Abstract
The annual fixation of dissolved Si (DSi) into terrestrial vegetation has been estimated to range from 60 to 200 Tmole, or 10–40 times more than the yearly export of DSi and biogenic Si (BSi) from the terrestrial geobiosphere to the coastal zone. Ecosystems form a large filter between primary mobilization of DSi from silicate weathering and its eventual export to the oceans, and a large reservoir of BSi accumulates in aquatic and terrestrial ecosystems. Although a number of synthesis activities within the last decade have discussed biological transformations in the terrestrial Si cycle, the timescales at which BSi is stored and recycled within ecosystems, BSi persistence and reactivity throughout soil profiles, the dependence of the BSi storage and recycling on ecological processes, the feedbacks to hydrology, the interaction with man’s activities and ultimately the global relevance in Si budgets are poorly constrained. Here we discuss 5 key controls on the ability of ecosystems to filter and control the export of DSi: ecosystem biodiversity, BSi dissolution rates and reactivity, hydrology, interaction with the geosphere and anthropogenic impacts. These controls need to be further studied to better quantify the global and local importance of the terrestrial biogeochemical Si cycle and specifically the BSi reservoir in ecosystems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Traditionally transport of dissolved Si (DSi) from the continents through ecosystems to rivers and eventually the coastal zone has been considered a strict geochemical process controlled solely by the chemical weathering of silicate minerals (e.g. White and Blum 1995). Under this assumption the eventual export of DSi to the oceans is controlled by the relatively slow weathering processes involving primary and secondary silicate minerals. However, this assumption of steady-state is violated by the important role of vegetation in terrestrial ecosystems (Bartoli 1983). The annual fixation of DSi into terrestrial vegetation has been estimated to range from 60 to 200 Tmole, or 10–40 times more than the yearly export of DSi and suspended biogenic Si from the terrestrial geobiosphere to the coastal zone (Conley 2002). Ecosystems thus form a potentially large filter between primary mobilization of DSi from weathering and eventual export to the oceans. Conley (2002) hypothesized that biological recycling of DSi is the main driver determining the size of continental Si fluxes. The amorphous form in which Si occurs in biota (biogenic Si, further referred to as BSi) is several orders of magnitude more susceptible to dissolution than mineral silicates (Van Cappellen 2003). Recent budget studies on basaltic watersheds in Hawaii (Derry et al. 2005), in forested watersheds (Fulweiler and Nixon 2005), in forests (Gérard et al. 2008) and boreal wetlands (Struyf et al. 2010a) have confirmed the biologically controlled export of DSi from the continents, with 30–90% of all DSi fluxes linked to ecosystem BSi pools.
Although a number of synthesis activities within the last decade have discussed biological transformations in the terrestrial Si cycle (Clarke 2003; Conley et al. 2006; Sommer et al. 2006; Street-Perrott and Barker 2008; Struyf and Conley 2009; Struyf et al. 2010b), the timescales associated with BSi storage and recycling of Si in soils, the persistence and reactivity of BSi throughout the soil profile, the dependence of storage on ecological processes, the feedbacks to hydrology, the interaction with man’s activities and ultimately the global relevance of BSi storage and recycling in Si budgets are poorly constrained.
Here we aim to identify the major gaps in our understanding of biogeochemical Si processing in terrestrial ecosystems. Through identification of key controls in the biogeochemical Si cycle, we hope to inspire research focusing on the intricate interactions between ecological, biological, chemical and physical processes controlling the global Si cycle. The impact of ecosystems on weathering intensity of silicate minerals, the role of ecosystems as filters in continental Si transport and the relative magnitude of the biologically controlled Si cycle compared to basic weathering should be a focus of future research efforts. We feel this can be achieved by addressing 5 key controls on ecosystem BSi cycling: ecosystem biodiversity, BSi dissolution rates and reactivity, hydrology, interaction with the geosphere and anthropogenic impacts. This is not purely an academic question. The Si cycle is closely connected to the carbon cycle (Street-Perrott and Barker 2008). Mineral weathering of silicates is an important sink for atmospheric CO2: incomplete knowledge of the ecosystem Si filter impedes the accurate quantification of this sink. Moreover, the import of Si into coastal zones from the terrestrial environment is essential to sustain diatom growth. Diatoms play a key role in the oceanic C-sink and eutrophication of coastal zones (Rabosky and Sorhannus 2009).
