487–1500. https://doi.org/10.1038/s41375-018-0310-y
Boada, M., Echarte, L., Guillermo, C., Diaz, L., Touriño, C., & Grille, S. (2021). 5-Azacytidine restores interleukin 6-increased production in mesenchymal stromal cells from myelodysplastic patients. Hematology, Transfusion and Cell Therapy, 43(1), 35–42. https://doi.org/10.1016/j.htct.2019.12.0
Lopes, M. R., Pereira, J. K., de Melo Campos, P., Machado-Neto, J. A., Traina, F., Saad, S. T., & Favaro, P. (2017). De novo AML exhibits greater microenvironment dysregulation compared to AML with myelodysplasia-related changes. Scientific Reports, 7, 40707. https://doi.org/10.1038/srep40707
von der Heide, E. K., Neumann, M., Vosberg, S., James, A. R., Schroeder, M. P., Ortiz-Tanchez, J., Isaakidis, K., Schlee, C., Luther, M., Johrens, K., Anagnostopoulos, I., Mochmann, L. H., Nowak, D., Hofmann, W. K., Greif, P. A., & Baldus, C. D. (2017). Molecular alterations in bone marrow mesenchymal stromal cells derived from acute myeloid leukemia patients. Leukemia, 31(5), 1069–1078. https://doi.org/10.1038/leu.2016.324
Cheng, J., Li, Y., Liu, S., Jiang, Y., Ma, J., Wan, L., Li, Q., & Pang, T. (2019). CXCL8 derived from mesenchymal stromal cells supports survival and proliferation of acute myeloid leukemia cells through the PI3K/AKT pathway. The Federation of American Societies of Experimental Biology Journal, 33(4), 4755–4764. https://doi.org/10.1096/fj.201801931R
Pardanani, A., Finke, C., Lasho, T. L., Al-Kali, A., Begna, K. H., Hanson, C. A., & Tefferi, A. (2012). IPSS-independent prognostic value of plasma CXCL10, IL-7 and IL-6 levels in myelodysplastic syndromes. Leukemia, 26(4), 693–699. https://doi.org/10.1038/leu.2011.251
Shi, X., Zheng, Y., Xu, L., Cao, C., Dong, B., & Chen, X. (2019). The inflammatory cytokine profile of myelodysplastic syndromes: A meta-analysis. Medicine (Baltimore), 98(22), e15844. https://doi.org/10.1097/MD.0000000000015844
Desbourdes, L., Javary, J., Charbonnier, T., Ishac, N., Bourgeais, J., Iltis, A., Chomel, J. C., Turhan, A., Guilloton, F., Tarte, K., Demattei, M. V., Ducrocq, E., Rouleux-Bonnin, F., Gyan, E., Herault, O., & Domenech, J. (2017). Alteration analysis of bone marrow mesenchymal stromal cells from de novo acute myeloid leukemia patients at diagnosis. Stem Cells and Development, 26(10), 709–722. https://doi.org/10.1089/scd.2016.0295
Abdul-Aziz, A. M., Sun, Y., Hellmich, C., Marlein, C. R., Mistry, J., Forde, E., Piddock, R. E., Shafat, M. S., Morfakis, A., Mehta, T., Di Palma, F., Macaulay, I., Ingham, C. J., Haestier, A., Collins, A., Campisi, J., Bowles, K. M., & Rushworth, S. A. (2019). Acute myeloid leukemia induces protumoral p16INK4a-driven senescence in the bone marrow microenvironment. Blood, 133(5), 446–456. https://doi.org/10.1182/blood-2018-04-845420
Su, Y. C., Li, S. C., Wu, Y. C., Wang, L. M., Chao, K. S., & Liao, H. F. (2013). Resveratrol downregulates interleukin-6-stimulated sonic hedgehog signaling in human acute myeloid leukemia. Evidence-Based Complementary and Alternative Medicine, 2013, 547430. https://doi.org/10.1155/2013/547430
Saily, M., Koistinen, P., Zheng, A., & Savolainen, E. R. (1998). Signaling through interleukin-6 receptor supports blast cell proliferation in acute myeloblastic leukemia. European Journal of Haematology, 61(3), 190–196. https://doi.org/10.1111/j.1600-0609.1998.tb01083.x
Abdul-Aziz, A. M., Shafat, M. S., Mehta, T. K., Di Palma, F., Lawes, M. J., Rushworth, S. A., & Bowles, K. M. (2017). MIF-Induced Stromal PKCbeta/IL8 Is Essential in Human Acute Myeloid Leukemia. Cancer Research, 77(2), 303–311. https://doi.org/10.1158/0008-5472.CAN-16-1095
Hallek, M., Cheson, B. D., Catovsky, D., Caligaris-Cappio, F., Dighiero, G., Dohner, H., Hillmen, P., Keating, M. J., Montserrat, E., Rai, K. R., Kipps, T. J., & International Workshop on Chronic Lymphocytic, L. (2008). Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: A report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood, 111(12), 5446–5456. https://doi.org/10.1182/blood-2007-06-093906
