Abstract
Transesophageal echocardiography (TEE) is the gold standard imaging study used in the diagnosis of infective endocarditis (IE). Computed tomography angiography (CTA) has undergone rapid advancement as a cardiac imaging technique and has previously shown promise in small non-randomized studies for evaluation of IE. We hypothesized that cardiac CTA would perform similarly to TEE in the detection of endocarditic lesions and that there would be no difference in clinical outcomes whether the coronary arteries were evaluated by CTA or invasive coronary angiography (ICA). 255 adults who underwent surgery for IE at the Mayo Clinic Rochester between January 1, 2006 and June 1, 2014 were identified retrospectively. 251 patients underwent TEE and 34 patients underwent cardiac CTA. TEE had statistically higher detection of vegetations (95.6 vs. 70.0%, p < 0.0001) and leaflet perforations (81.3 vs. 42.9%, p = 0.02) as compared to cardiac CTA. For detection of abscess/pseudoaneurysm TEE had a similar sensitivity to cardiac CTA (90.5 vs. 78.4%, p = 0.21). There was no significant difference in peri-operative outcomes whether coronary arteries were evaluated by CTA or ICA. The greatest advantage of cardiac CT in the setting of IE is its ability to couple the detection of complex cardiac anatomic abnormalities with coronary artery delineation, serving two important components of the diagnostic evaluation, particularly among patients who will require surgical intervention due to IE complications. Cardiac CTA may be considered as an alternate coronary artery imaging modality in IE patients with low to intermediate risk of disease but meet guideline recommendations for coronary artery imaging.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Transesophageal echocardiography (TEE) is the gold standard diagnostic tool for identifying structural abnormalities in infective endocarditis (IE) [1,2,3]. Classic features of IE on TEE include vegetations, ring abscess, pseudoaneurysm, fistula, leaflet perforation, and valvular dehiscence [4]. In recent years, rapid advancement in computed tomography (CT) scanning has led to markedly improved utility in cardiac and coronary artery imaging since it was first shown to be of use in quantifying coronary calcium over 25 years ago [5, 6]. Prior to surgery for IE, a proportion of patients will undergo invasive coronary angiography (ICA) to determine if there is a need for intra-operative coronary artery bypass grafting (CABG). Current indications for coronary assessment include: men > 40 years old, post-menopausal women, and those with a history of coronary artery disease (CAD) or multiple risk factors for CAD [7, 8].
Although there have been improvements in the safety of angiography, it remains an invasive procedure with associated risks, particularly in critically ill patients with IE who require urgent surgery [9]. Of particular concern in patients with IE, are reports of fatal coronary embolization of mobile vegetative material that occurred during ICA [10]. Cardiac CT angiography (CTA) has been validated previously as a non-invasive option to evaluate for CAD [11]. Prospective clinical trials as well as meta-analyses have repeatedly demonstrated high sensitivity and negative predictive values (NPV) of cardiac CTA for excluding coronary stenoses > 50% in intermediate and low-risk patients [11,12,13,14,15,16,17].
Therefore, the current investigation sought to compare the diagnostic accuracy of cardiac CTA with TEE for detecting imaging features of IE and compare perioperative outcomes of patients who underwent cardiac CTA vs. ICA for assessment of coronary anatomy prior to surgery for IE.
Methods
A retrospective cohort of 255 adult (18 and older) consecutive patients who underwent cardiac surgery for IE between January 1, 2006 and June 1, 2014 at Mayo Clinic Rochester was identified.
Cardiac CT scanners over the study period ranged from 64 to 128 slice dual-source machines. All scans were ECG-gated and 3D post-processing was performed. Multiphase datasets were obtained in the majority of cases. Beta-blocker use was part of the cardiac CT protocol but was excluded if the patient had a contraindication to their use. All reading physicians had received advanced training in cardiac CT imaging (SCCT Level 3 certified). All echocardiographers were COCATS level III trained. Echo machines were GE, Phillips, or Acuson devices.