The key controls
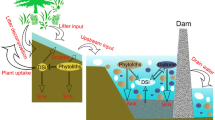
Nutrient cycling in soils is controlled by four key processes: external nutrient input, nutrient export, internal nutrient translocation and nutrient transformations (e.g. Sommer et al. 2006). Mineral weathering of primary and secondary silicates constitutes the prime external source of DSi to ecosystems (Fig. 1). Plant uptake translocates DSi to BSi and is a key factor determining the uptake into the ecosystem Si reservoir. Other important biogenic pools are diatoms, sponges and testate amoebae (Clarke 2003; Aoki et al. 2007). We will discuss the factors that current research indicates are the main controllers of the processes occurring within the ecosystem BSi reservoir; these factors will ultimately determine its importance in global Si cycling. We have not incorporated climate as a specific factor, although the impact of climate will be indirect through the key factors we identified. Climate will impact hydrology, ecosystem diversity, dissolution rates (through pH and temperature) and silicate mineral weathering. Understanding the effect of climate on the ecosystem Si filter will therefore require a thorough understanding of the effect of the key factors.
Key aspects in the ecosystem BSi filter represented in a generalized ecosystem soil profile. In soils, ASi consists of both BSi and pedogenic ASi (see “Dissolution rates and reactivity” section). Key aspects as identified in the manuscript text are presented. Full arrows indicate hydrology related fluxes, dotted arrows represent impacts
Ecosystem biodiversity
We hypothesize that the magnitude and the size of the ecosystem Si reservoir will be strongly dependent on the occurrence of organisms specialized in the biological processing of Si. This may sound obvious, but surprisingly few studies have actually focused on biological Si storage and cycling at the ecosystem scale. Strong variability exists in plant biomass Si concentrations (Hodson et al. 2005). BSi concentrations in aboveground biomass can impact directly on the BSi storage in ecosystem soils (e.g. Struyf et al. 2009). Other Si accumulating organisms also impact soil BSi content, especially sponges and diatoms in wetlands and in wet soils (Struyf and Conley 2009) and testate amoebae in forest soils and wetlands (Cary et al. 2005; Aoki et al. 2007). Increased diatom abundance during periods characterized by a transition in vegetation in Northern peatlands led to pronounced storage peaks of BSi in soil profiles (Kokfelt et al. 2009). Quantification of BSi (or amorphous Si, ASi, see “Dissolution rates and reactivity” section) storage on the ecosystem scale is needed, but has been performed in only a small number of studies (Table 1), e.g. forest systems (e.g. Bartoli 1983; Conley 2002; Gérard et al. 2008), a limited number of grasslands (Blecker et al. 2006) and a small set of wetland types (Struyf and Conley 2009). Recent research has also indicated the capacity of wetland and aquatic macrophyte communities to store BSi (Struyf et al. 2005; Schoelynck et al. 2010).
Ecosystem Si storage can directly impact watershed scale Si fluxes. A rare case study was described for an African mountain lake where the development of high biomass vegetation dominated by grasses led to decreased DSi fluxes from the surrounding catchment into the lake, while nutrient poor vegetation in the catchments led to higher input of DSi into the lake (Street-Perrott et al. 2008). A similar observation was made in a Northern Sweden peatland-lake continuum (Kokfelt et al. 2010).
Quantitative mass-balance studies are necessary to improve our estimates of BSi storage and cycling within terrestrial ecosystems. However, the focus should now also shift to the impact of ecological processes on local BSi storage. We hypothesize that research focusing on connections between ecosystem biodiversity and the ecosystem BSi reservoir could well reveal that species’ effects can be more important than abiotic factors, e.g. weathering and climate, in controlling ecosystem BSi storage, as was previously observed for other nutrients (Hobbie 1992). We hypothesize that the increased storage of BSi in soils will favour dominance of Si accumulating plants. BSi concentrations in aboveground biomass can impact directly on BSi storage in ecosystem soils (e.g. Struyf et al. 2009). Accumulation of BSi in soils will increase DSi availability in soil water (Farmer et al. 2005; Struyf et al. 2009), due to the high solubility of BSi. Si-accumulators’ ability to concentrate BSi in soils effectively engineers the accumulators’ environment and increases their potential fitness, by creating a soil solution rich in a key nutrient. Uptake of Si by grasses and other accumulators positively impacts their competitive strength, and provides protection against several stress factors such as pollution, drought and physical damage (Richmond and Sussman 2003; Epstein 2009). Wetlands are a good example; they are rich in grasses and sedges and their occurrence is positively linked to BSi storage and DSi availability in soils (Struyf et al. 2009). A similar mechanism could be true for grass tussocks where tussocks potentially alter Si availability for grasses by recycling of BSi to DSi even before reaching the soil profile (as observed for other nutrients, e.g. Jonasson and Chapin 1991). Grasses are well-known to exert a strong influence on cycling and storage of other nutrients in soils, with impact greater than expected based on their biomass contribution to communities (McLaren and Turkington 2010).