Pontikoglou, C., Kastrinaki, M. C., Klaus, M., Kalpadakis, C., Katonis, P., Alpantaki, K., Pangalis, G. A., & Papadaki, H. A. (2013). Study of the quantitative, functional, cytogenetic, and immunoregulatory properties of bone marrow mesenchymal stem cells in patients with B-cell chronic lymphocytic leukemia. Stem Cells and Development, 22(9), 1329–1341. https://doi.org/10.1089/scd.2012.0255
Janel, A., Dubois-Galopin, F., Bourgne, C., Berger, J., Tarte, K., Boiret-Dupre, N., Boisgard, S., Verrelle, P., Dechelotte, P., Tournilhac, O., & Berger, M. G. (2014). The chronic lymphocytic leukemia clone disrupts the bone marrow microenvironment. Stem Cells and Development, 23(24), 2972–2982. https://doi.org/10.1089/scd.2014.0229
Kay, N. E., Shanafelt, T. D., Strege, A. K., Lee, Y. K., Bone, N. D., & Raza, A. (2007). Bone biopsy derived marrow stromal elements rescue chronic lymphocytic leukemia B-cells from spontaneous and drug induced cell death and facilitates an “angiogenic switch.” Leukemia Research, 31(7), 899–906. https://doi.org/10.1016/j.leukres.2006.11.024
Fayad, L., Keating, M. J., Reuben, J. M., O’Brien, S., Lee, B. N., Lerner, S., & Kurzrock, R. (2001). Interleukin-6 and interleukin-10 levels in chronic lymphocytic leukemia: Correlation with phenotypic characteristics and outcome. Blood, 97(1), 256–263. https://doi.org/10.1182/blood.v97.1.256
Molica, S., Vitelli, G., Levato, D., Gandolfo, G. M., & Liso, V. (1999). Increased serum levels of vascular endothelial growth factor predict risk of progression in early B-cell chronic lymphocytic leukaemia. British Journal of Haematology, 107(3), 605–610. https://doi.org/10.1046/j.1365-2141.1999.01752.x
Wierda, W. G., Johnson, M. M., Do, K. A., Manshouri, T., Dey, A., O’Brien, S., Giles, F. J., Kantarjian, H., Thomas, D., Faderl, S., Lerner, S., Keating, M., & Albitar, M. (2003). Plasma interleukin 8 level predicts for survival in chronic lymphocytic leukaemia. British Journal of Haematology, 120(3), 452–456. https://doi.org/10.1046/j.1365-2141.2003.04118.x
Yoon, J. Y., Lafarge, S., Dawe, D., Lakhi, S., Kumar, R., Morales, C., Marshall, A., Gibson, S. B., & Johnston, J. B. (2012). Association of interleukin-6 and interleukin-8 with poor prognosis in elderly patients with chronic lymphocytic leukemia. Leukemia and Lymphoma, 53(9), 1735–1742. https://doi.org/10.3109/10428194.2012.666662
Lai, R., O’Brien, S., Maushouri, T., Rogers, A., Kantarjian, H., Keating, M., & Albitar, M. (2002). Prognostic value of plasma interleukin-6 levels in patients with chronic lymphocytic leukemia. Cancer, 95(5), 1071–1075. https://doi.org/10.1002/cncr.10772
Ferrajoli, A., Keating, M. J., Manshouri, T., Giles, F. J., Dey, A., Estrov, Z., Koller, C. A., Kurzrock, R., Thomas, D. A., Faderl, S., Lerner, S., O’Brien, S., & Albitar, M. (2002). The clinical significance of tumor necrosis factor-alpha plasma level in patients having chronic lymphocytic leukemia. Blood, 100(4), 1215–1219. https://doi.org/10.1182/blood.V100.4.1215.h81602001215_1215_1219
Zhu, F., McCaw, L., Spaner, D. E., & Gorczynski, R. M. (2018). Targeting the IL-17/IL-6 axis can alter growth of chronic lymphocytic leukemia in vivo/in vitro. Leukemia Research, 66, 28–38. https://doi.org/10.1016/j.leukres.2018.01.006
Panayiotidis, P., Jones, D., Ganeshaguru, K., Foroni, L., & Hoffbrand, A. V. (1996). Human bone marrow stromal cells prevent apoptosis and support the survival of chronic lymphocytic leukaemia cells in vitro. British Journal of Haematology, 92(1), 97–103. https://doi.org/10.1046/j.1365-2141.1996.00305.x
Trimarco, V., Ave, E., Facco, M., Chiodin, G., Frezzato, F., Martini, V., Gattazzo, C., Lessi, F., Giorgi, C. A., Visentin, A., Castelli, M., Severin, F., Zambello, R., Piazza, F., Semenzato, G., & Trentin, L. (2015). Cross-talk between chronic lymphocytic leukemia (CLL) tumor B cells and mesenchymal stromal cells (MSCs): implications for neoplastic cell survival. Oncotarget, 6(39), 42130–42149. https://doi.org/10.18632/oncotarget.6239
