Abstract
PIK3CA mutations occur frequently in breast cancer, predominantly in exons 9 and 20. The aim of this retrospective study is to evaluate the PIK3CA mutation status for its relationship with prognosis and first-line endocrine therapy outcome. PIK3CA exon 9 and 20 were evaluated for mutations in 1,352 primary breast cancer specimens by SnaPshot multiplex analyses. The mutation status was studied for their relationship with metastasis-free survival (MFS) in 342 untreated lymph node-negative (LNN) patients and to time to progression (TTP) in estrogen receptor (ER)-positive patients with metastatic disease treated with first-line tamoxifen (N = 447) or aromatase inhibitors (AIs; N = 84). We detected in 423 patients hotspot mutations for PIK3CA (31 %). Mutations in exon 20 were detected in 251 patients (59 %), with H1047L and H1047R mutations in 37 (15 %) and 214 (85 %) cases, respectively. Mutations in PIK3CA exon 9 were discovered in 173 patients (41 %), with E542K and E545K mutations in 57 (32 %) and 104 (60 %) cases as most prevalent ones. Evaluation of the untreated LNN patients for prognosis showed no relationship between MFS and PIK3CA mutations, neither for exon 9 [HR = 1.04 (95 % CI 0.57–1.89), P = 0.90] nor for exon 20 [HR = 0.98 (95 % CI 0.63–1.54); P = 0.94] when compared to wild-type. The PIK3CA mutation status was also not associated with treatment outcome after first-line tamoxifen. On the other hand, patients treated with first-line AIs showed a longer TTP when having a PIK3CA mutation in exon 9 [HR = 0.40 (95 % CI 0.17–0.95); P = 0.038] or exon 20 [HR = 0.50 (95 % CI 0.27–0.91); P = 0.024] compared to wild-types, both significant in uni- and multivariate analysis including traditional predictive factors. All results remained when only HER2-negative patients were evaluated for each cohort. PIK3CA mutations in ER-positive tumors were significantly associated with a favorable outcome after first-line AIs, which needs further confirmation in other datasets. Mutations were not associated with prognosis in untreated LNN patients nor predictive outcome after first-line tamoxifen therapy in advanced disease patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The phosphatidylinositol-3-kinase (PI3K) pathway regulates several cellular processes critical for cancer progression, i.e., protein synthesis, growth, metabolism, proliferation, cell survival, apoptosis, motility and angiogenesis [1]. Since this pathway is frequently deregulated in breast cancer, it is an attractive pharmacologic target to investigate. The PI3K is a complex of regulatory and catalytic proteins and one of the mechanisms of abnormal PI3K pathway activation is through mutations in the 110 kDa catalytic protein encoded by PIK3CA or in the 85 kDa regulatory protein encoded by PIK3R1. PIK3CA is the most frequent (30 %) mutated oncogene in breast cancer (http://www.sanger.ac.uk/genetics/CGP/cosmic), especially in ER-positive tumors [2, 3]. Approximately 90 % of PIK3CA mutations [4], are clustered in two hotspot regions: exon 9 (E542K and E545K) encoding the helical domain and exon 20 (H1047R and H1047L) encoding the kinase domain [2, 5].
PIK3CA mutations may play an important role in the carcinogenesis and development of breast cancer and has been correlated with clinical and treatment outcome [6]. Several studies presented, however, contradicting results for the relationship between PIK3CA mutation status and clinical outcome. Some studies showed no correlation between PIK3CA mutations and clinicopathologic variables [4, 7], while others have shown a worse prognosis after treatment with HER2-inhibitors for patients with HER2+ tumors and PIK3CA mutations [8, 9]. Moreover, PIK3CA alterations are mainly present in invasive lobular (46 %) and ductal (22 %) breast carcinoma [10]. In lobular tumors, PIK3CA mutations have been found to be associated with tumor size, ER+ tumors and poor survival [6, 10], whereas other histological types with a low incidence of PIK3CA mutations have shown to be associated with a favorable prognosis [10]. Especially tumors bearing exon 20 PIK3CA mutations are related with poor prognosis compared to those having wild type PIK3CA and other PIK3CA mutations [11]. Finally, whole genome DNA analysis of a cohort of breast cancers revealed that PIK3CA mutations occur predominantly in ER-positive breast cancers of the molecular luminal subtype, whereas PI3K-pathway activation was especially observed in ER-negative basal breast cancers [3]. In this study, however, relationships with clinical and treatment outcome were not investigated.
Breast cancer patients with tumors expressing the ER are treated with endocrine therapies, i.e., with tamoxifen or AIs. Unfortunately, not all patients respond (de novo resistance) while in the metastatic setting, patients who do initially respond will eventually relapse (acquired resistance). Endocrine therapy resistance may occur through activation of the PI3K- and MAPK-pathways and/or their downstream targets AKT and mTOR [12–16]. In this respect it is of interest to note that phase III clinical trials showed substantial benefit when mTOR-inhibitors were added to AI treatment [17]. Also for survival after adjuvant tamoxifen therapy contradicting results have been published. Lai et al. [11] showed in a cohort of 152 patients that invasive ductal carcinoma with exon 20 PIK3CA mutation had a significant shorter survival after adjuvant tamoxifen therapy compared to wild type tumors. On the other hand, Loi et al. [18] observed no relation with PIK3CA mutation status but a beneficial outcome after adjuvant tamoxifen therapy when applying a PI3K exon 20 gene expression signature.