Another link between ecosystem engineering and Si accumulation in plants has recently been shown for the aquatic species Nuphar lutea (spatterdock) (Schoelynck et al. submitted). In this species, the underwater leaves create a low-dynamic environment ideal for the effective floating of the surficial photosynthesizing leaves. BSi storage in the underwater shoots and leaves is highest, when water current is highest. Si is thus directly stored to increase resistance to physical damage and to create a suitable living environment. This raises questions whether other ecological adaptations might also alter Si storage in ecosystems. Studies focusing on ecological adaptations and BSi storage, will surely lead to the discovery of BSi storage gradients which are currently unaccounted for.
Patterns in plant BSi storage can also have a significant impact on ecosystem functioning. Grasses are well-known to employ Si-based defences against herbivores (McNaughton and Tarrants 1983; Massey et al. 2007). Recent work in Kielder Forest in northern England indicates that BSi defences in grasses play a key role in field vole (Microtus agrestis) populations (Massey et al. 2008). Grass-feeding mammals such as voles show dramatic population cycles which population ecologists have tried to understand for decades. Massey et al. (2008) now show that declining vole populations were associated with high BSi content in the grass species Deschampsia caespitose (tufted hair-grass), their winter food plant, while increasing vole populations occurred when BSi content in the grass was low. This is a dramatic example of how BSi content in vegetation serves as an ecological adaptation, and how changing environmental pressures may thus strongly alter Si uptake in the ecosystem BSi reservoir.
Dissolution rate and reactivity
Knowledge regarding the variability of BSi reactivity through soil profiles and across ecosystems is key to understanding the functioning of the ecosystem BSi reservoir, and should receive immediate attention from biogeochemists. Numerous BSi dissolution studies have been conducted for coastal, estuarine and oceanic diatom BSi in particulate matter and sediments, highlighting the importance of ambient DSi-concentrations and of external factors (pH, salinity) (e.g. Yamada and D’Elia 1984; Van Cappellen et al. 2002; Truesdale et al. 2005; Moriceau et al. 2007; Loucaides et al. 2008) on BSi reactivity. Despite all these processes affecting dissolution and preservation rates of BSi, BSi reactivity in terrestrial samples remains literally unstudied. This is partly attributable to the fact that terrestrial BSi storage is currently studied using simple alkaline extraction methods (DeMaster 1981; Saccone et al. 2007) or estimated from phytolith extraction (Alexandre et al. 1997), providing little insight in its composition and reactivity. Yet, as in the coastal environment, one expects a strong effect of ambient variables (temperature, pH) on the dissolution of terrestrial BSi. Studies combining quantification of BSi dissolution rates and its microscopic identification will potentially link key control one, ecosystem biodiversity, to key control two, reactivity.
We hypothesize that large reactivity gradients for BSi exist across ecosystems, but also within ecosystems. BSi is deposited by a diverse collection of organisms including diatoms, sponges, testate amoebae and plants (as phytoliths). Experimental studies have shown that structural changes to the BSi can affect the dissolution rate, and this directly affects its recycling and preservation in earth surface environments (Loucaides et al. 2010). Aluminum substitution into the surface matrix of BSi increases its preservation efficiency (Van Cappellen et al. 2002) and is an important mechanism for preservation of BSi in marine sediments. There is also a range of pedogenic amorphous materials in soils (Conley et al. 2006) ranging from allophanes, imogolites to clay minerals that differ greatly in their structure and reactivity (Sauer et al. 2006; Sommer et al. 2006). These can interfere with extraction procedures and BSi has been referred to as ASi (amorphous Si, the sum of biogenic amorphous Si and pedogenic amorphous Si). It is indeed important to realize that current alkaline extraction techniques are unable to distinguish such non-biogenic ASi fractions from BSi (Saccone et al. 2007) and non-biogenic fractions could be a substantial component of extracted Si in some soils where non-biological ASi fractions dominate (Lee and Gilkes 2005). A reliable, yet easily performed method that could distinguish between both BSi and pedogenic ASi fractions, would be a great asset to our understanding of the ecosystem BSi reservoir.
It is also likely that micro-organisms will impact the dissolution and/or the retention of BSi in ecosystem soils. Microbial communities influence a range of different soil functions, including organic matter decomposition (Nannipieri et al. 2003), nitrogen fixation, carbon fixation and related trace gas fluxes (Conrad 1996). Bacterial microfilms have been associated with the formation of secondary Fe- and Al-silicates (Konhauser and Urrutia 1999) and bacteria are well-known to impact diatom BSi dissolution in coastal ecosystems (Bidle et al. 2003), by removing organic coatings from diatom frustules in seawater. A similar role is likely important in the terrestrial biosphere, especially when physical dissolution fluxes are slowed down due to high ambient DSi concentrations. Fungi could play a similar role, with fungi playing a key role in the weathering of mineral silicates (Landeweert et al. 2001; Bonneville et al. 2009). Still, we know of no studies that have investigated effects of either fungi or microbial communities on terrestrial ecosystem soil BSi storage.