The comprehensive TEE closest to the time of cardiac surgery was used for comparison. Cardiac CT data was included only if both coronary artery and IE findings were assessed on the same study (i.e. if a CTA was ordered solely for coronary assessment and IE findings were not assessed, the study was excluded). The IE features of interest on echocardiogram included vegetations, abscess/pseudoaneurysm, fistula, leaflet perforation, and valvular dehiscence. Surgical observations were established by review of operative and pathology reports. Findings were reported on a per-patient basis.
For patients who underwent cardiac CTA or ICA, coronary arteries were evaluated for the number of vessels with > 50% stenosis. ICA was further evaluated to determine if it changed management. A change in management was defined as the decision to pursue revascularization at the time of cardiac surgery regardless of whether bypass was aborted at the time of procedure for technical reasons. For evaluation of acute kidney injury (AKI), the pre-angiogram serum creatinine level was compared to a 3 day post-angiogram creatinine level. An increase of greater than or equal to 0.3 mg/dL was defined as AKI. Patients who underwent surgery during this time were excluded from this analysis. Additionally, patients on dialysis were excluded from this analysis.
For statistical analysis, the diagnostic accuracy of cardiac CTA for IE as compared with operative findings was assessed. The same analysis was performed for TEE to serve as a reference in this patient population. Second, clinical outcomes were assessed for patients undergoing cardiac CTA as compared with ICA. Third, coronary artery disease findings were compared between patients undergoing cardiac CTA compared with ICA. Sensitivity and specificity analyses were performed. Population proportions were compared using the Pearson Chi square test. A value of p < 0.05 was considered significant for all tests (These values have been made bold in the tables). All analyses were performed using JMP (SAS Institute, Cary, NC, United States). This study was approved by the Mayo Clinic Institutional Review Board.
Results
Of the 255 patients with IE undergoing surgery, 34 (13%) had cardiac CTA pre-operatively and 103 (40%) underwent ICA. Baseline characteristics for these groups were similar except for a statistically significant younger age (54.0 vs. 64.1 years, p < 0.001) and larger male predominance (91.2% CTA vs. 74.8% ICA, p = 0.04) in the cardiac CTA group as shown in Table 1.
There were no significant differences among cardiac risk factors. The CTA group had a larger proportion of aortic valve involvement than the ICA group (82.4 vs. 58.3%, p = 0.01). Fig. 1 depicts the number of patients undergoing each method of preoperative coronary evaluation. The group of patients that did not have documented preoperative coronary evaluation was heterogeneous and included 4 main clinical scenarios: (1) young patients with no risk factors; (2) urgent or emergent timing of surgery; (3) recent coronary evaluation performed for an unrelated reason; (4) coronary evaluation was performed at outside facility but textual report was not available in the medical record. Of note, 7 patients proceeded to ICA after CTA. Additionally, 5 patients had contraindications to ICA and 23 patients did not undergo coronary evaluation for unclear reasons.
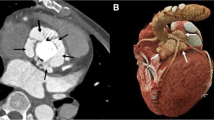
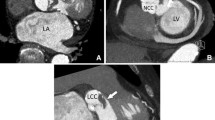
Among 34 patients who underwent pre-operative cardiac CTA, the median number of days from CT to surgery was 4 and 47.1% had prosthetic valves. Sensitivity and specificity for detection of vegetations were 70 and 92.9%, respectively (Table 2). However, for detection of abscess/pseudoaneurysm, these were 90.5 and 92.3%, respectively. Sensitivity for detection of fistula, leaflet perforation, or dehiscence ranged from 40 to 50%. Specificity of cardiac CTA was high (> 90%) for all endocarditic findings. Figure 2 demonstrates a perivalvular abscess identified by cardiac CTA. Figure 3 compares the CTA and TEE imaging of a patient with peri-prosthetic abscess/pseudoaneurysm. Figure 4 depicts a pseudoaneurysm of the intervalvular fibrosa and an aortic valve vegetation imaged by both CTA and TEE.