To address above contradicting findings, we examined retrospectively the PIK3CA mutations in a cohort of 1,352 breast cancer patients to establish the prognostic and predictive significance of these mutations in tumors of 342 untreated LNN patients as well as 532 ER-positive patients with advanced disease treated with first-line endocrine therapy, i.e., tamoxifen (N = 447) or aromatase inhibitors (N = 84). Moreover, we correlated patient and tumor characteristics and clinical outcome with the PIK3CA mutation status, stratified for helical (exon 9) or kinase (exon 20) domain hotspot mutations, respectively.
Patients
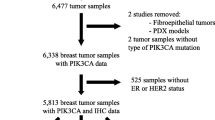
The Erasmus University Medical Center (EMC; N = 1,031), at Rotterdam, the Netherlands Cancer Institute (NKI; N = 159), at Amsterdam, and the Radboud University Nijmegen Medical Centre at Nijmegen (N = 77) all located in the Netherlands, and the Sint Augustinus Hospital at Antwerpen (N = 85) in Belgium participated in this study. Primary breast cancer tissue specimens were collected from 1,352 female patients with primary or advanced breast cancer that entered the hospitals between 1978 and 2007. Patients and tumor characteristics are presented in Table 1 for all patients for which information was available and for the three different patient cohorts analyzed in this study.
Follow-up, tumor staging, and response to therapy was defined by standard International Union against Cancer (Geneva, Switzerland) classification criteria [19]. This retrospective study has been approved by the local medical ethics committees, performed in accordance with the Code of Conduct of the Federation of Medical Scientific Societies in the Netherlands (http://www.fmwv.nl) and Belgium, and reported following the REMARK recommendations [20]. DNA isolated from primary breast tumor specimens were evaluated for PIK3CA exon 9 and exon 20 hotspot mutation status to assess relationships with patient and tumor characteristics and with clinical and treatment outcome. ER, progesterone receptor (PR), and HER2 status of the primary tumor tissue specimens were established as described previously [21–24].
The prognostic value of PIK3CA mutations was assessed in 342 LNN breast cancer patients. These patients had no metastatic disease at time of diagnosis and received no adjuvant systemic therapy. In addition, ER-positive patients with advanced disease treated with first-line tamoxifen (N = 447) or aromatase inhibitors (N = 84) were analyzed to determine the predictive value of PIK3CA mutations. Patients treated with endocrine therapy were selected based on the following inclusion criteria: Invasive ER-positive breast carcinoma; advanced disease deemed not curable by surgery and/or radiotherapy for which first-line tamoxifen or AI therapy had been given for at least 4 weeks; and frozen (N = 1,193) or paraffin-embedded (N = 159) primary tumor specimens were available. Detailed patient characteristics for the cohort of tamoxifen treated patients have been previously described [25]. The cohort of 84 metastatic breast cancer patients treated with first-line AIs received either steroidal (15 exemestane) or non-steroidal AIs (43 anastrozole, 26 letrozole). Nine of these patients presented with metastatic disease at time of diagnosis, 52 patients had modified mastectomy and 23 patients underwent breast-conserving lumpectomy. Sixty-four patients received adjuvant endocrine therapy and 17 patients were treated with adjuvant chemotherapy, however, all developed metastatic disease that was treated with first-line aromatase inhibitors.
Methods
Multiplex PCR amplification and SnaPshot analysis
For the detection of mutations, stored DNA was amplified for exons 9 and 20 of PIK3CA using earlier published PCR primers [26]. The amplified exons were assessed for mutations at the following nucleotide positions (with corresponding amino acid changes) G1624 (E542K, Q), G1633 (E545K, Q), A1634 (E545G, A), A3140 (H1047R, L) using the SnaPshot® multiplex system (Life Technologies) as described previously [26, 27]. All specimens with a mutation or that failed initially, were re-analyzed with the SnaPshot to validate the mutation status of the tumor.
Statistics
The relationship of mutation status with patient and tumor characteristics was investigated using nonparametric methods, i.e., Spearman rank correlations for continuous variables and Wilcoxon rank-sum or Kruskal–Wallis and χ 2 square test for ordered variables. The Cox proportional hazards model was used to compute the hazard ratio (HR) in the analysis of metastasis-free survival (MFS) and time to progression (TTP). MFS was defined as the time elapsed between the surgery and the first distant metastasis. TTP was defined as the time elapsed between initiation of endocrine therapy and the first detection of disease progression. In multivariate analysis, the predictive value of PIK3CA mutation status was compared to the base model of traditional clinicopathological factors for metastatic breast cancer, i.e., age at start of therapy, disease free interval, dominant site of relapse and PR and HER2 status. The HR was presented with its 95 % confidence intervals (95 % CI). Survival curves were generated using the Kaplan–Meier method and a log rank test was used to test for differences. Computations were done with the STATA statistical package, release 12SE (STATA Corp., College Station, TX). All P-values were two-sided and P < 0.05 was considered statistically significant.