Hydrology
Hydrology by definition controls water fluxes through ecosystems and hence the potential DSi export. The terrestrial biosphere can be seen as a series of interconnected spatially distributed subsystems (Fisher et al. 1998). These subsystems are distributed both horizontally (adjacent ecosystems, uplands and wetlands…) and vertically (litter, soil, sediment, bedrock…) and hydrology mediates material fluxes between the different subsystems. Although dust-related transport of Si exists between terrestrial and ocean ecospheres (Tréguer et al. 1995), this is only a minor fraction of BSi in the terrestrial biosphere, with the major part of ecosystem BSi either dissolving into soil water or remaining buried in ecosystem soils. The impact of hydrology is even more intricate if one considers that recycling of BSi to DSi is positively correlated to ambient DSi concentration (Rickert et al. 2002; Truesdale et al. 2005). Longer water residence time will allow higher DSi concentrations to develop in soil water and will slow down the actual dissolution processes. Ground water table and hydrology also partly control ecosystem biodiversity and hence impact on potential DSi uptake into the biological BSi buffer. Hydrologists have already taken advantage of the fact that different flow paths produce distinct Si concentrations, and have therefore used DSi as a hydrologic tracer (e.g. Hornberger et al. 2001; Scanlon et al. 2001). During snow-melt or precipitation related high-discharge events (Smedberg et al. 2006; Smis et al. in press), DSi concentrations drop due to dilution.
Quantification and qualification of flow paths of DSi through ecosystems via different hydrological pathways (leaching, surface flow, lateral flows) should be coupled to ecosystem BSi balances. This is a necessary prerequisite to understand the residence time of BSi in ecosystem soils. The effect of hydrology on BSi storage and recycling within soils could be addressed by focusing on hydrological gradients within ecosystems and by conducting experiments artificially impacting hydrology, thus providing an understanding how changing flow paths will alter BSi storage in ecosystems.
Addressing issues of the influence of hydrology will allow us to quantify key factors that land cover changes have on Si fluxes. For example, impervious cover in urban landscapes has been shown to alter the base-flow to surface run-off ratio as well as increased erosion in urban landscapes (Loucaides et al. 2007). Understanding changing hydraulic pathways is essential to increase our understanding of ecosystem Si filtering with human alteration of landscapes (see “Anthropogenic influences” section).
Interaction with the geosphere
The weatherability of silicate containing minerals, as well as the pedogenic ASi and the BSi content of soils, will determine how much DSi is available for biogeochemical cycling in an ecosystem (Conley et al. 2006). On a global scale lithology is an important component describing Si fluxes with volcanic material in the watershed a key parameter determining the export of DSi (e.g. Beusen et al. 2009). However, we can not understand the role of the mineral Si cycle solely from the perspective of the lithology. Different mineral pools of Si develop over time, especially with soil formation, with different solubilities and reaction rates because of their chemical and mineralogical composition (Sauer et al. 2006). We need a better understanding of the range of pedogenic ASi fractions and their reactivity in terrestrial environments (see “Dissolution rates and reactivity” section) including the role of parent material in providing the initial substrate for pedogenic ASi formation.
Recent research has shown that tropical vegetation can differ in isotopic Si content, depending on the weathering stage of the parent material, indicating a direct link between vegetation Si and weathered Si (Opfergelt et al. 2008; Henriet et al. 2008). Impacts of morphoclimate zonation on the size of the ecosystem BSi reservoir is expected with weathering rates and weathering stage differing between tropical and temperate regions. In an exhaustive review, Cornelis et al. (2010) hypothesize a tight link between weathering and Si fluxes from the soil–plant system. Further studies are especially needed in soil–plant systems at high latitudes, as global change will most strongly impact temperature, hydrology and vegetation in these regions (Cornelis et al. 2010), and in systems with high weathering rates, since most studies focused on the biological control on Si cycling and export in highly weathered tropical systems with a low availability of mineral-derived Si.
Anthropogenic influences
We hypothesize that anthropogenic disturbances in landscapes will strongly impact the ecosystem BSi processing, as they will affect the key factors identified here. Before the impact man, ecosystem BSi was widely distributed (Struyf et al. 2010a). For example, a typical West-European floodplain would consist of a mosaique of deciduous forest, grasslands, heathlands, with wetlands in the riverine floodplain. In the current anthropocene, man-made ecosystems appear, such as croplands and pastures, and urban landscapes with roads, villages and cities, with floodplains drained and diked. The remaining ecosystems are now only isolated BSi hot-spots. In the new ecosystems, uptake of BSi into the soil BSi reservoir will be absent or reduced since urban areas have limited ability to take up DSi into vegetation, cropland and pastures have reduced soil accumulation, and BSi in vegetation is removed frequently through harvesting. Increased soil erosion in deforested areas can result in the loss of the bio-active soil layers and remove the high BSi concentrations found in surficial soils (Saccone et al. 2007). Land-use by humans also changes hydrological flow paths, by increasing surface flows, decreasing precipitation infiltration and increasing river canalization. Changes in precipitation patterns as expected with climate change (increased importance of event-like, intense precipitation) will increase surface run-off and top-soil erosion reducing BSi in surface soils (Conley et al. 2008; Struyf et al. 2010b).
