Abstract
Purpose
Breast cancer has become the leading cause of cancer mortality in women. Although immune checkpoint inhibitors targeting programmed death-1 (PD-1) are promising, it remains unclear whether PD-L1 expression on circulating tumor cells (CTCs) has predictive and prognostic values in predicting and stratifying metastatic breast cancer (MBC) patients who can benefit from anti-PD-1 immunotherapy.
Methods
Twenty six MBC patients that received anti-PD-1 immunotherapy were enrolled in this study. The peptide-based Pep@MNPs method was used to isolate and enumerate CTCs from 2.0 ml of peripheral venous blood. The expression of PD-L1 on CTCs was evaluated by an established immunoscoring system categorizing into four classes (negative, low, medium, and high).
Results
Our data showed that 92.3% (24/26) of patients had CTCs, 83.3% (20/26) of patients had PD-L1-positive CTCs, and 65.4% (17/26) of patients had PD-L1-high CTCs. We revealed that the clinical benefit rate (CBR) of patients with a cut-off value of ≥ 35% PD-L1-high CTCs (66.6%) was higher than the others (29.4%). We indicated that PD-L1 expression on CTCs from MBC patients treated with anti-PD-1 monotherapy was dynamic. We demonstrated that MBC patients with a cut-off value of ≥ 35% PD-L1-high CTCs had longer PFS (P = 0.033) and OS (P = 0.00058) compared with patients with a cut-off value of < 35% PD-L1-high CTCs.
Conclusion
Our findings suggested that PD-L1 expression on CTCs could predict the therapeutic response and clinical outcomes, providing a valuable predictive and prognostic biomarker for patients treated with anti-PD-1 immunotherapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer (BC) is the most common cancers diagnosed in women [1]. In recent years, the mortality of BC has decreased with the advancements in the early diagnosis and therapeutics, but approximately 20% of the patients will experience relapse with distant metastatic disease within 5 years [2]. Although endocrine therapy is the standard of care for hormone receptor-positive BC, the risks of distant recurrence up to 41% depending on tumor characteristics were reported [3, 4]. Significantly poorer clinical outcomes were shown in patients with triple-negative BC [5, 6]. 10% to 20% of BC patients were defined as having TNBC, who had younger mean age at diagnosis and increased likelihood of distant recurrence compared with individuals with other types of BC [7]. Unfortunately, median overall survival (OS) for patients with metastatic triple-negative BC were only about 1 year compared with 5 years of other BC subtypes [8]. Thus, novel therapeutic strategies are needed to overcome the limitations of current BC therapies.
Immunotherapy has shown noticeable efficacy in several types of cancers, and immune checkpoint inhibitor (ICI) therapy is revolutionizing the BC treatment landscape [9]. In view of toxicity, immune-related adverse events, cost and healthcare utilization associated with the immune checkpoint inhibitors [10, 11], it is urgent to develop a means of predicting and stratifying patients who can benefit from immunotherapy [12]. Currently, assessment of programmed death-ligand 1 (PD-L1) on tissue sections using immunohistochemistry (IHC) assays is the most widely used [12, 13]. However, tumor heterogeneity, tissue sampling bias, and dynamic conversion of PD-L1 status limit the clinical practice of PD-L1 IHC as a reliable predictive biomarker [13, 14], emphasizing the importance of exploring more valuable and accessible methods.
Detection of circulating tumor cells (CTCs) is increasingly promising since it is a minimally invasive, repeatable, cost-effective and real-time “liquid biopsy”, which has been demonstrated to predict the outcome for patients with BC [15,16,17,18,19,20,21,22,23,24,25]. PD-L1 expression on CTCs was first reported in BC patients in 2015 [26] and subsequent studies have supported the predictive value of PD-L1-positive CTCs [27,28,29]. Nevertheless, these patients included were treated with conventional therapies rather than immunotherapies. In recent years, by taking advantages of the Pep@MNPs method, we have revealed that PD-L1 status in CTCs could predict treatment response in patients with various gastrointestinal cancers who received immune checkpoint blockade immunotherapies [30,31,32]. Whether PD-L1 status in CTCs has a predictive and prognostic value in BC patients treated with immunotherapies remains unclear.
In this study, we aimed to detect the expression of PD-L1 on CTCs using the Pep@MNPs method from MBC patients treated with anti-PD-1 immunotherapy. Moreover, the correlation of PD-L1 expression between CTCs and tumor tissues was evaluated. Furthermore, the predictive and prognostic values of PD-L1 expression on CTCs were explored. The study brings new insights into immunotherapy for patients with MBC.
Materials and methods
Patients
Eligible patients were required to have measurable or evaluable disease, with an Eastern Cooperative Oncology Group (ECOG) performance status score of 0 to 3. A total of 26 MBC patients involved in anti-PD-1 immunotherapy were enrolled in this study from the Fifth Medical Center of Chinese PLA General Hospital. 67 female controls were also included. The baseline samples were collected from October 2019 to October 2020. Blood samples from healthy controls were harvested for the establishment of CTC baseline. This study was reviewed and approved by the Ethics Committees of the Fifth Medical Center of Chinese PLA General Hospital. All patients and healthy individuals had given written informed consent. In this study, 24 patients had TNBC, whereas 2 patients with hormone receptor-positive and HER2-negative BC received immunotherapy since they were TMB-high [33, 34]. Among 6 patients with primary hormone receptor-positive BC, there were 4 patients with metastatic TNBC. The other 2 patients were treated with immunotherapy due to high tumor mutation burden.