Crompot, E., Van Damme, M., Pieters, K., Vermeersch, M., Perez-Morga, D., Mineur, P., Maerevoet, M., Meuleman, N., Bron, D., Lagneaux, L., & Stamatopoulos, B. (2017). Extracellular vesicles of bone marrow stromal cells rescue chronic lymphocytic leukemia B cells from apoptosis, enhance their migration and induce gene expression modifications. Haematologica, 102(9), 1594–1604. https://doi.org/10.3324/haematol.2016.163337
Gehrke, I., Gandhirajan, R. K., Poll-Wolbeck, S. J., Hallek, M., & Kreuzer, K. A. (2011). Bone marrow stromal cell-derived vascular endothelial growth factor (VEGF) rather than chronic lymphocytic leukemia (CLL) cell-derived VEGF is essential for the apoptotic resistance of cultured CLL cells. Molecular Medicine, 17(7–8), 619–627. https://doi.org/10.2119/molmed.2010.00210
Lee, Y. K., Bone, N. D., Strege, A. K., Shanafelt, T. D., Jelinek, D. F., & Kay, N. E. (2004). VEGF receptor phosphorylation status and apoptosis is modulated by a green tea component, epigallocatechin-3-gallate (EGCG). B-cell chronic lymphocytic leukemia. Blood, 104(3), 788–794. https://doi.org/10.1182/blood-2003-08-2763
Paesler, J., Gehrke, I., Gandhirajan, R. K., Filipovich, A., Hertweck, M., Erdfelder, F., Uhrmacher, S., Poll-Wolbeck, S. J., Hallek, M., & Kreuzer, K. A. (2010). The vascular endothelial growth factor receptor tyrosine kinase inhibitors vatalanib and pazopanib potently induce apoptosis in chronic lymphocytic leukemia cells in vitro and in vivo. Clinical Cancer Research, 16(13), 3390–3398. https://doi.org/10.1158/1078-0432.CCR-10-0232
Francia di Celle, P., Mariani, S., Riera, L., Stacchini, A., Reato, G., & Foa, R. (1996). Interleukin-8 induces the accumulation of B-cell chronic lymphocytic leukemia cells by prolonging survival in an autocrine fashion. Blood, 87(10), 4382–4389. https://doi.org/10.1182/blood.V87.10.4382.bloodjournal87104382
Blanco, G., Puiggros, A., Sherry, B., Nonell, L., Calvo, X., Puigdecanet, E., Chiu, P. Y., Kieso, Y., Ferrer, G., Palacios, F., Arnal, M., Rodriguez-Rivera, M., Gimeno, E., Abella, E., Rai, K. R., Abrisqueta, P., Bosch, F., Calon, A., Ferrer, A., Chiorazzi, N., et al. (2021). Chronic lymphocytic leukemia-like monoclonal B-cell lymphocytosis exhibits an increased inflammatory signature that is reduced in early-stage chronic lymphocytic leukemia. Experimental Hematology, 95, 68–80. https://doi.org/10.1016/j.exphem.2020.12.007
Aguilar-Santelises, M., Loftenius, A., Ljungh, C., Svenson, S. B., Andersson, B., Mellstedt, H., & Jondal, M. (1992). Serum levels of helper factors (IL-1α, IL-1β and IL-6), T-cell products (sCD4 and sCD8), sIL-2R and β2-microglobulin in patients with B-CLL and benign B lymphocytosis. Leukemia Research, 16(6–7), 607–613. https://doi.org/10.1016/0145-2126(92)90009-v
Rajkumar, S. V., Dimopoulos, M. A., Palumbo, A., Blade, J., Merlini, G., Mateos, M. V., Kumar, S., Hillengass, J., Kastritis, E., Richardson, P., Landgren, O., Paiva, B., Dispenzieri, A., Weiss, B., LeLeu, X., Zweegman, S., Lonial, S., Rosinol, L., Zamagni, E., Jagannath, S., et al. (2014). International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncology, 15(12), e538-548. https://doi.org/10.1016/S1470-2045(14)70442-5
Andre, T., Meuleman, N., Stamatopoulos, B., De Bruyn, C., Pieters, K., Bron, D., & Lagneaux, L. (2013). Evidences of early senescence in multiple myeloma bone marrow mesenchymal stromal cells. PLoS ONE, 8(3), e59756. https://doi.org/10.1371/journal.pone.0059756
Garderet, L., Mazurier, C., Chapel, A., Ernou, I., Boutin, L., Holy, X., Gorin, N. C., Lopez, M., Doucet, C., & Lataillade, J. J. (2007). Mesenchymal stem cell abnormalities in patients with multiple myeloma. Leukemia Lymphoma, 48(10), 2032–2041. https://doi.org/10.1080/10428190701593644