The spatial distribution of BSi hotspots in the landscape is important to BSi storage and recycling. Riparian wetlands, for example, have a well-known buffering effect on the nitrogen and phosphorus cycle, but also on the Si cycle (Struyf and Conley 2009). The age of the ecosystems will equally be an important factor with older systems building up a significant BSi reservoir in soils, which might differ strongly in function and biomass from younger, less developed ecosystems.
Whether land-use impacts by humans will induce a net increase or reduction of Si fluxes depends on the combination of key controls we identified. This change adds to other anthropogenic impacts on Si fluxes through the riverine continuum. Important reductions in riverine Si transport have been observed by deposition in reservoirs. Damming of rivers leads to increased trapping of BSi in the lake sediments, reducing downstream transport of Si (Humborg et al. 1997).
Conclusions
Budget studies that do not incorporate the influence of terrestrial biological Si storage and processing are biased in deducting the origin and size of Si fluxes and the associated carbon sinks (e.g. Wolff-Boenisch et al. 2009; Gao et al. 2009; Noh et al. 2009). We realize such recent studies are currently based on best available knowledge. Increasing our knowledge on the mechanisms of the ecosystem Si filter, and quantifying its importance in global Si budgets, is therefore essential to avoid potential bias. Accurately quantifying human influence on the ecosystem Si filter is also necessary in models of coastal productivity (Conley et al. 1993; Laruelle et al. 2009). An estimate of past Si fluxes to the coastal zone is impossible without a good concept of biological Si buffering along the land–ocean continuum. As small changes in nutrient input parameters can have complex results for productivity (Denman 2003), incorporating knowledge on the ecosystem Si filter in models for continental Si mobilization could shed new light on our concept of eutrophication and associated shifts in coastal phytoplankton (Cloern 2001). The same holds true for the carbon pump in the ocean, which is controlled by the burial of diatoms in the deep ocean. The silica-associated carbon pump partly is dependent upon the annual input of terrestrial Si into the coastal zone and eventually the ocean (Tréguer et al. 1995; Laruelle et al. 2009).
We encourage research focusing on our key topics—ecosystem biodiversity, BSi dissolution rates and reactivity, hydrology, weatherability of parent material and anthropogenic landscape impacts—to better quantify the global and local importance of the terrestrial biogeochemical Si cycle, and especially the ecosystem Si filter. Research focusing on these topics, both individually or on synergetic effects, will allow us to constrain the timescales at which biological Si storage and recycling is occurring, its persistence and reactivity throughout the soil profile, its dependence on ecological processes, its feedbacks to hydrology, its interaction with man’s activities and ultimately its global relevance in Si budgets.
References
Alexandre A, Meunier J-D, Colin F, Koud J-M (1997) Plant impact on the biogeochemical cycle of silicon and related weathering processes. Geochim Cosmochim Acta 61:677–682
Aoki Y, Hoshino M, Matsubara T (2007) Silica and testate amoebae in a soil under pine-oak forest. Geoderma 142:29–35
Bartoli F (1983) The biogeochemical cycle of silicon in two temperate forest ecosystems. Ecol Bull 35:469–476
Beusen AHW, Bouwman AF, Durr HH, Dekkers ALM, Hartmann J (2009) Global patterns of dissolved silica export to the coastal zone: results from a spatially explicit global model. Global Biogeochem Cycles 23:GB0A02. doi:10.1029/2008GB00328
Bidle KD, Brzezinski MA, Long RA, Jones JL, Azam F (2003) Diminished efficiency in the oceanic silica pump caused by bacteria-mediated silica dissolution. Limnol Oceanogr 48:1855–1868
Blecker SW, McCulley RL, Chadwick OA, Kelly EF (2006) Biologic cycling of silica across a grassland bioclimosequence. Global Biogeochem Cycles 20:GB3023