Study design
For CTC detection, 2.0 ml of peripheral venous blood was drawn for further enumeration and analysis of PD-L1 expression. The assessment of PD-L1 in tissue biopsies was determined by IHC in the Department of Pathology. Evaluation of disease status was carried out according to Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 criteria. After follow-up, the correlation between PD-L1 expression on CTCs and clinical outcomes was assessed by statistical analysis as described previously [32].
Isolation and characterization of CTCs
CTCs were isolated and enumerated from 2.0 ml of peripheral venous blood using our previously established Pep@MNPs method [23,24,25, 35]. Briefly, the streptavidin-conjugated iron oxide magnetic nanoparticles (MNPs) are equipped with specialized peptides that target epithelial cell adhesion molecule (EpCAM). For each blood sample, 10 μl Pep@MNPs was added, and incubated at 37 °C for 1 h. Under a magnetic field, isolated CTCs were captured. For selection of CTCs, DAPI and multiple antibodies such as cytokeratin mix (Abcam, ab264485, 1:200) and CD45-phycoerythrin (Abcam, ab223183, 1:200) were employed. CTCs were identified as CKmix+/DAPI+/CD45-cells under OLYMPUS IX73 fluorescence microscope.
Semiquantitative assessment of PD-L1 on CTCs
For detection of PD-L1 expression on CTCs, we have constructed an efficient PD-L1 evaluation system based on mean fluorescence intensity [30,31,32]. Accordingly, different PD-L1 expression on CTCs was categorized into four groups: negative (−), low (+), medium (++), and high (+++).
Statistical analysis
All statistical computations were performed based on R version 3.6.3. Receiver operating characteristic (ROC) analyses were implemented in python3.7.6. Correlation between PD-L1 expression on CTCs and disease status was analyzed using Spearman’s rank correlation coefficient test. PFS and OS curves were estimated using the Kaplan–Meier test. P values < 0.05 were considered statistically significant.
Results
MBC patient information
Patient and tumor characteristics are summarized in Table 1. Totally 26 patients with MBC were included and the samples were harvested from November 2016 to January 2021. The median age of the enrolled patients was 47 years (range: 33 to 60 years). Among them, 15 patients were treated with anti-PD-1 monotherapy, while 11 patients received anti-PD-1 immunotherapy in combination with chemotherapy.
CTC enumeration between MBC patients and controls
In our previous studies, we have developed the Pep@MNPs method to isolate and analyze CTCs from BC patients, providing valuable prognostic insights into conventional therapies [23,24,25]. To examine the efficacy of CTC detection, we firstly enumerated the number of CTCs. Among 26 patients with MBC, CTCs were detected in 24 (92.3%) patients, with the median number of CTCs being 5 (range 0 to 53) (Fig. 1A). In contrast, the median number of CTCs in volunteers was 0 (range 0 to 3). The difference of CTC enumeration within 2 ml of blood between MBC patients and female controls was significant (P = 0.0000000239) (Fig. 1A), with area under the curve (AUC) being 0.9022 as indicated by the receiver operating characteristic (ROC) curve (Fig. 1B).
Comparisons of CTC numbers between MBC patients and female controls. A The difference of CTC enumeration detected within 2 ml of blood in MBC patients and healthy controls was significant (P = 0.0000000239). B The receiver operating characteristic curve indicated that the area under the curve was 0.9022. ROC, receiver operating characteristic; AUC, area under the curve
PD-L1 expression on CTCs and the IHC results
Recently, we have established a quantitative system utilizing the peptide-based assay for the evaluation of PD-L1 and verified it in several types of gastrointestinal cancers [30,31,32]. According to the scoring system, PD-L1 expression was defined by four categories: negative (−), low (+), medium (++), and high (+++) [30]. To assess the correlation of PD-L1 expression between CTCs and the IHC results, CTC samples and tumor tissues were analyzed in MBC patients treated with anti-PD-1 immunotherapy. 83.3% (20/26) of patients had PD-L1-positive CTCs, including PD-L1-low CTCs (CTCPD-L1+), PD-L1-medium CTCs (CTCPD-L1++), and PD-L1-high CTCs (CTCPD-L1+++). 17 of 26 (65.4%) patients had at least one CTCPD-L1+++. The expression of PD-L1 was also tested in 14 patients with available tissue biopsies. Surprisingly, the IHC results were positive for PD-L1 in 3 of 14 (21.4%) patients, and only one patient showed high expression of PD-L1 in the IHC results (Fig. 2, Table 2). Notably, all of the 3 patients were found to have PD-L1-positive CTCs. These observations indicated that the expression of PD-L1 was more frequent and higher on CTCs compared with the IHC results.
Representative IHC staining of PD-L1 expression in tumor tissues from 3 MBC patients whose IHC results were positive for PD-L1. A Only one patient (No. 51970837) had high PD-L1 expression in the IHC results. B, C The other two patients (No. 52147425 and No. 52003347) showed low expression of PD-L1 in the IHC results. IHC immunohistochemistry
Correlation between levels of PD-L1 on CTCs prior to immunotherapy and drug response
Among the 26 patients, 15 (57.7%) were with progressive disease (PD), 8 (30.8%) had a stable disease (SD), and the other 3 (11.5%) achieved a partial response (PR) (Fig. 3A). To ascertain whether PD-L1 expression on CTCs was correlated with the response of patients to anti-PD-1 immunotherapy, we carried out a thorough assessment of the ratio distribution of PD-L1-high CTCs among total CTCs and defined a cut-off value as previously described [30,31,32]. Interestingly, when a cut-off value of 35% for the ratio of PD-L1-high CTCs at baseline was defined, the clinical benefit rate (CBR) below and above the cut-off value was 29.4% (5/17) and 66.6% (6/9), respectively (Fig. 3B). These data suggested that the CBR of patients with a cut-off value of ≥ 35% PD-L1-high CTCs might be higher than that of patients with a cut-off value of < 35% PD-L1-high CTCs.