Xu, S., Evans, H., Buckle, C., De Veirman, K., Hu, J., Xu, D., Menu, E., De Becker, A., Vande Broek, I., Leleu, X., Camp, B. V., Croucher, P., Vanderkerken, K., & Van Riet, I. (2012). Impaired osteogenic differentiation of mesenchymal stem cells derived from multiple myeloma patients is associated with a blockade in the deactivation of the Notch signaling pathway. Leukemia, 26(12), 2546–2549. https://doi.org/10.1038/leu.2012.126
Corre, J., Mahtouk, K., Attal, M., Gadelorge, M., Huynh, A., Fleury-Cappellesso, S., Danho, C., Laharrague, P., Klein, B., Reme, T., & Bourin, P. (2007). Bone marrow mesenchymal stem cells are abnormal in multiple myeloma. Leukemia, 21(5), 1079–1088. https://doi.org/10.1038/sj.leu.2404621
Guo, J., Fei, C., Zhao, Y., Zhao, S., Zheng, Q., Su, J., Wu, D., Li, X., & Chang, C. (2017). Lenalidomide restores the osteogenic differentiation of bone marrow mesenchymal stem cells from multiple myeloma patients via deactivating Notch signaling pathway. Oncotarget, 8(33), 55405–55421. https://doi.org/10.18632/oncotarget.19265
Mehdi, S. J., Johnson, S. K., Epstein, J., Zangari, M., Qu, P., Hoering, A., van Rhee, F., Schinke, C., Thanendrarajan, S., Barlogie, B., Davies, F. E., Morgan, G. J., & Yaccoby, S. (2019). Mesenchymal stem cells gene signature in high-risk myeloma bone marrow linked to suppression of distinct IGFBP2-expressing small adipocytes. British Journal of Haematology, 184(4), 578–593. https://doi.org/10.1111/bjh.15669
Berenstein, R., Blau, O., Nogai, A., Waechter, M., Slonova, E., Schmidt-Hieber, M., Kunitz, A., Pezzutto, A., Doerken, B., & Blau, I. W. (2015). Multiple myeloma cells alter the senescence phenotype of bone marrow mesenchymal stromal cells under participation of the DLK1-DIO3 genomic region. BMC Cancer, 15(1), 68. https://doi.org/10.1186/s12885-015-1078-3
Fairfield, H., Costa, S., Falank, C., Farrell, M., Murphy, C. S., D’Amico, A., Driscoll, H., & Reagan, M. R. (2020). Multiple Myeloma Cells Alter Adipogenesis, Increase Senescence-Related and Inflammatory Gene Transcript Expression, and Alter Metabolism in Preadipocytes. Frontiers in Oncology, 10, 584683. https://doi.org/10.3389/fonc.2020.584683
Mahtouk, K., Moreaux, J., Hose, D., Reme, T., Meissner, T., Jourdan, M., Rossi, J. F., Pals, S. T., Goldschmidt, H., & Klein, B. (2010). Growth factors in multiple myeloma: A comprehensive analysis of their expression in tumor cells and bone marrow environment using Affymetrix microarrays. BMC Cancer, 10(1), 198. https://doi.org/10.1186/1471-2407-10-198
Wallace, S. R., Oken, M. M., Lunetta, K. L., Panoskaltsis-Mortari, A., & Masellis, A. M. (2001). Abnormalities of bone marrow mesenchymal cells in multiple myeloma patients. Cancer, 91(7), 1219–1230. https://doi.org/10.1002/1097-0142(20010401)91:7%3c1219::aid-cncr1122%3e3.0.co;2-1
Arnulf, B., Lecourt, S., Soulier, J., Ternaux, B., Lacassagne, M. N., Crinquette, A., Dessoly, J., Sciaini, A. K., Benbunan, M., Chomienne, C., Fermand, J. P., Marolleau, J. P., & Larghero, J. (2007). Phenotypic and functional characterization of bone marrow mesenchymal stem cells derived from patients with multiple myeloma. Leukemia, 21(1), 158–163. https://doi.org/10.1038/sj.leu.2404466
Tanno, T., Lim, Y., Wang, Q., Chesi, M., Bergsagel, P. L., Matthews, G., Johnstone, R. W., Ghosh, N., Borrello, I., Huff, C. A., & Matsui, W. (2014). Growth differentiating factor 15 enhances the tumor-initiating and self-renewal potential of multiple myeloma cells. Blood, 123(5), 725–733. https://doi.org/10.1182/blood-2013-08-524025
Bataille, R., Jourdan, M., Zhang, X. G., & Klein, B. (1989). Serum levels of interleukin 6, a potent myeloma cell growth factor, as a reflect of disease severity in plasma cell dyscrasias. Journal of Clinical Investigation, 84(6), 2008–2011. https://doi.org/10.1172/JCI114392
Ludwig, H., Nachbaur, D. M., Fritz, E., Krainer, M., & Huber, H. (1991). Interleukin-6 is a prognostic factor in multiple myeloma. Blood, 77(12), 2794–2795. https://doi.org/10.1182/blood.V77.12.2794.2794