Perivalvular abscess/pseudoaneurysm imaged by cardiac CTA and TEE. a Axial cut at the level of the aortic valve. b Sagittal cut demonstrating a long-axis view of the left ventricle. c Right ventricular outflow view via TEE. d Left ventricular long-axis view via TEE. Ao aorta, AV aortic valve, LA left atrium, LV left ventricle, PSA pseudoaneurysm, RA right atrium, RVOT right ventricular outflow tract
Pseudoaneurysm and associated aortic valve vegetation imaged by cardiac CTA and TEE. a, b CTA images in oblique and sagittal sections. c, d TEE images of the left ventricular outflow tract, intervalvular fibrosa, and aortic valve with and without color flow doppler. Ao aorta, Comm communication, LA left atrium, LV left ventricle, PSA pseudoaneurysm, Veg vegetation
Of the 255 patients included, 251 underwent pre-operative TEE and 42.9% had prosthetic valves. The median number of days from TEE to surgery was 1. Sensitivity and specificity for detection of vegetations were 95.6 and 93.0%, respectively. For the detection of abscess/pseudoaneurysm, these were 78.4 and 98.8%, respectively. For the other endocarditic findings, sensitivities ranged from 70 to 80%. Specificity of TEE for detection of fistula, leaflet perforation, and dehiscence were all high (> 90%).
When comparing the performance of cardiac CTA and TEE, statistically significant differences were demonstrated in the sensitivity for detection of vegetations and leaflet perforations with TEE performing superiorly. The sensitivity for detection of abscess/pseudoaneurysm, fistula, and dehiscence did not differ significantly between the two groups. The findings remained unchanged when an analysis was limited to the subgroup of patients with prosthetic valve IE (Data not shown).
Table 3 compares the CAD findings between patients undergoing CTA and ICA. The proportion of patients with less than 50% coronary stenosis in the cardiac CTA group was 70.6% compared with 60.2% in the ICA group (p = 0.28). Of all the patients who underwent ICA, including the 7 previously evaluated by CTA, the proportion whose management was changed by the study was 17.5% (10.0–24.9%, 95% CI). Patients undergoing cardiac CTA were less likely to have an elevated serum creatinine (> 1.5 mg/dL) at baseline (2.9 vs. 18.4%, p = 0.03). The mean difference in serum creatinine after contrast exposure was 0.03 mg/dL in the CT group and − 0.02 mg/dL in the catheterization group (p = 0.30). The percent of patients with a serum creatinine increase > 0.3 mg/dL after contrast exposure was 3.5 and 3.3% respectively (p = 0.97). There was one adverse event documented in the ICA group that was an allergic reaction attributed to iodinated contrast. There were no adverse events attributed to cardiac CTA.
Post-operative clinical outcomes in patients undergoing cardiac CTA vs. ICA are listed in Table 3. The rate of intra-operative bypass grafting for coronary disease was 14.7% in the cardiac CTA group (n = 5) and 18.4% in ICA group with no statistically significant difference. Of the 5 patients in the CTA group who underwent intra-operative CABG, 4 were also evaluated with ICA pre-operatively. The remaining patient underwent CABG due to anatomical complications of IE rather than due to obstructive CAD. The 1-year mortality rate post-operatively was low at 5.9 and 11.7% (p = 0.34) for the cardiac CTA and ICA groups, respectively. There was no significant difference in peri-operative stroke (n = 0 and n = 3, respectively) or mortality (n = 1 and n = 3, respectively) between the CTA and ICA groups. No peri-operative myocardial infarctions were noted in either group. Patients without a preoperative coronary evaluation demonstrated increased peri-operative and 1 year all-cause mortality rates of 10.4 and 20.0%, respectively. This difference in peri-operative mortality was statistically significant compared with ICA (p = 0.03) but not with CTA (p = 0.17). When peri-operative mortality was excluded from the 1-year mortality analysis, there were no longer any statistically significant differences between the CT group, the ICA group, and the no index evaluation group (3.0 vs. 9.0 vs. 10.7%, respectively; p = 0.42).
Discussion
The current study is the largest to date comparing cardiac CTA to TEE in IE patients. Cardiac CTA was similar to TEE in identification of abscess/pseudoaneurysm in the current investigation, while, as expected, TEE demonstrated significantly higher sensitivity for vegetations and leaflet perforation. Of note, the specificity of both tests for all IE lesions was > 90%. This is despite almost one-half of patients having prosthetic valves with recent evidence demonstrating that CTA has the ability to change diagnosis/treatment with echocardiography in 20–25% of prosthetic IE cases [18]. In patients with difficult to characterize perivalvular extension of infection by TEE, cardiac CTA is a reasonable next diagnostic step with the ability to define the coronary arterial anatomy concomitantly, thus avoiding an additional procedure (ICA) and contrast exposure.