Correlation between PD-L1 expression on CTCs at baseline and disease status. A 26 MBC patients were categorized into 3 groups based on the drug response, and proportional distribution of PD-L1 on CTCs prior to anti-PD-1 immunotherapy was listed. The dashed line referred to the cut-off value of 35% for the ratio of PD-L1-high CTCs at baseline. B Response rate of patients below and above the cut-off value was analyzed. PR partial response, SD stable disease, DC disease control, PD progressive disease
Dynamic changes of PD-L1 expression on CTCs before and after anti-PD-1 monotherapy
To determine whether the expression of PD-L1 on CTCs alters due to immunotherapy, we detected PD-L1 expression on CTCs from the 14 patients treated with anti-PD-1 monotherapy who were available for the second blood draw. Among the 14 patients, 11 were with PD and 3 had a disease control (DC). PD-L1 distribution on CTCs and clinical response were further compared. Interestingly, the ratio of PD-L1-high CTCs were found to be significantly decreased in DC group compared with that in PD group (P = 0.015) (Fig. 4). The findings showed that PD-L1 expression on CTCs during anti-PD-1 treatment was dynamic and the real-time assay might have a monitoring value.
Dynamic changes of PD-L1-high CTCs before and after anti-PD-1 immunotherapy. The ratio of PD-L1-high CTCs from 14 patients treated with anti-PD-1 monotherapy at T0 (baseline) and T1 (after treatment) were analyzed. The ratio of PD-L1-high CTCs were decreased in DC group compared with that in PD group (P = 0.015). DC disease control, PD progressive disease
Evaluation of PD-L1 expression on CTCs to predict PFS and OS
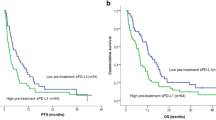
Accumulative data have confirmed important correlations between PD-L1 overexpression on tumor cells and clinical outcomes, but the prognostic value remains controversial due to different types of cancers [36,37,38]. To investigate the potential prognostic roles of PD-L1 expression on CTCs in predicting the clinical outcomes of BC patients treated with anti-PD-1 immunotherapy, we evaluated the PFS and OS of 26 patients based on the expression of PD-L1 on CTCs at baseline. An obvious but not significant improved PFS was observed in patients with PD-L1-positive CTCs compared with that in patients with PD-L1-negative CTCs (P = 0.075) (Fig. 5A). However, patients with a cut-off value of ≥ 35% PD-L1-high CTCs had significantly longer PFS compared with patients with a cut-off value of < 35% PD-L1-high CTCs (P = 0.033) (Fig. 5B). The OS of patients with PD-L1-positive CTCs was significantly longer compared with that of patients without PD-L1-positive CTCs (P = 0.000505) (Fig. 5C). Significantly longer OS was also observed in patients with a cut-off value of ≥ 35% PD-L1-high CTCs compared with that in patients with a cut-off value of < 35% PD-L1-high CTCs (P = 0.00058) (Fig. 5D). Collectively, these results showed that PD-L1 expression on CTCs might predict the clinical prognosis of patients treated with anti-PD-1-directed therapy.
Correlation between PD-L1 expression on CTCs at baseline and clinical outcomes. Kaplan–Meier estimates of PFS and OS of patients in different subgroups were analyzed. A The PFS of patients with PD-L1-positive CTCs was obviously but insignificantly improved compared with that of patients with PD-L1-negative CTCs (P = 0.075). B The PFS of patients with a cut-off value of ≥ 35% PD-L1-high CTCs was significantly longer compared with that of patients with a cut-off value of < 35% PD-L1-high CTCs (P = 0.033). C The OS of patients with PD-L1-positive CTCs was significantly longer compared with that of patients without PD-L1-positive CTCs (P = 0.000505). D The OS of patients with a cut-off value of ≥ 35% PD-L1-high CTCs was significantly longer compared with that of patients with a cut-off value of < 35% PD-L1-high CTCs (P = 0.00058). PFS progression-free survival, OS overall survival
Discussion
The clinical applications of immune checkpoint inhibitors (ICIs) targeting PD-1 and PD-L1 have revolutionized the therapeutics of several cancer types, including lung cancer and melanoma. ICI therapy is a milestone in the treatment of malignancies, as the patient’s prognosis and quality of life are significantly improved. ICI-based immunotherapy of BC has mainly focused on TNBC, which is the most difficult type of BC to treat. Meanwhile, compared with patients with other BC subtypes, patients with TNBC might be more likely to benefit from ICI therapies, due to the expression of PD-L1, tumor mutational burden (TMB), and the high level of tumor infiltrating lymphocytes [9, 39]. Currently anti-PD-1 monoclonal antibody has been shown to exert antitumor activity for the first-line treatment of PD-L1-positive metastatic TNBC and neoadjuvant treatment in patients with high-risk early TNBC [40, 41]. The therapeutic value of immunotherapy has also been investigated in second-line and later-line treatments. There is no satisfactory indicator for the treatment efficacy prediction. Testing the expression of PD-L1 on the tumor cell surface by immunohistochemistry holds great promise. But current data are not ideal as the factors including technology and biological issues might both influence the results. For instance, the cut-off value of PD-L1 staining positivity is indeterminate, testing antibodies lack standardization, and more importantly, the tumor heterogeneity exists and the expression of PD-1/PD-L1 is dynamic [42].