Pelliniemi, T. T., Irjala, K., Mattila, K., Pulkki, K., Rajamaki, A., Tienhaara, A., Laakso, M., & Lahtinen, R. (1995). Immunoreactive interleukin-6 and acute phase proteins as prognostic factors in multiple myeloma. Blood, 85(3), 765–771. https://doi.org/10.1182/blood.V85.3.765.bloodjournal853765
Thaler, J., Fechner, F., Herold, M., & Huber, H. (1994). Interleukin-6 in multiple myeloma: Correlation with disease activity and Ki-67 proliferation index. Leukemia & Lymphoma, 12(3–4), 265–271. https://doi.org/10.3109/10428199409059598
Banaszkiewicz, M., Malyszko, J., Batko, K., Koc-Zorawska, E., Zorawski, M., Dumnicka, P., Jurczyszyn, A., Woziwodzka, K., Tisonczyk, J., Krzanowski, M., Malyszko, J., Waszczuk-Gajda, A., Drozdz, R., Kuzniewski, M., & Krzanowska, K. (2020). Evaluating the relationship of GDF-15 with clinical characteristics, cardinal features, and survival in multiple myeloma. Mediators of Inflammation, 5657864. https://doi.org/10.1155/2020/5657864
Alexandrakis, M. G., Passam, F. H., Boula, A., Christophoridou, A., Aloizos, G., Roussou, P., & Kyriakou, D. S. (2003). Relationship between circulating serum soluble interleukin-6 receptor and the angiogenic cytokines basic fibroblast growth factor and vascular endothelial growth factor in multiple myeloma. Annals of Hematology, 82(1), 19–23. https://doi.org/10.1007/s00277-002-0558-0
Corre, J., Labat, E., Espagnolle, N., Hebraud, B., Avet-Loiseau, H., Roussel, M., Huynh, A., Gadelorge, M., Cordelier, P., Klein, B., Moreau, P., Facon, T., Fournie, J. J., Attal, M., & Bourin, P. (2012). Bioactivity and prognostic significance of growth differentiation factor GDF15 secreted by bone marrow mesenchymal stem cells in multiple myeloma. Cancer Research, 72(6), 1395–1406. https://doi.org/10.1158/0008-5472.CAN-11-0188
Tarkun, P., Birtas Atesoglu, E., Mehtap, O., Musul, M. M., & Hacihanefioglu, A. (2014). Serum growth differentiation factor 15 levels in newly diagnosed multiple myeloma patients. Acta Haematologica, 131(3), 173–178. https://doi.org/10.1159/000354835
Mei, S., Wang, H., Fu, R., Qu, W., Xing, L., Wang, G., Song, J., Liu, H., Li, L., Wang, X., Wu, Y., Guan, J., Ruan, E., & Shao, Z. (2014). Hepcidin and GDF15 in anemia of multiple myeloma. International Journal of Hematology, 100(3), 266–273. https://doi.org/10.1007/s12185-014-1626-7
Zingone, A., Wang, W., Corrigan-Cummins, M., Wu, S. P., Plyler, R., Korde, N., Kwok, M., Manasanch, E. E., Tageja, N., Bhutani, M., Mulquin, M., Zuchlinski, D., Yancey, M. A., Roschewski, M., Zhang, Y., Roccaro, A. M., Ghobrial, I. M., Calvo, K. R., & Landgren, O. (2014). Altered cytokine and chemokine profiles in multiple myeloma and its precursor disease. Cytokine, 69(2), 294–297. https://doi.org/10.1016/j.cyto.2014.05.017
Stasi, R., Brunetti, M., Parma, A., Di Giulio, C., Terzoli, E., & Pagano, A. (1998). The prognostic value of soluble interleukin-6 receptor in patients with multiple myeloma. Cancer, 82(10), 1860–1866. https://doi.org/10.1002/(SICI)1097-0142(19980515)82:10%3c1860::AID-CNCR7%3e3.0.CO;2-R
Uchiyama, H., Barut, B. A., Mohrbacher, A. F., Chauhan, D., & Anderson, K. C. (1993). Adhesion of human myeloma-derived cell lines to bone marrow stromal cells stimulates interleukin-6 secretion. Blood, 82(12), 3712–3720. https://doi.org/10.1182/blood.V82.12.3712.3712
Attar-Schneider, O., Zismanov, V., Dabbah, M., Tartakover-Matalon, S., Drucker, L., & Lishner, M. (2016). Multiple myeloma and bone marrow mesenchymal stem cells’ crosstalk: Effect on translation initiation. Molecular Carcinogenesis, 55(9), 1343–1354. https://doi.org/10.1002/mc.22378
Roccaro, A. M., Sacco, A., Maiso, P., Azab, A. K., Tai, Y. T., Reagan, M., Azab, F., Flores, L. M., Campigotto, F., Weller, E., Anderson, K. C., Scadden, D. T., & Ghobrial, I. M. (2013). BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression. Journal of Clinical Investigation, 123(4), 1542–1555. https://doi.org/10.1172/JCI66517