With regard to preoperative coronary evaluation, both groups had similar cardiac risk factors. Cardiac CTA has excellent negative predictive value in low to intermediate CAD risk patients. The evidence has been less robust in high risk patients with some studies demonstrating a continued high NPV while others maintaining a high sensitivity, but a decline in NPV due to the increased prevalence of disease [19,20,21,22,23]. Among patients undergoing ICA, 60% had no coronary stenosis > 50% and over 80% had no change in management. This implies that there are opportunities to increase the utilization of cardiac CTA and decrease the proportion of patients requiring an invasive procedure prior to cardiac surgery for IE. A cost effectiveness study incorporating CTA as a first line test in pre-operative coronary evaluation for non-cardiac surgery demonstrated a decreased cost associated with the work-up and perioperative period [24]. Therefore, in addition to avoiding an invasive procedure with a low risk for adverse events, this strategy may be fiscally prudent as well.
The low rate of cardiac CTA utilization in the current study population is noteworthy. While this fluctuated from year-to-year, there was no clear trend towards increased utilization from 2007 to 2014, even after the 2009 prospective study by Feuchtner et al. suggested its utility [25]. Potential factors that account for this low rate of utilization are likely uncertainty about the clinical value of cardiac CTA given the small studies to date, a long history of preoperative evaluation of coronaries by cardiac catheterization, and an understandable desire to avoid a double contrast load if a study is positive or non-diagnostic, particularly in severely ill patients with renal dysfunction.
Figure 5 displays an algorithm adapted from the 2014 AHA/ACC guideline for valvular heart disease by Nishimura et al. and our proposal for the approach to preoperative coronary assessment for patients with IE [26]. Unless patients have symptoms of active ischemic coronary disease or are at high pre-test probability, then performing a cardiac CTA may be reasonable. This will reliably exclude significant CAD, potentially avoid ICA with its associated costs/potential complications, and may assist with surgical planning.
There are several studies published to date to support this strategy of pre-operative CTA in non-cardiac as well as cardiac surgery [27]. Jakamy et al. evaluated 48 patients who underwent elective valve surgery and found that CTA had an NPV of 91% and ICA could have been avoided in 65% of patients [28]. Ciolina et al. demonstrated the additive value of cardiac CTA for assessing the aortic valve in pre-operative work-up for aortic stenosis [29]. In 42 patients, CTA correctly graded aortic valve calcification, sized the aortic annulus and sinotubular junction, detected thoracic aortic aneurysms, and correctly assessed aortic valve area over 90% of the time while also being used to assess coronary status. The ability of cardiac CTA to simultaneously provide additive diagnostic information and reliably assess coronary status makes it a versatile diagnostic test with significant yield in the pre-operative work-up of patients with IE and other valvular disorders.
Our study has limitations that deserve address. The study of IE has been hindered for decades by its relatively uncommon nature and the vast heterogeneity in microbiology, clinical presentation, disease manifestations, and patient factors. This has historically resulted in small study populations and a general lack of prospective randomized studies (Online Resource 1). Therefore, while the current investigation is limited by its size and retrospective nature, this is in line with how IE has historically been studied. Next, by identifying patients who underwent surgery for IE, we excluded patients who may have had a rarely reported fatal coronary embolic event during ICA or other severe complications including AKI resulting in dialysis dependence. Therefore, our study did not include identification of significant adverse events at the time of cardiac catheterization. Finally, the rates of perioperative mortality, stroke, and MI were low for both groups, accounting for a reduced statistical power for detecting differences.
Conclusion
The greatest advantage of cardiac CT in the setting of IE is its ability to couple the detection of complex cardiac anatomic abnormalities with coronary artery delineation. Cardiac CTA may be considered as an alternate coronary artery imaging modality in IE patients with low to intermediate risk of disease but meet guideline recommendations for coronary artery imaging.