Though the presence of PD-L1 on CTCs in BC patients treated with conventional therapies has been found [26,27,28,29], whether PD-L1 expression on CTCs is correlated with the therapeutic response and prognosis in MBC patients treated with immunotherapy remains largely unknown. Herein, using the Pep@MNPs method and an established quantitative system, we showed that PD-L1 was expressed more frequently and higher on CTCs compared with that in tumor tissues. Moreover, we indicated that PD-L1 expression on CTCs might help stratify and monitor patients that can benefit from the therapeutics. Furthermore, we suggested that PD-L1 expression on CTCs might serve as a potential prognostic indicator of PFS and OS. To the best of our knowledge, we demonstrated for the first time that PD-L1 expression on CTCs has predictive and prognostic values in MBC patients treated with anti-PD-1 immunotherapy.
Tumor cells can evade immune surveillance by overexpressing PD-L1 [12, 43]. Given that the response rate to anti-PD-1 antibodies is still low, a better understanding of PD-L1 would be beneficial for the prediction of tumor response and enhancement in treatment efficacy [43]. For MBC patients, tumor cells of primary lesion may be different from those of metastatic lesion, which may take years to develop and is difficult to biopsy [26]. Considerable discordance of PD-L1 expression has been observed between primary tumors and metastasis in BC [14]. Notably, CTCs are able to provide valuable real-time information. Our results showed that PD-L1 was expressed frequently on CTCs from patients with BC, which are consistent with recent reports [26,27,28,29]. Although the correlation of PD-L1 expression between tissue biopsies and CTCs was not considered significant [27], we found that 3 patients with PD-L1-positive IHC results also had PD-L1-positive CTCs. However, further research is needed to investigate the positive concordance.
It is noteworthy that the percentage of PD-L1-positive CTCs in patients with BC varies among different research groups, which might be explained by patient populations included and the differences in CTC enrichment methods applied such as CellSearch® system and Ficoll-Hypaque density gradient centrifugation [26,27,28,29]. The FDA-cleared CellSearch® system has been tested for the enumeration of CTCs in MBC patients [44]. However, these CTC enrichment methods need 7.5 ml to 10 ml of whole blood samples [26,27,28,29]. Compared with these methods, the Pep@MNPs method employed in this work only used 2.0 ml of blood, which reduced the amount of samples and might be beneficial for the real-time monitoring of disease status. Moreover, the capture efficiency of the Pep@MNPs method was high in this work, which is consistent with our previous studies [30, 35, 45]. Making use of the highly sensitive Pep@MNPs method, our data suggested a higher proportion of PD-L1-positive CTCs, which are in line with previously published results [30,31,32]. Interestingly, when PD-L1 expression on CTCs was first confirmed, the investigators developed immunoscores for various levels of PD-L1 defined as no, low, and high PD-L1 expression [26]. To better assess the value of PD-L1 distribution, our group established a scoring system categorizing PD-L1 expression on CTCs into four classes: negative, low, medium, and high [30], which are consistent with subsequent reports [27, 31, 32].
Since considerable variation in the intensity of PD-L1 staining among patients and within single CTC had been found [26], the ratio distribution of PD-L1-high CTCs was identified as a plausible biomarker [30]. Furthermore, the usage of optimal cut-off values of PD-L1-high CTCs has been demonstrated to efficiently predict the response to immunotherapies [30,31,32]. Our study defined a cut-off value of 35% for the ratio of PD-L1-high CTCs, and suggested that the cut-off value might help select MBC patients that most likely benefit from immunotherapies. The dynamic changes of PD-L1 expression on CTCs were observed during anti-PD-1 treatment. It might be important to assess the PD-L1 dynamic changes in CTCs, which could harvest the first-hand knowledge and help monitor the oncologic shifts induced by immunotherapies. Importantly, the dynamic changes of PD-L1 status on CTCs might be a real-time monitor for treatments, which could help mirroring the therapeutic response. The availability of resampling might be further improved using the Pep@MNPs method with the reduced demand for the amount of blood samples.
The prognostic value of PD-L1 expression remains controversial depending on different types of cancers [38]. PD-L1 distribution on CTC, however, has been shown to have prognostic significance in several types of cancers [30, 32]. In MBC patients treated with conventional therapies, PD-L1-positive CTCs were associated with shorter PFS [27, 28]. By contrast, our results indicated that in MBC patients who received anti-PD-1 immunotherapy, individuals with a cut-off value of ≥ 35% PD-L1-high CTCs had longer PFS and OS. Considering that PD-L1 expression helps tumor cells evade immune surveillance and inhibition of PD-L1 promotes antitumor immunity [12, 43], PD-L1 expression on CTCs might serve as a potential prognostic indicator.