Bonneville S, Smits MM, Brown A, Harrington J, Leake JR, Brydson R, Benning LG (2009) Plant-driven fungal weathering: early stages of mineral alteration at the nanometer scale. Geology 37:615–618
Cary L, Alexandre A, Meunier J-D, Boeglin J-L, Braun J-J (2005) Contribution of phytoliths to the suspended load of biogenic silica in the Nyong basin rivers (Cameroon). Biogeochemistry 74:101–114
Clarke J (2003) The occurence and significance of biogenic opal in the regolith. Earth Sci Rev 60:175–194
Cloern JE (2001) Our evolving conceptual model of the coastal eutrophication problem. Mar Ecol Prog Ser 210:223–253
Conley DJ (2002) Terrestrial ecosystems and the global biogeochemical silica cycle. Global Biogeochem Cycles 16:1121. doi:10.1029/2002GB001894
Conley DJ, Schelske CL, Stoermer EF (1993) Modification of silica biogeochemistry with eutrophication in aquatic systems. Mar Ecol Prog Ser 101:179–192
Conley DJ, Sommer M, Meunier J-D, Kaczorek D, Saccone L (2006) Silicon in the terrestrial biogeosphere. In: Ittekot V, Unger D, Humborg C, An NT (eds) The silicon cycle: human perturbations, impacts on aquatic Systems. Island Press, Washington, pp 13–28
Conley DJ, Likens GE, Buso DC, Saccone L, Bailey SW, Johnson CE (2008) Deforestation causes increased dissolved silicate losses in the Hubbard Brook Experimental Forest. Glob Change Biol 14:2548–2554
Conrad R (1996) Soil microorganisms as controllers of atmospheric trace gases (H2, CO, CH4, OCS, N2O and NO). Microbiol Rev 60:609
Cornelis J-T, Delvaux B, Georg RB, Lucas Y, Ranger J, Opfergelt S (2010) Tracing the origin of dissolved silicon transferred from various soil-plant systems towards rivers: a review. Biogeosci Discuss 7:5873–5930
Demaster DJ (1981) The supply and accumulation of silica in the marine-environment. Geochim Cosmochim Acta 45:1715–1732
Denman KL (2003) Modelling planktonic ecosystems: parameterizing complexity. Prog Oceeanogr 57:429–452
Derry LA, Kurtz AC, Ziegler K, Chadwick OA (2005) Biological control of terrestrial silica cycling and export fluxes to watersheds. Nature 433:728–731
Epstein E (2009) Silicon: its manifold roles in plants. Ann Appl Biol 155:155–160
Farmer VC, Delbos E, Miller JD (2005) The role of phytolith formation and dissolution in controlling concentrations of silica in soil solutions and streams. Geoderma 127:71–79
Fisher SG, Grimm NB, Marti E, Holmes RM, Jones JB (1998) Material spiralling in stream corridors: a telescoping ecosystem model. Ecosystems 1:19–34
Fulweiler RW, Nixon S (2005) Terrestrial vegetation and the seasonal cycle of dissolved silica in a southern New England coastal river. Biogeochemistry 74:115–130
Gao QZ, Tao Z, Huang XK, Na L, Yu KF, Wang ZG (2009) Chemical weathering and CO2 consumption in the Xijiang River basin, South China. Geomorphology 106:324–332
Gérard F, Mayer KU, Hodson MJ, Ranger J (2008) Modelling the biogeochemical cycle of silicon in soils: application to a temperate forest ecosystem. Geochim Cosmochim Acta 72:741–758
Henriet C, De Jaeger N, Dorel M, Opfergelt S, Delvaux B (2008) The reserve of weatherable primary silicates impacts the accumulation of biogenic silicon in volcanic ash soils. Biogeochemistry 90:209–223
Hobbie SE (1992) Effects of plant species on nutrient cycling. Trends Ecol Evol 7:336–339
Hodson MJ, White PJ, Mead A, Broadley MR (2005) Phylogenetic variation in the silicon composition of plants. Ann Bot 96:1027–1046
Hornberger GM, Scanlon TM, Raffensperger JP (2001) Modelling transport of dissolved silica in a forested headwater catchment: the effect of hydrological and chemical time scales on hysteresis in the concentration-discharge relationship. Hydrol Process 15:2029–2038
Humborg C, Ittekkot V, Cociasu A, Von Bodungen B (1997) Effect of Danube River dam on Black Sea biogeochemistry and ecosystem structure. Nature 386:385–388
Jonasson S, Chapin FS (1991) Seasonal uptake and allocation of phosphorus in Eriophorum vaginatum L. measured by labelling with P-32. New Phytol 118:349–357
Kokfelt U, Struyf E, Randsalu U (2009) Diatoms in peat—dominant producers in a changing environment? Soil Biol Biochem 41:1764–1766
Kokfelt U, Struyf E, Reuss N, Sonesson M, Rundgren M, Skog G, Rosén P, Hammarlund D (2010) Wetland development, permafrost history and nutrient cycling inferred from late Holocene peat and lake sediment records in subarctic Sweden. J Paleolimnol 44:327–342