Kanehira, M., Fujiwara, T., Nakajima, S., Okitsu, Y., Onishi, Y., Fukuhara, N., Ichinohasama, R., Okada, Y., & Harigae, H. (2017). An lysophosphatidic acid receptors 1 and 3 axis governs cellular senescence of mesenchymal stromal cells and promotes growth and vascularization of multiple myeloma. Stem Cells, 35(3), 739–753. https://doi.org/10.1002/stem.2499
Urashima, M., Ogata, A., Chauhan, D., Vidriales, M., Teoh, G., Hoshi, Y., Schlossman, R., DeCaprio, J., & Anderson, K. (1996). Interleukin-6 promotes multiple myeloma cell growth via phosphorylation of retinoblastoma protein. Blood, 88(6), 2219–2227. https://doi.org/10.1182/blood.V88.6.2219.bloodjournal8862219
Lokhorst, H. M., Lamme, T., de Smet, M., Klein, S., de Weger, R. A., van Oers, R., & Bloem, A. C. (1994). Primary tumor cells of myeloma patients induce interleukin-6 secretion in long-term bone marrow cultures. Blood, 84(7), 2269–2277. https://doi.org/10.1182/blood.V84.7.2269.2269
Klein, B., Zhang, X. G., Jourdan, M., Content, J., Houssiau, F., Aarden, L., Piechaczyk, M., & Bataille, R. (1989). Paracrine rather than autocrine regulation of myeloma-cell growth and differentiation by interleukin-6. Blood, 73(2), 517–526. https://doi.org/10.1182/blood.V73.2.517.517
Zhang, X. G., Klein, B., & Bataille, R. (1989). Interleukin-6 is a potent myeloma-cell growth factor in patients with aggressive multiple myeloma. Blood, 74(1), 11–13. https://doi.org/10.1182/blood.V74.1.11.11
Fulciniti, M., Hideshima, T., Vermot-Desroches, C., Pozzi, S., Nanjappa, P., Shen, Z., Patel, N., Smith, E. S., Wang, W., Prabhala, R., Tai, Y. T., Tassone, P., Anderson, K. C., & Munshi, N. C. (2009). A high-affinity fully human anti-IL-6 mAb, 1339, for the treatment of multiple myeloma. Clinical Cancer Research, 15(23), 7144–7152. https://doi.org/10.1158/1078-0432.CCR-09-1483
Teoh, H. K., Chong, P. P., Abdullah, M., Sekawi, Z., Tan, G. C., Leong, C. F., & Cheong, S. K. (2016). Small interfering RNA silencing of interleukin-6 in mesenchymal stromal cells inhibits multiple myeloma cell growth. Leukemia Research, 40, 44–53. https://doi.org/10.1016/j.leukres.2015.10.004
Rossi, J. F., Fegueux, N., Lu, Z. Y., Legouffe, E., Exbrayat, C., Bozonnat, M. C., Navarro, R., Lopez, E., Quittet, P., Daures, J. P., Rouille, V., Kanouni, T., Widjenes, J., & Klein, B. (2005). Optimizing the use of anti-interleukin-6 monoclonal antibody with dexamethasone and 140 mg/m2 of melphalan in multiple myeloma: Results of a pilot study including biological aspects. Bone Marrow Transplantation, 36(9), 771–779. https://doi.org/10.1038/sj.bmt.1705138
Moreau, P., Harousseau, J. L., Wijdenes, J., Morineau, N., Milpied, N., & Bataille, R. (2000). A combination of anti-interleukin 6 murine monoclonal antibody with dexamethasone and high-dose melphalan induces high complete response rates in advanced multiple myeloma. British Journal of Haematology, 109(3), 661–664. https://doi.org/10.1046/j.1365-2141.2000.02093.x
Voorhees, P. M., Manges, R. F., Sonneveld, P., Jagannath, S., Somlo, G., Krishnan, A., Lentzsch, S., Frank, R. C., Zweegman, S., Wijermans, P. W., Orlowski, R. Z., Kranenburg, B., Hall, B., Casneuf, T., Qin, X., van de Velde, H., Xie, H., & Thomas, S. K. (2013). A phase 2 multicentre study of siltuximab, an anti-interleukin-6 monoclonal antibody, in patients with relapsed or refractory multiple myeloma. British Journal of Haematology, 161(3), 357–366. https://doi.org/10.1111/bjh.12266
Brighton, T. A., Khot, A., Harrison, S. J., Ghez, D., Weiss, B. M., Kirsch, A., Magen, H., Gironella, M., Oriol, A., Streetly, M., Kranenburg, B., Qin, X., Bandekar, R., Hu, P., Guilfoyle, M., Qi, M., Nemat, S., & Goldschmidt, H. (2019). Randomized, double-blind, placebo-controlled, multicenter study of siltuximab in high-risk smoldering multiple myeloma. Clinical Cancer Research, 25(13), 3772–3775. https://doi.org/10.1158/1078-0432.CCR-18-3470
Jourdan, M., Tarte, K., Legouffe, E., Brochier, J., Rossi, J. F., & Klein, B. (1999). Tumor necrosis factor is a survival and proliferation factor for human myeloma cells. European Cytokine Network, 10(1), 65–70.