References
Baddour LM, Wilson WR, Bayer AS, Fowler VG Jr, Tleyjeh IM, Rybak MJ, Barsic B, Lockhart PB, Gewitz MH, Levison ME, Bolger AF, Steckelberg JM, Baltimore RS, Fink AM, O’Gara P, Taubert KA (2015) Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation 132(15):1435–1486. https://doi.org/10.1161/cir.0000000000000296
Baddour LM, Wilson WR, Bayer AS, Fowler VG Jr, Bolger AF, Levison ME, Ferrieri P, Gerber MA, Tani LY, Gewitz MH, Tong DC, Steckelberg JM, Baltimore RS, Shulman ST, Burns JC, Falace DA, Newburger JW, Pallasch TJ, Takahashi M, Taubert KA (2005) Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America. Circulation 111(23):e394-434. https://doi.org/10.1161/circulationaha.105.165564
Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta JP, Del Zotti F, Dulgheru R, El Khoury G, Erba PA, Iung B, Miro JM, Mulder BJ, Plonska-Gosciniak E, Price S, Roos-Hesselink J, Snygg-Martin U, Thuny F, Tornos Mas P, Vilacosta I, Zamorano JL, Erol C, Nihoyannopoulos P, Aboyans V, Agewall S, Athanassopoulos G, Aytekin S, Benzer W, Bueno H, Broekhuizen L, Carerj S, Cosyns B, De Backer J, De Bonis M, Dimopoulos K, Donal E, Drexel H, Flachskampf FA, Hall R, Halvorsen S, Hoen B, Kirchhof P, Lainscak M, Leite-Moreira AF, Lip GY, Mestres CA, Piepoli MF, Punjabi PP, Rapezzi C, Rosenhek R, Siebens K, Tamargo J, Walker DM (2015) 2015 ESC Guidelines for the management of infective endocarditis: the task force for the management of infective endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J 36(44):3075–3128. https://doi.org/10.1093/eurheartj/ehv319
Entrikin DW, Gupta P, Kon ND, Carr JJ (2012) Imaging of infective endocarditis with cardiac CT angiography. J Cardiovasc Comput Tomogr 6(6):399–405. https://doi.org/10.1016/j.jcct.2012.10.001
Sun Z, Ng KH (2010) Multislice CT angiography in cardiac imaging. Part II: clinical applications in coronary artery disease. Singap Med J 51(4):282–289
Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M Jr, Detrano R (1990) Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 15(4):827–832
Prendergast BD, Tornos P (2010) Surgery for infective endocarditis: who and when? Circulation 121(9):1141–1152. https://doi.org/10.1161/CIRCULATIONAHA.108.773598
Byrne JG, Rezai K, Sanchez JA, Bernstein RA, Okum E, Leacche M, Balaguer JM, Prabhakaran S, Bridges CR, Higgins RS (2011) Surgical management of endocarditis: the society of thoracic surgeons clinical practice guideline. Ann Thorac Surg 91(6):2012–2019. https://doi.org/10.1016/j.athoracsur.2011.01.106
Bashore TM, Balter S, Barac A, Byrne JG, Cavendish JJ, Chambers CE, Hermiller JB Jr, Kinlay S, Landzberg JS, Laskey WK, McKay CR, Miller JM, Moliterno DJ, Moore JW, Oliver-McNeil SM, Popma JJ, Tommaso CL (2012) 2012 American College of Cardiology Foundation/Society for Cardiovascular Angiography and Interventions expert consensus document on cardiac catheterization laboratory standards update: a report of the American College of Cardiology Foundation Task Force on Expert Consensus documents developed in collaboration with the Society of Thoracic Surgeons and Society for Vascular Medicine. J Am Coll Cardiol 59(24):2221–2305. https://doi.org/10.1016/j.jacc.2012.02.010
Shamsham F, Safi AM, Pomerenko I, Salciccioli L, Feit A, Clark LT, Alam M (2000) Fatal left main coronary artery embolism from aortic valve endocarditis following cardiac catheterization. Catheter Cardiovasc Interv 50(1):74–77