Results
PIK3CA mutation frequencies and clinicopathological features
We evaluated the DNA of 1,352 primary breast tumor specimens for hotspot mutations in the PIK3CA gene using SnaPshot multiplex analysis (Fig. 1a). No hotspot mutations for PIK3CA were detected in 928 tumors, here referred to as wild-type, although these tumors may harbor mutations outside the analyzed hotspots. For 423 patients a mutation in exon 9 or exon 20 (31 %) was discovered. Exon 20 mutations were detected in 251 patients (59 %), with a H1047L in 37 (15 %) and a H1047R mutation in 214 (85 %) cases. Mutations in PIK3CA exon 9 were detected in 173 patients (41 %), with E542K and E545K mutations in 57 (32 %) and 104 (60 %) cases, respectively, as the most prevalent ones (Fig. 1b).
PIK3CA mutation frequencies. a The frequencies of PIK3CA exon 9 and 20 mutations as detected in the total cohort and in subsets of breast cancer patients. Mutations occur more frequently in estrogen receptor positive (ER+) compared to ER-negative (ER−) tumors. Lymph node-negative (LNN) breast cancer patients that received no adjuvant systemic therapy (N = 342) were investigated for the prognostic value of the PIK3CA mutation status. ER-positive breast cancer patients with metastatic disease treated with first-line tamoxifen (N = 447) or aromatase inhibitors (N = 84) were evaluated for a relation between PIK3CA mutation status with treatment outcome. WT are patients who have no PIK3CA hotspot mutation in exon 9 or exon 20, defined as wild-type. b The type and number of mutations detected in exon 9 (all starting with E) or in exon 20 (H1047R, H1047L) for the 423 patients with a PIK3CA mutation in their primary tumor
Clinicopathological characteristics in relation with PIK3CA status for all patients and the three distinguished sub-cohorts of patients are shown in Table 1 and in more detail in Supplemental Table S1. PIK3CA mutations, both in exon 9 and 20, are most frequently observed in luminal ER-positive and/or in PR-positive tumors (all P < 0.001), in agreement with observations by others. Moreover, tumors with an exon 9 mutation appeared to be smaller (<2 cm; P = 0.009) and tend to metastasize preferentially to bone (P = 0.071) than those with a wild-type or exon 20 mutant PIK3CA gene. These cohorts included a cohort for prognosis of untreated LNN patients and two cohorts for treatment outcome of ER-positive patients with metastatic disease treated with endocrine therapy. For the prognostic cohort the PIK3CA mutations were predominantly observed in luminal (P = 0.001), ER-positive (P = 0.007), PR-positive (P < 0.001), and HER2-negative (P = 0.041) tumors. In the tamoxifen treated cohort, PIK3CA exon 20 mutations were especially detected in postmenopausal women (P = 0.003), whereas for the AI cohort exon 9 and 20 mutations were overrepresented in PR-positive tumors (P = 0.004). Since HER2 affects prognosis and response to endocrine therapy, survival analyses in the three sub-cohorts below were performed on all tumors (Table 1) and on HER2-negative tumors only (Supplemental Table S2).
PIK3CA mutation status and prognosis
To assess the prognostic value of PIK3CA mutation status, a subset of 342 LNN patients who received no adjuvant systemic therapy have been evaluated for the relation between mutation status and MFS. No significant differences were observed between wild-type and mutated PIK3CA tumors with regard to traditional prognostic factors age and menopausal status at diagnosis, tumor size, and grade. No association between PIK3CA mutation and MFS was found, neither for exon 9 [HR = 1.04 (95 % CI 0.57–1.90); P = 0.90] nor for exon 20 [HR = 0.98 (95 % CI 0.63–1.54); P = 0.94] when compared to wild-type (Fig. 2a). Since mutations occurred predominantly in luminal tumors, the prognostic value was also established in 262 ER-positive LNN patients of this sub-cohort. Again, for these 262 patients, no association between PIK3CA mutation and MFS was found, neither for tumors with exon 9 mutations [HR = 1.03 (95 % CI 0.55–1.93); P = 0.94] nor for tumors with exon 20 mutations [HR = 1.04 (95 % CI 0.65–1.66); P = 0.88] when compared to wild-type tumors. Mutation status and MFS showed also no relationships in the subsets of (ER-positive) HER2-negative tumors only (Supplemental Table S2).