Konhauser KO, Urrutia MM (1999) Bacterial clay authigenesis: a common biogeochemical process. Chem Geol 161:399–413
Landeweert R, Hoffland E, Finlay RD, Kuyper TW, van Breemen N (2001) Linking plants to rocks: ectomycorrhizal fungi mobilize nutrients from minerals. Trends Ecol Evol 16:248–254
Laruelle GG, Roubeix V, Sferratore A, Brodhern B, Ciuffa D, Conley DJ, Duerr H, Garnier J, Lancelot C, Phong QLT, Meybeck M, Michalopoulos P, Moriceau B, Ni Longphuirt S, Loucaides S, Papush L, Presti M, Ragueneau O, Regnier PAG, Saccone L, Slomp CP, Spiteri C, Van Cappellen P (2009) Anthropogenic perturbations of the silicon cycle at the global scale: key role of the land-ocean transition. Global Biogeochem Cycles 23:GB4031. doi:10.1029/2008GB003267
Lee SY, Gilkes RJ (2005) Groundwater geochemistry and composition of hardpans in southwestern Australian regolith. Geoderma 126:59–84
Loucaides S, Cahoon LB, Henry EJ (2007) Effects of watershed impervious cover on dissolved silica loading in storm flow. J Am Water Resour Assoc 43:841–849
Loucaides S, Van Cappellen P, Behrends T (2008) Dissolution of biogenic silica from land to ocean: role of salinity and pH. Limnol Oceanogr 53:1614–1621
Loucaides S, Behrends T, Van Cappellen P (2010) Reactivity of biogenic silica: surface versus bulk charge density. Geochim Cosmochim Acta 74:517–530
Markewitz D, Richter DD (1998) The bio in aluminum and silicon geochemistry. Biogeochemistry 42:235–252
Massey FP, Ennos AR, Hartley SE (2007) Grasses and the resource availability hypothesis: the importance of silica-based defences. J Ecol 95:414–424
Massey FP, Smith MJ, Lambin X, Hartley SE (2008) Are silica defences in grasses driving vole population cycles? Biol Lett 4:419–422
McLaren JR, Turkington R (2010) Ecosystem properties determined by plant functional group identity. J Ecol 98:459–469
McNaughton SJ, Tarrants JL (1983) Grass leaf silicification: natural selection for an inducible defense against herbivores. Proc Natl Acad Sci USA 80:790–791
Moriceau B, Garvey M, Ragueneau O, Passow U (2007) Evidence for reduced biogenic silica dissolution rates in diatom aggregates. Mar Ecol Prog Ser 333:129–142
Nannipieri P, Ascher J, Ceccherini MT, Landi L, Pietramellara G, Renella G (2003) Microbial diversity and soil functions. Eur J Soil Sci 54:655–670
Noh H, Huh Y, Qin JH, Ellis A (2009) Chemical weathering in the Three Rivers region of Eastern Tibet. Geochim Cosmochim Acta 73:1857–1877
Opfergelt S, Delvaux B, Andre L, Cardinal D (2008) Plant silicon isotopic signature might reflect soil weathering degree. Biogeochemistry 91:163–175
Rabosky DL, Sorhannus U (2009) Diversity dynamics of marine planktonic diatoms across the Cenozoic. Nature 457:183–186
Richmond KE, Sussman M (2003) Got silicon? The non-essential beneficial plant nutrient. Curr Opin Plant Biol 6:268–272
Rickert D, Schlüter M, Wallmann K (2002) Dissolution kinetics of biogenic silica from the water column to the sediments. Geochim Cosmochim Acta 66:439–455
Saccone L, Conley DJ, Koning E, Sauer D, Sommer M, Kaczorek D, Blecker SW, Kelly EF (2007) Assessing the extraction and quantification of amorphous silica in soils of forest and grassland ecosystems. Eur J Soil Sci 58:1446–1459
Saccone L, Conley DJ, Likens GE, Bailey SW, Buso DC, Johnson CE (2008) Distribution of amorphous silica in soils of the Hubbard Brook Experimental Forest. Soil Sci Soc Am J 72:1637–1644
Sauer D, Saccone L, Conley DJ, Herrmann L, Sommer M (2006) Review of methodologies for extracting plant-available and amorphous Si from soils and aquatic sediments. Biogeochemistry 80:89–108
Scanlon TM, Raffensperger JP, Hornberger GM (2001) Modeling transport of dissolved silica in a forested headwater catchment: implications for defining the hydrochemical response of observed flow pathways. Water Resour Res 37:1071–1082
Schoelynck J, Bal K, Backx H, Okruszko T, Meire P, Struyf E (2010) Silica uptake in aquatic and wetland macrophytes: a strategic choice between silica, lignin and cellulose? New Phytol 186:385–391