Bosseboeuf, A., Allain-Maillet, S., Mennesson, N., Tallet, A., Rossi, C., Garderet, L., Caillot, D., Moreau, P., Piver, E., Girodon, F., Perreault, H., Brouard, S., Nicot, A., Bigot-Corbel, E., Hermouet, S., & Harb, J. (2017). Pro-inflammatory state in monoclonal gammopathy of undetermined significance and in multiple myeloma is characterized by low sialylation of pathogen-specific and other monoclonal immunoglobulins. Frontiers in Immunology, 8, 1347. https://doi.org/10.3389/fimmu.2017.01347
Tsirakis, G., Pappa, C. A., Spanoudakis, M., Chochlakis, D., Alegakis, A., Psarakis, F. E., Stratinaki, M., Stathopoulos, E. N., & Alexandrakis, M. G. (2012). Clinical significance of sCD105 in angiogenesis and disease activity in multiple myeloma. European Journal of Internal Medicine, 23(4), 368–373. https://doi.org/10.1016/j.ejim.2012.01.012
Schneiderova, P., Pika, T., Gajdos, P., Fillerova, R., Kromer, P., Kudelka, M., Minarik, J., Papajik, T., Scudla, V., & Kriegova, E. (2017). Serum protein fingerprinting by PEA immunoassay coupled with a pattern-recognition algorithms distinguishes MGUS and multiple myeloma. Oncotarget, 8(41), 69408–69421. https://doi.org/10.18632/oncotarget.11242
Merico, F., Bergui, L., Gregoretti, M. G., Ghia, P., Aimo, G., Lindley, I. J., & Caligaris-Cappio, F. (1993). Cytokines involved in the progression of multiple myeloma. Clinical Experimental Immunology, 92(1), 27–31. https://doi.org/10.1111/j.1365-2249.1993.tb05943.x
Tarantini, S., Balasubramanian, P., Delfavero, J., Csipo, T., Yabluchanskiy, A., Kiss, T., Nyul-Toth, A., Mukli, P., Toth, P., Ahire, C., Ungvari, A., Benyo, Z., Csiszar, A., & Ungvari, Z. (2021). Treatment with the BCL-2/BCL-xL inhibitor senolytic drug ABT263/Navitoclax improves functional hyperemia in aged mice. Geroscience, 43(5), 2427–2440. https://doi.org/10.1007/s11357-021-00440-z
Pan, J., Li, D., Xu, Y., Zhang, J., Wang, Y., Chen, M., Lin, S., Huang, L., Chung, E. J., Citrin, D. E., Wang, Y., Hauer-Jensen, M., Zhou, D., & Meng, A. (2017). Inhibition of Bcl-2/xl with ABT-263 selectively kills senescent type II pneumocytes and reverses persistent pulmonary fibrosis induced by ionizing radiation in mice. International Journal of Radiation Oncology Biology Physics, 99(2), 353–361. https://doi.org/10.1016/j.ijrobp.2017.02.216
Chang, J., Wang, Y., Shao, L., Laberge, R.-M., Demaria, M., Campisi, J., Janakiraman, K., Sharpless, N. E., Ding, S., Feng, W., Luo, Y., Wang, X., Aykin-Burns, N., Krager, K., Ponnappan, U., Hauer-Jensen, M., Meng, A., & Zhou, D. (2016). Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nature Medicine, 22(1), 78–83. https://doi.org/10.1038/nm.4010
Kim, H. N., Chang, J., Shao, L., Han, L., Iyer, S., Manolagas, S. C., O’Brien, C. A., Jilka, R. L., Zhou, D., & Almeida, M. (2017). DNA damage and senescence in osteoprogenitors expressing Osx1 may cause their decrease with age. Aging Cell, 16(4), 693–703. https://doi.org/10.1111/acel.12597
Sharma, A. K., Roberts, R. L., Benson, R. D., Jr., Pierce, J. L., Yu, K., Hamrick, M. W., & McGee-Lawrence, M. E. (2020). The senolytic drug navitoclax (ABT-263) causes trabecular bone loss and impaired osteoprogenitor function in aged mice. Frontiers in Cell and Developmental Biology, 8(354). https://doi.org/10.3389/fcell.2020.00354
Grezella, C., Fernandez-Rebollo, E., Franzen, J., Ventura Ferreira, M. S., Beier, F., & Wagner, W. (2018). Effects of senolytic drugs on human mesenchymal stromal cells. Stem Cell Research & Therapy, 9(1), 108. https://doi.org/10.1186/s13287-018-0857-6
Song, X., Dai, J., Li, H., Li, Y., Hao, W., Zhang, Y., Zhang, Y., Su, L., & Wei, H. (2019). Anti-aging effects exerted by Tetramethylpyrazine enhances self-renewal and neuronal differentiation of rat bMSCs by suppressing NF-kB signaling. Bioscience Reports, 39(6). https://doi.org/10.1042/BSR20190761