PIK3CA mutation status: Prognosis and treatment outcome after first-line tamoxifen. a MFS analysis in 342 LNN breast cancer patients who had no adjuvant systemic therapy as a function of PIK3CA mutation status. b TTP analysis in 447 ER-positive breast cancer patients with advanced disease treated with first-line tamoxifen as a function of PIK3CA mutation status
PIK3CA mutation status and outcome after first-line tamoxifen therapy
Next, a subset of 447 ER-positive patients with advanced disease treated with first-line tamoxifen has been evaluated for the relationship between PIK3CA mutation status and treatment outcome. No significant differences were observed between PIK3CA wild-type and mutated tumors in relation to traditional predictive factors age at start of tamoxifen treatment, dominant site of relapse, disease-free interval and PR. The tumors with PIK3CA exon 9 and 20 mutations did not associate with TTP after tamoxifen, i.e., for exon 9 [HR = 1.17 (95 % CI 0.87–1.57); P = 0.30] and for exon 20 [HR = 1.01 (95 % CI 0.78–1.31); P = 0.93] (Fig. 2b). The relationship between PIK3CA mutation status and TTP was also not observed in the subset of HER2-negative tumors (Supplemental Table S2). The PIK3CA mutation status showed a relation with menopausal status at start of therapy in this subset (P = 0.003), which was also confirmed by a significant test of interaction (P = 0.010) for exon 20 mutation status and menopausal status. For this reason, the mutation status in relation to TTP was also evaluated independently for pre- and postmenopausal patients. No associations were observed for PIK3CA exon 9 mutations in premenopausal [N = 77; HR = 0.94 (95 % CI 0.50–1.78); P = 0.85] and postmenopausal [N = 369; HR = 1.22 (95 % CI 0.87–1.71); P = 0.25] women. The PIK3CA exon 20 mutations, on the other hand, showed a significant relation with TTP in premenopausal women [HR = 2.59 (95 % CI 1.08–6.25); P = 0.034] but not in postmenopausal women [HR = 1.00 (95 % CI 0.76–1.31); P = 0.99], although exon 20 mutations were especially detected in postmenopausal women.
PIK3CA mutation status and outcome after first-line aromatase inhibitor therapy
Finally, the PIK3CA mutation status was evaluated for a relationship with treatment outcome in a set of 84 ER-positive patients with advanced disease treated with first-line AIs. The PR status in this cohort was the only traditional predictive factor for metastatic disease that linked to the PIK3CA exon 9 and 20 mutation status (P = 0.004). Interestingly, patients with a PIK3CA mutation when compared with those being wild-type had a prolonged TTP after AI treatment, both for exon 9 [HR = 0.40 (95 % CI 0.17–0.95); P = 0.038] as well as for exon 20 [HR = 0.50 (95 % CI 0.27–0.91); P = 0.024] mutations (Fig. 3). These associations between mutation status and TTP were still significant when analyzed in the subset of HER2-negative tumors (Supplemental Table S2). An explanatory analysis in only patients treated with non-steroidal AIs (N = 69) demonstrated that the relation between PIK3CA mutation and prolonged TTP kept significant, i.e., for exon 9 [HR = 0.27 (95 % CI 0.08–0.88); P = 0.030] and for exon 20 [HR = 0.45 (95 % CI 0.23–0.87); P = 0.019]. The subset of steroidal AI treated patients (N = 15) was too small to draw conclusions. In multivariate analysis including age, disease-free interval, dominant site of relapse and PR and HER2 status as traditional predictive factors, the association with AI treatment outcome remained significant for both exon 9 and exon 20 mutants (see Table 2). These significant preliminary findings, however, need further validation in additional larger datasets.
Discussion
The purpose of this study was to evaluate PIK3CA exon 9 and exon 20 hotspot mutations in 1,352 primary breast tumor tissue specimens and to associate the mutation status with clinicopathological characteristics and with clinical outcome. Using SnaPshot analyses we identified PIK3CA mutations in 31 % of the patients with 13 % mutations in exon 9 and 18 % mutations in exon 20. The mutations were mainly detected in the ER-positive tumors (35 %) compared to the triple-negative tumors (11 %) (Supplemental Table S1). Moreover, fewer mutated tumors were observed for patients with LRR (29 %) compared to those patients with bone metastasis (40 %; N = 356, P = 0.056) and to those patients with bone or other distant metastases (35 %: N = 696, P = 0.17). The frequency and distribution of PIK3CA hotspot mutations are comparable to those reported (Supplementary Figure S1) [3].
We evaluated exon 9 (helical) and exon 20 (kinase) domain PIK3CA mutations separately since exon 9 mutations have been reported mainly in lobular carcinomas and to associate with poor prognosis [28] and AKT-independent downstream signaling [29]. Although exon 9 mutations occur less frequent than exon 20 mutations, no significant differences were observed for almost all clinicopathological characteristics and clinical outcome in the sub-cohorts when stratified for PIK3CA exon 9 and 20 mutation status. We only observed in ER-positive tumors that PIK3CA exon 9 mutated tumors showed a trend to metastasize more often to bone and are smaller when compared to wild-type or exon 20 mutant tumors.
The PIK3CA mutant tumors in our analyses are mainly of the luminal subtype and PR-positive (80–83 %) in contrast to only 65 % PR-positive wild-type tumors. Other studies observed also significantly more PR-positive tumors in the PIK3CA mutant tumor group compared to the PIK3CA wild-type group, i.e., in a meta-analysis in 252 of the 333 mutant tumors (76 %) versus 374 of the 708 wild-type tumors (53 %) are PR-positive [5, 30, 31], respectively. However, presence of PR recently has shown to be only prognostic but not predictive for adjuvant tamoxifen therapy [32]. Moreover, the uni- and multivariate analyses for TTP after AI treatment in our cohort revealed no significant relationship between PR and outcome and PIK3CA mutation status remained independently from PR predictive for response to AI. In line with this, whole-genome analysis of breast cancers of patients treated with AI in the neoadjuvant setting showed that mutations in the PI3K-pathway mainly occur in luminal A breast tumors with low recurrence risk [33]. Additionally, invasive ductal and lobular carcinomas did not differ in PIK3CA mutation frequencies and distributions in all our specimens (Supplemental Table S1).