Among the patients, the objective response rate (ORR) was 11.5%, the median PFS was 2.1 months, and the median OS was 8.75 months. The results were associated with the facts that 96% of the patients received second-line and later-line treatments, 58% of the patients received anti-PD-1 antibody monotherapy, and only one patient had high histological PD-L1 expression, which were similar to previous research data [46,47,48,49]. Although our data revealed potential predictive and prognostic importance of PD-L1 expression on CTCs from MBC patients, there were some limitations. The number of the enrolled patients who received anti-PD-1 immunotherapy was limited, influencing further statistical analysis. The tissue biopsies available for the assessment of PD-L1 expression were also limited in this study. The relatively small number of patients may limit the interpretability of these results. Since this exploratory study mainly focused on the patients with second-line and later-line treatments, and 58% of the patients (15/26) had at least three metastatic sites, the application of anti-PD-1 immunotherapy was limited for second-line and later-line treatments, resulting in a relatively small size of the study population. In this work, wide heterogeneity was observed in the expression of PD-L1 between CTCs and IHC results. Interestingly, when the patients were included accordingly, only one patient showed high expression of PD-L1 in the IHC results (TPS = 70%). Given that several reports have recently demonstrated responses to immunotherapies in PD-L1-negative BC patients [50,51,52], in an attempt to receive the immunotherapies for second-line and later-line treatments, the patients provided written informed consent. However, the number of these patients was also limited. Among them, 15 patients received anti-PD-1 monotherapy, whereas 11 patients were treated with anti-PD-1 monotherapy together with chemotherapies such as Taxane and Apatinib, which may prevent firm conclusions from being drawn regarding our evaluation of the immunotherapies. Nevertheless, our findings indicated that patients with PD-L1-high CTCs were associated with a significant improvement, providing a valuable biomarker worthy to be noted. To obtain more robust evidence, further investigations were required in a larger cohort. Moreover, heterogeneity in the study population may not allow an accurate evaluation to be made. Therefore, due to the inherent weaknesses of this work, cautious interpretation of our data are warranted. It will be interesting to evaluate the therapeutic effects and safety in MBC patients who were treated with anti-PD-1 monotherapy avoiding the heterogeneity in the study population. We plan to further explore the number of CTCs as well as the expression status of PD-L1 in a real-time manner among MBC patients who received anti-PD-1 monotherapy in the near future. Further assessment of the relationship between PD-L1 expression in blood/tissue and TMB was also significant. With better and more accurate evaluation data, optimal therapeutics could be provided for MBC patients.
Conclusions
In summary, by utilizing the Pep@MNPs method and an established immunoscoring system, we demonstrated that PD-L1-high CTCs was frequent and dynamic in MBC patients treated with anti-PD-1 immunotherapy. The ratio distribution of PD-L1-high CTCs in combination with the defined cut-off value might help predict the drug response, as well as PFS and OS. All these observations suggested that PD-L1 expression on CTCs might present a valuable predictive and prognostic tool in stratifying MBC patients who are most likely to benefit from anti-PD-1 immunotherapy.
Data availability
All data are available within the manuscript or may be obtained from the corresponding authors upon reasonable request.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics. CA Cancer J Clin 72(1):7–33. https://doi.org/10.3322/caac.21708
Zou Y, Zou X, Zheng S, Tang H, Zhang L, Liu P, Xie X (2020) Efficacy and predictive factors of immune checkpoint inhibitors in metastatic breast cancer: a systematic review and meta-analysis. Ther Adv Med Oncol 12:1758835920940928. https://doi.org/10.1177/1758835920940928
Pan H, Gray R, Braybrooke J, Davies C, Taylor C, McGale P, Peto R, Pritchard KI, Bergh J, Dowsett M, Hayes DF (2017) 20-Year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med 377(19):1836–1846. https://doi.org/10.1056/NEJMoa1701830
Salvo EM, Ramirez AO, Cueto J, Law EH, Situ A, Cameron C, Samjoo IA (2021) Risk of recurrence among patients with HR-positive, HER2-negative, early breast cancer receiving adjuvant endocrine therapy: a systematic review and meta-analysis. Breast 57:5–17. https://doi.org/10.1016/j.breast.2021.02.009
Bonotto M, Gerratana L, Poletto E, Driol P, Giangreco M, Russo S, Minisini AM, Andreetta C, Mansutti M, Pisa FE, Fasola G, Puglisi F (2014) Measures of outcome in metastatic breast cancer: insights from a real-world scenario. Oncologist 19(6):608–615. https://doi.org/10.1634/theoncologist.2014-0002