Budoff MJ, Dowe D, Jollis JG, Gitter M, Sutherland J, Halamert E, Scherer M, Bellinger R, Martin A, Benton R, Delago A, Min JK (2008) Diagnostic performance of 64-multidetector row coronary computed tomographic angiography for evaluation of coronary artery stenosis in individuals without known coronary artery disease: results from the prospective multicenter ACCURACY (Assessment by Coronary Computed Tomographic Angiography of Individuals Undergoing Invasive Coronary Angiography) trial. J Am Coll Cardiol 52(21):1724–1732. https://doi.org/10.1016/j.jacc.2008.07.031
Abdulla J, Abildstrom SZ, Gotzsche O, Christensen E, Kober L, Torp-Pedersen C (2007) 64-multislice detector computed tomography coronary angiography as potential alternative to conventional coronary angiography: a systematic review and meta-analysis. Eur Heart J 28(24):3042–3050. https://doi.org/10.1093/eurheartj/ehm466
Hamon M, Biondi-Zoccai GG, Malagutti P, Agostoni P, Morello R, Valgimigli M (2006) Diagnostic performance of multislice spiral computed tomography of coronary arteries as compared with conventional invasive coronary angiography: a meta-analysis. J Am Coll Cardiol 48(9):1896–1910. https://doi.org/10.1016/j.jacc.2006.08.028
Miller JM, Rochitte CE, Dewey M, Arbab-Zadeh A, Niinuma H, Gottlieb I, Paul N, Clouse ME, Shapiro EP, Hoe J, Lardo AC, Bush DE, de Roos A, Cox C, Brinker J, Lima JA (2008) Diagnostic performance of coronary angiography by 64-row CT. N Engl J Med 359(22):2324–2336. https://doi.org/10.1056/NEJMoa0806576
Nieman K, Cademartiri F, Lemos PA, Raaijmakers R, Pattynama PM, de Feyter PJ (2002) Reliable noninvasive coronary angiography with fast submillimeter multislice spiral computed tomography. Circulation 106(16):2051–2054
Sun Z, Almutairi AM (2010) Diagnostic accuracy of 64 multislice CT angiography in the assessment of coronary in-stent restenosis: a meta-analysis. Eur J Radiol 73(2):266–273. https://doi.org/10.1016/j.ejrad.2008.10.025
Sun Z, Lin C, Davidson R, Dong C, Liao Y (2008) Diagnostic value of 64-slice CT angiography in coronary artery disease: a systematic review. Eur J Radiol 67(1):78–84. https://doi.org/10.1016/j.ejrad.2007.07.014
Habets J, Tanis W, van Herwerden LA, van den Brink RB, Mali WP, de Mol BA, Chamuleau SA, Budde RP (2014) Cardiac computed tomography angiography results in diagnostic and therapeutic change in prosthetic heart valve endocarditis. Int J Cardiovasc Imaging 30(2):377–387. https://doi.org/10.1007/s10554-013-0335-2
Arbab-Zadeh A, Miller JM, Rochitte CE, Dewey M, Niinuma H, Gottlieb I, Paul N, Clouse ME, Shapiro EP, Hoe J, Lardo AC, Bush DE, de Roos A, Cox C, Brinker J, Lima JA (2012) Diagnostic accuracy of computed tomography coronary angiography according to pre-test probability of coronary artery disease and severity of coronary arterial calcification. The CORE-64 (Coronary Artery Evaluation Using 64-Row Multidetector Computed Tomography Angiography) International Multicenter Study. J Am Coll Cardiol 59(4):379–387. https://doi.org/10.1016/j.jacc.2011.06.079
Sara L, Rochitte CE, Lemos PA, Niinuma H, Dewey M, Shapiro EP, Gottlieb I, Mansur AP, Nicolau JC, Lardo AC, Azevedo CF, Kalil-Filho R, Vavere AL, Cohn S, Cox C, Brinker J, Miller JM, Lima JA (2014) Accuracy of multidetector computed tomography for detection of coronary artery stenosis in acute coronary syndrome compared with stable coronary disease: a CORE64 multicenter trial substudy. Int J Cardiol 177(2):385–391. https://doi.org/10.1016/j.ijcard.2014.08.130