Our study shows that patients with a PIK3CA mutation in their tumors do have a favorable outcome on first-line AI therapy, whereas PIK3CA mutations are not prognostic nor related to first-line tamoxifen outcome. These findings are unexpected since somatic mutations in PIK3CA have been shown to activate AKT and induce oncogenic transformation of breast cancer cells [34] in vitro [35] and in vivo [36]. Additionally, in vitro studies demonstrated that resistance to endocrine therapy might be due to activation of the PI3K-pathway and/or it downstream targets AKT and mTOR [13, 15, 37]. Moreover, recent phase II and III clinical trials of metastatic breast cancer patients treated with tamoxifen or exemestane showed benefit from addition of everolimus, an mTOR-inhibitor, further stressing the role of the PI3K-pathway activation in endocrine resistance [17, 38]. On the other hand, no improve in progression free survival after first-line letrozole plus temsirolimus, also an inhibitor of mTOR, was observed in a randomized phase III trial of postmenopausal metastatic breast cancer patients [39]. Additionally, mutations in the kinase domain of PIK3CA were associated with favorable relapse-free survival and weakly with clinical response in a neoadjuvant endocrine therapy trial [40]. Finally, comprehensive reviews of a large number of clinical studies regarding PIK3CA mutations in breast carcinomas [2, 41] indicate that PIK3CA mutations are associated with favorable prognosis in ER-positive breast cancer. All these studies, however, included lymph node-positive and/or (neo) adjuvant treated patients, which may explain why we could not confirm this association with prognosis in our study on untreated LNN patients. The contradicting findings might also be explained by recent views that PIK3CA mutations not always translate into a downstream activated PI3K pathway [3]. A system biological approach was applied in this study to reveal a mechanism of action in clinical samples, which showed that while PIK3CA mutations are predominantly present in luminal breast tumors the PI3K pathway activation mainly occurs in basal-like tumors. Luminal A PIK3CA mutated tumors in this study were low in PI3K pathway activation markers pAKT, pS6 and p4EBP1 and showed less PI3K-gene signature activity when compared to basal PIK3CA wild-type tumors. Analyses of down-stream PI3K pathway activation might be informative in our sub-cohorts for prognosis and tamoxifen, since no relationship with PIK3CA mutation status was observed for these cohorts, however, a system biological approach on these two cohorts was not applicable because genome wide RNA and/or protein data were not available.
We investigated primary tumor specimens and related their PIK3CA status with treatment outcome for advanced disease, nevertheless, it has been shown that the PIK3CA status can differ between primary tumor and metastatic lesions [42]. Biopsies of metastatic lesions, however, are often difficult to obtain due to their localization. Additionally, serum/plasma-derived circulating free DNA (cfDNA) from a subset of metastatic breast cancer patients contained PIK3CA mutations whereas no mutations were detected in cfDNA from patients with localized breast cancer [43]. These studies also identified wild-type primary tumors with matched metastatic lesions or cfDNAs that harbored PIK3CA mutations [42, 43]. All this indicates that primary disease may differ sometimes from metastatic disease with regard to PIK3CA status. It may affect slightly our findings and might explain partially differences between our observations in metastatic disease from those obtained by others in the adjuvant setting [11, 18].
Interestingly, we found that patients with a PIK3CA mutation in primary tumors have longer TTP after AI therapy when compared to wild-type tumors whereas there is no association with treatment outcome after tamoxifen treatment. The association with TTP after aromatase inhibitor therapy remained significant in the subset of patients treated with non-steroidal AI (letrozole, anastrozole) and after multivariate analysis of the PIK3CA mutation status together with the traditional predictive factors. The discrepancy in PIK3CA mutation status relationship with treatment outcome after AI and tamoxifen therapy may be of relevance for the choice of treatment of ER-positive breast cancer patients. This discrepancy needs further evaluation since the AI-cohort is relatively small and because these sub-cohorts were not controlled for differences that could affect response to tamoxifen or AI for metastatic disease due to the retrospective design of the study. Our analyses indicate, however, that HER2 status does not affect the relationships between mutation status and outcome after AI and tamoxifen. Moreover, none of metastatic breast cancer patients treated with tamoxifen whereas 64 patients treated with AI received adjuvant endocrine therapy, but Cox regression analyses for adjuvant tamoxifen in the AI-cohort revealed no significant relationship with TTP [HR = 1.66, (95 % CI 0.95–2.89); P = 0.074]. Based on these observations we do not believe that adjuvant endocrine therapy could explain the differences in response for metastatic disease for our analyzed cohorts of patients.