Schmid P, Adams S, Rugo HS, Schneeweiss A, Barrios CH, Iwata H, Dieras V, Hegg R, Im SA, Shaw Wright G, Henschel V, Molinero L, Chui SY, Funke R, Husain A, Winer EP, Loi S, Emens LA, Investigators IMT (2018) Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer. N Engl J Med 379(22):2108–2121. https://doi.org/10.1056/NEJMoa1809615
Dent R, Trudeau M, Pritchard KI, Hanna WM, Kahn HK, Sawka CA, Lickley LA, Rawlinson E, Sun P, Narod SA (2007) Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res 13(15 Pt 1):4429–4434. https://doi.org/10.1158/1078-0432.CCR-06-3045
Waks AG, Winer EP (2019) Breast cancer treatment: a review. JAMA 321(3):288–300. https://doi.org/10.1001/jama.2018.19323
Barzaman K, Moradi-Kalbolandi S, Hosseinzadeh A, Kazemi MH, Khorramdelazad H, Safari E, Farahmand L (2021) Breast cancer immunotherapy: current and novel approaches. Int Immunopharmacol 98:107886. https://doi.org/10.1016/j.intimp.2021.107886
Chennamadhavuni A, Abushahin L, Jin N, Presley CJ, Manne A (2022) Risk factors and biomarkers for immune-related adverse events: a practical guide to identifying high-risk patients and rechallenging immune checkpoint inhibitors. Front Immunol 13:779691. https://doi.org/10.3389/fimmu.2022.779691
Gunturu KS, Pham TT, Shambhu S, Fisch MJ, Barron JJ, Debono D (2022) Immune checkpoint inhibitors: immune-related adverse events, healthcare utilization, and costs among commercial and Medicare Advantage patients. Support Care Cancer 30(5):4019–4026. https://doi.org/10.1007/s00520-022-06826-9
Doroshow DB, Bhalla S, Beasley MB, Sholl LM, Kerr KM, Gnjatic S, Wistuba II, Rimm DL, Tsao MS, Hirsch FR (2021) PD-L1 as a biomarker of response to immune-checkpoint inhibitors. Nat Rev Clin Oncol 18(6):345–362. https://doi.org/10.1038/s41571-021-00473-5
Hofman P, Heeke S, Alix-Panabieres C, Pantel K (2019) Liquid biopsy in the era of immuno-oncology: is it ready for prime-time use for cancer patients? Ann Oncol 30(9):1448–1459. https://doi.org/10.1093/annonc/mdz196
Boman C, Zerdes I, Martensson K, Bergh J, Foukakis T, Valachis A, Matikas A (2021) Discordance of PD-L1 status between primary and metastatic breast cancer: a systematic review and meta-analysis. Cancer Treat Rev 99:102257. https://doi.org/10.1016/j.ctrv.2021.102257
Wang Y, Zhou Y, Hu Z (2017) The functions of circulating tumor cells in early diagnosis and surveillance during cancer advancement. J Transl Int Med 5(3):135–138. https://doi.org/10.1515/jtim-2017-0029
Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, Reuben JM, Doyle GV, Allard WJ, Terstappen LW, Hayes DF (2004) Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 351(8):781–791. https://doi.org/10.1056/NEJMoa040766
Cristofanilli M, Hayes DF, Budd GT, Ellis MJ, Stopeck A, Reuben JM, Doyle GV, Matera J, Allard WJ, Miller MC, Fritsche HA, Hortobagyi GN, Terstappen LW (2005) Circulating tumor cells: a novel prognostic factor for newly diagnosed metastatic breast cancer. J Clin Oncol 23(7):1420–1430. https://doi.org/10.1200/JCO.2005.08.140
Hayes DF, Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Miller MC, Matera J, Allard WJ, Doyle GV, Terstappen LW (2006) Circulating tumor cells at each follow-up time point during therapy of metastatic breast cancer patients predict progression-free and overall survival. Clin Cancer Res 12(14 Pt 1):4218–4224. https://doi.org/10.1158/1078-0432.CCR-05-2821
Budd GT, Cristofanilli M, Ellis MJ, Stopeck A, Borden E, Miller MC, Matera J, Repollet M, Doyle GV, Terstappen LW, Hayes DF (2006) Circulating tumor cells versus imaging–predicting overall survival in metastatic breast cancer. Clin Cancer Res 12(21):6403–6409. https://doi.org/10.1158/1078-0432.CCR-05-1769
Liu Y, Liu Q, Wang T, Bian L, Zhang S, Hu H, Li S, Hu Z, Wu S, Liu B, Jiang Z (2013) Circulating tumor cells in HER2-positive metastatic breast cancer patients: a valuable prognostic and predictive biomarker. BMC Cancer 13:202. https://doi.org/10.1186/1471-2407-13-202
Jiang ZF, Cristofanilli M, Shao ZM, Tong ZS, Song EW, Wang XJ, Liao N, Hu XC, Liu Y, Wang Y, Zeng L, Zhang M (2013) Circulating tumor cells predict progression-free and overall survival in Chinese patients with metastatic breast cancer, HER2-positive or triple-negative (CBCSG004): a multicenter, double-blind, prospective trial. Ann Oncol 24(11):2766–2772. https://doi.org/10.1093/annonc/mdt246
Zhang S, Li L, Wang T, Bian L, Hu H, Xu C, Liu B, Liu Y, Cristofanilli M, Jiang Z (2016) Real-time HER2 status detected on circulating tumor cells predicts different outcomes of anti-HER2 therapy in histologically HER2-positive metastatic breast cancer patients. BMC Cancer 16:526. https://doi.org/10.1186/s12885-016-2578-5
Liu XR, Shao B, Peng JX, Li HP, Yang YL, Kong WY, Song GH, Jiang HF, Liang X, Yan Y (2017) Identification of high independent prognostic value of nanotechnology based circulating tumor cell enumeration in first-line chemotherapy for metastatic breast cancer patients. Breast 32:119–125. https://doi.org/10.1016/j.breast.2017.01.007