Meijboom WB, Meijs MF, Schuijf JD, Cramer MJ, Mollet NR, van Mieghem CA, Nieman K, van Werkhoven JM, Pundziute G, Weustink AC, de Vos AM, Pugliese F, Rensing B, Jukema JW, Bax JJ, Prokop M, Doevendans PA, Hunink MG, Krestin GP, de Feyter PJ (2008) Diagnostic accuracy of 64-slice computed tomography coronary angiography: a prospective, multicenter, multivendor study. J Am Coll Cardiol 52(25):2135–2144. https://doi.org/10.1016/j.jacc.2008.08.058
Malagutti P, Nieman K, Meijboom WB, van Mieghem CA, Pugliese F, Cademartiri F, Mollet NR, Boersma E, de Jaegere PP, de Feyter PJ (2007) Use of 64-slice CT in symptomatic patients after coronary bypass surgery: evaluation of grafts and coronary arteries. Eur Heart J 28(15):1879–1885. https://doi.org/10.1093/eurheartj/ehl155
Cademartiri F, Maffei E, Palumbo A, Seitun S, Martini C, Tedeschi C, La Grutta L, Midiri M, Weustink AC, Mollet NR, Krestin GP (2010) Coronary calcium score and computed tomography coronary angiography in high-risk asymptomatic subjects: assessment of diagnostic accuracy and prevalence of non-obstructive coronary artery disease. Eur Radiol 20(4):846–854. https://doi.org/10.1007/s00330-009-1612-2
Catalan P, Callejo D, Blasco JA (2013) Cost-effectiveness analysis of 64-slice computed tomography vs. cardiac catheterization to rule out coronary artery disease before non-coronary cardiovascular surgery. Eur Heart J Cardiovasc Imaging 14(2):149–157. https://doi.org/10.1093/ehjci/jes121
Feuchtner GM, Stolzmann P, Dichtl W, Schertler T, Bonatti J, Scheffel H, Mueller S, Plass A, Mueller L, Bartel T, Wolf F, Alkadhi H (2009) Multislice computed tomography in infective endocarditis: comparison with transesophageal echocardiography and intraoperative findings. J Am Coll Cardiol 53(5):436–444. https://doi.org/10.1016/j.jacc.2008.01.077
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP 3rd, Guyton RA, O’Gara PT, Ruiz CE, Skubas NJ, Sorajja P, Sundt TM, 3rd, Thomas JD (2014) 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 63 (22):e57-185. https://doi.org/10.1016/j.jacc.2014.02.536
Bettencourt N, Rocha J, Carvalho M, Leite D, Toschke AM, Melica B, Santos L, Rodrigues A, Goncalves M, Braga P, Teixeira M, Simoes L, Rajagopalan S, Gama V (2009) Multislice computed tomography in the exclusion of coronary artery disease in patients with presurgical valve disease. Circ Cardiovasc Imaging 2(4):306–313. https://doi.org/10.1161/CIRCIMAGING.108.827717
Jakamy R, Barthelemy O, Le Feuvre C, Berman E, Boutekadjirt R, Cluzel P, Metzger JP, Helft G (2012) Accuracy of multislice computed tomography in the preoperative assessment of coronary disease in patients scheduled for heart valve surgery. Arch Cardiovasc Dis 105(8–9):424–431. https://doi.org/10.1016/j.acvd.2012.05.004
Ciolina F, Sedati P, Zaccagna F, Galea N, Noce V, Miraldi F, Cavarretta E, Francone M, Carbone I (2015) Aortic valve stenosis: non-invasive preoperative evaluation using 64-slice CT angiography. J Cardiovasc Surg 56(5):799–808
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10554_2018_1318_MOESM1_ESM.tif
Supplementary material 1 (TIF 1112 KB). Timeline of major advances in infective endocarditis with the corresponding study and sample size. IE infective endocarditis, TTE transthoracic echocardiography, TEE transesophageal echocardiography, CT computed tomography, PET positron emission tomography
Rights and permissions
About this article
Cite this article
Sims, J.R., Anavekar, N.S., Chandrasekaran, K. et al. Utility of cardiac computed tomography scanning in the diagnosis and pre-operative evaluation of patients with infective endocarditis. Int J Cardiovasc Imaging 34, 1155–1163 (2018). https://doi.org/10.1007/s10554-018-1318-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-018-1318-0