In a neoadjuvant study on ER-positive breast tumors, it has been reported that everolimus increases the efficacy of letrozole [44]. Tumors with exon 20 PIK3CA mutations in this study showed less proliferation based upon Ki67-measurements after letrozole therapy compared to wild-type tumors and tumors carrying exon 9 mutations. Unfortunately, for analyses of overall survival, patients were not stratified for PIK3CA mutation status. Endocrine therapy combined with mTOR inhibitors, such as everolimus, has also been studied in cell line models. Cell lines harboring the PIK3CA mutation are sensitive to this combined therapy, however, cell lines with an active PI3K pathway due to PTEN deletions or activating mutations in KRAS or BRAF have no additional benefit from this combined therapy [45, 46]. Further studies are needed to evaluate treatment opportunities for ER-positive breast cancer patients with and without a PIK3CA mutation and PI3K/AKT/mTOR pathway activation.
Conclusions
This study is the first to report that metastatic breast cancer patients with PIK3CA mutations in ER-positive primary tumors show favorable outcome after first-line aromatase inhibitor treatment. This significant preliminary association should be verified in randomized prospective clinical trials to establish predictive significance [47]. Moreover, PIK3CA mutations in luminal ER-positive tumors have no prognostic value and are not predictive for first-line tamoxifen treatment.
References
Vivanco I, Sawyers CL (2002) The phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat Rev Cancer 2(7):489–501
Troxell ML (2012) PIK3CA/AKT1 mutations in breast carcinoma: a comprehensive review of experimental and clinical studies. J Clinic Experiment Pathol S1:002
Cancer Genome Atlas N (2012) Comprehensive molecular portraits of human breast tumours. Nature 490(7418):61–70
Campbell IG, Russell SE, Choong DY, Montgomery KG, Ciavarella ML, Hooi CS, Cristiano BE, Pearson RB, Phillips WA (2004) Mutation of the PIK3CA gene in ovarian and breast cancer. Cancer Res 64(21):7678–7681
Kalinsky K, Jacks LM, Heguy A, Patil S, Drobnjak M, Bhanot UK, Hedvat CV, Traina TA, Solit D, Gerald W et al (2009) PIK3CA mutation associates with improved outcome in breast cancer. Clin Cancer Res 15(16):5049–5059
Li SY, Rong M, Grieu F, Iacopetta B (2006) PIK3CA mutations in breast cancer are associated with poor outcome. Breast Cancer Res Treat 96(1):91–95
Dunlap J, Le C, Shukla A, Patterson J, Presnell A, Heinrich MC, Corless CL, Troxell ML (2010) Phosphatidylinositol-3-kinase and AKT1 mutations occur early in breast carcinoma. Breast Cancer Res Treat 120(2):409–418
Dave B, Migliaccio I, Gutierrez MC, Wu MF, Chamness GC, Wong H, Narasanna A, Chakrabarty A, Hilsenbeck SG, Huang J et al (2011) Loss of phosphatase and tensin homolog or phosphoinositol-3 kinase activation and response to trastuzumab or lapatinib in human epidermal growth factor receptor 2-overexpressing locally advanced breast cancers. J Clin Oncol 29(2):166–173
Esteva FJ, Guo H, Zhang S, Santa-Maria C, Stone S, Lanchbury JS, Sahin AA, Hortobagyi GN, Yu D (2010) PTEN, PIK3CA, p-AKT, and p-p70S6K status: association with trastuzumab response and survival in patients with HER2-positive metastatic breast cancer. Am J Pathol 177(4):1647–1656
Buttitta F, Felicioni L, Barassi F, Martella C, Paolizzi D, Fresu G, Salvatore S, Cuccurullo F, Mezzetti A, Campani D et al (2006) PIK3CA mutation and histological type in breast carcinoma: high frequency of mutations in lobular carcinoma. J Pathol 208(3):350–355
Lai YL, Mau BL, Cheng WH, Chen HM, Chiu HH, Tzen CY (2008) PIK3CA exon 20 mutation is independently associated with a poor prognosis in breast cancer patients. Ann Surg Oncol 15(4):1064–1069
Musgrove EA, Sutherland RL (2009) Biological determinants of endocrine resistance in breast cancer. Nat Rev Cancer 9(9):631–643
Sun M, Paciga JE, Feldman RI, Yuan Z, Coppola D, Lu YY, Shelley SA, Nicosia SV, Cheng JQ (2001) Phosphatidylinositol-3-OH kinase (PI3K)/AKT2, activated in breast cancer, regulates and is induced by estrogen receptor alpha (ERalpha) via interaction between ERalpha and PI3K. Cancer Res 61(16):5985–5991
Stemke-Hale K, Gonzalez-Angulo AM, Lluch A, Neve RM, Kuo WL, Davies M, Carey M, Hu Z, Guan Y, Sahin A et al (2008) An integrative genomic and proteomic analysis of PIK3CA, PTEN, and AKT mutations in breast cancer. Cancer Res 68(15):6084–6091