Liu X, Ran R, Shao B, Rugo HS, Yang Y, Hu Z, Wei Z, Wan F, Kong W, Song G, Jiang H, Liang X, Zhang R, Yan Y, Xu G, Li H (2018) Combined peripheral natural killer cell and circulating tumor cell enumeration enhance prognostic efficiency in patients with metastatic triple-negative breast cancer. Chin J Cancer Res 30(3):315–326. https://doi.org/10.21147/j.issn.1000-9604.2018.03.04
Zhou Y, Zhou J, Xiao J, Wang Y, Wang H, Shi H, Yue C, Jia F, Li P, Hu Z, Yang Y, Jiang Z, Wang T (2022) Prognostic relevance of estrogen receptor status in circulating tumor cells in breast cancer patients treated with endocrine therapy. Front Oncol 12:866293. https://doi.org/10.3389/fonc.2022.866293
Mazel M, Jacot W, Pantel K, Bartkowiak K, Topart D, Cayrefourcq L, Rossille D, Maudelonde T, Fest T, Alix-Panabieres C (2015) Frequent expression of PD-L1 on circulating breast cancer cells. Mol Oncol 9(9):1773–1782. https://doi.org/10.1016/j.molonc.2015.05.009
Jacot W, Mazel M, Mollevi C, Pouderoux S, D’Hondt V, Cayrefourcq L, Bourgier C, Boissiere-Michot F, Berrabah F, Lopez-Crapez E, Bidard FC, Viala M, Maudelonde T, Guiu S, Alix-Panabieres C (2020) Clinical correlations of programmed cell death ligand 1 status in liquid and standard biopsies in breast cancer. Clin Chem 66(8):1093–1101. https://doi.org/10.1093/clinchem/hvaa121
Papadaki MA, Koutsopoulos AV, Tsoulfas PG, Lagoudaki E, Aggouraki D, Monastirioti A, Koutoulaki C, Apostolopoulou CA, Merodoulaki AC, Papadaki C, Mavroudis D, Agelaki S (2020) Clinical relevance of immune checkpoints on circulating tumor cells in breast cancer. Cancers 12(2):376. https://doi.org/10.3390/cancers12020376
Darga EP, Dolce EM, Fang F, Kidwell KM, Gersch CL, Kregel S, Thomas DG, Gill A, Brown ME, Gross S, Connelly M, Holinstat M, Cobain EF, Rae JM, Hayes DF, Paoletti C (2021) PD-L1 expression on circulating tumor cells and platelets in patients with metastatic breast cancer. PLoS ONE 16(11):e0260124. https://doi.org/10.1371/journal.pone.0260124
Yue C, Jiang Y, Li P, Wang Y, Xue J, Li N, Li D, Wang R, Dang Y, Hu Z, Yang Y, Xu J (2018) Dynamic change of PD-L1 expression on circulating tumor cells in advanced solid tumor patients undergoing PD-1 blockade therapy. Oncoimmunology 7(7):e1438111. https://doi.org/10.1080/2162402X.2018.1438111
Xu J, Zhang Y, Jia R, Yue C, Chang L, Liu R, Zhang G, Zhao C, Zhang Y, Chen C, Wang Y, Yi X, Hu Z, Zou J, Wang Q (2019) Anti-PD-1 Antibody SHR-1210 combined with apatinib for advanced hepatocellular carcinoma, gastric, or esophagogastric junction cancer: an open-label, dose escalation and expansion study. Clin Cancer Res 25(2):515–523. https://doi.org/10.1158/1078-0432.CCR-18-2484
Tan Z, Yue C, Ji S, Zhao C, Jia R, Zhang Y, Liu R, Li D, Yu Q, Li P, Hu Z, Yang Y, Xu J (2021) Assessment of PD-L1 expression on circulating tumor cells for predicting clinical outcomes in patients with cancer receiving PD-1/PD-L1 blockade therapies. Oncologist 26(12):e2227–e2238. https://doi.org/10.1002/onco.13981
Marcus L, Fashoyin-Aje LA, Donoghue M, Yuan M, Rodriguez L, Gallagher PS, Philip R, Ghosh S, Theoret MR, Beaver JA, Pazdur R, Lemery SJ (2021) FDA approval summary: pembrolizumab for the treatment of tumor mutational burden-high solid tumors. Clin Cancer Res 27(17):4685–4689. https://doi.org/10.1158/1078-0432.CCR-21-0327
O’Meara TA, Tolaney SM (2021) Tumor mutational burden as a predictor of immunotherapy response in breast cancer. Oncotarget 12(5):394–400. https://doi.org/10.18632/oncotarget.27877
Bai L, Du Y, Peng J, Liu Y, Wang Y, Yang Y, Wang C (2014) Peptide-based isolation of circulating tumor cells by magnetic nanoparticles. J Mater Chem B 2(26):4080–4088. https://doi.org/10.1039/c4tb00456f
Patel SP, Kurzrock R (2015) PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther 14(4):847–856. https://doi.org/10.1158/1535-7163.MCT-14-0983
Meng X, Huang Z, Teng F, Xing L, Yu J (2015) Predictive biomarkers in PD-1/PD-L1 checkpoint blockade immunotherapy. Cancer Treat Rev 41(10):868–876. https://doi.org/10.1016/j.ctrv.2015.11.001
Wang X, Teng F, Kong L, Yu J (2016) PD-L1 expression in human cancers and its association with clinical outcomes. Onco Targets Ther 9:5023–5039. https://doi.org/10.2147/OTT.S105862
Agostinetto E, Losurdo A, Nader-Marta G, Santoro A, Punie K, Barroso R, Popovic L, Solinas C, Kok M, de Azambuja E, Lambertini M (2022) Progress and pitfalls in the use of immunotherapy for patients with triple negative breast cancer. Expert Opin Investig Drugs 31(6):567–591. https://doi.org/10.1080/13543784.2022.2049232
Cortes J, Cescon DW, Rugo HS, Nowecki Z, Im SA, Yusof MM, Gallardo C, Lipatov O, Barrios CH, Holgado E, Iwata H, Masuda N, Otero MT, Gokmen E, Loi S, Guo Z, Zhao J, Aktan G, Karantza V, Schmid P, Investigators K (2020) Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer (KEYNOTE-355): a randomised, placebo-controlled, double-blind, phase 3 clinical trial. Lancet 396(10265):1817–1828. https://doi.org/10.1016/S0140-6736(20)32531-9
Schmid P, Cortes J, Pusztai L, McArthur H, Kummel S, Bergh J, Denkert C, Park YH, Hui R, Harbeck N, Takahashi M, Foukakis T, Fasching PA, Cardoso F, Untch M, Jia L, Karantza V, Zhao J, Aktan G, Dent R, O’Shaughnessy J, Investigators K (2020) Pembrolizumab for early triple-negative breast cancer. N Engl J Med 382(9):810–821. https://doi.org/10.1056/NEJMoa1910549