Schoelynck J, Bal K, Struyf E, Meire P (submitted) A long-armed non-essential element: far-reaching effects of plant silicon uptake. J Veg Sci
Smedberg E, Morth CM, Swaney DP, Humborg C (2006) Modeling hydrology and silicon–carbon interactions in taiga and tundra biomes from a landscape perspective: implications for global warming feedbacks. Global Biogeochem Cycles 20:GB2014
Smis A, Van Damme S, Struyf E, Govers G, Van Wesemael B, Clymans W, Frot E, Vandevenne F, Van Hoestenberghe T, Meire P (in press) A trade-off between dissolved and amorphous silica transport during peak-flow events (Scheldt river basin, Belgium): impacts of precipitation intensity on terrestrial Si dynamics in strongly cultivated catchments. Biogeochemistry (in press). doi:10.1007/s10533-010-9527-1
Sommer M, Kaczorek D, Kuzyakov Y, Breuer J (2006) Silicon pools and fluxes in soils and landscapes—a review. J Plant Nutr Soil Sci 169:310–329
Street-Perrott AF, Barker PA (2008) Biogenic silica: a neglected component of the coupled global continental biogeochemical cycles of carbon and silicon. Earth Surf Process Landf 33:1436–1457
Street-Perrott FA, Barker PA, Leng MJ, Sloane HJ, Wooller MJ, Ficken KJ, Swain DL (2008) Towards an understanding of late quaternary variations in the continental biogeochemical cycle of silicon: multi-isotope and sediment-flux data for Lake Rutundu, Mt Kenya, East Africa, since 38 ka BP. J Quat Sci 23:375–387
Struyf E, Conley DJ (2009) Silica: an essential nutrient in wetland biogeochemistry. Front Ecol Environ 2(7):88–94
Struyf E, Van Damme S, Gribsholt B, Middelburg JJ, Meire P (2005) Biogenic silica in freshwater marsh sediments and vegetation. Mar Ecol Prog Ser 303:51–60
Struyf E, Opdekamp W, Backx H, Jacobs S, Conley DJ, Meire P (2009) Vegetation and proximity to the river control amorphous Si storage in a riparian wetland (Bierbza National Park, Poland). Biogeosciences 6:623–631
Struyf E, Mörth C-M, Humborg C, Conley DJ (2010a) An enormous amorphous silica stock in boreal wetlands. J Geophys Res 115:G04008
Struyf E, Smis A, Van Damme S, Garnier J, Govers G, Van Wesemael B, Conley DJ, Batelaan O, Frot E, Clymans W, Vandevenne F, Lancelot C, Goos P, Meire P (2010b) Historical land use change has lowered base-line silica mobilization from landscapes. Nat Commun 1:129. doi:10.1038/ncomms1128
Tréguer P, Nelson DM, Vanbennekom AJ, Demaster DJ, Leynaert A, Queguiner B (1995) The silica balance in the world ocean—a reestimate. Science 268:375–379
Truesdale VW, Greenwood JE, Rendell AR (2005) In vitro, batch-dissolution of biogenic silica in seawater—the application of recent modelling to real data. Prog Oceanogr 66:1–24
Van Cappellen P (2003) Biomineralization and global biogeochemical cycles. Rev Miner Geochem 54:357–381
Van Cappellen P, Dixit S, van Beusekom J (2002) Biogenic silica dissolution in the oceans: reconciling experimental and field-based dissolution rates. Global Biogeochem Cycles 16:10
White AF, Blum AE (1995) Effects of climate on chemical weathering in watersheds. Geochim Cosmochim Acta 59:1729–1747
Wolff-Boenisch D, Gabet EJ, Burbank DW, Langner H, Putkonen J (2009) Spatial variations in chemical weathering and CO2 consumption in Nepalese High Himalayan catchments during the monsoon season. Geochim Cosmochim Acta 73:3148–3170
Yamada SS, D’Elia CF (1984) Silicic acid regeneration from estuarine sediment cores. Mar Ecol Prog Ser 18:113–118
Acknowledgments
Eric Struyf would like to thank FWO (Research Foundation Flanders) for personal post doc research funding. We would like to acknowledge Belgian Science Policy (BELSPO) for funding the project “LUSi: Land use changes and silica fluxes in the Scheldt river basin” and FWO for funding project “Tracking the biological control on Si mobilisation in upland ecosystems” (Project nr. G014609N). This project was partially funded by a grant to D. Conley from the Crawfoord Foundation and the Swedish National Science Foundation (VR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Struyf, E., Conley, D.J. Emerging understanding of the ecosystem silica filter. Biogeochemistry 107, 9–18 (2012). https://doi.org/10.1007/s10533-011-9590-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-011-9590-2