Al Saleh S, Sharaf LH, Luqmani YA (2011) Signalling pathways involved in endocrine resistance in breast cancer and associations with epithelial to mesenchymal transition (Review). Int J Oncol 38(5):1197–1217
Miller TW, Perez-Torres M, Narasanna A, Guix M, Stal O, Perez-Tenorio G, Gonzalez-Angulo AM, Hennessy BT, Mills GB, Kennedy JP et al (2009) Loss of phosphatase and tensin homologue deleted on chromosome 10 engages ErbB3 and insulin-like growth factor-I receptor signaling to promote antiestrogen resistance in breast cancer. Cancer Res 69(10):4192–4201
Baselga J, Campone M, Piccart M, Burris HA 3rd, Rugo HS, Sahmoud T, Noguchi S, Gnant M, Pritchard KI, Lebrun F et al (2012) Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N Engl J Med 366(6):520–529
Loi S, Haibe-Kains B, Majjaj S, Lallemand F, Durbecq V, Larsimont D, Gonzalez-Angulo AM, Pusztai L, Symmans WF, Bardelli A et al (2010) PIK3CA mutations associated with gene signature of low mTORC1 signaling and better outcomes in estrogen receptor-positive breast cancer. Proc Natl Acad Sci USA 107(22):10208–10213
Hayward JL, Rubens RD, Carbone PP, Heuson JC, Kumaoka S, Segaloff A (1978) Assessment of response to therapy in advanced breast cancer. A project of the programme on clinical oncology of the International Union against Cancer, Geneva, Switzerland. Eur J Cancer 14(11):1291–1292
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM, Statistics Subcommittee of NCIEWGoCD (2006) REporting recommendations for tumor MARKer prognostic studies (REMARK). Breast Cancer Res Treat 100(2):229–235
Foekens JA, Portengen H, van Putten WL, Trapman AM, Reubi JC, Alexieva-Figusch J, Klijn JG (1989) Prognostic value of receptors for insulin-like growth factor 1, somatostatin, and epidermal growth factor in human breast cancer. Cancer Res 49(24 Pt 1):7002–7009
Berns EM, Klijn JG, van Staveren IL, Portengen H, Noordegraaf E, Foekens JA (1992) Prevalence of amplification of the oncogenes c-myc, HER2/neu, and int-2 in one thousand human breast tumours: correlation with steroid receptors. Eur J Cancer 28(2–3):697–700
Roepman P, Horlings HM, Krijgsman O, Kok M, Bueno-de-Mesquita JM, Bender R, Linn SC, Glas AM, van de Vijver MJ (2009) Microarray-based determination of estrogen receptor, progesterone receptor, and HER2 receptor status in breast cancer. Clin Cancer Res 15(22):7003–7011
Nguyen B, Cusumano PG, Deck K, Kerlin D, Garcia AA, Barone JL, Rivera E, Yao K, de Snoo FA, van den Akker J et al (2012) Comparison of molecular subtyping with BluePrint, MammaPrint, and TargetPrint to local clinical subtyping in breast cancer patients. Ann Surg Oncol 19(10):3257–3263
van Schaik RH, Kok M, Sweep FC, van Vliet M, van Fessem M, Meijer-van Gelder ME, Seynaeve C, Lindemans J, Wesseling J, Van ‘t Veer LJ et al (2011) The CYP2C19*2 genotype predicts tamoxifen treatment outcome in advanced breast cancer patients. Pharmacogenomics 12(8):1137–1146
Lurkin I, Stoehr R, Hurst CD, van Tilborg AA, Knowles MA, Hartmann A, Zwarthoff EC (2010) Two multiplex assays that simultaneously identify 22 possible mutation sites in the KRAS, BRAF, NRAS and PIK3CA genes. PLoS ONE 5(1):e8802
Kompier LC, Lurkin I, van der Aa MN, van Rhijn BW, van der Kwast TH, Zwarthoff EC (2010) FGFR3, HRAS, KRAS, NRAS and PIK3CA mutations in bladder cancer and their potential as biomarkers for surveillance and therapy. PLoS ONE 5(11):e13821
Barbareschi M, Buttitta F, Felicioni L, Cotrupi S, Barassi F, Del Grammastro M, Ferro A, Dalla Palma P, Galligioni E, Marchetti A (2007) Different prognostic roles of mutations in the helical and kinase domains of the PIK3CA gene in breast carcinomas. Clin Cancer Res 13(20):6064–6069
Vasudevan KM, Barbie DA, Davies MA, Rabinovsky R, McNear CJ, Kim JJ, Hennessy BT, Tseng H, Pochanard P, Kim SY et al (2009) AKT-independent signaling downstream of oncogenic PIK3CA mutations in human cancer. Cancer Cell 16(1):21–32
Cizkova M, Susini A, Vacher S, Cizeron-Clairac G, Andrieu C, Driouch K, Fourme E, Lidereau R, Bieche I (2012) PIK3CA mutation impact on survival in breast cancer patients and in ERalpha, PR and ERBB2-based subgroups. Breast Cancer Res 14(1):R28
Boyault S, Drouet Y, Navarro C, Bachelot T, Lasset C, Treilleux I, Tabone E, Puisieux A, Wang Q (2012) Mutational characterization of individual breast tumors: TP53 and PI3K pathway genes are frequently and distinctively mutated in different subtypes. Breast Cancer Res Treat 132(1):29–39