Rzhevskiy A, Kapitannikova A, Malinina P, Volovetsky A, Aboulkheyr Es H, Kulasinghe A, Thiery JP, Maslennikova A, Zvyagin AV, Ebrahimi Warkiani M (2021) Emerging role of circulating tumor cells in immunotherapy. Theranostics 11(16):8057–8075. https://doi.org/10.7150/thno.59677
Chen G, Huang AC, Zhang W, Zhang G, Wu M, Xu W, Yu Z, Yang J, Wang B, Sun H, Xia H, Man Q, Zhong W, Antelo LF, Wu B, Xiong X, Liu X, Guan L, Li T, Liu S, Yang R, Lu Y, Dong L, McGettigan S, Somasundaram R, Radhakrishnan R, Mills G, Lu Y, Kim J, Chen YH, Dong H, Zhao Y, Karakousis GC, Mitchell TC, Schuchter LM, Herlyn M, Wherry EJ, Xu X, Guo W (2018) Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 560(7718):382–386. https://doi.org/10.1038/s41586-018-0392-8
Lee JS, Magbanua MJM, Park JW (2016) Circulating tumor cells in breast cancer: applications in personalized medicine. Breast Cancer Res Treat 160(3):411–424. https://doi.org/10.1007/s10549-016-4014-6
Li W, Wang H, Zhao Z, Gao H, Liu C, Zhu L, Wang C, Yang Y (2019) Emerging nanotechnologies for liquid biopsy: the detection of circulating tumor cells and extracellular vesicles. Adv Mater 31(45):e1805344. https://doi.org/10.1002/adma.201805344
Nanda R, Chow LQ, Dees EC, Berger R, Gupta S, Geva R, Pusztai L, Pathiraja K, Aktan G, Cheng JD, Karantza V, Buisseret L (2016) Pembrolizumab in patients with advanced triple-negative breast cancer: phase Ib KEYNOTE-012 study. J Clin Oncol 34(21):2460–2467. https://doi.org/10.1200/JCO.2015.64.8931
Adams S, Loi S, Toppmeyer D, Cescon DW, De Laurentiis M, Nanda R, Winer EP, Mukai H, Tamura K, Armstrong A, Liu MC, Iwata H, Ryvo L, Wimberger P, Rugo HS, Tan AR, Jia L, Ding Y, Karantza V, Schmid P (2019) Pembrolizumab monotherapy for previously untreated, PD-L1-positive, metastatic triple-negative breast cancer: cohort B of the phase II KEYNOTE-086 study. Ann Oncol 30(3):405–411. https://doi.org/10.1093/annonc/mdy518
Adams S, Schmid P, Rugo HS, Winer EP, Loirat D, Awada A, Cescon DW, Iwata H, Campone M, Nanda R, Hui R, Curigliano G, Toppmeyer D, O’Shaughnessy J, Loi S, Paluch-Shimon S, Tan AR, Card D, Zhao J, Karantza V, Cortes J (2019) Pembrolizumab monotherapy for previously treated metastatic triple-negative breast cancer: cohort A of the phase II KEYNOTE-086 study. Ann Oncol 30(3):397–404. https://doi.org/10.1093/annonc/mdy517
Winer EP, Lipatov O, Im SA, Goncalves A, Munoz-Couselo E, Lee KS, Schmid P, Tamura K, Testa L, Witzel I, Ohtani S, Turner N, Zambelli S, Harbeck N, Andre F, Dent R, Zhou X, Karantza V, Mejia J, Cortes J, Investigators Group (2021) Pembrolizumab versus investigator-choice chemotherapy for metastatic triple-negative breast cancer (KEYNOTE-119): a randomised, open-label, phase 3 trial. Lancet Oncol 22(4):499–511. https://doi.org/10.1016/S1470-2045(20)30754-3
Chumsri S, Sokol ES, Soyano-Muller AE, Parrondo RD, Reynolds GA, Nassar A, Thompson EA (2020) Durable complete response with immune checkpoint inhibitor in breast cancer with high tumor mutational burden and APOBEC signature. J Natl Compr Canc Netw 18(5):517–521. https://doi.org/10.6004/jnccn.2020.7543
Braso-Maristany F, Sanso M, Chic N, Martinez D, Gonzalez-Farre B, Sanfeliu E, Ghiglione L, Carcelero E, Garcia-Corbacho J, Sanchez M, Soy D, Jares P, Peg V, Saura C, Munoz M, Prat A, Vivancos A (2021) Case report: a case study documenting the activity of atezolizumab in a PD-L1-negative triple-negative breast cancer. Front Oncol 11:710596. https://doi.org/10.3389/fonc.2021.710596
Chic N, Braso-Maristany F, Prat A (2022) Biomarkers of immunotherapy response in breast cancer beyond PD-L1. Breast Cancer Res Treat 191(1):39–49. https://doi.org/10.1007/s10549-021-06421-2
Funding
This work was supported by grants from Research Foundation for Advanced Talents of Fujian Medical University (XRCZX2017020, XRCZX2019005), Beijing Natural Science Foundation (7192198), Strategic Priority Research Program of Chinese Academy of Sciences (XDB36000000, XDB38010400), the National Natural Science Foundation of China (32027801, 31870992, 21775031), the Natural Science Foundation of Fujian Province (2022J01203), CAS-JSPS (GJHZ2094), Science and Technology Service Network Initiative of the Chinese Academy of Sciences (KFJ-STS-ZDTP-079).
Author information
Authors and Affiliations
Contributions
TW, ZJ, YY, and ZH contributed to the study conception and design. Material preparation, data collection and analysis were performed by YZ, JZ, XH, HS, XL, and AW. The first draft of the manuscript was written by YZ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Consent to participate
Written informed consent was obtained from the parents.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, Y., Zhou, J., Hao, X. et al. Efficacy relevance of PD-L1 expression on circulating tumor cells in metastatic breast cancer patients treated with anti-PD-1 immunotherapy. Breast Cancer Res Treat 200, 281–291 (2023). https://doi.org/10.1007/s10549-023-06972-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-023-06972-6