Gao, B., Lin, X., Jing, H., Fan, J., Ji, C., Jie, Q., Zheng, C., Wang, D., Xu, X., Hu, Y., Lu, W., Luo, Z., & Yang, L. (2018). Local delivery of tetramethylpyrazine eliminates the senescent phenotype of bone marrow mesenchymal stromal cells and creates an anti-inflammatory and angiogenic environment in aging mice. Aging Cell, 17(3), e12741. https://doi.org/10.1111/acel.12741
Zhu, Y., Tchkonia, T., Pirtskhalava, T., Gower, A. C., Ding, H., Giorgadze, N., Palmer, A. K., Ikeno, Y., Hubbard, G. B., Lenburg, M., O’Hara, S. P., LaRusso, N. F., Miller, J. D., Roos, C. M., Verzosa, G. C., LeBrasseur, N. K., Wren, J. D., Farr, J. N., Khosla, S., Stout, M. B., et al. (2015). The Achilles’ heel of senescent cells: From transcriptome to senolytic drugs. Aging Cell, 14(4), 644–658. https://doi.org/10.1111/acel.12344
Farr, J. N., Xu, M., Weivoda, M. M., Monroe, D. G., Fraser, D. G., Onken, J. L., Negley, B. A., Sfeir, J. G., Ogrodnik, M. B., Hachfeld, C. M., LeBrasseur, N. K., Drake, M. T., Pignolo, R. J., Pirtskhalava, T., Tchkonia, T., Oursler, M. J., Kirkland, J. L., & Khosla, S. (2017). Targeting cellular senescence prevents age-related bone loss in mice. Nature Medicine, 23(9), 1072–1079. https://doi.org/10.1038/nm.4385
Hickson, L. J., Langhi Prata, L. G. P., Bobart, S. A., Evans, T. K., Giorgadze, N., Hashmi, S. K., Herrmann, S. M., Jensen, M. D., Jia, Q., Jordan, K. L., Kellogg, T. A., Khosla, S., Koerber, D. M., Lagnado, A. B., Lawson, D. K., LeBrasseur, N. K., Lerman, L. O., McDonald, K. M., McKenzie, T. J., Passos, J. F., et al. (2019). Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of dasatinib plus quercetin in individuals with diabetic kidney disease. eBioMedicine, 47, 446–456. https://doi.org/10.1016/j.ebiom.2019.08.069
Justice, J. N., Nambiar, A. M., Tchkonia, T., LeBrasseur, N. K., Pascual, R., Hashmi, S. K., Prata, L., Masternak, M. M., Kritchevsky, S. B., Musi, N., & Kirkland, J. L. (2019). Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human, open-label, pilot study. eBioMedicine, 40, 554–563. https://doi.org/10.1016/j.ebiom.2018.12.052
Thaler, J., Fechner, F., Herold, M., & Huber, H. (1994). Interleukin-6 in multiple myeloma: Correlation with disease activity and Ki-67 proliferation index. Leukaemia & Lymphoma, 12(3–4), 265–271. https://doi.org/10.3109/10428199409059598
Banaszkiewicz, M., Malyszko, J., Batko, K., Koc-Zorawska, E., Zorawski, M., Dumnicka, P., Jurczyszyn, A., Woziwodzka, K., Tisonczyk, J., Krzanowski, M., Malyszko, J., Waszczuk-Gajda, A., Drozdz, R., Kuzniewski, M., & Krzanowska, K. (2020). Evaluating the relationship of GDF-15 with clinical characteristics, cardinal features, and survival in multiple myeloma. Mediators of Inflammation, 2020, 5657864. https://doi.org/10.1155/2020/5657864
Bonilla, X., Vanegas, N. P., & Vernot, J. P. (2019). Acute leukemia induces senescence and impaired osteogenic differentiation in mesenchymal stem cells endowing leukemic cells with functional advantages. Stem Cells International, 2019, 3864948. https://doi.org/10.1155/2019/3864948
Acknowledgements
K.V. and K.M.M. are supported by Early Career Cancer Research Fellowships from the Cancer Council SA Beat Cancer Project on behalf of its donors and the State Government of South Australia through the Department of Health. V.P. is supported by a National Health and Medical Research Council Early Career Fellowship. This research was supported in part by a research grant from the Ray and Shirl Norman Cancer Research Trust and by grant 2013025 awarded through the 2021 Priority-driven Collaborative Cancer Research Scheme and co-funded by Cancer Australia and the Leukaemia Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Plakhova, N., Panagopoulos, V., Vandyke, K. et al. Mesenchymal stromal cell senescence in haematological malignancies. Cancer Metastasis Rev 42, 277–296 (2023). https://doi.org/10.1007/s10555-022-10069-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-022-10069-9