Davies C, Godwin J, Gray R, Clarke M, Cutter D, Darby S, McGale P, Pan HC, Taylor C, Early Breast Cancer Trialists’ Collaborative G et al (2011) Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet 378(9793):771–784
Ellis MJ, Ding L, Shen D, Luo J, Suman VJ, Wallis JW, Van Tine BA, Hoog J, Goiffon RJ, Goldstein TC et al (2012) Whole-genome analysis informs breast cancer response to aromatase inhibition. Nature 486(7403):353–360
Isakoff SJ, Engelman JA, Irie HY, Luo J, Brachmann SM, Pearline RV, Cantley LC, Brugge JS (2005) Breast cancer-associated PIK3CA mutations are oncogenic in mammary epithelial cells. Cancer Res 65(23):10992–11000
Kang S, Bader AG, Vogt PK (2005) Phosphatidylinositol 3-kinase mutations identified in human cancer are oncogenic. Proc Natl Acad Sci USA 102(3):802–807
Bader AG, Kang S, Vogt PK (2006) Cancer-specific mutations in PIK3CA are oncogenic in vivo. Proc Natl Acad Sci USA 103(5):1475–1479
Zhang Y, Moerkens M, Ramaiahgari S, de Bont H, Price L, Meerman J, van de Water B (2011) Elevated insulin-like growth factor 1 receptor signaling induces antiestrogen resistance through the MAPK/ERK and PI3K/Akt signaling routes. Breast Cancer Res 13(3):R52
Bachelot T, Bourgier C, Cropet C, Ray-Coquard I, Ferrero JM, Freyer G, Abadie-Lacourtoisie S, Eymard JC, Debled M, Spaeth D et al (2012) Randomized phase II trial of everolimus in combination with tamoxifen in patients with hormone receptor-positive, human epidermal growth factor receptor 2-negative metastatic breast cancer with prior exposure to aromatase inhibitors: a GINECO study. J Clin Oncol 30(22):2718–2724
Wolff AC, Lazar AA, Bondarenko I, Garin AM, Brincat S, Chow L, Sun Y, Neskovic-Konstantinovic Z, Guimaraes RC, Fumoleau P et al (2013) Randomized phase III placebo-controlled trial of letrozole plus oral temsirolimus as first-line endocrine therapy in postmenopausal women with locally advanced or metastatic breast cancer. J Clin Oncol 31(2):195–202
Ellis MJ, Lin L, Crowder R, Tao Y, Hoog J, Snider J, Davies S, DeSchryver K, Evans DB, Steinseifer J et al (2010) Phosphatidyl-inositol-3-kinase alpha catalytic subunit mutation and response to neoadjuvant endocrine therapy for estrogen receptor positive breast cancer. Breast Cancer Res Treat 119(2):379–390
Dumont AG, Dumont SN, Trent JC (2012) The favorable impact of PIK3CA mutations on survival: an analysis of 2587 patients with breast cancer. Chin J Cancer 31(7):327–334
Dupont Jensen J, Laenkholm AV, Knoop A, Ewertz M, Bandaru R, Liu W, Hackl W, Barrett JC, Gardner H (2011) PIK3CA mutations may be discordant between primary and corresponding metastatic disease in breast cancer. Clin Cancer Res 17(4):667–677
Board RE, Wardley AM, Dixon JM, Armstrong AC, Howell S, Renshaw L, Donald E, Greystoke A, Ranson M, Hughes A et al (2010) Detection of PIK3CA mutations in circulating free DNA in patients with breast cancer. Breast Cancer Res Treat 120(2):461–467
Baselga J, Semiglazov V, van Dam P, Manikhas A, Bellet M, Mayordomo J, Campone M, Kubista E, Greil R, Bianchi G et al (2009) Phase II randomized study of neoadjuvant everolimus plus letrozole compared with placebo plus letrozole in patients with estrogen receptor-positive breast cancer. J Clin Oncol 27(16):2630–2637
Meric-Bernstam F, Akcakanat A, Chen H, Do KA, Sangai T, Adkins F, Gonzalez-Angulo AM, Rashid A, Crosby K, Dong M et al (2012) PIK3CA/PTEN mutations and Akt activation as markers of sensitivity to allosteric mTOR inhibitors. Clin Cancer Res 18(6):1777–1789
Mohseni M, Park BH (2010) PIK3CA and KRAS mutations predict for response to everolimus therapy: now that’s RAD001. J Clin Invest 120(8):2655–2658
Beelen K, Zwart W, Linn SC (2012) Can predictive biomarkers in breast cancer guide adjuvant endocrine therapy? Nat Rev Clin Oncol 9(9):529–541
Acknowledgments
We are especially grateful to Ellen Zwarthof, Anieta Sieuwerts, Anne van Galen, Marion Meijer-van Gelder, Mieke Timmermans, Vanja de Weerd, Cesar Payan for their continued contribution and technical support. DR is a recipient of the ERACOL program, which gives her the opportunity to perform this research in the Netherlands, MJ and EB are funded in part by the TI Pharma projects T3-108 and T3-502.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ramirez-Ardila, D.E., Helmijr, J.C., Look, M.P. et al. Hotspot mutations in PIK3CA associate with first-line treatment outcome for aromatase inhibitors but not for tamoxifen. Breast Cancer Res Treat 139, 39–49 (2013). https://doi.org/10.1007/s10549-013-2529-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-013-2529-7